Submitted:
30 October 2024
Posted:
31 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Treatment Preparation
2.2. Subjects and PBMC Isolation
2.3. Cytotoxic Activity
2.4. Cell Culture
2.5. MTT Assay
2.4. Cellular Reactive Oxygen Species Production
H2DCFDA (2’,7’-Dichlorofluorescin Diacetate) Assay
2.5. The Alkaline Comet Assay
2.6. Statistical Analysis
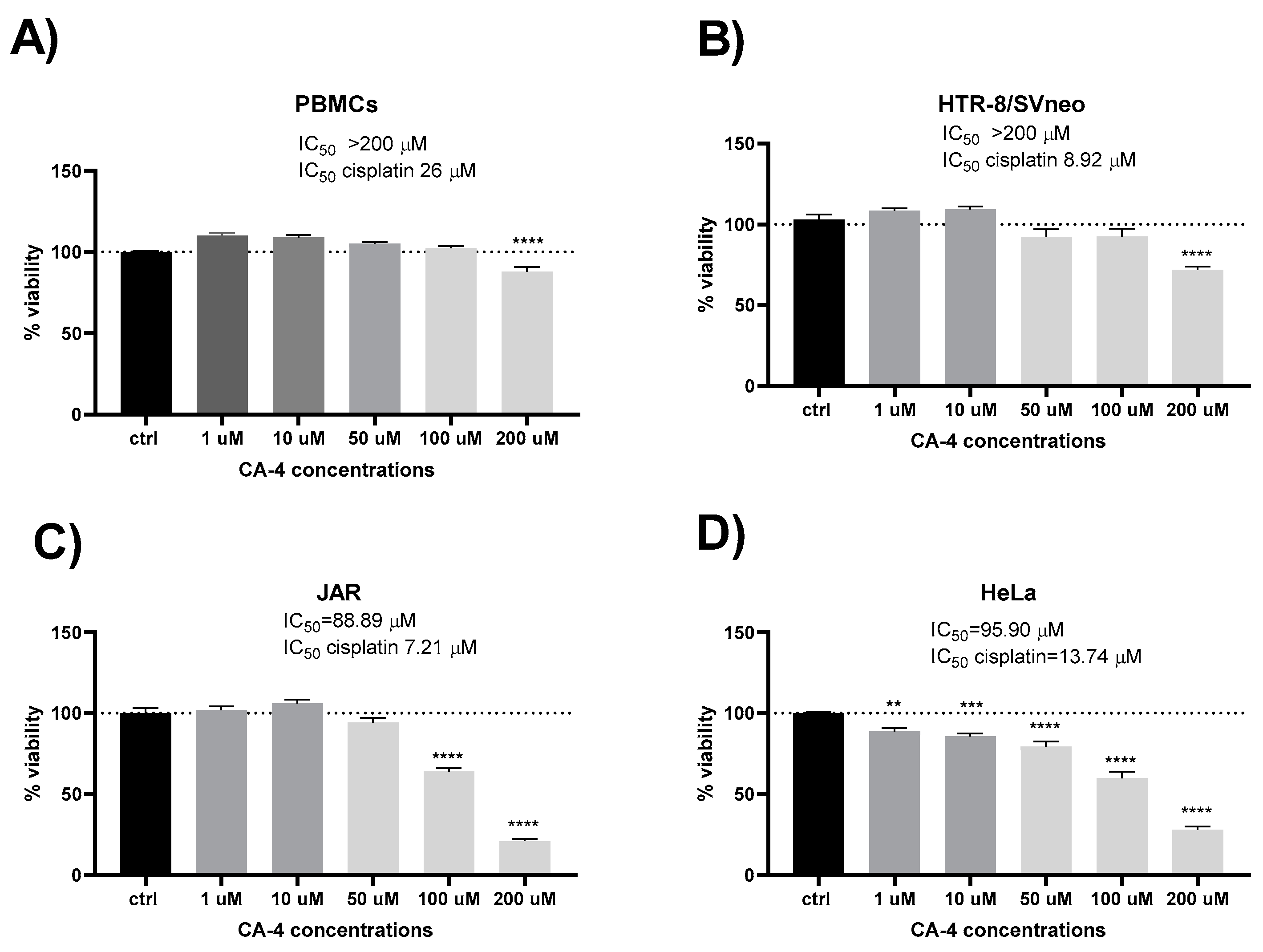
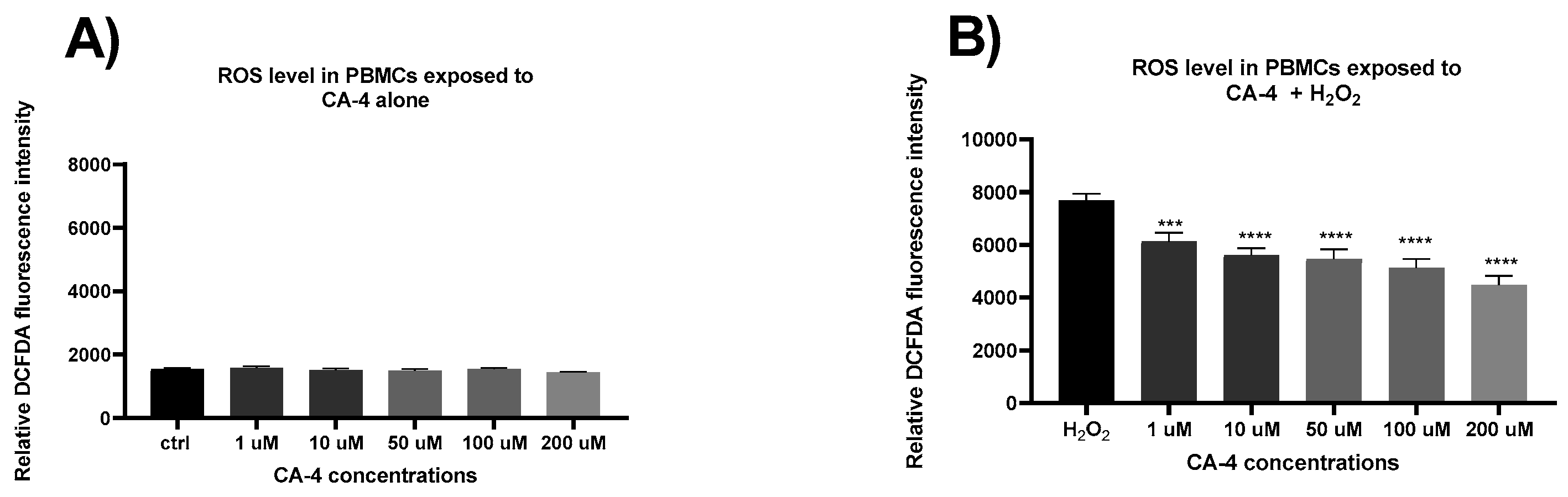
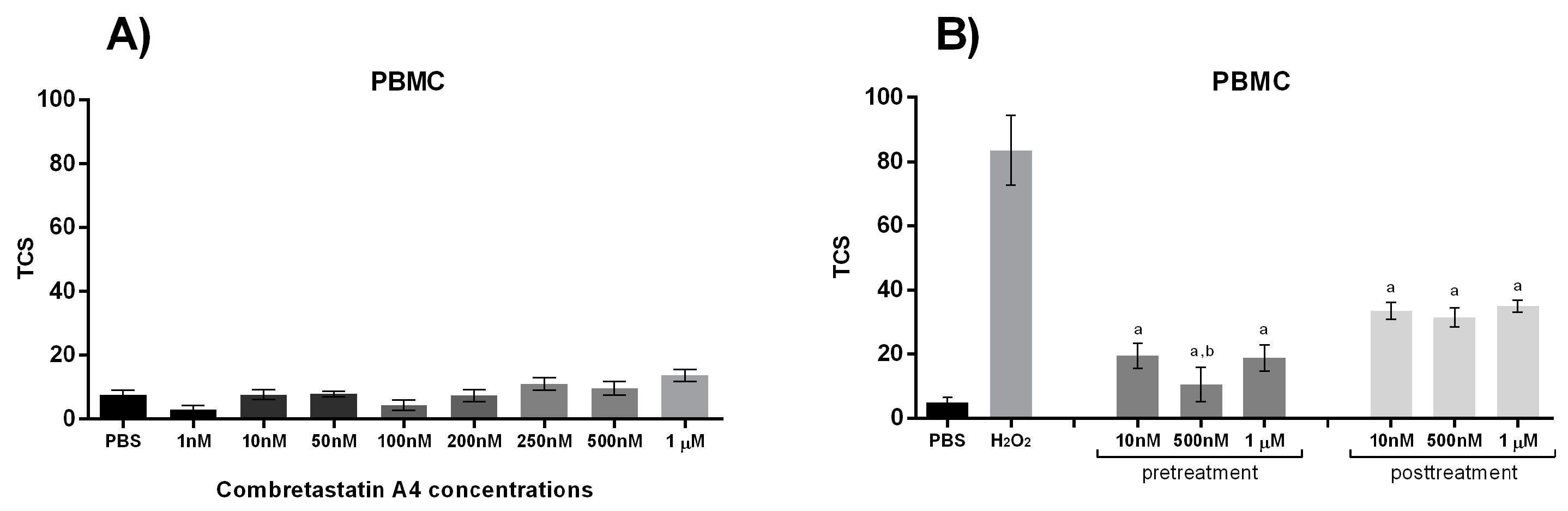
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef] [PubMed]
- Pettit, G.R.; Singh, S.B.; Hamel, E.; Lin, C.M.; Alberts, D.S.; Garcia-Kendal, D. Isolation and Structure of the Strong Cell Growth and Tubulin Inhibitor Combretastatin A-4. Experientia 1989, 45, 209–211. [Google Scholar] [CrossRef] [PubMed]
- Nam, N.-H. Combretastatin A-4 Analogues as Antimitotic Antitumor Agents. Curr. Med. Chem. 2003, 10, 1697–1722. [Google Scholar] [CrossRef] [PubMed]
- Karatoprak, G.Ş.; Küpeli Akkol, E.; Genç, Y.; Bardakcı, H.; Yücel, Ç.; Sobarzo-Sánchez, E. Combretastatins: An Overview of Structure, Probable Mechanisms of Action and Potential Applications. Molecules 2020, 25, 2560. [Google Scholar] [CrossRef] [PubMed]
- Dark, G.G.; Hill, S.A.; Prise, V.E.; Tozer, G.M.; Pettit, G.R.; Chaplin, D.J. Combretastatin A-4, an Agent That Displays Potent and Selective Toxicity toward Tumor Vasculature. Cancer Res. 1997, 57, 1829–1834. [Google Scholar]
- Guo, K.; Ma, X.; Li, J.; Zhang, C.; Wu, L. Recent Advances in Combretastatin A-4 Codrugs for Cancer Therapy. Eur. J. Med. Chem. 2022, 241, 114660. [Google Scholar] [CrossRef]
