Submitted:
10 November 2024
Posted:
11 November 2024
You are already at the latest version
Abstract
Keywords:
Background
Metabolic Dysfunction-Associated Steatotic Liver Disease
Heart Failure
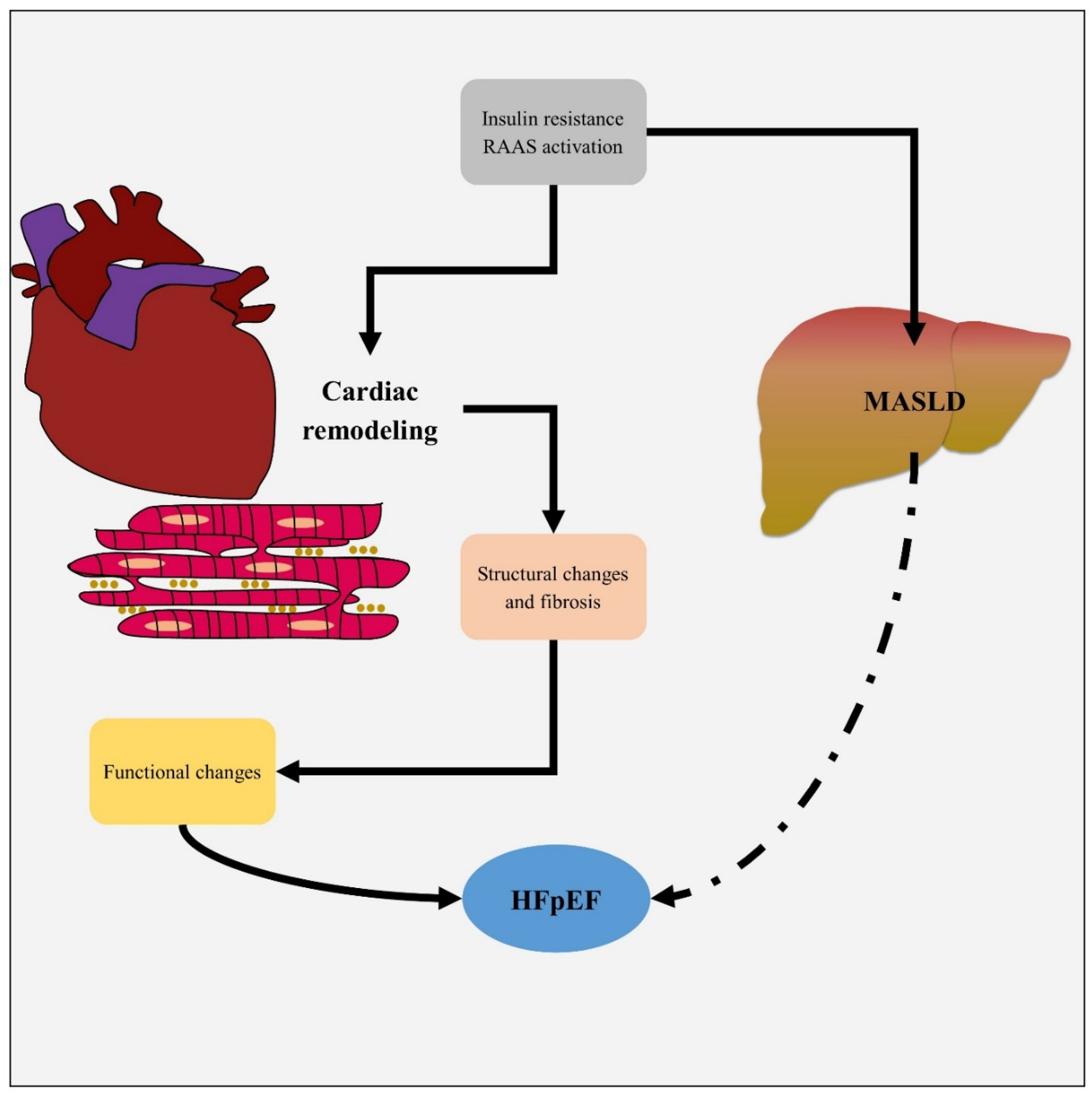
Metabolic Dysfunction-Associated Steatotic Liver Disease and Heart Failure Pathophysiology
Metabolic Dysfunction-Associated Steatotic Liver Disease and Heart Failure Association
Conclusion
Authors’ Contributions
Funding
Acknowledgments
Conflict of Interest
References
- Lekakis V, Papatheodoridis GV. Natural history of metabolic dysfunction-associated steatotic liver disease. European Journal of Internal Medicine 2024, 122, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Corrao S, Calvo L, Granà W, Scibetta S, Mirarchi L, Amodeo S, et al. Metabolic dysfunction-associated steatotic liver disease: A pathophysiology and clinical framework to face the present and the future. Nutrition, Metabolism and Cardiovascular Diseases 2024.
