Submitted:
21 October 2024
Posted:
24 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Data analysis and Statistics
2.3. Ethics
3. Results
3.1. Cohort
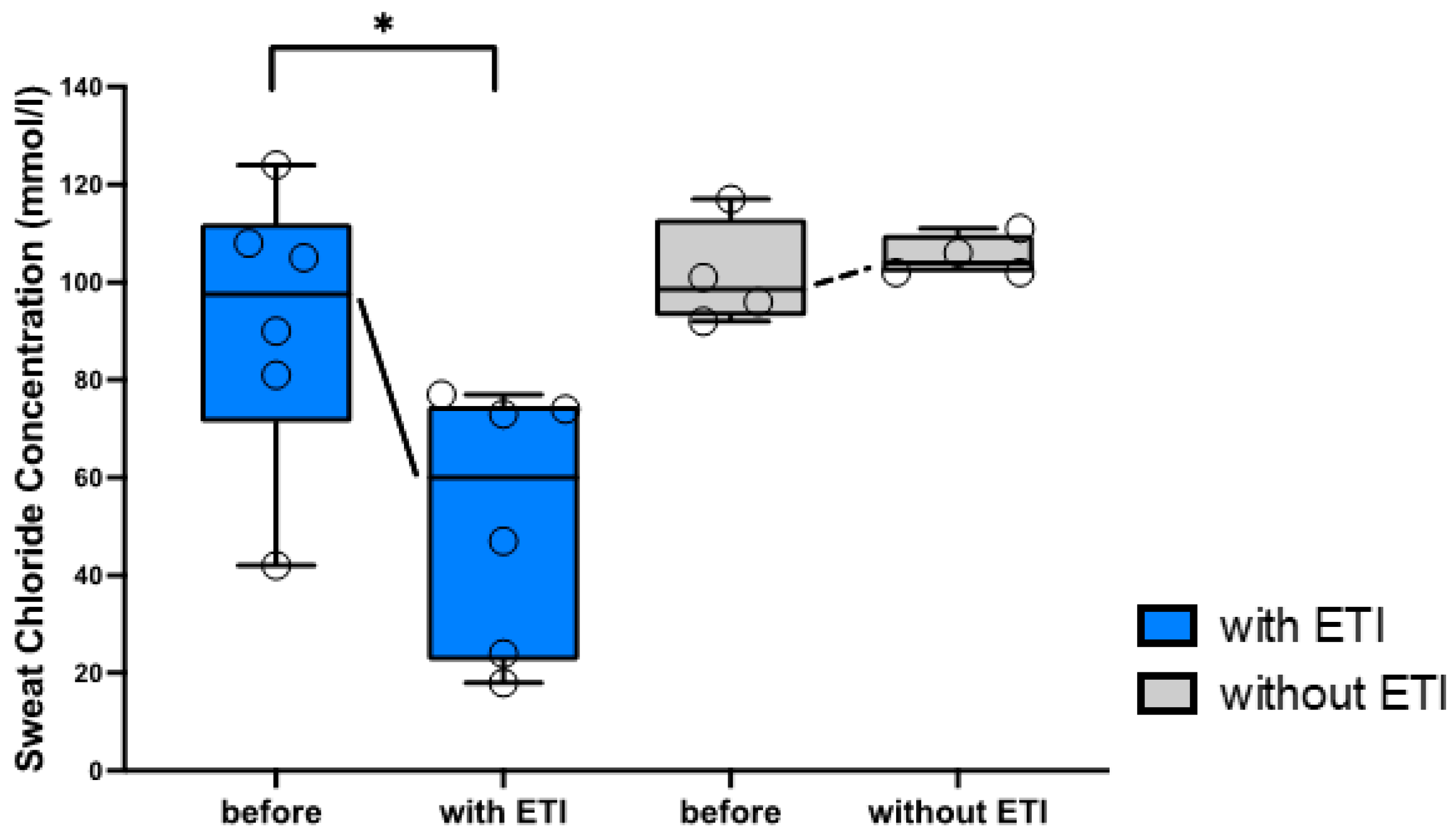
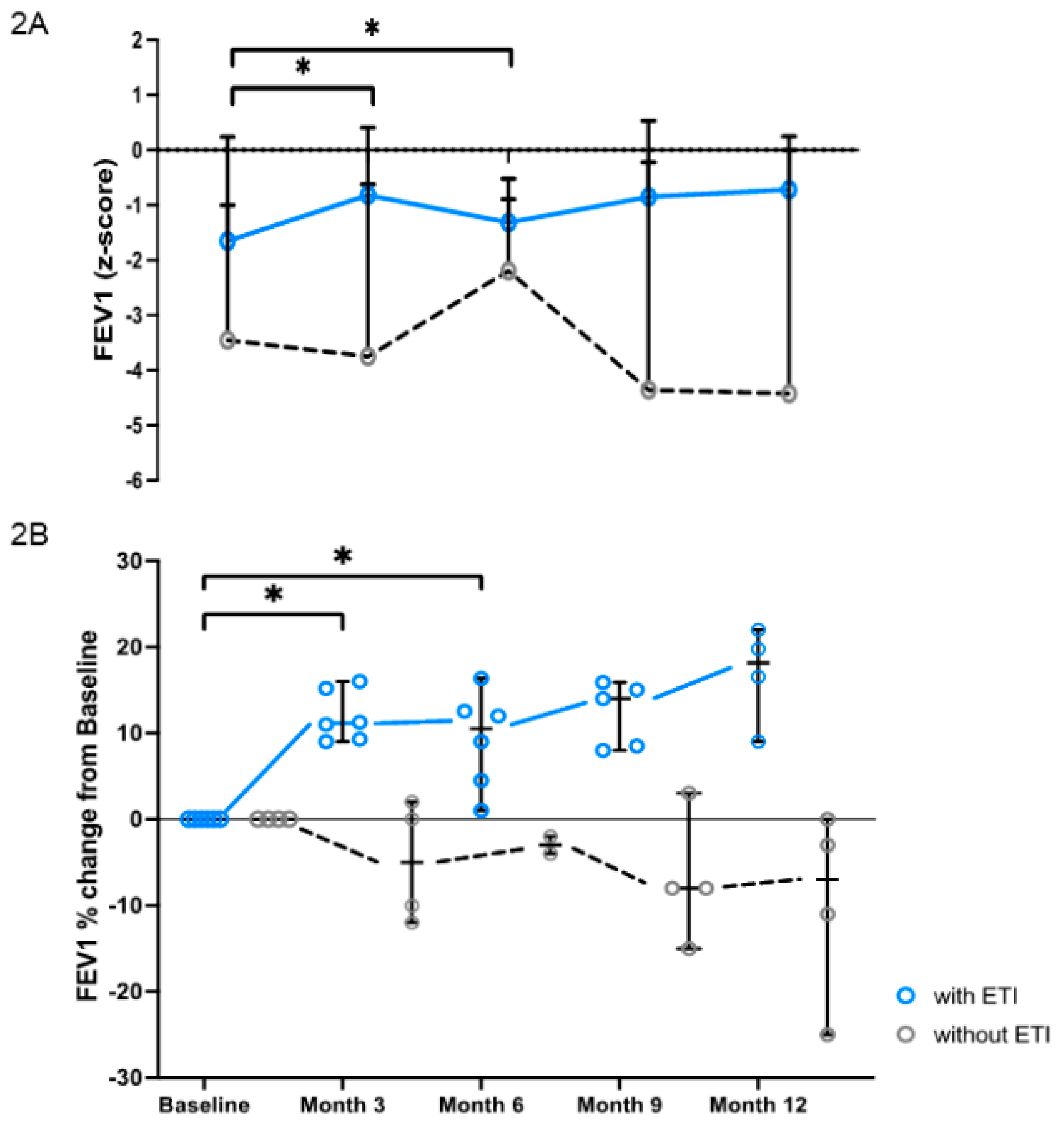
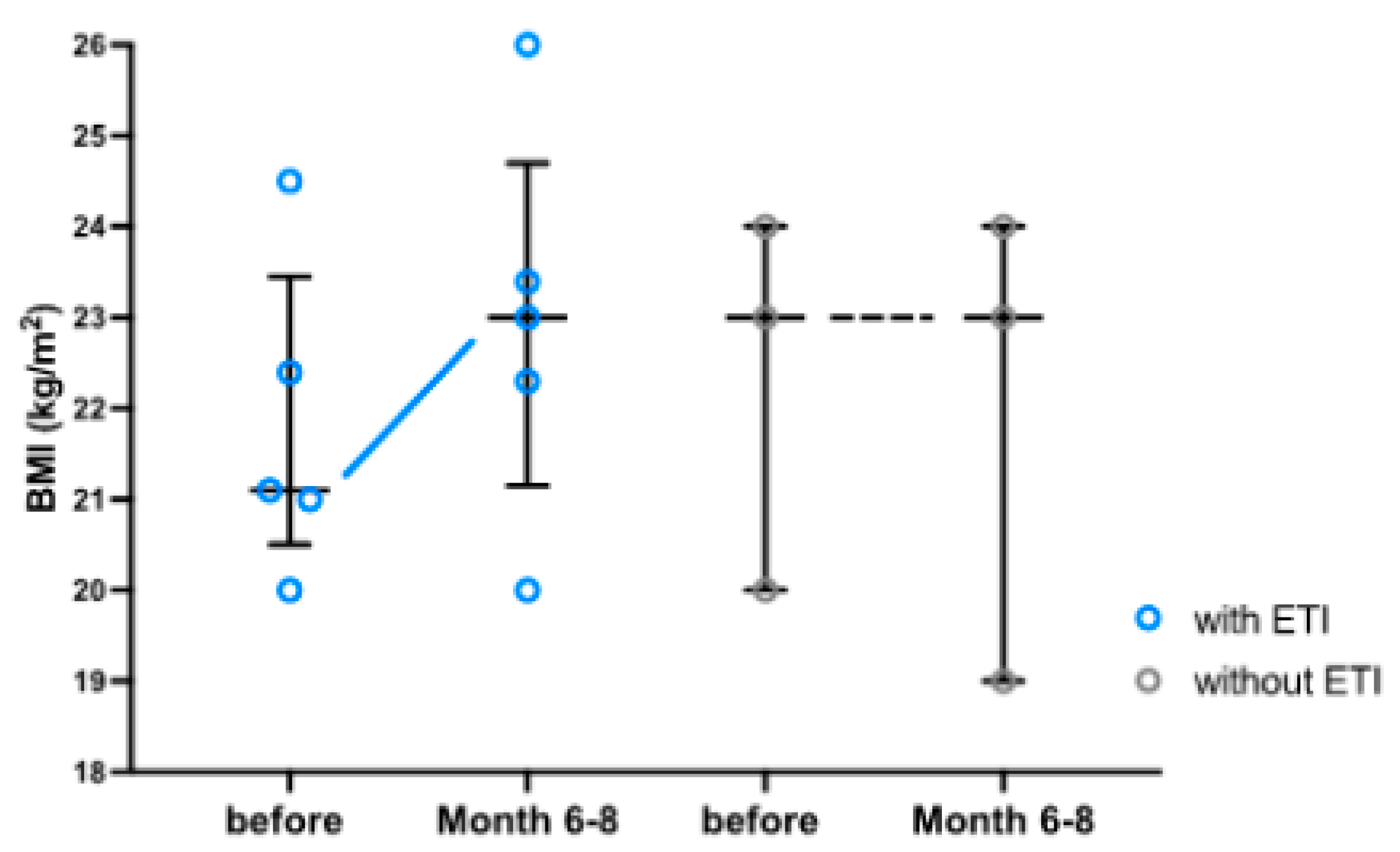
3.1. Clinical Parameters
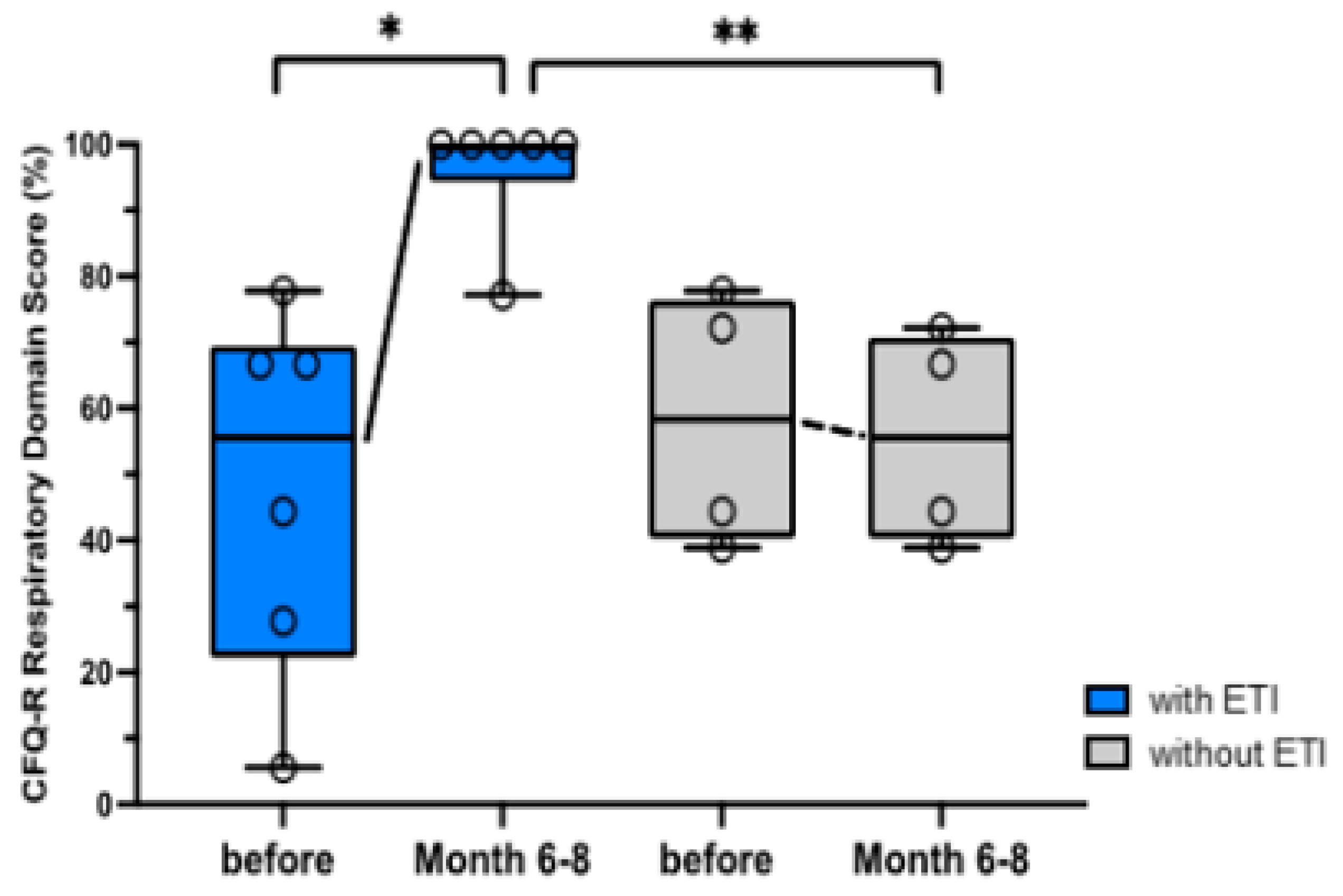
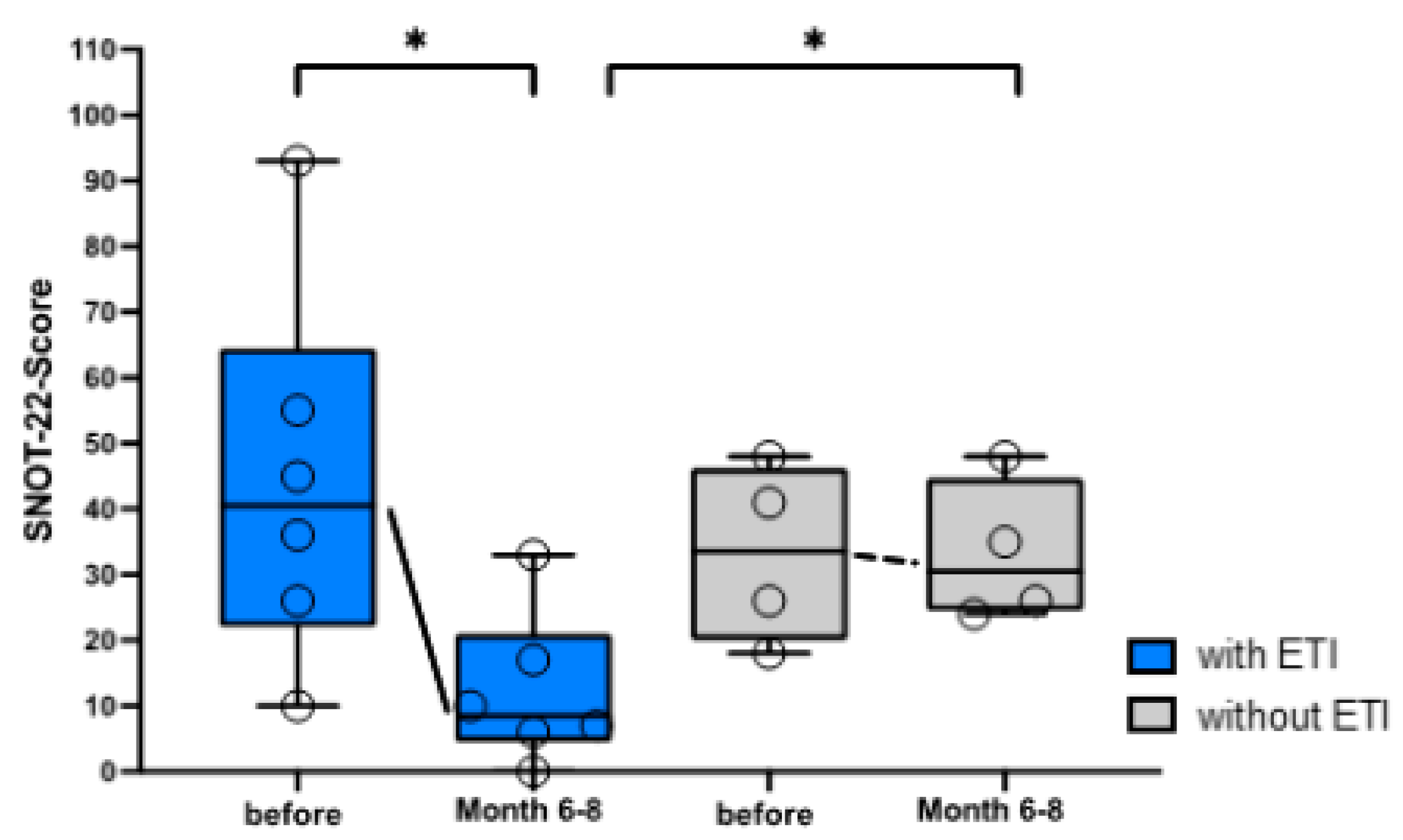
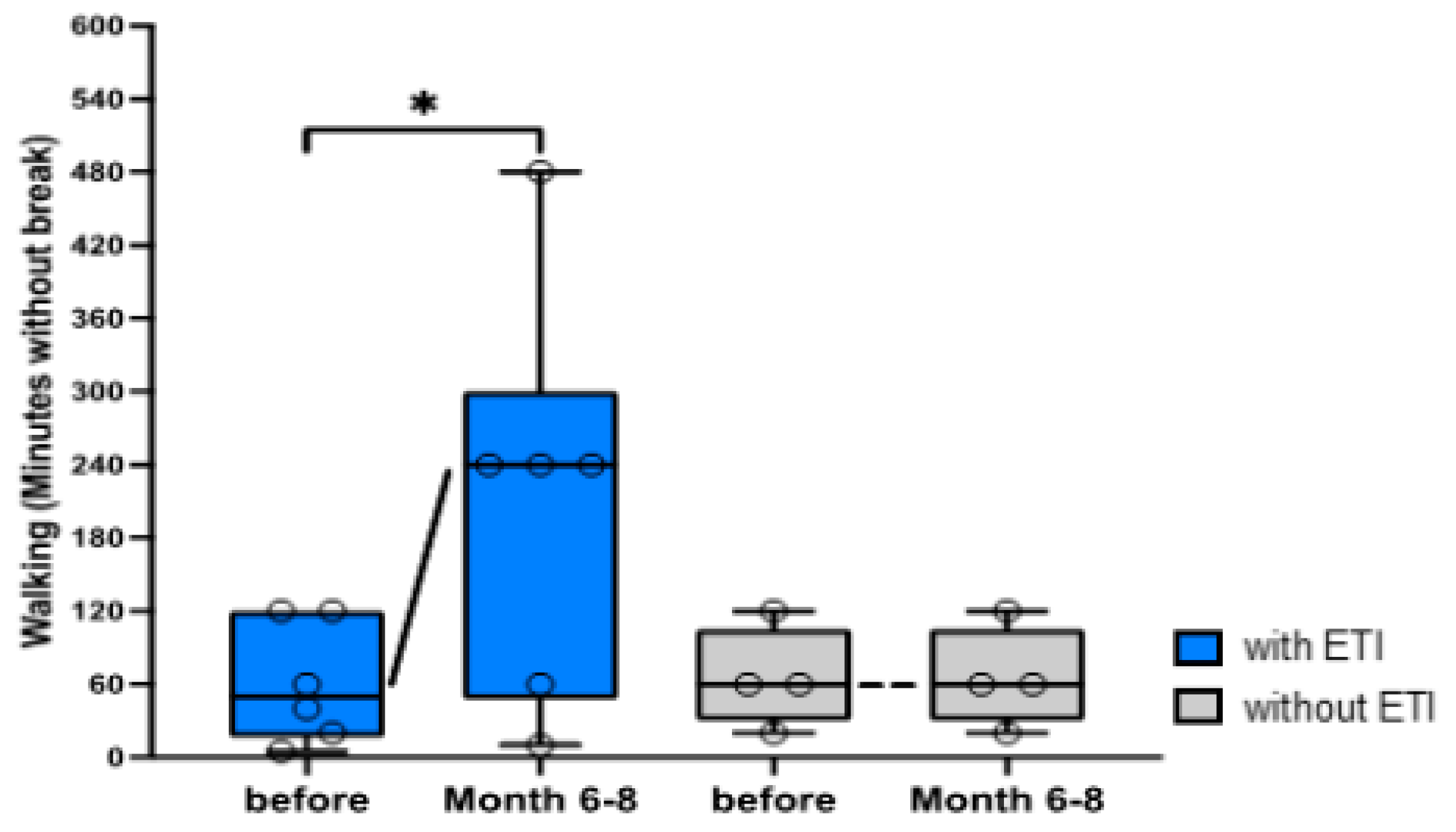
3.2. Questionnaires
