Submitted:
14 October 2024
Posted:
15 October 2024
You are already at the latest version
Abstract
Keywords:
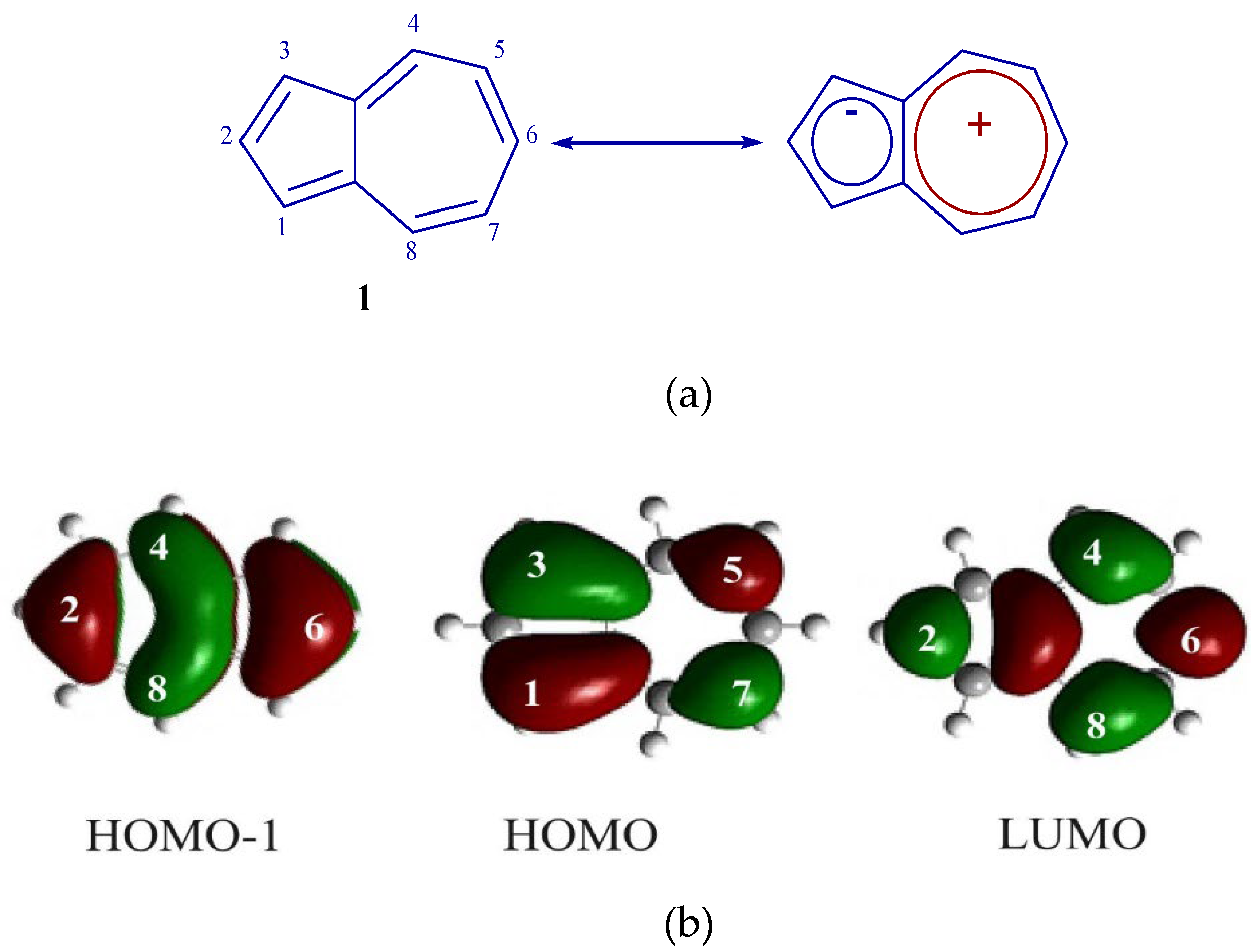
1. Introduction
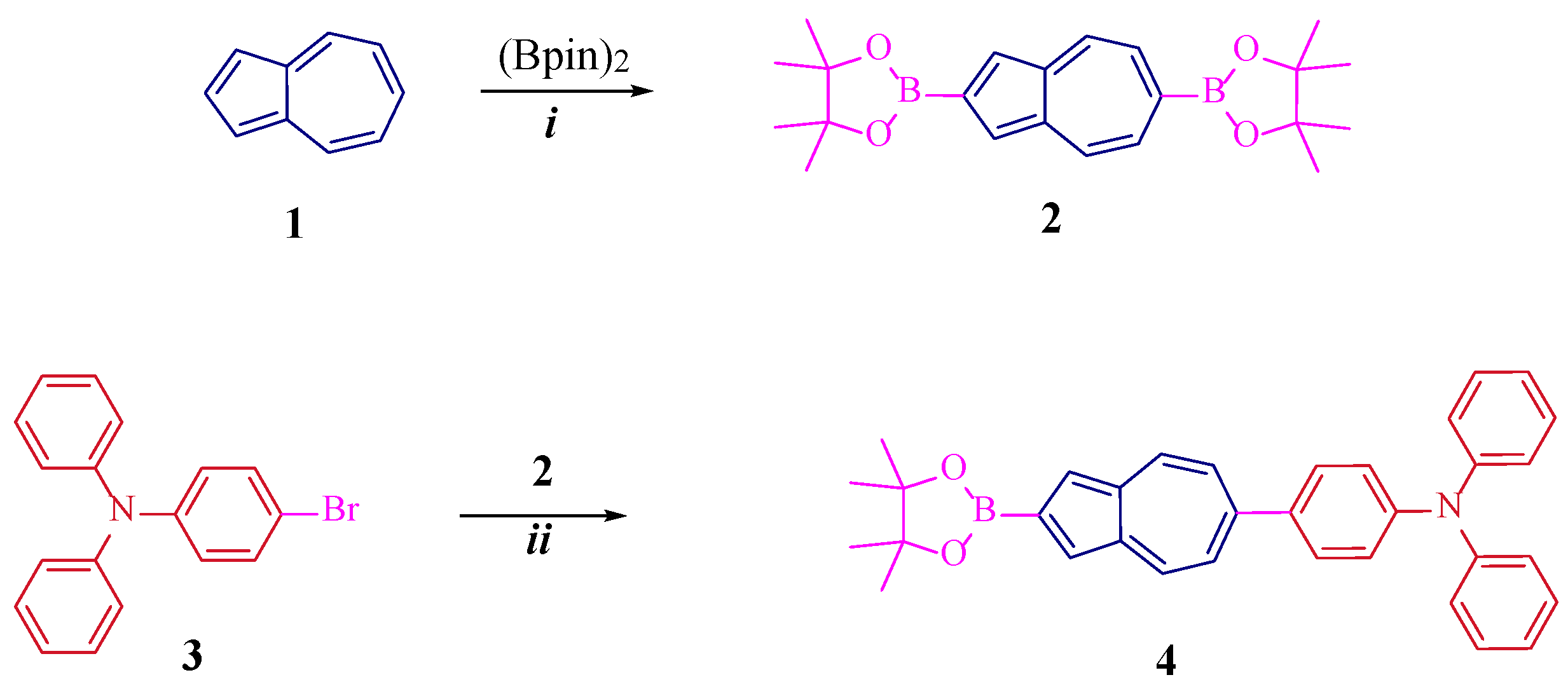
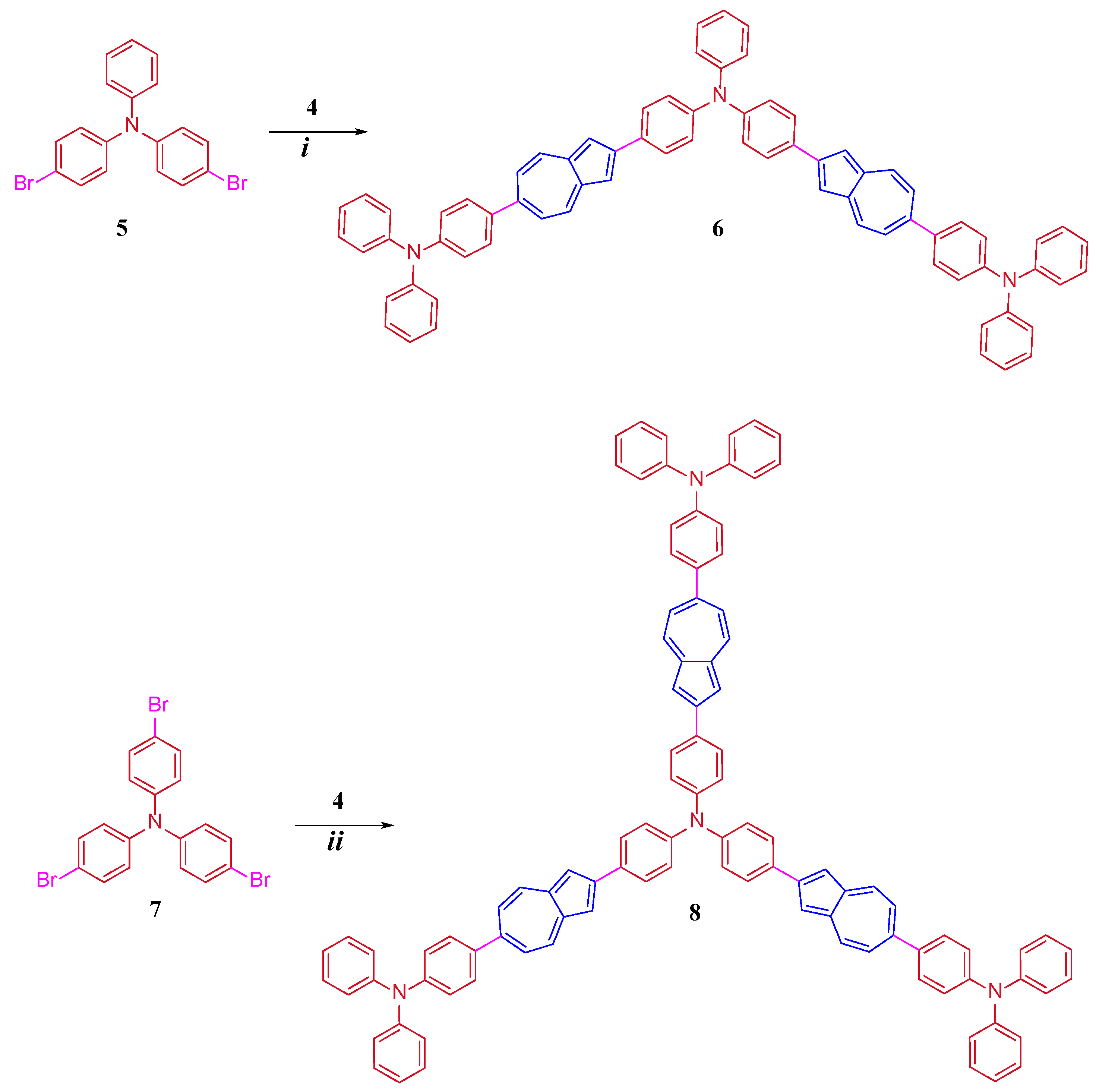
2. Results and Discussion
3. Materials and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zaumseil, J.; Sirringhaus, H. Electron and ambipolar transport in organic field-effect transistors. Chem. Rev. 2007, 107, 1296–1323. [Google Scholar] [PubMed]
- Duan, C.; Huang, F.; Cao, Y. Recent development of push–pull conjugated polymers for bulk-heterojunction photovoltaics: rational design and fine tailoring of molecular structures. J. Mater. Chem. 2012, 22, 10416–10434. [Google Scholar]
- Lash, T.D.; El-Beck, J.A.; Ferrence, G.M. Syntheses and Reactivity of meso-Unsubstituted Azuliporphyrins Derived from 6-tert-Butyl- and 6-Phenylazulene. J. Org. Chem. 2007, 72, 8402–8415. [Google Scholar] [PubMed]
- Wakabayashi, S.; Kato, Y.; Mochizuki, K.; Suzuki, R.; Matsumoto, M.; Sugihara, Y.; Shimizu, M. Pyridylazulenes: Synthesis, Color Changes, and Structure of the Colored Product. J. Org. Chem. 2007, 72, 744–749. [Google Scholar] [PubMed]
- Amatatsu, Y. Theoretical Prediction of the S1−S0 Internal Conversion of 6-Cyanoazulene. J. Phys. Chem. A 2007, 111, 5327–5332. [Google Scholar]
