Submitted:
22 October 2024
Posted:
25 October 2024
You are already at the latest version
Abstract
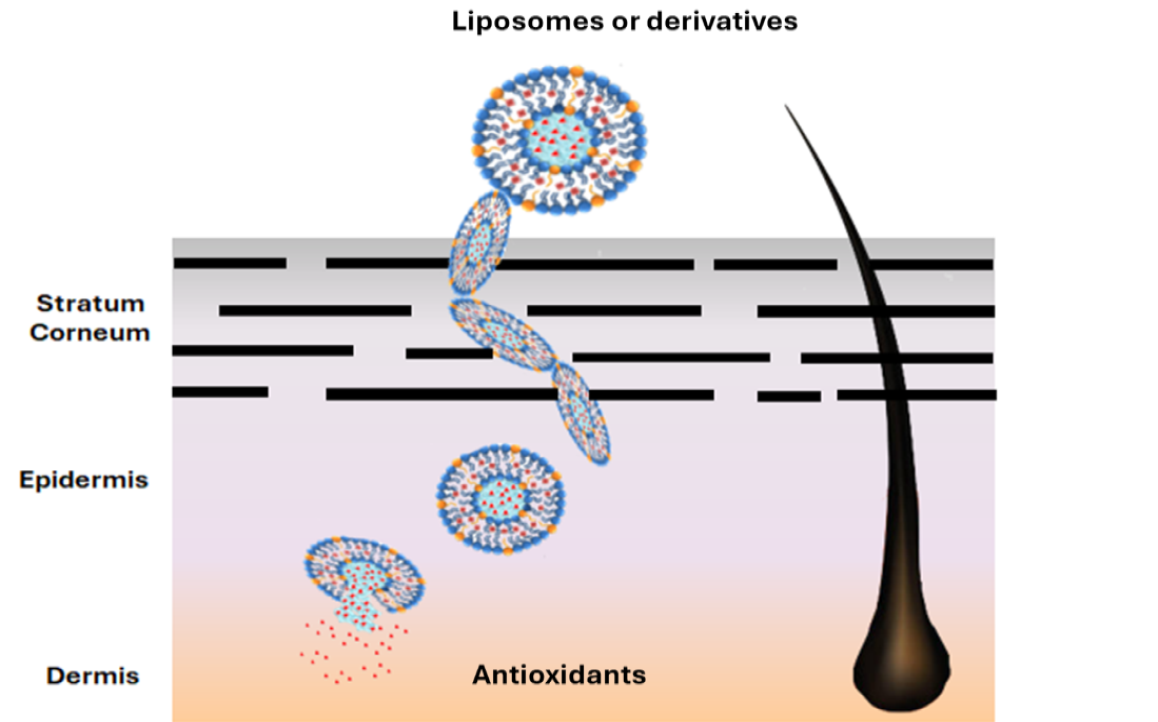
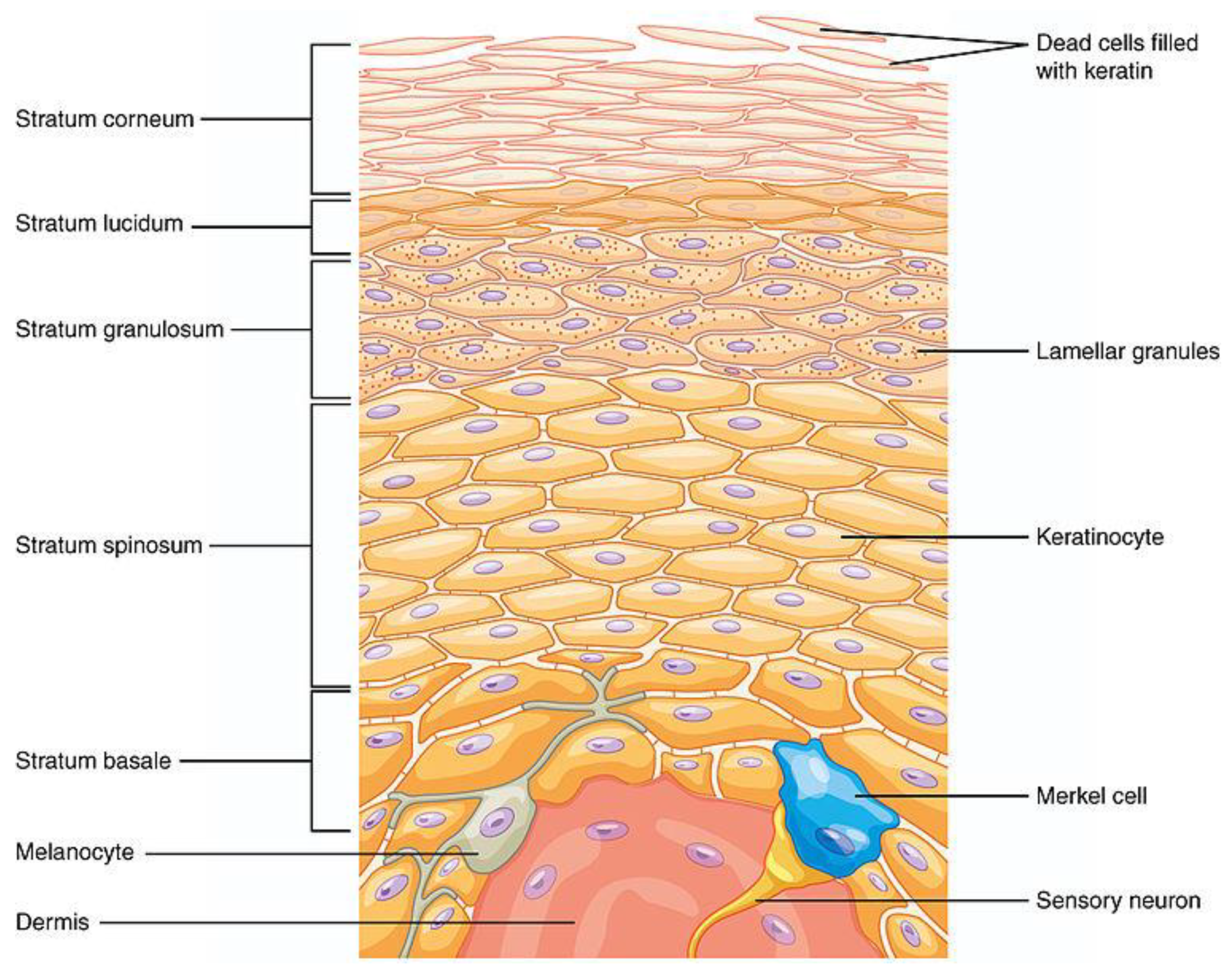
Keywords:
1. Introduction
2. Natural and Synthetic Vesicles
2.1.1. Liposomes
2.1.2. Ethosomes and Transethosomes
2.1.3. Niosomes
2.1.4. Unsaturated Fatty Acid Liposomes
3. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Sies, H.; Jones, D.P. Reactive Oxygen Species (ROS) as Pleiotropic Physiological Signalling Agents. Nat. Rev. Mol. Cell Biol. 2020, 21, 363–383. [Google Scholar] [CrossRef] [PubMed]
- Snezhkina, A.V.; Kudryavtseva, A.V.; Kardymon, O.L.; Savvateeva, M.V.; Melnikova, N.V.; Krasnov, G.S.; Dmitriev, A.A. ROS Generation and Antioxidant Defense Systems in Normal and Malignant Cells. Oxid. Med. Cell. Longev. 2019, 2019, e6175804. [Google Scholar] [CrossRef] [PubMed]
- Juan, C.A.; Pérez de la Lastra, J.M.; Plou, F.J.; Pérez-Lebeña, E. The Chemistry of Reactive Oxygen Species (ROS) Revisited: Outlining Their Role in Biological Macromolecules (DNA, Lipids and Proteins) and Induced Pathologies. Int. J. Mol. Sci. 2021, 22, 4642. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Understanding Mechanisms of Antioxidant Action in Health and Disease. Nat. Rev. Mol. Cell Biol. 2024, 25, 13–33. [Google Scholar] [CrossRef] [PubMed]
- Anik, M.I.; Mahmud, N.; Masud, A.A.; Khan, M.I.; Islam, M.N.; Uddin, S.; Hossain, M.K. Role of Reactive Oxygen Species in Aging and Age-Related Diseases: A Review. ACS Appl. Bio Mater. 2022, 5, 4028–4054. [Google Scholar] [CrossRef]
- Rinnerthaler, M.; Bischof, J.; Streubel, M.K.; Trost, A.; Richter, K. Oxidative Stress in Aging Human Skin. Biomolecules 2015, 5, 545–589. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.-M.; Cheng, M.-Y.; Xun, M.-H.; Zhao, Z.-W.; Zhang, Y.; Tang, W.; Cheng, J.; Ni, J.; Wang, W. Possible Mechanisms of Oxidative Stress-Induced Skin Cellular Senescence, Inflammation, and Cancer and the Therapeutic Potential of Plant Polyphenols. Int. J. Mol. Sci. 2023, 24, 3755. [Google Scholar] [CrossRef] [PubMed]