- Dowlati, A.; Robertson, K.; Cooney, M.; Petros, W.P.; Stratford, M.; Jesberger, J.; Rafie, N.; Overmoyer, B.; Makkar, V.; Stambler, B.; et al. A Phase I Pharmacokinetic and Translational Study of the Novel Vascular Targeting Agent Combretastatin A-4 Phosphate on a Single-Dose Intravenous Schedule in Patients with Advanced Cancer. Cancer Res. 2002, 62, 3408–3416. [Google Scholar]
- Grisham, R.; Ky, B.; Tewari, K.S.; Chaplin, D.J.; Walker, J. Clinical Trial Experience with CA4P Anticancer Therapy: Focus on Efficacy, Cardiovascular Adverse Events, and Hypertension Management. Gynecol. Oncol. Res. Pract. 2018, 5, 1. [Google Scholar] [CrossRef]
- Tozer, G.M.; Prise, V.E.; Wilson, J.; Cemazar, M.; Shan, S.; Dewhirst, M.W.; Barber, P.R.; Vojnovic, B.; Chaplin, D.J. Mechanisms Associated with Tumor Vascular Shut-down Induced by Combretastatin A-4 Phosphate: Intravital Microscopy and Measurement of Vascular Permeability. Cancer Res. 2001, 61, 6413–6422. [Google Scholar]
- McGown, A.T.; Fox, B.W. Structural and Biochemical Comparison of the Anti-Mitotic Agents Colchicine, Combretastatin A4 and Amphethinile. Anticancer. Drug Des. 1989, 3, 249–254. [Google Scholar]
- Griggs, J.; Metcalfe, J.C.; Hesketh, R. Targeting Tumour Vasculature: The Development of Combretastatin A4. Lancet Oncol. 2001, 2, 82–87. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, Y.F. Combretastatin A4-Based Coumarins: Synthesis, Anticancer, Oxidative Stress-Relieving, Anti-Inflammatory, Biosafety, and in Silico Analysis. Chem. Pap. 2024, 78, 3705–3720. [Google Scholar] [CrossRef]
