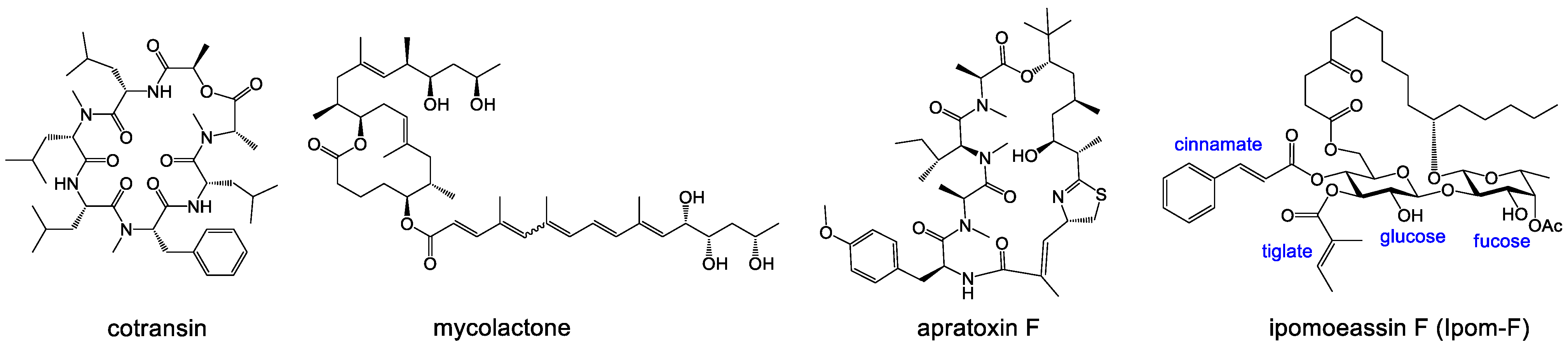
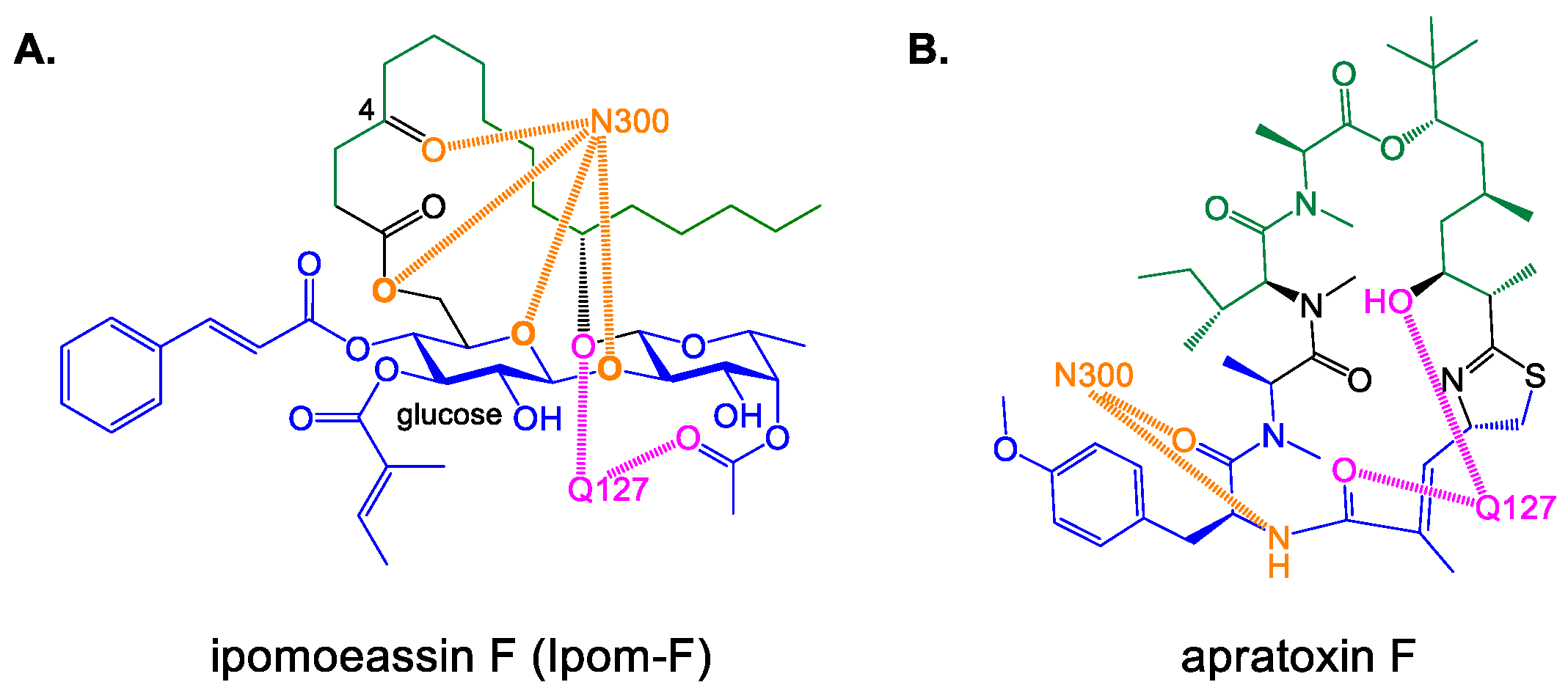
3.1. Chemical Synthesis General Methods
Unless otherwise stated, all commercially obtained reagents were used without further purification and all reactions were conducted under argon/nitrogen atmosphere. Reaction progress was monitored by TLC using silica gel F254 glass back plates with detection under UV lamp (254 nm) or charring with 5 % (v/v) H2SO4 (sulfuric acid) in EtOH (ethanol). Column chromatographic purifications were performed using silica gel (70 – 230 mesh) with a ratio that spanned from 100 to 50: 1 (w/w) between the silica gel and crude products. All 1H NMR spectra were obtained in deuterated chloroform (CDCl3), using chloroform (CHCl3, δ = 7.26) or tetramethylsilane (TMS, δ = 0) as an internal reference. All 13C NMR spectra were proton decoupled and obtained in CDCl3 with CHCl3 (δ = 77.16) as internal references. NMR data are reported in the form: chemical shifts (δ) in ppm, multiplicity, coupling constants (J) in Hz, and integrations. 1H data are reported as though they were first order. Other 1D and 2D NMR spectra like 135DEPT, COSY, HMQC, and HMBC were collected in addition to 1H and 13C for new compounds. High resolution mass spectrometry (HRMS) data were acquired by the Mass Spectrometry Lab at the University of Illinois Urbana-Champaign. Purity was analyzed using a Shimadzu HPLC with a dual wavelength UV detector set at 254 nm and 280 nm, a RESTEK Ultra reverse phase column (C18, 5 μm, 4.6x200 mm) and an isocratic mobile phase of acetonitrile in water.