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Guo, J.; Garratt, A.; Hill, A. Worldwide rates of diagnosis and effective treatment for cystic fibrosis. J Cyst Fibros 2022, 21, 456–462. [Google Scholar] [CrossRef] [PubMed]
- Shteinberg, M.; Haq, I.J.; Polineni, D.; Davies, J.C. Cystic fibrosis. Lancet 2021, 397, 2195–2211. [Google Scholar] [CrossRef] [PubMed]
- Nährlich, L.; Burkhart, M.; Wosniok, J. German Cystic Fibrosis Registry Annual Report 2022. Available online: https://www.muko.info/fileadmin/user_upload/was_wir_tun/register/berichtsbaende/annual_report_2022.pdf (accessed on 8 Jul 2024).
- Lord, L.; McKernon, D.; Grzeskowiak, L.; Kirsa, S.; Ilomaki, J. Depression and anxiety prevalence in people with cystic fibrosis and their caregivers: a systematic review and meta-analysis. Soc Psychiatry Psychiatr Epidemiol 2023, 58, 287–298. [Google Scholar] [CrossRef] [PubMed]
- Veit, G.; Roldan, A.; Hancock, M.A.; da Fonte, D.F.; Xu, H.; Hussein, M. , et al. Allosteric folding correction of F508del and rare CFTR mutants by elexacaftor-tezacaftor-ivacaftor (Trikafta) combination. JCI Insight 2020, 5. [Google Scholar] [CrossRef]
- Middleton, P.G.; Mall, M.A.; Dřevínek, P.; Lands, L.C.; McKone, E.F.; Polineni, D. , et al. Elexacaftor-Tezacaftor-Ivacaftor for Cystic Fibrosis with a Single Phe508del Allele. N Engl J Med 2019, 381, 1809–1819. [Google Scholar] [CrossRef]
- Heijerman, H.G.M.; McKone, E.F.; Downey, D.G.; Van Braeckel, E.; Rowe, S.M.; Tullis, E. , et al. Efficacy and safety of the elexacaftor plus tezacaftor plus ivacaftor combination regimen in people with cystic fibrosis homozygous for the F508del mutation: a double-blind, randomised, phase 3 trial. Lancet 2019, 394, 1940–1948. [Google Scholar] [CrossRef] [PubMed]
- Sutharsan, S.; McKone, E.F.; Downey, D.G.; Duckers, J.; MacGregor, G.; Tullis, E. , et al. Efficacy and safety of elexacaftor plus tezacaftor plus ivacaftor versus tezacaftor plus ivacaftor in people with cystic fibrosis homozygous for F508del-CFTR: a 24-week, multicentre, randomised, double-blind, active-controlled, phase 3b trial. Lancet Respir Med 2022, 10, 267–277. [Google Scholar] [CrossRef]
- Nährlich, L.; Burkhart, M.; Wosniok, J. German Cystic Fibrosis Registry Annual Report 2019. Available online: https://www.muko.info/fileadmin/user_upload/was_wir_tun/register/berichtsbaende/annual_report_2019.pdf (accessed on 20 Sep 2024).
