Submitted:
11 November 2024
Posted:
12 November 2024
You are already at the latest version
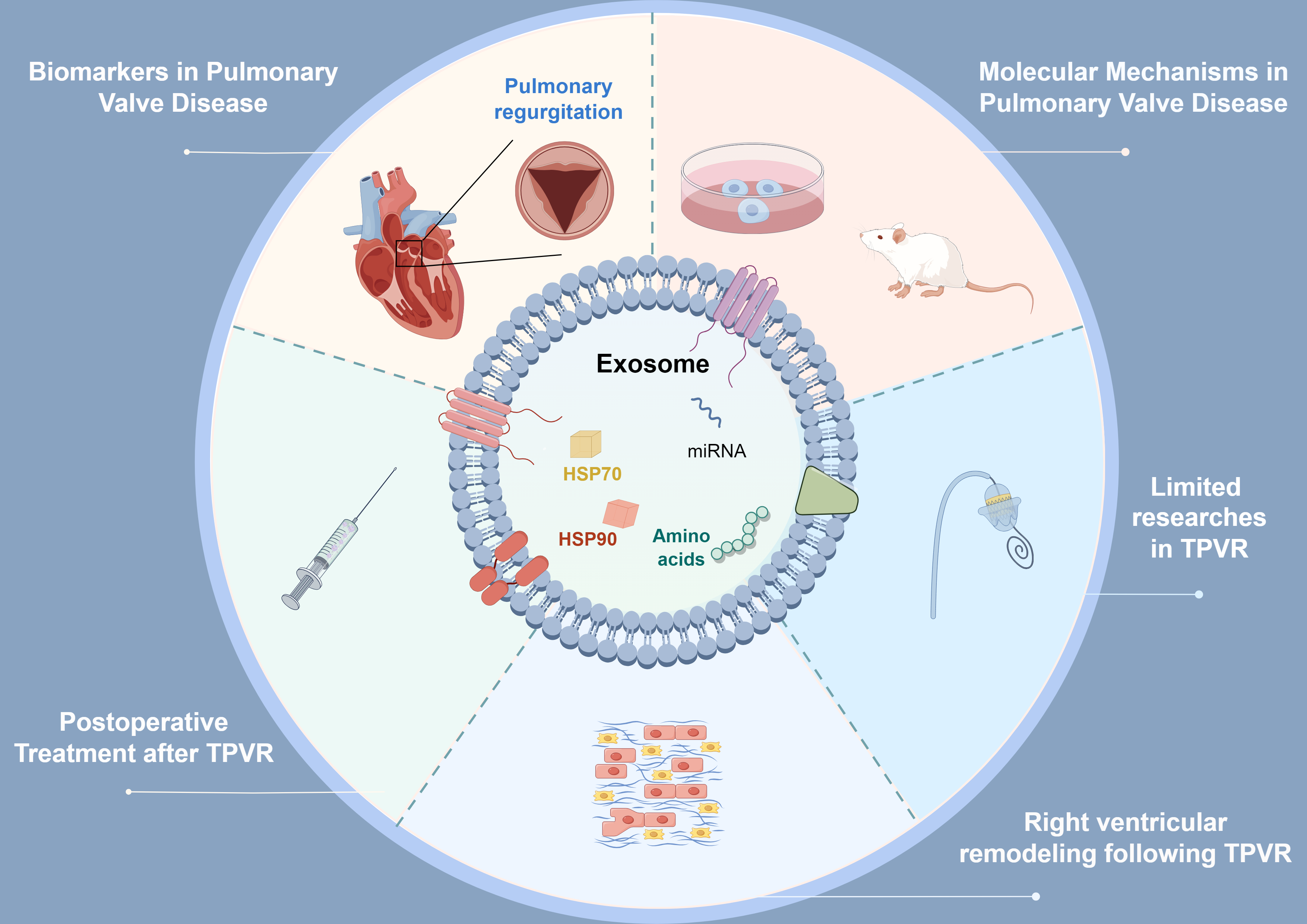
Abstract
Keywords:
1. Introduction
2. Physiological Characteristics of Exosomes
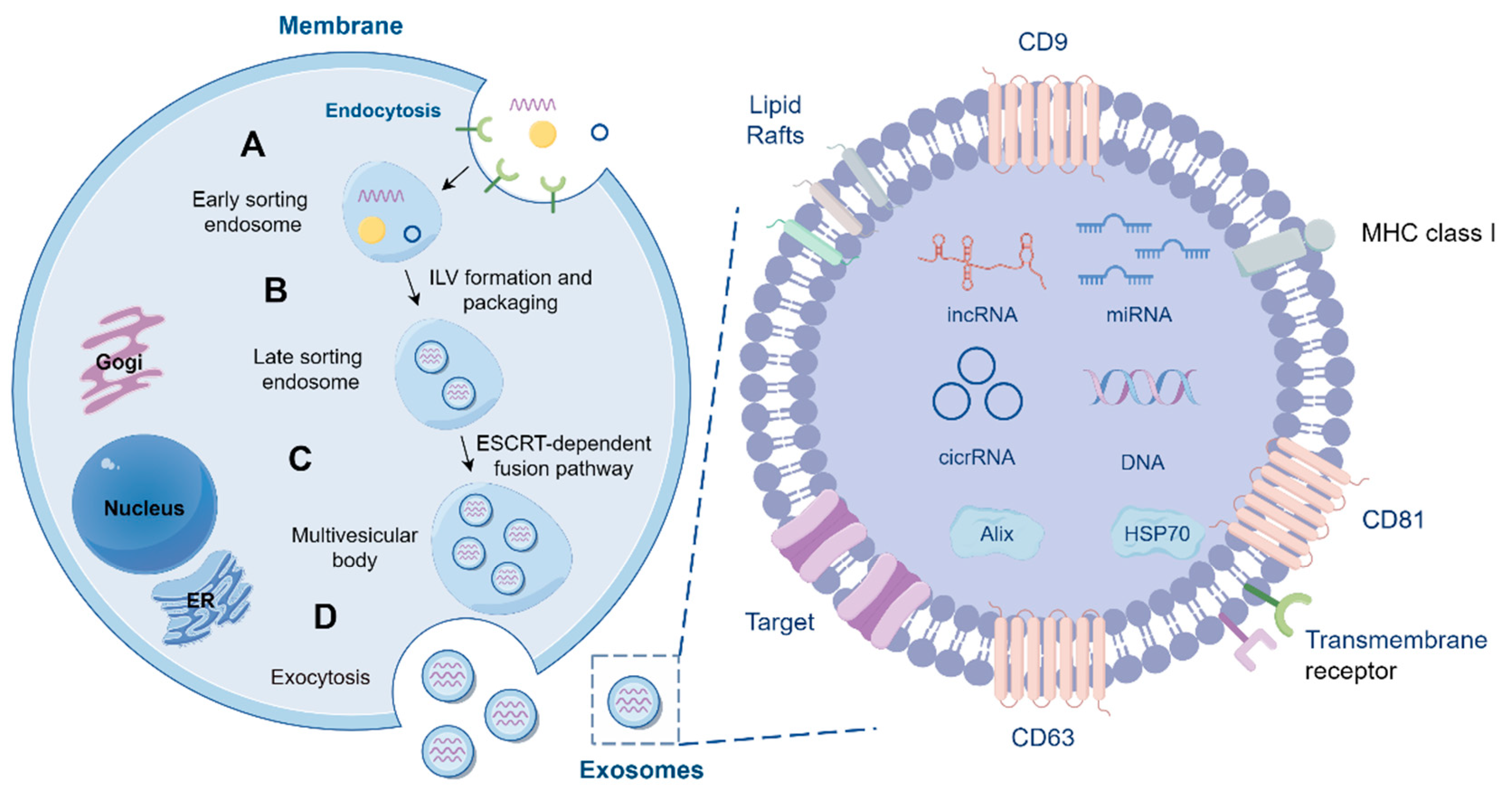
2.1. The Biogenesis of Exosomes
2.2. The Molecular Composition of EVs
3. The Role of miRNA in PVD
4. miRNA Aids in Determining the Optimal Timing for TPVR Intervention
5. Exosomes and RV Remodeling Following TPVR
6. Exosomes and Postoperative Treatment After TPVR
6.1. The Impact of TPVR on RV Function
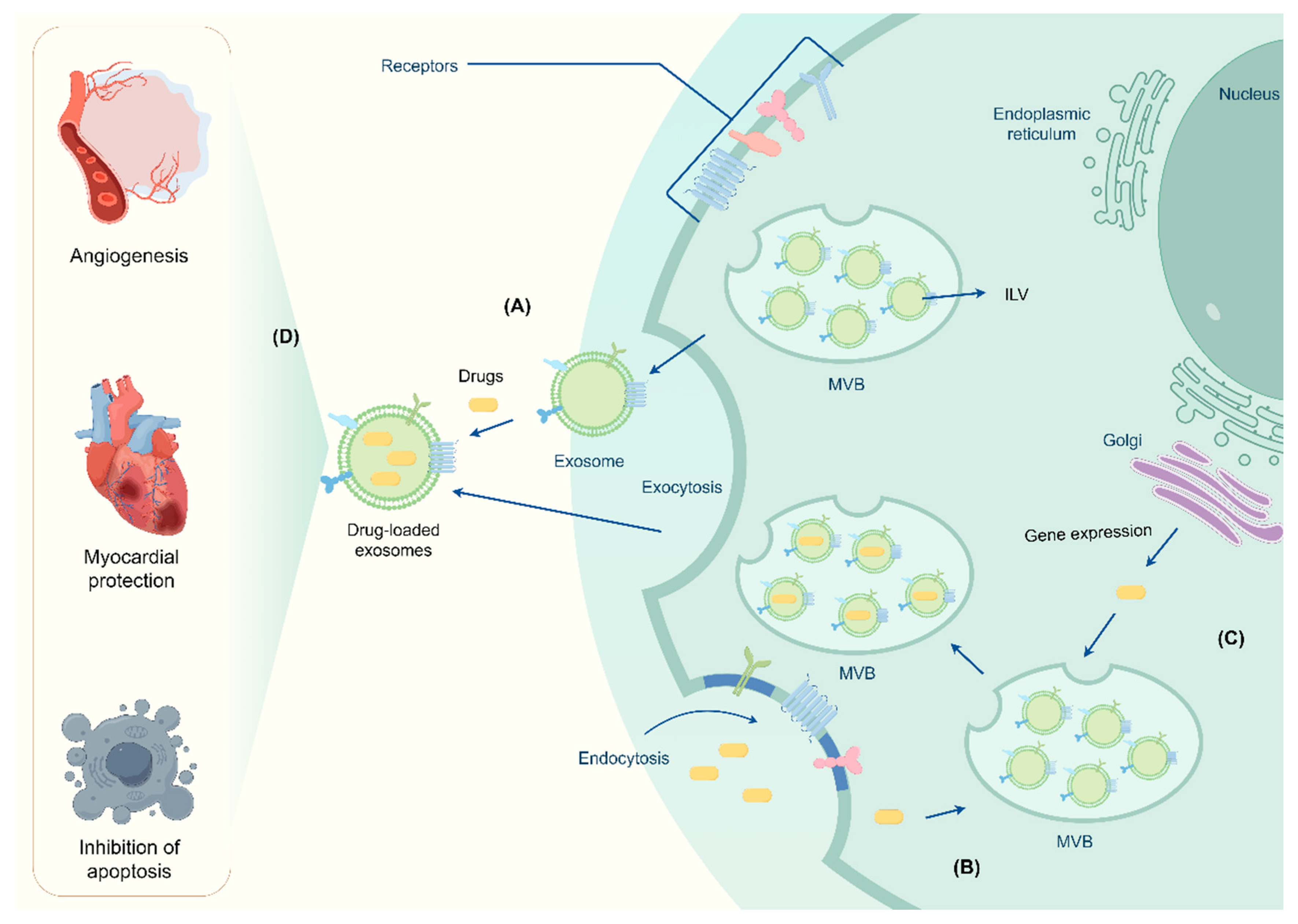
6.2. Exosomes as Drug Carriers to Improve Right Heart Function
7. Conclusion and Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Colombo, M.; Raposo, G.; Thery, C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu Rev Cell Dev Biol 2014, 30, 255–289. [Google Scholar] [CrossRef] [PubMed]
- Yanez-Mo, M.; Siljander, P.R.; Andreu, Z.; Zavec, A.B.; Borras, F.E.; Buzas, E.I.; Buzas, K.; Casal, E.; Cappello, F.; Carvalho, J.; et al. Biological properties of extracellular vesicles and their physiological functions. J Extracell Vesicles 2015, 4, 27066. [Google Scholar] [CrossRef] [PubMed]
- Livshits, M.A.; Khomyakova, E.; Evtushenko, E.G.; Lazarev, V.N.; Kulemin, N.A.; Semina, S.E.; Generozov, E.V.; Govorun, V.M. Isolation of exosomes by differential centrifugation: Theoretical analysis of a commonly used protocol. Sci Rep 2015, 5, 17319. [Google Scholar] [CrossRef] [PubMed]
- Tenchov, R.; Sasso, J.M.; Wang, X.; Liaw, W.S.; Chen, C.A.; Zhou, Q.A. Exosomes horizontal line Nature's Lipid Nanoparticles, a Rising Star in Drug Delivery and Diagnostics. ACS Nano 2022, 16, 17802–17846. [Google Scholar] [CrossRef]
- Rai, A.; Claridge, B.; Lozano, J.; Greening, D.W. The Discovery of Extracellular Vesicles and Their Emergence as a Next-Generation Therapy. Circ Res 2024, 135, 198–221. [Google Scholar] [CrossRef]
- Trams, E.G.; Lauter, C.J.; Salem, N., Jr.; Heine, U. Exfoliation of membrane ecto-enzymes in the form of micro-vesicles. Biochim Biophys Acta 1981, 645, 63–70. [Google Scholar] [CrossRef]
- Valadi, H.; Ekstrom, K.; Bossios, A.; Sjostrand, M.; Lee, J.J.; Lotvall, J.O. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol 2007, 9, 654–659. [Google Scholar] [CrossRef]
- Beaumont, J.; Lopez, B.; Ravassa, S.; Hermida, N.; San Jose, G.; Gallego, I.; Valencia, F.; Gomez-Doblas, J.J.; de Teresa, E.; Diez, J.; et al. MicroRNA-19b is a potential biomarker of increased myocardial collagen cross-linking in patients with aortic stenosis and heart failure. Sci Rep 2017, 7, 40696. [Google Scholar] [CrossRef]
- Zhang, W.; Hua, Y.; Zheng, D.; Chen, Q.; Huang, R.; Wang, W.; Li, X. Expression and clinical significance of miR-8078 in patients with congenital heart disease-associated pulmonary arterial hypertension. Gene 2024, 896, 147964. [Google Scholar] [CrossRef]
- Goren, Y.; Kushnir, M.; Zafrir, B.; Tabak, S.; Lewis, B.S.; Amir, O. Serum levels of microRNAs in patients with heart failure. Eur J Heart Fail 2012, 14, 147–154. [Google Scholar] [CrossRef]
- Wang, G.K.; Zhu, J.Q.; Zhang, J.T.; Li, Q.; Li, Y.; He, J.; Qin, Y.W.; Jing, Q. Circulating microRNA: a novel potential biomarker for early diagnosis of acute myocardial infarction in humans. Eur Heart J 2010, 31, 659–666. [Google Scholar] [CrossRef] [PubMed]
- Fusco, C.; De Rosa, G.; Spatocco, I.; Vitiello, E.; Procaccini, C.; Frige, C.; Pellegrini, V.; La Grotta, R.; Furlan, R.; Matarese, G.; et al. Extracellular vesicles as human therapeutics: A scoping review of the literature. J Extracell Vesicles 2024, 13, e12433. [Google Scholar] [CrossRef]