- Peart, P.A.; Repka, L.M.; Tovar, J.D. Emerging Prospects for Unusual Aromaticity in Organic Electronic Materials: The Case for Methano[10]annulene. Eur. J. Org. Chem. 2008, 28, 2193–2206. [Google Scholar]
- Shoji, T.; Ito, S.; Toyota, K.; Yasunami, M.; Morita, N. Synthesis, Properties, and Redox Behavior of Mono-, Bis-, and Tris[1,1,4,4,-tetracyano-2-(1-azulenyl)-3-butadienyl] Chromophores Binding with Benzene and Thiophene Cores. Chem. Eur. J. 2008, 14, 8398–8408. [Google Scholar]
- Ito, S.; Kubo, T.; Morita, N.; Ikoma, T.; Tero-Kubota, S.; Kawakami, J. ajiri, Azulene-Substituted Aromatic Amines. Synthesis and Amphoteric Redox Behavior of N,N-Di(6-azulenyl)-p-toluidine and N,N,N‘,N‘-Tetra(6-azulenyl)-p-phenylenediamine and Their Derivatives A. J. Org. Chem. 2005, 70, 2285–2293. [Google Scholar]
- Colby, D.A.; Ferrence, G.M.; Lash, T.D. Oxidative Metalation of Azuliporphyrins with Copper(II) Salts: Formation of a Porphyrin Analogue System with a Unique Fully Conjugated Nonaromatic Azulene Subunit. Angew. Chem. Int. Ed. 2004, 43, 1346–1349. [Google Scholar]
- Lopez-Alled, C.M.; Park, S.J.; Lee, D.J.; Murfin, L.C.; Kociok-Kohn, G.; Hann, J.L.; Wenk, J.; James, T.D.; Kim, H.M.; Lewis, S.E. Azulene-based fluorescent chemosensor for adenosine diphosphate. Chem. Commun. 2021, 57, 10608–10611. [Google Scholar]