- El-Kassas M, Othman HA, Elbadry M, Alswat K, Yilmaz Y. Risk Stratification of Patients with Metabolic Dysfunction-Associated Steatotic Liver Disease: Steatohepatitis, Fibrosis, and Hepatocellular Carcinoma. Journal of Clinical and Experimental Hepatology 2024, 102415.
- Chan W-K, Chuah K-H, Rajaram RB, Lim L-L, Ratnasingam J, Vethakkan SR. Metabolic dysfunction-associated steatotic liver disease (MASLD): a state-of-the-art review. Journal of Obesity & Metabolic Syndrome 2023, 32, 197. [Google Scholar]
- Khan MS, Shahid I, Bennis A, Rakisheva A, Metra M, Butler J. Global epidemiology of heart failure. Global epidemiology of heart failure. Nature Reviews Cardiology. 2024, 1–18. [Google Scholar]
- Doehner W, Frenneaux M, Anker SD. Metabolic impairment in heart failure: the myocardial and systemic perspective. Journal of the American College of Cardiology 2014, 64, 1388–1400. [Google Scholar] [CrossRef]
- Liu Z, Li Z, Li X, Yan Y, Liu J, Wang J, et al. Global trends in heart failure from 1990 to 2019, An age-period-cohort analysis from the Global Burden of Disease study. ESC Heart Failure. 2024.
- Younossi ZM, Kalligeros M, Henry L. Epidemiology of Metabolic Dysfunction Associated Steatotic Liver Disease. Clinical and Molecular Hepatology 2024. [Google Scholar]
- Younossi ZM, Golabi P, Paik J, Owrangi S, Yilmaz Y, El-Kassas M, et al. Prevalence of metabolic dysfunction-associated steatotic liver disease in the Middle East and North Africa. Liver International 2024, 44, 1061–1070. [Google Scholar] [CrossRef]
- Miao L, Targher G, Byrne CD, Cao Y-Y, Zheng M-H. Current status and future trends of the global burden of MASLD. Trends in Endocrinology & Metabolism 2024.
- Manikat R, Ahmed A, Kim D. Current epidemiology of chronic liver disease. Gastroenterology Report 2024, 12, goae069. [Google Scholar]
- He Q-J, Li Y-F, Zhao L-T, Lin C-T, Yu C-Y, Wang D. Recent advances in age-related metabolic dysfunction-associated steatotic liver disease. World Journal of Gastroenterology 2024, 30, 652. [Google Scholar] [CrossRef] [PubMed]
- Kim Y, Charlton M, Rezvani G, Saal GB. S1826 Prevalence of Metabolic-Associated Steatohepatitis (MASH) and Advanced Fibrosis in At-Risk Pediatric Populations. Official journal of the American College of Gastroenterology| ACG 2024, 119, S1316–S7.
- Shen T-H, Wu C-H, Lee Y-W, Chang C-C. Prevalence, trends, and characteristics of metabolic dysfunction-associated steatotic liver disease among the US population aged 12–79 years. European Journal of Gastroenterology & Hepatology 2024, 36, 636–645. [Google Scholar]
- Gancheva S, Roden M, Castera L. Diabetes as a risk factor for MASH progression. Diabetes Research and Clinical Practice 2024, 111846. [Google Scholar]
- Driessen S, Francque SM, Anker SD, Cabezas MC, Grobbee DE, Tushuizen ME, et al. Metabolic dysfunction associated steatotic liver disease and the heart. Hepatology 2023, 10, 1097. [Google Scholar]
- Targher G, Byrne CD, Tilg H. MASLD: a systemic metabolic disorder with cardiovascular and malignant complications. Gut 2024, 73, 691–702. [Google Scholar]
- Gulati R, Moylan CA, Wilder J, Wegermann K. Racial and ethnic disparities in metabolic dysfunction-associated steatotic liver disease. Metabolism and Target Organ Damage 2024, 4, N/A–N/A. [Google Scholar]
- Kardashian A, Serper M, Terrault N, Nephew LD. Health disparities in chronic liver disease. Hepatology 2023, 77, 1382–1403. [Google Scholar] [CrossRef]
- Díaz LA, Lazarus JV, Fuentes-López E, Idalsoaga F, Ayares G, Desaleng H, et al. Disparities in steatosis prevalence in the United States by Race or Ethnicity according to the 2023 criteria. Communications Medicine 2024, 4, 219. [Google Scholar] [CrossRef]
- Saini A, Rutledge B, Damughatla AR, Rasheed M, Naylor P, Mutchnick M. Manifestation and Progression of Metabolic Dysfunction-Associated Steatotic Liver Disease in a Predominately African American Population at a Multi-Specialty Healthcare Organization. Healthcare 2024, 1478.
