Submitted:
28 October 2024
Posted:
30 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Cochineal Extraction Yield Obtained by the Conventional Method
2.2. Fractional Factorial Design
2.3. Statistical Analysis
2.4. Analysis of Response Surface Plots
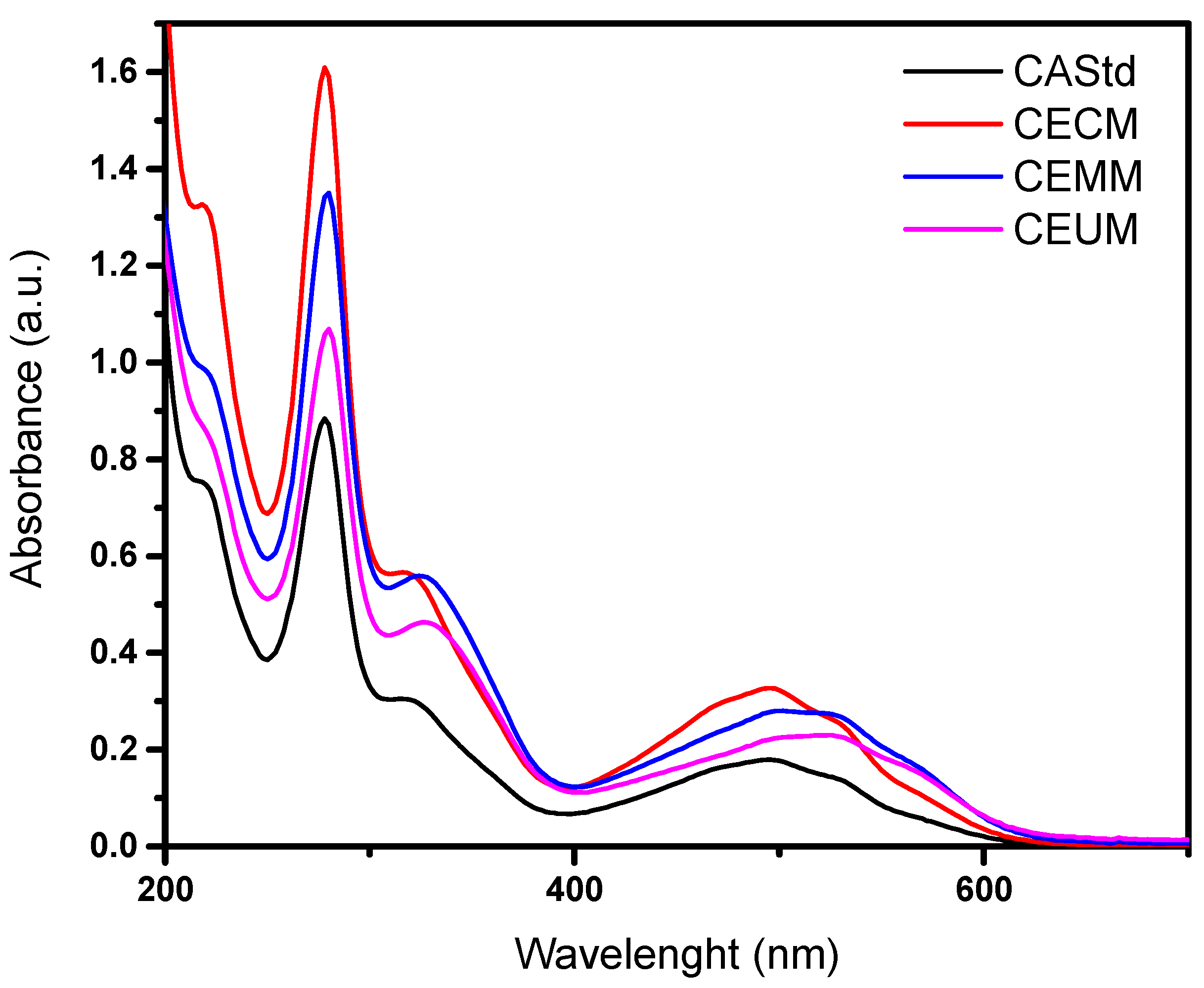
2.5. Comparison Between the Conventional Method and Green Extraction Techniques
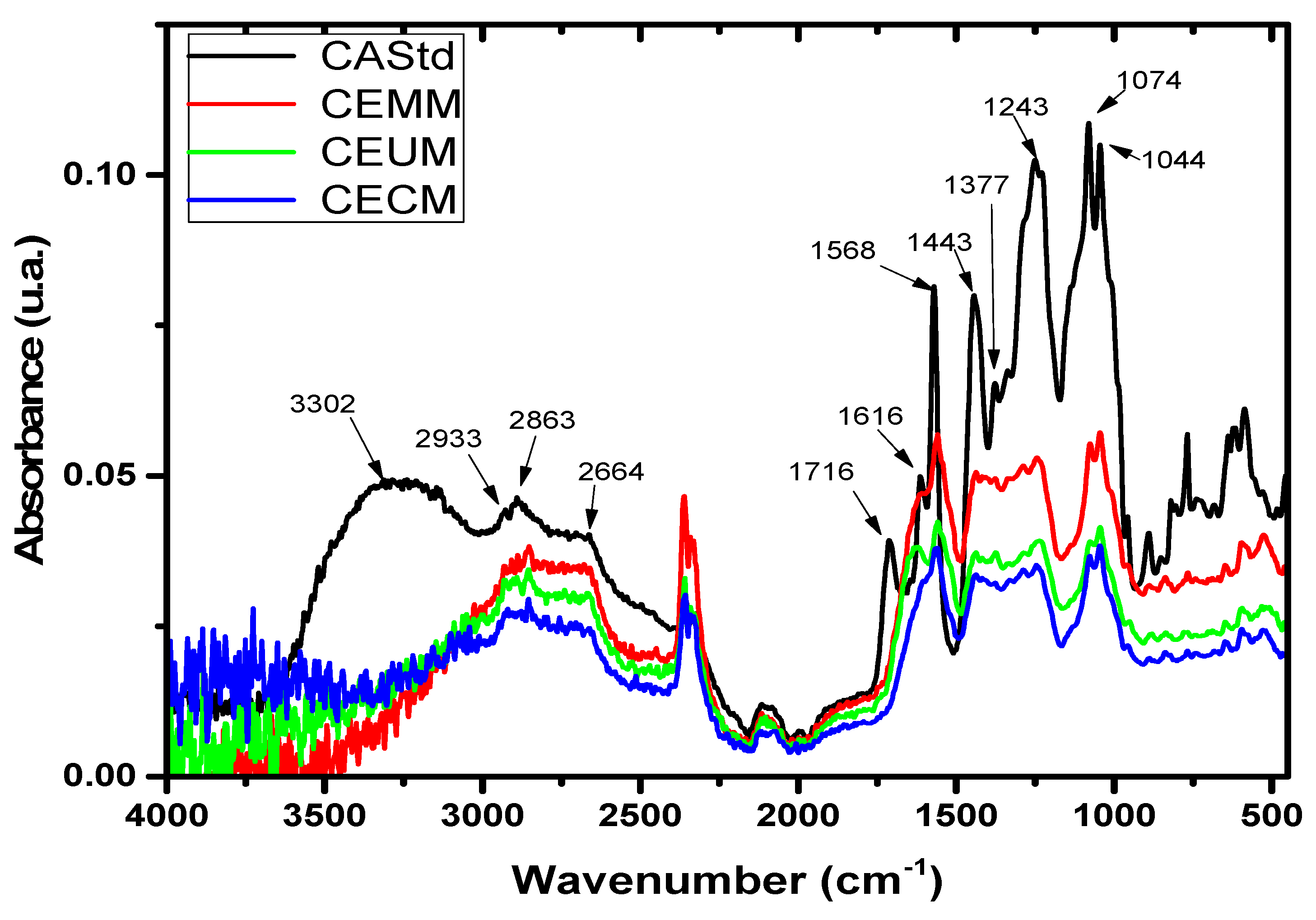
2.6. Infrared Spectra of Carminic Acid Extracts
3. Discussion
4. Materials and Methods
4.1. Cochineal Specimen Data
4.2. Carminic acid Determination
4.3. Carminic Acid Extraction by the Conventional Method
4.4. Extraction Assisted by Ultrasound (EAU)
4.6. Extraction Assisted by Microwave (EAM)
4.7. Carminic Acid Analysis by FTIR Spectroscopy
4.8. Analysis and Quantification of Carminic Acid by UV-vis Spectroscopy
4.9. Experimental Design and Statistical Analysis
5. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gonzalez, Monica & Méndez-Gallegos, Santiago De Jesús & Carnero, Aurelio & Lobo, Gloria & Afonso, Ana. (2002). Optimizing Conditions for the Extraction of Pigments in Cochineals (Dactylopius coccus Costa) Using Response Surface Methodology. Journal of agricultural and food chemistry. 50. 6968-74. [CrossRef]
- Nambela, Lutamyo, Liberato Venant Haule, and Quintino Mgani. 2020. “A Review on Source, Chemistry, Green Synthesis and Application of Textile Colorants.” Journal of Cleaner Production 246: 119036. [CrossRef]
