Submitted:
30 October 2024
Posted:
31 October 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Results
Study Population and Setting
Statistical Analysis
Adverse Effects
Methods
Study Design and Ethical Approval
Study Population and Setting
Treatment
Follow-Up
Statistical Analyses
Author Contributions
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maldonado, V. V., et al. Clinical utility of mesenchymal stem/stromal cells in regenerative medicine and cellular therapy. J. Biol. Eng. 17, 44 (2023).
- Clavellina, D., Balkan, W. & Hare, J. M. Stem cell therapy for acute myocardial infarction: mesenchymal stem cells and induced pluripotent stem cells. Exp. Opin. Biol. Ther. 23, 951-967 (2023).
- Saadh, M. J., et al. Therapeutic potential of mesenchymal stem/stromal cells (MSCs)-based cell therapy for inflammatory bowel diseases (IBD) therapy. Eur. J. Med. Res. 28, 47 (2023).
- Montoto-Meijide, R., Meijide-Faílde, R., Díaz-Prado, S. M., Montoto-Marqués, A. Mesenchymal stem cell therapy in traumatic spinal cord injury: a systematic review. Int. J. Mol. Sci. 24, 11719 (2023).
- Mikłosz, A. & Chabowski, A. Adipose-derived mesenchymal stem cells therapy as a new treatment option for diabetes mellitus. J. Clin. Endocrinol. Metab. 108, 1889-1897 (2023).
- Zhu, Y., Ge, J., Huang, C., Liu, H. & Jiang, H. Application of mesenchymal stem cell therapy for aging frailty: from mechanisms to therapeutics. Theranostics 11, 5675-5685 (2021).
- Schulman, I. H., Balkan, W. & Hare, J. M. Mesenchymal stem cell therapy for aging frailty. Front. Nutr. 5, 108 (2018).
- Fernández-Garza, L. E., Barrera-Barrera, S. A., & Barrera-Saldaña, H. A. Mesenchymal stem cell therapies approved by regulatory agencies around the world. Pharmaceuticals 16, 1334 (2023).
- Liu, H., et al. The association of triglyceride-glucose index with major adverse cardiovascular and cerebrovascular events after acute myocardial infarction: a meta-analysis of cohort studies. Nutr. Diabetes, 14, 39 (2024).
- Wang, W. T., et al. Comparative effectiveness of blood pressure-lowering drugs in patients who have already suffered from stroke: traditional and Bayesian network meta-analysis of randomized trials. Medicine 95, e3302 (2016).
- Evans, C. E., Iruela-Arispe, M. L., & Zhao, Y. Y. Mechanisms of endothelial regeneration and vascular repair and their application to regenerative medicine. Am. J. Pathol. 191, 52-65 (2021).
- Franceschi, C., et al. Inflamm-aging. An evolutionary perspective on immunosenescence. Ann. N. Y. Acad. Sci. 908, 244-254 (2000).
- Tompkins, B. A., et al. Allogeneic mesenchymal stem cells ameliorate aging frailty: a phase ii randomized, double-blind, placebo-controlled clinical trial. J. Gerontol. A Biol. Sci. Med. Sci. 72, 1513-1522 (2017).
