Submitted:
04 November 2024
Posted:
05 November 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
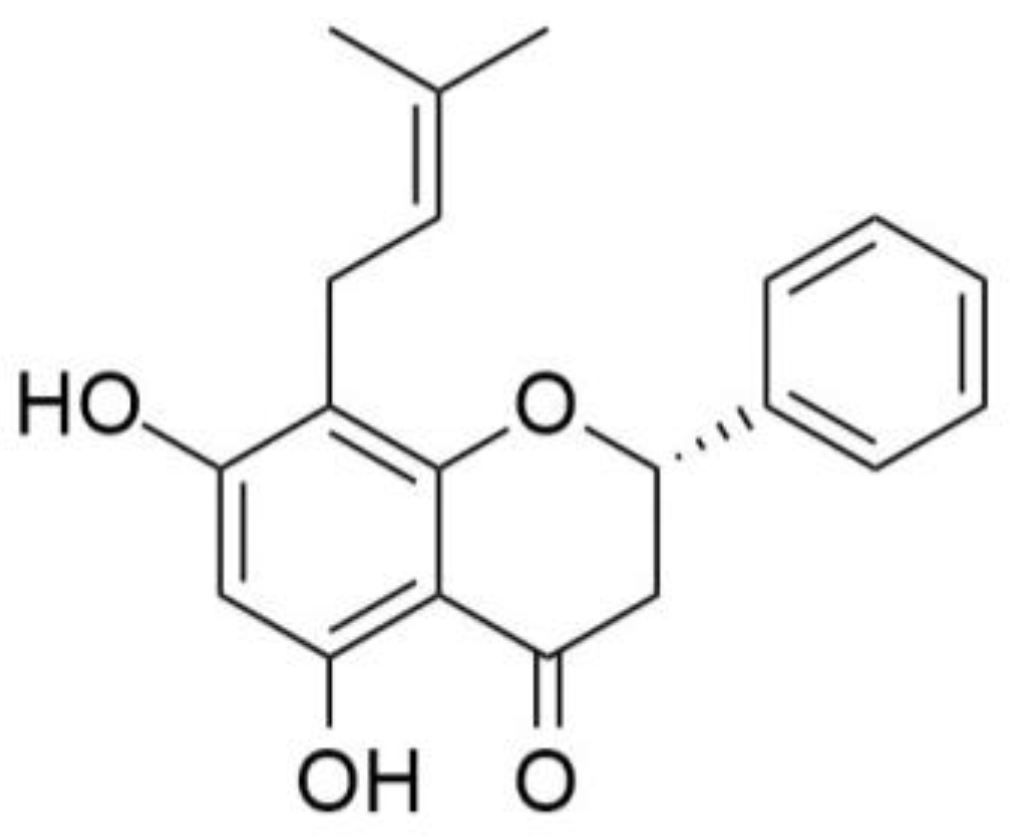
2.1 Chemistry
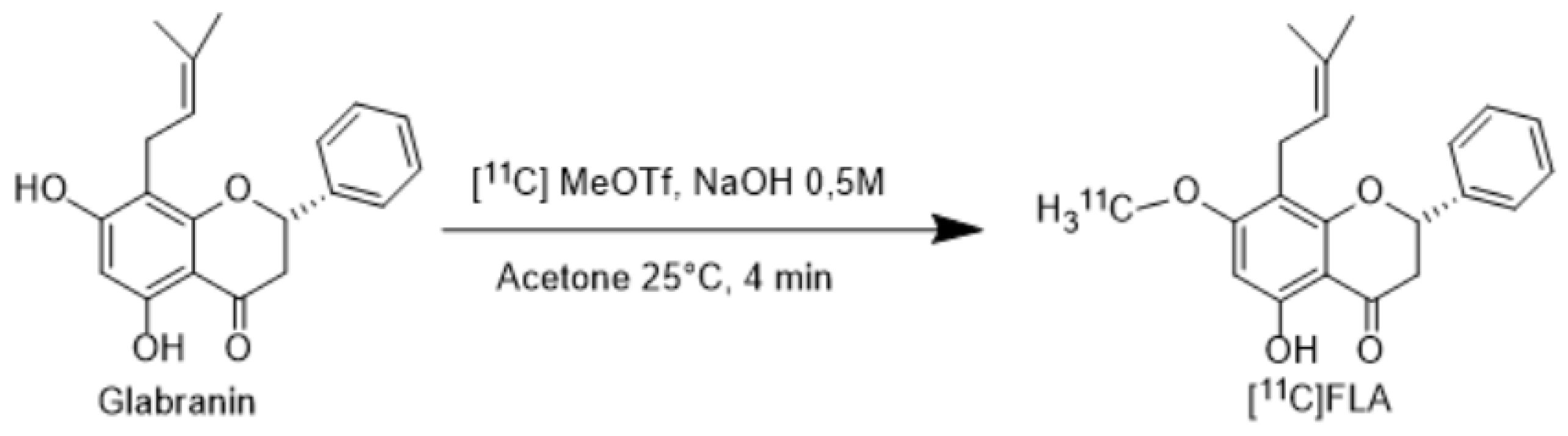
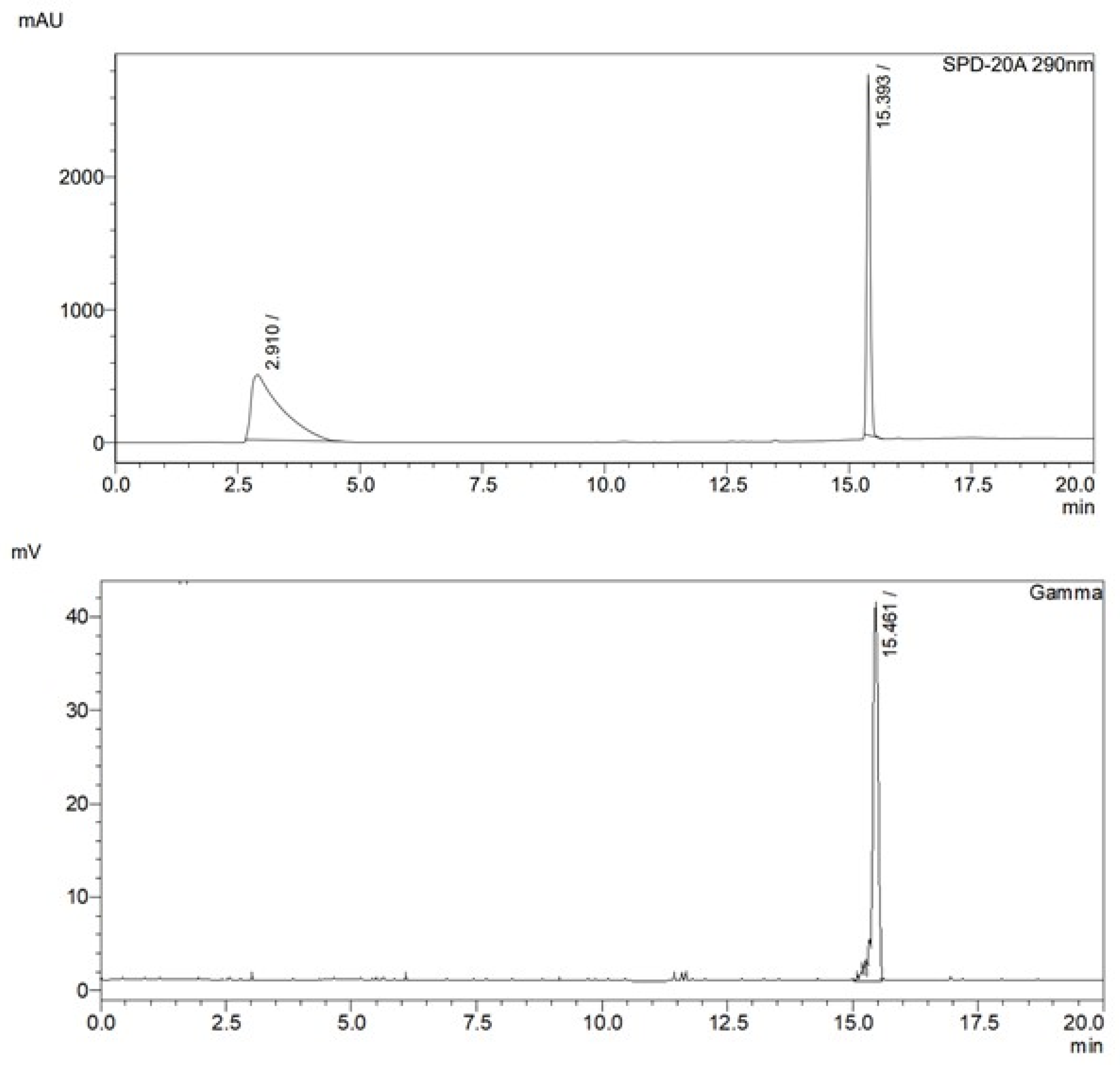
2.2. Radiochemistry
