1. Introduction
Inflammatory bowel disease (IBD) is an immune-mediated chronic disease characterized by gastrointestinal and extra digestive manifestations, alternating between periods of remission and active disease. It encompasses Crohn’s disease (CD), ulcerative colitis (UC) and unclassified IBD (IBD-U). In approximately 25% of IBD patients, the diagnosis is established at the pediatric age (1). Currently, these chronic conditions have no available cure and are widely recognized for their potentially negative impact on quality of life (QOL) [
1,
2].
Diagnosing IBD in pediatric patients may be quite challenging for both families and patients, as it is often more severe and progressive, with an increased risk of surgery, compared with adult IBD. Additionally, pediatric IBD patients are recognized to have age-specific complications, namely, linear growth impairment, malnutrition, pubertal delay, and decreased bone mineral density [
1,
3]. In addition to uncomfortable and sometimes disabling symptoms, invasive diagnostic procedures, complex and time-consuming treatment options and medication side effects, patients and families experience limitations in everyday activities and restrictions in school/professional and social activities [
2,
4].
Several pediatric studies have documented impaired health-related quality of life (HRQOL) in the physical, psychological, and autonomy domains related to disease activity, requiring specific coping strategies [
2,
4,
5,
6,
7]. When addressing an IBD patient, a comprehensive approach to function and disability, along with traditional clinical parameters, is desirable. The primary goal of treatment is to achieve and maintain disease control, thereby improving QOL and diminishing the disease burden. A systematic review published in 2014 (8) examined the evidence on HRQOL in pediatric IBD patients and concluded that HRQOL is lower in children and young adults than in adult patients. Understanding pediatric patients’ perspectives requires knowledge of their cognitive and developmental characteristics. Moreover, parents often provide most of the information concerning the child's health and tend to underestimate the patient's QOL [
7]. Involving the pediatric patient in the management of the disease contributes to a better transition to adult healthcare.
In pediatric IBD, specific QOL instruments such as the IMPACT questionnaire [
9,
10] have been developed, validated and used in several studies, both retrospective and prospective, as well as in clinical practice. Furthermore, in the context of pediatric IBD, to address other domains of health and functioning, including mood, sleep, and fatigue disorders, it is often necessary to utilize specific tools in addition to specific IBD tools, increasing the complexity of the assessment process and the respondent burden. Moreover, it is difficult to compare findings across studies in pediatric IBD patients and other pediatric chronic IBD patients.
Generic measures that can be applied to individuals and populations with several health conditions, differentiating groups of overall health and well-being [
11], may be useful in IBD patients, as they can capture specific morbidities (such as anxiety and depression) that are not captured by disease-specific measures [
12]. Additionally, they allow for comparability across patients and populations with different conditions.
2. Patient-Reported Outcomes
Patient-reported outcomes (PROs) are health experiences and evaluations assessed by patient reports, including symptoms, assessments of functioning, well-being, health perceptions, and satisfaction with care [
13]. They measure the outcomes of treatments and disease management reported by the patient and/or caregivers [
4,
13,
14,
15].
The measurement of PROs may be performed via interviews or, more frequently, via written questionnaires delivered through computers or telephone [
11]. Initially, developed as an additional outcome tool in clinical trials, their importance was reinforced in 2009 by FDA guidance [
16], which advocated for the incorporation of the patient’s perspective in clinical care and research [
17].
In the past two decades, PROs have become important tools in healthcare, attracting increasing professional interest as tools for day-to-day management decisions and driving healthcare quality [
7,
13,
14,
18,
19]. They represent a good model of patient-centered care [13, 15, 17, 20, 21).
The use of PROs in a clinical setting is particularly relevant in chronic conditions where patient involvement is central to successful disease management. They provide additional information regarding the burden of the disease or treatment that might be relevant for clinical decisions. PROs assess a person’s feelings, functional status, and well-being and not only traditional clinical outcomes, such as organ system function, disease activity and comorbidities, allowing a more comprehensive understanding of the impact of chronic disease on patients’ health [
22]. As PROs can be monitored without in-office evaluations, they can potentially determine the interval between medical visits [
20,
21,
23,
24,
25,
26]. However, the implementation of PROs in clinical settings faces several barriers, such as time and respondent burden, a lack of time during medical visits and the absence of standardized measures applicable to different chronic diseases in both research and clinical care [
11,
27].
In the pediatric setting, PROs are also becoming important endpoints in several chronic conditions (28-35). In pediatric IBD, different authors have concluded that well-defined, reliable, sensitive, and globally recognized PROs are needed, alongside traditional markers of the disease (endoscopy-based endpoints and/or biomarkers), to facilitate pediatric drug development [
18] and clinical practice [
36]. Additionally, highlighting the relevance of PROs in IBD, the Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE-II) recommendations [
36], has added that the restoration of HRQOL and absence of disability has formal long-term targets, along with endoscopic healing. The STRIDE-II recommends that this target be assessed at an early stage and on a regular basis throughout the disease course, irrespective of other objective markers of inflammation. The authors consider that the development of PROs for use in clinical practice, with high reliability, face validity, construct validity, responsiveness, and feasibility, is an important area of future research, as they should be developed.
An international, cross-disciplinary consensus, including an international working group representing patients, patient associations, gastroenterologists, surgeons, specialist nurses, IBD registries and PRO measurement methodologists, [
37] developed and proposed a standard set of patient-centered outcomes for IBD for patients aged ≥16 years. The suggested outcome domains important in adolescent and adult IBD patients include survival and disease control (disease activity/remission, colorectal cancer, anemia), disutility of care (treatment-related complications), healthcare utilization (IBD-related admissions, emergency room visits) and patient-reported outcomes (including quality of life, nutritional status and impact of fistulae) [
37].
Despite the potential impact on care, no PROMs are yet systematically used in routine clinical practice.