| Variables | Subcategory | Number of patients (%) |
|---|---|---|
| Sex | Female | 1276 (50.96%) |
| Male | 1225 (48.92%) | |
| Unknown | 3 (0.12%) | |
| Age (years) | Mean ± SD | 54.09 ± 11.65 |
| Median (Min, Max) | 54 (21, 104) | |
| MSC IV Dose (in billions) | Mean ± SD | 1.49 ± 0.75 |
| Median (Min, Max) | 2 (0.1, 2) | |
| Indications for MSC therapy | Allergy | 23 (0.92%) |
| ALS | 4 (0.16%) | |
| AMI | 60 (2.40%) | |
| AMI/Brain stroke | 1 (0.04%) | |
| AMI/DM | 7 (0.28%) | |
| AMI/DM/HT/HL | 1 (0.04%) | |
| Anti-aging | 1700 (67.89%) | |
| Arrythmia | 8 (0.32%) | |
| Arteriosclerosis | 1 (0.04%) | |
| Asthma | 16 (0.64%) | |
| Atherosclerosis | 51 (2.04%) | |
| Brain stroke | 35 (1.40%) | |
| Brain stroke/renal failure | 1 (0.04%) | |
| Brain stroke/AMI | 1 (0.04%) | |
| Brain stroke/DM | 1 (0.04%) | |
| Cancer | 10 (0.40%) | |
| Cancer/DM | 1 (0.04%) | |
| Cardiomyopathy | 2 (0.08%) | |
| CNS disorder | 1 (0.04%) | |
| Chronic obstructive pulmonary disease | 2 (0.08%) | |
| Chronic obstructive pulmonary disease/AMI | 1 (0.04%) | |
| Depression | 5 (0.20%) | |
| DM | 168 (6.71%) | |
| DM/arrythmia/hepatitis | 1 (0.04%) | |
| DM/HT | 1 (0.04%) | |
| DM/Renal failure | 1 (0.04%) | |
| Emphysema | 1 (0.04%) | |
| Heart valve disorder | 3 (0.12%) | |
| Hepatitis | 93 (3.71%) | |
| HL | 116 (4.63%) | |
| HT | 43 (1.72%) | |
| HT/DM | 1 (0.04%) | |
| HT/Hepatitis | 1 (0.04%) | |
| HT/HL | 2 (0.08%) | |
| Insomnia | 51 (2.04%) | |
| Infertility | 2 (0.08%) | |
| Leukaemia | 1 (0.04%) | |