- Sotoudeheian, M. Galectin-3 and Severity of Liver Fibrosis in Metabolic Dysfunction-Associated Fatty Liver Disease. Protein and Peptide Letters 2024, 31, 290–304. [Google Scholar] [CrossRef] [PubMed]
- Mokhtare M, Abdi A, Sadeghian AM, Sotoudeheian M, Namazi A, Sikaroudi MK. Investigation about the correlation between the severity of metabolic-associated fatty liver disease and adherence to the Mediterranean diet. Clinical Nutrition ESPEN 2023, 58, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Mohammadjavad, S. Agile 3+ and Metabolic Dysfunction-Associated Fatty Liver Disease: Detecting Advanced Fibrosis based on Reported Liver Stiffness Measurement in FibroScan and Laboratory Findings. The International Journal of Gastroenterology and Hepatology Diseases 2024, 3, 43–54. [Google Scholar]
- Lee EC, Anand VV, Razavi AC, Alebna PL, Muthiah MD, Siddiqui MS, et al. The global epidemic of metabolic fatty liver disease. Current Cardiology Reports 2024, 26, 199–210. [Google Scholar] [CrossRef] [PubMed]
- El-Kassas M, Awad A, Elbadry M, Arab JP. Tailored Model of Care for Patients with Metabolic Dysfunction-Associated Steatotic Liver Disease. Seminars in Liver Disease: Thieme Medical Publishers, Inc.; 2024.
- Sotoudeheian M, Hoseini S. Understanding the Pathophysiology of Heart Failure with Mid-Range Ejection Fraction: A Comprehensive Narrative Review. 2023.
- Sotoudeheian MJ, Mirahmadi S-M-S, Pirhayati M, Azarbad R, Nematollahi S, Taghizadeh M, et al. Understanding the Role of Galectin-1 in Heart Failure: A Comprehensive Narrative Review. Current Cardiology Reviews 2024, 20, 82–90. [Google Scholar]
- Murphy SP, Ibrahim NE, Januzzi JL. Heart failure with reduced ejection fraction: a review. Jama 2020, 324, 488–504. [Google Scholar] [CrossRef]
- Severino P, Maestrini V, Mariani MV, Birtolo LI, Scarpati R, Mancone M, et al. Structural and myocardial dysfunction in heart failure beyond ejection fraction. Heart failure reviews 2020, 25, 9–17. [Google Scholar] [CrossRef]
- Savarese G, Becher PM, Lund LH, Seferovic P, Rosano GM, Coats AJ. Global burden of heart failure: a comprehensive and updated review of epidemiology. Cardiovascular research 2022, 118, 3272–3287. [Google Scholar]
- Triposkiadis F, Xanthopoulos A, Parissis J, Butler J, Farmakis D. Pathogenesis of chronic heart failure: cardiovascular aging, risk factors, comorbidities, and disease modifiers. Heart failure reviews 2022, 27, 337–344. [Google Scholar] [CrossRef]
- Heidenreich PA, Fonarow GC, Opsha Y, Sandhu AT, Sweitzer NK, Warraich HJ, et al. Economic issues in heart failure in the United States. Journal of cardiac failure 2022, 28, 453–466. [Google Scholar] [CrossRef]
- Ziaeian B, Fonarow GC. Epidemiology and aetiology of heart failure. Nature Reviews Cardiology 2016, 13, 368–378. [Google Scholar] [CrossRef] [PubMed]
- Groenewegen A, Rutten FH, Mosterd A, Hoes AW. Epidemiology of heart failure. European journal of heart failure 2020, 22, 1342–1356. [Google Scholar] [CrossRef] [PubMed]
- Dharmarajan K, Rich MW. Epidemiology, pathophysiology, and prognosis of heart failure in older adults. Heart failure clinics 2017, 13, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Dunlay SM, Roger VL, Redfield MM. Epidemiology of heart failure with preserved ejection fraction. Nature Reviews Cardiology 2017, 14, 591–602. [Google Scholar] [CrossRef] [PubMed]
- Shi K, Yang M-X, Huang S, Yan W-F, Qian W-L, Li Y, et al. Effect of diabetes mellitus on the development of left ventricular contractile dysfunction in women with heart failure and preserved ejection fraction. Cardiovascular diabetology 2021, 20, 1–10. [Google Scholar]
