Submitted:
24 October 2024
Posted:
25 October 2024
You are already at the latest version
Abstract
Keywords:
1. Clinical Challenges in Myocardial Regeneration
2. The Promise of Cardiac Cellular Reprogramming
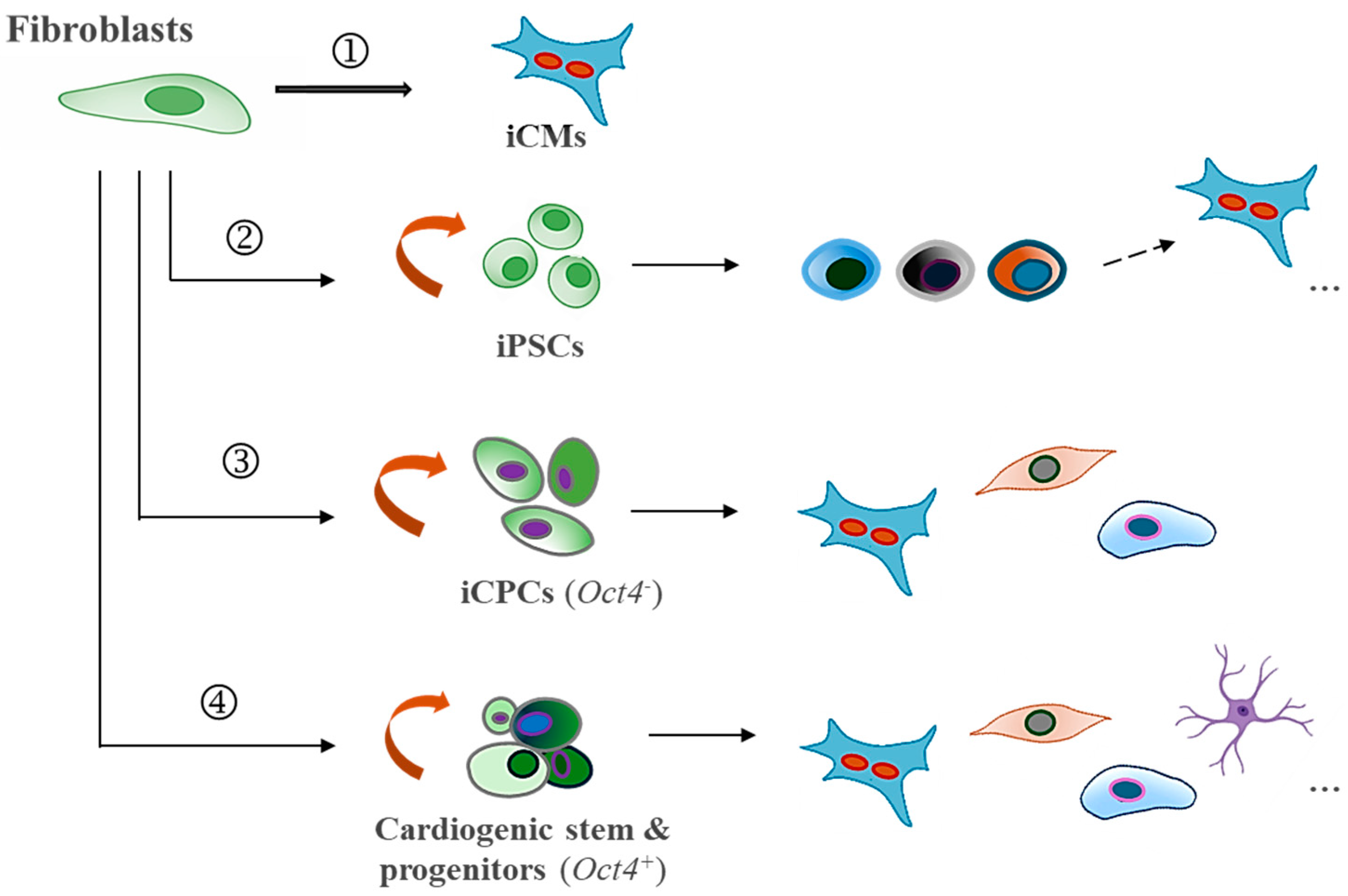
3. Induced Partial Cellular Fate Transitions and Recent Advances
3.1. Partial Reprogramming of Cardiomyocytes In Vitro
3.2. In Vivo Models of Partial Reprogramming
4. Advancing the Cardiac Partial Reprogramming Strategies
5. Prospective Considerations for Enhancing Therapeutic Cardiac Reprogramming
6. Conclusions
Funding
Informed Consent Statement
Conflicts of Interest
References
- Savarese, G. & Lund, L.H. Global Public Health Burden of Heart Failure. Card Fail Rev 3, 7-11 (2017). [CrossRef]
- Benjamin, E.J. et al. Heart Disease and Stroke Statistics-2018 Update: A Report From the American Heart Association. Circulation 137, e67-e492 (2018). [CrossRef]
- Aronow, W.S. Heart-failure-complicating acute myocardial infarction. Clin Geriatr Med 23, 123-139 (2007). [CrossRef]
- Jackson, S.L. et al. National Burden of Heart Failure Events in the United States, 2006 to 2014. Circ Heart Fail 11, e004873 (2018). [CrossRef]
- Akhtar, K.H. et al. The spectrum of post-myocardial infarction care: From acute ischemia to heart failure. Prog Cardiovasc Dis 82, 15-25 (2024). [CrossRef]
- Shah, D. & Sen, J. Mechanical Circulatory Support in Cardiogenic Shock: A Narrative Review. Cureus 16, e69379 (2024). [CrossRef]
- Heidenreich, P.A. et al. 2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation 145, e876-e894 (2022). [CrossRef]
- Carberry, J., Marquis-Gravel, G., O'Meara, E. & Docherty, K.F. Where Are We With Treatment and Prevention of Heart Failure in Patients Post-Myocardial Infarction? JACC Heart Fail 12, 1157-1165 (2024). [CrossRef]
- Dar, J.A. & Jacob, J.R. Beta Blockers in Contemporary Cardiology: Is It Better to Cast Them Out? Korean Circ J 54, 165-171 (2024). [CrossRef]
- Rosenzweig, A. Cardiac cell therapy--mixed results from mixed cells. N Engl J Med 355, 1274-1277 (2006). [CrossRef]
- Nguyen, P.K., Rhee, J.W. & Wu, J.C. Adult Stem Cell Therapy and Heart Failure, 2000 to 2016: A Systematic Review. JAMA Cardiol 1, 831-841 (2016). [CrossRef]
- Rosengart, T.K., Patel, V. & Sellke, F.W. Cardiac stem cell trials and the new world of cellular reprogramming: Time to move on. J Thorac Cardiovasc Surg 155, 1642-1646 (2018). [CrossRef]
- Abouzid, M.R. et al. Stem Cell Therapy for Myocardial Infarction and Heart Failure: A Comprehensive Systematic Review and Critical Analysis. Cureus 16, e59474 (2024). [CrossRef]
- Tsai, I.T. & Sun, C.K. Stem Cell Therapy against Ischemic Heart Disease. Int J Mol Sci 25 (2024). [CrossRef]
