Submitted:
31 October 2024
Posted:
31 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
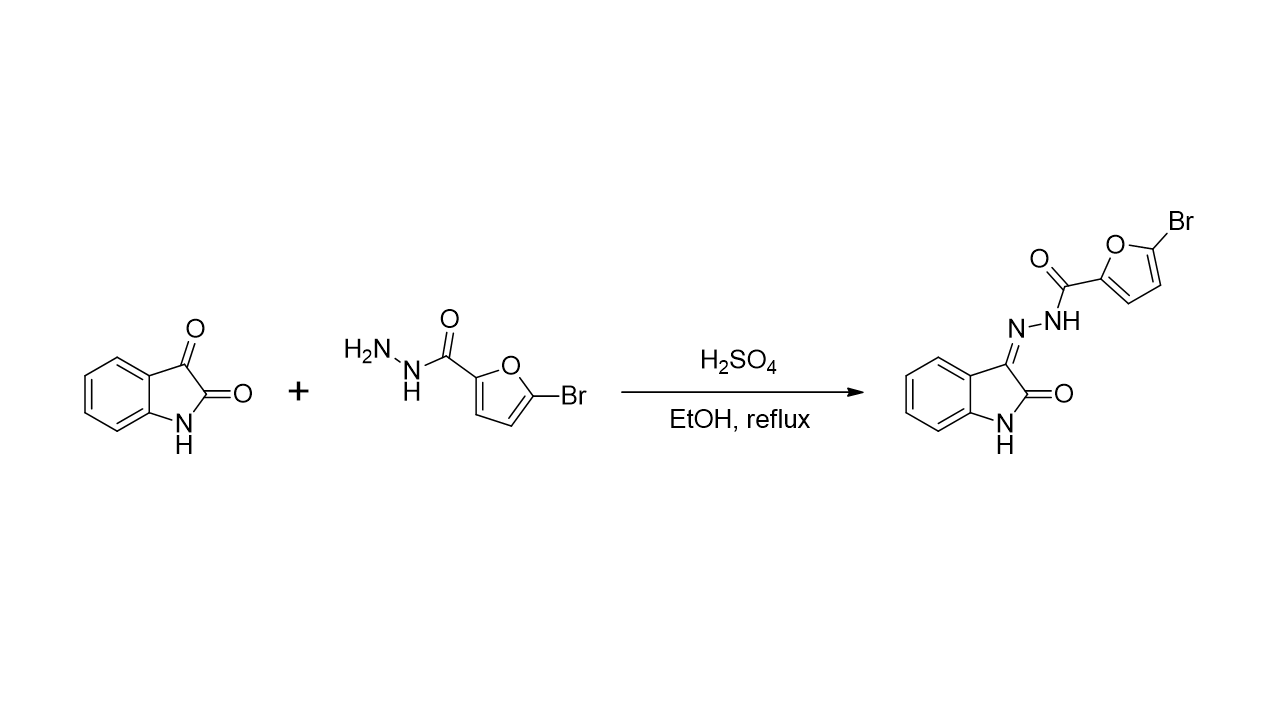
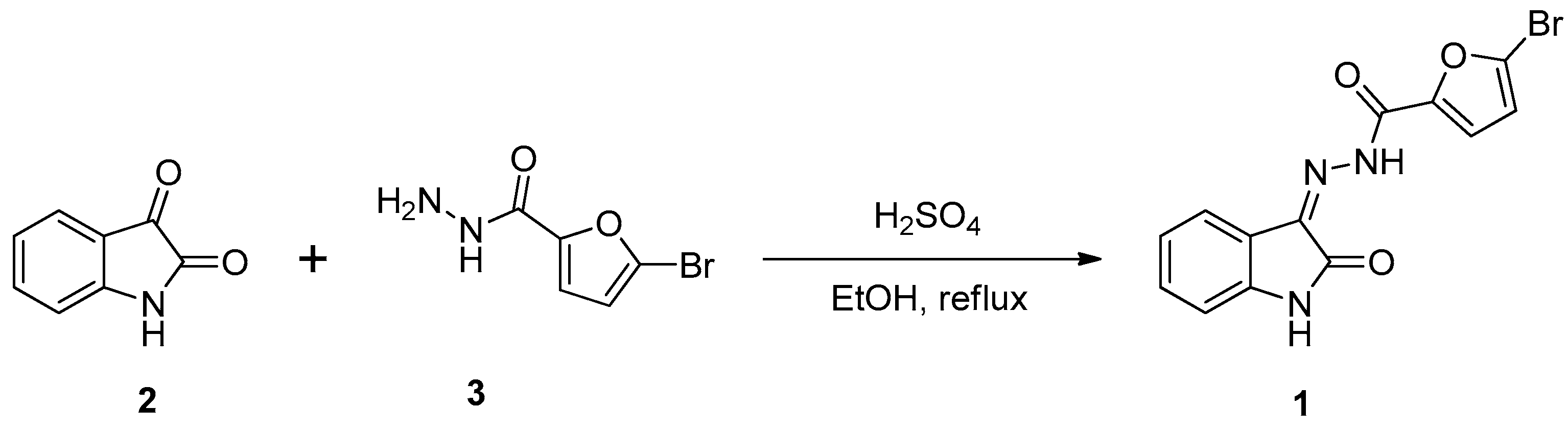
2.1. Chemistry
2.2. In vitro Anti-inflammatory Properties
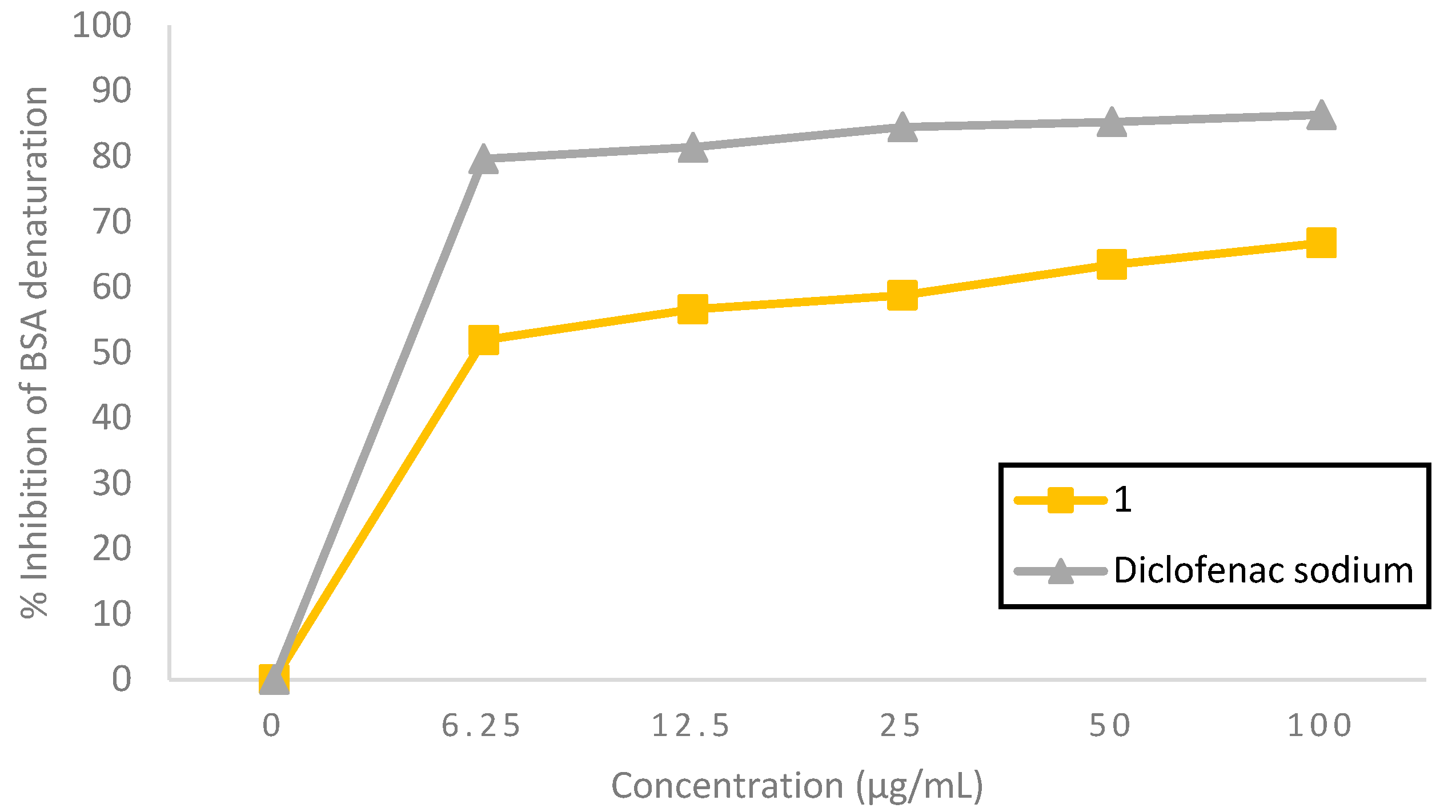
2.2.1. Bovine Serum Albumin (BSA) Denaturation Effect
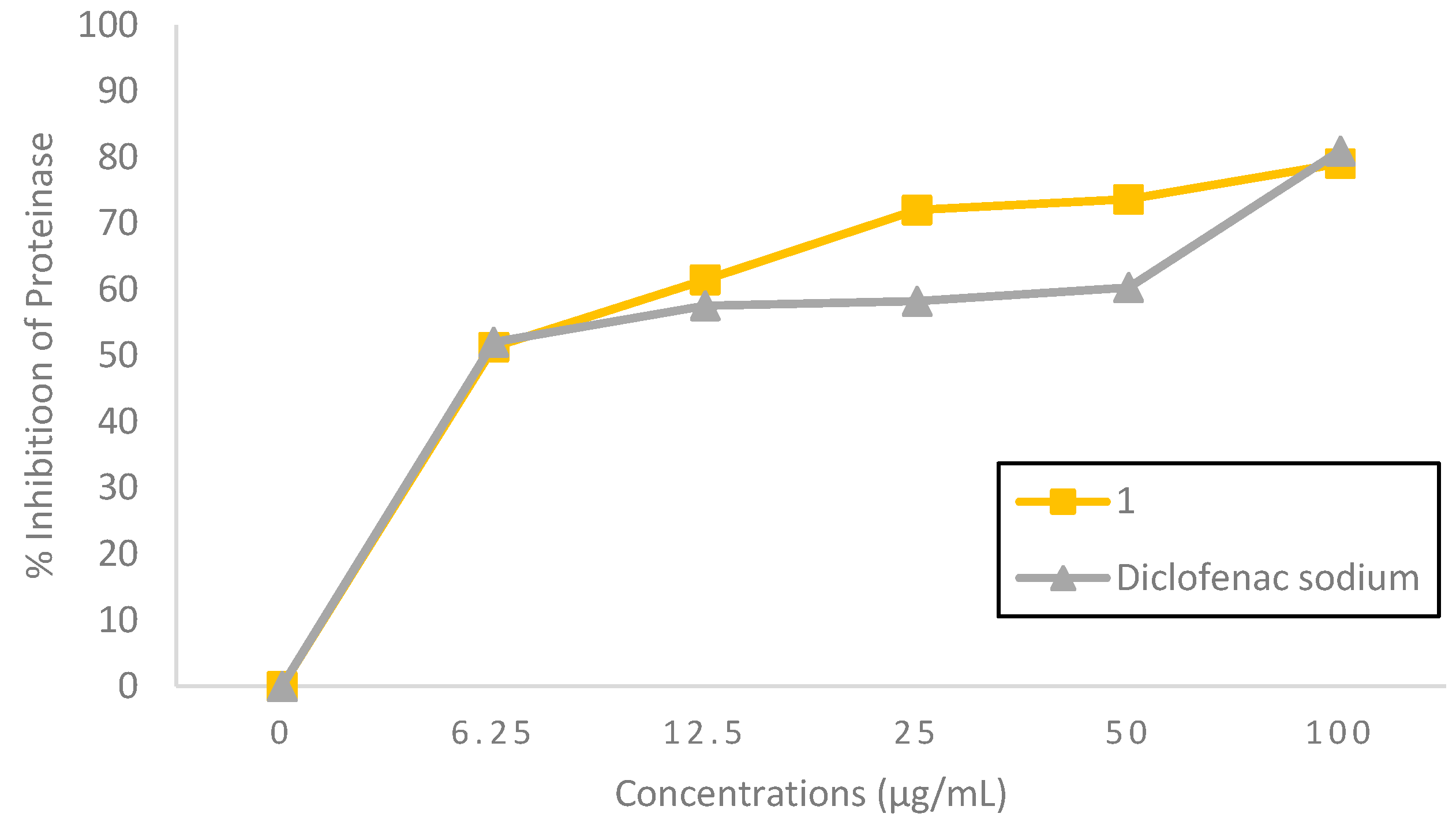
2.2.2. Protein Inhibition Effect
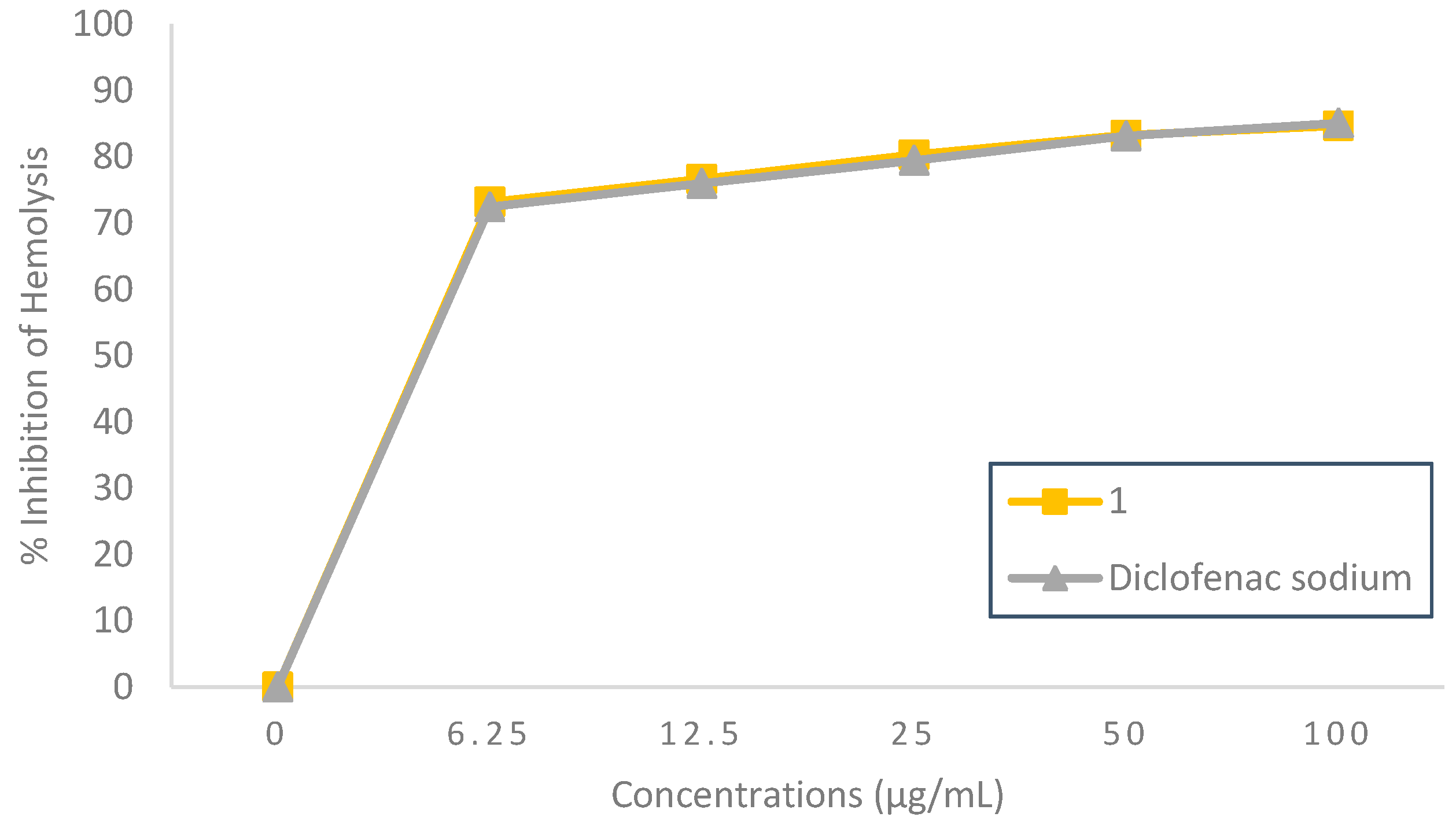
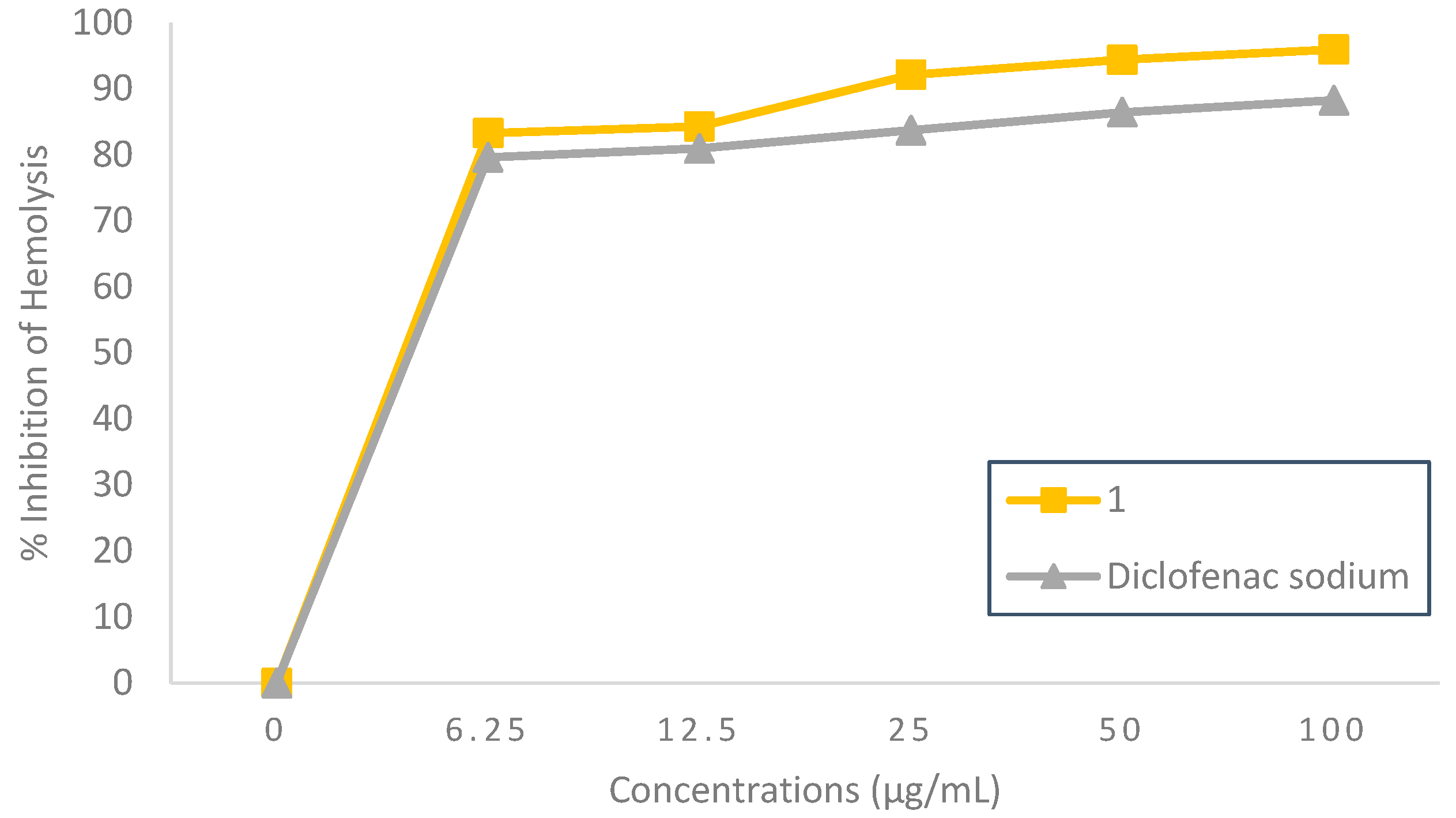
2.2.3. Membrane Stabilizing Effect
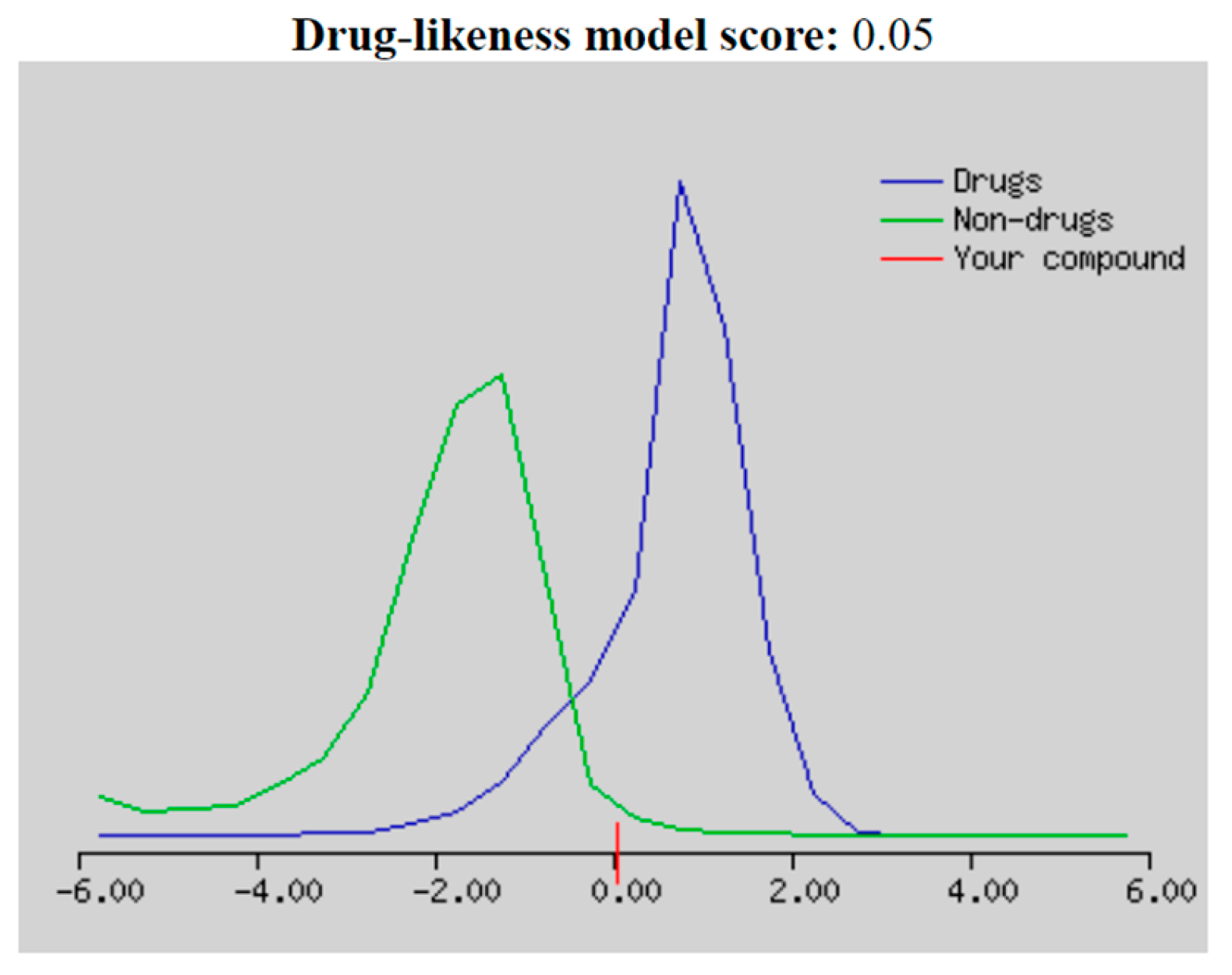
2.3. Drug-likeness, Molecular and ADMET Properties
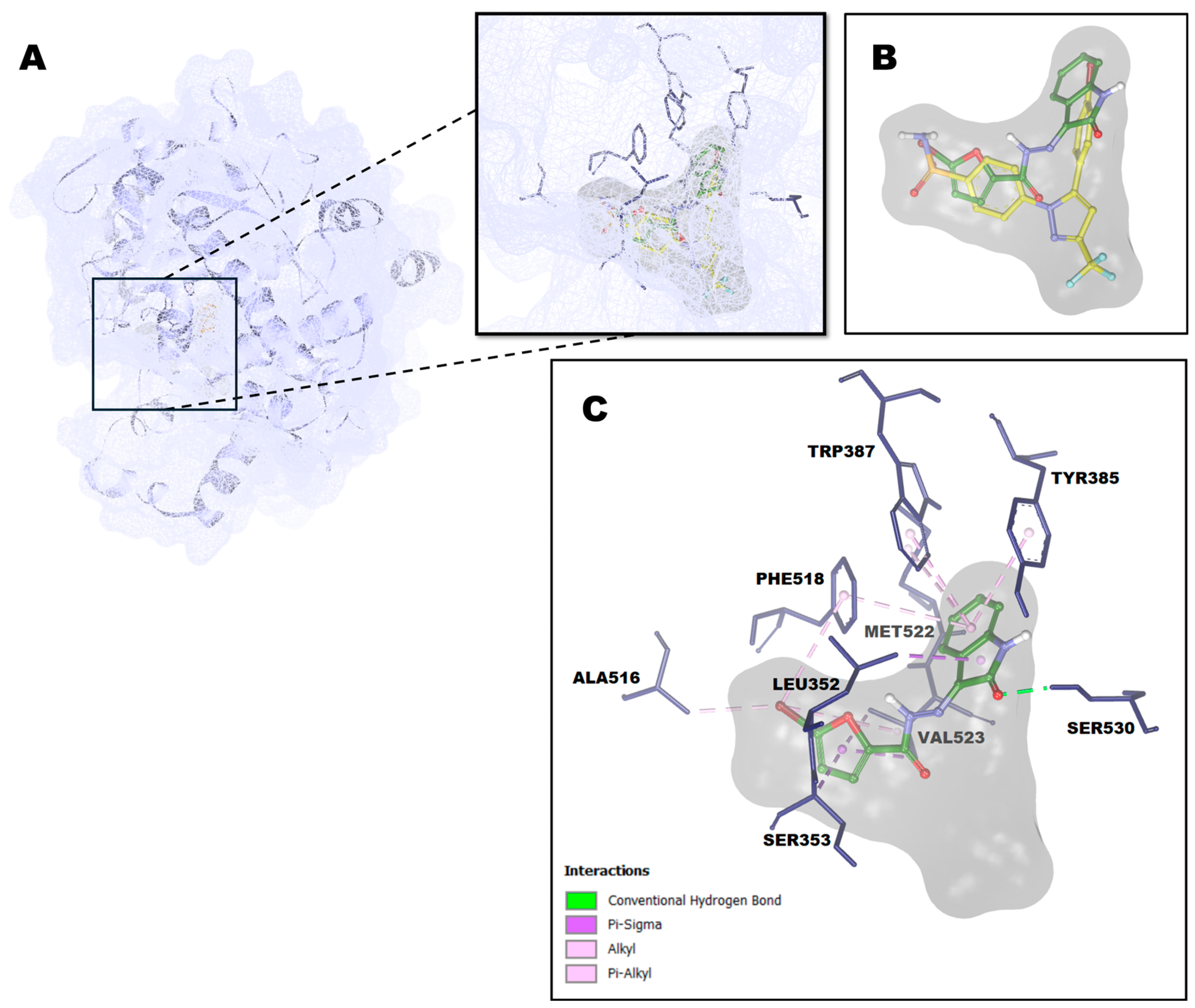
2.4. Molecular Docking Study
3. Materials and Methods
3.1. Materials
3.2. Synthesis of 5-bromo-N’-(2-oxoindolin-3-ylidene)furan-2-carbohydrazide (1)
3.3. In vitro Anti-inflammatory Activity Assay
3.3.1. Bovine Serum Albumin Denaturation Assay
3.3.2. Proteinase Activity Inhibition Assay
3.3.3. Hemolysis Inhibition Assay
Preparation of Human Red Blood Cells (HRBCs) Suspension
Heat-Induced Hemolysis Inhibition
Hypotonicity-Induced Hemolysis Inhibition
3.4. Drug-Likeness, Molecular and ADMET Prediction
3.5. Molecular Docking Study
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L. Inflammatory Responses and Inflammation-Associated Diseases in Organs. Oncotarget 2017, 9, 7204–7218. [Google Scholar] [CrossRef] [PubMed]
