Submitted:
15 October 2024
Posted:
16 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Culture, Virus and Reagents
2.2. Viral Stock and Titration Assay
2.3. Infection Experiments
2.4. RNA Extraction and Reverse Transcription Assay
2.5. PCR Assay
2.6. qPCR Assay
2.7. Inhibition of PERK Pathway
2.8. DNA Fragmentation Assay
2.9. Statistical Analysis
3. Results
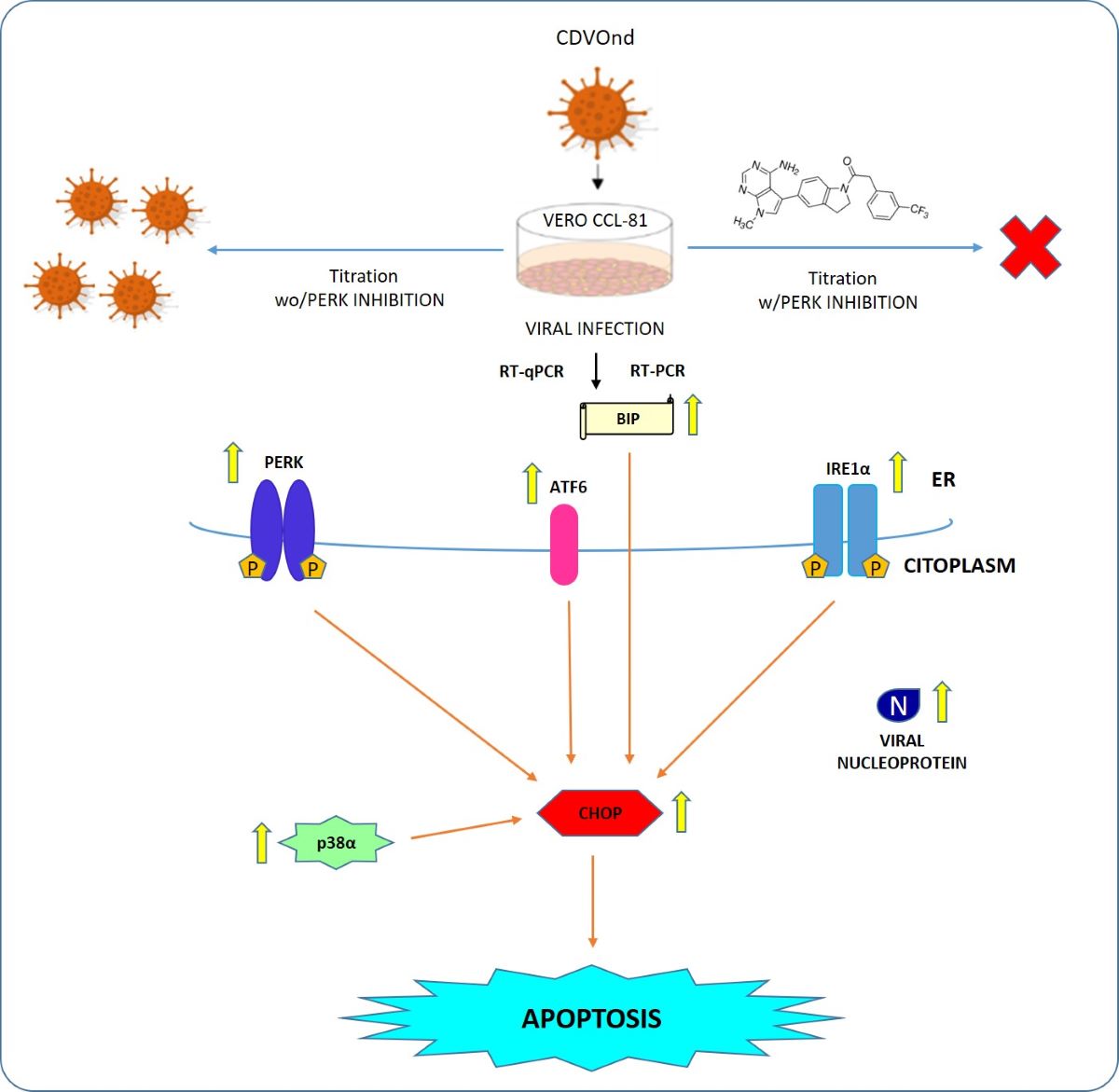
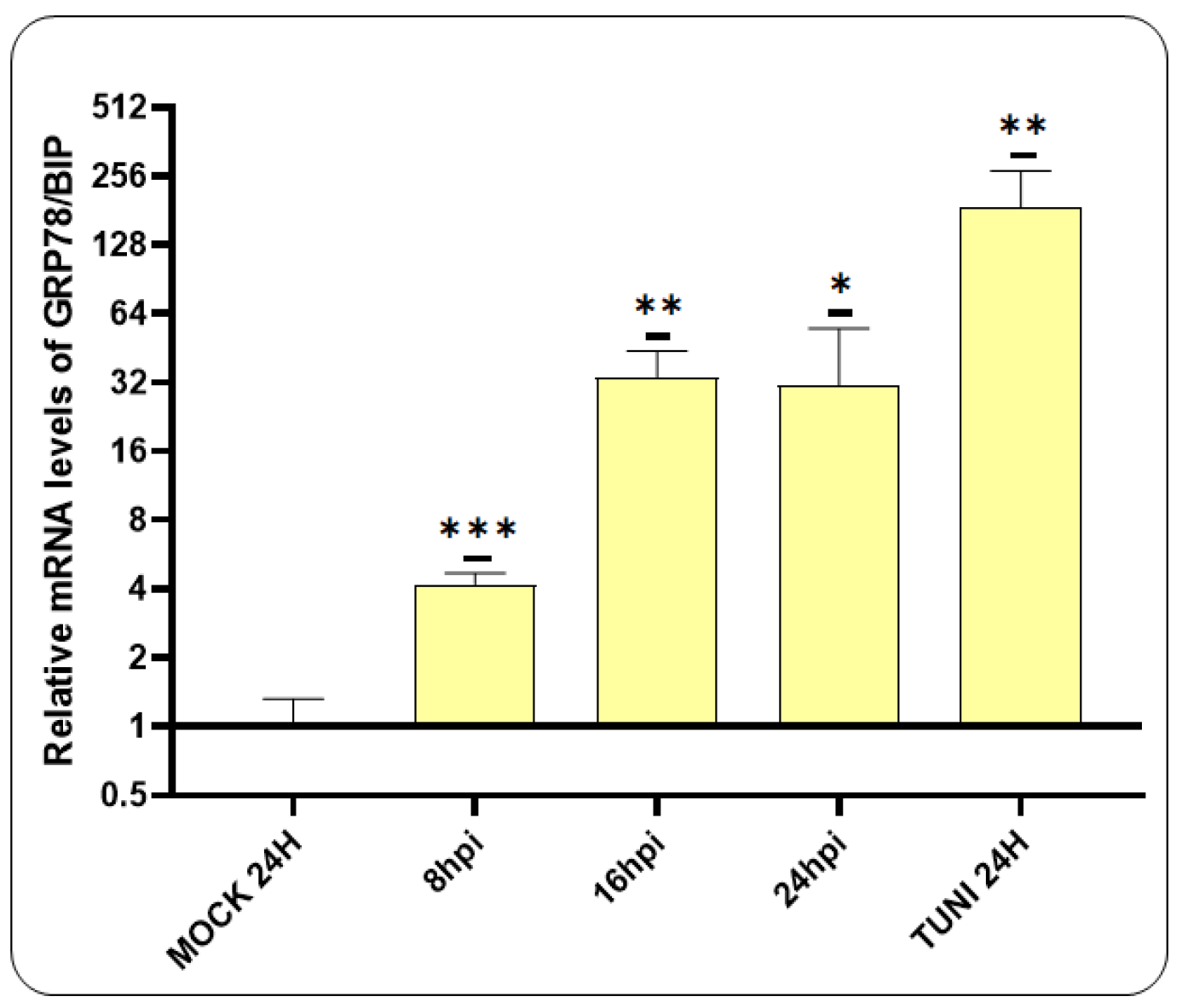
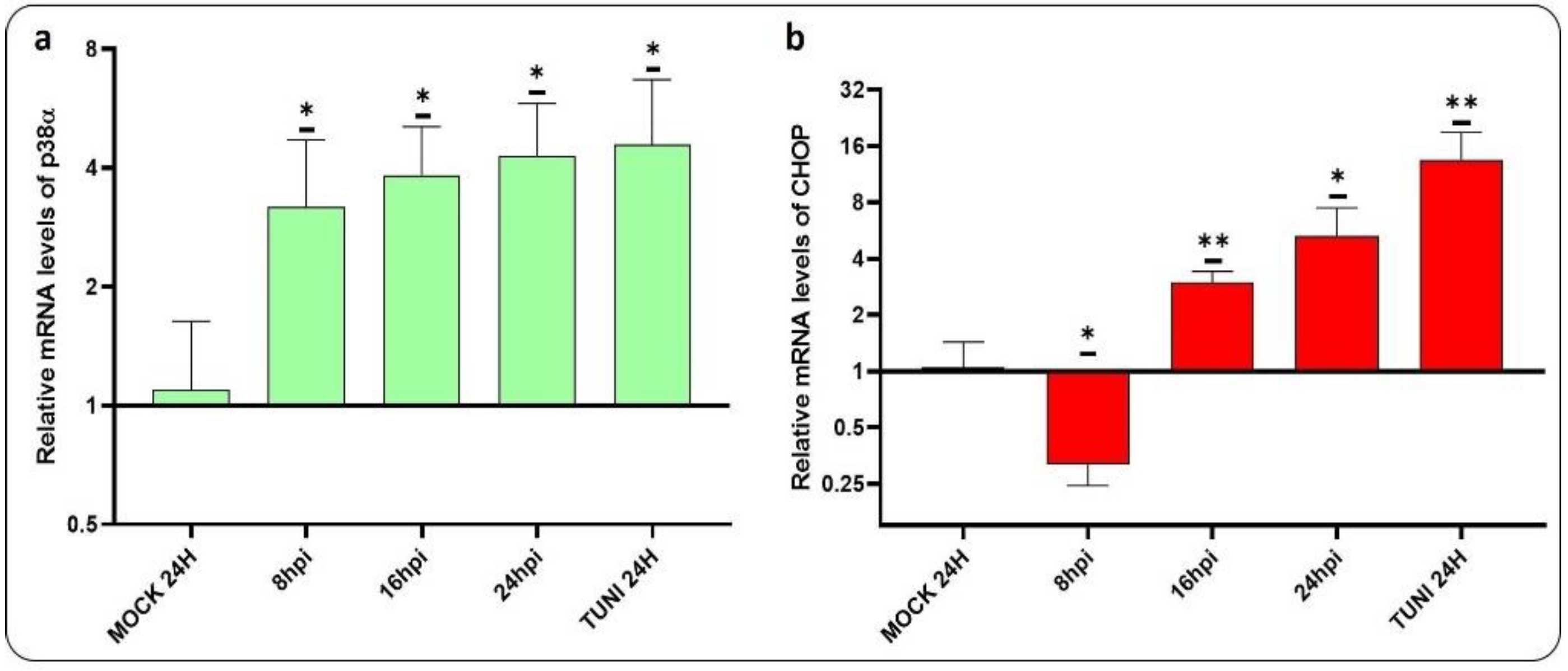
3.1. ER Stress Induced by CDV Infection
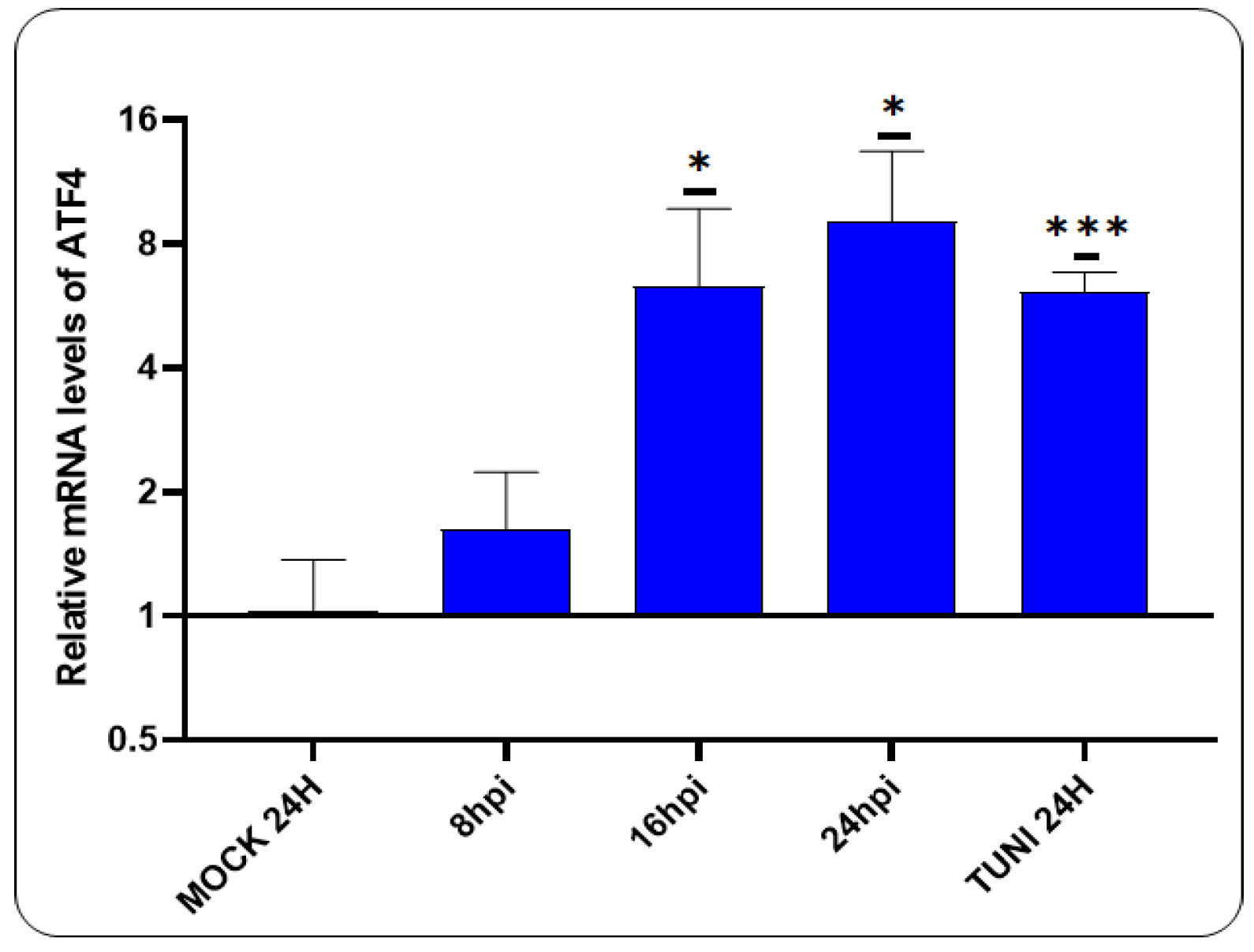
3.2. CDV Infection Activates PERK Pathway
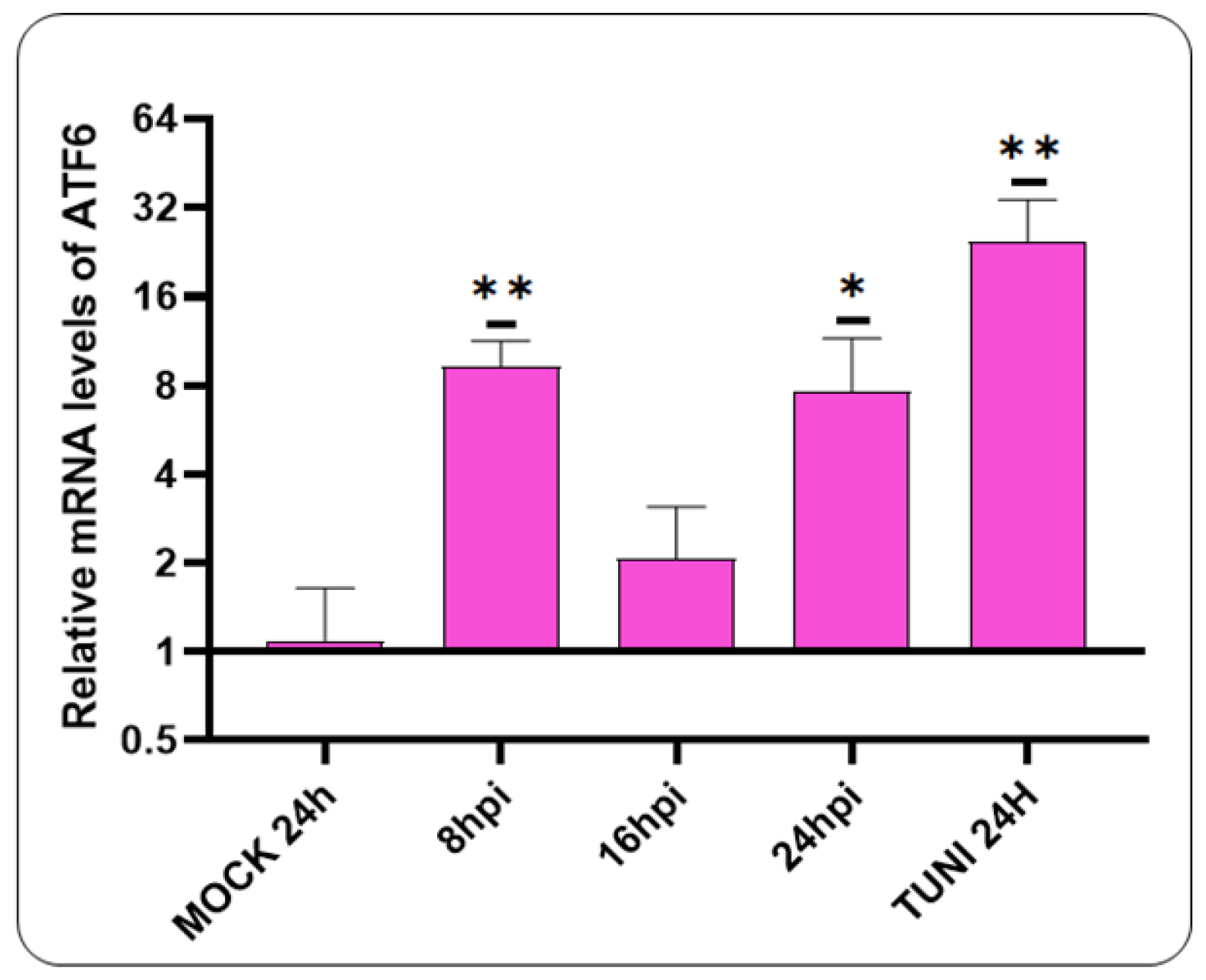
3.3. Activation of ATF6 Pathway after CDV Infection
3.4. IRE1⍺ Pathway Is Activated by CDVOnd Infection
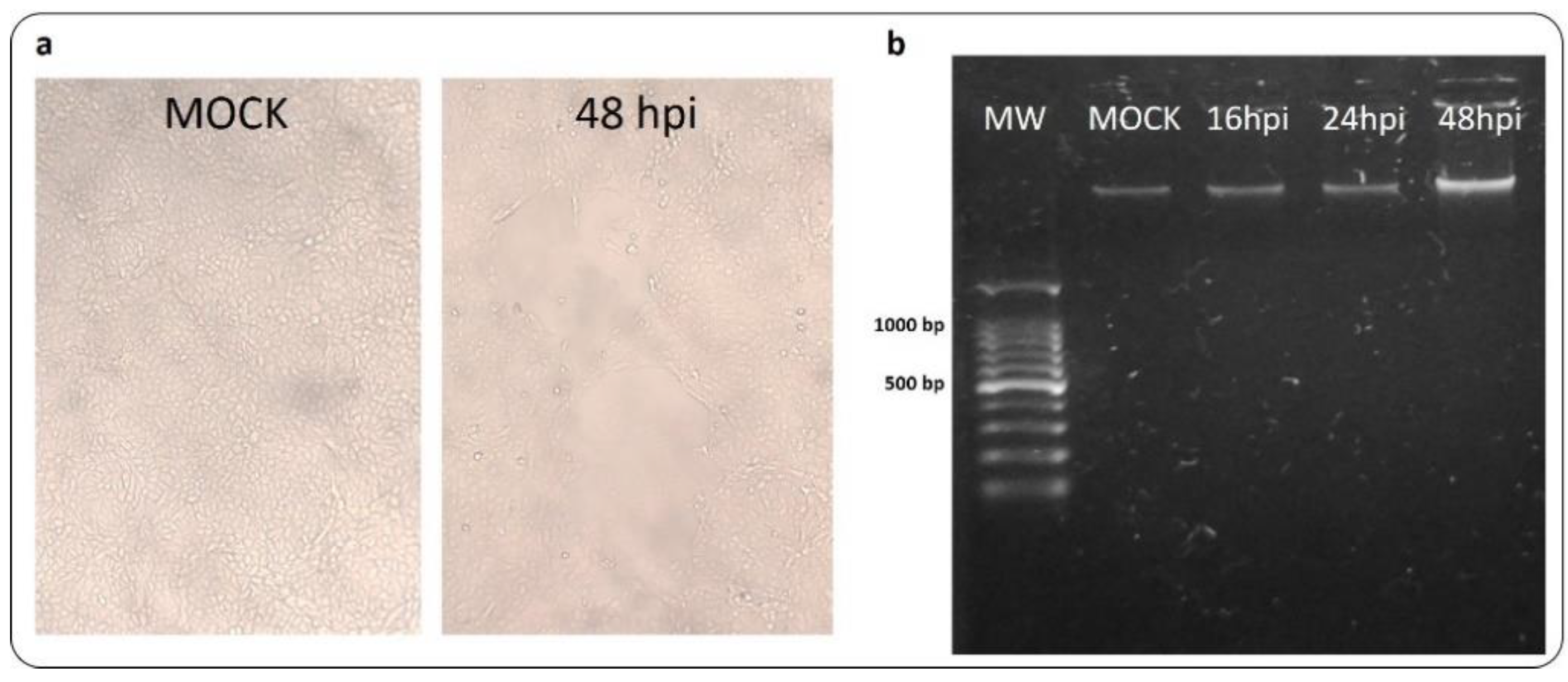
3.5. CDVOnd Infection Induces Apoptosis in VERO CCL-81
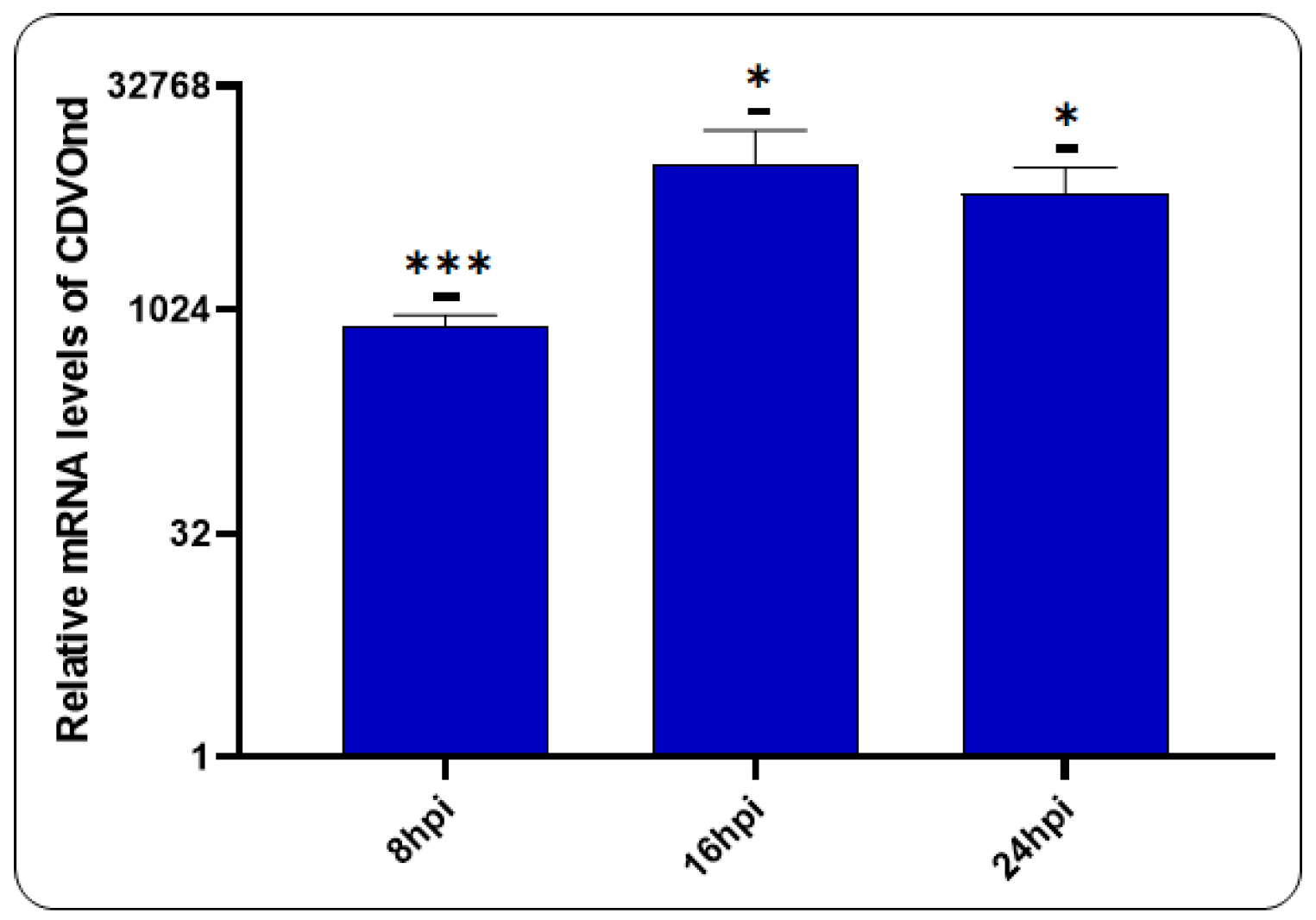
3.6. Viral Replication after CDVOnd Infection
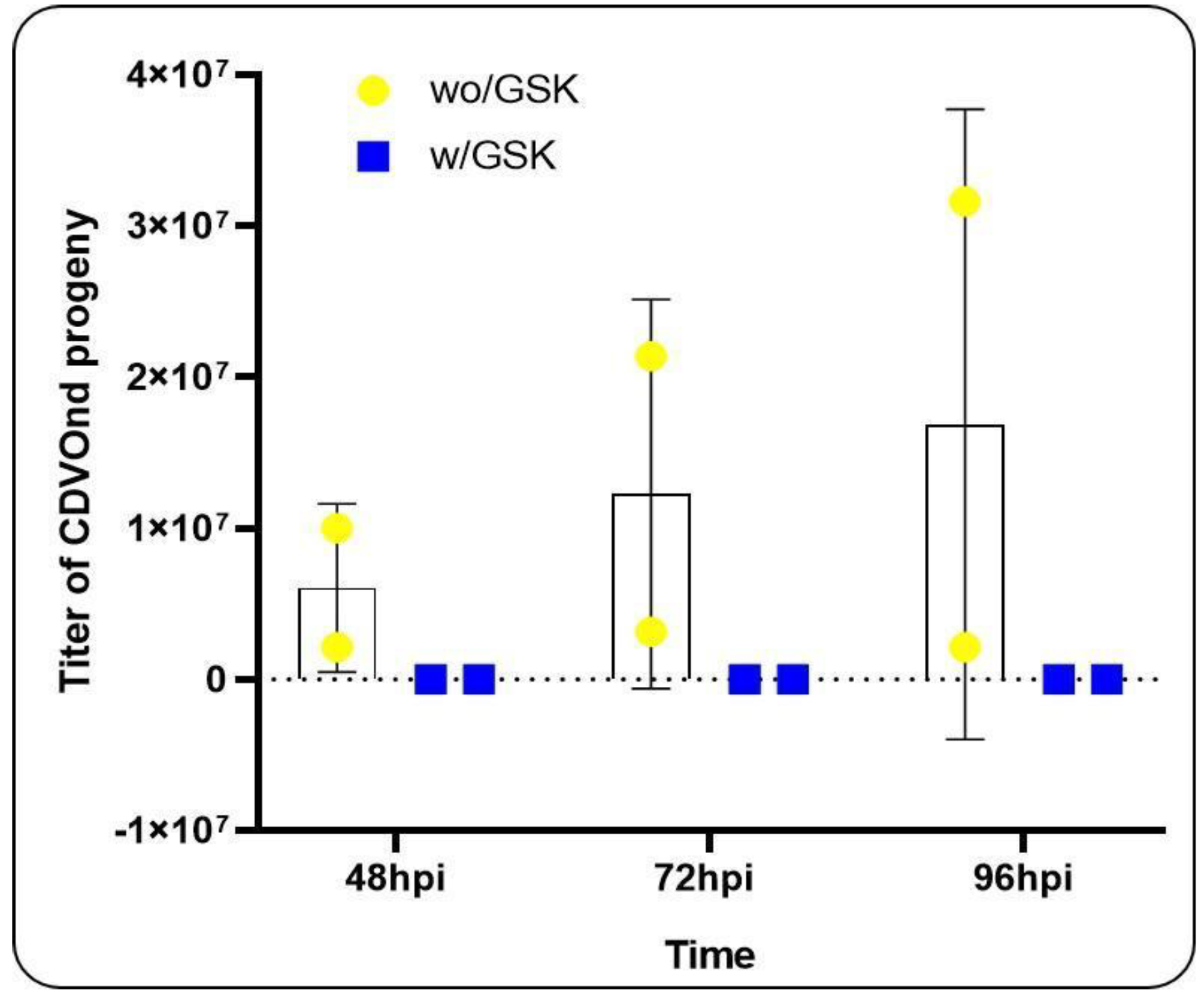
3.7. PERK Inhibition and Viral Progeny
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- da Fontoura Budaszewski, R.; von Messling, V. Morbillivirus Experimental Animal Models: Measles Virus Pathogenesis Insights from Canine Distemper Virus. Viruses. 2016, 8, 274. [Google Scholar] [CrossRef]
- Martella, V.; Elia, G.; Buonavoglia, C. Canine distemper virus. Vet. Clin. North. Am. Small. Anim. Pract. 2008, 38, 787-viii. [Google Scholar] [CrossRef] [PubMed]