- Jia, L.; Limeng, D.; Xiaoyin, T.; Junwen, W.; Xintong, Z.; Gang, X.; Yun, B.; Hong, G. A Novel Splicing Mutation c.335-1 G > A in the Cardiac Transcription Factor NKX2-5 Leads to Familial Atrial Septal Defect Through miR-19 and PYK2. Stem Cell Rev Rep 2022, 18, 2646–2661. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Higgins, H.; Guo, J.; Harrison, K.; Schultz, E.N.; Hales, B.J.; Moses, E.K.; Goldblatt, J.; Pachter, N.; Zhang, G. Clinical significance of circulating microRNAs as markers in detecting and predicting congenital heart defects in children. J Transl Med 2018, 16, 42. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.; Rajakaruna, C.; Caputo, M.; Emanueli, C. MicroRNAs in congenital heart disease. Ann Transl Med 2015, 3, 333. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Peng, B.; Han, Y. MiR-182 alleviates the development of cyanotic congenital heart disease by suppressing HES1. Eur J Pharmacol 2018, 836, 18–24. [Google Scholar] [CrossRef]
- Batkai, S.; Bar, C.; Thum, T. MicroRNAs in right ventricular remodelling. Cardiovasc Res 2017, 113, 1433–1440. [Google Scholar] [CrossRef]
- DiLorenzo, M.P.; DeCost, G.; Mai, A.D.; Hughes, N.; Goldmuntz, E.; Jones, A.; Fogel, M.A.; Mercer-Rosa, L. Comparison of serum biomarkers of myocardial fibrosis with cardiac magnetic resonance in patients operated for tetralogy of Fallot. Int J Cardiol 2022, 358, 27–33. [Google Scholar] [CrossRef]
- McElhinney, D.B.; Hennesen, J.T. The Melody(R) valve and Ensemble(R) delivery system for transcatheter pulmonary valve replacement. Ann N Y Acad Sci 2013, 1291, 77–85. [Google Scholar] [CrossRef]
- Gatzoulis, M.A.; Balaji, S.; Webber, S.A.; Siu, S.C.; Hokanson, J.S.; Poile, C.; Rosenthal, M.; Nakazawa, M.; Moller, J.H.; Gillette, P.C.; et al. Risk factors for arrhythmia and sudden cardiac death late after repair of tetralogy of Fallot: a multicentre study. Lancet 2000, 356, 975–981. [Google Scholar] [CrossRef]
- Bonhoeffer, P.; Boudjemline, Y.; Saliba, Z.; Merckx, J.; Aggoun, Y.; Bonnet, D.; Acar, P.; Le Bidois, J.; Sidi, D.; Kachaner, J. Percutaneous replacement of pulmonary valve in a right-ventricle to pulmonary-artery prosthetic conduit with valve dysfunction. Lancet 2000, 356, 1403–1405. [Google Scholar] [CrossRef] [PubMed]
- Bonhoeffer, P.; Boudjemline, Y.; Saliba, Z.; Hausse, A.O.; Aggoun, Y.; Bonnet, D.; Sidi, D.; Kachaner, J. Transcatheter implantation of a bovine valve in pulmonary position: a lamb study. Circulation 2000, 102, 813–816. [Google Scholar] [CrossRef] [PubMed]
- Georgiev, S.; Ewert, P.; Eicken, A.; Hager, A.; Horer, J.; Cleuziou, J.; Meierhofer, C.; Tanase, D. Munich Comparative Study: Prospective Long-Term Outcome of the Transcatheter Melody Valve Versus Surgical Pulmonary Bioprosthesis With Up to 12 Years of Follow-Up. Circ Cardiovasc Interv 2020, 13, e008963. [Google Scholar] [CrossRef] [PubMed]
- Gould, G.W.; Lippincott-Schwartz, J. New roles for endosomes: from vesicular carriers to multi-purpose platforms. Nat Rev Mol Cell Biol 2009, 10, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Klumperman, J.; Raposo, G. The complex ultrastructure of the endolysosomal system. Cold Spring Harb Perspect Biol 2014, 6, a016857. [Google Scholar] [CrossRef]
- Kalluri, R.; LeBleu, V.S. The biology, function, and biomedical applications of exosomes. Science 2020, 367. [Google Scholar] [CrossRef]
- Stoorvogel, W.; Strous, G.J.; Geuze, H.J.; Oorschot, V.; Schwartz, A.L. Late endosomes derive from early endosomes by maturation. Cell 1991, 65, 417–427. [Google Scholar] [CrossRef]
- Babst, M.; Katzmann, D.J.; Estepa-Sabal, E.J.; Meerloo, T.; Emr, S.D. Escrt-III: an endosome-associated heterooligomeric protein complex required for mvb sorting. Dev Cell 2002, 3, 271–282. [Google Scholar] [CrossRef]
- Kahlert, C.; Kalluri, R. Exosomes in tumor microenvironment influence cancer progression and metastasis. J Mol Med (Berl) 2013, 91, 431–437. [Google Scholar] [CrossRef]