- Pérez Q. 2014. Estudio Técnico para la implementación de una planta procesadora de cochinilla para la obtención del carmín. Thesis. Pontificia Universidad Católica del Perú. Lima, Perú.
- Borges, M. E., Tejera, R. L., Díaz, L., Esparza, P., & Ibáñez, E. (2012). Natural dyes extraction from cochineal (Dactylopius coccus). New extraction methods. Food Chemistry, 132(4), 1855–1860. [CrossRef]
- Ferreyra-Suarez, D., Paredes-Vargas, L., Jafari, S. M., García-Depraect, O., & Castro-Muñoz, R. (2024). Extraction pathways and purification strategies towards carminic acid as natural-based food colorant: A comprehensive review. Advances in Colloid and Interface Science, 323(October 2023). [CrossRef]
- Chemat, F., Vian, M. A., & Cravotto, G. (2012). Green extraction of natural products: Concept and principles. International Journal of Molecular Sciences, 13(7), 8615–8627. [CrossRef]
- Cristianini, M., & Guillén Sánchez, J. S. (2020). Extraction of bioactive compounds from purple corn using emerging technologies: A review. Journal of Food Science, 85(4), 862–869. [CrossRef]
- Bouloumpasi, E., Skendi, A., Christaki, S., Biliaderis, C. C., & Irakli, M. (2024). Optimizing conditions for the recovery of lignans from sesame cake using three green extraction methods: Microwave-, ultrasound- and accelerated-assisted solvent extraction. Industrial Crops and Products, 207(P2), 117770. [CrossRef]