- Appel, M.J.; Summers, B.A. Pathogenicity of morbilliviruses for terrestrial carnivores. Vet. Microbiol. 1995, 44, 187–191. [Google Scholar] [CrossRef]
- Deem, S.L.; Spelman, L.H.; Yates, R.A.; Montali, R.J. Canine distemper in terrestrial carnivores: a review. J. Zoo. Wildl. Med. 2000, 31, 441–451. [Google Scholar] [CrossRef]
- Li, S.; Kong, L.; Yu, X. The expanding roles of endoplasmic reticulum stress in virus replication and pathogenesis. Crit. Rev. Microbiol. 2015, 41, 150–164. [Google Scholar] [CrossRef]
- He, B. Viruses, endoplasmic reticulum stress, and interferon responses. Cell Death Differ. 2006, 13(3), 393–403. [Google Scholar] [CrossRef]
- Morishima, N.; Nakanishi, K.; Takenouchi, H.; Shibata, T.; Yasuhiko, Y. An endoplasmic reticulum stress-specific caspase cascade in apoptosis. Cytochrome c-independent activation of caspase-9 by caspase-12. J. Biol. Chem. 2002, 277, 34287–34294. [Google Scholar] [CrossRef]
- Szegezdi, E.; Fitzgerald, U.; Samali, A. Caspase-12 and ER-stress-mediated apoptosis: the story so far. Ann. N. Y. Acad. Sci. 2003, 1010, 186–194. [Google Scholar] [CrossRef]
- Zhang, Q.; Liu, J.; Chen, S. Liu, J.; Liu, L.; Liu, G.; Wang, F.; Jiang, W.; Zhang, C.; Wang, S.; Yuan, X. Caspase-12 is involved in stretch-induced apoptosis mediated endoplasmic reticulum stress. Apoptosis. 2016, 21, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Hetz, C.; Zhang, K.; Kaufman, R.J. Mechanisms, regulation and functions of the unfolded protein response. Nat. Rev. Mol. Cell Biol. 2020, 21, 421–438. [Google Scholar] [CrossRef]