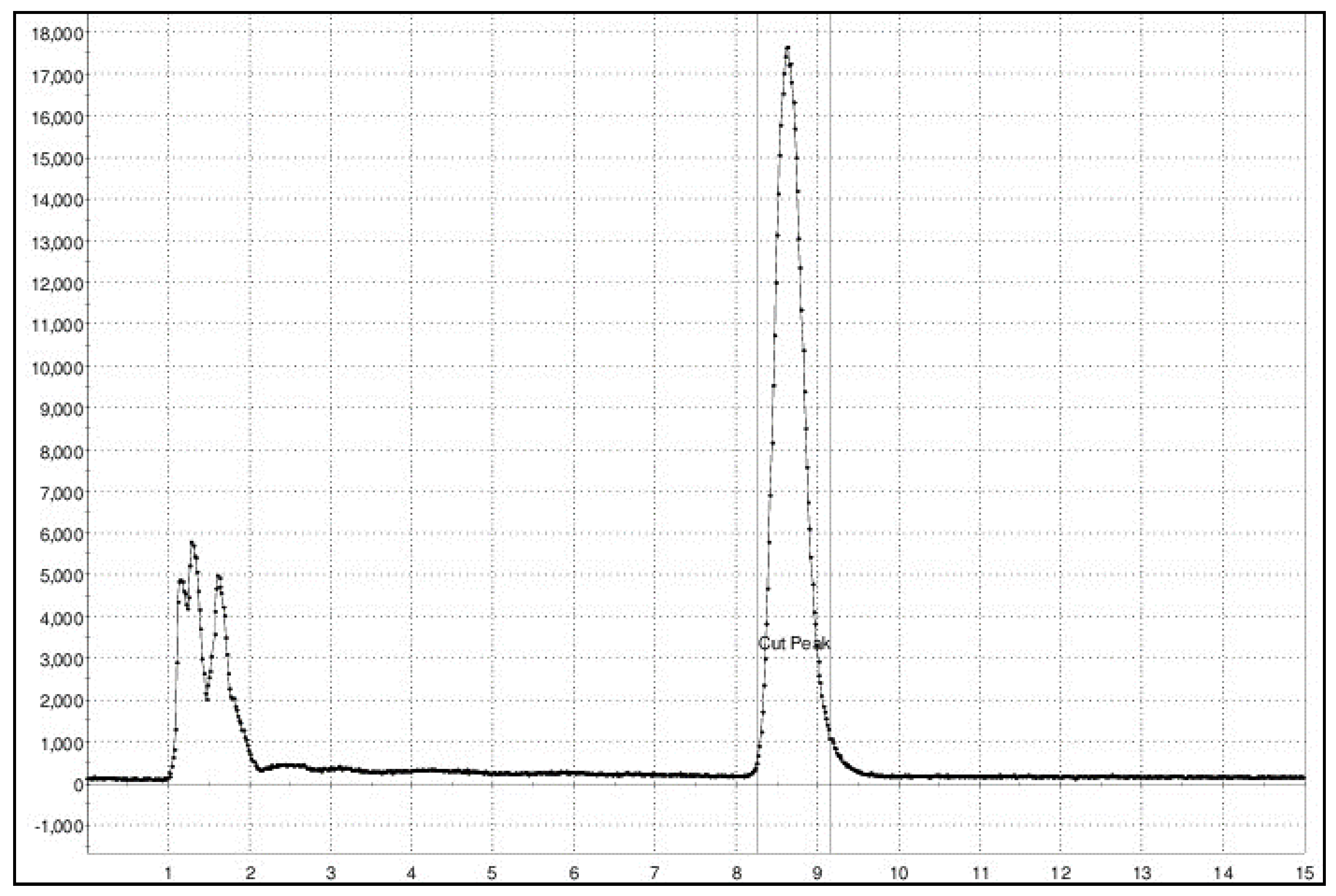
2.3. Physicochemical Evaluation
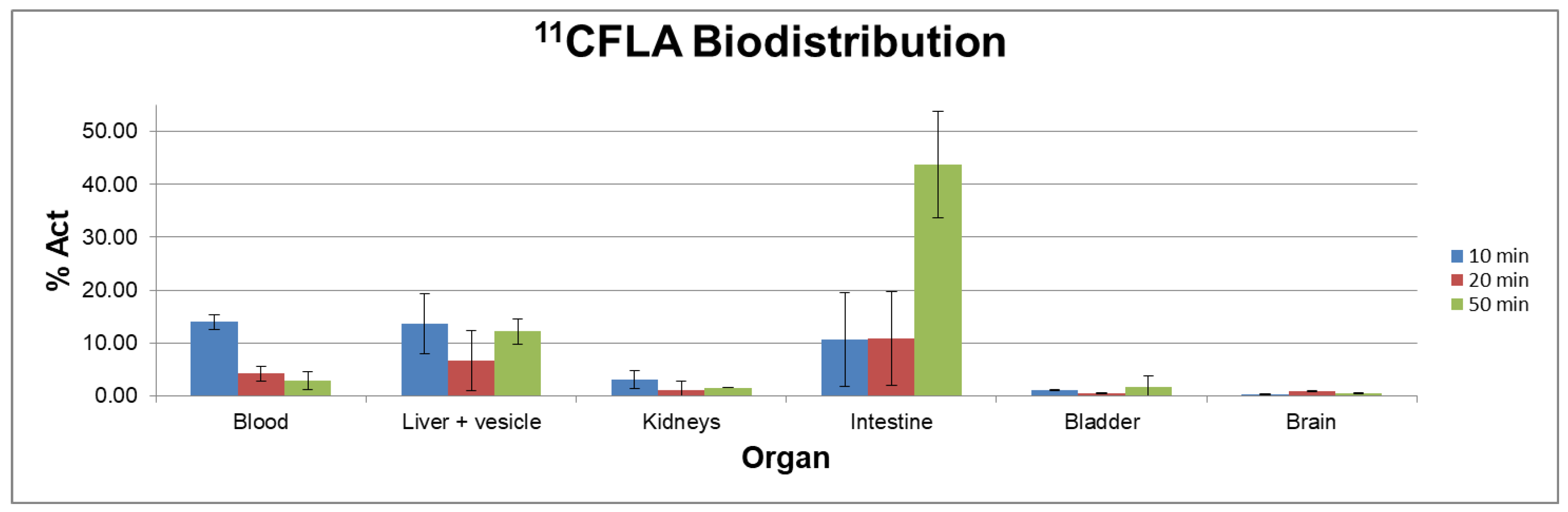
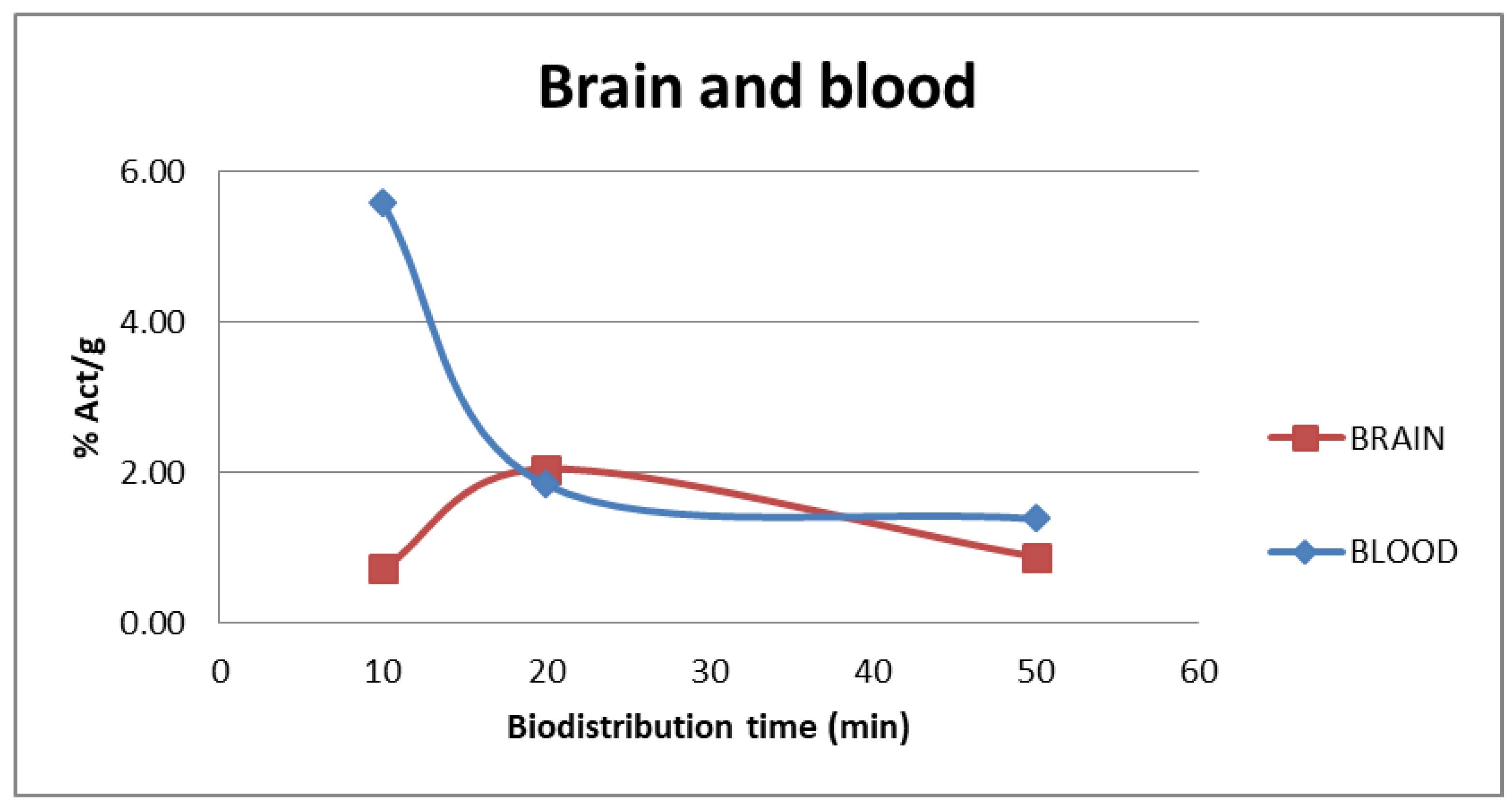
2.4. Ex Vivo Biodistribution Studies
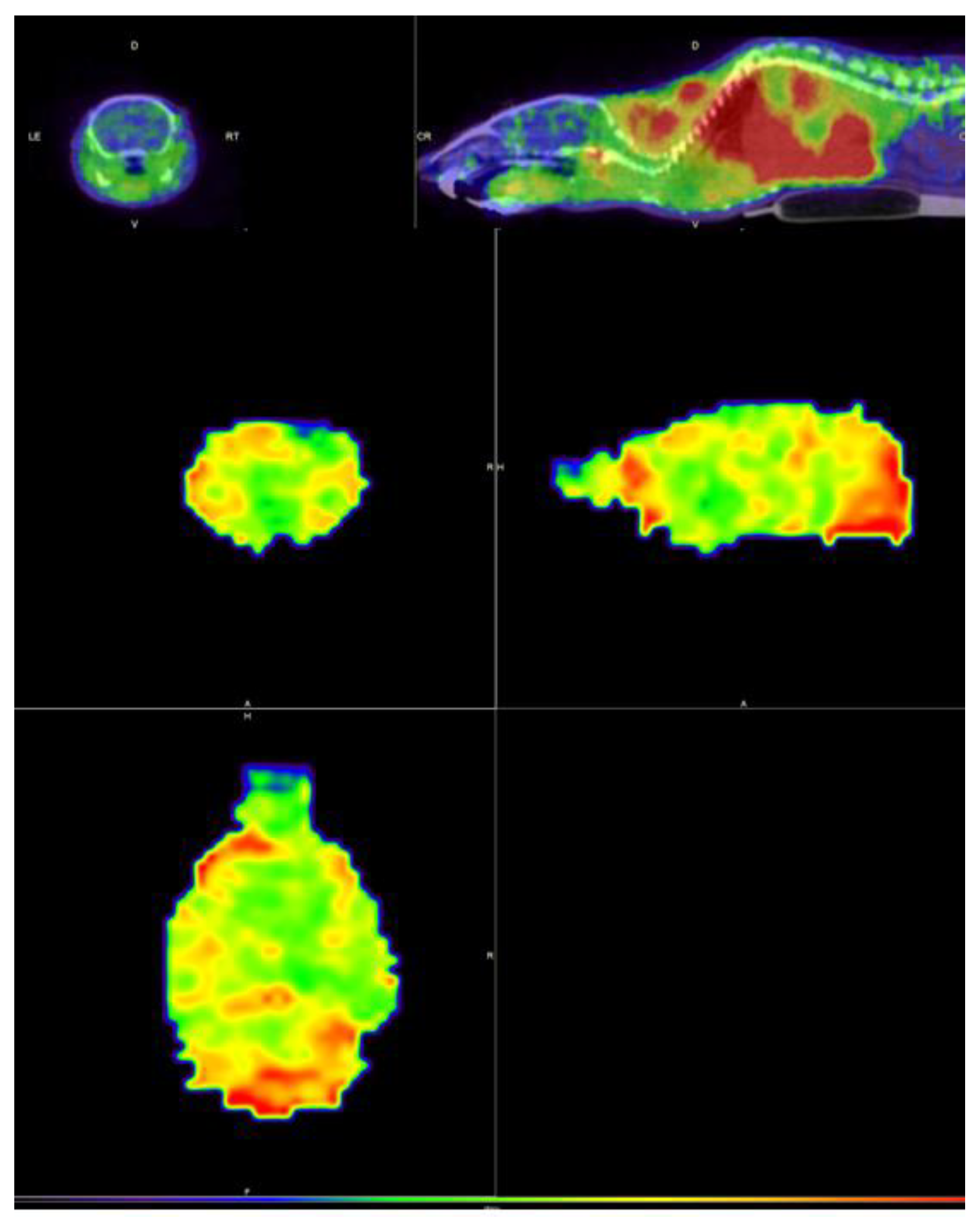
2.5. In Vivo Imagining Studies:
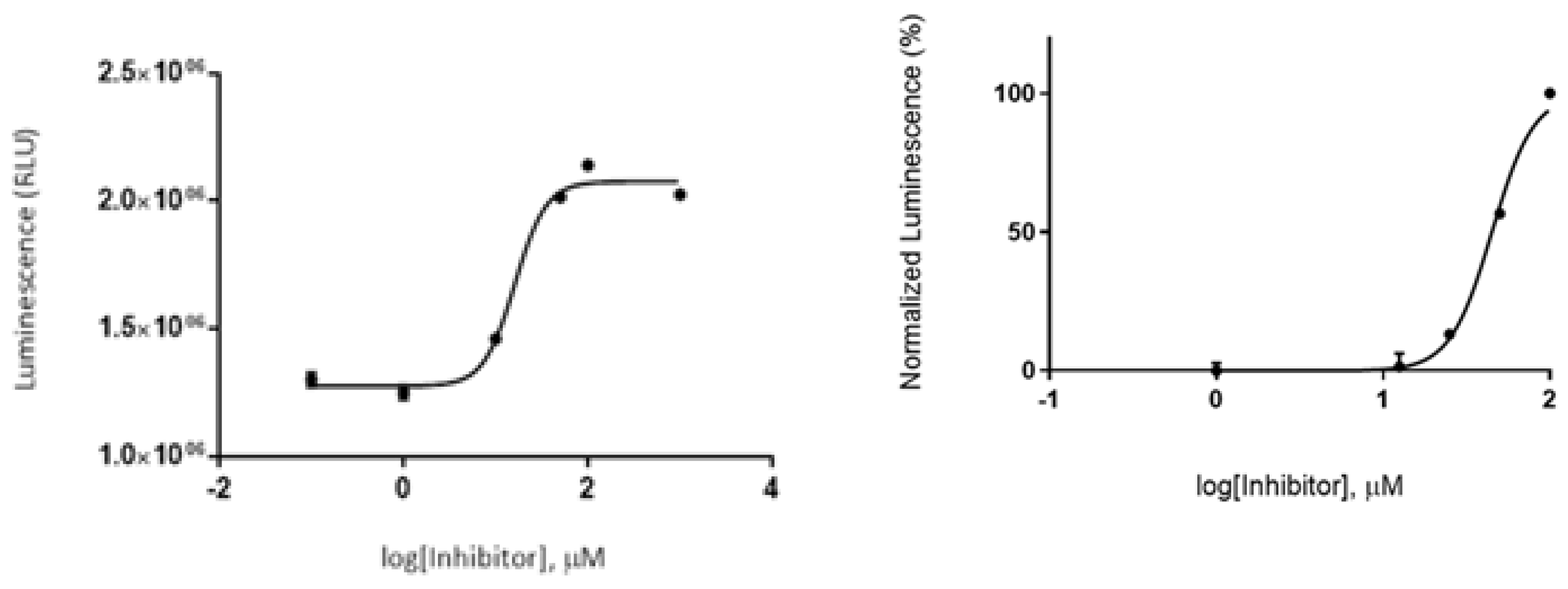
2.6. In Vitro Evaluation of GSK-3β Inhibition
2.7. In Vitro Evaluation of [11C]FLA Uptake by Neurons
2.8. In Vitro Evaluation of [11C]FLA Uptake by Astrocytes
2.9. Competition Assay
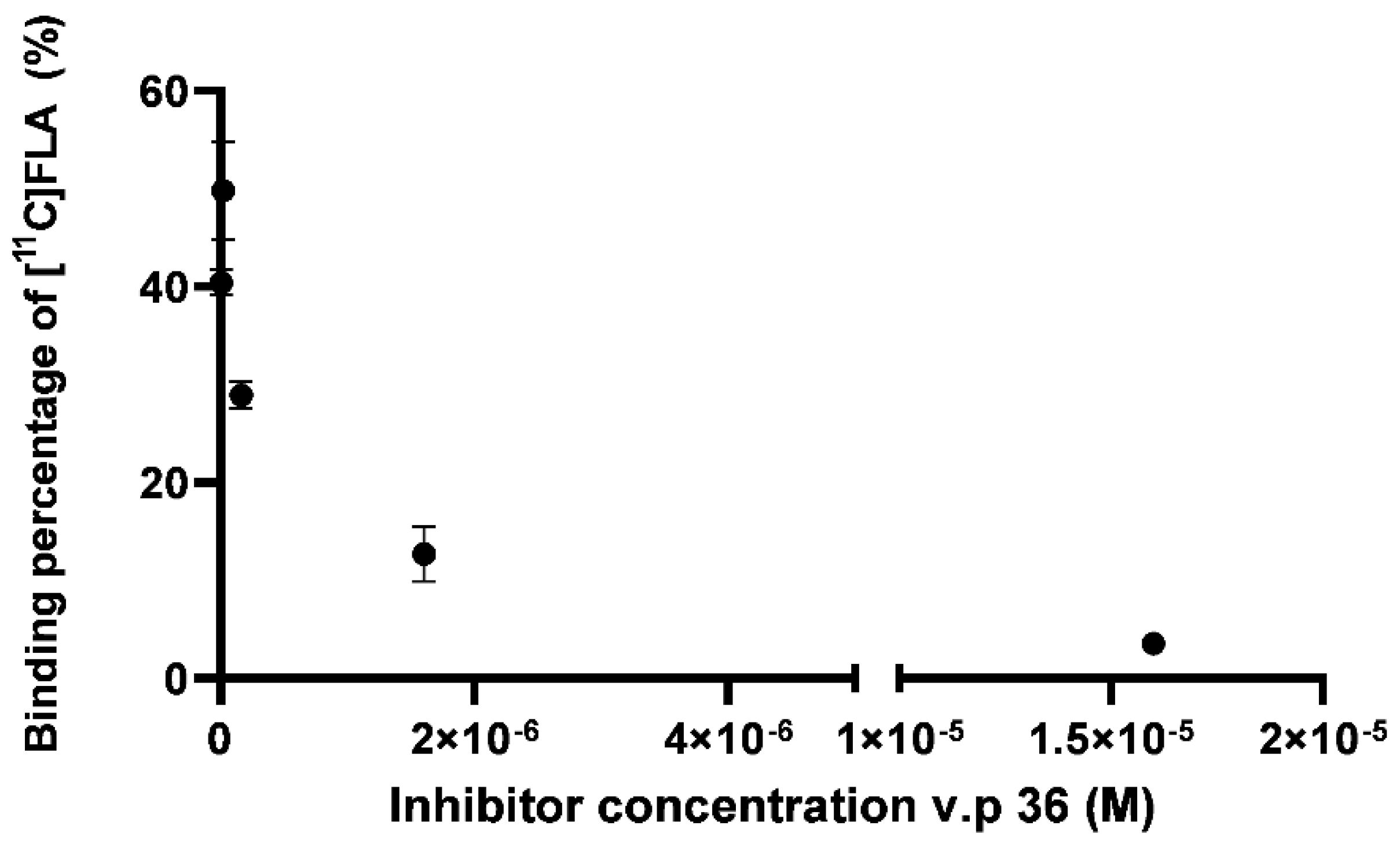
2.9.1. [11C]FLA Interaction with GSK-3β Enzyme Was Evaluated
3. Discussion
4. Materials and Methods
4.1. Chemistry
4.2. Radiochemistry
4.3. Physicochemical Evaluation
4.4. Biological Evaluation
4.5. Ex Vivo Biodistribution Studies
4.6. In Vivo Imaging Studies
4.7. In Vitro GSK-3β Inhibition Assay
4.8. Neurons and Astrocytes Primary Cultures
4.9. Uptake Studies in Neurons and Astrocytes
4.10. Competition Assay
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Singh DB, Gupta MK, Kesharwani RK, Sagar M, Dwivedi S, Misra K. Molecular drug targets and therapies for Alzheimer’s disease. Transl Neurosci. 2014;5(3):203-217. [CrossRef]
- Alzheimer Disease International. Informe Mundial sobre el Alzheimer 2019 Actitudes hacia la demencia. Glob voice Dement. Published online 2019:13.