- Poljšak, B.; Fink, R. The Protective Role of Antioxidants in the Defence against ROS/RNS-Mediated Environmental Pollution. Oxid. Med. Cell. Longev. 2014, 2014, 671539. [Google Scholar] [CrossRef] [PubMed]
- Ndhlala, A.R.; Moyo, M.; Van Staden, J. Natural Antioxidants: Fascinating or Mythical Biomolecules? Molecules 2010, 15, 6905–6930. [Google Scholar] [CrossRef] [PubMed]
- Afzal, O.; Altamimi, A.S.A.; Nadeem, M.S.; Alzarea, S.I.; Almalki, W.H.; Tariq, A.; Mubeen, B.; Murtaza, B.N.; Iftikhar, S.; Riaz, N.; et al. Nanoparticles in Drug Delivery: From History to Therapeutic Applications. Nanomaterials 2022, 12, 4494. [Google Scholar] [CrossRef]
- Li, D.; Martini, N.; Liu, M.; Falconer, J.R.; Locke, M.; Wu, Z.; Wen, J. Non-Ionic Surfactant Vesicles as a Carrier System for Dermal Delivery of (+)-Catechin and Their Antioxidant Effects. J. Drug Target. 2021, 29, 310–322. [Google Scholar] [CrossRef] [PubMed]
- Sguizzato, M.; Ferrara, F.; Mariani, P.; Pepe, A.; Cortesi, R.; Huang, N.; Simelière, F.; Boldrini, P.; Baldisserotto, A.; Valacchi, G.; et al. “Plurethosome” as Vesicular System for Cutaneous Administration of Mangiferin: Formulative Study and 3D Skin Tissue Evaluation. Pharmaceutics 2021, 13, 1124. [Google Scholar] [CrossRef]
- Gimenis, J.M.; Figueiredo, C.C.M.; Gomes, A. da C.; Granero, F.O.; Ximenes, V.F.; Silva, L.P.; Bronzel Junior, J.L.; Leite e Silva, V.R.; Silva, R.M.G. da Microencapsulation of Moringa Oleifera L. Extract in Liposomes: Evaluation of Antioxidant and Antiglycation Activities, Cytotoxicity and Skin Biometric Parameters. J. Drug Deliv. Sci. Technol. 2023, 88, 104972. [Google Scholar] [CrossRef]
- Elmowafy, M.; Shalaby, K.; Elkomy, M.H.; Alsaidan, O.A.; Gomaa, H.A.M.; Abdelgawad, M.A.; Mostafa, E.M. Polymeric Nanoparticles for Delivery of Natural Bioactive Agents: Recent Advances and Challenges. Polymers 2023, 15, 1123. [Google Scholar] [CrossRef] [PubMed]
- Sguizzato, M.; Mariani, P.; Ferrara, F.; Drechsler, M.; Hallan, S.S.; Huang, N.; Simelière, F.; Khunti, N.; Cortesi, R.; Marchetti, N.; et al. Nanoparticulate Gels for Cutaneous Administration of Caffeic Acid. Nanomaterials 2020, 10, 961. [Google Scholar] [CrossRef]