- Sano, R.; Reed, J.C. ER stress-induced cell death mechanisms. Biochim. Biophys. Acta. 2013, 1833, 3460–3470. [Google Scholar] [CrossRef]
- Guo, A.; Lu, C. Canine distemper virus causes apoptosis of Vero cells. J. Vet. Med. B. Infect. Dis. Vet. Public Health. 2000, 47, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Kajita, M.; Katayama, H.; Murata, T.; Kai, C.; Hori, M.; Ozaki, H. Canine distemper virus induces apoptosis through caspase-3 and -8 activation in vero cells. J. Vet. Med. B. Infect. Di.s Vet. Public Health. 2006, 53, 273–277. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Deka, D. Studies on the molecular mechanisms involved in apoptosis triggered by Canine Distemper Virus (CDV). I.J.B.B. 2023, 60, 525–544. [Google Scholar] [CrossRef]
- Li, P.; Wang, J.; Chen, G.; Zhang, X.; Lin, D.; Zhou, Y.; Yu, Y.; Liu, W.; Zhang, D. Oncolytic activity of canine distemper virus in canine mammary tubular adenocarcinoma cells. Vet. Comp. Oncol. 2019, 17, 174–183. [Google Scholar] [CrossRef]
- Brunner, J.M.; Plattet, P.; Doucey, M.A.; Rosso, L.; Curie, T.; Montagner, A.; Wittek, R.; Vandelvelde, M.; Zurbriggen, A.; Hirling, H.; Desvergne, B. Morbillivirus glycoprotein expression induces ER stress, alters Ca2+ homeostasis and results in the release of vasostatin. PLoS One. 2012, 7, e32803. [Google Scholar] [CrossRef]
- Wang, W.; Bi, Z.; Liu, Y.; Xia, X.; Qian, J.; Tan, Y.; Zhao, J.; Song, S. The H protein of attenuated canine distemper virus is degraded via endoplasmic reticulum-associated protein degradation. Front. Vet. Sci. 2023, 10, 1214318. [Google Scholar] [CrossRef] [PubMed]
