Submitted:
31 October 2024
Posted:
04 November 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
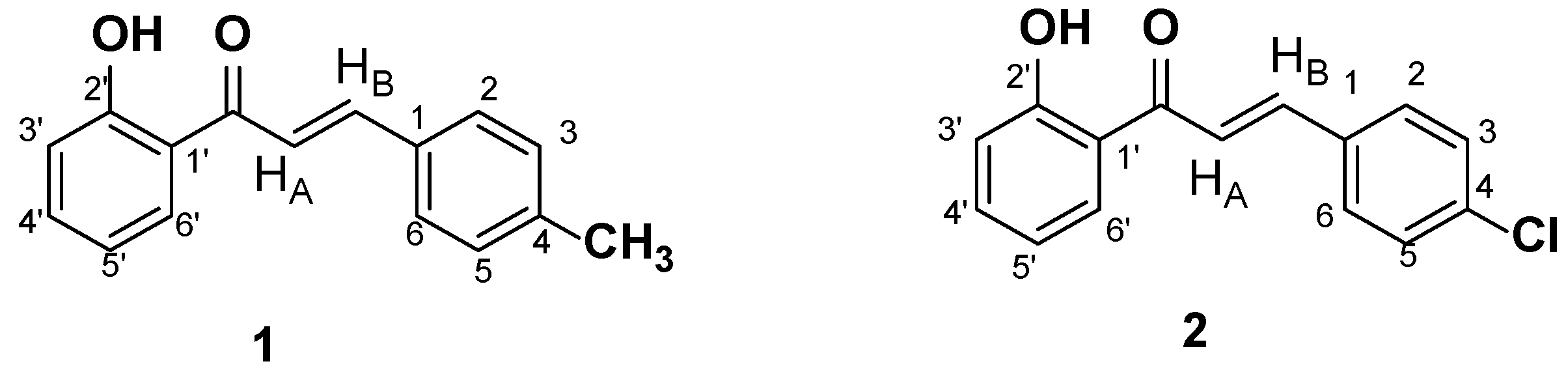
2. Materials and Methods
General Procedure for the Synthesis of Chalcones
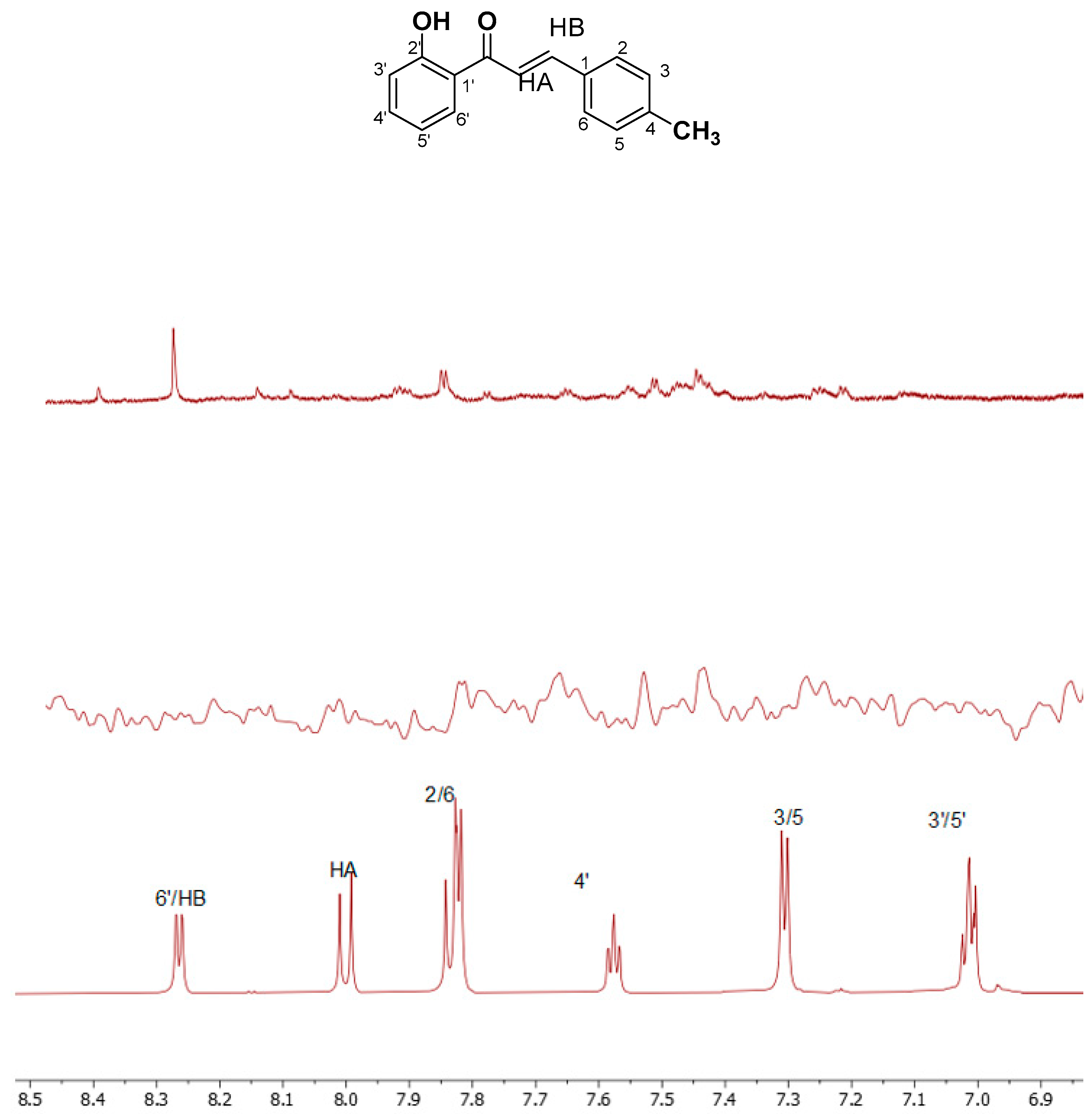
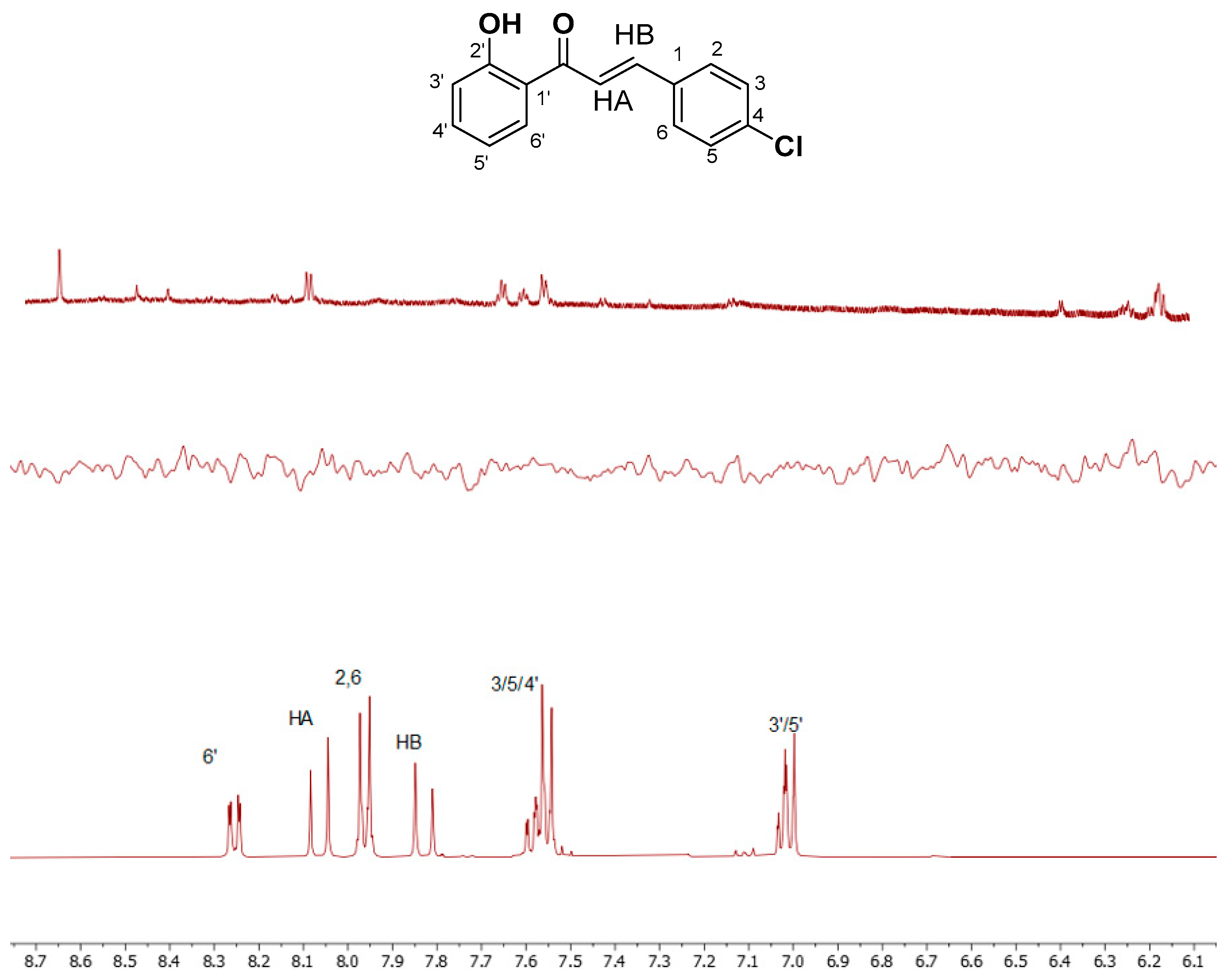
Structure Assignment
Saturation Transfer Difference (STD) NMR
Conformational Analysis
Induced Fit Docking
Molecular Dynamics
Theoretical Absorption and Fluorescence Spectra
Enzyme inhibition studies
3. Results
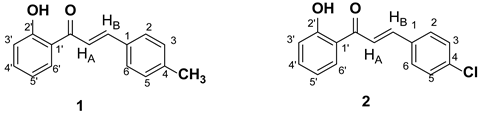
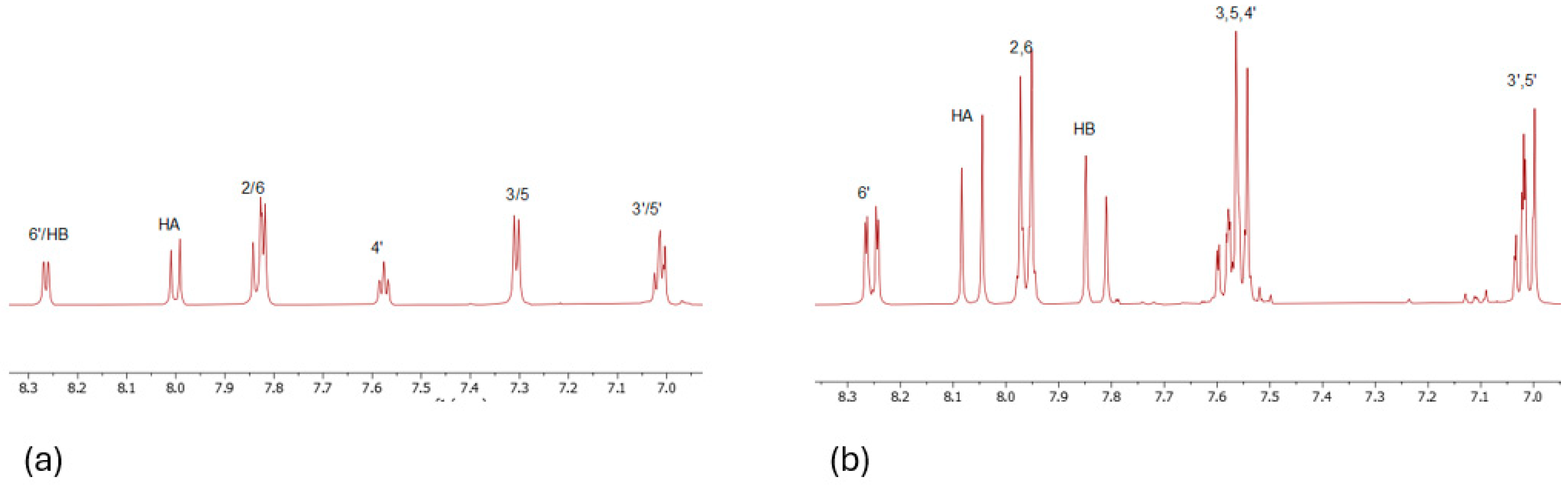
|
Hydrogen 1 |
Chemical shift (ppm) |
Hydrogen 1 |
Chemical shift (ppm) |
Hydrogen 1 |
Chemical shift (ppm) |
| CH3 | 2.36 | HA | 8.01 | 5′ | 6.96 |
| 2,6 | 7.82 | HB | 8.25 | 4′ | 7.57 |
| 3,5 | 7.29 | 3′ | 7.05 | 6′ | 8.27 |
| OH | 12.56 | ||||
|
Hydrogen 2 |
Chemical shift (ppm) |
Hydrogen 2 |
Chemical shift (ppm) |
Hydrogen 2 |
Chemical shift (ppm) |
| 2,6 | 7.96 | HB | 7.83 | 4′ | 7.58 |
| 3,5 | 7.55 | 3′ | 7.01 | 6′ | 8.25 |
| HA | 8.05 | 5′ | 7.01 | ||
| OH | 12.43 |
Conformational Analysis
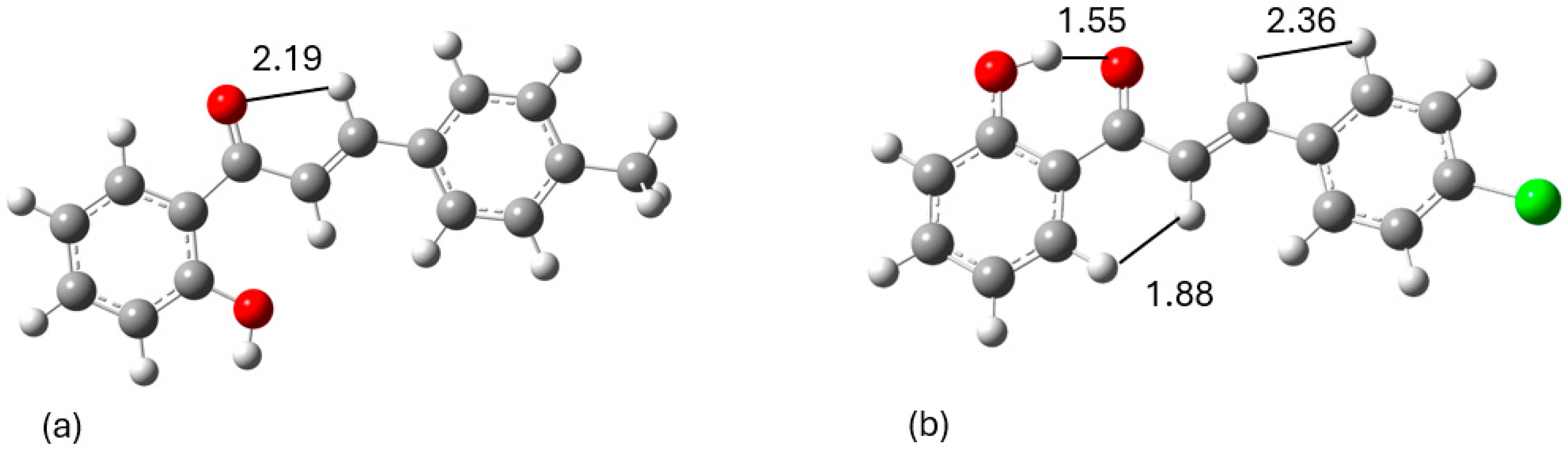
| Observed signal in 2D-NOESY spectrum | Distance in Å |
|---|---|
| 1 | |
| H2-HB | 2.192 |
| 2 | |
| H2-HB | 2.363 |
| H6′-HA | 1.882 |
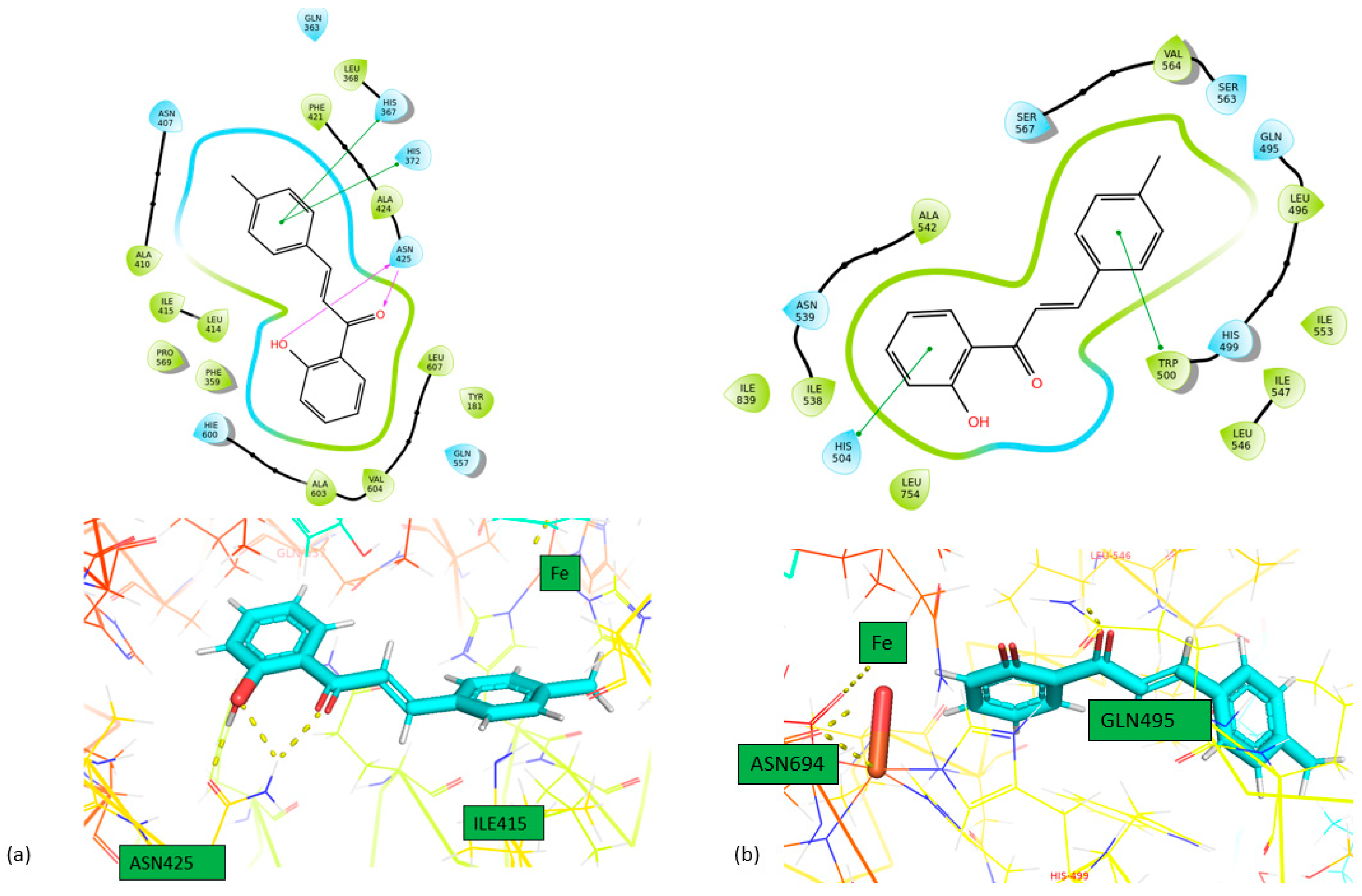
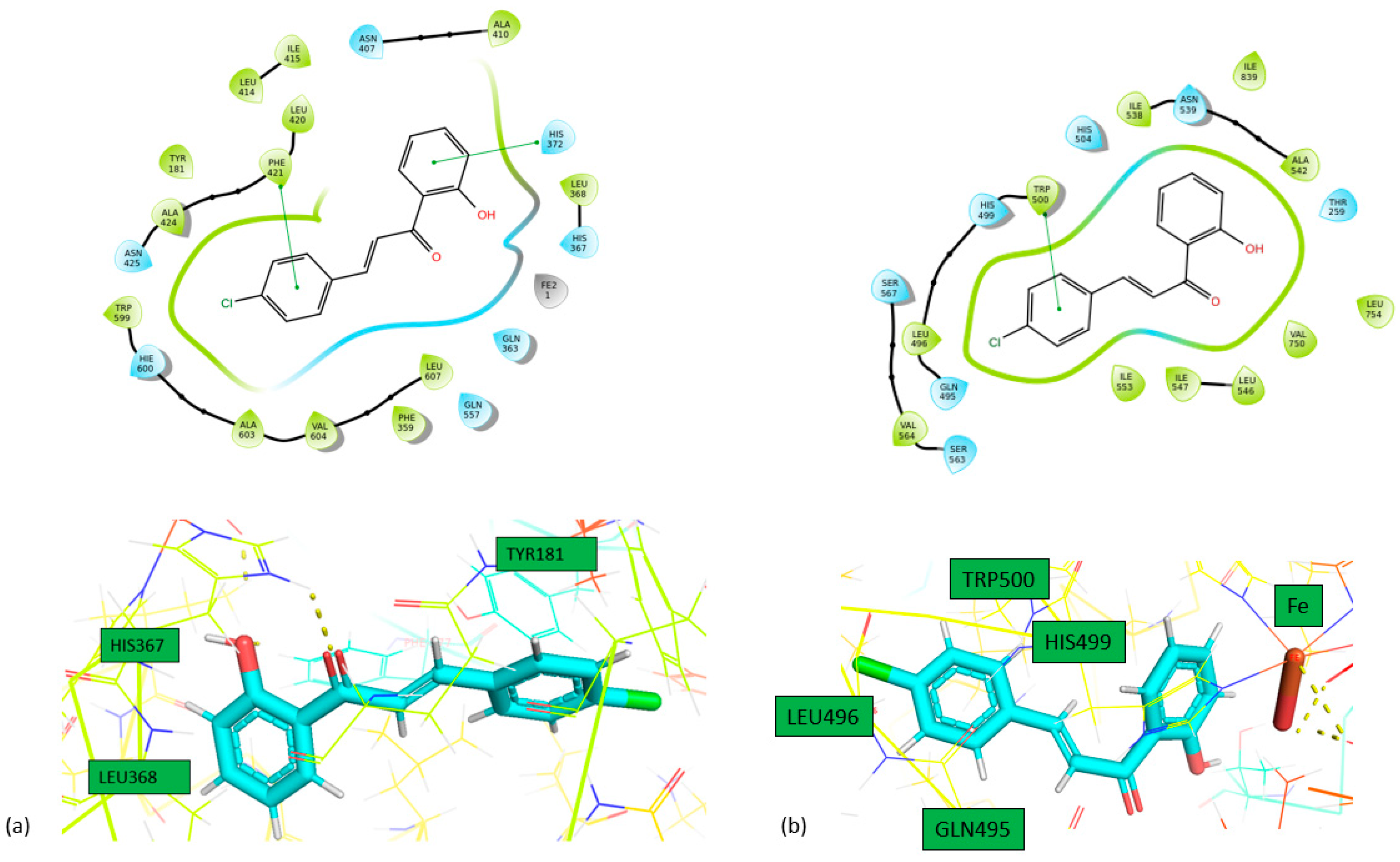
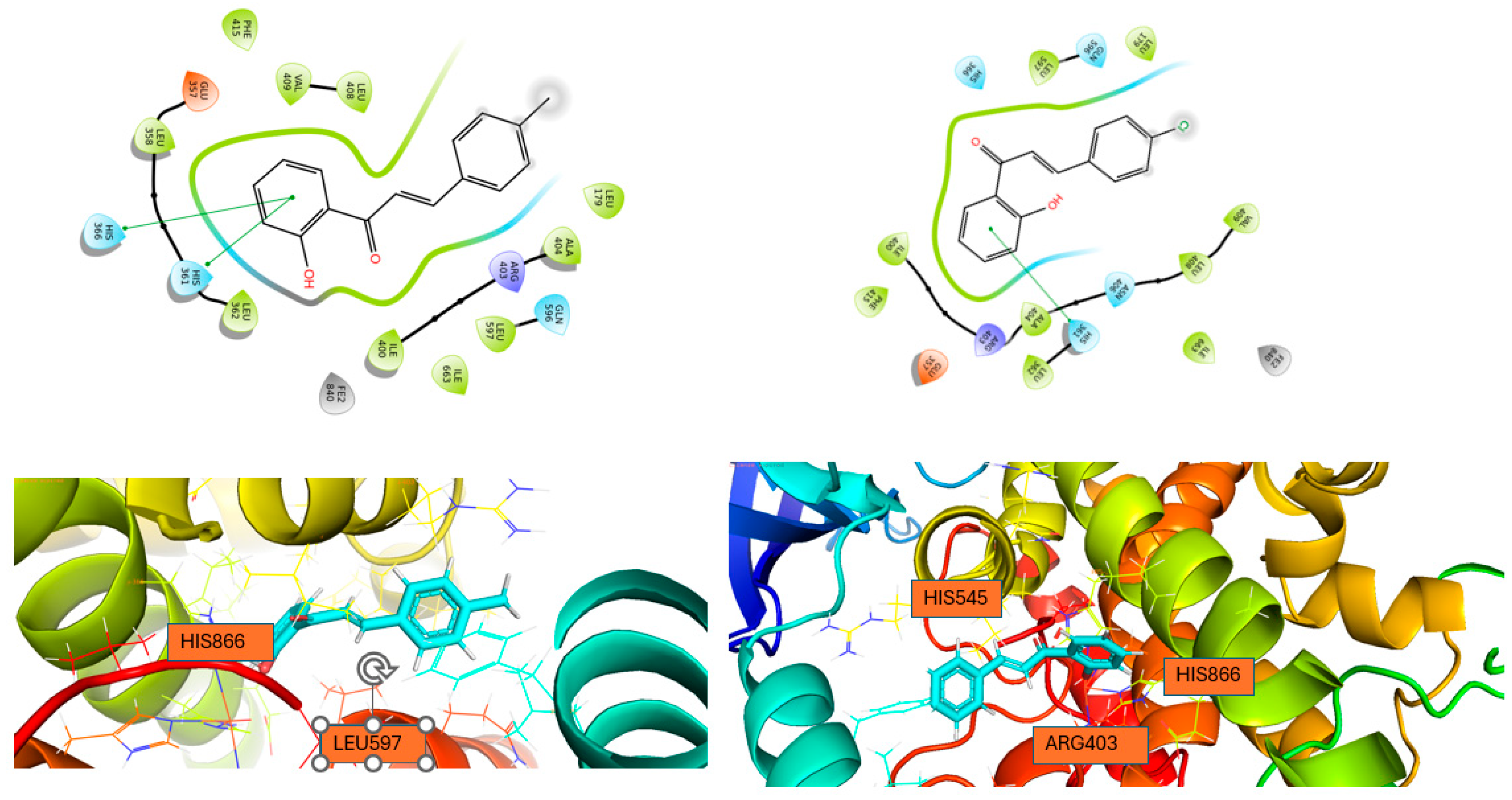
Docking Calculations
| Compound | IFD docking score (5-LOX) |
IFD docking score (LOX-1) | IFD docking score (15-LOX) |
|---|---|---|---|
|
1 |
-10.778 | -9.657 | -7.581 |
|
2 |
-11.630 | -10.256 | -7.634 |
| Compound | Enzyme | Hydrogen bonds | π-πstacking | |
| 1 | 5-LOX | ASN425 | HIS367, HIS372 | |
| LOX-1 | HIS504, TRP500 | |||
| 15-LOX | HIS361, HIS366 | |||
