Submitted:
09 October 2024
Posted:
10 October 2024
You are already at the latest version
Abstract
Keywords:
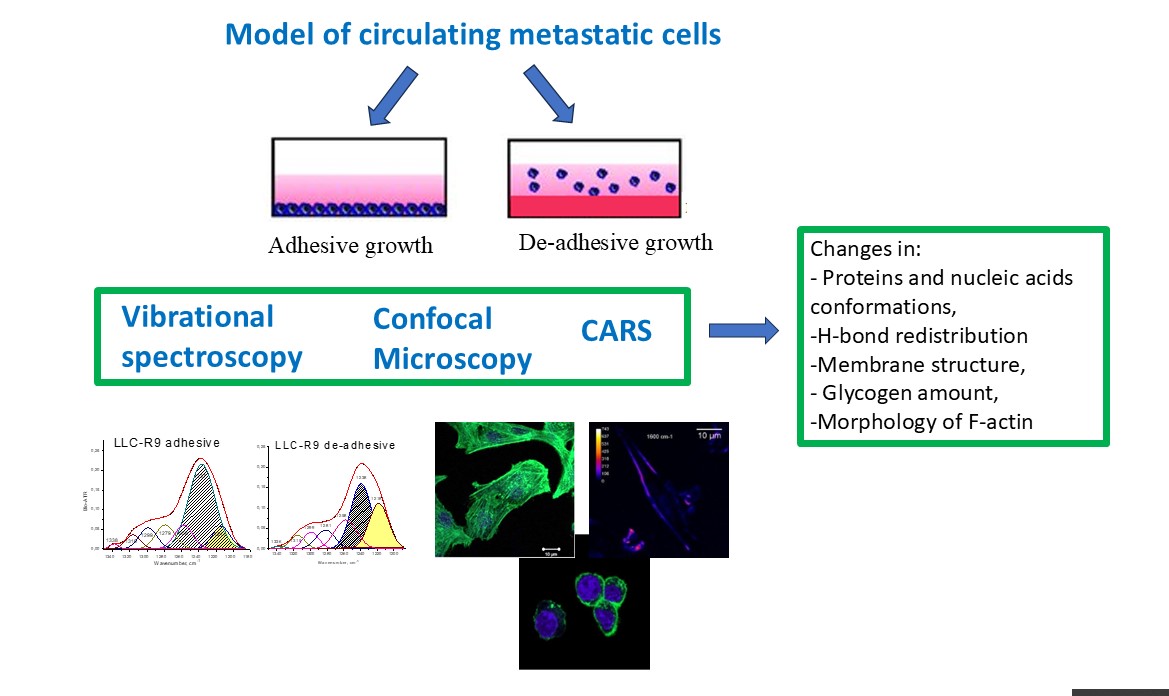
1. Introduction
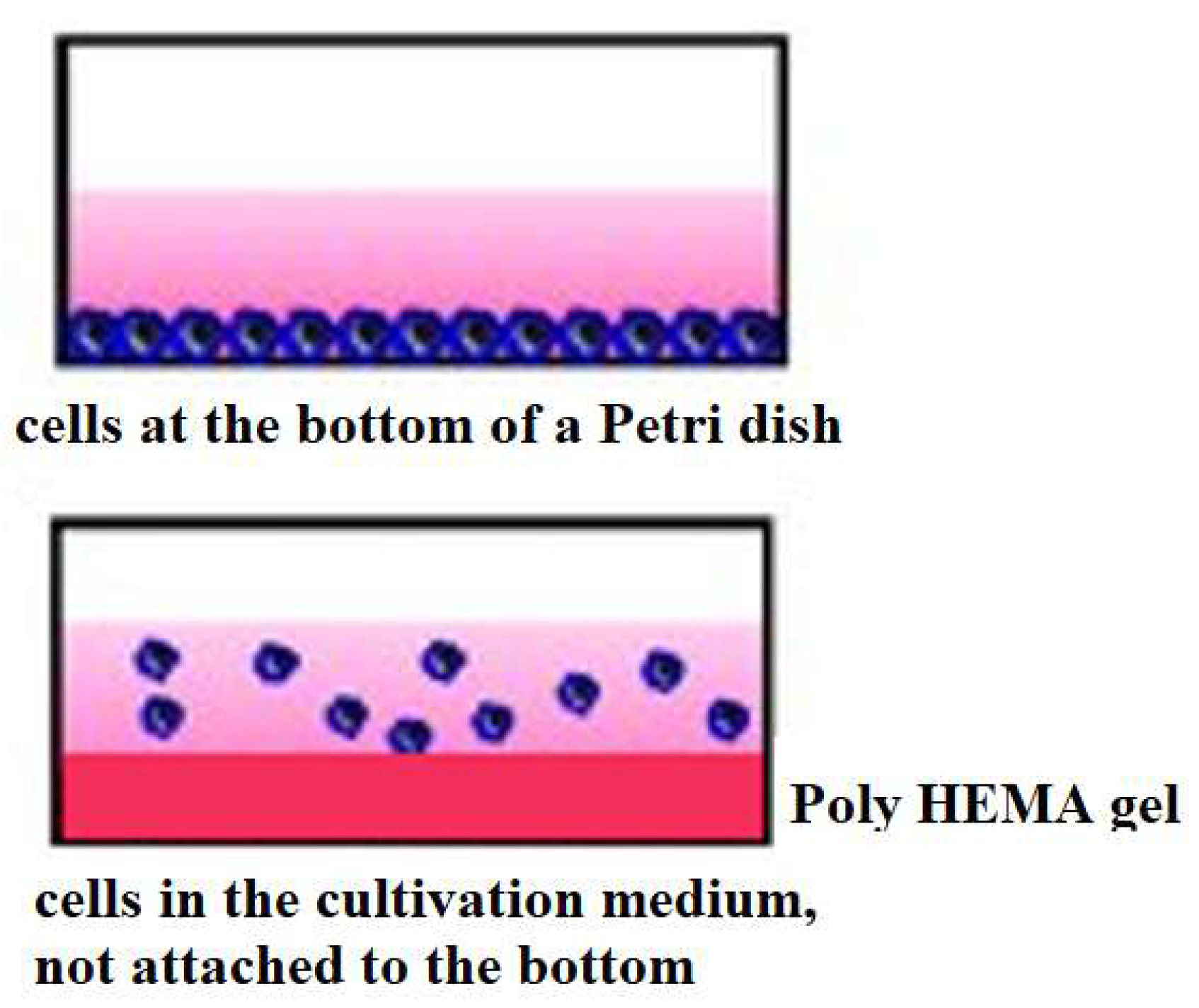
2. Materials and Methods
3. Results
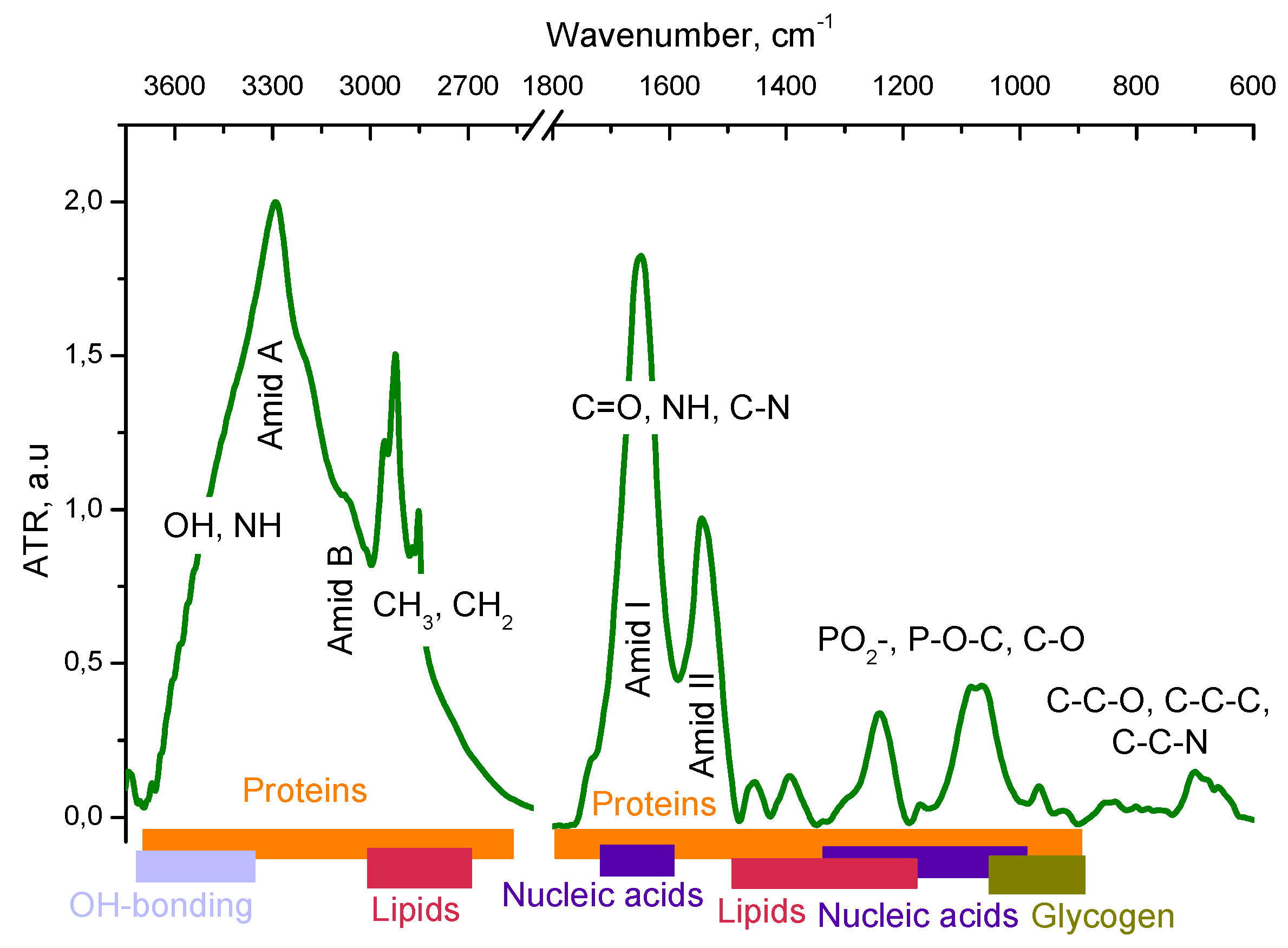
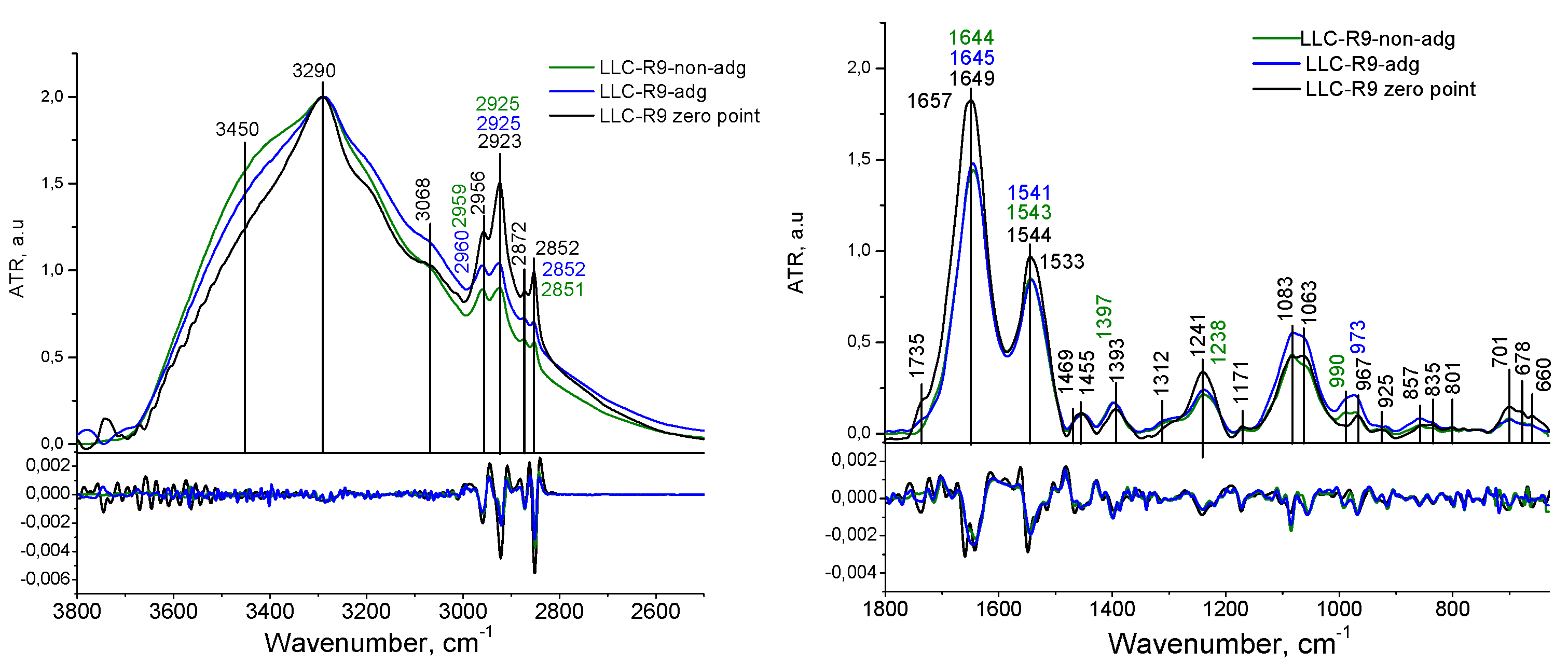
3.1. IR spectral Analysis of Tumor Cells LLC-R9
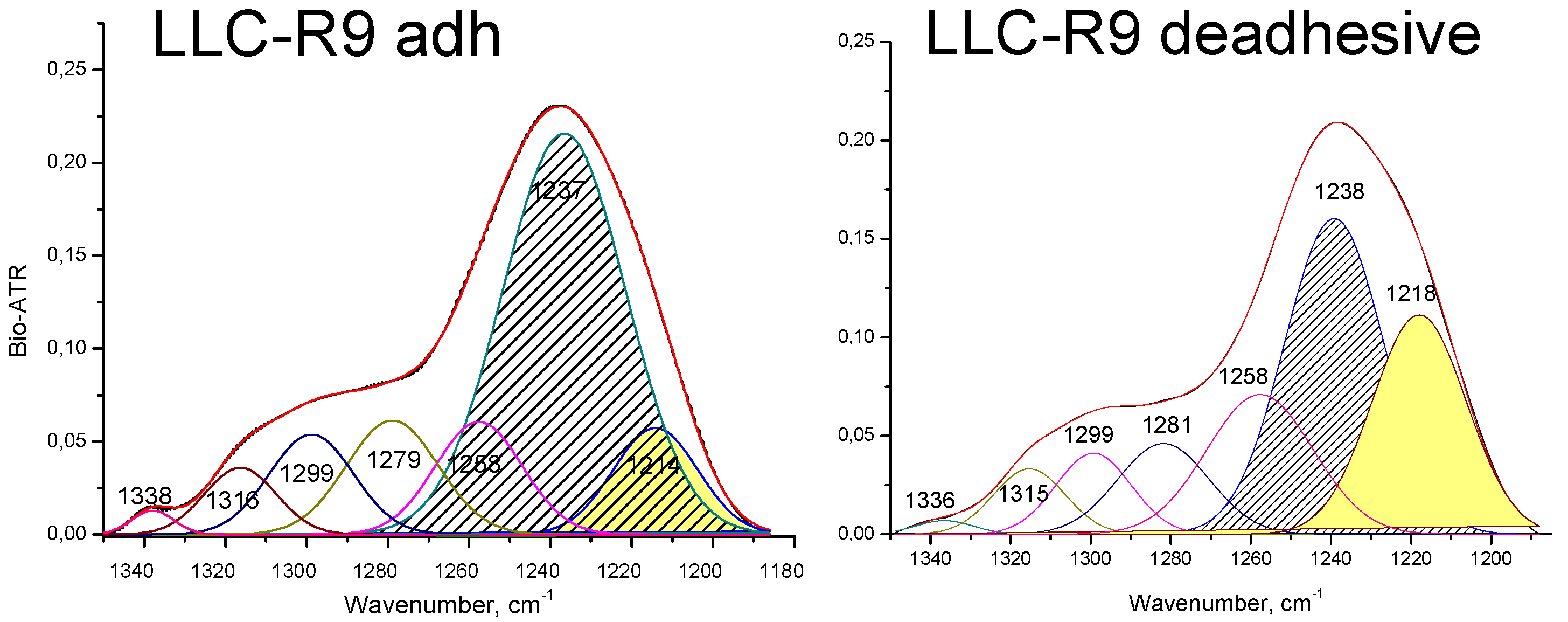
| LLC-R9 | |||
| De-adhesive | Adhesive | Zero point | Assignment |
| 3450 | 3450 | 3450 | Str OH |
| 3288 | 3286 | 3290 | Amid A, Str NH, |
| 3066 | 3068 | 3068 | Amid B, Fermi resonance Amid II |