- Virgin, H.W.; Wherry, E.J.; Ahmed, R. Redefining chronic viral infection. Cell. 2009, 138, 30–50. [Google Scholar] [CrossRef]
- Wang, Y.; Li, J.R.; Sun, M.X.; Ni, B.; Huan, C.; Huang, L.; Li, C.; Fan, H.J.; Ren, X.F.; Mao, X. Triggering unfolded protein response by 2-Deoxy-D-glucose inhibits porcine epidemic diarrhea virus propagation. Antiviral Res. 2014, 106, 33–41. [Google Scholar] [CrossRef]
- Wolfson, J.J.; May, K.L.; Thorpe, C.M.; Jandhyala, D.M.; Paton, J.C.; Paton, A.W. Subtilase cytotoxin activates PERK, IRE1 and ATF6 endoplasmic reticulum stress-signalling pathways. Cell. Microbiol. 2008, 10, 1775–1786. [Google Scholar] [CrossRef]
- Zhang, C.; Hu, J.; Wang, X.; Wang, Y.; Guo, M.; Zhang, X.; Wu, Y. Avian reovirus infection activate the cellular unfold protein response and induced apoptosis via ATF6-dependent mechanism. Virus Res. 2021, 297, 198346. [Google Scholar] [CrossRef]
- Echavarría-Consuegra, L.; Cook, G.M.; Busnadiego, I.; Lefèvre, C.; Keep, S.; Brown, K.; Doyle, N.; Dowgier, G.; Franaszek, K.; Moore, N.A.; Siddell, S.G.; Bickerton, E.; Hale, B.G.; Firth, A.E.; Brierley, I.; Irigoyen, N. Manipulation of the unfolded protein response: A pharmacological strategy against coronavirus infection. PLoS Pathog. 2021, 17, e1009644. [Google Scholar] [CrossRef]