- Hasan, Md.M.; Islam, Md.E.; Hossain, Md.S.; Akter, M.; Rahman, Md.A.A.; Kazi, M.; Khan, S.; Parvin, M.S. Unveiling the Therapeutic Potential: Evaluation of Anti-Inflammatory and Antineoplastic Activity of Magnolia champaca Linn’s Stem Bark Isolate through Molecular Docking Insights. Heliyon 2024, 10, e22972. [Google Scholar] [CrossRef] [PubMed]
- Rane, M.A.; Foster, J.G.; Wood, S.K.; Hebert, P.R.; Hennekens, C.H. Benefits and Risks of Nonsteroidal Anti-Inflammatory Drugs: Methodologic Limitations Lead to Clinical Uncertainties. Drug. Inf. J. 2019, 53, 502–505. [Google Scholar] [CrossRef] [PubMed]
- Jarapula, R.; Gangarapu, K.; Manda, S.; Rekulapally, S. Synthesis, In Vivo Anti-Inflammatory Activity, and Molecular Docking Studies of New Isatin Derivatives. Int. J. Med. Chem. 2016, 2016, 2181027. [Google Scholar] [CrossRef]
- Hassanzadeh, F.; Jafari, E.; Khayambashi, N.; Hajhashemi, V. Synthesis and Anti-Inflammatory Effects Evaluation of 1,3 Substituted Isatin Derivatives:(TJPS-2020-0290.R1). TJPS 2021, 45, 248–252. [Google Scholar] [CrossRef]
- Cheke, R.S.; Patil, V.M.; Firke, S.D.; Ambhore, J.P.; Ansari, I.A.; Patel, H.M.; Shinde, S.D.; Pasupuleti, V.R.; Hassan, M.I.; Adnan, M.; et al. Therapeutic Outcomes of Isatin and Its Derivatives against Multiple Diseases: Recent Developments in Drug Discovery. Pharmaceuticals (Basel) 2022, 15, 272. [Google Scholar] [CrossRef]
- Alizadeh, M.; Jalal, M.; Hamed, K.; Saber, A.; Kheirouri, S.; Pourteymour Fard Tabrizi, F.; Kamari, N. Recent Updates on Anti-Inflammatory and Antimicrobial Effects of Furan Natural Derivatives. J. Inflamm. Res. 2020, 13, 451–463. [Google Scholar] [CrossRef]
