Submitted:
14 November 2024
Posted:
19 November 2024
You are already at the latest version
Abstract
Machine learning has witnessed a notable increase in significance within the medical field, primarily due to the increasing availability of health-related data and the progressive enhancements in machine learning algorithms. It can be utilized to formulate predictive models that aid in disease diagnosis, anticipate disease progression, tailor treatment to fulfill individual patient needs and improve the operational efficiency of healthcare systems. The strategic utilization of data can considerably elevate the quality of patient care, reduce healthcare costs, and promote the formulation of personalized and effective medical interventions. The healthcare industry reaps considerable benefits from the meticulous analysis of medical data, as it plays an integral role in promptly identifying diseases in patients. Timely detection of a disease can contribute to effective symptom management and guarantee that appropriate treatment is provided. The pronounced association between evoked potentials (EPs) and Expanded Disability Status Scale (EDSS) scores in individuals diagnosed with multiple sclerosis (MS) indicates that EPs may serve as dependable predictive markers for the progression of disability. Numerous studies have confirmed that variations in somatosensory evoked potentials (SEPs) demonstrate a relationship with EDSS scores, particularly during the early stages of the disease. The present study aims to apply artificial intelligence techniques to identify predictors linked to the progression of Multiple Sclerosis (MS) as assessed by the disability index (EDSS). It is essential to clarify the role of evoked potentials (EPs) in the prognostication of MS. We analyzed empirical data obtained from a medical database of 125 records. Our primary objective is to construct an expert Artificial Intelligence system capable of predicting the EDSS index by applying advanced knowledge-mining algorithms. We have developed intelligent systems that predict the progression of MS utilizing machine learning algorithms, specifically Decision Trees and Neural Networks. In our experimental evaluation, Decision Trees, Neural Networks, and Bayes for EPs achieved accuracies of 88.9%, 92.9%, and 88.2% respectively, which are comparable to MRI which obtained accuracies of 88.2%, 96.0%, and 85.0%. The EPs can be established as predictors of MS with efficacy analogous to that of MRI findings. Further investigation is necessary to validate EPs, which are significantly less expensive, portable, and simpler to administer than MRI, as equally effective as imaging or biochemical methods in functioning as biomarkers for MS.