- Biswas, R., Sarkar, A., Alam, M., Roy, M., & Mahdi Hasan, M. M. (2023). Microwave and ultrasound-assisted extraction of bioactive compounds from Papaya: A sustainable green process. Ultrasonics Sonochemistry, 101(May), 106677. [CrossRef]
- Sharma, M., & Dash, K. K. (2022). Microwave and ultrasound assisted extraction of phytocompounds from black jamun pulp: Kinetic and thermodynamics characteristics. Innovative Food Science and Emerging Technologies, 75(December 2021), 102913. [CrossRef]
- Chan, C. H., Yusoff, R., Ngoh, G. C., & Kung, F. W. L. (2011). Microwave-assisted extractions of active ingredients from plants. Journal of Chromatography A, 1218(37), 6213–6225. [CrossRef]
- Bansod, S. P., Parikh, J. K., & Sarangi, P. K. (2023). Pineapple peel waste valorization for extraction of bio-active compounds and protein: Microwave assisted method and Box Behnken design optimization. Environmental Research, 221(October 2022), 115237. [CrossRef]
- Muruganandam L., Krishna, A., Reddy, J., & Nirmala, G. S. (2017). Optimization studies on extraction of phytocomponents from betel leaves. Resource-Efficient Technologies, 3(4), 385–393. [CrossRef]
- Centeno, M. M. (2003). Extracción, estabilización y evaluaciones analíticas del carmín. Tesis de Maestría. Instituto Politécnico Nacional, México. (justificación: información amplia sobre métodos ocupados por industria que por lo regular son secretos industriales).