| 2 | 5-LOX | HIS372, PHE421 | ||
| LOX-1 | TRP500 | |||
| 15-LOX | HIS361 | |||
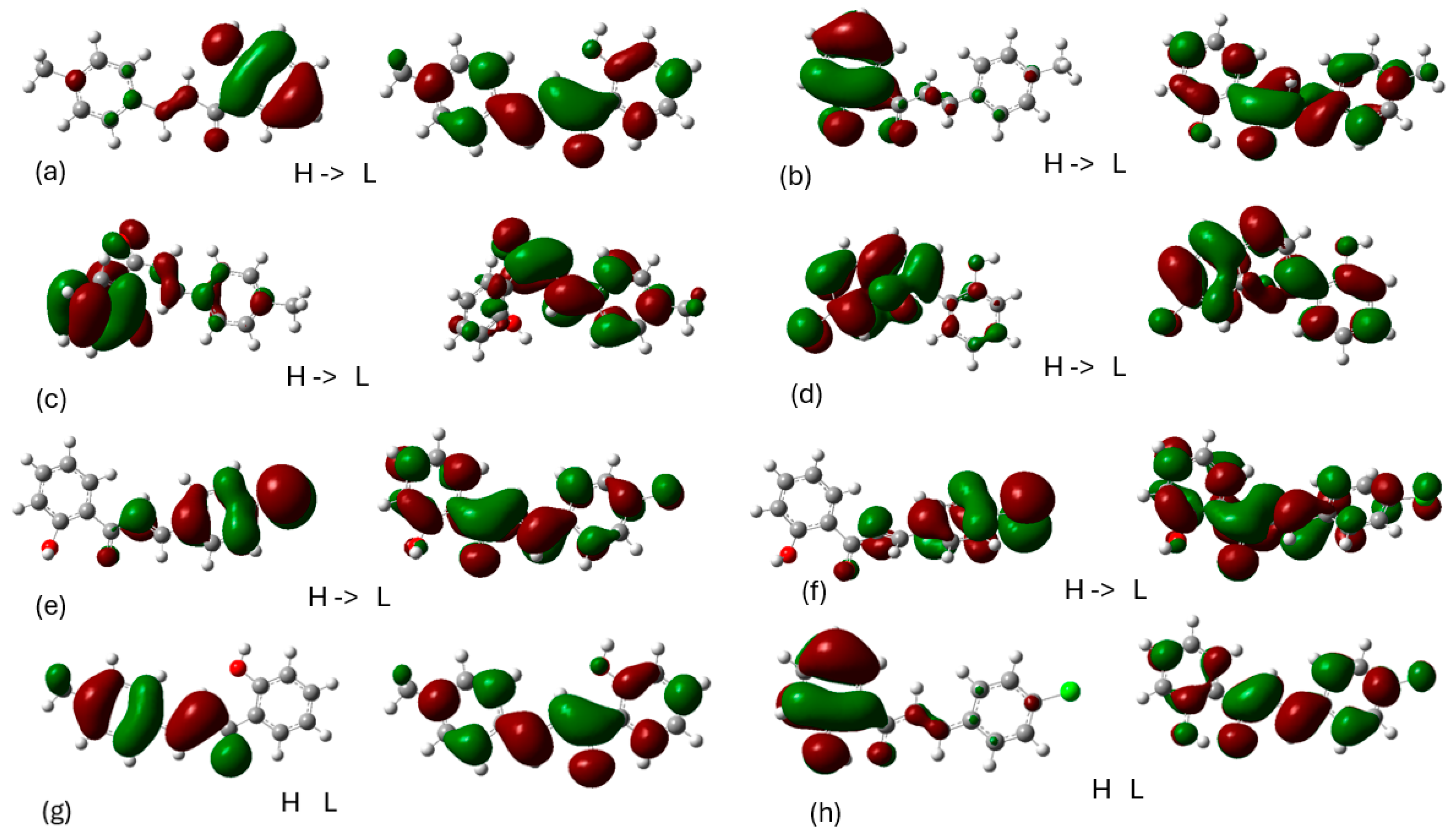
Molecular Dynamics
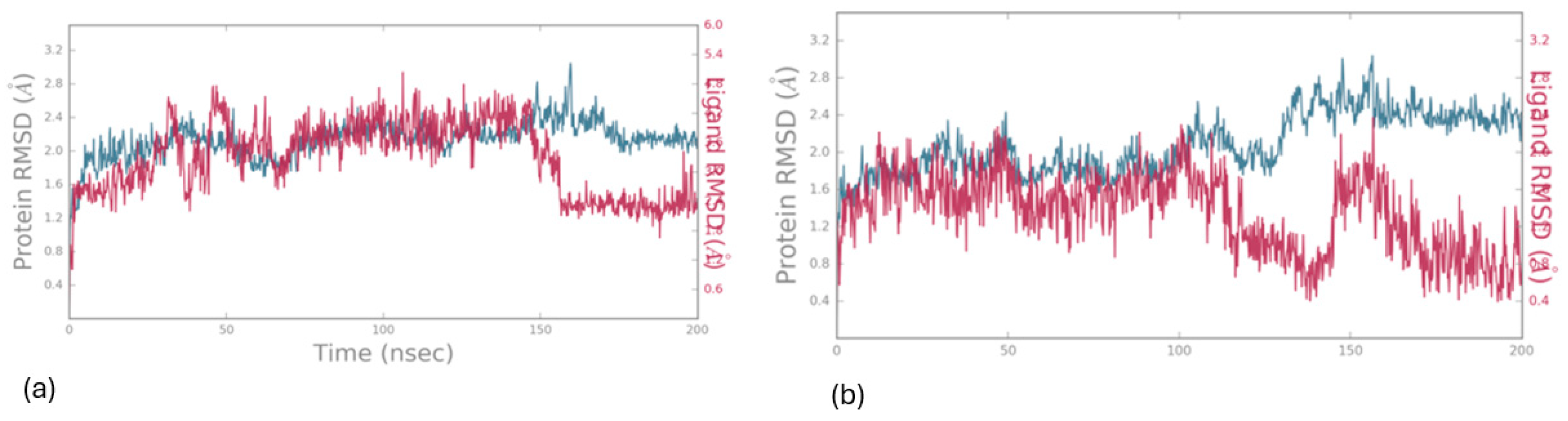
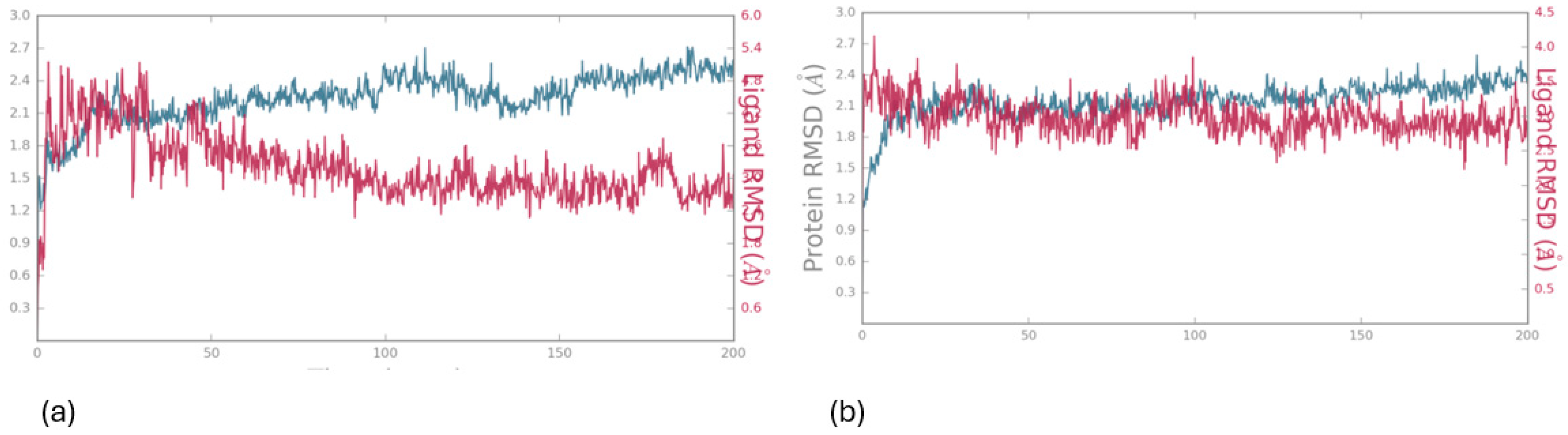
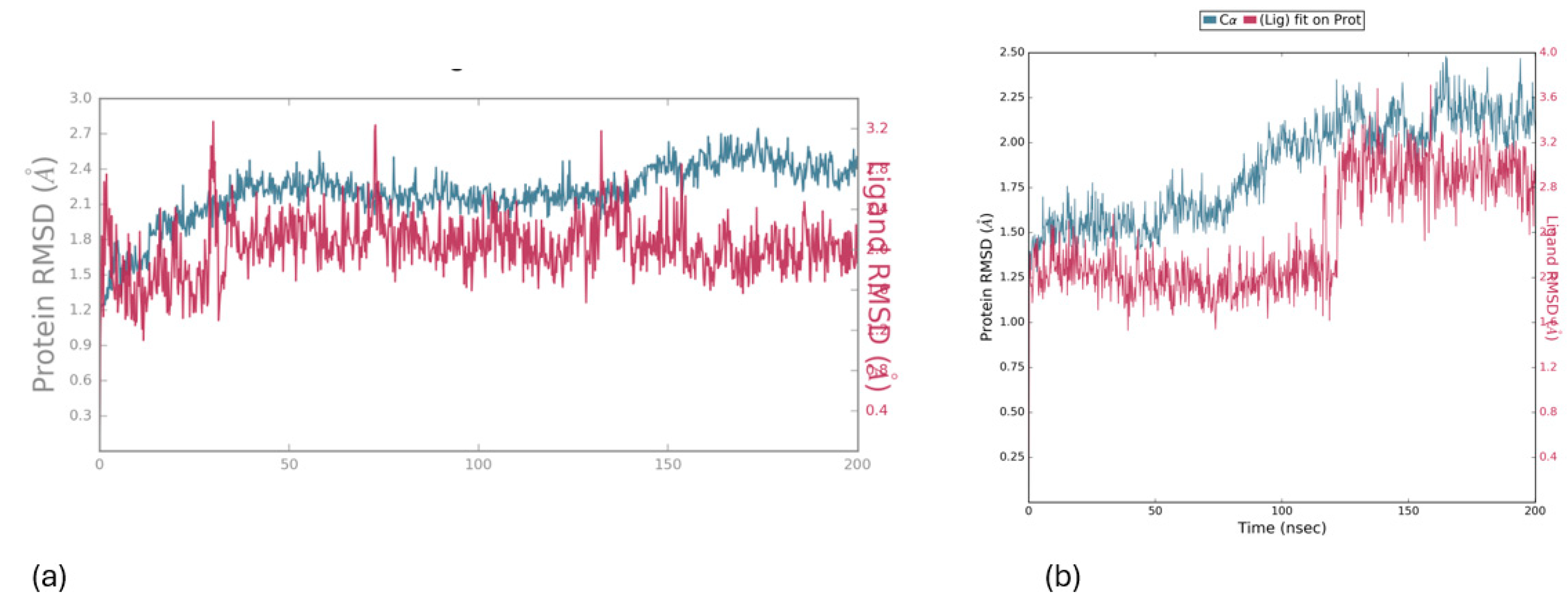
Charting Compounds - LOX-1/5-LOX Interaction Through Saturation Transfer Difference (STD) NMR Experiment
Absorption and Emission Spectra
| State | λ | ΔΕ | f | |
| 1 | S1 | 383.5 | 3.755 | 0.3485 |
| 1_LOX-1 | S1 | 403.1 | 3.076 | 0.0059 |
| 1_LOX-1 | S3 | 306.3 | 4.048 | 0.4806 |
| 1_5-LOX | S1 | 385.6 | 3.215 | 0.0005 |
| 2 | S1 | 485.3 | 2.555 | 0.2922 |
| 2_LOX-1 | S1 | 380.8 | 3.256 | 0.0043 |
| 2_LOX-1 | S3 | 305.2 | 4.062 | 0.2364 |
| 2_5-LOX | S1 | 386.1 | 3.211 | 0.0081 |
| λ | ΔΕ | f | |
|---|---|---|---|
| 1 | 532.5 | 2.328 | 0.0000 |
| a | 431.9 | 2.871 | 0.0030 |
| 337.2 | 3.677 | 0.5410 | |
| 2 | 521.0 | 2.379 | 0.1712 |
| a | 459.4 | 2.699 | 0.0299 |
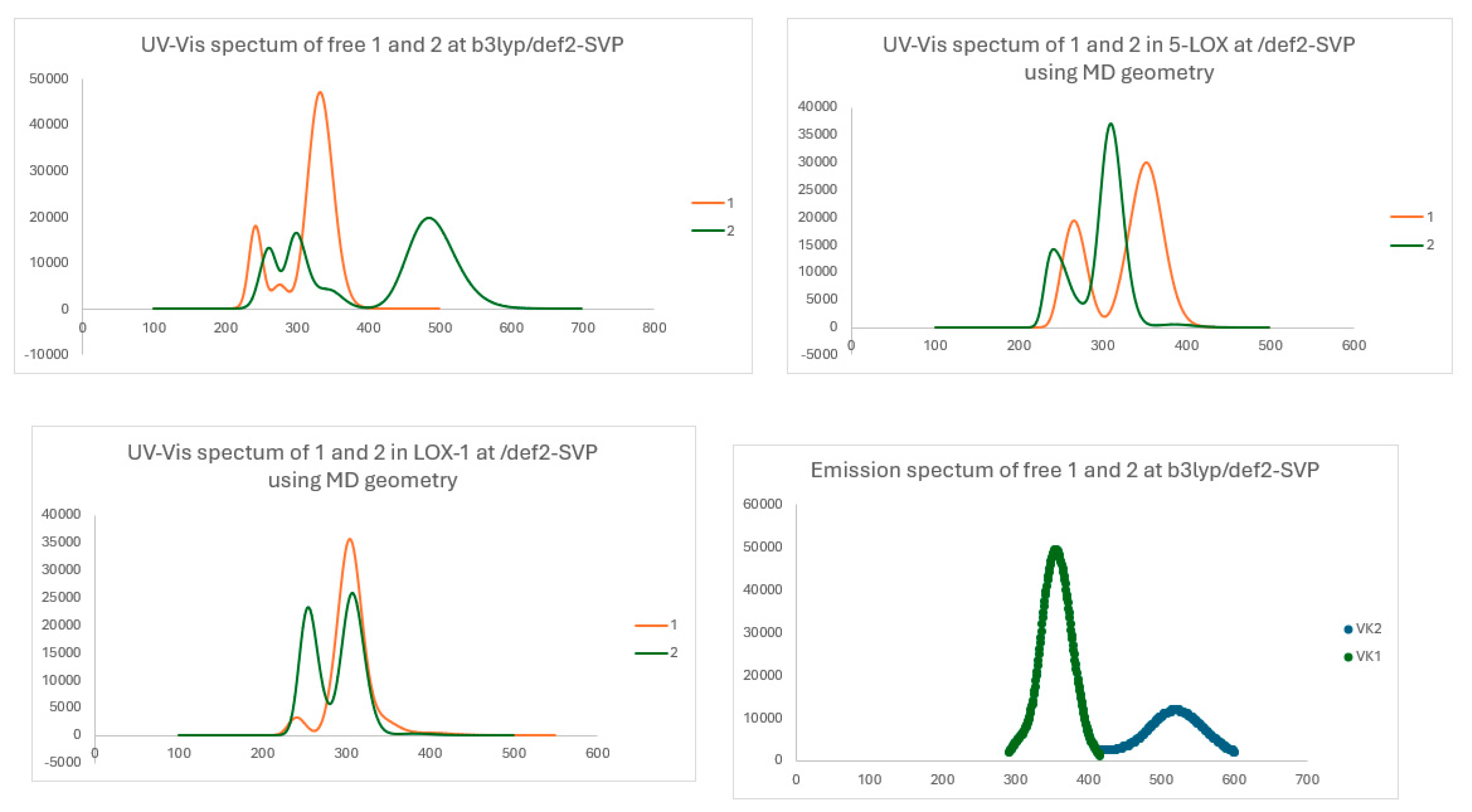
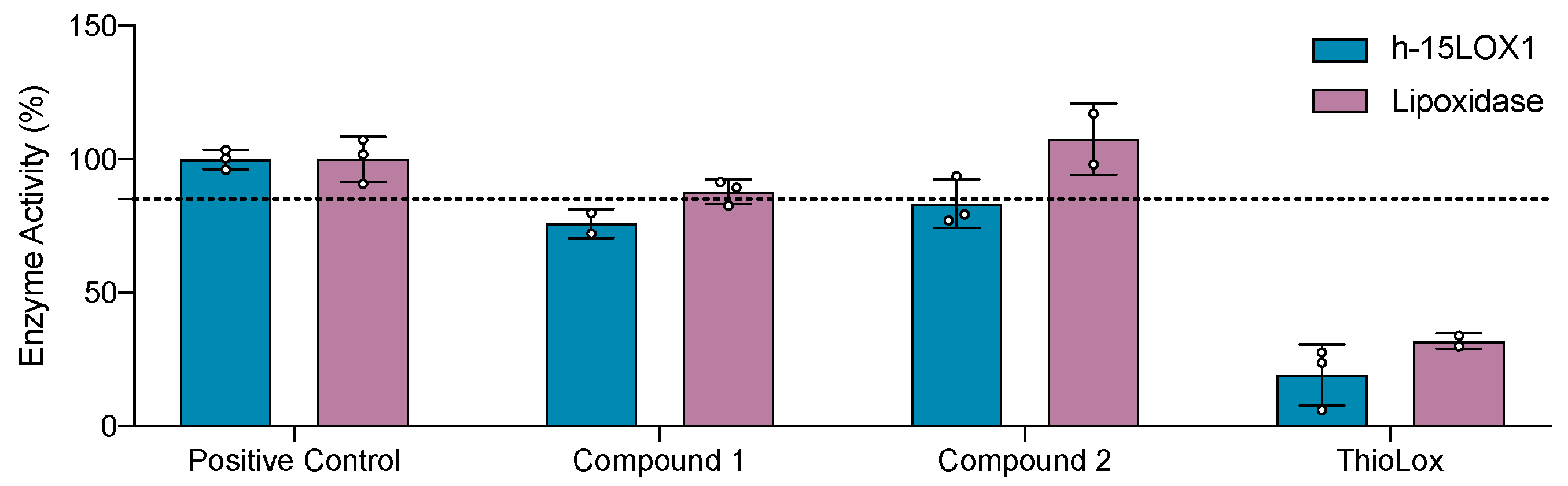
In Vitro Evaluation Against Human 15-LOX-1 and Lipoxidases Enzymes
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Qin, H.-L.; Zhang, Z.-W.; Lekkala, R.; Alsulami, H.; Rakesh, K. P. Chalcone Hybrids as Privileged Scaffolds in Antimalarial Drug Discovery: A Key Review. Eur. J. Med. Chem. 2020, 193, 112215. [CrossRef]
- Papaemmanouil, C.; Chatziathanasiadou, M. V.; Chatzigiannis, C.; Chontzopoulou, E.; Mavromoustakos, T.; Grdadolnik, S. G.; Tzakos, A. G. Unveiling the Interaction Profile of Rosmarinic Acid and Its Bioactive Substructures with Serum Albumin. J. Enzyme Inhib. Med. Chem. 2020, 35 (1), 786–804. [CrossRef]