- Xin, H.; Hou, B.; Gao, X. Azulene-Based π-Functional Materials: Design, Synthesis, and Applications. Acc. of Chem. Res. 2021, 54, 1737–1753. [Google Scholar]
- Anderson, A.G.; Steckler, B.M. Azulene. VIII. A Study of the Visible Absorption Spectra and Dipole Moments of Some 1- and 1,3-Substituted Azulenes. J. Am. Chem. Soc. 1959, 81, 4941–4946. [Google Scholar]
- Tomin, V.I.; Włodarkiewicz, A. Anti-Kasha behavior of DMABN dual fluorescence. J. Lumin. 2018, 198, 220–225. [Google Scholar]
- Nenov, A.; Borrego-Varillas, R.; Oriana, A.; et al. UV-Light-Induced Vibrational Coherences: The Key to Understand Kasha Rule Violation in trans-Azobenzene. J. Phys. Chem. Lett. 2018, 9, 1534–1541. [Google Scholar] [PubMed]
- Del Valle, J.C.; Catalán, J. Kasha’s Rule: a Reappraisal. Phys. Chem. Chem. Phys. 2019, 21, 10061–10069. [Google Scholar]
- Behera, S.K.; Park, S.Y.; Gierschner, J. Dual Emission: Classes, Mechanisms, and Conditions. Angew. Chem. Int. Ed. 2021, 60, 22624–22638. [Google Scholar]
- Dunlop, D.; Ludvikova, L.; Banerjee, A.; Ottosson, H. Slanina Excited-State (Anti)Aromaticity Explains Why Azulene Disobeys Kasha’s Rule. J. Am. Chem. Soc. 2023, 145, 21569–21575. [Google Scholar]