- Mancuso, A.; Tarsitano, M.; Cavaliere, R.; Fresta, M.; Cristiano, M.C.; Paolino, D. Gelled Liquid Crystal Nanocarriers for Improved Antioxidant Activity of Resveratrol. Gels 2023, 9, 872. [Google Scholar] [CrossRef] [PubMed]
- Nsairat, H.; Khater, D.; Sayed, U.; Odeh, F.; Al Bawab, A.; Alshaer, W. Liposomes: Structure, Composition, Types, and Clinical Applications. Heliyon 2022, 8, e09394. [Google Scholar] [CrossRef] [PubMed]
- Kraft, J.C.; Freeling, J.P.; Wang, Ziyao. ; Ho, R.J.Y. Emerging Research and Clinical Development Trends of Liposome and Lipid Nanoparticle Drug Delivery Systems. J. Pharm. Sci. 2014, 103, 29–52. [Google Scholar] [CrossRef] [PubMed]
- Akombaetwa, N.; Ilangala, A.B.; Thom, L.; Memvanga, P.B.; Witika, B.A.; Buya, A.B. Current Advances in Lipid Nanosystems Intended for Topical and Transdermal Drug Delivery Applications. Pharmaceutics 2023, 15, 656. [Google Scholar] [CrossRef]
- Paolino, D.; d’Avanzo, N.; Canato, E.; Ciriolo, L.; Grigoletto, A.; Chiara Cristiano, M.; Mancuso, A.; Celia, C.; Pasut, G.; Fresta, M. Improved Anti-Breast Cancer Activity by Doxorubicin-Loaded Super Stealth Liposomes. Biomater. Sci. 2024, 12, 3933–3946. [Google Scholar] [CrossRef] [PubMed]
- Zarrabi, A.; Zarepour, A.; Khosravi, A.; Alimohammadi, Z.; Thakur, V.K. Synthesis of Curcumin Loaded Smart pH-Responsive Stealth Liposome as a Novel Nanocarrier for Cancer Treatment. Fibers 2021, 9, 19. [Google Scholar] [CrossRef]
- Bavarsad, N.; Karampour, N.S.; Hemmati, G.; Rezaie, A. The Effect of Topical Quercetin Loaded Liposome on Pressure Ulcer Healing in Rats. Nanomedicine J. 2021, 8, 187–199. [Google Scholar] [CrossRef]
- De Luca, M.; Lucchesi, D.; Tuberoso, C.I.G.; Fernàndez-Busquets, X.; Vassallo, A.; Martelli, G.; Fadda, A.M.; Pucci, L.; Caddeo, C. Liposomal Formulations to Improve Antioxidant Power of Myrtle Berry Extract for Potential Skin Application. Pharmaceutics 2022, 14, 910. [Google Scholar] [CrossRef] [PubMed]