| 2959 | 2959 | 2956 | Str CH3 asym |
| 2923 | 2925 | 2923 | Str CH2 asym |
| 2873 | 2872 | 2872 | Str CH3 sym |
| 2851 | 2852 | 2852 | Str CH2 sym |
| 1735 | 1735 | Str C=O | |
| - | - | 1657 | Amid I, Str C=O, Str C-N, Def N-H |
| 1644 | 1645 | 1649 | Amid I, Str C=O, Str C-N, Def N-H, def OH |
| 1543 | 1542 | 1544 | Amid II, Str C-N, Def N-H |
| - | - | 1533 | Amid II, Str C-N, Def N-H |
| 1451 | 1454 | 1455 | Def CH2 |
| 1398 | 1396 | 1393 | Def CH3 |
| 1238 | 1237 | 1240 | Str РО2- asym |
| 1218 | 1214 | Str РО2- asym | |
| 1170 | 1171 | 1171 | Str C-C, def C-OH, strC-O, C-OH |
| 1084 | 1081 | 1083 | Str РО2- sym |
| 1058 | 1062 | 1063 | Str C-O deoxyribose |
| 990 | 987 | C-C, C-O DNA and deoxyribose | |
| 970 | 973 | 967 | C-O DNA and deoxyribose |
| 917 | 917 | 925 | C-C, C-O |
| 858 | 858 | 854 | C3’ endo/anti (A-helix of DNA) |
| 834 | 835 | 835 | C2’ endo/anti (B-helix of DNA) |
| 778 | 780 | 782 | Out-of-plane bend |
| 699 | 701 | Out-of-plane bend | |
| 678 | C-C-H out-of-plane bend | ||
| 655 | 660 | C-C-O out-of-plane bend | |
3.2. Analysis of Raman Spectra
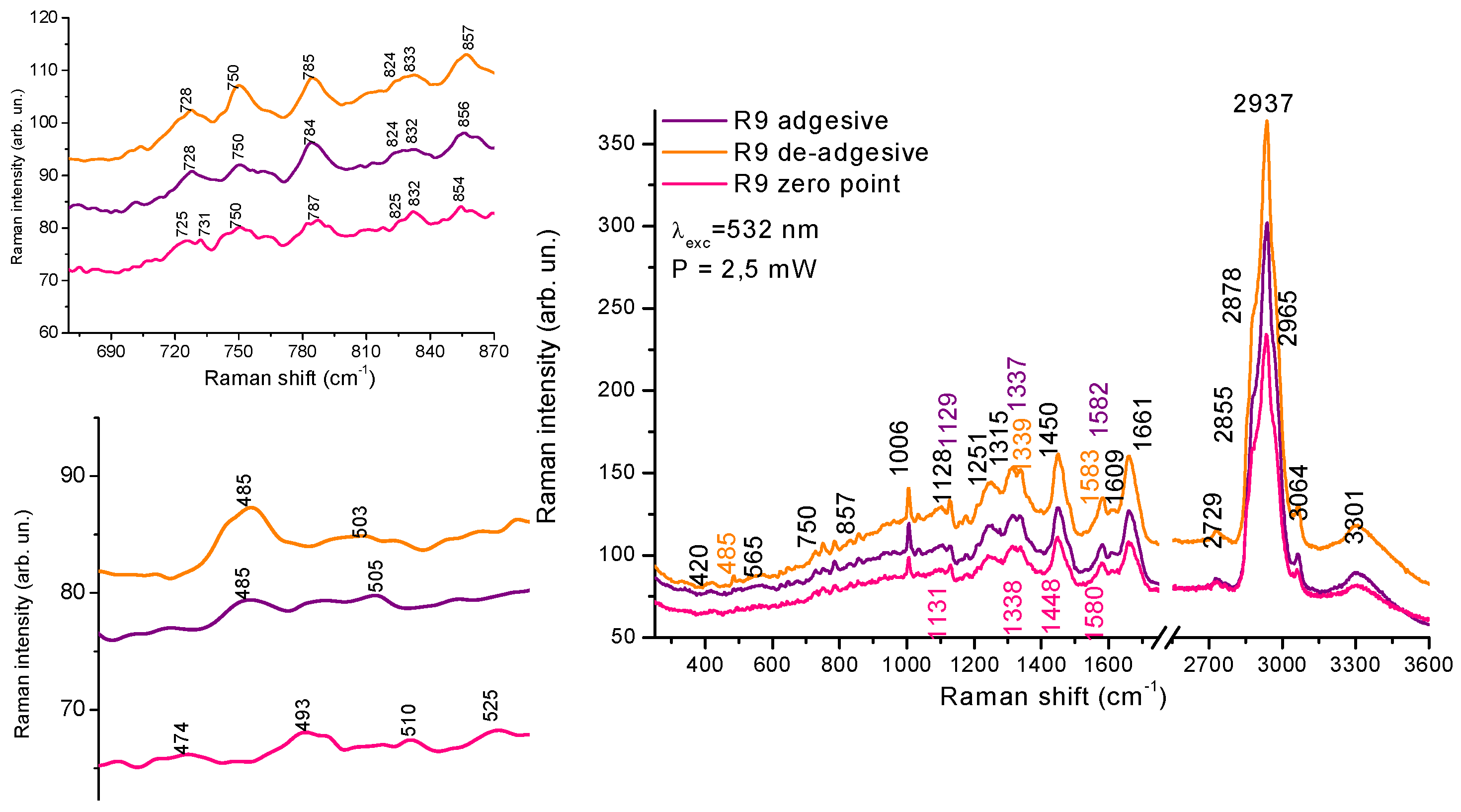
| 825 Tyr/750Trp | 854Tyr/825 Tyr | 750 Trp/785 DNA | Glycogen, a.u. | |