- Wang, F.; Lai, Y.-H. Conducting Azulene−Thiophene Copolymers with Intact Azulene Units in the Polymer Backbone. Macromolecules 2003, 36, 536–538. [Google Scholar]
- Wang, F.; Lai, Y.-H.; Han, M.-Y. Stimuli-Responsive Conjugated Copolymers Having Electro-Active Azulene and Bithiophene Units in the Polymer Skeleton: Effect of Protonation and p-Doping on Conducting Properties. Macromolecules 2004, 37, 3222–3230. [Google Scholar]
- Mrozek, T.; Daub, H.G.J. Multimode-Photochromism Based on Strongly Coupled Dihydroazulene and Diarylethene. J. Chem. Eur. J. 2001, 7, 1028–1040. [Google Scholar]
- Dong, J.X.; Zhang, H.L. Azulene-based organic functional molecules for optoelectronics. Chin. Chem. Lett. 2016, 27, 1097–1104. [Google Scholar] [CrossRef]
- Ou, L.; Zhou, Y.; Wu, B.; Zhu, L. The unusual physicochemical properties of azulene and azulene-based compounds. Chin. Chem. Lett. 2019, 30, 1903–1907. [Google Scholar] [CrossRef]
- Kurotobi, K.; Kim, K.S.; Noh, S.B.; et al. A Quadruply Azulene-Fused Porphyrin with Intense Near-IR Absorption and a Large Two-Photon Absorption Cross Section. Angew. Chem. Int. Ed. 2006, 45, 3944–3947. [Google Scholar] [CrossRef] [PubMed]
- Cristian, L.; Sasaki, I.; Lacroix, P.G.; et al. Donating Strength of Azulene in Various Azulen-1-yl-Substituted Cationic Dyes: Application in Nonlinear Optics. Chem. Mater. 2004, 16, 3543–3551. [Google Scholar] [CrossRef]