- Detsi, A.; Majdalani, M.; Kontogiorgis, C. A.; Hadjipavlou-Litina, D.; Kefalas, P. Natural and Synthetic 2′-Hydroxy-Chalcones and Aurones: Synthesis, Characterization and Evaluation of the Antioxidant and Soybean Lipoxygenase Inhibitory Activity. Bioorganic and Medicinal Chemistry. 2009, pp 8073–8085. [CrossRef]
- Kostopoulou, I.; Tzani, A.; Polyzos, N. I.; Karadendrou, M. A.; Kritsi, E.; Pontiki, E.; Liargkova, T.; Hadjipavlou-Litina, D.; Zoumpoulakis, P.; Detsi, A. Exploring the 2′-Hydroxy-Chalcone Framework for the Development of Dual Antioxidant and Soybean Lipoxygenase Inhibitory Agents. Molecules 2021, 26 (9). [CrossRef]
- Fu, Y.; Liu, D.; Zeng, H.; Ren, X.; Song, B.; Hu, D.; Gan, X. New Chalcone Derivatives: Synthesis, Antiviral Activity and Mechanism of Action. RSC Adv. 2020, 10 (41), 24483–24490. [CrossRef]
- Nowakowska, Z. A Review of Anti-Infective and Anti-Inflammatory Chalcones. Eur. J. Med. Chem. 2007, 42 (2), 125–137. [CrossRef]
- Rosa, G. P.; Seca, A. M. L.; Barreto, M. D.; Silva, A. M. S.; Pinto, D. C. G. A. Chalcones and Flavanones Bearing Hydroxyl and/or Methoxyl Groups: Synthesis and Biological Assessments. Applied Sciences . 2019. [CrossRef]
- Tran, T.-D.; Park, H.; Kim, H. P.; Ecker, G. F.; Thai, K.-M. Inhibitory Activity of Prostaglandin E2 Production by the Synthetic 2′-Hydroxychalcone Analogues: Synthesis and SAR Study. Bioorg. Med. Chem. Lett. 2009, 19 (6), 1650–1653. [CrossRef]
- Pande, A. N.; Biswas, S.; Reddy, N. D.; Jayashree, B. S.; Kumar, N.; Rao, C. M. In Vitro and in Vivo Anticancer Studies of 2′-Hydroxy Chalcone Derivatives Exhibit Apoptosis in Colon Cancer Cells by HDAC Inhibition and Cell Cycle Arrest. EXCLI J. 2017, 16, 448.
- Kuhn, H.; Banthiya, S.; Van Leyen, K. Mammalian Lipoxygenases and Their Biological Relevance. Biochim. Biophys. Acta (BBA)-Molecular Cell Biol. Lipids 2015, 1851 (4), 308–330.