| Long COVID | 1 (0.04%) | |
| Mental disorder | 2 (0.08%) | |
| Pancreatitis | 1 (0.04%) | |
| Parkinson’s disease | 6 (0.24%) | |
| Post cancer | 17 (0.68%) | |
| Post cancer surgery | 1 (0.04%) | |
| Post pancreatitis | 1 (0.04%) | |
| Renal failure | 4 (0.16%) | |
| System lupus erythematosus | 4 (0.16%) | |
| Type-1 diabetes | 1 (0.04%) | |
| Thyroid disease | 45 (1.80%) | |
| Tuberculosis | 4 (0.16%) | |
| Side effects | Disrupted menstrual cycles | 4 (0.16%) |
| Dizziness | 2 (0.08%) | |
| Fatigue | 1 (0.04%) | |
| Hives | 1 (0.04%) | |
| Insomnia | 10 (0.4%) | |
| Itch for 1week | 1 (0.04%) | |
| Psoriasis flare-up | 1 (0.04%) | |
| None | 2484 (99.2%) | |
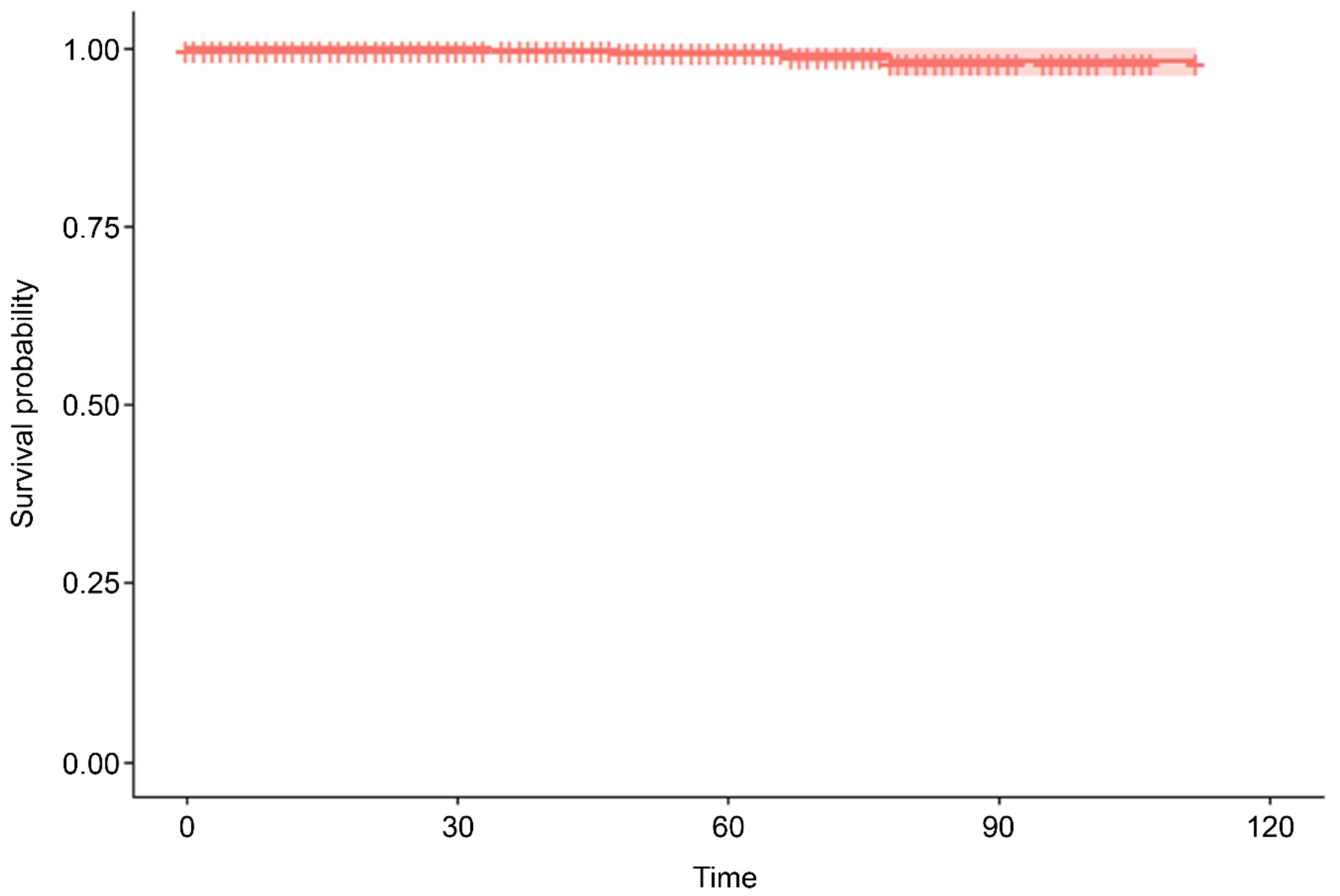
| MACCE-free duration (months) | Mean ± SD | 15.88 ± 26.85 |