- Azuola, R., & Vargas Aguilar, P. (2007). Extracción de sustancias asistida por ultrasonido (EUA). Tecnología En Marcha, 20(4), 1. ISSN: 0379-3962.
- Cravotto, C. y Cintas, P. (2006). Power ultrasound in organic synthesis: moving cavitational chemistry from academia to innovative and large-scale applications. Chemical Society. Reviews, 35: 180-196.
- Zhang, W., Boateng, I. D., Zhang, W., Jia, S., Wang, T., & Huang, L. (2022). Effect of ultrasound-assisted ionic liquid pretreatment on the structure and interfacial properties of soy protein isolate. Process Biochemistry, 115(May 2021), 160–168. [CrossRef]
- Cañamares, M. V., García-ramos, J. V., Domingo, C. y Sanchez-Cortes, S. (2006). Surface-enhanced Raman scattering study of the anthraquinone red pigment carminic acid. Vibrational Spectroscopy, 40, 161–167.
- Guo, Yuan & Zheng, Hua & Zhang, Hong & Li-Yi, Ma & Han, Juan & Li, Kun. (2012). Optimization of Combined Microwave-Ultrasonic Wave Extraction of Cochineal Dye by Response Surface Methodology. Applied Mechanics and Materials. 161. 82-87. 10.4028/www.scientific.net/AMM.161.82.
- Anastas, P., Bastlett, L., Kirchoff, M., y Williamson T. (2000). The role of catalysis in the design, development, and implementation of green chemistry. Catalysis Today, 55, 11 -22.
- Robles-Azuna, L. E., & Ochoa-Martínez, L. A. (2012). Disponible en: http://www.redalyc.org/articulo.oa?id=81325441002. Revista Iberoamericana de Tecnología Postcosecha, 13(2), 109–122.
- Pájaro Nerlis Paola, & Olivero Jesús. (2011). Química Verde: Un nuevo reto. Ciencia e Ingeniería Neogranadina, 21(2), 169–182. http://www.redalyc.org/articulo.oa?id=91123440009.
- Flores-Hernández, Arnoldo, Murillo-Amador, Bernardo, Rueda-Puente, Edgar Omar, Salazar-Torres, José Cruz, García-Her nández, José Luis, & Troyo-Diéguez, Enrique. (2006). Reproducción de cochinilla silvestre Dactylopius opuntiae (Homóptera: Dactylopiidae). Revista mexicana de biodiversidad, 77(1), 97-102. Recuperado en 18 de diciembre de 2023, de http://www.scielo.org.mx/scielo.php?script=sci_arttext&pid=S1870-34532006000100011&lng=es&tlng=es.