- van Niel, G.; D'Angelo, G.; Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat Rev Mol Cell Biol 2018, 19, 213–228. [Google Scholar] [CrossRef]
- Jaiswal, J.K.; Andrews, N.W.; Simon, S.M. Membrane proximal lysosomes are the major vesicles responsible for calcium-dependent exocytosis in nonsecretory cells. J Cell Biol 2002, 159, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Keerthikumar, S.; Chisanga, D.; Ariyaratne, D.; Al Saffar, H.; Anand, S.; Zhao, K.; Samuel, M.; Pathan, M.; Jois, M.; Chilamkurti, N.; et al. ExoCarta: A Web-Based Compendium of Exosomal Cargo. J Mol Biol 2016, 428, 688–692. [Google Scholar] [CrossRef] [PubMed]
- Pathan, M.; Fonseka, P.; Chitti, S.V.; Kang, T.; Sanwlani, R.; Van Deun, J.; Hendrix, A.; Mathivanan, S. Vesiclepedia 2019: a compendium of RNA, proteins, lipids and metabolites in extracellular vesicles. Nucleic Acids Res 2019, 47, D516–D519. [Google Scholar] [CrossRef] [PubMed]
- van Balkom, B.W.; Eisele, A.S.; Pegtel, D.M.; Bervoets, S.; Verhaar, M.C. Quantitative and qualitative analysis of small RNAs in human endothelial cells and exosomes provides insights into localized RNA processing, degradation and sorting. J Extracell Vesicles 2015, 4, 26760. [Google Scholar] [CrossRef] [PubMed]
- Bellingham, S.A.; Coleman, B.M.; Hill, A.F. Small RNA deep sequencing reveals a distinct miRNA signature released in exosomes from prion-infected neuronal cells. Nucleic Acids Res 2012, 40, 10937–10949. [Google Scholar] [CrossRef]
- Hemler, M.E. Tetraspanin proteins mediate cellular penetration, invasion, and fusion events and define a novel type of membrane microdomain. Annu Rev Cell Dev Biol 2003, 19, 397–422. [Google Scholar] [CrossRef]
- Hemler, M.E. Targeting of tetraspanin proteins--potential benefits and strategies. Nat Rev Drug Discov 2008, 7, 747–758. [Google Scholar] [CrossRef]
- Kowal, J.; Arras, G.; Colombo, M.; Jouve, M.; Morath, J.P.; Primdal-Bengtson, B.; Dingli, F.; Loew, D.; Tkach, M.; Thery, C. Proteomic comparison defines novel markers to characterize heterogeneous populations of extracellular vesicle subtypes. Proc Natl Acad Sci U S A 2016, 113, E968–977. [Google Scholar] [CrossRef]
- Viereck, J.; Thum, T. Circulating Noncoding RNAs as Biomarkers of Cardiovascular Disease and Injury. Circ Res 2017, 120, 381–399. [Google Scholar] [CrossRef]
- Thum, T.; Condorelli, G. Long noncoding RNAs and microRNAs in cardiovascular pathophysiology. Circ Res 2015, 116, 751–762. [Google Scholar] [CrossRef]
- Xie, W.Q.; Zhou, L.; Chen, Y.; Ni, B. Circulating microRNAs as potential biomarkers for diagnosis of congenital heart defects. World J Emerg Med 2016, 7, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Kalayinia, S.; Arjmand, F.; Maleki, M.; Malakootian, M.; Singh, C.P. MicroRNAs: roles in cardiovascular development and disease. Cardiovasc Pathol 2021, 50, 107296. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.; Xu, X.; Deng, F.; Feng, J.; Zhang, H.; Liu, Y.; Zhang, Y.; Pan, L.; Liu, Y.; Zhang, D.; et al. miRNA-940 reduction contributes to human Tetralogy of Fallot development. J Cell Mol Med 2014, 18, 1830–1839. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Ma, X.J.; Wang, H.J.; Li, W.C.; Chen, L.; Ma, D.; Huang, G.Y. Expression of Cx43-related microRNAs in patients with tetralogy of Fallot. World J Pediatr 2014, 10, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Mosquera-Heredia, M.I.; Morales, L.C.; Vidal, O.M.; Barcelo, E.; Silvera-Redondo, C.; Velez, J.I.; Garavito-Galofre, P. Exosomes: Potential Disease Biomarkers and New Therapeutic Targets. Biomedicines 2021, 9. [Google Scholar] [CrossRef]