3.1.1. Synthesis of Compound 14
The fucoside diol acceptor
13 (1.10 g, 2.54 mmol), glucoside trichloroacetimidate donor
12 (1.57 g, 2.80 mmol, 1.1 equiv), and crushed activated 4Å molecular sieves (2.8 g) were suspended in 40 ml anhydrous CH
2Cl
2. The mixture was stirred under an argon atmosphere for ~30 min at room temperature and then cooled to −52 °C to −58 °C. To the cold reaction mixture was added TMSOTf (41 μl, 0.229 mmol, 0.09 equiv) via a syringe. The reaction was left to stir for 18 mins at this temperature and then quenched by addition of a few drops of TEA. The mixture was filtered through a pad of celite, and the filtrate was then concentrated. The resulting residue was purified by column chromatography (10:1→2:1 hexanes–EtOAc) to afford the product
14 as a yellow oil sticky oil (1.82 g, 86%). Its NMR data match with the literature-reported data [
26].
3.1.2. Synthesis of Compound 15
To an ice-cold solution of 14 (1.82 g, 2.19 mmol), TEA (2.44 ml, 17.52 mmol, 8 equiv), and DMAP (26.8 mg, 0.219 mmol, 0.1 equiv) in DCM (14 ml) was added Ac2O (1.76 ml, 18.61 mmol, 8.5 equiv) dropwise and the reaction was warmed to room temperature overnight. At this point, TLC (Hex: EtOAc 3:1) showed the reaction was complete. Methanol was added to quench the reaction. DCM was added and the mixture was washed with 1N HCl (2x) followed by saturated NaHCO3 wash. The aqueous layers were extracted with DCM twice and the combined organic layers were dried over Na2SO4. Rotary evaporation of the solvents yielded the crude product as a pale-yellow syrup (1.82 g 95%), which was moved to the next step without further purification.
To an ice-cold solution of the syrup obtained above (350.0 mg, 0.40 mmol) in DCM (5 ml) was added a buffer solution of hydrazine monohydrate (110 mg, 2.20 mmol, 5.5 equiv) in acetic acid (1.47 ml, 25.6 mmol, 64 equiv) and pyridine (2.20 ml, 68 equiv). The mixture was stirred at 0 °C for 30 minutes and then at room temperature for 1 hour. At this point, TLC showed the reaction was complete. The mixture was washed with 1M HCl (3x), followed by brine wash. All the aqueous layers were extracted twice by DCM and the combined organic layers were dried over Na2SO4. After solvent evaporation, the residue was purified by column chromatography (2:1, hexanes–EtOAc) to afford 15 as a white foam (265.0 mg, 98%): 1H NMR (400 MHz, CDCl3, δH) 4.98 (d, J = 3.3 Hz, 1H), 4.54 (d, J = 7.7 Hz, 1H), 4.29 (d, J = 7.4 Hz, 1H), 3.65–3.88 (m, 4H), 3.44–3.62 (m, 5H), 3.31 (t, J = 8.4 Hz, 1H), 3.20 (td, J = 9.9, 5.2 Hz, 1H), 3.01 (br s, 1H), 2.04 (s, 3H), 1.32–1.56 (m, 10H), 1.12–1.31 (m, 12H),1.05 (d, J = 6.4 Hz, 3H), 0.74–0.87 (m, 15H), 0.09 (s, 3H), 0.07 (s, 3H); 13C NMR (100 MHz, CDCl3, δC) 170.7 (C=O), 104.2 (O2C), 101.1 (O2CH), 99.7 (O2CH), 79.8 (OCH), 79.0 (OCH), 76.5 (OCH), 73.3 (OCH), 72.9 (OCH), 72.6 (OCH), 72.5 (OCH), 68.6 (OCH), 68.2 (OCH), 62.2 (OCH2), 34.0 (CH2), 33.4 (CH2), 32.1 (CH2), 31.9 (CH2), 29.0 (CH3), 25.8 (C(CH3)3), 24.8 (CH2), 24.2 (CH2), 22.5(9) (CH2), 22.5(7) (CH2), 20.8 (CH3), 19.0 (CH3), 17.8 (SiC(CH3)3), 16.4 (CH3), 14.1 (2xCH3), -4.5 (SiCH3), -4.7 (SiCH3).