The variety of IBD specific PROMs available underscores the complexity and multifaceted nature of the disease's impact on QOL and highlight the difficulty in choosing an adequate tool (table 1). Most PROMs (pediatric and adult) were designed to capture HRLQOL, however, some assess specific aspects of the disease experience, ranging from overall life satisfaction and stress (IBDSI) to detailed concerns about treatment (RFIPC) and the impact on work productivity (WPAI:CD). A minority evaluates PRO regarding disease symptoms (TUMMY-UC and CD-HI)
The diversity in the number of items (ranging from 3 to 71), modes of administration (self-administered vs. interviewer-administered), and response options (from Likert scales to visual analog scales) reflects the complexity of score interpretation and limits everyday use, as the more comprehensive tools have higher respondent-burden. For example, the Crohn’s Disease–Health Index (CD-HI) is notable for its comprehensive nature, comprising 71 items, which allows for a detailed assessment of various health domains. In contrast, shorter forms, such as the SIBDQ, provide a more streamlined evaluation, which is suitable for quicker assessments.
The Crohn’s Disease Health Index (CD-HI) has recently been developed and validated as a disease-specific PROM with the aim of measuring the symptomatic burden of CD in adults [
38]. Although the research results supported the use of the CD-HI as a valid, sensitive, reliable, and relevant PRO in CD [
38], its use is not yet generalized. It was only tested in adult CD patients, and thus is not applicable to pediatric CD patients.
Language availability is also a crucial consideration, as multilanguage translation and validation is fundamental to accommodate diverse patient populations. Finaly the scarcity of pediatric PROMs underscores the necessity of age-appropriate assessments, acknowledging that IBD's impact can differ significantly between these groups and emphasizing the importance of developing and validating age-specific tools.
The plethora of IBD specific- PROMs suggests the necessity for continued refinement and standardization to ensure comprehensive, reliable, and universally applicable assessment tools, thus suggesting the usefulness of more generic PROMs. This is further reinforced by STRIDE II initiative. It recommends that QOL (including food-related QOL), disability, fatigue, depression, anxiety, sexual dysfunction, and body image, should be considered important PROMs that must be regularly assessed in patients with IBD (both adult and pediatric) [
36].
Current generic PROs measuring tools were developed as research tools and may not be appropriate to routine clinical practice, shorter and simpler measuring tools for everyday use are of the utmost importance. Also, useful generic PROs for clinical practice must have high reliability, face validity, construct validity, responsiveness, and feasibility [
36].
3. Patient-Reported Outcome Measurement Information System (PROMIS)
In 2004, the National Institutes of Health (NIH) from the United States launched the Patient-Reported Outcome Measurement Information System (PROMIS), which was developed to address, investigate and promote the use of PROs among patients with chronic conditions and to provide the next generation of standardized PRO measures in pediatric and adult health with improved reliability and validity (39-41). Their key objective is to provide PROs for research and clinical practice across the life course.
The PROMIS is a set of nondisease-specific instruments, one for adults and one for pediatric patients, that assess domains of physical, psychological, and social health, as well as QoL. These instruments are designed to be applicable to a range of chronic diseases, offering advantages over disease-specific instruments, as they allow for comparisons between different domains of health and across a wide range of adult and pediatric chronic diseases [19, 28-30, 32-35, 42, 43).
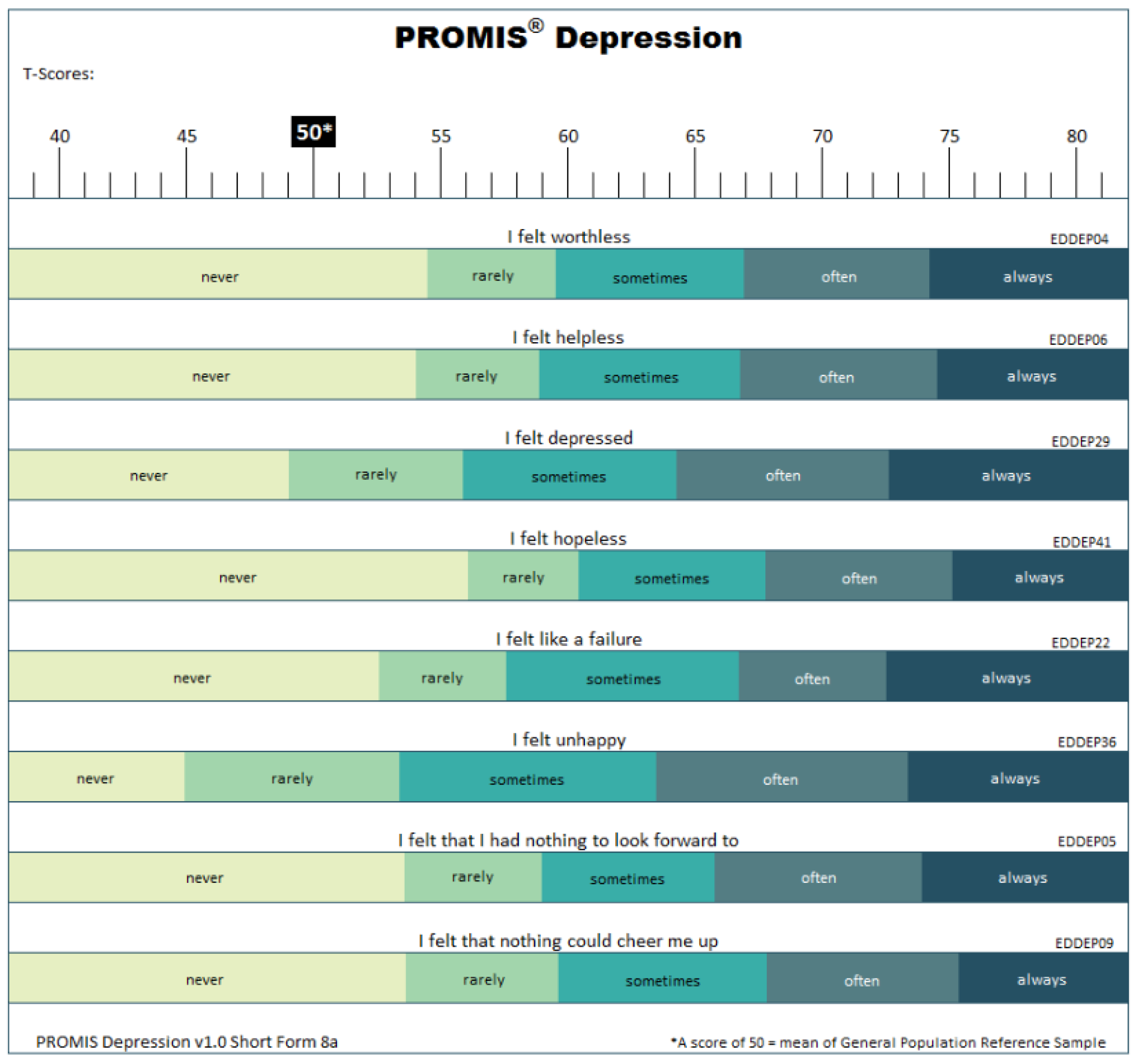
The PROMIS instruments are calibrated via a T score metric, with the mean of the original calibration population equal to 50 and the standard deviation (SD) equal to 10. Higher scores in any PROMIS domain indicate that more of the domain is being measured; for example, higher scores for anxiety, depression, fatigue, and pain interference indicate poorer well-being, whereas higher scores for peer relationships indicate better well-being [
44].