- Hardy J. Has the Amyloid Cascade Hypothesis for Alzheimers Disease been Proved? Curr Alzheimer Res. 2006;3(1):71-73. [CrossRef]
- Durães F, Pinto M, Sousa E. Old Drugs as New Treatments for Neurodegenerative Diseases. Pharmaceuticals. 2018;11(2):44. [CrossRef]
- Mahase E. Three FDA advisory panel members resign over approval of Alzheimer’s drug. BMJ. Published online June 11, 2021:n1503. [CrossRef]
- Day GS, Scarmeas N, Dubinsky R, et al. Aducanumab Use in Symptomatic Alzheimer Disease Evidence in Focus. Neurology. 2022;98(15):619-631. [CrossRef]
- Reardon S. FDA approves Alzheimer’s drug lecanemab amid safety concerns. Nature. 2023;613(7943):227-228. [CrossRef]
- Alghamdi SS, Suliman RS, Aljammaz NA, Kahtani KM, Aljatli DA, Albadrani GM. Natural Products as Novel Neuroprotective Agents; Computational Predictions of the Molecular Targets, ADME Properties, and Safety Profile. Plants. 2022;11(4):549. [CrossRef]
- Newman DJ, Cragg GM. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J Nat Prod. 2020;83(3):770-803. [CrossRef]
- Hong J. Role of natural product diversity in chemical biology. Curr Opin Chem Biol. 2011;15(3):350-354. [CrossRef]
- Dzobo K. The Role of Natural Products as Sources of Therapeutic Agents for Innovative Drug Discovery. In: Comprehensive Pharmacology. Elsevier; 2022:408-422. [CrossRef]
- Santi MD, Peralta MA, Mendoza CS, Cabrera JL, Ortega MG. Chemical and bioactivity of flavanones obtained from roots of Dalea pazensis Rusby. Bioorg Med Chem Lett. 2017;27(8):1789-1794. [CrossRef]
- Santi MD, Arredondo F, Carvalho D, et al. Neuroprotective effects of prenylated flavanones isolated from Dalea species, in vitro and in silico studies. Eur J Med Chem. 2020;206:112718. [CrossRef]
- Eldar-Finkelman H. Glycogen synthase kinase 3: an emerging therapeutic target. Trends Mol Med. 2002;8(3):126-132. [CrossRef]
- Hur E-M, Zhou F-Q. GSK3 signalling in neural development. Nat Rev Neurosci. 2010;11(8):539-551. [CrossRef]
- Beurel E, Grieco SF, Jope RS. Glycogen synthase kinase-3 (GSK3): Regulation, actions, and diseases. Pharmacol Ther. 2015;148:114-131. [CrossRef]
- Cormier KW, Woodgett JR. Recent advances in understanding the cellular roles of GSK-3. F1000Research. 2017;6:167. [CrossRef]
- Eldar-Finkelman H, Martinez A. GSK-3 Inhibitors: Preclinical and Clinical Focus on CNS. Front Mol Neurosci. 2011;4. [CrossRef]
- Kim YT, Hur E-M, Snider WD, Zhou F-Q. Role of GSK3 Signaling in Neuronal Morphogenesis. Front Mol Neurosci. 2011;4. [CrossRef]
- Kaidanovich-Beilin O, Beaulieu J-M, Jope RS, Woodgett JR. Neurological Functions of the Masterswitch Protein Kinase – Gsk-3. Front Mol Neurosci. 2012;5. [CrossRef]
- Llorens-Martà n M, Blazquez-Llorca L, Benavides-Piccione R, et al. Selective alterations of neurons and circuits related to early memory loss in Alzheimerâ€TMs disease. Front Neuroanat. 2014;8. [CrossRef]
- Pandey MK, DeGrado TR. Glycogen Synthase Kinase-3 (GSK-3)-Targeted Therapy and Imaging. Theranostics. 2016;6(4):571-593. [CrossRef]
- Sayas CL, Ávila J. GSK-3 and Tau: A Key Duet in Alzheimer’s Disease. Cells. 2021;10(4):721. [CrossRef]
- Panche AN, Diwan AD, Chandra SR. Flavonoids: an overview. J Nutr Sci. 2016;5:e47. [CrossRef]
- Cassidy A, Kay C. Phytochemicals: Classification and Occurrence. In: Encyclopedia of Human Nutrition. Elsevier; 2013:39-46. [CrossRef]
- Ferrieri RA. Production and Application of Synthetic Precursors Labeled with Carbon-l 1 and Fluorine-18.