3.1.3. Synthesis of Compound 16
To an ice-cold solution of 15 (230.0 mg, 0.334 mmol), DMAP (8.3 mg, 0.2 equiv) and tiglic acid (40.8 mg, 1.2 equiv) in DCM (5 ml) was added DCC (140.0 mg, 2.0 equiv). The reaction mixture was stirred overnight while warming up to room temperature. TLC showed the reaction was complete. Hexanes (5 mL) and ether (10 ml) were added, and the mixture was stirred for 20 more minutes. The resulting suspension was then filtered through a pad of celite. After solvent evaporation, the residue was purified by column chromatography (30:1→20:1, hexanes–EtOAc) to afford 16 as a white foam (217.0 mg, 84%): 1H NMR (400 MHz, CDCl3, δH) 6.80 (q, J = 7.1 Hz, 1H), 5.09 (t, J = 9.5 Hz, 1H), 5.00 (d, J = 3.3 Hz, 1H), 4.66 (d, J = 7.5 Hz, 1H), 4.30 (d, J = 7.5 Hz, 1H), 3.78–3.94 (m, 2H), 3.68–3.77 (m, 2H), 3.50–3.67 (m, 3H), 3.40–3.49 (m, 2H), 3.30 (td, J = 9.9, 5.1 Hz, 1H), 2.04 (s, 3H), 1.72–1.80 (m, 6H), 1.12–1.59 (m, 22H), 1.07 (d, J = 6.3 Hz, 3H), 0.74–0.91 (d, J = 11.2 Hz, 15H), 0.11 (s, 3H), 0.08 (s, 3H); 13C NMR (100 MHz, CDCl3, δC) 170.8 (C=O), 167.5 (C=O), 137.3 (=CH), 128.4 (=C), 104.4 (O2C), 101.3 (O2CH), 99.6 (O2CH), 79.8 (OCH), 78.8 (OCH), 75.3 (OCH), 72.9 (OCH), 72.8 (OCH), 72.7 (OCH), 71.7 (OCH), 68.6 (OCH), 68.3 (OCH), 62.3 (OCH2), 34.0 (CH2), 33.4 (CH2), 32.2 (CH2), 31.9 (CH2), 28.9 (CH3), 25.8 (C(CH3)3), 25.0 (CH2), 24.8 (CH2), 24.2 (CH2), 22.6(5) (CH2), 22.6(1) (CH2), 20.9 (CH3), 18.9 (CH3), 17.8 (SiC(CH3)3), 16.4 (CH3), 14.3 (CH3), 14.1 (CH3), 12.1 (CH3), -4.5(2) (SiCH3), -4.5(8) (SiCH3).
3.1.4. Synthesis of Compound 17
To a solution of 16 (217.0 mg, 0.286 mmol) and 2,6 lutidine (153.0 mg, 5 equiv) in DCM (3.0 ml) at room temperature was added TESOTf (151.0 mg, 2.0 equiv). The mixture was stirred for one hour. At this point, TLC showed the reaction was complete. The mixture was washed with 1M HCl (2x), followed by brine wash. All the aqueous layers were extracted by DCM twice and the combined organic layers were dried over Na2SO4. After the solvents were removed by rotary evaporation, the residue was purified by column chromatography (30:1→20:1, hexanes–EtOAc) to afford 17 as a yellow sticky oil (200.1 mg, 80%): H NMR (400 MHz, CDCl3, δH) 6.79 (qd, J = 7.0, 1.6 Hz, 1H), 5.00 (t, J = 9.3 Hz, 1H), 4.88–4.96 (m, 2H), 4.20 (d, J = 7.7 Hz, 1H), 3.87–4.01 (m, 2H), 3.81 (dd, J = 9.2, 3.6 Hz, 1H), 3.64 (t, J = 10.5 Hz, 1H), 3.57 (q, J = 6.4 Hz, 1H), 3.41–3.52 (m, 3H), 3.20 (td, J = 9.9, 5.2 Hz, 1H), 2.06 (s, 3H), 1.72–1.82 (m, 6H), 1.14–1.66 (m, 22H), 1.07 (d, J = 6.4 Hz, 3H), 0.78–0.96 (m, 24H), 0.40–0.63 (m, 6H), 0.11 (s, 3H), 0.05 (s, 3H); 13C NMR (100 MHz, CDCl3, δC) 170.9 (C=O), 166.8 (C=O), 137.0 (=CH), 128.7 (=C), 101.6 (O2CH), 101.1 (O2C), 99.3 (O2CH), 82.0 (OCH), 74.9 (OCH), 74.5 (OCH), 74.4 (OCH), 74.0 (OCH), 73.4 (OCH), 72.5 (OCH), 68.8 (OCH), 67.2 (OCH), 62.6 (OCH2), 34.6 (CH2), 34.2 (CH2), 32.4 (CH2), 32.0 (CH2), 29.0 (CH3), 25.9 (C(CH3)3), 25.2 (CH2), 24.6 (CH2), 23.1 (CH2), 22.7 (CH2), 21.0 (CH3), 18.8 (CH3), 17.7 (SiC(CH3)3), 16.8 (CH3), 14.4 (CH3), 14.3 (CH3), 14.1 (CH3), 12.1 (CH3), 6.9 (3xSiCH2CH3), 5.0 (3xSiCH2CH3), -4.1 (SiCH3), -4.3 (SiCH3).
3.1.5. Synthesis of Compound 18
To an ice-cold solution of 17 (168.0 mg, 0.229 mmol) in CHCl3 (3.00 ml) was added TFA (130 mg, 5 equiv). The mixture was stirred at 0 °C for two hours. At this point, TLC showed the reaction was complete. Water was added. The mixture was then extracted with DCM and washed with NaHCO3. All the aqueous layers were extracted with DCM twice and the combined organic layers were dried over Na2SO4. After the solvents were removed by rotary evaporation, the residue was purified by column chromatography (6:1→4:1, hexanes–EtOAc) to afford 18 as a white foam (168.0 mg, 88%): 1H NMR (400 MHz, CDCl3, δH) 6.88 (q, J = 7.0 Hz, 1H), 4.85–4.99 (m, 3H), 4.21 (dd, J = 7.8, 1.9 Hz, 1H), 3.94 (td, J = 8.3, 1.9 Hz, 1H), 3.78–3.90 (m, 2H), 3.71 (dd, J = 12.2, 4.3 Hz, 1H), 3.41–3.63 (m, 4H), 3.25–3.35 (m, 1H), 2.93 (d, J = 5.4 Hz, 1H), 2.60 (s, 1H), 2.09 (s, 3H), 1.77–1.85 (m, 6H), 1.38–1.53 (m, 4H), 1.17–1.38 (m, 12H), 1.08 (d, J = 6.4 Hz, 3H), 0.76–0.95 (m, 24H), 0.45–0.63 (m, 6H), 0.11 (s, 3H), 0.07 (s, 3H); 13C NMR (100 MHz, CDCl3, δC) 171.1 (C=O), 168.8 (C=O), 138.4 (=CH), 128.3 (=C), 101.6 (O2CH), 100.5 (O2CH), 81.9 (OCH), 79.0 (OCH), 75.3 (OCH), 74.5 (OCH), 74.0 (OCH), 73.7 (OCH), 73.4 (OCH), 70.7 (OCH), 68.9 (OCH), 62.3 (OCH2), 34.6 (CH2), 34.2 (CH2), 32.4 (CH2), 32.0 (CH2), 25.9 (C(CH3)3), 25.2 (CH2), 24.7 (CH2), 22.9 (CH2), 22.7 (CH2), 21.1 (CH3), 17.7 (SiC(CH3)3), 16.8 (CH3), 14.5 (CH3), 14.3 (CH3), 14.1 (CH3), 12.1 (CH3), 6.9 (3xSiCH2CH3), 5.1 (3xSiCH2CH3), -4.1 (SiCH3), -4.3 (SiCH3).