Figure 1 illustrates the measurement of the domains.
The ability of a PRO measure to detect change over time, when expected, defines responsiveness and supports the efficacy of the intervention or the disease treatment impact.
Minimally important differences (MIDs) are the smallest differences in PRO scores that detect a clinically meaningful change in the outcome the PRO is designed to measure [
45,
46]. Any differences in PRO scores smaller than the MID are likely due to measurement error rather than a true change in the outcome. Studies in adults suggest that MIDs for many PROMIS items range from 2–6 [
45] MIDs are an objective measure of assessing responsiveness over time.
4. Pediatric PROMIS
Like the adult PROMIS project, the PROMIS Pediatric multisite initiative developed, over the past ten years, several pediatric self-report item banks for individuals aged 8-17 years across five general health domains, namely, physical function, pain, fatigue, emotional health, and social health, on the basis of the larger PROMIS network, creating pediatric self-report scales measuring unidimensional health attributes (domains) of depressive symptoms, anxiety, anger, pain interference, peer relationships, fatigue, and physical functioning [44, 47, 48).
PROMIS pediatric measures were developed via qualitative and quantitative methods, including focus groups, expert item reviews, cognitive interviews, and item administration to a large population of North American children and adolescents. These methods created item banks specific to selected domains [
44,
48]. Like Adult PROMIS measures, pediatric measures are publicly available at no cost, offer flexible modes of administration, provide parent‒proxy report scales for children aged 5--7 years, can be customized to the disorder's unique features and potentially link measures across pediatric and adult age groups [
49].
Pediatric PROMIS instruments are calibrated via the same methodology as adult PROMIS instruments. Although MIDs are not yet well established in PROMIS pediatric measures, new research utilizing adolescent patients, parents and physicians as judges of clinically important differences in scores suggests MIDs of 2–3 for multiple pediatric PROMIS instruments. [
45]. A recent published study highlighted another possible approach, the use of disease-specific MIDs [
46]. The authors stated that some of the samples utilized to establish the metric and to determine the mean and standard deviation of the distributions were not representative of the general population, nor were they representative of a clinical population. Thus, they suggest the possibility of establishing disease-specific percentiles for PROMIS Pediatric measures and estimating clinically important changes by percentile changes (approximately one-half standard deviation units) [
46].
Data from several studies reinforce that Pediatric PROMIS measures are efficient, precise and valid across various diseases for assessing patient-reported symptoms and quality of life. They have been studied in chronic diseases, including sickle cell disease [
30,
33], kidney disease [
31,
33], cancer [
35,
50], rehabilitative needs [
28,
42], obesity [
33], asthma [
33,
34], rheumatic disease [
33,
34] and IBD [
22,
32,
46,
51,
52]. These cross-sectional studies validated the PROMIS Pediatric measures and demonstrated good discrimination among groups in terms of disease activity and severity [
33]. Furthermore, Pediatric PROMIS measures are discriminative among different clinically meaningful subgroups within several chronic illnesses.
A recent study [
22] reported that PROMIS® pediatric measures a particular ability to assess individual symptoms and functional impacts and to evaluate the effects of chronic disease activity on PROs in different chronic pediatric conditions, such as asthma, atopic dermatitis, cancer, cancer survivors, chronic kidney disease, Crohn’s disease, juvenile idiopathic arthritis, lupus, sickle cell disease, and type 1 diabetes mellitus. In this study, in all chronic conditions, children/adolescents reported more anxiety, fatigue, pain, and mobility restrictions than did the general pediatric population did, and disease activity was consistently associated with poorer self-reported health [
22], emphasizing the functional burden of chronic conditions and the importance of integrating self-reported PROs in the clinical assessment.
Furthermore, ongoing research has revealed evidence for the responsiveness of PROMIS Pediatric measures to clinical disease states in several chronic pediatric illnesses [
12,
30,
34,
52,
53], supporting the utilization of PROMIS Pediatric measures in clinical and research contexts. A multisite, multidisease longitudinal study evaluating the responsiveness of eight PROMIS Pediatric measures [
30] in children and adolescents (aged 8--17 years) with cancer (n=96), sickle cell disease (n=121), and nephrotic syndrome (n=127) provided evidence supporting the responsiveness of the PROMIS Pediatric measures for fatigue, pain interference, anxiety, anger, depressive symptoms, mobility, and upper extremity function. Additionally, they reported that the magnitude of change in these measures exceeded the MID of 3 points for all domains anticipated to change, thus indicating significant responsiveness.
This study also validates the ability of PROMIS measures to compare results across different chronic diseases. Using mixed modeling methods, the authors compared PROMIS scores in children and adolescents with cancer, nephrotic syndrome, and sickle cell disease in stable health states with scores when the child was experiencing a deteriorating health event.
Further evidence from studies evaluating the responsiveness of the PROMIS Pediatric measures has been reported. In a study involving 229 children (aged 8–17 years) with asthma [
34], data from the PROMIS Pediatric measures of pain interference, fatigue, depressive symptoms, mobility and peer relationships, and measures of asthma control were collected across four time points over 2 years. The study revealed that children with worsened asthma control and poorer overall health reported deteriorated function and greater symptom burden on the PROMIS Pediatric measures, with fatigue showing the greatest change.