- Weldy, C.S.; Syed, S.A.; Amsallem, M.; Hu, D.Q.; Ji, X.; Punn, R.; Taylor, A.; Navarre, B.; Reddy, S. Circulating whole genome miRNA expression corresponds to progressive right ventricle enlargement and systolic dysfunction in adults with tetralogy of Fallot. PLoS One 2020, 15, e0241476. [Google Scholar] [CrossRef]
- Abu-Halima, M.; Meese, E.; Abdul-Khaliq, H.; Raedle-Hurst, T. MicroRNA-183-3p Is a Predictor of Worsening Heart Failure in Adult Patients With Transposition of the Great Arteries and a Systemic Right Ventricle. Front Cardiovasc Med 2021, 8, 730364. [Google Scholar] [CrossRef]
- Tsao, C.W.; Aday, A.W.; Almarzooq, Z.I.; Anderson, C.A.M.; Arora, P.; Avery, C.L.; Baker-Smith, C.M.; Beaton, A.Z.; Boehme, A.K.; Buxton, A.E.; et al. Heart Disease and Stroke Statistics-2023 Update: A Report From the American Heart Association. Circulation 2023, 147, e93–e621. [Google Scholar] [CrossRef]
- Yanagawa, B.; Lovren, F.; Pan, Y.; Garg, V.; Quan, A.; Tang, G.; Singh, K.K.; Shukla, P.C.; Kalra, N.P.; Peterson, M.D.; et al. miRNA-141 is a novel regulator of BMP-2-mediated calcification in aortic stenosis. J Thorac Cardiovasc Surg 2012, 144, 256–262. [Google Scholar] [CrossRef]
- Zhang, M.; Liu, X.; Zhang, X.; Song, Z.; Han, L.; He, Y.; Xu, Z. MicroRNA-30b is a multifunctional regulator of aortic valve interstitial cells. J Thorac Cardiovasc Surg 2014, 147, 1073–1080.e1072. [Google Scholar] [CrossRef] [PubMed]
- Ohukainen, P.; Syvaranta, S.; Napankangas, J.; Rajamaki, K.; Taskinen, P.; Peltonen, T.; Helske-Suihko, S.; Kovanen, P.T.; Ruskoaho, H.; Rysa, J. MicroRNA-125b and chemokine CCL4 expression are associated with calcific aortic valve disease. Ann Med 2015, 47, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Hosen, M.R.; Goody, P.R.; Zietzer, A.; Xiang, X.; Niepmann, S.T.; Sedaghat, A.; Tiyerili, V.; Chennupati, R.; Moore, J.B.t.; Boon, R.A.; et al. Circulating MicroRNA-122-5p Is Associated With a Lack of Improvement in Left Ventricular Function After Transcatheter Aortic Valve Replacement and Regulates Viability of Cardiomyocytes Through Extracellular Vesicles. Circulation 2022, 146, 1836–1854. [Google Scholar] [CrossRef] [PubMed]
- Kleeberger, J.A.; Neuser, J.; de Gonzalo-Calvo, D.; Kempf, T.; Bauersachs, J.; Thum, T.; Widder, J.D. microRNA-206 correlates with left ventricular function after transcatheter aortic valve implantation. Am J Physiol Heart Circ Physiol 2017, 313, H1261–H1266. [Google Scholar] [CrossRef] [PubMed]
- Toutouzas, K.; Stathogiannis, K.; Latsios, G.; Synetos, A.; Drakopoulou, M.; Penesopoulou, V.; Michelongona, A.; Tsiamis, E.; Tousoulis, D. Biomarkers in Aortic Valve Stenosis and their Clinical Significance in Transcatheter Aortic Valve Implantation. Curr Med Chem 2019, 26, 864–872. [Google Scholar] [CrossRef]
- Chen, M.C.; Chang, T.H.; Chang, J.P.; Huang, H.D.; Ho, W.C.; Lin, Y.S.; Pan, K.L.; Liu, W.H.; Huang, Y.K. Circulating miR-148b-3p and miR-409-3p as biomarkers for heart failure in patients with mitral regurgitation. Int J Cardiol 2016, 222, 148–154. [Google Scholar] [CrossRef]
- Pizzino, F.; Furini, G.; Casieri, V.; Mariani, M.; Bianchi, G.; Storti, S.; Chiappino, D.; Maffei, S.; Solinas, M.; Aquaro, G.D.; et al. Late plasma exosome microRNA-21-5p depicts magnitude of reverse ventricular remodeling after early surgical repair of primary mitral valve regurgitation. Front Cardiovasc Med 2022, 9, 943068. [Google Scholar] [CrossRef]
- Hinojar, R.; Moreno-Gomez-Toledano, R.; Conde, E.; Gonzalez-Gomez, A.; Garcia-Martin, A.; Gonzalez-Portilla, P.; Fernandez-Golfin, C.; Garcia-Bermejo, M.L.; Zaragoza, C.; Zamorano, J.L. Circulating miRNA in functional tricuspid regurgitation. Unveiling novel links to heart failure: A pilot study. ESC Heart Fail 2024, 11, 2272–2286. [Google Scholar] [CrossRef]
- Abu-Halima, M.; Meese, E.; Keller, A.; Abdul-Khaliq, H.; Radle-Hurst, T. Analysis of circulating microRNAs in patients with repaired Tetralogy of Fallot with and without heart failure. J Transl Med 2017, 15, 156. [Google Scholar] [CrossRef]