- Ito, S.; Inabe, H.; Morita, N.; et al. Synthesis of Poly(6-azulenylethynyl)benzene Derivatives as a Multielectron Redox System with Liquid Crystalline Behavior. J. Am. Chem. Soc. 2003, 125, 1669–1680. [Google Scholar] [CrossRef]
- Yamaguchi, Y.; Takubo, M.; Ogawa, K.; et al. Terazulene Isomers: Polarity Change of OFETs through Molecular Orbital Distribution Contrast. J. Am. Chem. Soc. 2016, 138, 1335–11343. [Google Scholar] [CrossRef]
- Nishimura, H.; Ishida, N.; Shimazaki, A.; et al. Hole-Transporting Materials with a Two-Dimensionally Expanded π-System around an Azulene Core for Efficient Perovskite Solar Cells. J. Am. Chem. Soc. 2015, 137, 5656–15659. [Google Scholar] [CrossRef]
- Zhou, Y.; Zou, Q.; Qiu, J.; et al. Rational Design of a Green-Light-Mediated Unimolecular Platform for Fast Switchable Acidic Sensing. J. Phys. Chem. Lett. 2018, 9, 550–556. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhu, L. Involving Synergy of Green Light and Acidic Responses in Control of Unimolecular Multicolor Luminescence. Chem.–Eur. J. 2018, 24, 10306–10309. [Google Scholar]
- Tsuchiya, T.; Higashibeppu, M.; Mazaki, Y. Synthesis and Properties of Twisted and Helical Azulene Oligomers and Azulene Based Polycyclic Hydrocarbons. ChemistryOpen 2023, 12, e202100298. [Google Scholar] [CrossRef]