| Median (Min, Max) | 0 (0, 112) |
| n | Events | Mean | SE (Mean) |
|---|---|---|---|
| 2504 | 5 | 111 | 0.435 |
| Characteristic | HR | 95% CI | p-value |
|---|---|---|---|
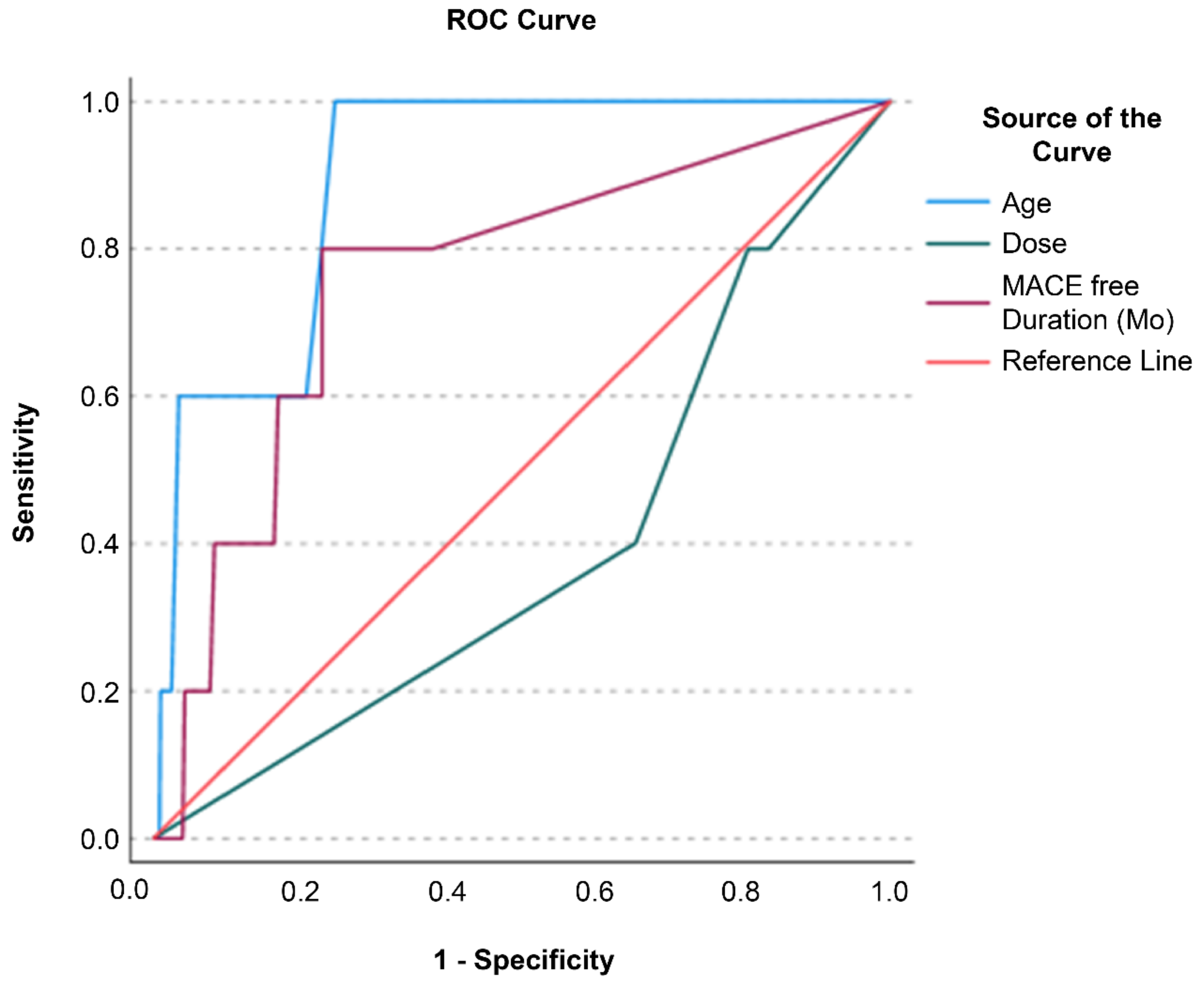
| Age (years) | 1.124 | 1.04, 1.214 | 0.0031* |
| Sex (Ref: Female) | |||
| Male | 1.528 | 0.2552, 9.153 | 0.642 |
| MSC IV dose (in billions) | 0.667 | 0.203, 2.191 | 0.504 |
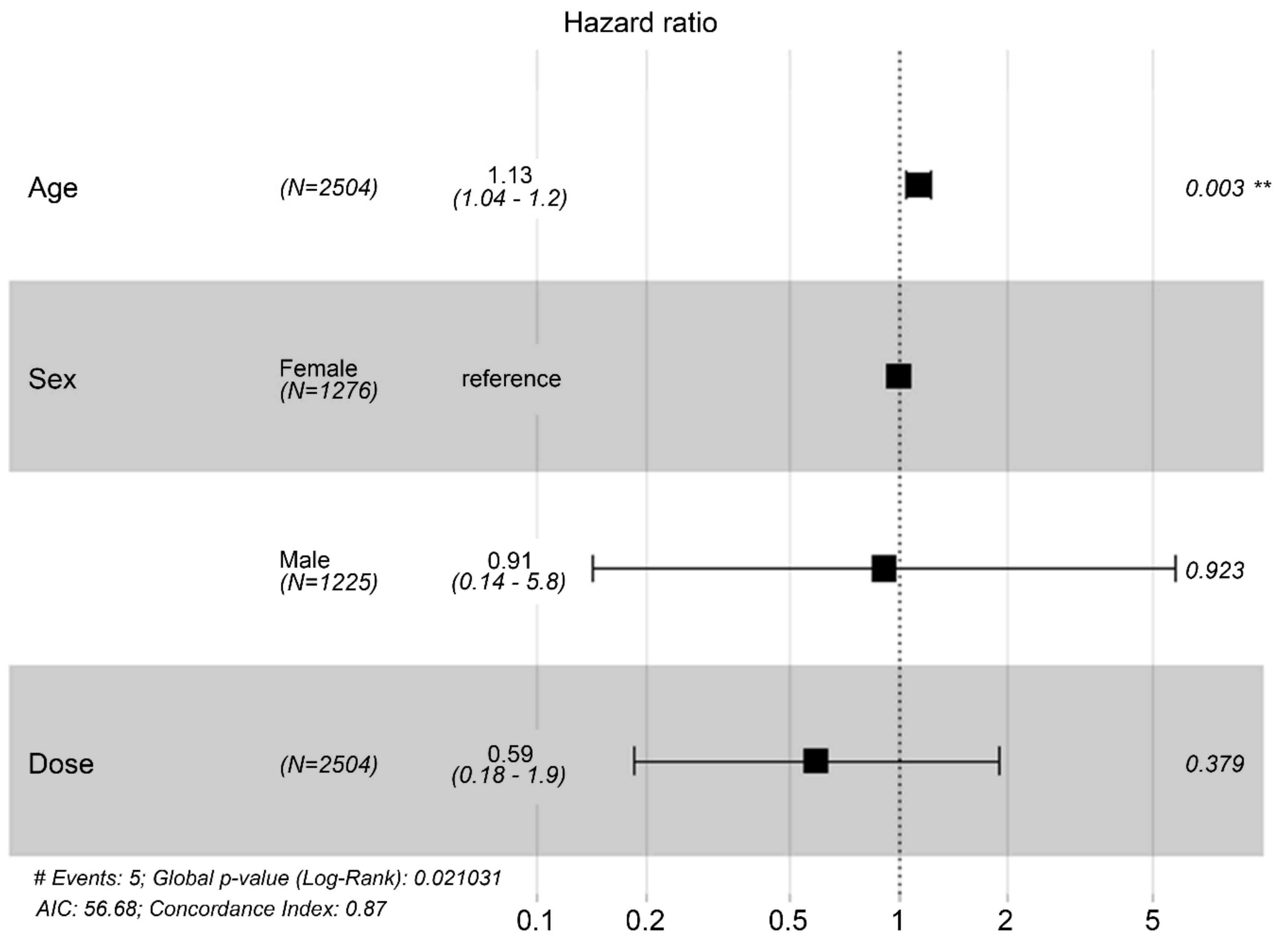
| Characteristic | HR | 95% CI | p-value |
|---|---|---|---|
| Age (years) | 1.127 | 1.0418, 1.219 | 0.0029* |
| Sex (Ref: Female) | |||
| Male | 0.9129 | 0.1429, 5.831 | 0.9232 |
| MSC IV dose (in billions) | 0.5928 | 0.1849, 1.900 | 0.379 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