- Qian, L. et al. In vivo reprogramming of murine cardiac fibroblasts into induced cardiomyocytes. Nature 485, 593-598 (2012). [CrossRef]
- Song, K. et al. Heart repair by reprogramming non-myocytes with cardiac transcription factors. Nature 485, 599-604 (2012). [CrossRef]
- Ieda, M. et al. Direct reprogramming of fibroblasts into functional cardiomyocytes by defined factors. Cell 142, 375-386 (2010). [CrossRef]
- Mathison, M. et al. In vivo cardiac cellular reprogramming efficacy is enhanced by angiogenic preconditioning of the infarcted myocardium with vascular endothelial growth factor. J Am Heart Assoc 1, e005652 (2012). [CrossRef]
- Mathison, M. et al. "Triplet" polycistronic vectors encoding Gata4, Mef2c, and Tbx5 enhances postinfarct ventricular functional improvement compared with singlet vectors. J Thorac Cardiovasc Surg 148, 1656-1664 e1652 (2014). [CrossRef]
- Mathison, M. et al. In situ reprogramming to transdifferentiate fibroblasts into cardiomyocytes using adenoviral vectors: Implications for clinical myocardial regeneration. J Thorac Cardiovasc Surg 153, 329-339 e323 (2017). [CrossRef]
- Tani, H. et al. Direct Reprogramming Improves Cardiac Function and Reverses Fibrosis in Chronic Myocardial Infarction. Circulation 147, 223-238 (2023). [CrossRef]
- Perveen, S., Vanni, R., Lo Iacono, M., Rastaldo, R. & Giachino, C. Direct Reprogramming of Resident Non-Myocyte Cells and Its Potential for In Vivo Cardiac Regeneration. Cells 12 (2023). [CrossRef]
- Cao, N. et al. Conversion of human fibroblasts into functional cardiomyocytes by small molecules. Science 352, 1216-1220 (2016). [CrossRef]
- Christoforou, N. et al. Core Transcription Factors, MicroRNAs, and Small Molecules Drive Transdifferentiation of Human Fibroblasts Towards The Cardiac Cell Lineage. Sci Rep 7, 40285 (2017). [CrossRef]
- Nam, Y.J. et al. Reprogramming of human fibroblasts toward a cardiac fate. Proc Natl Acad Sci U S A 110, 5588-5593 (2013). [CrossRef]
- Wada, R. et al. Induction of human cardiomyocyte-like cells from fibroblasts by defined factors. Proc Natl Acad Sci U S A 110, 12667-12672 (2013). [CrossRef]
- Singh, V.P. et al. Enhanced Generation of Induced Cardiomyocytes Using a Small-Molecule Cocktail to Overcome Barriers to Cardiac Cellular Reprogramming. J Am Heart Assoc 9, e015686 (2020). [CrossRef]
- Garry, G.A. & Olson, E.N. Cardiac Reprogramming: Toward a Total Eclipse of the Failing Heart. Circulation 147, 239-241 (2023). [CrossRef]
- Talkhabi, M., Zonooz, E.R. & Baharvand, H. Boosters and barriers for direct cardiac reprogramming. Life Sci 178, 70-86 (2017). [CrossRef]
- Singh, V.P. et al. MiR-590 Promotes Transdifferentiation of Porcine and Human Fibroblasts Toward a Cardiomyocyte-Like Fate by Directly Repressing Specificity Protein 1. J Am Heart Assoc 5 (2016). [CrossRef]