- Lai, C.T.; Ng, E.K.; Chow, P.C.; Kwong, A.; Cheung, Y.F. Circulating microRNA expression profile and systemic right ventricular function in adults after atrial switch operation for complete transposition of the great arteries. BMC Cardiovasc Disord 2013, 13, 73. [Google Scholar] [CrossRef]
- Lai, C.T.M.; Ng, E.K.O.; Chow, P.C.; Kwong, A.; Cheung, Y.F. Circulating MicroRNA in patients with repaired tetralogy of Fallot. Eur J Clin Invest 2017, 47, 574–582. [Google Scholar] [CrossRef]
- Shi, L.; Kojonazarov, B.; Elgheznawy, A.; Popp, R.; Dahal, B.K.; Bohm, M.; Pullamsetti, S.S.; Ghofrani, H.A.; Godecke, A.; Jungmann, A.; et al. miR-223-IGF-IR signalling in hypoxia- and load-induced right-ventricular failure: a novel therapeutic approach. Cardiovasc Res 2016, 111, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Ye, P.; Zhang, A.K.; Yu, W.D.; Lin, S.; Zheng, Y.G. Upregulation of miR-335-5p Contributes to Right Ventricular Remodeling via Calumenin in Pulmonary Arterial Hypertension. Biomed Res Int 2022, 2022, 9294148. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Liu, H.; Gao, W.; Zhang, L.; Ye, Y.; Yuan, L.; Ding, Z.; Wu, J.; Kang, L.; Zhang, X.; et al. MicroRNA-378 suppresses myocardial fibrosis through a paracrine mechanism at the early stage of cardiac hypertrophy following mechanical stress. Theranostics 2018, 8, 2565–2582. [Google Scholar] [CrossRef] [PubMed]
- Beltrami, C.; Besnier, M.; Shantikumar, S.; Shearn, A.I.; Rajakaruna, C.; Laftah, A.; Sessa, F.; Spinetti, G.; Petretto, E.; Angelini, G.D.; et al. Human Pericardial Fluid Contains Exosomes Enriched with Cardiovascular-Expressed MicroRNAs and Promotes Therapeutic Angiogenesis. Mol Ther 2017, 25, 679–693. [Google Scholar] [CrossRef] [PubMed]
- Barile, L.; Lionetti, V.; Cervio, E.; Matteucci, M.; Gherghiceanu, M.; Popescu, L.M.; Torre, T.; Siclari, F.; Moccetti, T.; Vassalli, G. Extracellular vesicles from human cardiac progenitor cells inhibit cardiomyocyte apoptosis and improve cardiac function after myocardial infarction. Cardiovasc Res 2014, 103, 530–541. [Google Scholar] [CrossRef]
- Chen, Y.T.; Wang, J.; Wee, A.S.; Yong, Q.W.; Tay, E.L.; Woo, C.C.; Sorokin, V.; Richards, A.M.; Ling, L.H. Differential MicroRNA Expression Profile in Myxomatous Mitral Valve Prolapse and Fibroelastic Deficiency Valves. Int J Mol Sci 2016, 17. [Google Scholar] [CrossRef]
- Kasner, M.; Gast, M.; Galuszka, O.; Stroux, A.; Rutschow, S.; Wang, X.; Dohmen, P.; Skurk, C.; Landmesser, U.; Poller, W.; et al. Circulating exosomal microRNAs predict functional recovery after MitraClip repair of severe mitral regurgitation. Int J Cardiol 2016, 215, 402–405. [Google Scholar] [CrossRef]
- Back, M.; Pizarro, R.; Clavel, M.A. Biomarkers in Mitral Regurgitation. Prog Cardiovasc Dis 2017, 60, 334–341. [Google Scholar] [CrossRef]
- Rodriguez, F.H., 3rd; Marelli, A.J. The epidemiology of heart failure in adults with congenital heart disease. Heart Fail Clin 2014, 10, 1–7. [Google Scholar] [CrossRef]
- Baumgartner, H.; De Backer, J.; Babu-Narayan, S.V.; Budts, W.; Chessa, M.; Diller, G.P.; Lung, B.; Kluin, J.; Lang, I.M.; Meijboom, F.; et al. 2020 ESC Guidelines for the management of adult congenital heart disease. Eur Heart J 2021, 42, 563–645. [Google Scholar] [CrossRef]
- Grunert, M.; Appelt, S.; Dunkel, I.; Berger, F.; Sperling, S.R. Altered microRNA and target gene expression related to Tetralogy of Fallot. Sci Rep 2019, 9, 19063. [Google Scholar] [CrossRef] [PubMed]
- Bittel, D.C.; Kibiryeva, N.; Marshall, J.A.; O'Brien, J.E. MicroRNA-421 Dysregulation is Associated with Tetralogy of Fallot. Cells 2014, 3, 713–723. [Google Scholar] [CrossRef] [PubMed]