- Wolf AP, Redvanly CS. Carbon-11 and radiopharmaceuticals. Int J Appl Radiat Isot. 1977;28(1-2):29-48. [CrossRef]
- Schmidt S, Gonzalez D, Derendorf H. Significance of Protein Binding in Pharmacokinetics and Pharmacodynamics. J Pharm Sci. 2010;99(3):1107-1122. [CrossRef]
- Pajouhesh H, Lenz GR. Medicinal chemical properties of successful central nervous system drugs. NeuroRX. 2005;2(4):541-553. [CrossRef]
- Yao HB, Shaw PC, Wong CC, Wan DCC. Expression of glycogen synthase kinase-3 isoforms in mouse tissues and their transcription in the brain. J Chem Neuroanat. 2002;23(4):291-297. [CrossRef]
- Wang X, L. Michaelis M, K. Michaelis E. Functional Genomics of Brain Aging and Alzheimers Disease: Focus on Selective Neuronal Vulnerability. Curr Genomics. 2010;11(8):618-633. [CrossRef]
- Muyllaert D, Kremer A, Jaworski T, et al. Glycogen synthase kinase-3β, or a link between amyloid and tau pathology? Genes, Brain Behav. 2008;7(SUPPL. 1):57-66. [CrossRef]
- Perez DI, Palomo V, Pérez C, et al. Switching Reversibility to Irreversibility in Glycogen Synthase Kinase 3 Inhibitors: Clues for Specific Design of New Compounds. J Med Chem. 2011;54(12):4042-4056. [CrossRef]
- Atanasov AG, Zotchev SB, Dirsch VM, Supuran CT. Natural products in drug discovery: advances and opportunities. Nat Rev Drug Discov. 2021;20(3):200-216. [CrossRef]
- Harvey AL, Edrada-Ebel R, Quinn RJ. The re-emergence of natural products for drug discovery in the genomics era. Nat Rev Drug Discov. 2015;14(2):111-129. [CrossRef]
- Newman DJ, Cragg GM. Natural Products as Sources of New Drugs from 1981 to 2014. J Nat Prod. 2016;79(3):629-661. [CrossRef]
- Atanasov AG, Waltenberger B, Pferschy-Wenzig E-M, et al. Discovery and resupply of pharmacologically active plant-derived natural products: A review. Biotechnol Adv. 2015;33(8):1582-1614. [CrossRef]
- Waltenberger B, Mocan A, Šmejkal K, Heiss E, Atanasov A. Natural Products to Counteract the Epidemic of Cardiovascular and Metabolic Disorders. Molecules. 2016;21(6):807. [CrossRef]
- Tintore M, Vidal-Jordana A, Sastre-Garriga J. Treatment of multiple sclerosis — success from bench to bedside. Nat Rev Neurol. 2019;15(1):53-58. [CrossRef]
- Di Paolo M, Papi L, Gori F, Turillazzi E. Natural Products in Neurodegenerative Diseases: A Great Promise but an Ethical Challenge. Int J Mol Sci. 2019;20(20):5170. [CrossRef]
- Nerella SG, Singh P, Sanam T, Digwal CS. PET Molecular Imaging in Drug Development: The Imaging and Chemistry Perspective. Front Med. 2022;9. [CrossRef]
- Brooks DJ. Positron emission tomography and single-photon emission computed tomography in central nervous system drug development. NeuroRX. 2005;2(2):226-236. [CrossRef]
- Nicolazzo JA, Charman SA, Charman WN. Methods to assess drug permeability across the blood-brain barrier. J Pharm Pharmacol. 2010;58(3):281-293. [CrossRef]
- Phelps ME. Positron emission tomography provides molecular imaging of biological processes. Proc Natl Acad Sci. 2000;97(16):9226-9233. [CrossRef]
- Youdim KA, Qaiser MZ, Begley DJ, Rice-Evans CA, Abbott NJ. Flavonoid permeability across an in situ model of the blood–brain barrier. Free Radic Biol Med. 2004;36(5):592-604. [CrossRef]
- Youdim KA, Dobbie MS, Kuhnle G, Proteggente AR, Abbott NJ, Rice-Evans C. Interaction between flavonoids and the blood–brain barrier: in vitro studies. J Neurochem. 2003;85(1):180-192. [CrossRef]
- Rangel-Ordóñez L, Nöldner M, Schubert-Zsilavecz M, Wurglics M. Plasma Levels and Distribution of Flavonoids in Rat Brain after Single and Repeated Doses of Standardized Ginkgo biloba Extract EGb 761®. Planta Med. 2010;76(15):1683-1690. [CrossRef]
- Lambrecht FY, Yilmaz O, Bayrak E, Kocagozoglu G, Durkan K. Could be radiolabeled flavonoid used to evaluate infection? J Radioanal Nucl Chem. 2010;283(2):503-506. [CrossRef]
- Domínguez JM, Fuertes A, Orozco L, del Monte-Millán M, Delgado E, Medina M. Evidence for Irreversible Inhibition of Glycogen Synthase Kinase-3β by Tideglusib. J Biol Chem. 2012;287(2):893-904. [CrossRef]
- Yao H-B, Shaw P-C, Wong C-C, Wan DC-C. Expression of glycogen synthase kinase-3 isoforms in mouse tissues and their transcription in the brain. J Chem Neuroanat. 2002;23(4):291-297. [CrossRef]
| Methylating agent | Base | Solvent | Temperature (°C) |
Time (min) |
Yield (%) |
|
|---|---|---|---|---|---|---|
| I | [11C]CH3I | NaOH 5M 5µL | DMSO 0.5 mL | 80 | 1 | 0 |
| II | [11C]CH3I | TBAF 1M 100µL | Acetone 0.4 mL and THF 0.15 mL | 80 | 5 | 0 |
| III | [11C]CH3OTf | NaOH 0.5M 2µL | Acetone 0.3 mL | 25 | 4 | 16 ± 3 (n=5) |
| Time (min) | Binding percentage (%) |
|---|---|
| 5 | 4.2 ± 0.7 |
| 10 | 8.4 ± 1.1 |
| 20 | 13.7 ± 1.5 |
| Activity (MBq) | Binding percentage (%) |
|---|---|
| 0.5 | 8.3 ± 0.1 |
| 1 | 6.5 ± 0.3 |
| 2 | 5.4 ± 0.1 |
| Time (min) | Binding percentage (%) |
|---|---|
| 5 | 0.8 ± 0.1 |
| 10 | 1.0 ± 0.2 |
| 20 | 6.4 ± 0.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