- Georgiou, N.; Chontzopoulou, E.; Cheilari, A.; Katsogiannou, A.; Karta, D.; Vavougyiou, K.; Hadjipavlou-Litina, D.; Javornik, U.; Plavec, J.; Tzeli, D. Thiocarbohydrazone and Chalcone-Derived 3, 4-Dihydropyrimidinethione as Lipid Peroxidation and Soybean Lipoxygenase Inhibitors. ACS omega 2023, 8 (13), 11966–11977.
- Tzani, A.; Kritsi, E.; Tsamantioti, L.; Kostopoulou, I.; Karadendrou, M.-A.; Zoumpoulakis, P.; Detsi, A. Synthesis, Conformational Analysis and CtDNA Binding Studies of Flavonoid Analogues Possessing the 3,5-Di-Tert-Butyl-4-Hydroxyphenyl Moiety. Antioxidants. 2022. [CrossRef]
- Lin, Y.-M.; Zhou, Y.; Flavin, M. T.; Zhou, L.-M.; Nie, W.; Chen, F.-C. Chalcones and Flavonoids as Anti-Tuberculosis Agents. Bioorg. Med. Chem. 2002, 10 (8), 2795–2802.
- Georgiou, N.; Katsogiannou, A.; Skourtis, D.; Iatrou, H.; Tzeli, D.; Vassiliou, S.; Javornik, U.; Plavec, J.; Mavromoustakos, T. Conformational Properties of New Thiosemicarbazone and Thiocarbohydrazone Derivatives and Their Possible Targets. Molecules 2022, 27 (8), 2537. [CrossRef]
- Georgiou, N.; Gouleni, N.; Chontzopoulou, E.; Skoufas, G. S.; Gkionis, A.; Tzeli, D.; Vassiliou, S.; Mavromoustakos, T. Structure Assignment, Conformational Properties and Discovery of Potential Targets of the Ugi Cinnamic Adduct NGI25. J. Biomol. Struct. Dyn. 2021, 0 (0), 1–14. [CrossRef]
- Georgiou, N.; Chontzopoulou, E.; Cheilari, A.; Katsogiannou, A.; Karta, D.; Vavougyiou, K.; Hadjipavlou-Litina, D.; Javornik, U.; Plavec, J.; Tzeli, D.; Vassiliou, S.; Mavromoustakos, T. Thiocarbohydrazone and Chalcone-Derived 3,4-Dihydropyrimidinethione as Lipid Peroxidation and Soybean Lipoxygenase Inhibitors. ACS Omega 2022. [CrossRef]