3.1.6. Synthesis of Compound 19
To a solution of diol 18 (168.0 mg, 0.201 mmol), TEA (40.8 mg, 2.0 equiv) and DMAP (2.5 mg, 0.1 equiv) at room temperature was added trityl chloride (78.7 mg, 1.40 equiv). After the reaction mixture was stirred at room temperature overnight, TLC showed the reaction was complete. After solvent evaporation, the residue was purified by column chromatography (40:1→20:1, hexanes–EtOAc) to afford 19 as a white foam (214.0 mg, 99%). The successful installation of the trityl group was confirmed by a quick NMR check. No full NMR characterization was conducted.
3.1.7. Synthesis of Compound 20
To an ice-cold solution of 19 (214.0 mg, 0.199 mmol), DMAP (4.9 mg, 0.2 equiv) and cinnamic acid (41 mg, 1.40 equiv) in DCM (4 ml) was added DCC (164.0 mg, 4.0 equiv). The mixture was stirred while warming up to room temperature overnight. At this point, TLC showed the reaction was complete. Hexanes (5 mL) and ether (10 ml) was added. The reaction mixture was stirred for another 20 minutes, and then filtered through a pad of celite. After the solvents were removed by rotary evaporation, the residue was purified by column chromatography (50:1→30:1, hexanes–EtOAc) to afford 20 as a white foam (189 mg, 79%). The presence of the cinnamate group was confirmed by a quick NMR check, but full NMR characterization was not conducted.
3.1.8. Synthesis of Compound 21
To an ice-cold solution of 20 (189 mg, 0.156 mmol) in DCM (2.00 ml) was added TFA (89.0 mg, 5 equiv). The mixture was stirred at 0 °C for 30 minutes. At this point, TLC showed the reaction was complete. The mixture was washed with saturated NaHCO 3 , followed by brine wash. All the aqueous layers were extracted twice with DCM and the combined organic layers were dried over Na2SO4. After solvents were evaporated, the residue was purified by column chromatography (30:1→5:1, hexanes–EtOAc) to afford 21 as a white foam (107 mg, 71%): 1H NMR (400 MHz, CDCl3, δH) 7.60 (d, J = 16.0 Hz, 1H), 7.42–7.52 (m, 2H), 7.29–7.40 (m, 3H), 6.79 (qd, J = 7.0, 1.6 Hz, 1H), 6.31 (d, J = 16.0Hz, 1H), 5.26 (t, J = 9.5 Hz, 1H), 4.96–5.09 (m, 3H), 4.26 (d, J = 7.6 Hz, 1H), 3.99 (dd, J = 9.1, 7.6 Hz, 1H), 3.87 (dd, J = 9.1, 3.7 Hz, 1H), 3.67–3.77 (m, 1H), 3.49–3.66 (m, 5H), 2.12 (s, 3H), 1.68–1.77 (m, 6H), 1.19–1.64 (m, 16H), 1.13 (d, J = 6.4 Hz, 3H), 0.80–1.00 (m, 24H), 0.46–0.66 (m, 6H), 0.18 (s, 3H), 0.11 (s, 3H); 13C NMR (100 MHz, CDCl3, δC) 171.0 (C=O), 167.0 (C=O), 166.0 (C=O), 146.2 (=CH), 138.2 (=CH), 134.2 (=C), 130.6 (=CH), 129.0 (2x=CH), 128.3 (2x=CH), 128.2 (=C), 116.9 (=CH), 101.4 (O2CH), 100.8 (O2CH), 81.8 (OCH), 75.0 (OCH), 74.5 (OCH), 74.4 (OCH), 74.0(8) (OCH), 74.0(4) (OCH), 73.5 (OCH), 69.8 (OCH), 68.9 (OCH), 61.9 (OCH2), 34.6 (CH2), 34.1 (CH2), 32.4 (CH2), 32.1 (CH2), 26.0 (C(CH3)3), 25.2 (CH2), 24.8 (CH2), 23.0 (CH2), 22.7 (CH2), 21.1 (CH3), 17.8 (SiC(CH3)3), 16.8 (CH3), 14.5 (CH3), 14.3 (CH3), 14.2 (CH3), 12.1 (CH3), 6.9 (3xSiCH2CH3), 5.0 (3xSiCH2CH3), -4.0 (SiCH3), -4.2 (SiCH3).
3.1.9. General Procedure for Steglich Esterification with EDC
At 0 °C, EDC (5 equiv) was added in one portion to a CH2Cl2 (4 mL) solution of alcohol (1 equiv), acid (~1.2 eq) and DMAP (4-dimethylaminopyridine, 2 equiv). The reaction was stirred overnight while warming to ambient temperature. At this point, TLC (silica, EtOAc–hexanes) showed the reaction was complete. The reaction mixture was quenched with a few drops of methanol and washed sequentially with 1M HCl and saturated aqueous NaHCO3. The aqueous layers were back extracted with CH2Cl2. The combined organic layer was dried over Na2SO4 and concentrated under reduced pressure. The residue was purified by column chromatography (silica, EtOAc–hexanes) to give desired compounds.