| De-adhesive | 0,43 | 1,67 | 1,0 | 4,77 |
| Adgesive | 0,50 | 1,33 | 0,85 | 1,85 |
| Zero-point | 0,60 | 1,33 | 0,83 | - |
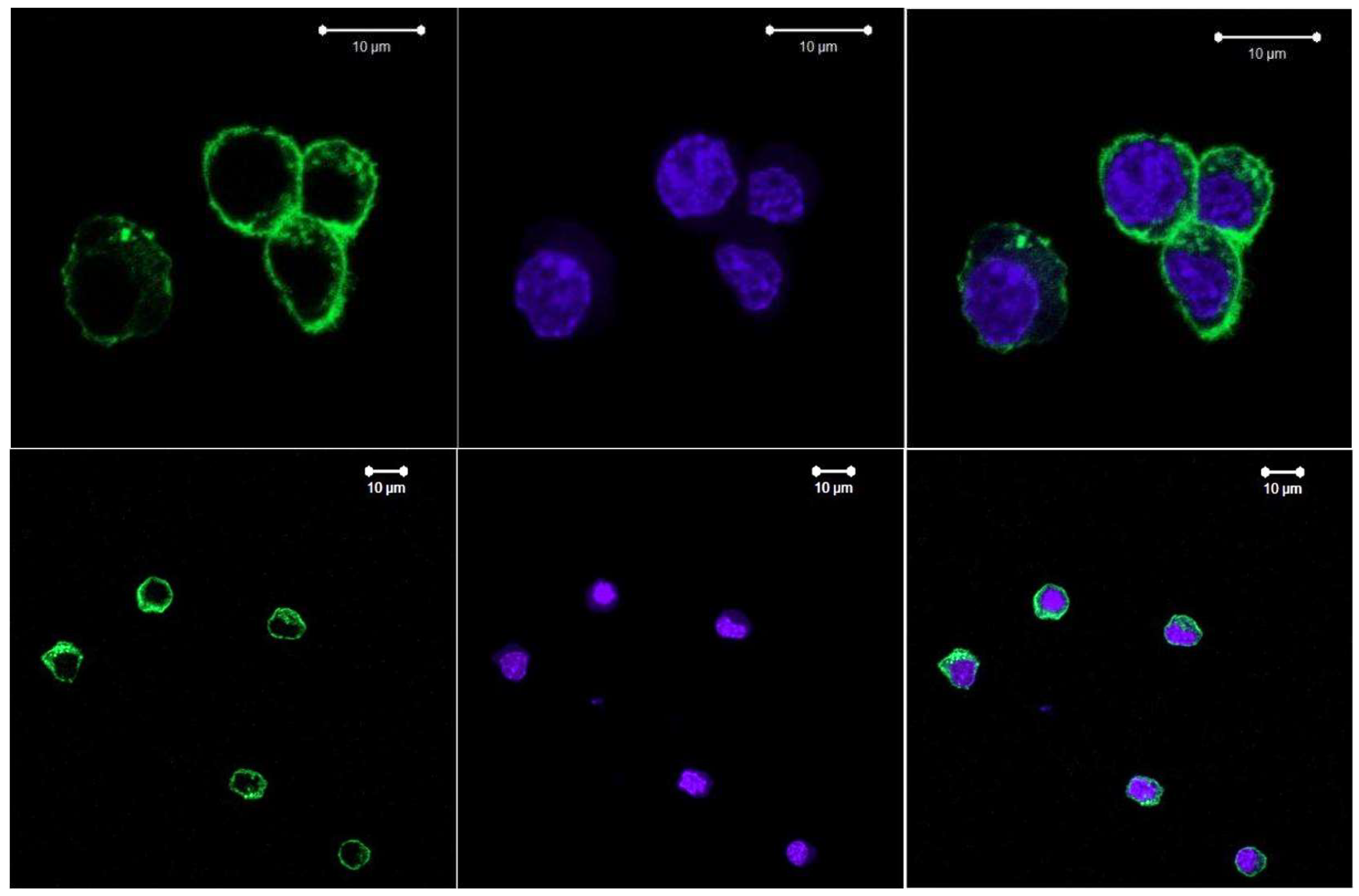
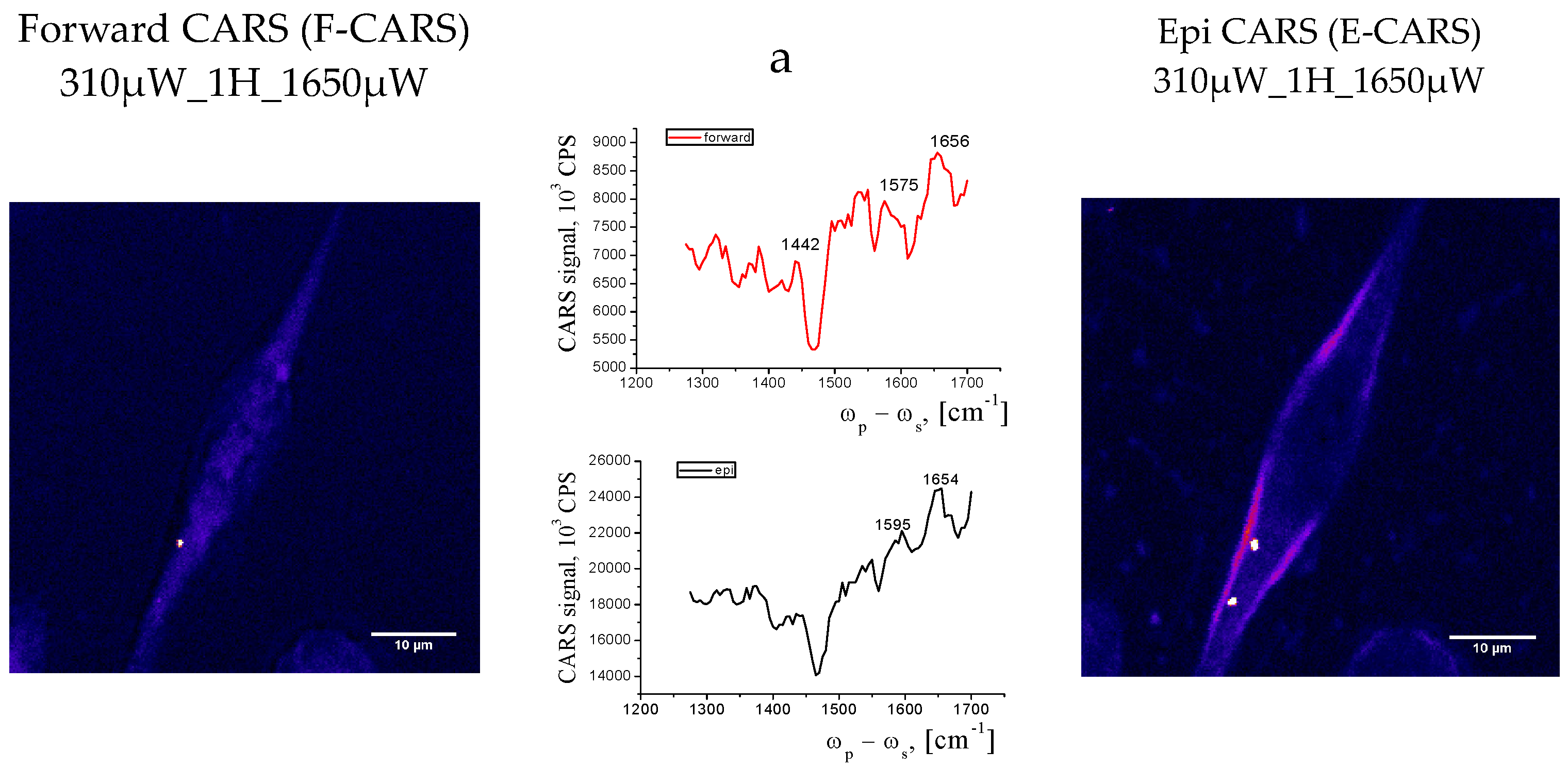
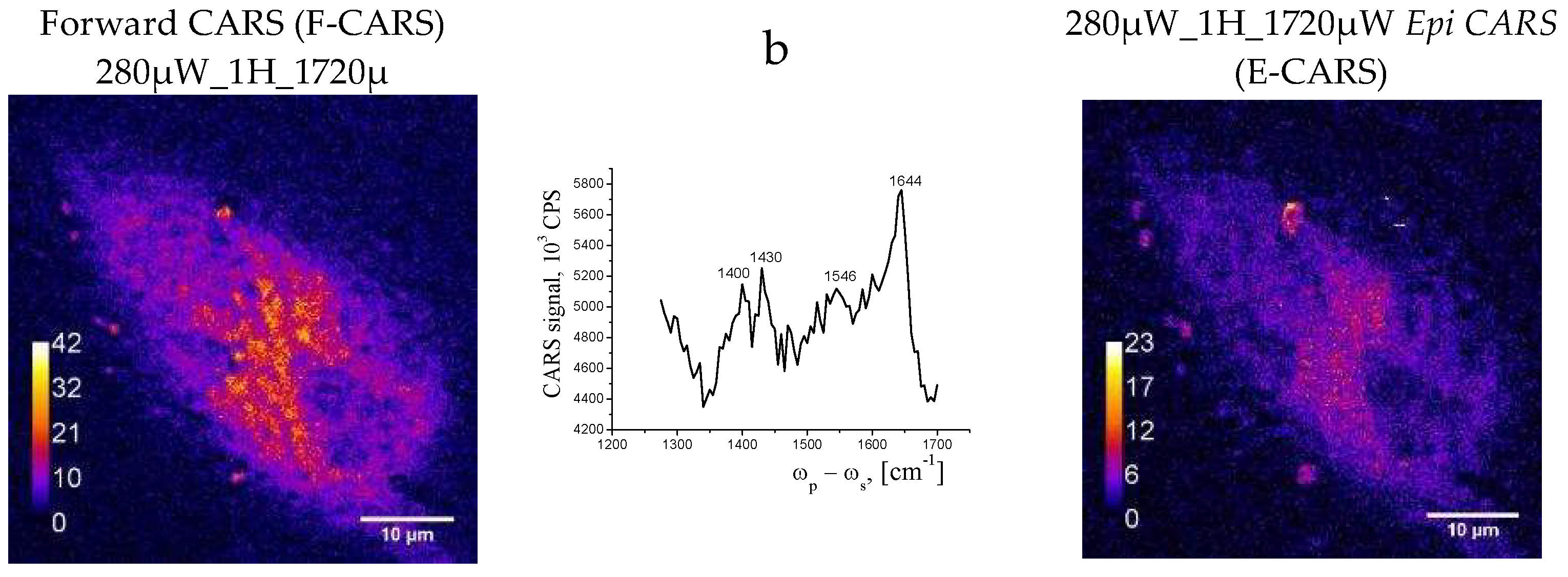
3.3. Experimental Data from CARS Microscopy
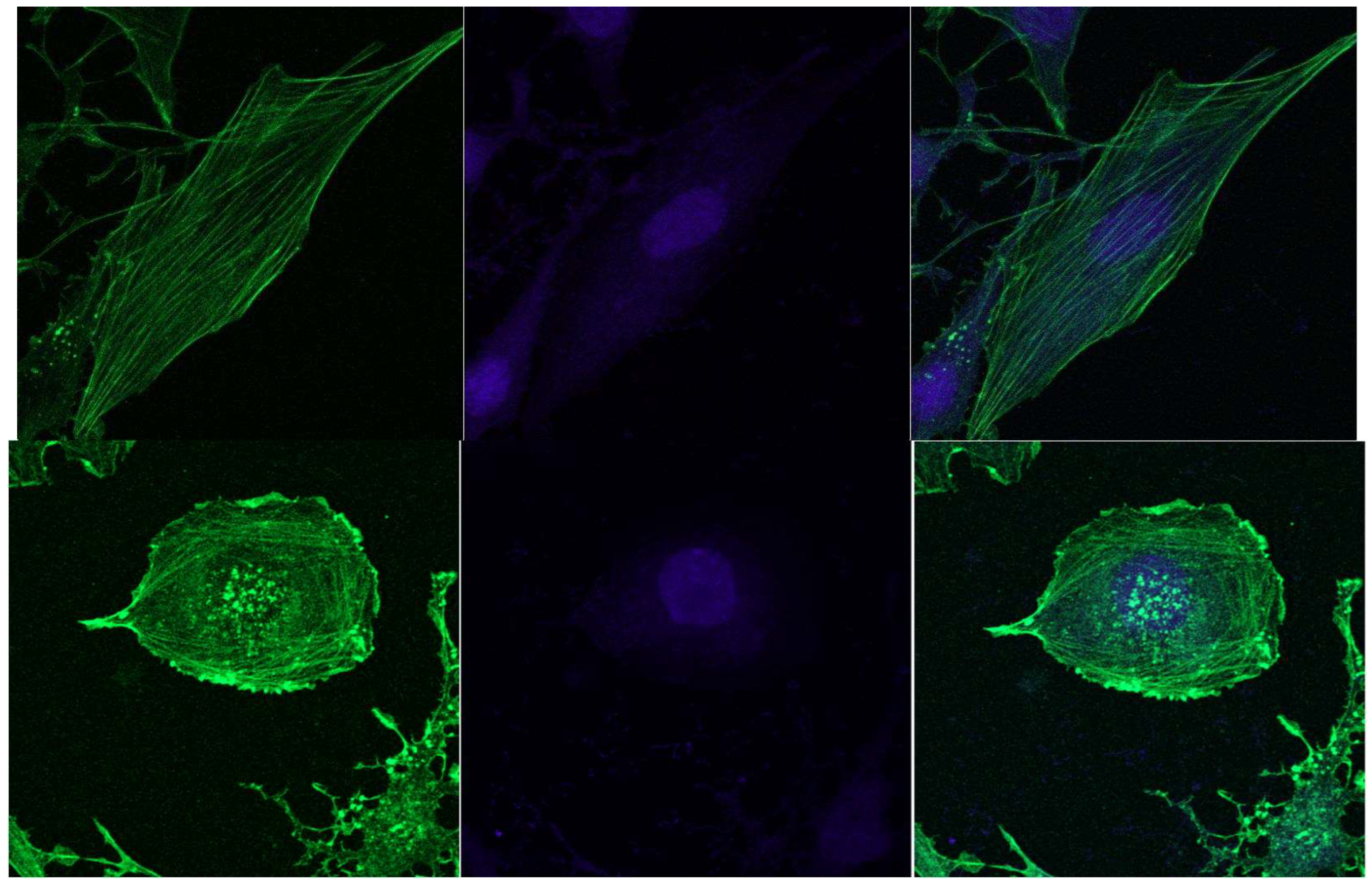
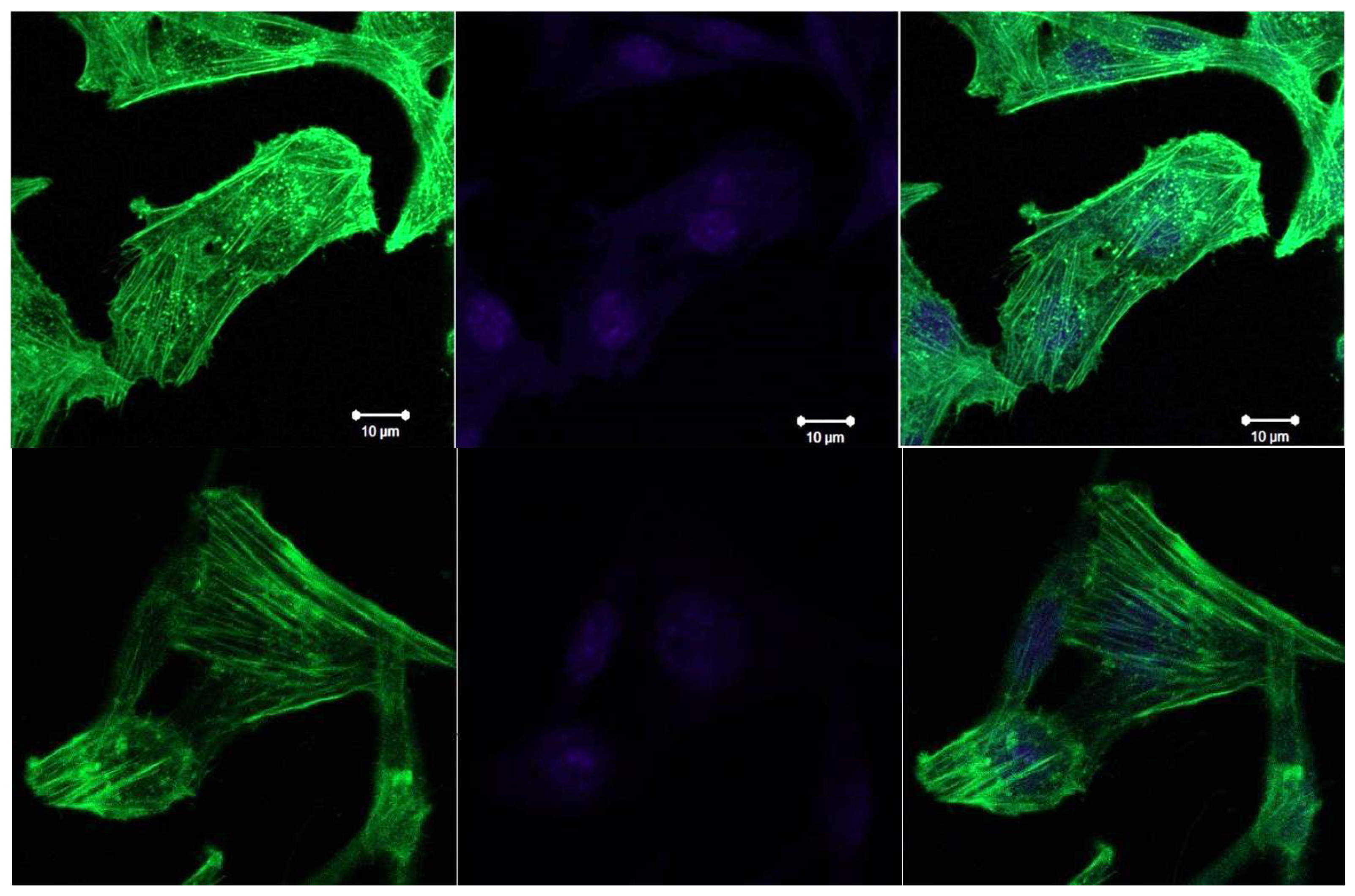
3.4. Confocal Microscopy Data
4. Conclusions
- FTIR spectra of adhesive and de-adhesive cells show protein conformations transformation, membrane changes and H-bond reorganization.
- From the Raman spectra in the region of 400-900 cm-1, Trp, Tyr aminoacids and glycogen level characterized special features which indicates changes in the environment for adhesive and de-adhesive cells, H-bonds redistribution, reorganization of proteins etc.
- CARS data correlate with Raman data and together with confocal microscopy data indicate that a small number of round cells can be detected in the population of adherent cells, which may be the result of apoptosis or necrosis or changes in their functional activity under the influence of external stimuli or the environment.
- LLC cells during de-adhesive growth are rounded, significantly reducing the cell surface. During adhesive growth, thin actin threads are clearly visible, which are part of the contractile and migration apparatus. During de-adhesive growth, F-actin is fixed in the form of microfilaments in the cytoskeleton of cells. F-actin in LLC/R9 cells for de-adhesive growth is statistically significantly reduced by more than 38% compared to adhesive growth. The nature of F-actin distribution under de-adhesive and adhesive growth conditions of LLC/R9 cells is also significantly different: F-actin is fixed in the form of microfilaments during de-adhesive growth and in the form of threads during adhesive growth.