- Manolov, S.; Ivanov, I.; Bojilov, D.; Nedialkov, P. Synthesis, In Silico, and In Vitro Biological Evaluation of New Furan Hybrid Molecules. Processes 2022, 10, 1997. [Google Scholar] [CrossRef]
- Yang, L.; He, J. Traditional Uses, Phytochemistry, Pharmacology and Toxicological Aspects of the Genus Hosta (Liliaceae): A Comprehensive Review. J. Ethnopharmacol. 2021, 265, 113323. [Google Scholar] [CrossRef]
- Banerjee, R.; Kumar, H.K.S.; Banerjee, M. Medicinal Significance of Furan Derivatives: A Review. Int. J. Rev. Life. Sci. 2012, 2, 7–16. [Google Scholar]
- Derbel, H.; Elleuch, J.; Mahfoudh, W.; Michaud, P.; Fendri, I.; Abdelkafi, S. In Vitro Antioxidant and Anti-Inflammatory Activities of Bioactive Proteins and Peptides from Rhodomonas sp. Appl. Sci. 2023, 13, 3202. [Google Scholar] [CrossRef]
- Karjalainen, M.J.; Neuvonen, P.J.; Backman, J.T. In Vitro Inhibition of CYP1A2 by Model Inhibitors, Anti-Inflammatory Analgesics, and Female Sex Steroids: Predictability of In Vivo Interactions. BCPT 2008, 103, 157–165. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Ruiz, J.C.; Matus-Basto, A.J.; Acereto-Escoffié, P.; Segura-Campos, M.R. Antioxidant and Anti-Inflammatory Activities of Phenolic Compounds Isolated from Melipona beecheii Honey. Food Agr. Immunol. 2017, 28, 1424–1437. [Google Scholar] [CrossRef]
- Moualek, I.; Iratni Aiche, G.; Mestar Guechaoui, N.; Lahcene, S.; Houali, K. Antioxidant and Anti-Inflammatory Activities of Arbutus unedo Aqueous Extract. Asian Pac. J. Trop. Biomed. 2016, 6, 937–944. [Google Scholar] [CrossRef]