- Vaseghi, H., Liu, J. & Qian, L. Molecular barriers to direct cardiac reprogramming. Protein Cell 8, 724-734 (2017). [CrossRef]
- Addis, R.C. et al. Optimization of direct fibroblast reprogramming to cardiomyocytes using calcium activity as a functional measure of success. J Mol Cell Cardiol 60, 97-106 (2013). [CrossRef]
- Zhou, H., Dickson, M.E., Kim, M.S., Bassel-Duby, R. & Olson, E.N. Akt1/protein kinase B enhances transcriptional reprogramming of fibroblasts to functional cardiomyocytes. Proceedings of the National Academy of Sciences of the United States of America 112, 11864-11869 (2015). [CrossRef]
- Patel, V. et al. p63 Silencing induces reprogramming of cardiac fibroblasts into cardiomyocyte-like cells. J Thorac Cardiovasc Surg 156, 556-565 e551 (2018). [CrossRef]
- Pinnamaneni, J.P. et al. p63 silencing induces epigenetic modulation to enhance human cardiac fibroblast to cardiomyocyte-like differentiation. Sci Rep 12, 11416 (2022). [CrossRef]
- Barreto, S., Hamel, L., Schiatti, T., Yang, Y. & George, V. Cardiac Progenitor Cells from Stem Cells: Learning from Genetics and Biomaterials. Cells 8 (2019). [CrossRef]
- Schwach, V. et al. Expandable human cardiovascular progenitors from stem cells for regenerating mouse heart after myocardial infarction. Cardiovasc Res 116, 545-553 (2020). [CrossRef]
- Xu, J., Lian, W., Li, L. & Huang, Z. Generation of induced cardiac progenitor cells via somatic reprogramming. Oncotarget 8, 29442-29457 (2017). [CrossRef]
- Moretti, A. et al. Mouse and human induced pluripotent stem cells as a source for multipotent Isl1+ cardiovascular progenitors. FASEB J 24, 700-711 (2010). [CrossRef]
- Parmacek, M.S. & Epstein, J.A. Pursuing cardiac progenitors: Regeneration redux. Cell 120, 295-298 (2005). [CrossRef]
- Lalit, P.A. et al. Lineage Reprogramming of Fibroblasts into Proliferative Induced Cardiac Progenitor Cells by Defined Factors. Cell Stem Cell 18, 354-367 (2016). [CrossRef]
- Zhang, Y. et al. Expandable Cardiovascular Progenitor Cells Reprogrammed from Fibroblasts. Cell Stem Cell 18, 368-381 (2016). [CrossRef]
- Wang, J. et al. Reprogramming of fibroblasts into expandable cardiovascular progenitor cells via small molecules in xeno-free conditions. Nat Biomed Eng 6, 403-420 (2022). [CrossRef]
- Islas, J.F. et al. Transcription factors ETS2 and MESP1 transdifferentiate human dermal fibroblasts into cardiac progenitors. Proc Natl Acad Sci U S A 109, 13016-13021 (2012). [CrossRef]
- Li, X.H. et al. Generation of Functional Human Cardiac Progenitor Cells by High-Efficiency Protein Transduction. Stem Cells Transl Med 4, 1415-1424 (2015). [CrossRef]