- Tadic M, Cuspidi C, Plein S, Belyavskiy E, Heinzel F, Galderisi M. Sex and heart failure with preserved ejection fraction: from pathophysiology to clinical studies. Journal of clinical medicine 2019, 8, 792. [Google Scholar] [CrossRef]
- Roger, VL. Epidemiology of heart failure: a contemporary perspective. Circulation research 2021, 128, 1421–1434. [Google Scholar] [CrossRef]
- Kilgore M, Patel HK, Kielhorn A, Maya JF, Sharma P. Economic burden of hospitalizations of Medicare beneficiaries with heart failure. Risk management and healthcare policy 2017, 63–70. [Google Scholar]
- Arora S, Patel P, Lahewala S, Patel N, Patel NJ, Thakore K, et al. Etiologies, trends, and predictors of 30-day readmission in patients with heart failure. The American journal of cardiology 2017, 119, 760–769. [Google Scholar] [CrossRef]
- Grosman-Rimon L, Billia F, Wright E, Carasso S, Elbaz-Greener G, Kachel E, et al. Neurohormones, inflammatory mediators, and cardiovascular injury in the setting of heart failure. Heart Failure Reviews 2020, 25, 685–701. [Google Scholar] [CrossRef]
- Chaudhry R, Mahmood A, Ullah I, Urra EP, Masmoum MD, Atif A, et al. Heart Failure: Causes, Symptoms, and Management Strategies. Journal of Advances in Medicine and Medical Research 2024, 36, 335–345. [Google Scholar] [CrossRef]
- Ge Z, Li A, McNamara J, Dos Remedios C, Lal S. Pathogenesis and pathophysiology of heart failure with reduced ejection fraction: translation to human studies. Heart failure reviews 2019, 24, 743–758. [Google Scholar] [CrossRef] [PubMed]
- Borlaug, BA. The pathophysiology of heart failure with preserved ejection fraction. Nature Reviews Cardiology 2014, 11, 507–515. [Google Scholar] [CrossRef] [PubMed]
- Gevaert AB, Boen JR, Segers VF, Van Craenenbroeck EM. Heart failure with preserved ejection fraction: a review of cardiac and noncardiac pathophysiology. Frontiers in physiology 2019, 10, 638. [Google Scholar] [CrossRef] [PubMed]
- Fayyaz AU, Eltony M, Prokop LJ, Koepp KE, Borlaug BA, Dasari S, et al. Pathophysiological insights into HFpEF from studies of human cardiac tissue. Nature Reviews Cardiology 2024, 1–15. [Google Scholar]
- Schiattarella GG, Rodolico D, Hill JA. Metabolic inflammation in heart failure with preserved ejection fraction. Cardiovascular research 2021, 117, 423–434. [Google Scholar] [CrossRef]
- Sandireddy R, Sakthivel S, Gupta P, Behari J, Tripathi M, Singh BK. Systemic impacts of metabolic dysfunction-associated steatotic liver disease (MASLD) and metabolic dysfunction-associated steatohepatitis (MASH) on heart, muscle, and kidney related diseases. Frontiers in Cell and Developmental Biology 2024, 12, 1433857. [Google Scholar]
- Leite AR, Dias J, Godinho T, Correia-Chaves J, Borges-Canha M, Lourenço IM, et al. Metabolic Dysfunction-associated Steatotic Liver Disease and Heart Failure with Preserved Ejection Fraction: a bidirectional relationship with clinical and therapeutic implications. Endocrinology Insights 2024, 1–17.
- Lopez-Candales A, Burgos PMH, Hernandez-Suarez DF, Harris D. Linking chronic inflammation with cardiovascular disease: from normal aging to the metabolic syndrome. Journal of nature and science 2017, 3.
- Gehrke N, Schattenberg JM. Metabolic inflammation—a role for hepatic inflammatory pathways as drivers of comorbidities in nonalcoholic fatty liver disease? Gastroenterology 2020, 158, 1929–1947.e6.