- Tsuchiya, T.; Katsuoka, Y.; Yoza, K.; Sato, H.; Mazaki, Y. Stereochemistry, Stereodynamics, and Redox and Complexation Behaviors of 2,2′ -Diaryl-1,1′ -Biazulenes. ChemPlusChem 2019, 84, 1659. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, T.; Umemura, R.; Kaminaga, M.; Kushida, S.; Ohkubo, K.; Noro, S.I.; Mazaki, Y. Paddlewheel Complexes with Azulenes: Electronic Interaction between Metal Centers and Equatorial Ligands. ChemPlusChem 2019, 84, 655. [Google Scholar] [CrossRef] [PubMed]
- Konishi, A.; Yasuda, M. Breathing new life into nonalternant hydrocarbon chemistry: Syntheses and properties of polycyclic hydrocarbons containing azulene, pentalene, and heptalene frameworks. Chem. Lett. 2021, 50, 195. [Google Scholar] [CrossRef]
- Xin, H.; Hou, B.; Gao, X. Azulene-Based π-Functional Materials: Design, Synthesis, and Applications. Acc. Chem. Res. 2021, 54, 1737. [Google Scholar] [CrossRef] [PubMed]
- Shevyakov, S.V.; Li, H.; Muthyala, R.; Asato, A.E.; Croney, J.C.; Jameson, D.M.; Liu, R.S. Orbital control of the color and excited state properties of formylated and fluorinated derivatives of azulene. J. Phys. Chem. A 2003, 107, 3295. [Google Scholar] [CrossRef]
- Fujinaga, M.; Murafuji, T.; Kurotobi, K.; Sugihara, Y. Polyborylation of azulenes. Tetrahedron 2009, 65, 7115–7121. [Google Scholar] [CrossRef]