- Jiang, L. et al. CRISPR activation of endogenous genes reprograms fibroblasts into cardiovascular progenitor cells for myocardial infarction therapy. Mol Ther 30, 54-74 (2022). [CrossRef]
- Jiang, L., Liang, J.L., Huang, W., Paul, C. & Wang, Y.G. Reprogramming of Fibroblasts Into Cardiac Progenitor Cells Using Crispr Activation System. Circulation 138 (2018).
- Liu, L., Guo, Y., Li, Z. & Wang, Z. Improving Cardiac Reprogramming for Heart Regeneration in Translational Medicine. Cells 10 (2021). [CrossRef]
- He, X. et al. Advances in Cellular Reprogramming-Based Approaches for Heart Regenerative Repair. Cells 11 (2022). [CrossRef]
- Zhao, H.Y. & Huang, C.X. Conversion of human cardiac progenitor cells using reprogramming factors into heterogeneous cardiac pacemaker-like cells. Journal of Molecular and Cellular Cardiology 141, 53-53 (2020). [CrossRef]
- Efe, J.A. et al. Conversion of mouse fibroblasts into cardiomyocytes using a direct reprogramming strategy. Nat Cell Biol 13, 215-222 (2011). [CrossRef]
- Wang, H. et al. Small molecules enable cardiac reprogramming of mouse fibroblasts with a single factor, Oct4. Cell Rep 6, 951-960 (2014). [CrossRef]
- Fu, Y. et al. Direct reprogramming of mouse fibroblasts into cardiomyocytes with chemical cocktails. Cell Res 25, 1013-1024 (2015). [CrossRef]
- Kong, Y.P., Carrion, B., Singh, R.K. & Putnam, A.J. Matrix identity and tractional forces influence indirect cardiac reprogramming. Sci Rep 3, 3474 (2013). [CrossRef]
- Smith, A.W. et al. Direct reprogramming of mouse fibroblasts to cardiomyocyte-like cells using Yamanaka factors on engineered poly(ethylene glycol) (PEG) hydrogels. Biomaterials 34, 6559-6571 (2013). [CrossRef]
- Yannarelli, G. et al. OCT4 expression mediates partial cardiomyocyte reprogramming of mesenchymal stromal cells. PLoS ONE 12, e0189131 (2017). [CrossRef]
- Kisby, T., de Lazaro, I., Stylianou, M., Cossu, G. & Kostarelos, K. Transient reprogramming of postnatal cardiomyocytes to a dedifferentiated state. PLoS ONE 16, e0251054 (2021). [CrossRef]
- Chuang, W. et al. Partial Reprogramming of Pluripotent Stem Cell-Derived Cardiomyocytes into Neurons. Sci Rep 7, 44840 (2017). [CrossRef]
- Puri, D. & Wagner, W. Epigenetic rejuvenation by partial reprogramming. Bioessays 45, e2200208 (2023). [CrossRef]
- Gill, D. et al. Multi-omic rejuvenation of human cells by maturation phase transient reprogramming. Elife 11 (2022). [CrossRef]
- Mendelsohn, A.R., Larrick, J.W. & Lei, J.L. Rejuvenation by Partial Reprogramming of the Epigenome. Rejuvenation Res 20, 146-150 (2017). [CrossRef]
- Olova, N., Simpson, D.J., Marioni, R.E. & Chandra, T. Partial reprogramming induces a steady decline in epigenetic age before loss of somatic identity. Aging Cell 18, e12877 (2019). [CrossRef]