- Clouthier, K.L.; Taylor, A.C.; Xuhuai, J.; Liu, Y.; Parker, S.; Van Eyk, J.; Reddy, S. A Noninvasive Circulating Signature of Combined Right Ventricular Pressure and Volume Overload in Tetralogy of Fallot/Pulmonary Atresia/Major Aortopulmonary Collateral Arteries. World J Pediatr Congenit Heart Surg 2024, 15, 162–173. [Google Scholar] [CrossRef] [PubMed]
- Tutarel, O.; Dangwal, S.; Bretthauer, J.; Westhoff-Bleck, M.; Roentgen, P.; Anker, S.D.; Bauersachs, J.; Thum, T. Circulating miR-423_5p fails as a biomarker for systemic ventricular function in adults after atrial repair for transposition of the great arteries. Int J Cardiol 2013, 167, 63–66. [Google Scholar] [CrossRef] [PubMed]
- Dimopoulos, K.; Wort, S.J.; Gatzoulis, M.A. Pulmonary hypertension related to congenital heart disease: a call for action. Eur Heart J 2014, 35, 691–700. [Google Scholar] [CrossRef] [PubMed]
- Pagourelias, E.D.; Daraban, A.M.; Mada, R.O.; Duchenne, J.; Mirea, O.; Cools, B.; Heying, R.; Boshoff, D.; Bogaert, J.; Budts, W.; et al. Right ventricular remodelling after transcatheter pulmonary valve implantation. Catheter Cardiovasc Interv 2017, 90, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Cools, B.; Nagaraju, C.K.; Vandendriessche, K.; van Puyvelde, J.; Youness, M.; Roderick, H.L.; Gewillig, M.; Sipido, K.; Claus, P.; Rega, F. Reversal of Right Ventricular Remodeling After Correction of Pulmonary Regurgitation in Tetralogy of Fallot. JACC Basic Transl Sci 2023, 8, 301–315. [Google Scholar] [CrossRef]
- Lunze, F.I.; Hasan, B.S.; Gauvreau, K.; Brown, D.W.; Colan, S.D.; McElhinney, D.B. Progressive intermediate-term improvement in ventricular and atrioventricular interaction after transcatheter pulmonary valve replacement in patients with right ventricular outflow tract obstruction. Am Heart J 2016, 179, 87–98. [Google Scholar] [CrossRef]
- Egbe, A.C.; Salama, A.A.; Miranda, W.R.; Karnakoti, S.; Anderson, J.H.; Jain, C.C.; Burchill, L.J.; Connolly, H.M. Right Heart Reverse Remodeling and Prosthetic Valve Function After Transcatheter vs Surgical Pulmonary Valve Replacement. JACC Cardiovasc Interv 2024, 17, 248–258. [Google Scholar] [CrossRef]
- Hasan, B.S.; Lunze, F.I.; Chen, M.H.; Brown, D.W.; Boudreau, M.J.; Rhodes, J.; McElhinney, D.B. Effects of transcatheter pulmonary valve replacement on the hemodynamic and ventricular response to exercise in patients with obstructed right ventricle-to-pulmonary artery conduits. JACC Cardiovasc Interv 2014, 7, 530–542. [Google Scholar] [CrossRef]
- Talner, C.N. Report of the New England Regional Infant Cardiac Program, by Donald C. Fyler, MD, Pediatrics, 1980;65(suppl):375-461. Pediatrics 1998, 102, 258–259. [Google Scholar] [CrossRef] [PubMed]
- Bashore, T.M. Adult congenital heart disease: right ventricular outflow tract lesions. Circulation 2007, 115, 1933–1947. [Google Scholar] [CrossRef] [PubMed]
- Alkashkari, W.; Albugami, S.; Abbadi, M.; Niyazi, A.; Alsubei, A.; Hijazi, Z.M. Transcatheter pulmonary valve replacement in pediatric patients. Expert Rev Med Devices 2020, 17, 541–554. [Google Scholar] [CrossRef] [PubMed]
- Ylitalo, P.; Nieminen, H.; Pitkanen, O.M.; Jokinen, E.; Sairanen, H. Need of transannular patch in tetralogy of Fallot surgery carries a higher risk of reoperation but has no impact on late survival: results of Fallot repair in Finland. Eur J Cardiothorac Surg 2015, 48, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.Y.; Jang, W.S.; Kim, Y.H.; Park, I.S.; Ko, J.K.; Lee, M.S.; Yun, T.J. Tetralogy of Fallot without the infundibular septum-restricted growth of the pulmonary valve annulus after annulus preservation may render the right ventricular outflow tract obstructive. J Thorac Cardiovasc Surg 2011, 141, 969–974. [Google Scholar] [CrossRef]