Another study in pediatric oncology also supported the responsiveness of the PROMIS to changes over time [
35]. In this study, which included 96 pediatric oncology patients, eight PROMIS pediatric short-form measures were administered and compared with the Symptom Distress Scale at three time points during a course of chemotherapy. Most of the PROMIS pediatric measures demonstrated responsiveness and had significant relationships with the Symptom Distress Scale.
All studies emphasize the necessity of future research, including a broader pediatric population (in number and with diverse demographic characteristics), as well as the incorporation of biological and/or objective clinical anchors of disease.
5. IBD and PROMIS
The management of adult and pediatric CD patients has advanced significantly, with evolving therapeutic options and ambitious treatment goals. There is a paradigm shift from a disease-centric approach to a patient-centric approach, as emphasized by the STRIDE II [
36] recommendations for treat-to-target strategies in IBD.
In adult IBD patients, the use of the PROMIS was first reported in a cross-sectional and longitudinal study [
19] aiming to analyze and evaluate associations between PROMIS measures and validated disease activity indices (Short Crohn’s Disease Activity Index and Simple Clinical Colitis Activity Index) and the Short IBD Questionnaire quality of life instrument. The measured PROMIS domains included anxiety, depression, fatigue, sleep disturbance, satisfaction with social role, and pain interference. The study concluded that disease control was related to several health outcome measures reported by patients, providing strong support for the construct validity of the PROMIS in the IBD population. This study included a large number of geographically diverse patients; however, it was not performed in a real clinical setting, the IBD type and activity were self-reported, and clinical anchor disease assessments were lacking. Despite these limitations, this study represents a significant advancement in the applicability of the PROMIS in IBD patients.
Other studies sought to validate different PROMIS patient-reported measures in IBD patients (54-56). These studies evaluated social and emotional functioning, [
55,
56], and sexual function [
56] in adult IBD patients, supporting the validity of PROMIS measures in assessing HRQOL and functioning and further supporting the potential interest and relevance of using PROMIS tools in the clinical setting.
Recent studies addressing the PROMIS in IBD patients have extended the measures applied to evaluate both gastrointestinal symptoms [
54] and the burden of disease in different areas [
12,
57], enhancing its validity, reliability, and responsiveness for assessing general HRQOL and treatment response in IBD clinical trials and in the clinical setting.
Data on PROs in pediatric IBD are emerging [
22,
32,
46,
52,
53]. These studies, which use several PROMIS pediatric measures, consistently document similar results to adult IBD data, enhancing reliability with good correlation with clinical disease activity scores and with pediatric IBD-specific HRQOL scales.
In this setting, PROMIS pediatric tools have been used for the first time in a web-based cohort of 276 North American children with CD276 (aged 9–17 years). and in their parents [
32]. Data on self-reported CD activity, HRQOL (IMPACT-III), and the PROMIS domains of pain interference, anxiety, depression, fatigue, and peer relationships were recorded at baseline and six months later. Furthermore, children with improved Crohn’s disease activity reported improved scores on all PROMIS Pediatric measures, and children with worsened Crohn’s disease activity reported worse scores for all domains except anxiety. This study revealed that PROMIS scores were significantly related to CD activity in a linear and clinically meaningful manner and reflected the change in CD activity over a 6-month period, supporting the validity and responsiveness of the pediatric PROMIS within this time period.
More recently, two longitudinal studies documented evidence for the longitudinal responsiveness of the PROMIS Pediatric measures to changes in disease status and HRQOL in pediatric IBD patients and in both CD and UC pediatric patients [
52,
53]. Both studies were cross-sectional and longitudinal (follow-up of 6 months) and recruited patients aged 9--17 years from a North American internet-based cohort of children with IBD (IBD Partners Kids & Teens). Disease activity scores (short Crohn's Disease Activity Index and Pediatric Ulcerative Colitis Activity Index), the IMPACT-III, and 5 PROMIS Pediatric measures (anxiety, depressive symptoms, pain interference, fatigue, and peer relationships) were compared to examine the extent to which the PROMIS Pediatric measures responded to changes in clinical scores and IMPACT-III scores. A total of 635 pediatric IBD patients were included (544 CD patients and 91 UC patients), and in both studies, the results were similar, documenting a change in all PROMIS pediatric domains related to changes in clinical scores and changes in IMPACT-III scores. In both the cross-sectional and longitudinal analyses, better PROMIS Pediatric scores were associated with lower disease activity and improved IMPACT-III scores. These studies support the use of the PROMIS in pediatric IBD patients in clinical research as well as in patient care.
6. Conclusions
The incorporation of PROs into clinical practice for pediatric IBD represents a significant advancement in patient-centered care. IBD, as with other chronic conditions, particularly benefits from the detailed insights provided by PROs. These tools permit a comprehensive assessment of the patient's health, encompassing not only clinical symptoms but also the broader physical, psychological, and social dimensions of the disease. This patient-centered approach ensures that treatment plans are more aligned with patients' needs and expectations, thereby improving adherence and satisfaction with care (added health value). It is often the case that traditional clinical measures fail to capture the full extent of the disease's impact on a patient's life. PROs address this gap by offering insights into areas such as emotional well-being, social functioning, and daily activities. Furthermore, PROs facilitate more effective communication between patients, caregivers, and healthcare providers. By regularly monitoring these aspects, clinicians can make more informed decisions that aim to enhance overall quality of life. The utilization of PROMIS measures in pediatric IBD patients has been demonstrated to be reliable and valid, facilitating comprehensive assessments. Despite initial concerns about the time and effort required to complete PRO questionnaires, the integration of technology in administering these measures has made the process more efficient. The integration of electronic health records and automated systems allows for seamless collection and analysis of PRO data, thereby reducing the burden on patients and clinicians. In conclusion, the implementation of PROs in clinical practice for pediatric IBD should not be regarded merely as an adjunct but rather as a fundamental component of modern healthcare. These measures permit a more nuanced and real understanding of the patient’s condition, thereby facilitating the implementation of more personalized and effective care strategies. By prioritizing the patient’s voice in disease management, healthcare providers can significantly enhance the quality of life and overall well-being of pediatric IBD patients. This approach leads to better long-term outcomes and a more patient-centered approach to care.