- Rahimi, K.; Botiz, I.; Agumba, J.O.; Motamen, S.; Stingelin, N.; Reiter, G. Light absorption of poly(3-hexylthiophene) single crystals. RSC Adv. 2014, 4, 11121. [Google Scholar] [CrossRef]
- Wang, Z.-S.; Koumura, N.; Cui, Y.; Takahashi, M.; Sekiguchi, H.; Mori, A.; Kubo, T.; Furube, A.; Hara, K. HexylthiopheneFunctionalized Carbazole Dyes for Efficient Molecular Photovoltaics: Tuning of Solar-Cell Performance by Structural Modification. Chem. Mater. 2008, 20, 3993. [Google Scholar] [CrossRef]
- Shirota, Y.; Kageyama, H. Charge Carrier Transporting Molecular Materials and Their Applications in Devices. Chem. Rev. 2007, 107, 953. [Google Scholar] [CrossRef]
- Ren, S.; Habibi, A.; Ni, P.; Nahdi, H.; Bouanis, F.Z.; Bourcier, S.; Clavier, G.; Frigoli, M.; Yassar, A. Synthesis and characterization of solution-processed indophenine derivatives for function as a hole transport layer for perovskite solar cells. Dye. Pigment. 2023, 213, 111136. [Google Scholar] [CrossRef]
- Ren, S.; Wang, Z.; Zhang, W.; Ding, Y.; Yi, Z. Donor-acceptor-based organic polymer semiconductor materials to achieve high hole mobility in organic field-effect transistors. Polymers 2023, 15, 3713. [Google Scholar] [CrossRef] [PubMed]