- Dong, X., Wang, J., & Raghavan, V. (2020). Effects of high-intensity ultrasound processing on the physiochemical and allergenic properties of shrimp. Innovative Food Science and Emerging Technologies, 65(July), 102441. [CrossRef]
- Food Chemicals Codex. 1981. Third Edition. National Research Council. Washington, DC: The National Academies Press. [CrossRef]
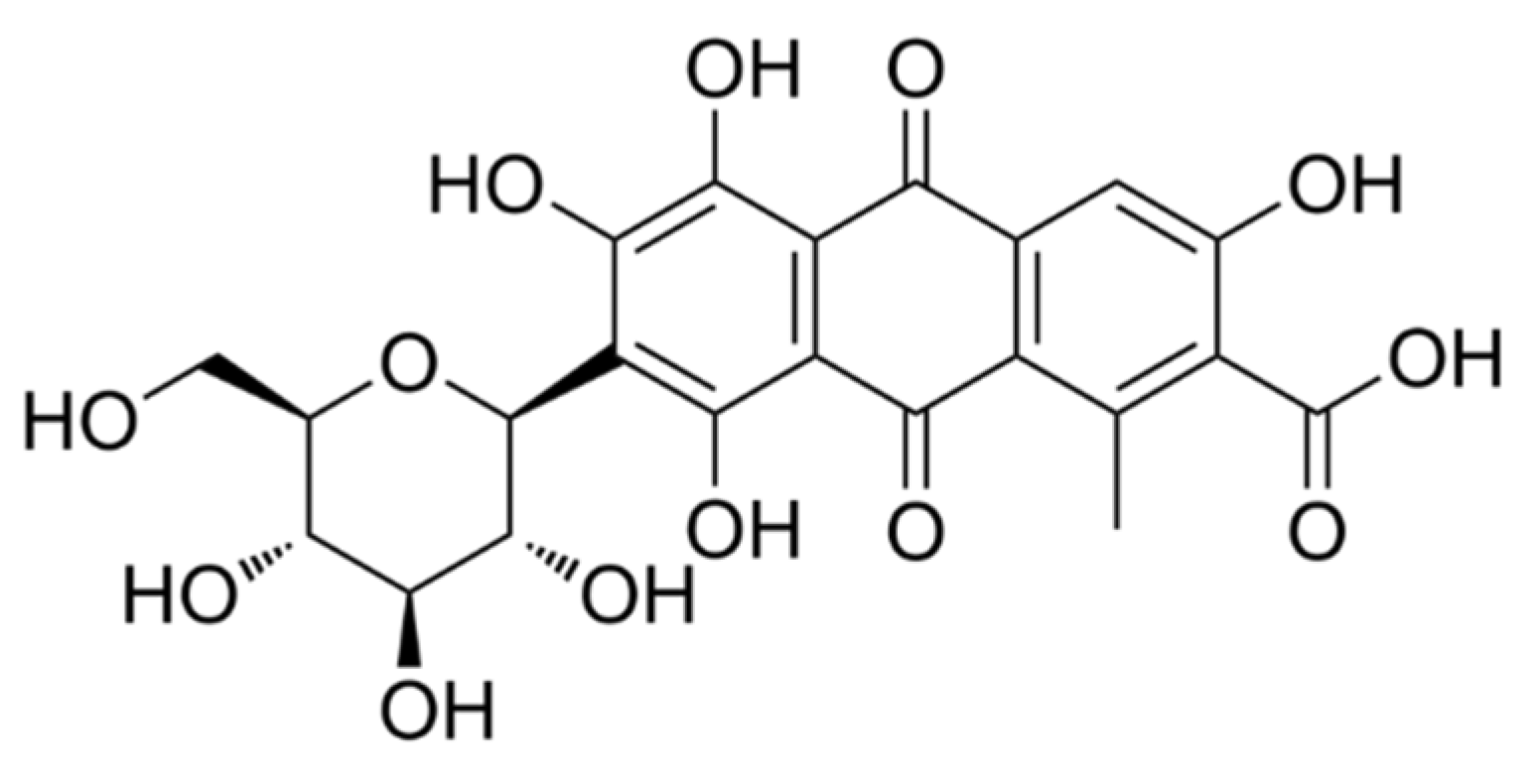
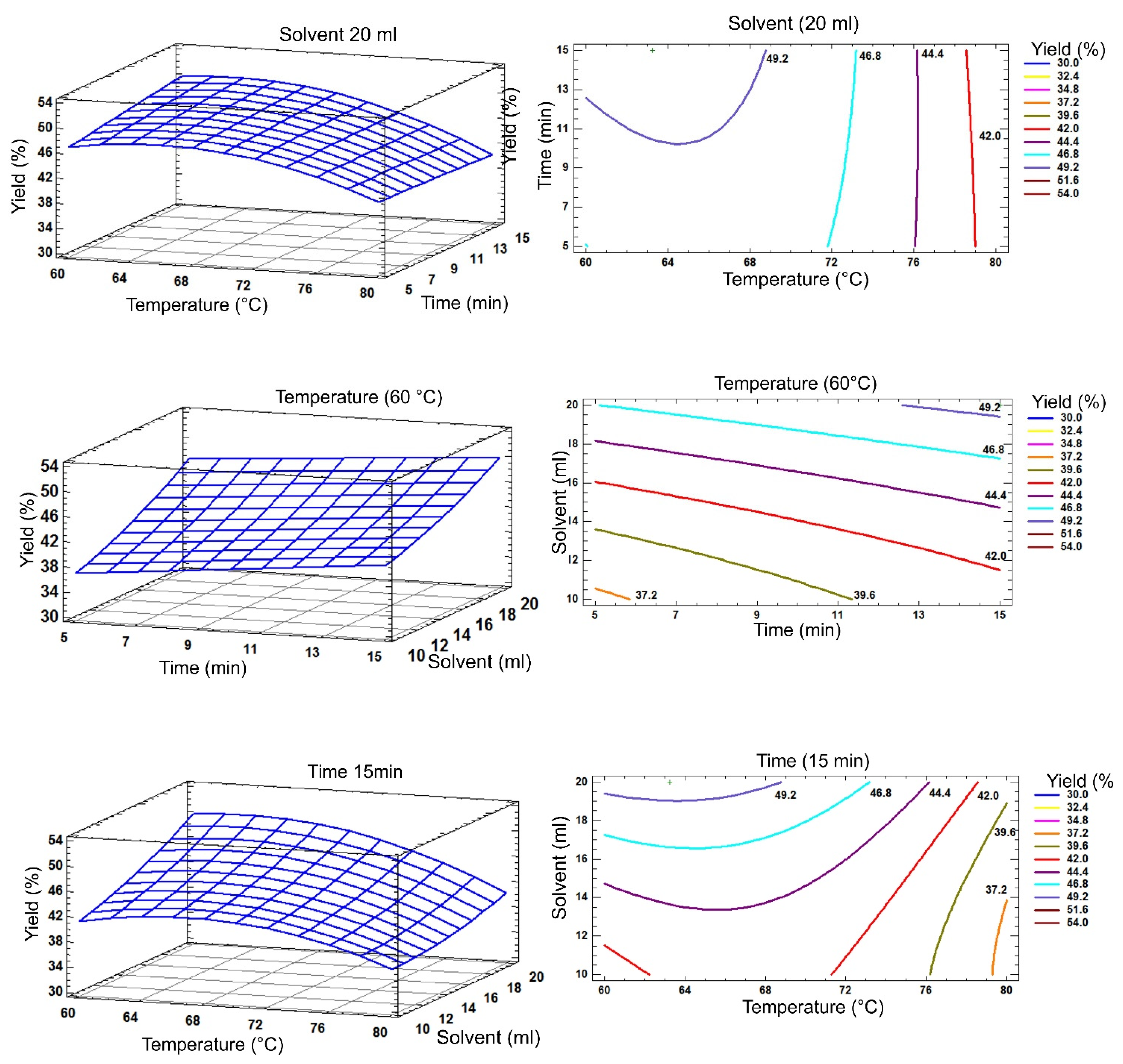
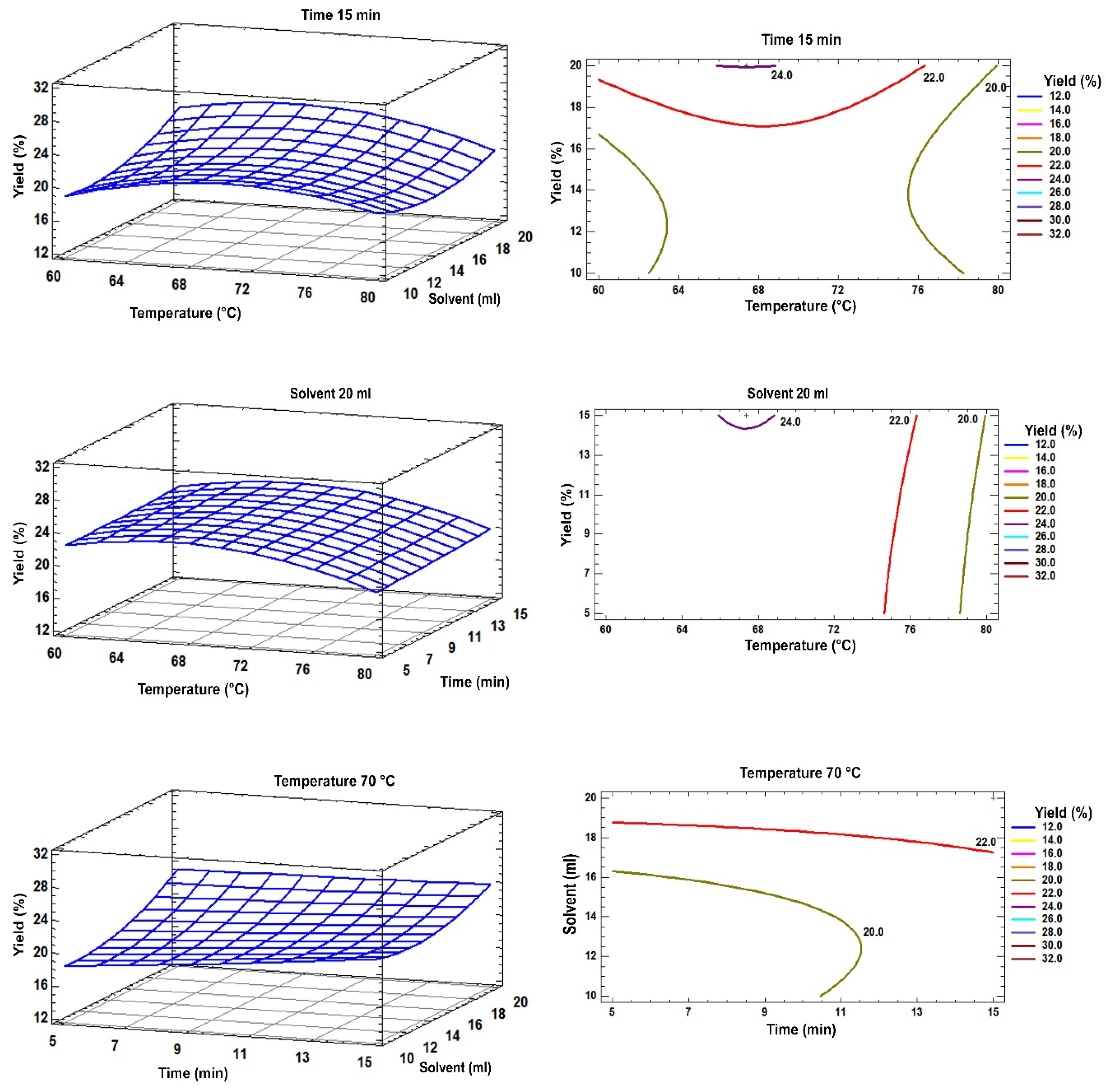
| Independent Variables | Levels codification | |||
| -1 | 0 | 1 | ||
| Temperature (ºC) | X1 | 60 | 70 | 80 |
| Irradiation time (min) | X2 | 5 | 10 | 15 |
| Solvent ratio (mL) | X3 | 10 | 15 | 20 |
| Run | X1 Temperature (o C) |
X2 Irradiation time (min) |
X3 Solvent volume (mL) |
Yield of cochineal extract (%) by Ultrasound |