- Ocampo, A. et al. In Vivo Amelioration of Age-Associated Hallmarks by Partial Reprogramming. Cell 167, 1719-1733 e1712 (2016). [CrossRef]
- Paine, P.T., Nguyen, A. & Ocampo, A. Partial cellular reprogramming: A deep dive into an emerging rejuvenation technology. Aging Cell 23, e14039 (2024). [CrossRef]
- Singh, P.B. & Zhakupova, A. Age reprogramming: Cell rejuvenation by partial reprogramming. Development 149 (2022). [CrossRef]
- Chondronasiou, D. et al. Multi-omic rejuvenation of naturally aged tissues by a single cycle of transient reprogramming. Aging Cell 21, e13578 (2022). [CrossRef]
- Lu, Y. et al. Reprogramming to recover youthful epigenetic information and restore vision. Nature 588, 124-129 (2020). [CrossRef]
- de Lazaro, I., Cossu, G. & Kostarelos, K. Transient transcription factor (OSKM) expression is key towards clinical translation of in vivo cell reprogramming. EMBO Mol Med 9, 733-736 (2017). [CrossRef]
- Wang, C. et al. In vivo partial reprogramming of myofibers promotes muscle regeneration by remodeling the stem cell niche. Nat Commun 12, 3094 (2021). [CrossRef]
- Hishida, T. et al. In vivo partial cellular reprogramming enhances liver plasticity and regeneration. Cell Rep 39, 110730 (2022). [CrossRef]
- Kim, J. et al. Partial in vivo reprogramming enables injury-free intestinal regeneration via autonomous Ptgs1 induction. Sci Adv 9, eadi8454 (2023). [CrossRef]
- Doeser, M.C., Scholer, H.R. & Wu, G. Reduction of Fibrosis and Scar Formation by Partial Reprogramming In Vivo. Stem Cells 36, 1216-1225 (2018). [CrossRef]
- Cho, H.E. et al. In Vivo Reprogramming Using Yamanaka Factors in the CNS: A Scoping Review. Cells 13 (2024). [CrossRef]
- Gao, X., Wang, X., Xiong, W. & Chen, J. In vivo reprogramming reactive glia into iPSCs to produce new neurons in the cortex following traumatic brain injury. Sci Rep 6, 22490 (2016). [CrossRef]
- Dehghan, S. et al. Oct4 transcription factor in conjunction with valproic acid accelerates myelin repair in demyelinated optic chiasm in mice. Neuroscience 318, 178-189 (2016). [CrossRef]
- Su, Z., Niu, W., Liu, M.L., Zou, Y. & Zhang, C.L. In vivo conversion of astrocytes to neurons in the injured adult spinal cord. Nat Commun 5, 3338 (2014). [CrossRef]
- Tai, W. et al. In vivo reprogramming of NG2 glia enables adult neurogenesis and functional recovery following spinal cord injury. Cell Stem Cell 28, 923-937 e924 (2021). [CrossRef]
- Seo, J.H. et al. In Situ Pluripotency Factor Expression Promotes Functional Recovery From Cerebral Ischemia. Mol Ther 24, 1538-1549 (2016). [CrossRef]
- Niu, W. et al. In vivo reprogramming of astrocytes to neuroblasts in the adult brain. Nat Cell Biol 15, 1164-1175 (2013). [CrossRef]