- Permana, A.D.; Utami, R.N.; Courtenay, A.J.; Manggau, M.A.; Donnelly, R.F.; Rahman, L. Phytosomal Nanocarriers as Platforms for Improved Delivery of Natural Antioxidant and Photoprotective Compounds in Propolis: An Approach for Enhanced Both Dissolution Behaviour in Biorelevant Media and Skin Retention Profiles. J. Photochem. Photobiol. B 2020, 205, 111846. [Google Scholar] [CrossRef]
- Spanidi, E.; Karapetsas, A.; Voulgaridou, G.-P.; Letsiou, S.; Aligiannis, N.; Tsochantaridis, I.; Kynigopoulos, S.; Lambropoulou, M.; Mourtzinos, I.; Pappa, A.; et al. A New Controlled Release System for Propolis Polyphenols and Its Biochemical Activity for Skin Applications. Plants 2021, 10, 420. [Google Scholar] [CrossRef]
- Oskoueian, E.; Karimi, E.; Noura, R.; Ebrahimi, M.; Shafaei, N.; Karimi, E. Nanoliposomes Encapsulation of Enriched Phenolic Fraction from Pistachio Hulls and Its Antioxidant, Anti-Inflammatory, and Anti-Melanogenic Activities. J. Microencapsul. 2020, 37, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Caddeo, C.; Lucchesi, D.; Fernàndez-Busquets, X.; Valenti, D.; Penno, G.; Fadda, A.M.; Pucci, L. Efficacy of a Resveratrol Nanoformulation Based on a Commercially Available Liposomal Platform. Int. J. Pharm. 2021, 608, 121086. [Google Scholar] [CrossRef]
- Marwah, M.; Badhan, R.K.S.; Lowry, D. Development of a Novel Polymer-Based Carrier for Deformable Liposomes for the Controlled Dermal Delivery of Naringenin. J. Liposome Res. 2022, 32, 181–194. [Google Scholar] [CrossRef]
- Sguizzato, M.; Mariani, P.; Spinozzi, F.; Benedusi, M.; Cervellati, F.; Cortesi, R.; Drechsler, M.; Prieux, R.; Valacchi, G.; Esposito, E. Ethosomes for Coenzyme Q10 Cutaneous Administration: From Design to 3D Skin Tissue Evaluation. Antioxidants 2020, 9, 485. [Google Scholar] [CrossRef]
- Cândido, T.M.; De Oliveira, C.A.; Ariede, M.B.; Velasco, M.V.R.; Rosado, C.; Baby, A.R. Safety and Antioxidant Efficacy Profiles of Rutin-Loaded Ethosomes for Topical Application. AAPS PharmSciTech 2018, 19, 1773–1780. [Google Scholar] [CrossRef]