- Cheng, Y.; Sun, F.; Wang, L.; Gao, M.; Xie, Y.; Sun, Y.; Liu, H.; Yuan, Y.; Yi, W.; Huang, Z.; Yan, H.; Peng, K.; Wu, Y.; Cao, Z. Virus-induced p38 MAPK activation facilitates viral infection. Theranostics. 2020, 10, 12223–12240. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Yan, X.; Chai, X.; Zhang, H., Zhao, J.; Wen, Y.; Wu, W. Differentiation of canine distemper virus isolates in fur animals from various vaccine strains by reverse transcription-polymerase chain reaction-restriction fragment length polymorphism according to phylogenetic relations in china. Virol. J. 2011, 8, 85. [CrossRef]
- Majtnerová, P.; Roušar, T. An overview of apoptosis assays detecting DNA fragmentation. Mol. Biol. Rep. 2018, 45, 1469–1478. [Google Scholar] [CrossRef] [PubMed]
- Shang, J.; Lehrman, M.A. Discordance of UPR signaling by ATF6 and Ire1p-XBP1 with levels of target transcripts. Biochem. Biophys. Res. Commun. 2004, 317, 390–396. [Google Scholar] [CrossRef]
- Choi, J.A.; Song, C.H. Insights Into the Role of Endoplasmic Reticulum Stress in Infectious Diseases. Front. Immunol. 2020, 10, 3147. [Google Scholar] [CrossRef]
- Park, S.W.; Ozcan, U. Potential for therapeutic manipulation of the UPR in disease. Semin. Immunopathol. 2013, 35, 351–373. [Google Scholar] [CrossRef]
- Ha, D.P.; Van Krieken, R.; Carlos, A.J.; Lee, A.S. The stress-inducible molecular chaperone GRP78 as potential therapeutic target for coronavirus infection. J. Infect. 2020, 81, 452–482. [Google Scholar] [CrossRef] [PubMed]