Keywords:
1. Introduction
1.1. Pathology of MS
1.2. Diagnosing MS
1.3. Identifying Biomarkers for Multiple Sclerosis progress
1.4. Classification in MS
1.5. Risk Factors for MS
1.6. Treatment in MS
Physiotherapy in MS Rehabilitation Strategy
1.7. Prognosis in MS
1.8. Evoked Potentials in MS
1.9. Artificial Intelligence Technological Approaches
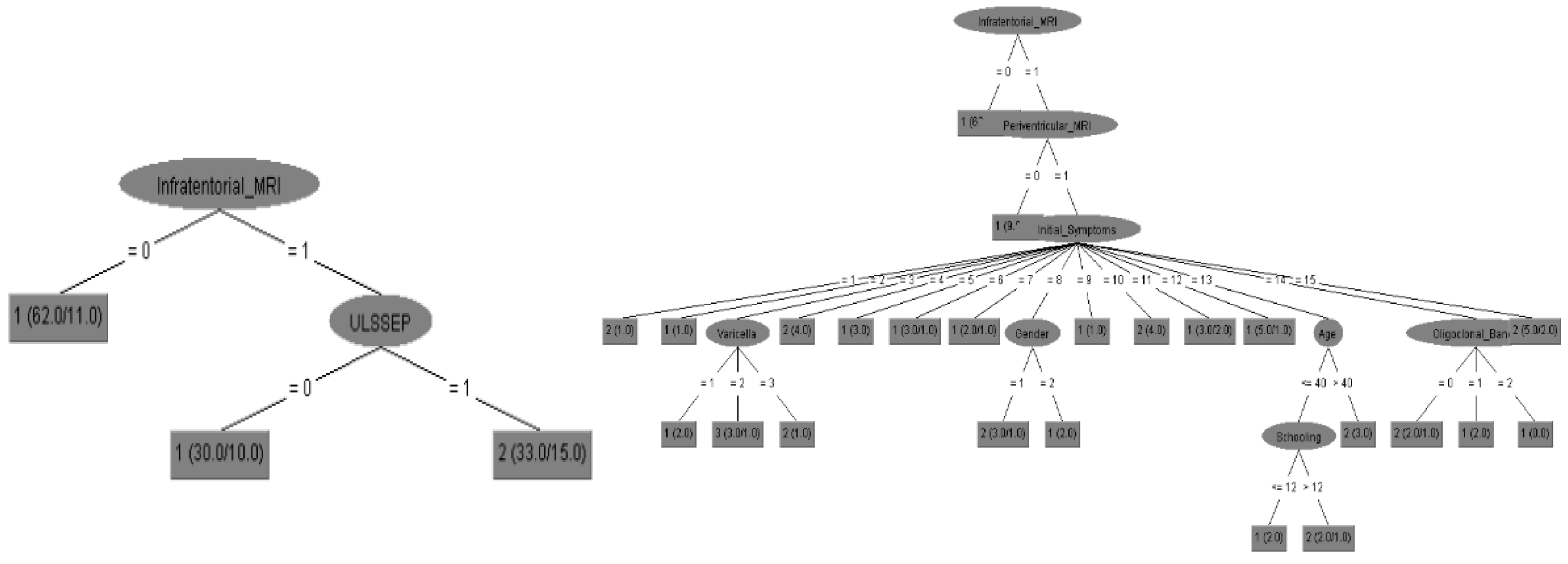
2. Materials and Methods
-
- 1.
- Age: Age of the patient (in years)
- 2.
- Schooling: time the patient spent in school (in years)
- 3.
- Gender: 1=male, 2=female
- 4.
- Breastfeeding: 1=yes, 2=no, 3=unknown
- 5.
- Varicella: 1=positive, 2=negative, 3=unknown
- 6.
- Initial_Symptoms: 1=visual, 2=sensory, 3=motor, 4=other, 5= visual and sensory, 6=visual and motor, 7=visual and others, 8=sensory and motor, 9=sensory and other, 10=motor and other, 11=Visual, sensory and motor, 12=visual, sensory and other, 13=Visual, motor and other, 14=Sensory, motor and other, 15=visual, sensory, motor and other
- 7.
- Mono _or_Polysymptomatic: 1=monosymptomatic, 2=polysymptomatic, 3=unknown
- 8.
- Oligoclonal_Bands: 0=negative, 1=positive, 2=unknown
- 9.
- LLSSEP: 0=negative, 1=positive
- 10.
- ULSSEP:0=negative, 1=positive
- 11.
- VEP:0=negative, 1=positive
- 12.
- BAEP: 0=negative, 1=positive
- 13.
- Periventricular_MRI:0=negative, 1=positive
- 14.
- Cortical_MRI: 0=negative, 1=positive
- 15.
- Infratentorial_MRI:0=negative, 1=positive
- 16.
- Spinal_Cord_MRI: 0=negative, 1=positive
- 17.
- initial_EDSS: EDSS index at the onset of the disease
- 18.
- final_EDSS: index after the disease progress
- Varicella: Another name for Chickenpox, or chicken pox, is a highly contagious disease caused by the initial infection with varicella zoster virus (VZV), a member of the herpesvirus family.
- BAEP: In human neuroanatomy, brainstem auditory evoked potentials (BAEPs), also called brainstem auditory evoked responses (BAERs), are very small auditory evoked potentials in response to an auditory stimulus, which are recorded by electrodes placed on the scalp.
- VEP: Visual evoked potential (VEP) is an evoked potential elicited by presenting light flash or pattern stimulus which can be used to confirm damage to visual pathway including retina, optic nerve, optic chiasm, optic radiations, and occipital cortex.
- Oligoclonal bands: Oligoclonal bands (OCBs) are bands of immunoglobulins that are seen when a patient’s blood serum, or cerebrospinal fluid (CSF) is analyzed. They are used in the diagnosis of various neurological and blood diseases. Oligoclonal bands are present in the CSF of more than 95% of patients with clinically definite multiple sclerosis.