- Gruber, W.; Stehling, F.; Blosch, C.; Dillenhoefer, S.; Olivier, M.; Brinkmann, F. , et al. Longitudinal changes in habitual physical activity in adult people with cystic fibrosis in the presence or absence of treatment with elexacaftor/tezacaftor/ivacaftor. Front Sports Act Living 2024, 6. [Google Scholar] [CrossRef]
- DiMango, E.; Overdevest, J.; Keating, C.; Francis, S.F.; Dansky, D.; Gudis, D. Effect of highly effective modulator treatment on sinonasal symptoms in cystic fibrosis. J Cyst Fibros 2021, 20, 460–463. [Google Scholar] [CrossRef]
- Douglas, J.E.; Civantos, A.M.; Locke, T.B.; Sweis, A.M.; Hadjiliadis, D.; Hong, G. , et al. Impact of novel CFTR modulator on sinonasal quality of life in adult patients with cystic fibrosis. Int Forum Allergy Rhinol 2021, 11, 201–203. [Google Scholar] [CrossRef]
- Shakir, S.; Echevarria, C.; Doe, S.; Brodlie, M.; Ward, C.; Bourke, S.J. Elexacaftor-Tezacaftor-Ivacaftor improve Gastro-Oesophageal reflux and Sinonasal symptoms in advanced cystic fibrosis. J Cyst Fibros 2022, 21, 807–810. [Google Scholar] [CrossRef] [PubMed]
- Beswick, D.M.; Humphries, S.M.; Balkissoon, C.D.; Strand, M.; Vladar, E.K.; Lynch, D.A. , et al. Impact of Cystic Fibrosis Transmembrane Conductance Regulator Therapy on Chronic Rhinosinusitis and Health Status: Deep Learning CT Analysis and Patient-reported Outcomes. Ann Am Thorac Soc 2022, 19, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Bode, S.F.N.; Rapp, H.; Lienert, N.; Appel, H.; Fabricius, D. Effects of CFTR-modulator triple therapy on sinunasal symptoms in children and adults with cystic fibrosis. Eur Arch Otorhinolaryngol 2023. [CrossRef]
- Arslan, M.; Chalmers, S.; Rentfrow, K.; Olson, J.M.; Dean, V.; Wylam, M.E. , et al. Suicide attempts in adolescents with cystic fibrosis on Elexacaftor/Tezacaftor/Ivacaftor therapy. J Cyst Fibros 2023, 22, 427–430. [Google Scholar] [CrossRef]
- Bathgate, C.J.; Muther, E.; Georgiopoulos, A.M.; Smith, B.; Tillman, L.; Graziano, S. , et al. Positive and negative impacts of elexacaftor/tezacaftor/ivacaftor: Healthcare providers’ observations across US centers. Pediatr Pulmonol 2023, 58, 2469–2477. [Google Scholar] [CrossRef]
- Piehler, L.; Thalemann, R.; Lehmann, C.; Thee, S.; Röhmel, J.; Syunyaeva, Z. , et al. Effects of elexacaftor/tezacaftor/ivacaftor therapy on mental health of patients with cystic fibrosis. Front Pharmacol 2023, 14. [Google Scholar] [CrossRef] [PubMed]
- Ramsey, B.; Correll, C.U.; DeMaso, D.R.; McKone, E.; Tullis, E.; Taylor-Cousar, J.L. , et al. Elexacaftor/Tezacaftor/Ivacaftor Treatment and Depression-related Events. Am J Respir Crit Care Med 2024, 209, 299–306. [Google Scholar] [CrossRef]
- Food and Drug Administration. HIGHLIGHTS OF PRESCRIBING INFORMATION; Available online:. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/212273s004lbl.pdf (accessed on 16 Aug 2024).
- Durmowicz, A.G.; Lim, R.; Rogers, H.; Rosebraugh, C.J.; Chowdhury, B.A. The U.S. Food and Drug Administration’s Experience with Ivacaftor in Cystic Fibrosis. Establishing Efficacy Using In Vitro Data in Lieu of a Clinical Trial. Ann Am Thorac Soc 2018, 15, 1–2. [Google Scholar] [CrossRef]
- Sosnay, P.R.; Siklosi, K.R.; Van Goor, F.; Kaniecki, K.; Yu, H.; Sharma, N. , et al. Defining the disease liability of variants in the cystic fibrosis transmembrane conductance regulator gene. Nat Genet [Internet]. 2023, 45, pp. 1160–1167. Available online: https://pubmed.ncbi.nlm.nih.gov/23974870/.