- Cristiano, M.C.; Barone, A.; Mancuso, A.; Torella, D.; Paolino, D. Rutin-Loaded Nanovesicles for Improved Stability and Enhanced Topical Efficacy of Natural Compound. J. Funct. Biomater. 2021, 12, 74. [Google Scholar] [CrossRef] [PubMed]
- Pintus, F.; Floris, S.; Fais, A.; Era, B.; Porcedda, C.; Tuberoso, C.I.G.; Caddeo, C. Euphorbia Characias Extract: Inhibition of Skin Aging-Related Enzymes and Nanoformulation. Plants 2022, 11, 1849. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, F.; Bondi, A.; Pula, W.; Contado, C.; Baldisserotto, A.; Manfredini, S.; Boldrini, P.; Sguizzato, M.; Montesi, L.; Benedusi, M.; et al. Ethosomes for Curcumin and Piperine Cutaneous Delivery to Prevent Environmental-Stressor-Induced Skin Damage. Antioxidants 2024, 13, 91. [Google Scholar] [CrossRef]
- Sguizzato, M.; Ferrara, F.; Hallan, S.S.; Baldisserotto, A.; Drechsler, M.; Malatesta, M.; Costanzo, M.; Cortesi, R.; Puglia, C.; Valacchi, G.; et al. Ethosomes and Transethosomes for Mangiferin Transdermal Delivery. Antioxidants 2021, 10, 768. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, F.; Benedusi, M.; Sguizzato, M.; Cortesi, R.; Baldisserotto, A.; Buzzi, R.; Valacchi, G.; Esposito, E. Ethosomes and Transethosomes as Cutaneous Delivery Systems for Quercetin: A Preliminary Study on Melanoma Cells. Pharmaceutics 2022, 14, 1038. [Google Scholar] [CrossRef] [PubMed]
- Al Saqr, A.; Annaji, M.; Poudel, I.; Rangari, S.; Boddu, S.H.S.; Tiwari, A.K.; Babu, R.J. Niosomal Formulation of Hydroxytyrosol, a Polyphenolic Antioxidant, for Enhancing Transdermal Delivery across Human Cadaver Skin. Pharm. Dev. Technol. 2022, 27, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Martini, N.; Wu, Z.; Chen, S.; Falconer, J.R.; Locke, M.; Zhang, Z.; Wen, J. Niosomal Nanocarriers for Enhanced Dermal Delivery of Epigallocatechin Gallate for Protection against Oxidative Stress of the Skin. Pharmaceutics 2022, 14, 726. [Google Scholar] [CrossRef]
- Cristiano, M.C.; Mancuso, A.; Fresta, M.; Torella, D.; De Gaetano, F.; Ventura, C.A.; Paolino, D. Topical Unsaturated Fatty Acid Vesicles Improve Antioxidant Activity of Ammonium Glycyrrhizinate. Pharmaceutics 2021, 13, 548. [Google Scholar] [CrossRef] [PubMed]