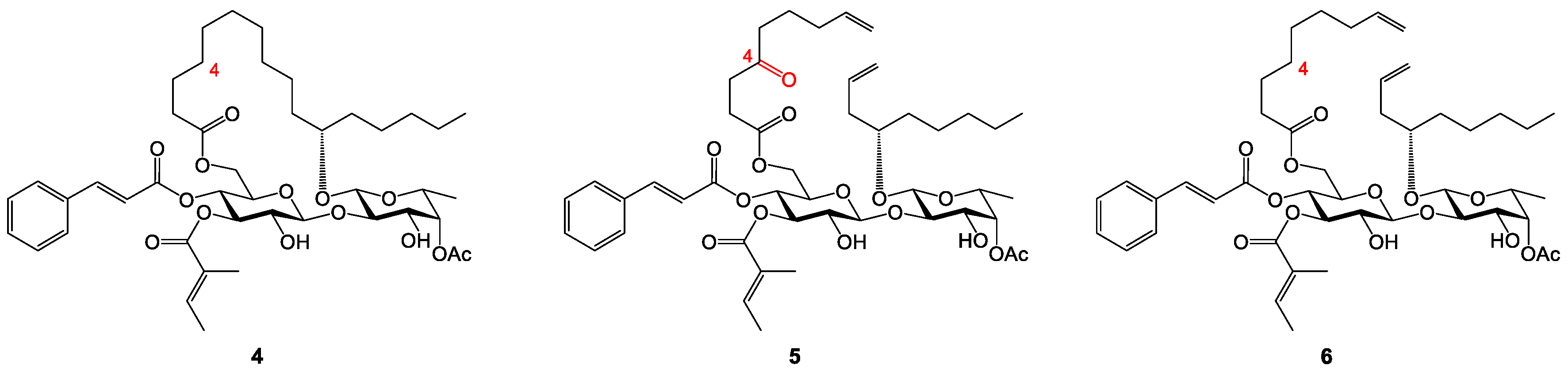
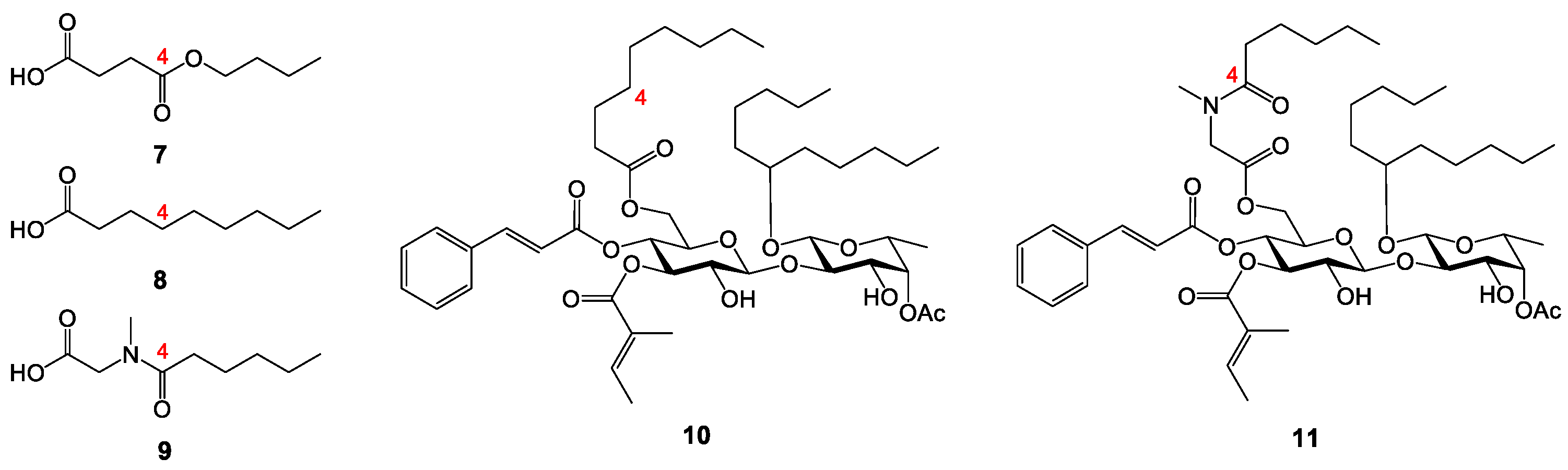
Analogue 22: Alcohol 21 (39.6 mg, 0.0411 mmol) was reacted with nonanoic acid 8 (8.0 mg, 0.0506 mmol) to make ester 22. The crude product was purified by column chromatography (100:1→15:1, hexanes–EtOAc) to afford 22 as a colorless syrup (45.4 mg, contaminated by 8 that could not be separated): Rf 0.70 (6:1 hexanes–EtOAc); 1H NMR (400 MHz, CDCl3, δH) 7.59 (d, J = 16.0 Hz, 1H), 7.43–7.52 (m, 2H), 7.32–7.40 (m, 3H), 6.79 (br q, J = 6.0 Hz, 1H), 6.31 (d, J = 16.0 Hz, 1H), 5.23 (t, J = 8.9 Hz, 1H), 5.09 (t, J = 9.8 Hz, 1H), 4.96–5.03 (m, 2H), 4.31 (d, J = 7.7 Hz, 1H), 4.17 (d, J = 4.0 Hz, 2H), 4.04 (d, J = 8.4 Hz, 1H), 3.89 (dd, J = 9.2, 3.6 Hz, 1H), 3.50–3.70 (m, 4H), 2.33 (t, J = 7.4 Hz, 2H), 2.11 (s, 3H), 1.68–1.78 (m, 6H), 1.17–1.66 (m, 28H), 1.13 (d, J = 6.4 Hz, 3H), 0.80–1.01 (m, 27H), 0.46–0.66 (m, 6H), 0.18 (s, 3H), 0.12 (s, 3H); 13C NMR (100 MHz, CDCl3, δC) 173.5 (C=O), 171.0 (C=O), 167.0 (C=O), 165.6 (C=O), 146.0 (=CH), 138.1 (=CH), 134.2 (=C), 130.6 (=CH), 128.9 (2x=CH), 128.3 (2x=CH), 128.2 (=C), 117.0 (=CH), 101.3 (O2CH), 100.6 (O2CH), 81.7 (OCH), 75.3 (OCH), 74.5 (OCH), 74.3 (OCH), 74.1 (OCH), 73.6 (OCH), 71.8 (OCH), 69.6 (OCH), 68.8 (OCH), 62.9(OCH2), 34.7 (CH2), 34.2 (CH2), 34.1 (CH2), 32.5 (CH2), 32.1 (CH2), 31.9 (CH2), 26.0 (C(CH3)3), 29.3 (CH2), 29.2 (CH2), 29.1 (CH2), 25.3 (CH2), 24.8(1) (CH2), 24.7(7) (CH2), 23.0 (CH2), 22.7(5) (CH2), 22.7(2) (CH2), 21.1 (CH3), 17.9 (SiC(CH3)3), 16.8 (CH3), 14.5 (CH3), 14.4 (CH3), 14.2 (2xCH3), 12.1 (CH3), 6.9 (3xSiCH2CH3), 5.0 (3xSiCH2CH3), -4.1 (SiCH3), -4.2 (SiCH3).