- Kumar, B.; Sharma, P.; Gupta, V.P.; Khullar, M.; Singh, S.; Dogra, N.; Kumar, V. Synthesis and Biological Evaluation of Pyrimidine Bridged Combretastatin Derivatives as Potential Anticancer Agents and Mechanistic Studies. Bioorg. Chem. 2018, 78, 130–140. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, V.; Hay, N. Molecular Pathways: Reactive Oxygen Species Homeostasis in Cancer Cells and Implications for Cancer Therapy. Clin. Cancer Res. 2013, 19, 4309–4314. [Google Scholar] [CrossRef] [PubMed]
- Weinberg, S.E.; Chandel, N.S. Targeting Mitochondria Metabolism for Cancer Therapy. Nat. Chem. Biol. 2015, 11, 9–15. [Google Scholar] [CrossRef]
- Aziz, G.; Odlo, K.; Hansen, T.V.; Paulsen, R.E.; Mathisen, G.H. Combretastatin A-4 and Structurally Related Triazole Analogues Induce Caspase-3 and Reactive Oxygen Species-Dependent Cell Death in PC12 Cells. Eur. J. Pharmacol. 2013, 703, 25–32. [Google Scholar] [CrossRef]
- Billard, C.; Menasria, F.; Quiney, C.; Faussat, A.-M.; Finet, J.-P.; Combes, S.; Kolb, J.-P. 4-Arylcoumarin Analogues of Combretastatins Stimulate Apoptosis of Leukemic Cells from Chronic Lymphocytic Leukemia Patients. Exp. Hematol. 2008, 36, 1625–1633. [Google Scholar] [CrossRef]
- ZHANG, C.; ZHOU, S.-S.; LI, X.-R.; WANG, B.-M.; LIN, N.-M.; FENG, L.-Y.; ZHANG, D.-Y.; ZHANG, L.-H.; WANG, J.-B.; PAN, J.-P. Enhanced Antitumor Activity by the Combination of Dasatinib and Combretastatin A-4 in Vitro and in Vivo. Oncol. Rep. 2013, 29, 2275–2282. [Google Scholar] [CrossRef]
- Kuzminov, A. Single-Strand Interruptions in Replicating Chromosomes Cause Double-Strand Breaks. Proc. Natl. Acad. Sci. USA 2001, 98, 8241–8246. [Google Scholar] [CrossRef]
- Moretton, A.; Loizou, J.I. Interplay between Cellular Metabolism and the DNA Damage Response in Cancer. Cancers 2020, 12, 2051. [Google Scholar] [CrossRef]