Acknowledgments
References
- Saxena, M.; Christofori, G. Rebuilding cancer metastasis in the mouse. Mol Oncol. 2013, 7, 283–296. [Google Scholar] [CrossRef] [PubMed]
- Eslami-S, Z.; Cortés-Hernández, L.E.; Thomas, F.; et al. Functional analysis of circulating tumour cells: the KEY to understand the biology of the metastatic cascade. Br J Cancer 2022, 127, 800–810. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.; Shen, L.; Luo, M.; et al. Circulating tumor cells: biology and clinical significance. Sig Transduct Target Ther 2021, 6, 404. [Google Scholar] [CrossRef]
- Nasr, M.M.; Lynch, C.C. How circulating tumor cluster biology contributes to the metastatic cascade: from invasion to dissemination and dormancy. Cancer Metastasis Rev 2023, 42, 1133–1146. [Google Scholar] [CrossRef]
- Solyanik, G.; Kolesnyk, D.; Gnatyuk, O.; Dovbeshko, G. Changes in glucose metabolism during detachment of metastatic cells. Scientific Collection «InterConf+» 2023, 35, 179–193. [Google Scholar] [CrossRef]
- Solyanik, I.; Kolesnik, D.L.; Prokhorova, I.V.; Yurchenko, O.V.; Pyaskovskaya, O.N. Mitochondrial dysfunction significantly contributes to the sensitivity of tumor cells to anoikis and their metastatic potential. Heliyon 2024, 10. [Google Scholar] [CrossRef] [PubMed]
- Mantsch, H.H. Biomedical Vibrational Spectroscopy in the Era of Artificial Intelligence. Molecules 2021, I, 1439. Biological and biomedical Infrared spectroscopy, edited by A.Barth P.Haris,IOS Press-Amsterdam-Berlin-Tokyo-Washington, DC, 2009,435P. [CrossRef]
- Helburn, R.; Nolan, K. Characterizing biological macromolecules with attenuated total reflectance–Fourier transform infrared spectroscopy provides hands-on spectroscopy experiences for undergraduates. Biochem Mol Biol Educ. 2022, 50, 381–392. [Google Scholar] [CrossRef]
- Finlayson, D.; Rinaldi, C.; Baker, M.J. Is Infrared Spectroscopy Ready for the Clinic? Anal. Chem. 2019, 91, 12117. [Google Scholar] [CrossRef]
- Dovbeshko, G.I.; Gridina, N.Y.; Kruglova, E.B.; et al. FTIR spectroscopy studies of nucleic acid damage. Talanta 2000, 53, 233–246. [Google Scholar] [CrossRef]
- Dovbeshko, G.I.; Chegel, V.I.; Gridina, N.Y.; Repnytska, O.P.; Shirshov, Y.M.; Tryndiak, V.P.; Todor, I.M.; Solyanik, G.I. Surface enhanced IR absorption of nucleic acids from tumor cells: FTIR reflectance study. Biopolymers 2002, 67, 470–486. [Google Scholar] [CrossRef]
- Dovbeshko, G.I.; Chegel, V.I.; Gridina, N.Y.; Repnytska, O.P.; Shirshov, Y.M.; Tryndiak, V.P.; Todor, I.M.; Zynio, S.A. Surface enhanced infrared absorption of nucleic acids on gold substrate. Semiconductor Physics Quantum Electronics & Optoelectronics 2001, 4, 202–206. [Google Scholar]
- Vo-Dinh, T.; Yan, F.; Wabuyele, M.B. Surface-Enhanced Raman Scattering for Biomedical Diagnostics and Molecular Imaging. In: Kneipp, K., Moskovits, M., Kneipp, H. (eds) Surface-Enhanced Raman Scattering. In Topics in Applied Physics; Springer: Berlin, Heidelberg, 2006; Volume 103. [Google Scholar] [CrossRef]
- Fesenko, O.; et al. Graphene-enhanced Raman spectroscopy of thymine adsorbed on single-layer graphene. Nanoscale Res. Lett 2015, 10, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Cox, G. Biological applications of second harmonic imaging. Biophys. Rev. 2011, 3, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Dovbeshko, G.; Fesenko, O.; Dementjev, A.; Karpicz, R.; Fedorov, V.; Posudievsky, O.Y. Coherent anti-Stokes Raman scattering enhancement of thymine adsorbed on graphene oxide. Nanoscale Res Lett 2014, 9, 263. [Google Scholar] [CrossRef] [PubMed]
- Dementjev, A.; Rutkauskas, D.; Polovy, I. Mindaugas Macernis, Darius Abramavicius, Leonas Valkunas & Galina Dovbeshko Characterization of thymine microcrystals by CARS and SHG microscopy. Sci Rep 2020, 10, 17097. [Google Scholar] [CrossRef]
- Hnatiuk, S.I.; Dovbeshko, G.I. Studies of mitochondria – FT-IR and nano-IR. Conference Proceedings of the Youth Science League. Priority directions and vectors of development of world science. November 19, Drohobych, Ukraine. 2021. 82-85.
- Chen, X.; Nadiarynkh, O.; Plotnikov, S.; Campagnola, P.J. Second harmonic generation microscopy for quantitative analysis of collagen fibrillar structure. Nat. Protoc 2012, 7, 654–669. [Google Scholar] [CrossRef] [PubMed]
- Wurpel, G.W.; Rinia, H.A.; Müller, M. Imaging orientational order and lipid density in multilamellar vesicles with multiplex CARS microscopy. J Microsc. 2005, 218 Pt 1, 37–45. [Google Scholar] [CrossRef]
- Chen, H.; Li, X.; Zhang, S.; Yang, H.; Gao, Q.; Zhou, F. Rapid and sensitive detection of esophageal cancer by FTIR spectroscopy of serum and plasma. Photodiagnosis and Photodynamic Therapy 2022, 40, 103177. [Google Scholar] [CrossRef]
- Malins, D.C.; Polissar, N.L.; Nishikida, K.; Holmes, E.H.; Gardner, H.S.; Gunselman, S.J. The etiology and prediction of breast cancer. Fourier transform-infrared spectroscopy reveals progressive alterations in breast DNA leading to a cancer-like phenotype in a high proportion of normal women. Cancer 1995, 75, 503–517. [Google Scholar] [CrossRef]
- Pyaskovskaya, O.N.; Dasyukevich, O.I.; Kolesnik, D.L.; Garmanchouk, L.V.; Todor, I.N.; Solyanik, G.I. Changes in VEGF level and tumor growth characteristics during lewis lung carcinoma progression towards cis-DDP resistance. Exp Oncol. 2007, 29, 197–202. [Google Scholar]
- Pyaskovskaya, O.N.; Kolesnik, D.L.; Garmanchouk, L.V.; Yanish, Y.u.V.; Solyanik, G.I. Role of tumor/endothelial cell interactions in tumor growth and metastasis. Exp Oncol 2021, 43, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Parker, F.S. Biochemical Applications of Infrared and Raman Spectroscopy / F.S. Parker. New York: Plenum Press. 1983. p. 528 p.