- Shin, J.; Toyoda, S.; Fukuhara, A.; Shimomura, I. GRP78, a novel host factor for SARS-coV-2: the emerging roles in COVID-19 related to metabolic risk factors. Biomedicines. 2022, 10, 1995. [Google Scholar] [CrossRef]
- Qiao, D.; Skibba, M.; Xu, X.; Garofalo, R.P.; Zhao, Y.; Brasier, A.R. Paramyxovirus replication induces the hexosamine biosynthetic pathway and mesenchymal transition via the IRE1α-XBP1s arm of the unfolded protein response. Am. J. Physiol. Lung Cell. Mol. Physiol. 2021, 321, L576–L594. [Google Scholar] [CrossRef]
- Han, C.; Xie, Z.; Lv, Y.; Liu, D.; Chen, R. Direct interaction of the molecular chaperone GRP78/BiP with the Newcastle disease virus hemagglutinin-neuraminidase protein plays a vital role in viral attachment to and infection of culture cells. Front. Immunol. 2023, 14, 1259237. [Google Scholar] [CrossRef] [PubMed]
- Watowich, S.S.; Morimoto, R.I.; Lamb, R.A. Flux of the paramyxovirus hemagglutinin-neuraminidase glycoprotein through the endoplasmic reticulum activates transcription of the GRP78-BiP gene. J. Vir. 1991, 65, 3590–3597. [Google Scholar] [CrossRef] [PubMed]
- Kohli, E.; Causse, S.; Baverel, V.; Dubrez, L.; Borges-Bonan, N.; Demidov, O.; Garrido, C. Endoplasmic Reticulum Chaperones in Viral Infection: Therapeutic Perspectives. Microbiol. Mol. Biol. Rev. 2021, 85, e0003521. [Google Scholar] [CrossRef]
- Zhang, R.; Lin, H.; You, Q.; Zhang, Z.; Bai, L.; Chen, F.; Zhu, X.; Zhang, Z.; Li, Y. Peste des Petits Ruminants Virus Upregulates STING to Activate ATF6-Mediated Autophagy. J. Virol. 2022, 96, e0137522. [Google Scholar] [CrossRef]