- Aidoo, D.B.; Konja, D.; Henneh, I.T.; Ekor, M. Protective Effect of Bergapten against Human Erythrocyte Hemolysis and Protein Denaturation In Vitro. Int. J. Inflam. 2021, 2021, 1279359. [Google Scholar] [CrossRef]
- Anwar, M.M.; Shalaby, M.; Embaby, A.M.; Saeed, H.; Agwa, M.M.; Hussein, A. Prodigiosin/PU-H71 as A Novel Potential Combined Therapy for Triple Negative Breast Cancer (TNBC): Preclinical Insights. Sci. Rep. 2020, 10, 14706. [Google Scholar] [CrossRef]
- Daina, A.; Michielin, O.; Zoete, V. SwissADME: A Free Web Tool to Evaluate Pharmacokinetics, Drug-Likeness and Medicinal Chemistry Friendliness of Small Molecules. Sci. Rep. 2017, 7, 42717. [Google Scholar] [CrossRef]
- Tsume, Y.; Langguth, P.; Garcia-Arieta, A.; Amidon, G.L. In Silico Prediction of Drug Dissolution and Absorption with Variation in Intestinal pH for BCS Class II Weak Acid Drugs: Ibuprofen and Ketoprofen. Biopharm. Drug Disp. 2012, 33, 366–377. [Google Scholar] [CrossRef]
- Hacker, K.; Maas, R.; Kornhuber, J.; Fromm, M.; Zolk, O. Substrate-Dependent Inhibition of the Human Organic Cation Transporter OCT2: A Comparison of Metformin with Experimental Substrates. PloS One 2015, 10, e0136451. [Google Scholar] [CrossRef]
- Domínguez-Villa, F.X.; Durán-Iturbide, N.A.; Ávila-Zárraga, J.G. Synthesis, Molecular Docking, and In Silico ADME/Tox Profiling Studies of New 1-aryl-5-(3-azidopropyl)indol-4-ones: Potential Inhibitors of SARS CoV-2 Main Protease. Bioorg. Chem. 2021, 106, 104497. [Google Scholar] [CrossRef]