Future Perspectives
Continued longitudinal studies and representative studies in the clinical setting are needed to further validate the clinical applicability in the real world and the responsiveness of PROMIS Pediatric measures across different stages of IBD.
It will be relevant to develop strategies to integrate PROs into routine clinical care. This encompasses the identification and resolution of obstacles, such as time constraints and respondent burden. The use of technology to facilitate the administration and monitoring of PROs has the potential to enhance patient engagement and data collection, which could result in the development of more personalized care plans. Finally, future research must define the ideal PROs for IBD [
37], which should be able to capture health status and impact QOL beyond specific disease-related symptoms; be applicable to both CD and UC patients and both adult and pediatric patients, including proxy measures; and be feasible for application in routine clinical practice, responsive to change and meaningful to pediatric patients.
Author Contributions
All authors read and approved the final manuscript. Conceptualization, data curation, investigation, methodology (SA), formal analysis (SA, AIL); supervision and validation (AIL). The first draft of the manuscript was written by (SA) and all authors commented on previous versions of the manuscript.
Funding
Not applicable. The authors declare that no funds, grants, or other support was received during the preparation of this manuscript.
DECLARATION OF INTEREST STATEMENT
ACKNOWLEDGMENTS.
Consent for publication
Not applicable.
Availability of data and materials
Not applicable; no additional data are available.
Competing interests
The authors declare that they have no competing interests.
Acknowledgments
Not applicable.
List of Abbreviations
Crohn’s disease (CD), health-related quality of life (HRQOL), inflammatory bowel disease (IBD), patient-reported outcomes (PROs), patient-reported outcome measurement information system (PROMIS), quality of life (QOL), therapeutic targets in inflammatory bowel disease (STRIDE-II), ulcerative colitis (UC), unclassified inflammatory bowel disease (IBD-U).
References
- van Rheenen, P.F.; Aloi, M.; Assa, A.; Bronsky, J.; Escher, J.C.; Fagerberg, U.L.; et al. The Medical Management of Paediatric Crohn's Disease: an ECCO-ESPGHAN Guideline Update. J Crohns Colitis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Herzer, M.; Denson, L.A.; Baldassano, R.N.; Hommel, K.A. Patient and parent psychosocial factors associated with health-related quality of life in pediatric inflammatory bowel disease. J Pediatr Gastroenterol Nutr. 2011, 52, 295–299. [Google Scholar] [CrossRef]
- Agrawal, M.; Spencer, E.A.; Colombel, J.F.; Ungaro, R.C. Approach to the Management of Recently Diagnosed Inflammatory Bowel Disease Patients: A User's Guide for Adult and Pediatric Gastroenterologists. Gastroenterology. 2021, 161, 47–65. [Google Scholar] [CrossRef] [PubMed]
- Achleitner, U.; Coenen, M.; Colombel, J.F.; Peyrin-Biroulet, L.; Sahakyan, N.; Cieza, A. Identification of areas of functioning and disability addressed in inflammatory bowel disease-specific patient reported outcome measures. J Crohns Colitis. 2012, 6, 507–517. [Google Scholar] [CrossRef] [PubMed]
- Gray, W.N.; Denson, L.A.; Baldassano, R.N.; Hommel, K.A. Disease activity, behavioral dysfunction, and health-related quality of life in adolescents with inflammatory bowel disease. Inflamm Bowel Dis. 2011, 17, 1581–1586. [Google Scholar] [CrossRef]
- Haapamäki, J.; Roine, R.P.; Sintonen, H.; Kolho, K.L. Health-related quality of life in paediatric patients with inflammatory bowel disease related to disease activity. J Paediatr Child Health. 2011, 47, 832–837. [Google Scholar] [CrossRef]
- Mueller, R.; Ziade, F.; Pittet, V.; Fournier, N.; Ezri, J.; Schoepfer, A.; et al. Quality of Life in Swiss Paediatric Inflammatory Bowel Disease Patients: Do Patients and Their Parents Experience Disease in the Same Way? J Crohns Colitis. 2016, 10, 269–276. [Google Scholar] [CrossRef]
- van der Have, M.; van der Aalst, K.S.; Kaptein, A.A.; Leenders, M.; Siersema, P.D.; Oldenburg, B.; et al. Determinants of health-related quality of life in Crohn's disease: a systematic review and meta-analysis. J Crohns Colitis. 2014, 8, 93–106. [Google Scholar] [CrossRef] [PubMed]
- Otley, A.; Smith, C.; Nicholas, D.; Munk, M.; Avolio, J.; Sherman, P.M.; et al. The IMPACT questionnaire: a valid measure of health-related quality of life in pediatric inflammatory bowel disease. J Pediatr Gastroenterol Nutr. 2002, 35, 557–563. [Google Scholar]
- Abdovic, S.; Mocic Pavic, A.; Milosevic, M.; Persic, M.; Senecic-Cala, I.; Kolacek, S. The IMPACT-III (HR) Questionnaire: A valid measure of health-related quality of life in Croatian children with inflammatory bowel disease☆. Journal of Crohn's and Colitis. 2013, 7, 908–915. [Google Scholar] [CrossRef]
- David Cella, E.A.H.; Sally, E. Jensen, Zeeshan Butt, Cindy J. Nowinski, Nan Rothrock, Kathleen N. Lohr. Patient-Reported Outcomes in Performance Measurement: RTI International; 2015. 95 p.