- SSEP : Somatosensory evoked potentials (SSEP) are recorded from the central nervous system following stimulation of peripheral nerves. ULSSEP (upper limb SSEP), LLSSEP (lower limb SSEP)
- EDSS: The Expanded Disability Status Scale (EDSS) is a method of quantifying disability in multiple sclerosis and monitoring changes in the level of disability over time. It is widely used in clinical trials and in the assessment of people with MS (Cadavid et al., 2017)
- Oligoclonal_Bands: Oligoclonal bands (OCBs) are bands of immunoglobulins that are seen when a patient’s blood serum, or cerebrospinal fluid (CSF) is analyzed. They are used in the diagnosis of various neurological and blood diseases. Oligoclonal bands are present in the CSF of more than 95% of patients with clinically definite multiple sclerosis (Haki et al., 2024).
3. Results
4. Discussion
References
- Arrambide, G., Iacobaeus, E., Amato, M. P., Derfuss, T., Vukusic, S., Hemmer, B., Brundin, L., Tintore, M., & 2018 ECTRIMS Focused Workshop Group (2020). Aggressive multiple sclerosis (2): Treatment. Multiple sclerosis (Houndmills, Basingstoke, England), 26(9), 1352458520924595. Advance online publication. [CrossRef]
- Bakirtzis, C., Evangelopoulos, M. E., & Grigoriadis, N. (2024). Multiple Sclerosis and Demyelinating Disorders: Past, Present, and Future. Journal of clinical medicine, 13(18), 5621. [CrossRef]
- Banitalebi, E., Ghahfarrokhi, M. M., Negaresh, R., Kazemi, A., Faramarzi, M., Motl, R. W., & Zimmer, P. (2020). Exercise improves neurotrophins in multiple sclerosis independent of disability status. Multiple sclerosis and related disorders, 43, 102143. [CrossRef]
- Cadavid, D., Cohen J., A., Freedman, M., S., et al. (2017) The EDSS-Plus, an improved endpoint for disability progression in secondary progressive multiple sclerosis. Multiple Sclerosis Journal. 23(1):94-105. [CrossRef]
- Casanova, B., Coret, F., Parra, S., Bosca, I., & Piera, A. (2005). Predictive value of evoked potentials in multiple sclerosis. A 12-years follow-up study. J Neurol, 252 (Suppl 2), 11.
- Corrini, C., Gervasoni, E., Perini, G., Cosentino, C., Putzolu, M., Montesano, A., Pelosin, E., Prosperini, L., & Cattaneo, D. (2023). Mobility and balance rehabilitation in multiple sclerosis: A systematic review and dose-response meta-analysis. Multiple sclerosis and related disorders, 69, 104424. [CrossRef]
- Dendrou, C., Fugger, L. & Friese, M. Immunopathology of multiple sclerosis. Nat Rev Immunol 15, 545–558 (2015). [CrossRef]
- Disanto, G., Morahan, J. M., & Ramagopalan, S. V. (2012). Multiple sclerosis: risk factors and their interactions. CNS & neurological disorders drug targets, 11(5), 545–555. [CrossRef]
- Etoom, M., Khraiwesh, Y., Lena, F., Hawamdeh, M., Hawamdeh, Z., Centonze, D., & Foti, C. (2018). Effectiveness of Physiotherapy Interventions on Spasticity in People With Multiple Sclerosis: A Systematic Review and Meta-Analysis. American journal of physical medicine & rehabilitation, 97(11), 793–807. [CrossRef]
- Fernández V. (2021). The Use of Motor-Evoked Potentials in Clinical Trials in Multiple Sclerosis. Journal of clinical neurophysiology: official publication of the American Electroencephalographic Society, 38(3), 166–170. [CrossRef]
- Garg, N., Hu, Y., & Rammohan, K., W. (2018). Biomarkers in Multiple Sclerosis. Practical Neurology https://practicalneurology.com/articles/2018-sept/biomarkers-in-multiple-sclerosis.
- Haki, M., Al-Biati, H. A., Al-Tameemi, Z. S., Ali, I. S., & Al-Hussaniy, H. A. (2024). Review of multiple sclerosis: Epidemiology, etiology, pathophysiology, and treatment. Medicine, 103(8), e37297. [CrossRef]
- Hardmeier, M., & Fuhr, P. (2021). Multimodal Evoked Potentials as Candidate Prognostic and Response Biomarkers in Clinical Trials of Multiple Sclerosis. Journal of clinical neurophysiology: official publication of the American Electroencephalographic Society, 38(3), 171–180. [CrossRef]
- Harris, V. K., Tuddenham, J. F., & Sadiq, S. A. (2017). Biomarkers of multiple sclerosis: current findings. Degenerative neurological and neuromuscular disease, 7, 19–29. [CrossRef]
- Hongmei, Y., Yingtao J. Zheng, J. Peng, C. Li, Q. (2006), “A multilayer perceptron-based medical decision support system for heart disease diagnosis”, Expert Systems with Applications, Volume 30, Issue 2, Pages 272-281.