- Ferenc K, Jarmakiewicz-Czaja S, Sokal-Dembowska A, Stasik K, Filip R. Common Denominator of MASLD and Some Non-Communicable Diseases. Current Issues in Molecular Biology 2024, 46, 6690–6709. [Google Scholar] [CrossRef] [PubMed]
- Jamalinia M, Lonardo A. determinants and assessment of cardiovascular risk in steatotic liver disease owing to metabolic dysfunction-addressing the challenge. Metabolism and Target Organ Damage 2024, 4, N/A-N/A.
- Yanai H, Adachi H, Hakoshima M, Iida S, Katsuyama H. Metabolic-Dysfunction-Associated Steatotic Liver Disease—Its Pathophysiology, Association with Atherosclerosis and Cardiovascular Disease, and Treatments. International journal of molecular sciences 2023, 24, 15473. [Google Scholar] [CrossRef] [PubMed]
- Shin S, Kim J, Lee JY, Kim J, Oh C-M. Mitochondrial quality control: its role in metabolic dysfunction-associated steatotic liver disease (MASLD). Journal of Obesity & Metabolic Syndrome 2023, 32, 289.
- Svobodová G, Horní M, Velecká E, Boušová I. Metabolic dysfunction-associated steatotic liver disease-induced changes in the antioxidant system: a review. Archives of Toxicology 2024, 1–22.
- Zhao Y, Zhou Y, Wang D, Huang Z, Xiao X, Zheng Q, et al. Mitochondrial Dysfunction in Metabolic Dysfunction Fatty Liver Disease (MAFLD). International Journal of Molecular Sciences 2023, 24, 17514. [Google Scholar] [CrossRef]
- Ma Y, Wang J, Xiao W, Fan X. A review of MASLD-related hepatocellular carcinoma: progress in pathogenesis, early detection, and therapeutic interventions. Frontiers in Medicine 2024, 11, 1410668. [Google Scholar] [CrossRef]
- He Y, Huang W, Zhang C, Chen L, Xu R, Li N, et al. Energy metabolism disorders and potential therapeutic drugs in heart failure. Acta Pharmaceutica Sinica B 2021, 11, 1098–1116. [Google Scholar] [CrossRef]
- Ramaccini D, Montoya-Uribe V, Aan FJ, Modesti L, Potes Y, Wieckowski MR, et al. Mitochondrial function and dysfunction in dilated cardiomyopathy. Frontiers in cell and developmental biology 2021, 8, 624216. [Google Scholar]
- Bansal SK, Bansal MB. Pathogenesis of MASLD and MASH–role of insulin resistance and lipotoxicity. Alimentary Pharmacology & Therapeutics 2024, 59, S10–S22. [Google Scholar]
- Meagher P, Adam M, Civitarese R, Bugyei-Twum A, Connelly KA. Heart failure with preserved ejection fraction in diabetes: mechanisms and management. Canadian Journal of Cardiology 2018, 34, 632–643. [Google Scholar] [CrossRef] [PubMed]
- Caturano A, Vetrano E, Galiero R, Sardu C, Rinaldi L, Russo V, et al. Advances in the Insulin–Heart Axis: Current Therapies and Future Directions. International Journal of Molecular Sciences 2024, 25, 10173. [Google Scholar] [CrossRef] [PubMed]
- Varghese TP, Tazneem B. Unraveling the complex pathophysiology of heart failure: insights into the role of renin-angiotensin-aldosterone system (RAAS) and sympathetic nervous system (SNS). Current Problems in Cardiology 2024, 49, 102411. [Google Scholar] [CrossRef] [PubMed]
- Manolis AA, Manolis TA, Manolis AS. Neurohumoral activation in heart failure. International Journal of Molecular Sciences 2023, 24, 15472. [Google Scholar] [CrossRef] [PubMed]
- McGrath MS, Wentworth BJ. The Renin–Angiotensin System in Liver Disease. International Journal of Molecular Sciences 2024, 25, 5807. [Google Scholar] [CrossRef]
- Akkız H, Gieseler RK, Canbay A. Liver fibrosis: From basic science towards clinical progress, focusing on the central role of hepatic stellate cells. International Journal of Molecular Sciences 2024, 25, 7873. [Google Scholar] [CrossRef]
- Hara A, Tallquist MD. Fibroblast and immune cell cross-talk in cardiac fibrosis. Current Cardiology Reports 2023, 25, 485–493. [Google Scholar] [CrossRef]
- Amrute JM, Luo X, Penna V, Bredemeyer A, Yamawaki T, Yang S, et al. Targeting immune-fibroblast crosstalk in myocardial infarction and cardiac fibrosis. Research square. 2023.