- Burgel, P.R.; Sermet-Gaudelus, I.; Durieu, I.; Kanaan, R.; Macey, J.; Grenet, D. , et al. The French Compassionate Program of elexacaftor-tezacaftor-ivacaftor in people with cystic fibrosis with advanced lung disease and no F508del CFTR variant. European Respiratory Journal 2024, 2202437. [Google Scholar]
- Livnat, G.; Dagan, A.; Heching, M.; Shmueli, E.; Prais, D.; Yaacoby-Bianu, K. , et al. Treatment effects of Elexacaftor/Tezacaftor/Ivacaftor in people with CF carrying non-F508del mutations. J Cyst Fibros 2023, 22, 450–455. [Google Scholar] [CrossRef]
- Leung, W.M.; Davoodi, P.M.; Langevin, A.; Smith, C.; Parkins, M.D. Elexacaftor-Tezacaftor-Ivacaftor in 2 cystic fibrosis adults homozygous for M1101K with end-stage lung disease. Respir Med Case Rep 2023, 46, 101938. [Google Scholar] [CrossRef] [PubMed]
- Pinto, M.O.; Santos, A.S.; Castanhinha, S. Efficacy of elexacaftor-tezacaftor-ivacaftor in portuguese adolescents and adults with cystic fibrosis carrying non-F508del variants. Pulmonology 2024. [CrossRef] [PubMed]
- Albrecht, T.; Beule, A.G.; Hildenbrand, T.; Gerstacker, K.; Praetorius, M.; Rudack, C. , et al. Cross-cultural adaptation and validation of the 22-item sinonasal outcome test (SNOT-22) in German-speaking patients: a prospective, multicenter cohort study. Eur Arch Otorhinolaryngol 2022, 279, 2433–2439. [Google Scholar] [CrossRef] [PubMed]
- Henry, B.; Aussage, P.; Grosskopf, C.; Goehrs, J. Development of the Cystic Fibrosis Questionnaire (CFQ) for assessing quality of life in pediatric and adult patients. Qual Life Res 2003, 12, 63–76. [Google Scholar] [CrossRef] [PubMed]
- Regnault, A.; Balp, M.M.; Kulich, K.; Viala-Danten, M. Validation of the Treatment Satisfaction Questionnaire for Medication in patients with cystic fibrosis. J Cyst Fibros 2012, 11, 494–501. [Google Scholar] [CrossRef]
- Fuchs, R.; Klaperski, S.; Gerber, M.; Seelig, H. Messung der Bewegungs- und Sportaktivität mit dem BSA-Fragebogen. Eur J Health Psychol 2015, 23, 60–76. [Google Scholar] [CrossRef]
- Causer, A.J.; Shute, J.K.; Cummings, M.H.; Shepherd, A.I.; Wallbanks, S.R.; Pulsford, R.M. , et al. Elexacaftor–Tezacaftor–Ivacaftor improves exercise capacity in adolescents with cystic fibrosis. Pediatr Pulmonol 2022, 57, 2652–2658. [Google Scholar] [CrossRef]
- Stastna, N.; Hrabovska, L.; Homolka, P.; Homola, L.; Svoboda, M.; Brat, K. , et al. The long-term effect of elexacaftor/tezacaftor/ivacaftor on cardiorespiratory fitness in adolescent patients with cystic fibrosis: a pilot observational study. BMC Pulm Med 2024, 24, 260. [Google Scholar] [CrossRef]
- Zemanick, E.T.; Taylor-Cousar, J.L.; Davies, J.; Gibson, R.L.; Mall, M.A.; McKone, E.F. , et al. A Phase 3 Open-Label Study of Elexacaftor/Tezacaftor/Ivacaftor in Children 6 through 11 Years of Age with Cystic Fibrosis and at Least One F508del Allele. Am J Respir Crit Care Med 2021, 203, 1522–1532. [Google Scholar] [CrossRef]
| Patient | Age (y) | Gender | CFTR-Mutation 1 | CFTR-Mutation 2 | ETI | Ivacaftor before ETI |
|---|---|---|---|---|---|---|
| 1 | 45 | f | R347P* | R1066C | Yes | No |
| 2 | 6 | m | Q220X | M1101K* | Yes | No |
| 3 | 66 | f | M1101K* | 2789+5G>A | Yes | No |
| 4 | 35 | m | G542X | M1101K* | Yes | No |
| 5 | 45 | m | CFTRdele2,3 | M1101K* | Yes | No |
| 6 | 29 | f | G551D* | M1101K* | Yes | Yes |
| A | 22 | f | Q39X | CFTRdele17a,17b | No | No |
| B | 7 | m | Q39X | R785X | No | No |
| C | 46 | f | phe508del* | phe508del* | No | No |
| D | 23 | f | I506S | CFTRdele14b-17b | No | No |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