Analogue 23: Alcohol 21 (39.1 mg, 0.0406 mmol) was reacted with acid 9 (10.6 mg, 0.0566 mmol) to make ester 23. The crude product was purified by column chromatography (50:1→10:1, hexanes–EtOAc) to afford 23 as a colorless syrup (46.0 mg, quantitative). The ratio of the two rotamers (~3:1) was estimated from the 13C NMR spectrum. 1H NMR and 13C NMR are reported for the major rotamer: Rf 0.36 (3:1 hexanes–EtOAc); 1H NMR (400 MHz, CDCl3, δH) 7.57 (d, J = 16.0 Hz, 1H), 7.42–7.51 (m, 2H), 7.31–7.39 (m, 3H), 6.77 (q, J = 7.0 Hz, 1H), 6.29 (d, J = 16.0 Hz, 1H), 4.95–5.30 (m, 4H), 4.22–4.43 (m, 3H), 4.07–4.19 (m, 1H), 3.82–4.06 (m, 3H), 3.45–3.72 (m, 4H), 3.04 (s, 3H), 2.33 (t, J = 7.5 Hz, 2H), 2.10 (s, 3H), 1.66–1.76 (m, 6H), 1.17–1.66 (m, 22H), 1.11 (d, J = 6.4 Hz, 3H), 0.80–1.01 (m, 27H), 0.45–0.64 (m, 6H), 0.17 (s, 3H), 0.10 (s, 3H); 13C NMR (100 MHz, CDCl3, δC) 174.0 (C=O), 170.9 (C=O), 169.1 (C=O), 166.9 (C=O), 165.7 (C=O), 146.2 (=CH), 138.2 (=CH), 134.2 (=C), 130.6 (=CH), 129.0 (2x=CH), 128.4 (2x=CH), 128.2 (=C), 116.9 (=CH), 101.4 (O2CH), 100.6 (O2CH), 81.8 (OCH), 75.1 (OCH), 74.5 (OCH), 74.4 (OCH), 74.0 (OCH), 73.6 (OCH), 71.7 (OCH), 69.6 (OCH), 68.8 (OCH), 61.5(OCH2), 48.9 (NCH2), 36.5 (CH3), 34.7 (CH2), 34.2 (CH2), 33.2 (CH2), 32.5 (CH2), 32.1 (CH2), 31.6 (CH2), 26.0 (C(CH3)3), 25.2 (CH2), 24.8 (CH2), 24.6 (CH2), 23.0 (CH2), 22.8 (CH2), 22.6 (CH2), 21.1 (CH3), 17.9 (SiC(CH3)3), 16.8 (CH3), 14.5 (CH3), 14.4 (CH3), 14.2 (CH3), 14.1 (CH3), 12.1 (CH3), 6.9 (3xSiCH2CH3), 6.9 (3xSiCH2CH3), -4.0 (SiCH3), -4.2 (SiCH3).
3.1.10. General Procedure for TES and TBS Removal Using TBAF/AcOH
To a solution of TES and TBS protected compounds (1 equiv) in THF (2 mL) was added AcOH (37 equiv) and TBAF (1M solution in THF, 22 equiv) at room temperature. The reaction was then stirred at room temperature for 8 h. At this point, TLC (silica, EtOAc–hexanes) showed the reaction was complete. The reaction mixture was diluted with CH2Cl2 and washed sequentially with 1M HCl, saturated aqueous NaHCO3 and brine. The aqueous layers were back extracted with CH2Cl2. The combined organic layer was dried over Na2SO4 and concentrated under reduced pressure. The residue was purified by column chromatography (silica, EtOAc–hexanes) to give desired analogues.