- Iacobaeus, E., Arrambide, G., Amato, M. P., Derfuss, T., Vukusic, S., Hemmer, B., Tintore, M., Brundin, L., & 2018 ECTRIMS Focused Workshop Group (2020). Aggressive multiple sclerosis (1): Towards a definition of the phenotype. Multiple sclerosis (Houndmills, Basingstoke, England), 26(9), 1352458520925369. Advance online publication. [CrossRef]
- Inojosa, H., Schriefer, D., & Ziemssen, T. (2020). Clinical outcome measures in multiple sclerosis: A review. Autoimmunity reviews, 19(5), 102512. [CrossRef]
- Kalb, R., Brown, T. R., Coote, S., Costello, K., Dalgas, U., Garmon, E., Giesser, B., Halper, J., Karpatkin, H., Keller, J., Ng, A. V., Pilutti, L. A., Rohrig, A., Van Asch, P., Zackowski, K., & Motl, R. W. (2020). Exercise and lifestyle physical activity recommendations for people with multiple sclerosis throughout the disease course. Multiple sclerosis (Houndmills, Basingstoke, England), 26(12), 1459–1469. [CrossRef]
- Kjaer M. (1980). The value of brain stem auditory, visual and somatosensory evoked potentials and blink reflexes in the diagnosis of multiple sclerosis. Acta Neurol Scand. 62(4):220-36. [CrossRef] [PubMed]
- Kraft G. H. (2013). Evoked potentials in multiple sclerosis. Physical medicine and rehabilitation clinics of North America, 24(4), 717–720. [CrossRef]
- Krishnani, D., Kumari, A., Dewangan, A., Singh, A., & Naik, N. S. (2019). Prediction of Coronary Heart Disease using Supervised Machine Learning Algorithms. IEEE Xplore. [CrossRef]
- Koehler, J., Faldum, A. & Hopf, H. (2000). EDSS correlated analysis of median nerve somatosensory evoked potentials in multiple sclerosis. Neurol Sci 21, 217–221 . [CrossRef]
- Lawal, A. I., & Kwon, S. (2021). Application of artificial intelligence to rock mechanics: An overview. Journal of Rock Mechanics and Geotechnical Engineering, 13(1), 248–266. [CrossRef]
- Margaritella N, Mendozzi L, Garegnani M, Nemni R, Colicino E, Gilardi E, Pugnetti L.( 2012) Exploring the predictive value of the evoked potentials score in MS within an appropriate patient population: a hint for an early identification of benign MS? BMC Neurol. Aug 22;12:80. [CrossRef]
- Mastaglia, F. (2006). Can abnormal evoked potentials predict future clinical disability in patients with multiple sclerosis? Nat Rev Neurol 2, 304–305. [CrossRef]
- Moxley-Wyles, B., Colling, R., & Verrill, C. (2020). Artificial intelligence in pathology: an overview. Diagnostic Histopathology, 26(11), 513–520. [CrossRef]
- McKay, K., A., Tremlett, H., Fisk, J., D., et al. (2018). Psychiatric comorbidity is associated with disability progression in multiple sclerosis. Neurology; 90(15):e1316-e1323. [CrossRef]
- Murdaca, G., Tonacci, A., Negrini, S., Greco, M., Borro, M., Puppo, F., & Gangemi, S. (2019). Emerging role of vitamin D in autoimmune diseases: An update on evidence and therapeutic implications. Autoimmunity reviews, 18(9), 102350. [CrossRef]
- Nourbakhsh, B., & Mowry, E. M. (2019). Multiple Sclerosis Risk Factors and Pathogenesis. Continuum (Minneapolis, Minn.), 25(3), 596–610. [CrossRef]
- Oh, J., Vidal-Jordana, A., & Montalban, X. (2018). Multiple sclerosis: clinical aspects. Current opinion in neurology, 31(6), 752–759. [CrossRef]
- Pineda, Benjamin; Flores Rivera, Jose De Jesus (2023), “Conversion predictors of Clinically Isolated Syndrome to Multiple Sclerosis in Mexican patients: a prospective study”, Mendeley Data, V1: https://10.17632/8wk5hjx7x2.1.