- Chandrakanthan, V. et al. PDGF-AB and 5-Azacytidine induce conversion of somatic cells into tissue-regenerative multipotent stem cells. Proc Natl Acad Sci U S A 113, E2306-2315 (2016). [CrossRef]
- Kisby, T. et al. Adenoviral Mediated Delivery of OSKM Factors Induces Partial Reprogramming of Mouse Cardiac Cells In Vivo. Adv Ther-Germany 4 (2021). [CrossRef]
- Chen, Y. et al. Reversible reprogramming of cardiomyocytes to a fetal state drives heart regeneration in mice. Science 373, 1537-1540 (2021). [CrossRef]
- Gao, H. et al. Sall4 and Gata4 induce cardiac fibroblast transition towards a partially multipotent state with cardiogenic potential. Sci Rep 14, 24182 (2024). [CrossRef]
- Aguila, J.R. et al. SALL4 is a robust stimulator for the expansion of hematopoietic stem cells. Blood 118, 576-585 (2011). [CrossRef]
- Wu, J. et al. Improved Factor Combination for In Vivo Reprogramming of Cardiac Myofibroblast to Cardiomyocyte-Like Cell With Dual Recombinase Tracing. Circulation 148, 1728-1731 (2023). [CrossRef]
- Tao, Y. et al. Robust small molecule-aided cardiac reprogramming systems selective to cardiac fibroblasts. iScience 26, 108466 (2023). [CrossRef]
- Furtado, M.B. et al. Cardiogenic genes expressed in cardiac fibroblasts contribute to heart development and repair. Circ Res 114, 1422-1434 (2014). [CrossRef]
- Tang, Y. et al. TBX20 Improves Contractility and Mitochondrial Function During Direct Human Cardiac Reprogramming. Circulation 146, 1518-1536 (2022). [CrossRef]
- Chong, J.J. et al. Adult cardiac-resident MSC-like stem cells with a proepicardial origin. Cell Stem Cell 9, 527-540 (2011). [CrossRef]
- Yu, J. et al. Topological Arrangement of Cardiac Fibroblasts Regulates Cellular Plasticity. Circ Res 123, 73-85 (2018). [CrossRef]
- Moretti, A. et al. Multipotent embryonic isl1+ progenitor cells lead to cardiac, smooth muscle, and endothelial cell diversification. Cell 127, 1151-1165 (2006). [CrossRef]
- Mehanna, R.A. et al. Cardiac stem cells: Current knowledge and future prospects. World J Stem Cells 14, 1-40 (2022). [CrossRef]
- Nemajerova, A., Kim, S.Y., Petrenko, O. & Moll, U.M. Two-factor reprogramming of somatic cells to pluripotent stem cells reveals partial functional redundancy of Sox2 and Klf4. Cell Death Differ 19, 1268-1276 (2012). [CrossRef]
- Aires, A. et al. Neurovascular Coupling Impairment in Heart Failure with Reduction Ejection Fraction. Brain Sci 10 (2020). [CrossRef]
- Moore, M., Ryzhov, S., Sawyer, D.B., Gartner, C. & Vary, C.P.H. ALK1 Signaling in Human Cardiac Progenitor Cells Promotes a Pro-angiogenic Secretome. J Cell Signal 5, 122-142 (2024). [CrossRef]
- Khan, A. et al. Biomimetic Approaches in Cardiac Tissue Engineering: Replicating the Native Heart Microenvironment. Cureus 15, e43431 (2023). [CrossRef]