- Boeckmans J, Sandrin L, Knackstedt C, Schattenberg JM. Liver stiffness as a cornerstone in heart disease risk assessment. Liver International 2024, 44, 344–356. [Google Scholar] [CrossRef]
- Macerola N, Riccardi L, Di Stasio E, Montalto M, Gasbarrini A, Pompili M, et al. Prognostic value of liver stiffness in patients hospitalized for acute decompensated heart failure: a meta-analysis. Journal of Ultrasound. 2024, 1–7.
- Mantovani A, Petracca G, Csermely A, Beatrice G, Bonapace S, Rossi A, et al. Non-alcoholic fatty liver disease and risk of new-onset heart failure: an updated meta-analysis of about 11 million individuals. Gut 2023, 72, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Inciardi RM, Mantovani A, Targher G. Non-alcoholic fatty liver disease as an emerging risk factor for heart failure. Current Heart Failure Reports 2023, 20, 308–319. [Google Scholar] [CrossRef] [PubMed]
- Petrescu M, Vlaicu SI, Ciumărnean L, Milaciu MV, Mărginean C, Florea M, et al. Chronic inflammation—A link between nonalcoholic fatty liver disease (NAFLD) and dysfunctional adipose tissue. Medicina 2022, 58, 641. [Google Scholar] [CrossRef] [PubMed]
- Chen L-z, Jing X-b, Wu C-f, Zeng Y-c, Xie Y-c, Wang M-q, et al. Nonalcoholic fatty liver disease-associated liver fibrosis is linked with the severity of coronary artery disease mediated by systemic inflammation. Disease Markers 2021, 2021, 6591784. [Google Scholar]
- Xie W, Gan J, Zhou X, Tian H, Pan X, Liu W, et al. Myocardial infarction accelerates the progression of MASH by triggering immunoinflammatory response and induction of periosti. Cell metabolism 2024, 36, 1269–1286. [Google Scholar] [CrossRef]
- Nasiri-Ansari N, Androutsakos T, Flessa C-M, Kyrou I, Siasos G, Randeva HS, et al. Endothelial cell dysfunction and nonalcoholic fatty liver disease (NAFLD): A concise review. Cells 2022, 11, 2511. [Google Scholar] [CrossRef]
- Garbuzenko, DV. Pathophysiological mechanisms of cardiovascular disorders in non-alcoholic fatty liver disease. Gastroenterology and Hepatology from Bed to Bench 2022, 15, 194. [Google Scholar]
- Sanyal AJ, Husain M, Diab C, Mangla KK, Shoeb A, Lingvay I, et al. Cardiovascular disease in patients with metabolic dysfunction-associated steatohepatitis compared with metabolic dysfunction-associated steatotic liver disease and other liver diseases: A systematic review. American Heart Journal Plus: Cardiology Research and Practice. 2024, 100386.
- Patel SS, Nabi E, Guzman L, Abbate A, Bhati C, Stravitz RT, et al. Coronary artery disease in decompensated patients undergoing liver transplantation evaluation. Liver transplantation 2018, 24, 333–342. [Google Scholar] [CrossRef]
- Simon TG, Roelstraete B, Hagström H, Sundström J, Ludvigsson JF. Non-alcoholic fatty liver disease and incident major adverse cardiovascular events: results from a nationwide histology cohort. Gut 2022, 71, 1867–1875. [Google Scholar] [CrossRef]
- VanWagner LB, Lapin B, Skaro AI, Lloyd-Jones DM, Rinella ME. Impact of renal impairment on cardiovascular disease mortality after liver transplantation for nonalcoholic steatohepatitis cirrhosis. Liver International 2015, 35, 2575–2583. [Google Scholar] [CrossRef] [PubMed]
- Wei Z, Huang Z, Song Z, Zhao W, Zhao D, Tan Y, et al. Metabolic Dysfunction-associated fatty liver disease and incident heart failure risk: the Kailuan cohort study. Diabetology & Metabolic Syndrome 2023, 15, 137. [Google Scholar]
- Nagai S, Collins K, Chau LC, Safwan M, Rizzari M, Yoshida A, et al. Increased risk of death in first year after liver transplantation among patients with nonalcoholic steatohepatitis vs liver disease of other etiologies. Clinical gastroenterology and hepatology 2019, 17, 2759–2768.e5. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