- Sangha, A., Quon, M., Pfeffer, G., & Orton, S. M. (2023). The Role of Vitamin D in Neuroprotection in Multiple Sclerosis: An Update. Nutrients, 15(13), 2978. [CrossRef]
- Šoda, J., Pavelin, S., Vujović, I., & Rogić Vidaković, M. (2023). Assessment of Motor Evoked Potentials in Multiple Sclerosis. Sensors (Basel, Switzerland), 23(1), 497. [CrossRef]
- Thompson, A. J., Banwell, B. L., Barkhof, F., Carroll, W. M., Coetzee, T., Comi, G., Correale, J., Fazekas, F., Filippi, M., Freedman, M. S., Fujihara, K., Galetta, S. L., Hartung, H. P., Kappos, L., Lublin, F. D., Marrie, R. A., Miller, A. E., Miller, D. H., Montalban, X., Mowry, E. M., … Cohen, J. A. (2018). Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. The Lancet. Neurology, 17(2), 162–173. [CrossRef]
- van Langelaar, J., van der Vuurst de Vries, R. M., Janssen, M., Wierenga-Wolf, A. F., Spilt, I. M., Siepman, T. A., Dankers, W., Verjans, G. M. G. M., de Vries, H. E., Lubberts, E., Hintzen, R. Q., & van Luijn, M. M. (2018). T helper 17.1 cells associate with multiple sclerosis disease activity: perspectives for early intervention. Brain : a journal of neurology, 141(5), 1334–1349. [CrossRef]
- Voigt, I., Inojosa, H., Dillenseger, A., Haase, R., Akgün, K., & Ziemssen, T. (2021). Digital Twins for Multiple Sclerosis. Frontiers in immunology, 12, 669811. [CrossRef]
- Wagner, C., A., Roqué, P., J., & Goverman, J., M. Pathogenic T cell cytokines in multiple sclerosis. J Exp Med 6 January 2020; 217 (1): e20190460. doi: . [CrossRef]
- Wang, G., Marrie, R., A., Fox, R., J., et al. Treatment satisfaction and bothersome bladder, bowel, sexual symptoms in multiple sclerosis (2018). Mult Scler Relat Disord 20:16-21. [CrossRef]
- Ward, M., & Goldman, M. D. (2022). Epidemiology and Pathophysiology of Multiple Sclerosis. Continuum (Minneapolis, Minn.), 28(4), 988–1005. [CrossRef]
- Warutkar, V., Gulrandhe, P., Morghade, S., Krishna Kovela, R., Qureshi, M., I. (2022) Physiotherapy for Multiple Sclerosis Patients From Early to Transition Phase: A Scoping Review. Cureus. 14(10):e30779. [CrossRef] [PubMed] [PubMed Central]
- WEKA, Holmes G, Donkin A. Witten I.H. (1994) Weka: A machine learning workbench. Proc Second Australia and New Zealand Conference on Intelligent Information Systems, Brisbane, Australia. WEKA. Application of artificial neural networks to clinical medicine. Lancet 1995q346:1135-1138.
- WEKA Machine Learning Group at the University of Waikato: http://www.cs.waikato.ac.nz/ml/index.