- Houeijeh, A.; Batteux, C.; Karsenty, C.; Ramdane, N.; Lecerf, F.; Valdeolmillos, E.; Lourtet-Hascoet, J.; Cohen, S.; Belli, E.; Petit, J.; et al. Long-term outcomes of transcatheter pulmonary valve implantation with melody and SAPIEN valves. Int J Cardiol 2023, 370, 156–166. [Google Scholar] [CrossRef]
- Ruzyllo, W.; Biernacka, E.K.; Wozniak, O.; Kowalski, M.; Spiewak, M.; Cicha-Mikolajczyk, A.; Szczesny, A.; Kusmierczyk, M.; Hoffman, P.; Demkow, M. Transcatheter pulmonary valve implantation in 100 patients: a 10-year single-center experience. Postepy Kardiol Interwencyjnej 2020, 16, 235–243. [Google Scholar] [CrossRef]
- McElhinney, D.B.; Zhang, Y.; Levi, D.S.; Georgiev, S.; Biernacka, E.K.; Goldstein, B.H.; Shahanavaz, S.; Qureshi, A.M.; Cabalka, A.K.; Bauser-Heaton, H.; et al. Reintervention and Survival After Transcatheter Pulmonary Valve Replacement. J Am Coll Cardiol 2022, 79, 18–32. [Google Scholar] [CrossRef]
- Alkashkari, W.; Alsubei, A.; Hijazi, Z.M. Transcatheter Pulmonary Valve Replacement: Current State of Art. Curr Cardiol Rep 2018, 20, 27. [Google Scholar] [CrossRef]
- Egidy Assenza, G.; Krieger, E.V.; Baumgartner, H.; Cupido, B.; Dimopoulos, K.; Louis, C.; Lubert, A.M.; Stout, K.K.; Valente, A.M.; Zeppenfeld, K.; et al. AHA/ACC vs ESC Guidelines for Management of Adults With Congenital Heart Disease: JACC Guideline Comparison. J Am Coll Cardiol 2021, 78, 1904–1918. [Google Scholar] [CrossRef]
- Regueiro, A.; Linke, A.; Latib, A.; Ihlemann, N.; Urena, M.; Walther, T.; Husser, O.; Herrmann, H.C.; Nombela-Franco, L.; Cheema, A.N.; et al. Association Between Transcatheter Aortic Valve Replacement and Subsequent Infective Endocarditis and In-Hospital Death. JAMA 2016, 316, 1083–1092. [Google Scholar] [CrossRef] [PubMed]
- McElhinney, D.B.; Zhang, Y.; Aboulhosn, J.A.; Morray, B.H.; Biernacka, E.K.; Qureshi, A.M.; Torres, A.J.; Shahanavaz, S.; Goldstein, B.H.; Cabalka, A.K.; et al. Multicenter Study of Endocarditis After Transcatheter Pulmonary Valve Replacement. J Am Coll Cardiol 2021, 78, 575–589. [Google Scholar] [CrossRef] [PubMed]
- El Harane, N.; Kervadec, A.; Bellamy, V.; Pidial, L.; Neametalla, H.J.; Perier, M.C.; Lima Correa, B.; Thiebault, L.; Cagnard, N.; Duche, A.; et al. Acellular therapeutic approach for heart failure: in vitro production of extracellular vesicles from human cardiovascular progenitors. Eur Heart J 2018, 39, 1835–1847. [Google Scholar] [CrossRef]
- Nakamura, Y.; Kita, S.; Tanaka, Y.; Fukuda, S.; Obata, Y.; Okita, T.; Nishida, H.; Takahashi, Y.; Kawachi, Y.; Tsugawa-Shimizu, Y.; et al. Adiponectin Stimulates Exosome Release to Enhance Mesenchymal Stem-Cell-Driven Therapy of Heart Failure in Mice. Mol Ther 2020, 28, 2203–2219. [Google Scholar] [CrossRef]
- Gao, L.; Gregorich, Z.R.; Zhu, W.; Mattapally, S.; Oduk, Y.; Lou, X.; Kannappan, R.; Borovjagin, A.V.; Walcott, G.P.; Pollard, A.E.; et al. Large Cardiac Muscle Patches Engineered From Human Induced-Pluripotent Stem Cell-Derived Cardiac Cells Improve Recovery From Myocardial Infarction in Swine. Circulation 2018, 137, 1712–1730. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Wen, J.; Lu, C.; Lin, B.; Xian, S.; Huang, F.; Wu, Y.; Zeng, Z. Inhibition of miR-155-5p attenuates the valvular damage induced by rheumatic heart disease. Int J Mol Med 2020, 45, 429–440. [Google Scholar] [CrossRef] [PubMed]
- Barile, L.; Cervio, E.; Lionetti, V.; Milano, G.; Ciullo, A.; Biemmi, V.; Bolis, S.; Altomare, C.; Matteucci, M.; Di Silvestre, D.; et al. Cardioprotection by cardiac progenitor cell-secreted exosomes: role of pregnancy-associated plasma protein-A. Cardiovasc Res 2018, 114, 992–1005. [Google Scholar] [CrossRef]
- Song, R.; Dasgupta, C.; Mulder, C.; Zhang, L. MicroRNA-210 Controls Mitochondrial Metabolism and Protects Heart Function in Myocardial Infarction. Circulation 2022, 145, 1140–1153. [Google Scholar] [CrossRef]