Yield of cochineal extract (%) by Microwave |
| 1 | 80 | 10 | 20 | 48.225 | 30.922 |
| 2 | 70 | 5 | 10 | 35.563 | 40.478 |
| 3 | 60 | 5 | 15 | 43.021 | 30.078 |
| 4 | 70 | 15 | 20 | 39.790 | 40.889 |
| 5 | 70 | 5 | 20 | 38.718 | 39.578 |
| 6 | 80 | 5 | 15 | 44.286 | 39.400 |
| 7 | 60 | 15 | 15 | 40.081 | 33.789 |
| 8 | 80 | 15 | 15 | 38.030 | 32.789 |
| 9 | 60 | 10 | 10 | 40.413 | 35.147 |
| 10 | 70 | 15 | 10 | 40.946 | 37.300 |
| 11 | 60 | 10 | 20 | 42.029 | 38.333 |
| 12 | 80 | 10 | 10 | 39.713 | 35.611 |
| 13 | 70 | 10 | 15 | 49.211 | 35.667 |
| 14 | 70 | 10 | 15 | 47.752 | 38.622 |
| 15 | 70 | 10 | 15 | 47.432 | 35.756 |
| ANOVA of ultrasound | ANOVA of microwave | |||||||
| Source | S.M. | C.M. | F-value | P-value | SM | C.M. | F-value | P-value |