- Aijijiyah, N.P.; Wati, F.A.; Rahayu, R.; Srilistiani, A.; Mahzumi, F.; Aulia, T.; Santoso, L.; Pamela, E.; Ramadhani, E.Y.; Ilfahmi, Y.A.; et al. Synthesis, α-Glucosidase Inhibitory Activity, and Molecular Docking of Cinnamamides. Med. Chem. Res. 2023, 32, 723–735. [Google Scholar] [CrossRef]
- Ejeh, S.; Uzairu, A.; Shallangwa, G.A.; Abechi, S.E. Computational Insight to Design New Potential Hepatitis C Virus NS5B Polymerase Inhibitors with Drug-Likeness and Pharmacokinetic ADMET Parameters Predictions. Futur. J. Pharm. Sci. 2021, 7, 219. [Google Scholar] [CrossRef]
- Kurumbail, R.G.; Stevens, A.M.; Gierse, J.K.; McDonald, J.J.; Stegeman, R.A.; Pak, J.Y.; Gildehaus, D.; Iyashiro, J.M.; Penning, T.D.; Seibert, K.; et al. Structural Basis for Selective Inhibition of Cyclooxygenase-2 by Anti-Inflammatory Agents. Nature 1996, 384, 644–648. [Google Scholar] [CrossRef] [PubMed]
- Elisha, I.L.; Dzoyem, J.-P.; McGaw, L.J.; Botha, F.S.; Eloff, J.N. The Anti-Arthritic, Anti-Inflammatory, Antioxidant Activity and Relationships with Total Phenolics and Total Flavonoids of Nine South African Plants Used Traditionally to Treat Arthritis. BMC Complement. Altern. Med. 2016, 16, 307. [Google Scholar] [CrossRef]
- Gunathilake, K.D.P.P.; Ranaweera, K.K.D.S.; Rupasinghe, H.P.V. In Vitro Anti-Inflammatory Properties of Selected Green Leafy Vegetables. Biomedicines 2018, 6, 107. [Google Scholar] [CrossRef]
| Sample | IC50 value (µg/mL) | |||
|---|---|---|---|---|