- Bernales, S.; Papa, F.R.; Walter, P. Intracellular signaling by the unfolded protein response. Annu. Rev. Cell Dev. Biol. 2006, 22, 487–508. [Google Scholar] [CrossRef]
- Yoshida, H.; Matsui, T.; Yamamoto, A.; Okada, T.; Mori, K. XBP1 mRNA is induced by ATF6 and spliced by IRE1 in response to ER stress to produce a highly active transcription factor. Cell. 2001, 107, 881–891. [Google Scholar] [CrossRef]
- Hu, H.; Tian, M.; Ding, C.; Yu, S. The C/EBP Homologous Protein (CHOP) Transcription Factor Functions in Endoplasmic Reticulum Stress-Induced Apoptosis and Microbial Infection. Front. Immunol. 2019, 9, 3083. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.J.; Ryu, S.W.; Song, B.J. JNK- and p38 kinase-mediated phosphorylation of Bax leads to its activation and mitochondrial translocation and to apoptosis of human hepatoma HepG2 cells. J. Biol. Chem. 2006, 281, 21256–21265. [Google Scholar] [CrossRef]
- Lumley, E.C.; Osborn, A.R.; Scott, J.E.; Scholl, A.G.; Mercado, V.; McMahan, Y.T.; Coffman, Z.G.; Brewster, J.L. Moderate endoplasmic reticulum stress activates a PERK and p38-dependent apoptosis. Cell Stress Chaperones. 2017, 22, 43–54. [Google Scholar] [CrossRef]
- Guo, X.; Meng, Y.; Sheng, X.; Guan, Y.; Zhang, F.; Han, Z.; Kang, Y.; Tai, G.; Zhou, Y.; Cheng, H. Tunicamycin enhances human colon cancer cells to TRAIL-induced apoptosis by JNK-CHOP-mediated DR5 upregulation and the inhibition of the EGFR pathway. Anticancer Drugs. 2017, 28, 66–74. [Google Scholar] [CrossRef]