| A: Temperature | 159.24 | 159.24 | 9.79 | 0.0036 | 20.47 | 20.47 | 3.08 | 0.0886 |
| B: Time | 19.84 | 19.84 | 1.22 | 0.2773 | 1.337 | 1.337 | 0.20 | 0.6567 |
| C: Solvent | 284.68 | 284.68 | 17.51 | 0.0002 | 376.83 | 376.83 | 56.68 | 0.0000 |
| AA | 137.88 | 137.88 | 8.48 | 0.0064 | 3.698 | 3.69 | 0.56 | 0.4610 |
| AB | 10.87 | 10.87 | 0.67 | 0.4192 | 3.484 | 3.48 | 0.52 | 0.4742 |
| AC | 18.60 | 18.605 | 1.14 | 0.2925 | 10.64 | 10.640 | 1.60 | 0.2147 |
| BB | 0.047 | 0.047 | 0.00 | 0.9572 | 0.092 | 0.093 | 0.01 | 0.0906 |
| BC | 0.977 | 0.979 | 0.06 | 0.8076 | 20.80 | 20.803 | 3.13 | 0.0862 |
| CC | 8.39 | 8.393 | 0.52 | 0.4775 | 8.86 | 8.860 | 1.33 | 0.2566 |
| Total error | 536.52 | 16.258 | 219.41 | 6.648 | ||||
| Total (corr.) | 1309.0 | 670.55 | ||||||
| Technique | Yield of Extraction (%) |