- Leonis, G.; Vakali, V.; Zoupanou, N.; Georgiou, N.; Diamantis, D. A.; Tzakos, A. G.; Mavromoustakos, T.; Tzeli, D. Computational and Spectroscopic Analysis of the Quercetin Encapsulation in (2HP-β-CD)2 and (2,6Me-β-CD)2 Complexes. J. Mol. Struct. 2023, 1294 (P2), 136430. [CrossRef]
- Willcott, M. R. MestRe Nova. J. Am. Chem. Soc. 2009, 131 (36), 13180–13180. [CrossRef]
- Chontzopoulou, E.; Papaemmanouil, C. D.; Chatziathanasiadou, M. V.; Kolokouris, D.; Kiriakidi, S.; Konstantinidi, A.; Gerogianni, I.; Tselios, T.; Kostakis, I. K.; Chrysina, E. D.; Hadjipavlou-Litina, D.; Tzeli, D.; Tzakos, A. G.; Mavromoustakos, T. Molecular Investigation of Artificial and Natural Sweeteners as Potential Anti-Inflammatory Agents. J. Biomol. Struct. Dyn. 2021, 0 (0), 1–13. [CrossRef]
- Hall, J. L.; Sohail, A.; Cabrita, E. J.; Macdonald, C.; Stockner, T.; Sitte, H. H.; Angulo, J.; MacMillan, F. Saturation Transfer Difference NMR on the Integral Trimeric Membrane Transport Protein GltPh Determines Cooperative Substrate Binding. Sci. Rep. 2020, 10 (1), 1–9. [CrossRef]
- Viegas, A.; Manso, J.; Nobrega, F. L.; Cabrita, E. J. Saturation-Transfer Difference (STD) NMR: A Simple and Fast Method for Ligand Screening and Characterization of Protein Binding. J. Chem. Educ. 2011, 88 (7), 990–994. [CrossRef]
- van Mourik, T.; Bühl, M.; Gaigeot, M.-P. Density Functional Theory across Chemistry, Physics and Biology. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2014, 372 (2011), 20120488. [CrossRef]
- Grimme, S.; Antony, J.; Ehrlich, S.; Krieg, H. A Consistent and Accurate Ab Initio Parametrization of Density Functional Dispersion Correction (DFT-D) for the 94 Elements H-Pu. J. Chem. Phys. 2010, 132 (15). [CrossRef]
- Weigend, F.; Ahlrichs, R. Balanced Basis Sets of Split Valence, Triple Zeta Valence and Quadruple Zeta Valence Quality for H to Rn: Design and Assessment of Accuracy. Phys. Chem. Chem. Phys. 2005, 7 (18), 3297. [CrossRef]