- Hu, S.; Huang, M.; Li, Z.; Jia, F.; Ghosh, Z.; Lijkwan, M.A.; Fasanaro, P.; Sun, N.; Wang, X.; Martelli, F.; et al. MicroRNA-210 as a novel therapy for treatment of ischemic heart disease. Circulation 2010, 122, S124–131. [Google Scholar] [CrossRef]
- Bittle, G.J.; Morales, D.; Pietris, N.; Parchment, N.; Parsell, D.; Peck, K.; Deatrick, K.B.; Rodriguez-Borlado, L.; Smith, R.R.; Marban, L.; et al. Exosomes isolated from human cardiosphere-derived cells attenuate pressure overload-induced right ventricular dysfunction. J Thorac Cardiovasc Surg 2021, 162, 975–986.e976. [Google Scholar] [CrossRef]
- Gong, Z.T.; Xiong, Y.Y.; Ning, Y.; Tang, R.J.; Xu, J.Y.; Jiang, W.Y.; Li, X.S.; Zhang, L.L.; Chen, C.; Pan, Q.; et al. Nicorandil-Pretreated Mesenchymal Stem Cell-Derived Exosomes Facilitate Cardiac Repair After Myocardial Infarction via Promoting Macrophage M2 Polarization by Targeting miR-125a-5p/TRAF6/IRF5 Signaling Pathway. Int J Nanomedicine 2024, 19, 2005–2024. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Sun, S.; Zhu, D.; Mei, X.; Lyu, Y.; Huang, K.; Li, Y.; Liu, S.; Wang, Z.; Hu, S.; et al. Inhalable Stem Cell Exosomes Promote Heart Repair After Myocardial Infarction. Circulation 2024, 150, 710–723. [Google Scholar] [CrossRef] [PubMed]
- Escude Martinez de Castilla, P.; Tong, L.; Huang, C.; Sofias, A.M.; Pastorin, G.; Chen, X.; Storm, G.; Schiffelers, R.M.; Wang, J.W. Extracellular vesicles as a drug delivery system: A systematic review of preclinical studies. Adv Drug Deliv Rev 2021, 175, 113801. [Google Scholar] [CrossRef] [PubMed]
- van Rooij, E.; Olson, E.N. MicroRNA therapeutics for cardiovascular disease: opportunities and obstacles. Nat Rev Drug Discov 2012, 11, 860–872. [Google Scholar] [CrossRef]
| miRNA | Expression level | Clinical significance | References |
|---|---|---|---|
| miRNA-141 | Down-regulation | Aortic valve calcification | [49] |
| miRNA-30b | Down-regulation | Aortic valve calcification | [50] |
| miRNA-125b | Up-regulation | Aortic valve calcification | [51] |
| miRNA-122-5p | Up-regulation | Lack of improvement in LV function after TAVR | [52] |
| miRNA-206 | Up-regulation | Decrease in LVEF after TAVR | [53] |
| miRNA-148b-3p, miRNA-409-3p | Down-regulation | HF in MR patients | [55] |
| miRNA-21-5p | Up-regulation | LV reverse remodeling | [56] |
| miRNA-186-5p, miRNA-30e-5p, miRNA-152-3p | Down-regulation | Diagnostic biomarkers for FTR | [57] |
| miRNA-421, miRNA-1233-3p, miRNA-625-5p | Down-regulation | Diagnostic biomarkers for TOF patients with symptomatic right HF | [58] |
| miRNA-18a, miRNA-486-5p | Up-regulation | Decrease in systemic ventricular contractility for patients with complete TGA after surgery | [59] |
| miRNA-99b | Up-regulation | Biomarkers for patients with repaired TOF and volume-overloaded RV | [60] |
| miRNA-766 | Down-regulation | Biomarkers for patients with repaired TOF and volume-overloaded RV | [60] |
| miRNA-940 | Down-regulation | Leading to the occurrence of TOF | [43] |
| miRNA-28-3p, miRNA-433-3p, miRNA-371b-3p | Up-regulation | Progressive RV enlargement and systolic dysfunction | [46] |
| miRNA-183-3p | Up-regulation | Biomarkers of worsening HF in adult patients with TGA and a systemic RV. | [47] |
| miRNA-223 | Down-regulation | Right HF | [61] |
| miRNA-335-5p | Up-regulation | RV remodeling | [62] |
| miRNA-378 | Up-regulation | Suppressing myocardial fibrosis | [63] |
| miRNA-let-7b-5p | Up-regulation | Promoting angiogenesis | [64] |
| miRNA-210 | Down-regulation | Inhibiting apoptosis in cardiomyocytic cells | [65] |
| miRNA-132 | Down-regulation | Enhancing tube formation in endothelial cells | [65] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).