Extract Efficiency (mg/min) | Carminic Acid Yield (%) | Carminic Acid Efficiency (mg/min) |
| Conventional Method | 31.9 | 3.5 | 17.8 | 2.0 |
| Pressurized Liquids |
42.4 | 14.1 | NR | NR |
| Supercritical Fluids | 39.4 | 1.6 | NR | NR |
| Microwaves | 41 | 27.3 | 18.0 | 36.0 |
| Ultrasound | 47 | 31.3 | 26.3 | 17.5 |
| Absorption bands reported by Cañamares (REF) (cm-1) |
Absorption bands reported in this work (cm-1) |
Assignments |
| 3423 | 3302 | ν(OH) |
| 2917 | 2933 | ν(CH3)/νGlu(CH) |
| 1716 | 1716 | νacid(C=O) |
| 1617 | 1616 | ν(C=O)/νI(C–C) |
| 1572 | 1568 | νI(C–C)/δ(C5OH)/δ(CH) |
| 1376 | 1377 | δGlu(CH)/δGlu(COH)/δ(COH) |
| 1255 | 1243 | ν(C–C)/δacid(COH)/δ(COH)/δGlu(CH) |
| 1083 | 1074 | νGlu(C–O)/δGlu(COH) |
| 1045 | 1044 | νGlu(C–C)/δGlu(COH) |
| 820 | 813 | γ(COH) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