- Neese, F.; Wennmohs, F.; Becker, U.; Riplinger, C. The ORCA Quantum Chemistry Program Package. J. Chem. Phys. 2020, 152 (22). [CrossRef]
- Zeraik, M. L.; Pauli, I.; Dutra, L. A.; Cruz, R. S.; Valli, M.; Paracatu, L. C.; de Faria, C. M. Q. G.; Ximenes, V. F.; Regasini, L. O.; Andricopulo, A. D.; Bolzani, V. S. Identification of a Prenyl Chalcone as a Competitive Lipoxygenase Inhibitor: Screening, Biochemical Evaluation and Molecular Modeling Studies. Molecules 2021, 26 (8), 2205. [CrossRef]
- Dubé, D.; Blouin, M.; Brideau, C.; Chan, C.-C.; Desmarais, S.; Ethier, D.; Falgueyret, J.-P.; Friesen, R. W.; Girard, M.; Girard, Y.; Guay, J.; Riendeau, D.; Tagari, P.; Young, R. N. Quinolines as Potent 5-Lipoxygenase Inhibitors: Synthesis and Biological Profile of L-746,530. Bioorg. Med. Chem. Lett. 1998, 8 (10), 1255–1260. [CrossRef]
- Offenbacher, A. R.; Hu, S.; Poss, E. M.; Carr, C. A. M.; Scouras, A. D.; Prigozhin, D. M.; Iavarone, A. T.; Palla, A.; Alber, T.; Fraser, J. S.; Klinman, J. P. Hydrogen–Deuterium Exchange of Lipoxygenase Uncovers a Relationship between Distal, Solvent Exposed Protein Motions and the Thermal Activation Barrier for Catalytic Proton-Coupled Electron Tunneling. ACS Cent. Sci. 2017, 3 (6), 570–579. [CrossRef]
- Choi, J.; Chon, J. K.; Kim, S.; Shin, W. Conformational Flexibility in Mammalian 15S-lipoxygenase: Reinterpretation of the Crystallographic Data. Proteins Struct. Funct. Bioinforma. 2008, 70 (3), 1023–1032. [CrossRef]
- Gilbert, N. C.; Bartlett, S. G.; Waight, M. T.; Neau, D. B.; Boeglin, W. E.; Brash, A. R.; Newcomer, M. E. The Structure of Human 5-Lipoxygenase. Science (80-. ). 2011, 331 (6014), 217–219. [CrossRef]
- Schrodinger, L.L.C. MacroModel, Version 10.2. New York 2013.