- Sands, B.E.; Panés, J.; Feagan, B.G.; Zhang, H.; Vetter, M.L.; Mathias, S.D.; et al. Qualitative and Psychometric Evaluation of PROMIS®-29 to Assess General Health-Related Quality of Life in Patients With Moderately to Severely Active Inflammatory Bowel Disease. Value Health. 2024. [Google Scholar] [CrossRef] [PubMed]
- Chouliaras, G.; Margoni, D.; Dimakou, K.; Fessatou, S.; Panayiotou, I.; Roma-Giannikou, E. Disease impact on the quality of life of children with inflammatory bowel disease. World J Gastroenterol. 2017, 23, 1067–1075. [Google Scholar] [CrossRef] [PubMed]
- El-Matary, W. Patient-reported outcome measures in inflammatory bowel disease. Can J Gastroenterol Hepatol. 2014, 28, 536–542. [Google Scholar] [CrossRef] [PubMed]
- Bodger, K.; Ormerod, C.; Shackcloth, D.; Harrison, M. Development and validation of a rapid, generic measure of disease control from the patient's perspective: the IBD-control questionnaire. Gut. 2014, 63, 1092–1102. [Google Scholar] [CrossRef]
- Bradley, C. Feedback on the FDA's February 2006 draft guidance on Patient Reported Outcome (PRO) measures from a developer of PRO measures. Health Qual Life Outcomes. 2006, 4, 78. [Google Scholar] [CrossRef]
- Reeve, B.B.; Burke, L.B.; Chiang, Y.P.; Clauser, S.B.; Colpe, L.J.; Elias, J.W.; et al. Enhancing measurement in health outcomes research supported by Agencies within the US Department of Health and Human Services. Qual Life Res. 2007, 16 (Suppl 1), 175–186. [Google Scholar] [CrossRef]
- Ruemmele, F.M.; Hyams, J.S.; Otley, A.; Griffiths, A.; Kolho, K.L.; Dias, J.A.; et al. Outcome measures for clinical trials in paediatric IBD: an evidence-based, expert-driven practical statement paper of the paediatric ECCO committee. Gut. 2015, 64, 438–446. [Google Scholar] [CrossRef]
- Kappelman, M.D.; Long, M.D.; Martin, C.; DeWalt, D.A.; Kinneer, P.M.; Chen, W.; et al. Evaluation of the patient-reported outcomes measurement information system in a large cohort of patients with inflammatory bowel diseases. Clin Gastroenterol Hepatol. 2014, 12, 1315–1323.e2. [Google Scholar] [CrossRef]
- Lohr, K.N.; Zebrack, B.J. Using patient-reported outcomes in clinical practice: challenges and opportunities. Qual Life Res. 2009, 18, 99–107. [Google Scholar] [CrossRef]
- Greenhalgh, J. The applications of PROs in clinical practice: what are they, do they work, and why? Qual Life Res. 2009, 18, 115–123. [Google Scholar] [CrossRef]
- Forrest, C.B.; Schuchard, J.; Bruno, C.; Amaral, S.; Cox, E.D.; Flynn, K.E.; et al. Self-Reported Health Outcomes of Children and Youth with 10 Chronic Diseases. J Pediatr. 2022, 246, 207–12.e1. [Google Scholar] [CrossRef] [PubMed]
- Fung, C.H.; Hays, R.D. Prospects and challenges in using patient-reported outcomes in clinical practice. Qual Life Res. 2008, 17, 1297–1302. [Google Scholar] [CrossRef] [PubMed]
- Osoba, D. Translating the science of patient-reported outcomes assessment into clinical practice. J Natl Cancer Inst Monogr. 2007, 5–11. [Google Scholar] [CrossRef]
- Valderas, J.M.; Kotzeva, A.; Espallargues, M.; Guyatt, G.; Ferrans, C.E.; Halyard, M.Y.; et al. The impact of measuring patient-reported outcomes in clinical practice: a systematic review of the literature. Qual Life Res. 2008, 17, 179–193. [Google Scholar] [CrossRef]
- Broderick, J.E.; DeWitt, E.M.; Rothrock, N.; Crane, P.K.; Forrest, C.B. Advances in Patient-Reported Outcomes: The NIH PROMIS(®) Measures. EGEMS (Wash DC). 2013, 1, 1015. [Google Scholar] [CrossRef]
- Rose, M.; Bezjak, A. Logistics of collecting patient-reported outcomes (PROs) in clinical practice: an overview and practical examples. Qual Life Res. 2009, 18, 125–136. [Google Scholar] [CrossRef]
- Mulcahey, M.J.; Haley, S.M.; Slavin, M.D.; Kisala, P.A.; Ni, P.; Tulsky, D.S.; et al. Ability of PROMIS Pediatric Measures to Detect Change in Children With Cerebral Palsy Undergoing Musculoskeletal Surgery. J Pediatr Orthop. 2016, 36, 749–756. [Google Scholar] [CrossRef] [PubMed]
- Gipson, D.S.; Selewski, D.T.; Massengill, S.F.; Wickman, L.; Messer, K.L.; Herreshoff, E.; et al. Gaining the PROMIS perspective from children with nephrotic syndrome: a Midwest pediatric nephrology consortium study. Health Qual Life Outcomes. 2013, 11, 30. [Google Scholar] [CrossRef]
- Reeve, B.B.; Edwards, L.J.; Jaeger, B.C.; Hinds, P.S.; Dampier, C.; Gipson, D.S.; et al. Assessing responsiveness over time of the PROMIS(®) pediatric symptom and function measures in cancer, nephrotic syndrome, and sickle cell disease. Qual Life Res. 2018, 27, 249–257. [Google Scholar] [CrossRef]
- Villegas, L.; Amaral, S. Patient-Reported Outcomes in Children With Chronic Kidney Disease. Semin Nephrol. 2021, 41, 476–484. [Google Scholar] [CrossRef]
- Arvanitis, M.; DeWalt, D.A.; Martin, C.F.; Long, M.D.; Chen, W.; Jaeger, B.; et al. Patient-Reported Outcomes Measurement Information System in Children with Crohn's Disease. J Pediatr. 2016, 174, 153–9.e2. [Google Scholar] [CrossRef] [PubMed]