- Patil, Y.P.; Jadhav, S. Novel Methods for Liposome Preparation. Chem. Phys. Lipids 2014, 177, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Has, C.; Sunthar, P. A Comprehensive Review on Recent Preparation Techniques of Liposomes. J. Liposome Res. 2020, 30, 336–365. [Google Scholar] [CrossRef]
- Jiang, Y.; Li, W.; Wang, Z.; Lu, J. Lipid-Based Nanotechnology: Liposome. Pharmaceutics 2024, 16, 34. [Google Scholar] [CrossRef] [PubMed]
- Trucillo, P.; Campardelli, R.; Reverchon, E. Liposomes: From Bangham to Supercritical Fluids. Processes 2020, 8, 1022. [Google Scholar] [CrossRef]
- Mouritsen, O.G. Lipids, Curvature, and Nano-Medicine. Eur. J. Lipid Sci. Technol. 2011, 113, 1174–1187. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, D.; Kiselev, M.A. Methods of Liposomes Preparation: Formation and Control Factors of Versatile Nanocarriers for Biomedical and Nanomedicine Application. Pharmaceutics 2022, 14, 543. [Google Scholar] [CrossRef]
- Andra, V.V.S.N.L.; Pammi, S.V.N.; Bhatraju, L.V.K.P.; Ruddaraju, L.K. A Comprehensive Review on Novel Liposomal Methodologies, Commercial Formulations, Clinical Trials and Patents. Bionanoscience 2022, 12, 274–291. [Google Scholar] [CrossRef]
- Miller, C.R.; Bondurant, B.; McLean, S.D.; McGovern, K.A.; O’Brien, D.F. Liposome−Cell Interactions in Vitro: Effect of Liposome Surface Charge on the Binding and Endocytosis of Conventional and Sterically Stabilized Liposomes. Biochemistry 1998, 37, 12875–12883. [Google Scholar] [CrossRef]
- Papagiannaros, A.; Dimas, K.; Papaioannou, G.T.; Demetzos, C. Doxorubicin-PAMAM Dendrimer Complex Attached to Liposomes: Cytotoxic Studies against Human Cancer Cell Lines. Int. J. Pharm. 2005, 302, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Ascenso, A.; Raposo, S.; Batista, C.; Cardoso, P.; Mendes, T.; Praça, F.G.; Bentley, M.V.L.B.; Simões, S. Development, Characterization, and Skin Delivery Studies of Related Ultradeformable Vesicles: Transfersomes, Ethosomes, and Transethosomes. Int. J. Nanomedicine 2015, 10, 5837–5851. [Google Scholar] [CrossRef]