- Mittal, D.; Gubin, M.M.; Schreiber, R.D.; Smyth, M.J. New Insights into Cancer Immunoediting and Its Three Component Phases—Elimination, Equilibrium and Escape. Curr. Opin. Immunol. 2014, 27, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, R.D.; Old, L.J.; Smyth, M.J. Cancer Immunoediting: Integrating Immunity’s Roles in Cancer Suppression and Promotion. Science 2011, 331, 1565–1570. [Google Scholar] [CrossRef] [PubMed]
- Collins, A.R.; Dobson, V.L.; Dušinská, M.; Kennedy, G.; Štětina, R. The Comet Assay: What Can It Really Tell Us? Mutat. Res. Mol. Mech. Mutagen. 1997, 375, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Collins, A.R. The Comet Assay for DNA Damage and Repair: Principles, Applications, and Limitations. Mol. Biotechnol. 2004, 26, 249–261. [Google Scholar] [CrossRef]
- Duan, Y.-T.; Man, R.-J.; Tang, D.-J.; Yao, Y.-F.; Tao, X.-X.; Yu, C.; Liang, X.-Y.; Makawana, J.A.; Zou, M.-J.; Wang, Z.-C.; et al. Design, Synthesis and Antitumor Activity of Novel Link-Bridge and B-Ring Modified Combretastatin A-4 (CA-4) Analogues as Potent Antitubulin Agents. Sci. Rep. 2016, 6, 25387. [Google Scholar] [CrossRef]
- Jadala, C.; Sathish, M.; Anchi, P.; Tokala, R.; Lakshmi, U.J.; Reddy, V.G.; Shankaraiah, N.; Godugu, C.; Kamal, A. Synthesis of Combretastatin-A4 Carboxamidest That Mimic Sulfonyl Piperazines by a Molecular Hybridization Approach: In Vitro Cytotoxicity Evaluation and Inhibition of Tubulin Polymerization. ChemMedChem 2019, 14, 2052–2060. [Google Scholar] [CrossRef]