- Yang, J., Hamade, M., Wu, Q., Wang, Q., Axtell, R., Giri, S., & Mao-Draayer, Y. (2022). Current and Future Biomarkers in Multiple Sclerosis. International journal of molecular sciences, 23(11), 5877. [CrossRef]
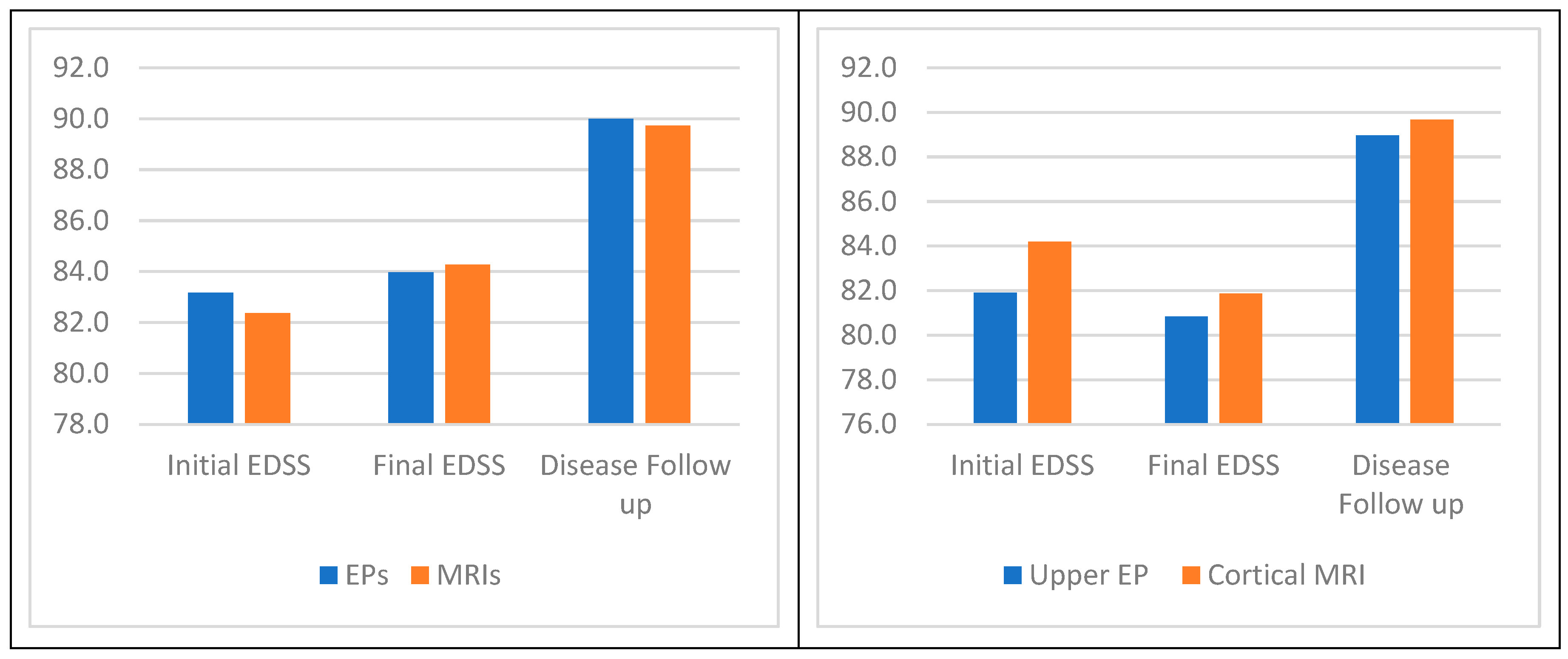
| a. Decision Trees (J48) | ||||||
| Biomarker accuracy (%) | All | EPs | MRIs | Upper EP | Cortical MRI | Other |
| Initial EDSS | 89,3 | 82,5 | 81,1 | 85,0 | 85,8 | 85,8 |
| Final EDSS | 94,5 | 85,8 | 82,7 | 82,7 | 82,6 | 81,8 |
| Disease Follow up | 88,1 | 88,9 | 88,2 | 85,8 | 85,8 | 85,8 |
| b. Neural Networks (Multilayer Perceptron) | ||||||
| Biomarker accuracy (%) | All | EPs | MRIs | Upper EP | Cortical MRI | Other |
| Initial EDSS | 95,3 | 95,3 | 97,5 | 90,6 | 96,8 | 85,8 |
| Final EDSS | 96,0 | 98,4 | 94,5 | 92,9 | 97,6 | 91,3 |
| Disease Follow up | 94,5 | 92,9 | 96,0 | 94,5 | 96,6 | 94,5 |
| c. Bayes (Naïve Bayes) | ||||||
| Biomarker accuracy (%) | All | EPs | MRIs | Upper EP | Cortical MRI | Other |
| Initial EDSS | 72,0 | 71,7 | 68,5 | 70,1 | 70,0 | 66,9 |
| Final EDSS | 78,4 | 67,7 | 75,6 | 66,9 | 65,4 | 63,8 |
| Disease Follow up | 85,0 | 88,2 | 85,0 | 86,6 | 86,6 | 86,6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