- Jorgensen, W.L.; Maxwell, D.S.; Tirado-Rives, J. Development and Testing of the OPLS All-Atom Force Field on Conformational Energetics and Properties of Organic Liquids. J. Am. Chem. Soc. 118, 11225–11236,. [CrossRef]
- Imtiaz, S.; Muzaffar, S.; Ali, S. M. Demonstrating Accuracy of the Already Proposed Protocol for Structure Elucidation of Cyclodextrin Inclusion Complexes by Validation Using Quantitative ROESY Analysis. J. Incl. Phenom. Macrocycl. Chem. 2021, 100 (1–2), 71–87. [CrossRef]
- Essmann, U.; Perera, L.; Berkowitz, M.L.; Darden, T.; Lee, H.; Pedersen, L. G. A Smooth Particle Mesh Ewald Method. J. Chem. Phys. 103, 8577,. [CrossRef]
- Martyna, G.J.; Tobias, D.J.; Klein, M. L. Constant Pressure Molecular Dynamics Algorithms. J. Chem. Phys. 1994, 101, 4177–4189,. [CrossRef]
- Humphreys, D.D.; Friesner, R.A.; Berne, B. J. A Multiple-Time-Step Molecular Dynamics Algorithm for Macromolecules. J. Phys. Chem. 1994. [CrossRef]
- Lyman, E.; Zuckerman, D. M. Ensemble-Based Convergence Analysis of Biomolecular Trajectories. Biophys. J. 2006 91, 164–172,. [CrossRef]
- Version, D.D.; Version, D. D. Desmond Tutorial. Schroedinger. [CrossRef]
- Lee, C.; Yang, W.; Parr, R. G. Development of the Colle-Salvetti Correlation-Energy Formula into a Functional of the Electron Density. Phys. Rev. B 1988, 37 (2), 785–789. [CrossRef]
- Becke, A. D. A New Mixing of Hartree–Fock and Local Density-functional Theories. J. Chem. Phys. 1993, 98 (2), 1372–1377. [CrossRef]
- Tzeli, D.; Theodorakopoulos, G.; Petsalakis, I. D.; Ajami, D.; Rebek, J. Conformations and Fluorescence of Encapsulated Stilbene. J. Am. Chem. Soc. 2012, 134 (9), 4346–4354. [CrossRef]
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V., D. J. F. Gaussian 16, Revision B.01. In Gaussian 09; 2016.