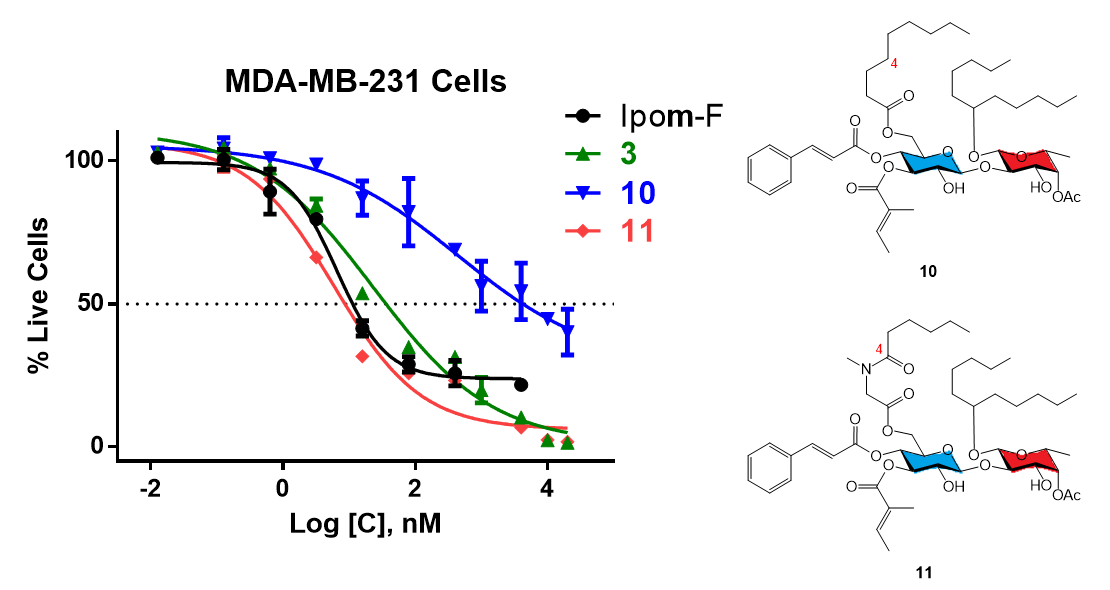
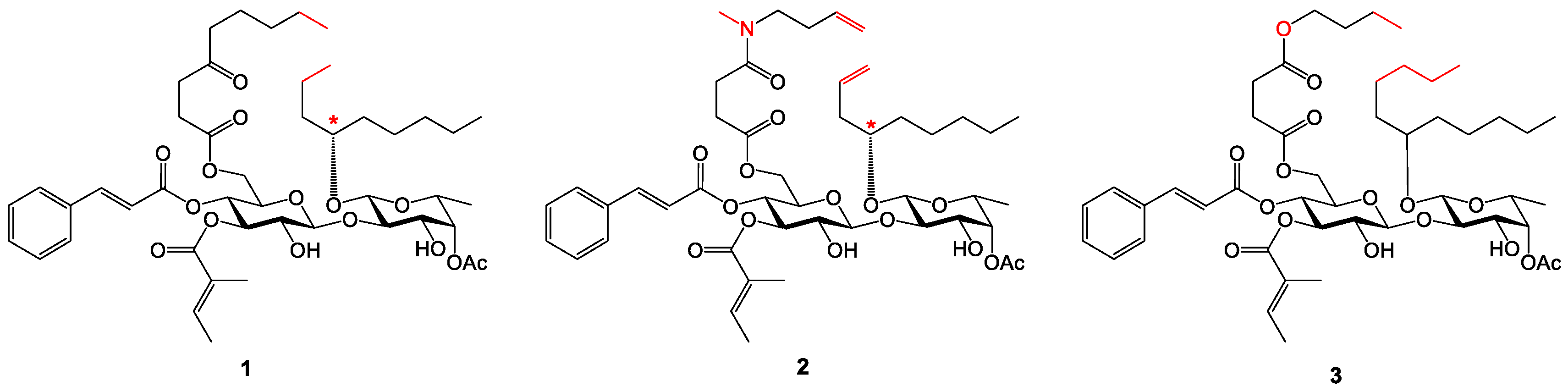
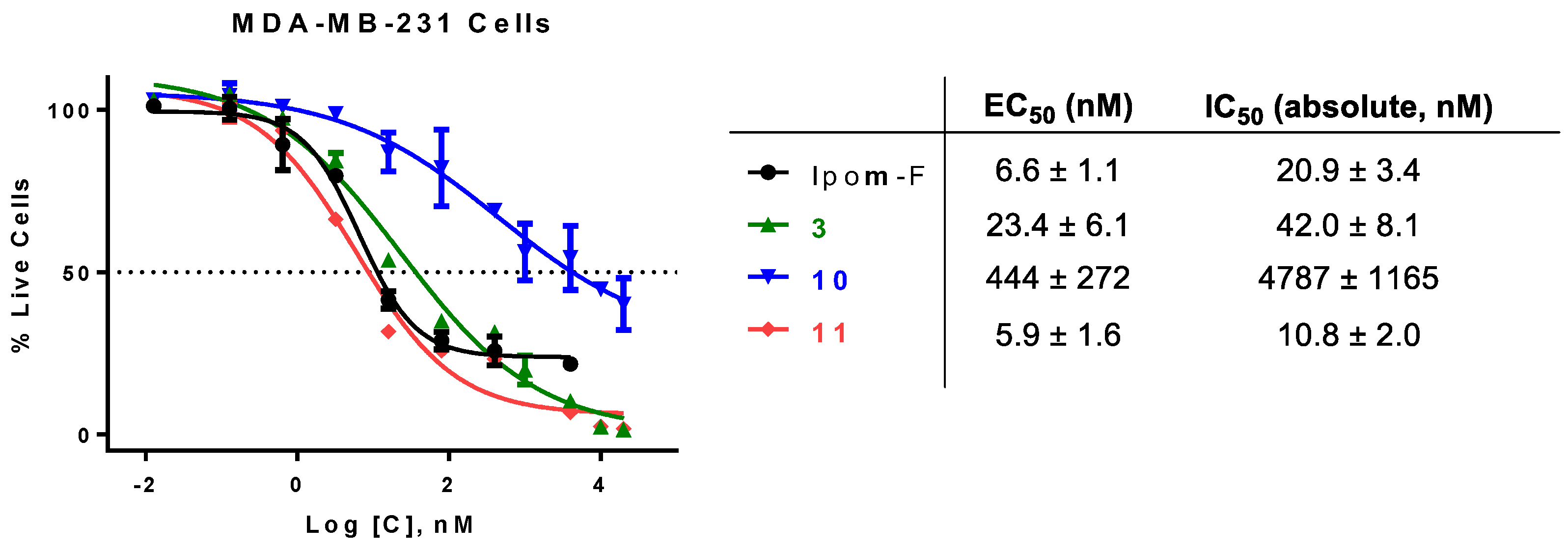
Analogue 10: The TES and TBS groups in 22 (45.4 mg, 0.0411 mmol) were removed by TBAF/AcOH. The crude product was purified by column chromatography (15:1→2:1, hexanes–EtOAc) to afford 11 as a colorless to pale yellow syrup (29.8 mg, 83%): Rf 0.55 (1:1 hexanes–EtOAc); 1H NMR (400 MHz, CDCl3, δH) 7.57 (d, J = 16.0 Hz, 1H), 7.38–7.48 (m, 2H), 7.26–7.38 (m, 3H), 6.75–6.87 (m, 1H), 6.27 (d, J = 16.0 Hz, 1H), 5.10–5.26 (m, 3H), 4.65 (d, J = 8.2 Hz, 1H), 4.38 (d, J = 7.3 Hz, 1H), 4.24 (br s, 1H), 4.13 (d, J = 3.8 Hz, 2H), 3.55–3.84 (m, 7H), 2.24 (td, J = 7.6, 3.2 Hz, 2H), 2.14 (s, 3H), 1.63–1.76 (m, 6H), 1.36–1.55 (m, 6H), 1.14–1.35 (m, 22H), 1.12 (d, J = 6.4 Hz, 3H), 0.73–0.88 (m, 9H); 13C NMR (100 MHz, CDCl3, δC) 173.6 (C=O), 171.4 (C=O), 168.1 (C=O), 165.6 (C=O), 146.6 (=CH), 139.2 (=CH), 134.1 (=C), 130.8 (=CH), 129.0 (2x=CH), 128.4 (2x=CH), 127.8 (=C), 116.6 (=CH), 102.8 (O2CH), 99.6 (O2CH), 79.3 (OCH), 77.7 (OCH), 74.4 (OCH), 72.8 (OCH), 72.2 (OCH), 71.8 (OCH), 70.8 (OCH), 69.3 (OCH), 68.3 (OCH), 62.3 (OCH2), 34.4 (CH2), 34.0 (CH2), 33.4 (CH2), 32.1 (CH2), 31.9 (2xCH2), 29.3 (CH2), 29.2(4) (CH2), 29.2(2) (CH2), 24.7(9) (CH2), 24.7(7) (2xCH2), 24.7(4) (CH2), 22.7(1) (CH2), 22.6(8) (CH2), 21.1 (CH3), 16.4 (CH3), 14.6 (CH3), 14.2(0) (2xCH3), 14.1(7) (CH3), 12.1 (CH3). HRMS m/z calcd for C48H74NaO14 [M+Na]+ 897.4969, found: 897.4976. Purity: 95.4% (MeCN/H2O 97:3; 1.5 mL/min, tR = 14.336 min).