- Gokce, E.H.; Korkmaz, E.; Tuncay-Tanrıverdi, S.; Dellera, E.; Sandri, G.; Bonferoni, M.C.; Ozer, O. A Comparative Evaluation of Coenzyme Q10-Loaded Liposomes and Solid Lipid Nanoparticles as Dermal Antioxidant Carriers. Int. J. Nanomedicine 2012, 7, 5109–5117. [Google Scholar] [CrossRef]
- Sohn, S.-I.; Priya, A.; Balasubramaniam, B.; Muthuramalingam, P.; Sivasankar, C.; Selvaraj, A.; Valliammai, A.; Jothi, R.; Pandian, S. Biomedical Applications and Bioavailability of Curcumin—An Updated Overview. Pharmaceutics 2021, 13, 2102. [Google Scholar] [CrossRef]
- Shi, L.; Xun, W.; Peng, W.; Hu, H.; Cao, T.; Hou, G. Effect of the Single and Combined Use of Curcumin and Piperine on Growth Performance, Intestinal Barrier Function, and Antioxidant Capacity of Weaned Wuzhishan Piglets. Front. Vet. Sci. 2020, 7, 418. [Google Scholar] [CrossRef]
- Jantarat, C.; Sirathanarun, P.; Boonmee, S.; Meechoosin, W.; Wangpittaya, H. Effect of Piperine on Skin Permeation of Curcumin from a Bacterially Derived Cellulose-Composite Double-Layer Membrane for Transdermal Curcumin Delivery. Sci. Pharm. 2018, 86, 39. [Google Scholar] [CrossRef] [PubMed]
- Ma, M.; Wang, J.; Guo, F.; Lei, M.; Tan, F.; Li, N. Development of Nanovesicular Systems for Dermal Imiquimod Delivery: Physicochemical Characterization and in Vitro/in Vivo Evaluation. J. Mater. Sci. Mater. Med. 2015, 26, 191. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, P.; Tripathi, P.; Gupta, R.; Pandey, S. Niosomes: A Review on Niosomal Research in the Last Decade. J. Drug Deliv. Sci. Technol. 2020, 56, 101581. [Google Scholar] [CrossRef]
- Liga, S.; Paul, C.; Moacă, E.-A.; Péter, F. Niosomes: Composition, Formulation Techniques, and Recent Progress as Delivery Systems in Cancer Therapy. Pharmaceutics 2024, 16, 223. [Google Scholar] [CrossRef] [PubMed]
- Wang, E.; Jiang, Y.; Zhao, C. Hydroxytyrosol Isolation, Comparison of Synthetic Routes and Potential Biological Activities. Food Sci. Nutr. n/a. [CrossRef] [PubMed]