- Eleftheriadis, N.; Poelman, H.; Leus, N. G. J.; Honrath, B.; Neochoritis, C. G.; Dolga, A.; Dömling, A.; Dekker, F. J. Design of a Novel Thiophene Inhibitor of 15-Lipoxygenase-1 with Both Anti-Inflammatory and Neuroprotective Properties. Eur. J. Med. Chem. 2016, 122, 786–801. [CrossRef]
- Spacho, N.; Casertano, M.; Imperatore, C.; Papadopoulos, C.; Menna, M.; Eleftheriadis, N. Investigating the Catalytic Site of Human 15-Lipoxygenase-1 via Marine Natural Products. Chem. – A Eur. J. 2024. [CrossRef]
- Eleftheriadis, N.; Thee, S. A.; Zwinderman, M. R. H.; Leus, N. G. J.; Dekker, F. J. Activity-Based Probes for 15-Lipoxygenase-1. Angew. Chemie Int. Ed. 2016, 55 (40), 12300–12305. [CrossRef]
- Eleftheriadis, N.; Neochoritis, C. G.; Leus, N. G. J.; van der Wouden, P. E.; Dömling, A.; Dekker, F. J. Rational Development of a Potent 15-Lipoxygenase-1 Inhibitor with in Vitro and Ex Vivo Anti-Inflammatory Properties. J. Med. Chem. 2015, 58 (19), 7850–7862. [CrossRef]
- Mayer, M.; Meyer, B. Characterization of Ligand Binding by Saturation Transfer Difference NMR Spectroscopy. Angew. Chemie Int. Ed. 1999, 38 (12), 1784–1788. [CrossRef]
- Dalvit, C.; Pevarello, P.; Tat, M.; Veronesi, M.; Vulpetti, A.; Sundström, M. 상아방2000-Identification+of+compounds+with+binding+affinity+to.PDF. 2000, 65–68.
- Snaith, H. J. Perovskites: The Emergence of a New Era for Low-Cost, High-Efficiency Solar Cells. J. Phys. Chem. Lett. 2013, 4 (21), 3623–3630. [CrossRef]
- Ethirajan, M.; Chen, Y.; Joshi, P.; Pandey, R. K. The Role of Porphyrin Chemistry in Tumor Imaging and Photodynamic Therapy. Chem. Soc. Rev. 2011, 40 (1), 340–362. [CrossRef]
- Weissleder, R. A Clearer Vision for in Vivo Imaging. Nat. Biotechnol. 2001, 19 (4), 316–317. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