- Yuko, A.E. et al. LIN28a induced metabolic and redox regulation promotes cardiac cell survival in the heart after ischemic injury. Redox Biol 47, 102162 (2021). [CrossRef]
- Tachibana, A. et al. Paracrine Effects of the Pluripotent Stem Cell-Derived Cardiac Myocytes Salvage the Injured Myocardium. Circ Res 121, e22-e36 (2017). [CrossRef]
- Stefanovic, S. et al. Interplay of Oct4 with Sox2 and Sox17: A molecular switch from stem cell pluripotency to specifying a cardiac fate. J Cell Biol 186, 665-673 (2009). [CrossRef]
- Shu, J. et al. GATA family members as inducers for cellular reprogramming to pluripotency. Cell Res 25, 169-180 (2015). [CrossRef]
- Katano, W. et al. Sall1 and Sall4 cooperatively interact with Myocd and SRF to promote cardiomyocyte proliferation by regulating CDK and cyclin genes. Development 150 (2023). [CrossRef]
- Chen, L., Fulcoli, F.G., Tang, S. & Baldini, A. Tbx1 regulates proliferation and differentiation of multipotent heart progenitors. Circ Res 105, 842-851 (2009). [CrossRef]
- Lighthouse, J.K. & Small, E.M. Transcriptional control of cardiac fibroblast plasticity. J Mol Cell Cardiol 91, 52-60 (2016). [CrossRef]
- Raghunathan, S. et al. Conversion of human cardiac progenitor cells into cardiac pacemaker-like cells. J Mol Cell Cardiol 138, 12-22 (2020). [CrossRef]
- Liang, X. et al. HCN4 dynamically marks the first heart field and conduction system precursors. Circ Res 113, 399-407 (2013). [CrossRef]
- Alibhai, F.J. & Li, R.K. Rejuvenation of the Aging Heart: Molecular Determinants and Applications. Can J Cardiol 40, 1394-1411 (2024). [CrossRef]
- He, X. et al. Direct cellular reprogramming techniques for cardiovascular regenerative therapeutics. Can J Physiol Pharmacol 102, 1-13 (2024). [CrossRef]
- Kablak-Ziembicka, A. et al. Cardiac microRNAs: Diagnostic and therapeutic potential. Arch Med Sci 19, 1360-1381 (2023). [CrossRef]
- Abouelnazar, F.A. et al. The new advance of SALL4 in cancer: Function, regulation, and implication. J Clin Lab Anal 37, e24927 (2023). [CrossRef]
- Medlej, A., Mohammad Soltani, B., Javad Mowla, S., Hosseini, S. & Baharvand, H. A novel miRNA located in the GATA4 gene regulates the expression of IGF-1R and AKT1/2 genes and controls cell proliferation. J Cell Biochem 121, 3438-3450 (2020). [CrossRef]
- Liu, T. et al. Advanced Cardiac Patches for the Treatment of Myocardial Infarction. Circulation 149, 2002-2020 (2024). [CrossRef]
- Razavi, Z.S. et al. Advancements in tissue engineering for cardiovascular health: A biomedical engineering perspective. Front Bioeng Biotechnol 12, 1385124 (2024). [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