- Kolesnik, D.L.; Pyaskovskaya, O.N.; Gnatyuk, O.P.; et al. The effect of 2D tungsten disulfide nanoparticles on Lewis lung carcinoma cells in vitro. RSC Advances 2021, 11, 16142–16150. [Google Scholar] [CrossRef]
- Gnatyuk, O.P.; Dovbeshko, G.I.; Yershov, A.; et al. 2D-BN nanoparticles as a spectroscopic marker and drug delivery system with protection properties. RSC Advanced 2018, 8, 30404–30411. [Google Scholar] [CrossRef]
- Mourant, J.R.; Yamada, Y.R.; Carpenter, S.; Dominique, L.R.; Freyer, J.P. FTIR spectroscopy demonstrates biochemical differences in mammalian cell cultures at different growth stages. Biophys J. 2003, 85, 1938–1947. [Google Scholar] [CrossRef]
- Barth, A. Infrared spectroscopy of proteins. Biochim Biophys Acta 2007, 1767, 1073–1101. [Google Scholar] [CrossRef] [PubMed]
- Movasaghi, Z.; Rehman, S.; Rehman, I. Fourier Transform Infrared (FTIR) Spectroscopy of Biological Tissues. Applied Spectroscopy Reviews 2008, 43, 134–179. [Google Scholar] [CrossRef]
- Badger, R.M.; Bauer, S.H. Spectroscopic studies of the hydrogen bond. II. The shift of the O\H vibrational frequency in the formation of the hydrogen bond. J. Chem. Phys. 1937, 5, 839–851. [Google Scholar] [CrossRef]
- Badger, R.M.; Bauer, S.H. Spectroscopic Studies of the Hydrogen Bond I. A Photometric Investigation of the Association Equilibrium in the Vapor of Acetic Acid. J. Chem. Phys. 1937, 5, 605–608. [Google Scholar] [CrossRef]
- Lutz, H.D.; Eckers, W.; Haeuseler, H. OH stretching frequencies of solid hydroxides and of free OH− ions. Journal of Molecular Structure 1982, 80, 221–224. [Google Scholar] [CrossRef]
- Kumar, S.; Verma, T.; Mukherjee, R.; Ariese, F.; Somasundaram, K.; Umapathy, S. Raman and infra-red microspectroscopy: towards quantitative evaluation for clinical research by ratiometric analysis. Chem. Soc. Rev. 2016, 10.1039, C5CS00540J. [Google Scholar] [CrossRef]
- Huang, Y.S.; Karashima, T.; Yamamoto, M.; Hamaguchi, H.O. Molecular-level investigation of the structure, transformation, and bioactivity of single living fission yeast cells by time- and space-resolved Raman spectroscopy. Biochemistry 2005, 44, 10009–10019. [Google Scholar] [CrossRef] [PubMed]
- Dovbeshko, G.; Gnatyuk, O.; Dementjev, A.; Rutkauskas, D.; Kovalska, E.; Baldycheva, A.; Ilchenko, O.; Krasnenkov, D.; Kaplas, T. Coherent anti-stokes Raman scattering spectroscopy (CARS) and imaging of DNA on graphene layers and glass covers. FlatChem. 2021, 27, 100243. [Google Scholar] [CrossRef]
- Izdebska, M.; Zielińska, W.; Grzanka, D.; Gagat, M. The Role of Actin Dynamics and Actin-Binding Proteins Expression in Epithelial-to-Mesenchymal Transition and Its Association with Cancer Progression and Evaluation of Possible Therapeutic Targets. BioMed Research International 2018, 4578373. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; et al. Fiji: an open-source platform for biological-image analysis. Nat Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
- Shihan, M.H.; Novo, S.G.; Le Marchand, S.J.; Wang, Y.; Duncan, M.K. A simple method for quantitating confocal fluorescent images. Biochem Biophys Rep. 2021, 25, 100916. [Google Scholar] [CrossRef]
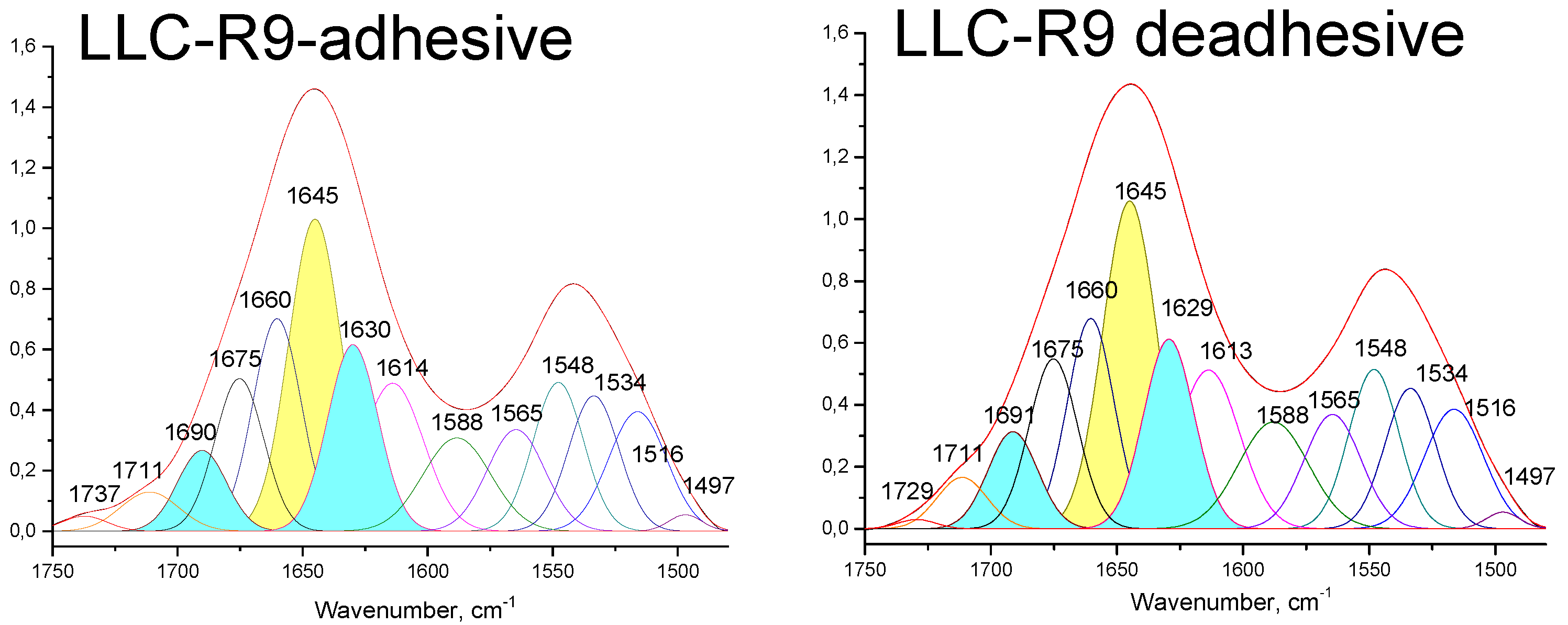
| Secondary structure | LLC-R9 adhesive | LLC-R9 de-adhesive | ||
| Position, cm-1 | Contribution$$$$$in % of total | Position, cm-1 | Contribution$$$$$in % of total | |
| α-helix | 1645 | 26.2 | 1645 | 26.4 |
| β-sheet | 1630, 1690 | 21.3 | 1629, 1691 | 21.5 |
| Turns | 1675 | 11.8 | 1676 | 12.3 |
| Side groups | 1614 | 14.6 | 1613 | 14.8 |
| Disordered form | 1660 | 16.3 | 1660 | 14.8 |
| Cell type | MFI |
| MAEC-2 | 45.6 |
| MAEC-5 | 42.1 |
| LLC-R9- adgesive -2 | 72.1 |
| LLC-R9- adgesive -4 | 67.3 |
| LLC-R9-de- adgesive -1 | 46.1 |
| LLC-R9-de- adgesive -2 | 38.0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