- Murphy, A.R.; Fréchet, J.M.J. Organic Semiconducting Oligomers for Use in Thin Film Transistors. Chem. Rev. 2007, 107, 1066. [Google Scholar] [PubMed]
- Zaumseil, J.; Sirringhaus, H. Electron and Ambipolar Transport in Organic Field-Effect Transistors. Chem. Rev. 2007, 107, 1296. [Google Scholar] [PubMed]
- Tsuchiya, T.; Hamano, T.; Inoue, M.; Nakamura, T.; Wakamiya, A.; Mazaki, Y. Intense absorption of azulene realized by molecular orbital inversion. Chem. Commun. 2023, 59, 10604–10607. [Google Scholar]
- Ren, S.; Wang, Z.; Zhang, W.; Yassar, A.; Chen, J.; Wang, S. Incorporation of diketopyrrolopyrrole into polythiophene for the preparation of organic polymer transistors. Molecules 2024, 29, 260. [Google Scholar] [CrossRef]
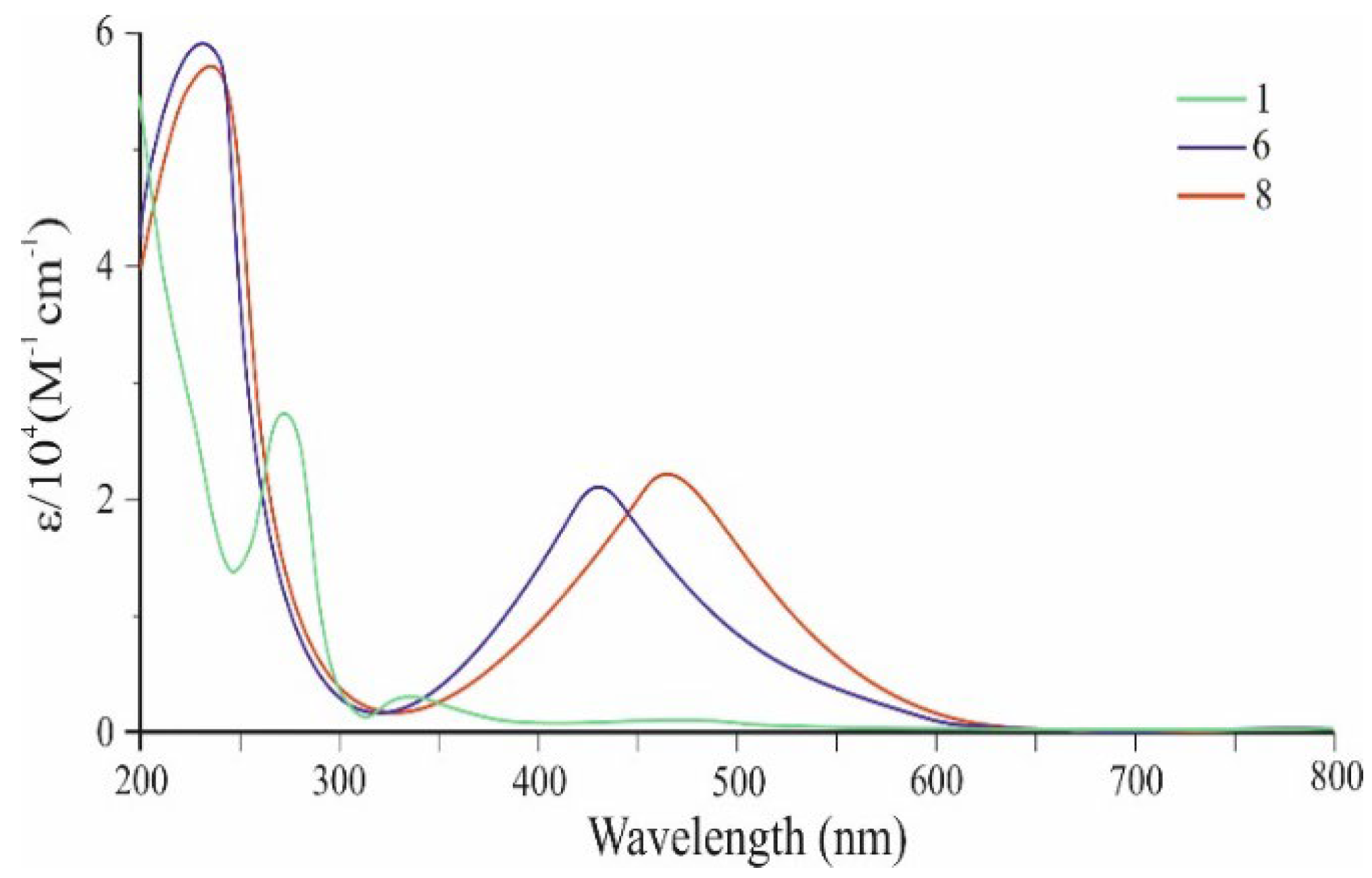
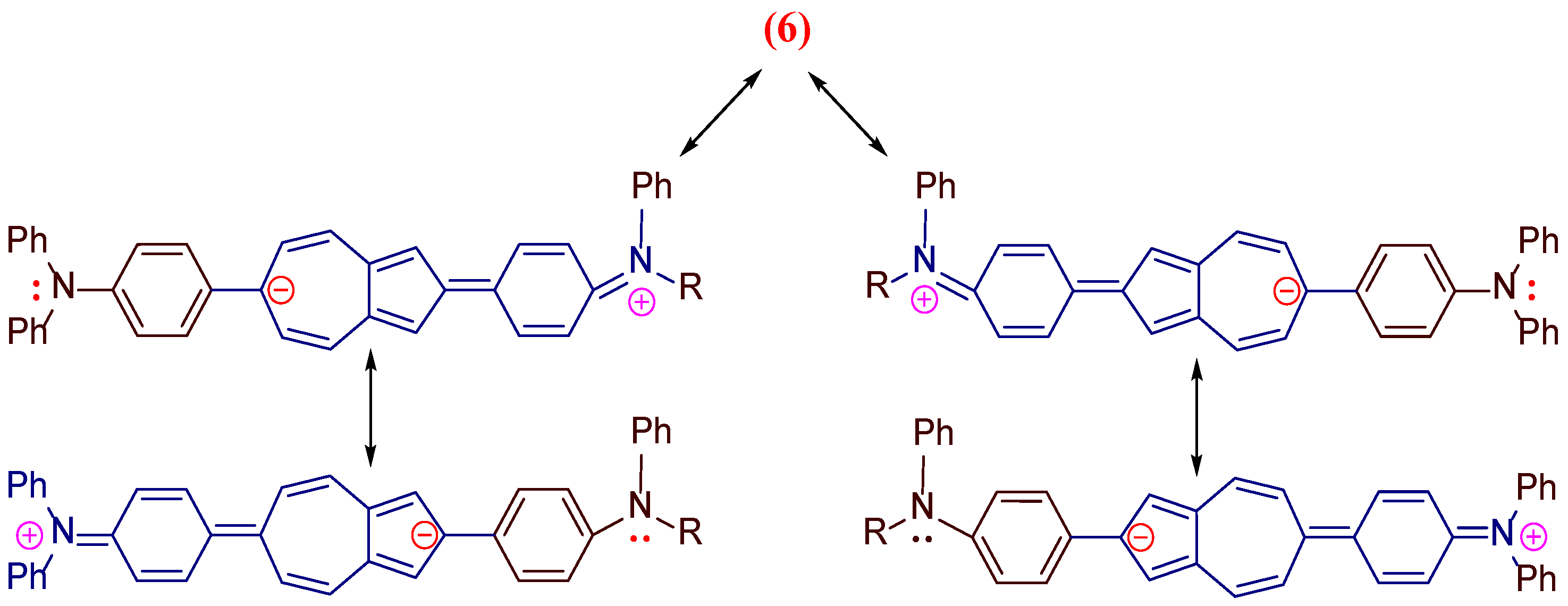
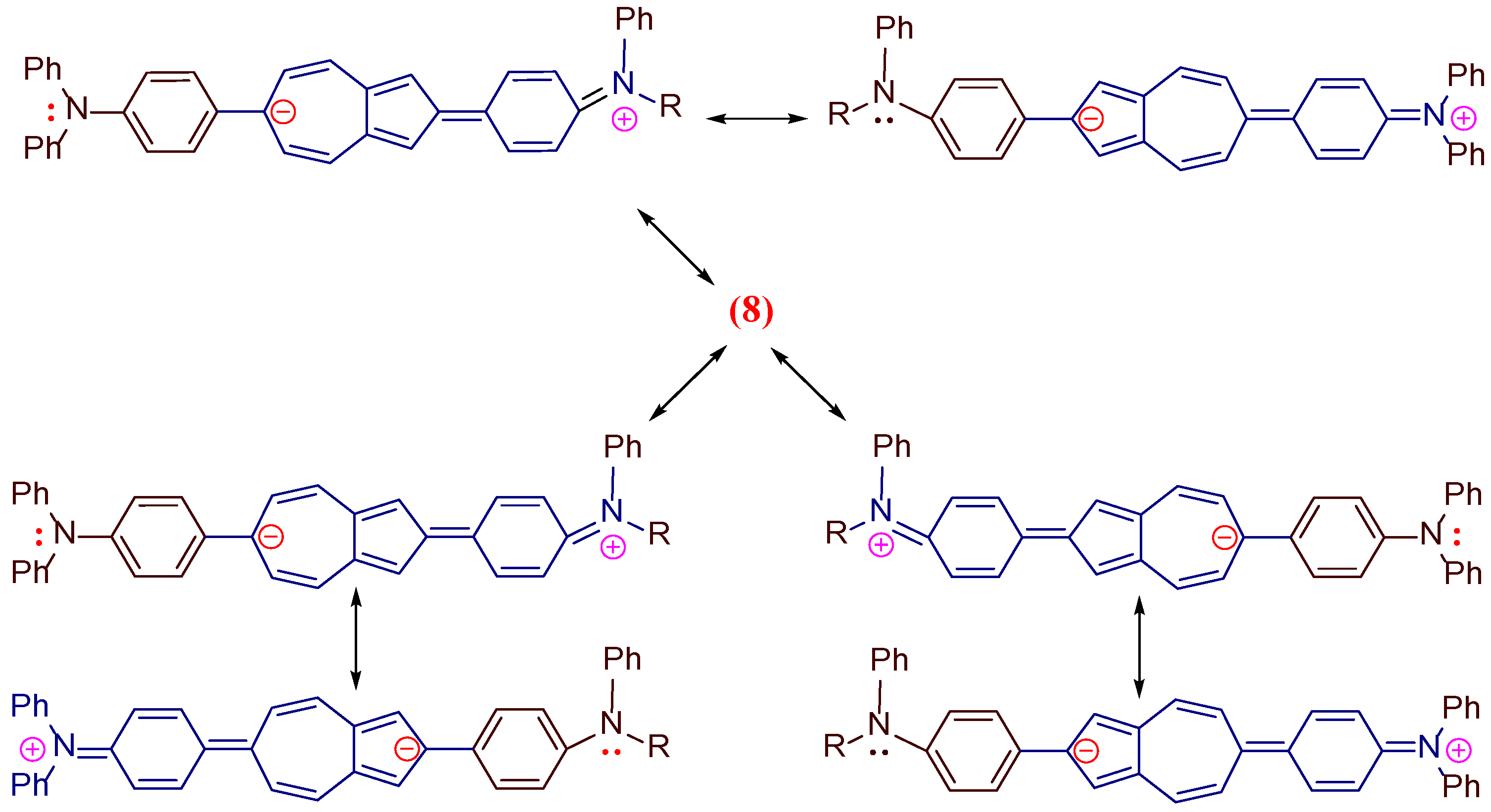
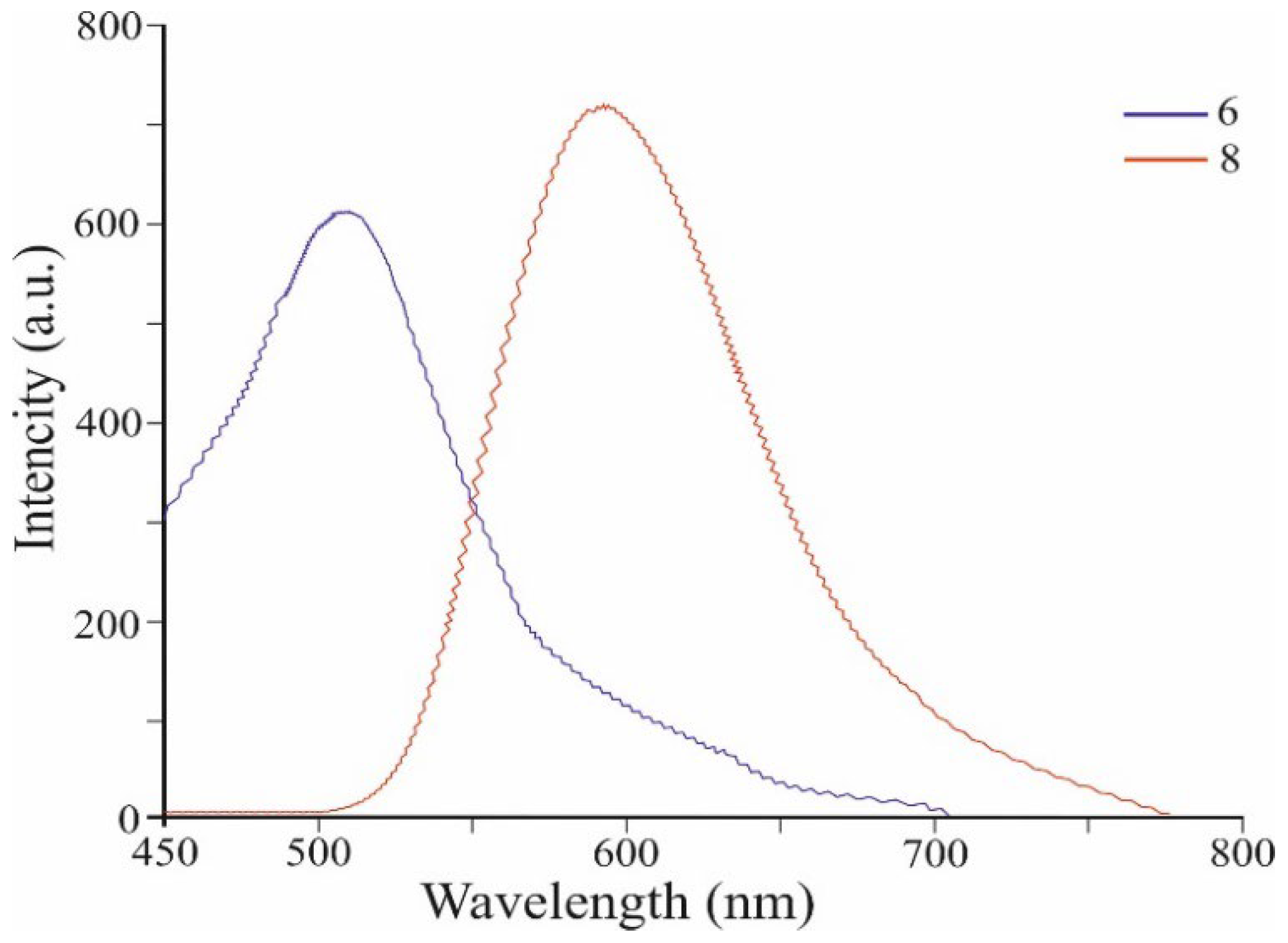
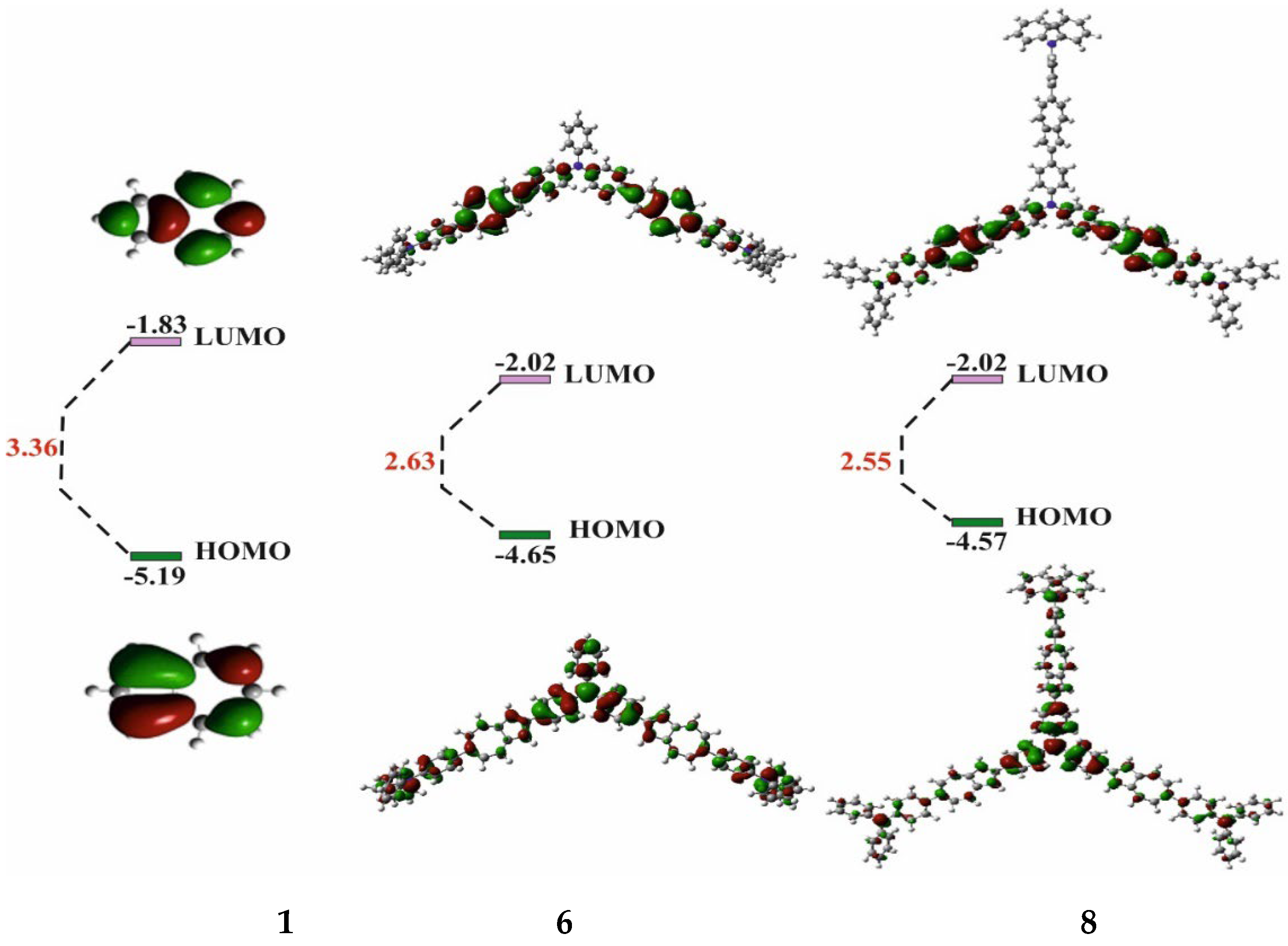
| Co-oligomers | Absorption data | Fluorescence data a | ||
|---|---|---|---|---|
| Absorption maximum (nm) | Molar coefficient (M-1cm-1) |
Emission maximum (nm) | Emission intensity (a.u) |
|
| 6 | 246 | 58 112 | 510 | 611 |
| 262 | 58 826 | |||
| 431 | 22 447 | |||
| 8 | 256 | 57 732 | 590 | 710 |
| 266 | 57 881 | |||
| 462 | 24 621 | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