| BSA Denaturation | Proteinase Activity | Heat-Induced Hemolysis | Hypotonicity-Induced Hemolysis |
|
| Compound 1 | 3.54 | 3.04 | 2.25 | 1.82 |
| Diclofenac sodium | 1.99 | 3.38 | 2.28 | 2.00 |
| Property | Predicted Value |
| Molecular weight | 332.97 g/mol |
| Number of HBA | 4 |
| Number of HBD | 2 |
| MolLogP | 2.44 |
| MolLogS | -3.07 (in Log(moles/L)) 285.52 (in mg/L) |
| MolPSA | 68.29 A2 |
| pKa | 0.61/8.59 (most basic/acidic group) |
| Property | Model name |
Predicted value |
Unit |
| Absorption | Water solubility | -3.562 | Numeric (log mol/L) |
| Absorption | Caco-2 permeability | 0.901 | Numeric (log Papp in 10-6 cm/s) |
| Absorption | Human intestinal absorption | 91.993 | Numeric (% absorbed) |
| Absorption | P-glycoprotein substrate | No | Categorical (Yes/No) |
| Distribution | VDss (human) | -0.375 | Numeric (log L/kg) |
| Metabolism | CYP1A2 inhibitor | No | Categorical (Yes/No) |
| Metabolism | CYP2C19 inhibitor | No | Categorical (Yes/No) |
| Metabolism | CYP2C9 inhibitor | No | Categorical (Yes/No) |
| Metabolism | CYP2D6 inhibitor | No | Categorical (Yes/No) |
| Metabolism | CYP34A inhibitor | No | Categorical (Yes/No) |
| Excretion | Total clearance | -0.286 | Numeric (log mL/min/kg) |
| Excretion | Renal OCT2 substrate | No | Categorical (Yes/No) |
| Toxicity | AMES toxicity | No | Categorical (Yes/No) |
| Toxicity | MRTDa (human) | -0.2 | Numeric (log mg/kg/day) |
| Toxicity | hERG I/II inhibitor | No | Categorical (Yes/No) |
| Toxicity | Oral rat acute toxicity (LD50) | 2.197 | Numeric (mol/kg) |