Analogue 11: The TES and TBS groups in 23 (46.0 mg, 0.0406 mmol) were removed by TBAF/AcOH. The crude product was purified by column chromatography (10:1→1:1, hexanes–EtOAc) to afford 11 as a colorless to pale yellow syrup (31.6 mg, 86%). The ratio of the two rotamers (~4:1) was estimated from the 13C NMR spectrum. 1H NMR and 13C NMR are reported for the major rotamer: Rf 0.57 (1:2 hexanes–EtOAc); 1H NMR (400 MHz, CDCl3, δH) 7.61 (d, J = 16.0 Hz, 1H), 7.44–7.52 (m, 2H), 7.32–7.41 (m, 3H), 6.78–6.88 (m, 1H), 6.31 (d, J = 16.0 Hz, 1H), 5.11–5.31 (m, 3H), 4.70 (d, J = 8.2 Hz, 1H), 4.38 (d, J = 6.7 Hz, 1H), 4.22–4.31 (m, 3H), 3.98 (d, J = 17.4 Hz, 1H), 3.61–3.90 (m, 8H), 3.03 (s, 3H), 2.33 (t, J = 7.4 Hz, 2H), 2.18 (s, 3H), 1.66–1.78 (m, 6H), 1.40–1.66 (m, 6H), 1.18–1.36 (m, 16H), 1.16 (d, J = 6.4 Hz, 3H), 0.78–0.90 (m, 9H); 13C NMR (100 MHz, CDCl3, δC) 174.1 (C=O), 171.4 (C=O), 169.3 (C=O), 167.9 (C=O), 165.7 (C=O), 146.7 (=CH), 139.0 (=CH), 134.0 (=C), 130.8 (=CH), 129.1 (2x=CH), 128.4 (2x=CH), 127.9 (=C), 116.5 (=CH), 102.5 (O2CH), 99.6 (O2CH), 79.3 (OCH), 77.3 (OCH), 74.2 (OCH), 72.7 (OCH), 72.2 (OCH), 71.8 (OCH), 70.7 (OCH), 69.3 (OCH), 68.3 (OCH), 62.8 (OCH2), 49.2 (NCH2), 36.5 (CH3), 34.4 (CH2), 33.5 (CH2), 33.2 (CH2), 32.0 (CH2), 31.9 (CH2), 31.6 (CH2), 24.8 (2xCH2), 24.6 (CH2), 22.7 (2xCH2), 22.6 (CH2), 21.1 (CH3), 16.4 (CH3), 14.6 (CH3), 14.1(8) (CH3), 14.1(5) (CH3), 14.0(6) (CH3), 12.1 (CH3). HRMS m/z calcd for C48H73NNaO15 [M+Na]+ 926.4871, found: 926.4878. Purity: 95.1% (MeCN/H2O 87:13; 1.0 mL/min, tR = 17.144 min).