- Morigaki, K.; Walde, P. Fatty Acid Vesicles. Curr. Opin. Colloid Interface Sci. 2007, 12, 75–80. [Google Scholar] [CrossRef]
- Kaur, M.; Kaur, L.; Singh, G.; Singh, L.; Kaur, A.; Dhawan, R.K. Recent Advancements in Biomimetic Drug Delivery System of Single- Chain Fatty Acids as Ufasomes and Ufosomes: A Comprehensive Review. Curr. Nanosci. 2023, 19, 362–371. [Google Scholar] [CrossRef]
- Ando, H.; Ryu, A.; Hashimoto, A.; Oka, M.; Ichihashi, M. Linoleic Acid and α-Linolenic Acid Lightens Ultraviolet-Induced Hyperpigmentation of the Skin. Arch. Dermatol. Res. 1998, 290, 375–381. [Google Scholar] [CrossRef]
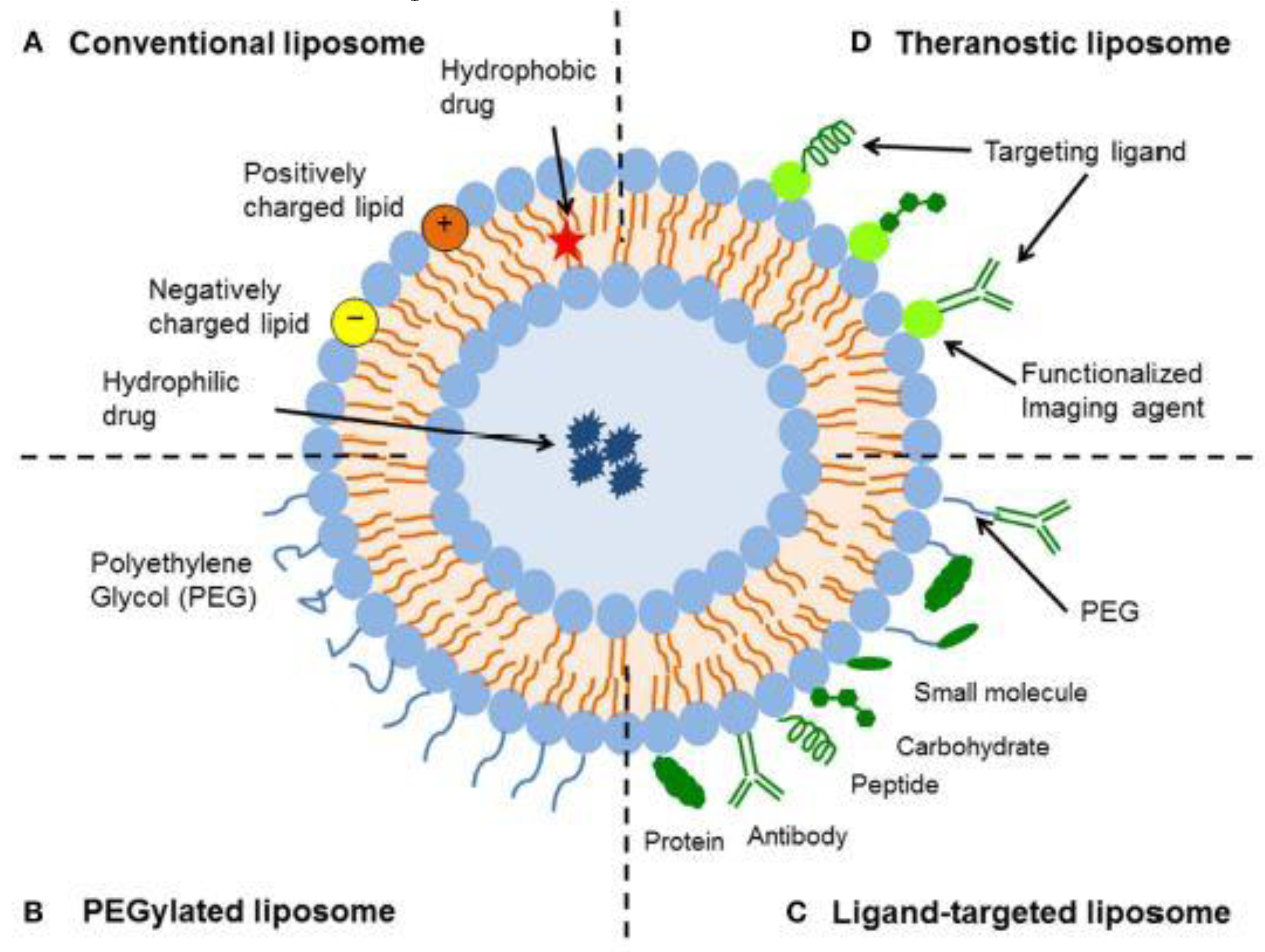
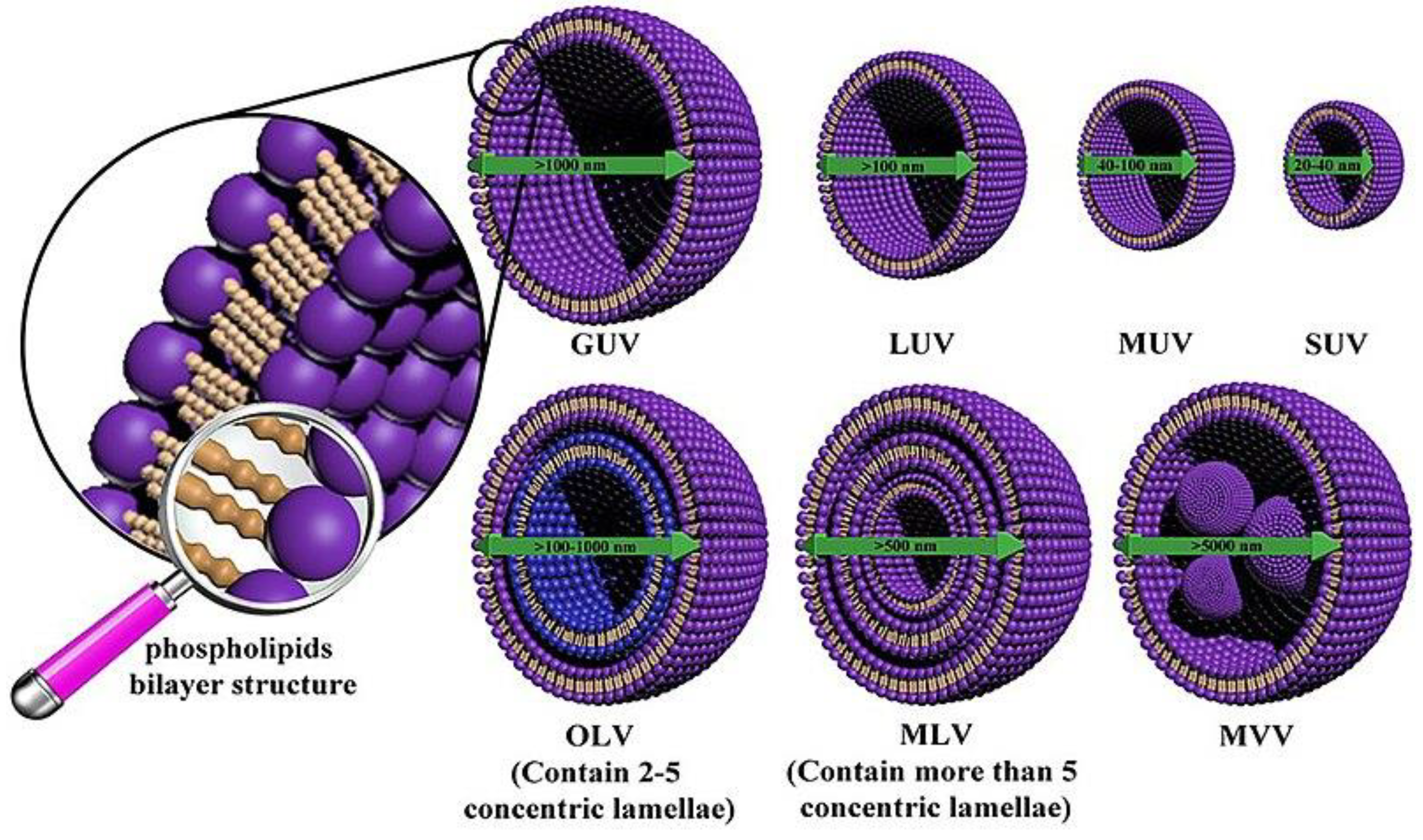
| entry | antioxidant | formulation | delivery system1 | properties analyzed2 | ref. |
|---|---|---|---|---|---|
| 1 | quercetin | pure comp. | LPS | A-Inf; VCir; | [22] |
| 2 | Moringa Oleifera | extract, cholesterol | LPS | A-ox | [13] |
| 3 | Myrtus communis | extract | LPS | A-ox | [23] |
| 4 | propolis | extract | LPS | A-ox; SAbs; PhP | [24] |
| 5 | propolis | extract, cyclodextrins |
LPS | A-ox; GR; PhP | [25] |
| 6 | onhadi pistachio | extract | LPS | A-ox; GR; A-mel | [26] |
| 7 | resveratrol | pure comp. | LPS | A-ox | [27] |
| 8 | naringenin | pure comp., Tween 20 |
TLPS | A-ox | [28] |
| 9 | coenzime Q10 | pure comp., EtOH | ETH | A-ox; SAbs | [29] |
| 10 | rutin | pure comp., EtOH | ETH | A-ox; SAbs | [30] |
| 11 | rutin | pure comp., EtOH | ETH | A-ox; A-Inf; PhP | [31] |
| 12 | euphorbia characias | pure comp., EtOH | ETH | A-ox; A-mel; PhP | [32] |
| 13 | curcumin, piperine | pure comp., EtOH | ETH | A-ox | [33] |
| 14 | mangiferin | pure comp., EtOH, surfactants | ETH, TETH | A-ox; A-Inf; SAbs; | [34] |
| 15 | quercetin | pure comp., EtOH, surfactants |
ETH, TETH | A-ox; SAbs; A-mel | [35] |
| 16 | hydroxytirosol | pure comp., EtOH, Span 60 |
NIS | SAbs | [36] |
| 17 | epigallocatechin gallate | pure comp., EtOH, surfactants |
NIS | A-ox; SAbs | [37] |
| 18 | glicirrate | pure comp., Fatty acids |
UFS | A-ox; SAbs; EM | [38] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