| Toxicity | Hepatotoxicity | No | Categorical (Yes/No) |
| Toxicity | Minnow toxicity | 1.463 | Numeric (log mM) |
| Property | Model name |
Predicted value |
Unit |
| Absorption | Water solubility | -3.562 | Numeric (log mol/L) |
| Absorption | Caco-2 permeability | 0.901 | Numeric (log Papp in 10-6 cm/s) |
| Absorption | Human intestinal absorption | 91.993 | Numeric (% absorbed) |
| Absorption | P-glycoprotein substrate | No | Categorical (Yes/No) |
| Distribution | VDss (human) | -0.375 | Numeric (log L/kg) |
| Metabolism | CYP1A2 inhibitor | No | Categorical (Yes/No) |
| Metabolism | CYP2C19 inhibitor | No | Categorical (Yes/No) |
| Metabolism | CYP2C9 inhibitor | No | Categorical (Yes/No) |
| Metabolism | CYP2D6 inhibitor | No | Categorical (Yes/No) |
| Metabolism | CYP34A inhibitor | No | Categorical (Yes/No) |
| Excretion | Total clearance | -0.286 | Numeric (log mL/min/kg) |
| Excretion | Renal OCT2 substrate | No | Categorical (Yes/No) |
| Toxicity | AMES toxicity | No | Categorical (Yes/No) |
| Toxicity | MRTDa (human) | -0.2 | Numeric (log mg/kg/day) |
| Toxicity | hERG I/II inhibitor | No | Categorical (Yes/No) |
| Toxicity | Oral rat acute toxicity (LD50) | 2.197 | Numeric (mol/kg) |
| Toxicity | Hepatotoxicity | No | Categorical (Yes/No) |
| Toxicity | Minnow toxicity | 1.463 | Numeric (log mM) |
| Compound | Binding Energy (kcal/mol) |
Cyclooxygenase Active Site Residues |
Interaction Unit of Compound | Interaction Type |
| 1 | -9.63 | Leu352 | benzene ring of isatin skeleton | π-σ hydrophobic |