- DeWalt, D.A.; Gross, H.E.; Gipson, D.S.; Selewski, D.T.; DeWitt, E.M.; Dampier, C.D.; et al. PROMIS(®) pediatric self-report scales distinguish subgroups of children within and across six common pediatric chronic health conditions. Qual Life Res. 2015, 24, 2195–2208. [Google Scholar] [CrossRef] [PubMed]
- Howell, C.R.; Thompson, L.A.; Gross, H.E.; Reeve, B.B.; DeWalt, D.A.; Huang, I.C. Responsiveness to Change in PROMIS(®) Measures among Children with Asthma: A Report from the PROMIS(®) Pediatric Asthma Study. Value Health. 2016, 19, 192–201. [Google Scholar] [CrossRef]
- Hinds, P.S.; Wang, J.; Cheng, Y.I.; Stern, E.; Waldron, M.; Gross, H.; et al. PROMIS pediatric measures validated in a longitudinal study design in pediatric oncology. Pediatr Blood Cancer. 2019, 66, e27606. [Google Scholar] [CrossRef] [PubMed]
- Turner, D.; Ricciuto, A.; Lewis, A.; D'Amico, F.; Dhaliwal, J.; Griffiths, A.M.; et al. STRIDE-II: An Update on the Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE) Initiative of the International Organization for the Study of IBD (IOIBD): Determining Therapeutic Goals for Treat-to-Target strategies in, I.B.D. Gastroenterology 2021, 160, 1570–1583. [Google Scholar] [CrossRef]
- Kim, A.H.; Roberts, C.; Feagan, B.G.; Banerjee, R.; Bemelman, W.; Bodger, K.; et al. Developing a Standard Set of Patient-Centred Outcomes for Inflammatory Bowel Disease-an International, Cross-disciplinary Consensus. J Crohns Colitis. 2018, 12, 408–418. [Google Scholar] [CrossRef]
- Varma, A.; Weinstein, J.; Seabury, J.; Rosero, S.; Wagner, E.; Zizzi, C.; et al. The Crohn's Disease-Health Index: Development and Evaluation of a Novel Outcome Measure. J Clin Gastroenterol. 2024. [Google Scholar] [CrossRef]
- Cella, D.; Yount, S.; Rothrock, N.; Gershon, R.; Cook, K.; Reeve, B.; et al. The Patient-Reported Outcomes Measurement Information System (PROMIS): progress of an NIH Roadmap cooperative group during its first two years. Med Care. 2007, 45, S3–S11. [Google Scholar] [CrossRef]
- Cella, D.; Riley, W.; Stone, A.; Rothrock, N.; Reeve, B.; Yount, S.; et al. The Patient-Reported Outcomes Measurement Information System (PROMIS) developed and tested its first wave of adult self-reported health outcome item banks: 2005-2008. J Clin Epidemiol. 2010, 63, 1179–1194. [Google Scholar] [CrossRef]
- Rothrock, N.E.; Hays, R.D.; Spritzer, K.; Yount, S.E.; Riley, W.; Cella, D. Relative to the general US population, chronic diseases are associated with poorer health-related quality of life as measured by the Patient-Reported Outcomes Measurement Information System (PROMIS). J Clin Epidemiol. 2010, 63, 1195–1204. [Google Scholar] [CrossRef]
- Kratz, A.L.; Slavin, M.D.; Mulcahey, M.J.; Jette, A.M.; Tulsky, D.S.; Haley, S.M. An examination of the PROMIS(®) pediatric instruments to assess mobility in children with cerebral palsy. Qual Life Res. 2013, 22, 2865–2876. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, M.T.; Block, A.M.; Ganapathy, A.K.; Huckleby, J.M.; Nepple, J.J. PROMIS Utilization in Pediatric Orthopaedics: A Scoping Review. J Pediatr Orthop. 2022, 42, 521–531. [Google Scholar] [CrossRef] [PubMed]
- Irwin, D.E.; Stucky, B.D.; Thissen, D.; Dewitt, E.M.; Lai, J.S.; Yeatts, K.; et al. Sampling plan and patient characteristics of the PROMIS pediatrics large-scale survey. Qual Life Res. 2010, 19, 585–594. [Google Scholar] [CrossRef] [PubMed]
- Thissen, D.; Liu, Y.; Magnus, B.; Quinn, H.; Gipson, D.S.; Dampier, C.; et al. Estimating minimally important difference (MID) in PROMIS pediatric measures using the scale-judgment method. Qual Life Res. 2016, 25, 13–23. [Google Scholar] [CrossRef]
- Schuchard, J.; Carle, A.C.; Kappelman, M.D.; Tucker, C.A.; Forrest, C.B. Interpreting Patient-Reported Outcome Scores: Pediatric Inflammatory Bowel Disease as a Use Case. Acad Pediatr. 2022, 22, 1520–1528. [Google Scholar] [CrossRef]
- Walsh, T.R.; Irwin, D.E.; Meier, A.; Varni, J.W.; DeWalt, D.A. The use of focus groups in the development of the PROMIS pediatrics item bank. Qual Life Res. 2008, 17, 725–735. [Google Scholar] [CrossRef]
- Irwin, D.E.; Varni, J.W.; Yeatts, K.; DeWalt, D.A. Cognitive interviewing methodology in the development of a pediatric item bank: a patient reported outcomes measurement information system (PROMIS) study. Health Qual Life Outcomes. 2009, 7, 3. [Google Scholar] [CrossRef]
- Varni, J.W.; Magnus, B.; Stucky, B.D.; Liu, Y.; Quinn, H.; Thissen, D.; et al. Psychometric properties of the PROMIS ® pediatric scales: precision, stability, and comparison of different scoring and administration options. Qual Life Res. 2014, 23, 1233–1243. [Google Scholar] [CrossRef]
- Daniel, L.C.; Gross, J.Y.; Meltzer, L.J.; Flannery, J.L.; Forrest, C.B.; Barakat, L.P. Clinical validity of the PROMIS pediatric sleep short forms in children receiving treatment for cancer. Pediatr Blood Cancer. 2020, 67, e28535. [Google Scholar] [CrossRef]
- Holbein, C.E.; Plevinsky, J.; Patel, T.; Conrad, M.C.; Kelsen, J.R. Pediatric Global Health in Children with Very Early-Onset Inflammatory Bowel Disease. J Pediatr Psychol. 2021, 46, 747–756. [Google Scholar] [CrossRef]