- do Amaral, D.N.; Cavalcanti, B.C.; Bezerra, D.P.; Ferreira, P.M.P.; Castro, R.d.P.; Sabino, J.R.; Machado, C.M.L.; Chammas, R.; Pessoa, C.; Sant’Anna, C.M.R.; et al. Docking, Synthesis and Antiproliferative Activity of N-Acylhydrazone Derivatives Designed as Combretastatin A4 Analogues. PLoS ONE 2014, 9, e85380. [Google Scholar] [CrossRef]
- Kwak, Y.-S.; Joo, S.-H.; Gansukh, E.; Mistry, B.M.; Keum, Y.S. Synthesis and Anticancer Activities of Polymethylenedioxy Analogues of Combretastatin A-2. Appl. Biol. Chem. 2019, 62, 25. [Google Scholar] [CrossRef]
- Song, M.-Y.; He, Q.-R.; Wang, Y.-L.; Wang, H.-R.; Jiang, T.-C.; Tang, J.-J.; Gao, J.-M. Exploring Diverse-Ring Analogues on Combretastatin A4 (CA-4) Olefin as Microtubule-Targeting Agents. Int. J. Mol. Sci. 2020, 21, 1817. [Google Scholar] [CrossRef]
- Sadhu, D.N.; Desai, L.; Young, S.; Randall, J.C. Lack of Clastogenicity of Combretastatin A-4 (CA4) - Microtubule Destabilizing Agent, in Human Peripheral Blood Lymphocytes in Vitro. Cancer Res. 2005, 65, 809. [Google Scholar]
- Carvalho, P.C.; Santos, E.A.; Schneider, B.U.C.; Matuo, R.; Pesarini, J.R.; Cunha-Laura, A.L.; Monreal, A.C.D.; Lima, D.P.; Antoniolli, A.C.M.B.; Oliveira, R.J. Diaryl Sulfide Analogs of Combretastatin A-4: Toxicogenetic, Immunomodulatory and Apoptotic Evaluations and Prospects for Use as a New Chemotherapeutic Drug. Environ. Toxicol. Pharmacol. 2015, 40, 715–721. [Google Scholar] [CrossRef] [PubMed]
- Petit, I.; Karajannis, M.A.; Vincent, L.; Young, L.; Butler, J.; Hooper, A.T.; Shido, K.; Steller, H.; Chaplin, D.J.; Feldman, E.; et al. The Microtubule-Targeting Agent CA4P Regresses Leukemic Xenografts by Disrupting Interaction with Vascular Cells and Mitochondrial-Dependent Cell Death. Blood 2008, 111, 1951–1961. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