| Tyr385 | benzene ring of isatin skeleton | π-alkyl hydrophobic | ||
| Trp387 | benzene ring of isatin skeleton | π-alkyl hydrophobic | ||
| Phe518 | benzene ring of isatin skeleton | π-alkyl hydrophobic | ||
| bromo | π-alkyl hydrophobic | |||
| Met552 | pyrrole ring of isatin skeleton | alkyl hydrophobic | ||
| Ser530 | C=O of isatin skeleton | H-bond | ||
| Val523 | furanyl | π-σ hydrophobic | ||
| bromo | alkyl hydrophobic | |||
| Ser353 | furanyl | π-σ hydrophobic | ||
| Ala516 | bromo | alkyl hydrophobic | ||
| SC-558 | -10.96 (0.89 Å) | Leu352 | N-H of sulphonamide | H-bond |
| Gln192 | N-H of sulphonamide | H-bond | ||
| Phe518 | S atom of sulphonamide | π-sulfur | ||
| His90 | C=O of sulphonamide | H-bond | ||
| Val523 | phenyl of phenylsulphonamide | π-alkyl hydrophobic | ||
| phenyl of bromophenyl | π-alkyl hydrophobic | |||
| Leu384 | bromo | alkyl hydrophobic | ||
| Tyr385 | bromo | π-alkyl hydrophobic | ||
| Trp387 | bromo | π-alkyl hydrophobic | ||
| Gly526 | phenyl of bromophenyl | Amide-π stacked hydrophobic | ||
| Ala527 | phenyl of bromophenyl | π-alkyl hydrophobic | ||
| pyrazole | π-alkyl hydrophobic | |||
| Val349 | pyrazole | π-alkyl hydrophobic | ||
| carbon atom of CF3 | alkyl hydrophobic | |||
| Leu359 | carbon atom of CF3 | alkyl hydrophobic | ||
| Leu531 | carbon atom of CF3 | alkyl hydrophobic | ||
| Arg120 | fluorine atom of CF3 | H-bond | ||
| pyrazole | π-cation charge attraction |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