- Fujita, E.; Kouroku, Y.; Jimbo, A.; Isoai, A.; Maruyama, K.; Momoi, T. Caspase-12 processing and fragment translocation into nuclei of tunicamycin-treated cells. Cell Death Differ. 2002, 9, 1108–1114. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Li, X.; Wang, Y.; Wang, H.; Huang, C.; Li, J. Endoplasmic reticulum stress-induced hepatic stellate cell apoptosis through calcium-mediated JNK/P38 MAPK and Calpain/Caspase-12 pathways. Mol. Cell. Biochem. 2014, 394, 1–12. [Google Scholar] [CrossRef] [PubMed]

| Use | Gene | Sequence (5’ to 3’) | Size (bp) | Reference |
|---|---|---|---|---|
| House- | GAPDH-Fw | AGGTCGGAGTCAACGGATTT | 112 | [19] |
| keeping | GAPDH-Rv | TAGTTGAGGTCAATGAAGGG | 112 | [19] |
| ER stress | BIP-Fw | ACCGCTGAGGCTTATTGGG | 147 | [19] |
| activation | BIP-Rv | TGCCGTAGGCTCGTTGATG | 147 | [19] |
| PERK | ATF4-Fw | CCAACAACAGCAAGGAGGAT | 143 | [20] |
| pathway | ATF4-Rv | GTGTCATCCAACGTGGTCAG | 143 | [20] |
| ATF6 | ATF6-Fw | CGAATAGCCCAGTGAA | 180 | [21] |
| pathway | ATF6-Rv | ATCTCGCCTCTAACCC | 180 | [21] |
| Apoptosis | CHOP-Fw | ACCAAGGGAGAACCAGGAAACG | 201 | [22] |
| Apoptosis | CHOP-Rv | TCACCATTCGGTCAATCAGAGC | 201 | [22] |
| MAPK | p38α-Fw | GCCCAAGCCCTTGCACAT | 156 | [23] |
| pathway | p38α-Rv | TGGTGGCACAAAGCTGATGAC | 156 | [23] |
| Viral | CDV-N-Fw | TTCTGAGGCAGATGAGTTCTTC | 829 | [24] |
| genome | CDV-N-Rv | CTTGGATGCTATTTCTGACACT | 829 | [24] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