- Brenner, E.J.; Long, M.D.; Mann, C.M.; Chen, W.; Reyes, C.; Lin, L.; et al. Responsiveness of the Patient-reported Outcomes Measurement Information System (PROMIS) Pediatric Measures to Changes in Disease Status and Quality of Life Among Children and Adolescents With Crohn's Disease. Inflamm Bowel Dis. 2021, 27, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Brenner, E.J.; Long, M.D.; Mann, C.M.; Lin, L.; Chen, W.; Reyes, C.; et al. Validity and Responsiveness of the Patient-reported Outcomes Measurement Information System in Children With Ulcerative Colitis. J Pediatr Gastroenterol Nutr. 2021, 73, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Kochar, B.; Martin, C.F.; Kappelman, M.D.; Spiegel, B.M.; Chen, W.; Sandler, R.S.; et al. Evaluation of Gastrointestinal Patient Reported Outcomes Measurement Information System (GI-PROMIS) Symptom Scales in Subjects With Inflammatory Bowel Diseases. Am J Gastroenterol. 2018, 113, 72–79. [Google Scholar] [CrossRef]
- Bernstein, C.N.; Zhang, L.; Lix, L.M.; Graff, L.A.; Walker, J.R.; Fisk, J.D.; et al. The Validity and Reliability of Screening Measures for Depression and Anxiety Disorders in Inflammatory Bowel Disease. Inflamm Bowel Dis. 2018, 24, 1867–1875. [Google Scholar] [CrossRef] [PubMed]
- Eluri, S.; Cross, R.K.; Martin, C.; Weinfurt, K.P.; Flynn, K.E.; Long, M.D.; et al. Inflammatory Bowel Diseases Can Adversely Impact Domains of Sexual Function Such as Satisfaction with Sex Life. Dig Dis Sci. 2018, 63, 1572–1582. [Google Scholar] [CrossRef]
- Feagan, B.G.; Sandborn, W.J.; Sands, B.E.; Liu, Y.; Vetter, M.; Mathias, S.D.; et al. Qualitative and psychometric evaluation of the PROMIS®-Fatigue SF-7a scale to assess fatigue in patients with moderately to severely active inflammatory bowel disease. J Patient Rep Outcomes. 2023, 7, 115. [Google Scholar] [CrossRef] [PubMed]
- Joachim, G.; Milne, B. Inflammatory bowel disease: effects on lifestyle. J Adv Nurs. 1987, 12, 483–487. [Google Scholar] [CrossRef]
- Guyatt, G.; Mitchell, A.; Irvine, E.J.; Singer, J.; Williams, N.; Goodacre, R.; et al. A new measure of health status for clinical trials in inflammatory bowel disease. Gastroenterology. 1989, 96, 804–810. [Google Scholar] [CrossRef]
- Drossman, D.A.; Leserman, J.; Li, Z.M.; Mitchell, C.M.; Zagami, E.A.; Patrick, D.L. The rating form of IBD patient concerns: a new measure of health status. Psychosom Med. 1991, 53, 701–712. [Google Scholar] [CrossRef]
- Fazio, V.W.; O'Riordain, M.G.; Lavery, I.C.; Church, J.M.; Lau, P.; Strong, S.A.; et al. Long-term functional outcome and quality of life after stapled restorative proctocolectomy. Ann Surg. 1999, 230, 575–584. [Google Scholar] [CrossRef]
- Love, J.R.; Irvine, E.J.; Fedorak, R.N. Quality of life in inflammatory bowel disease. J Clin Gastroenterol. 1992, 14, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Irvine, E.J. Development and subsequent refinement of the inflammatory bowel disease questionnaire: a quality-of-life instrument for adult patients with inflammatory bowel disease. J Pediatr Gastroenterol Nutr. 1999, 28, S23–S27. [Google Scholar] [CrossRef] [PubMed]
- Irvine, E.J.; Zhou, Q.; Thompson, A.K. The Short Inflammatory Bowel Disease Questionnaire: a quality of life instrument for community physicians managing inflammatory bowel disease. CCRPT Investigators. Canadian Crohn's Relapse Prevention Trial. Am J Gastroenterol. 1996, 91, 1571–1578. [Google Scholar] [PubMed]
- Reilly, M.C.; Gerlier, L.; Brabant, Y.; Brown, M. Validity, reliability, and responsiveness of the work productivity and activity impairment questionnaire in Crohn's disease. Clin Ther. 2008, 30, 393–404. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.; Leone, L.; Fries, W.; Naccarato, R. Quality of life in inflammatory bowel disease. Ital J Gastroenterol. 1995, 27, 450–454. [Google Scholar] [PubMed]
- Hjortswang, H.; Järnerot, G.; Curman, B.; Sandberg-Gertzén, H.; Tysk, C.; Blomberg, B.; et al. The Short Health Scale: a valid measure of subjective health in ulcerative colitis. Scand J Gastroenterol. 2006, 41, 1196–1203. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.D.; Watson, R.; Palmer, K.R. Inflammatory bowel disease: developing a short disease specific scale to measure health related quality of life. Int J Nurs Stud. 2002, 39, 583–590. [Google Scholar] [CrossRef]
- Alrubaiy, L.; Cheung, W.Y.; Dodds, P.; Hutchings, H.A.; Russell, I.T.; Watkins, A.; et al. Development of a short questionnaire to assess the quality of life in Crohn's disease and ulcerative colitis. J Crohns Colitis. 2015, 9, 66–76. [Google Scholar] [CrossRef]
- Griffiths, A.M.; Nicholas, D.; Smith, C.; Munk, M.; Stephens, D.; Durno, C.; et al. Development of a quality-of-life index for pediatric inflammatory bowel disease: dealing with differences related to age and IBD type. J Pediatr Gastroenterol Nutr. 1999, 28, S46–S52. [Google Scholar] [CrossRef]
- Marcovitch, L.; Nissan, A.; Mack, D.; Otley, A.; Hussey, S.; McLean, B.; et al. Item Generation and Reduction Toward Developing a Patient-reported Outcome for Pediatric Ulcerative Colitis (TUMMY-UC). J Pediatr Gastroenterol Nutr. 2017, 64, 373–377. [Google Scholar] [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).