Submitted:
07 October 2024
Posted:
07 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design and Ethical considerations
2.2. Patient Selection and Treatment
2.3. Sample Collection and Evaluation
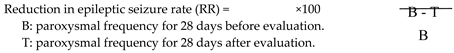
2.4. Statistical Analysis
3. Results
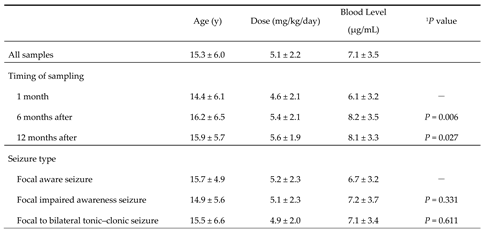
3.1. Details of Patients’ Age Ranges, Doses, and Blood Levels by Timing of Sampling and Seizure Type
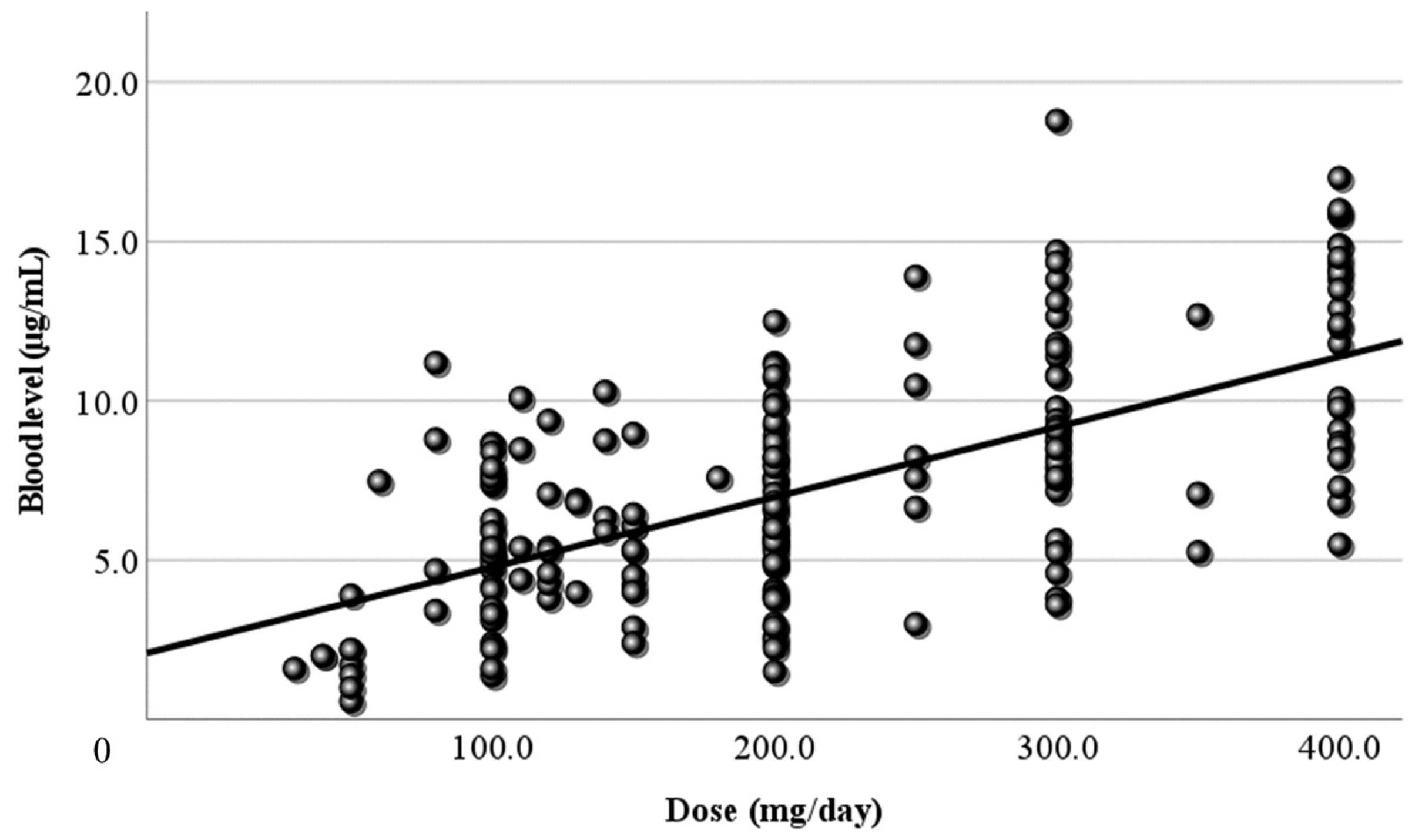
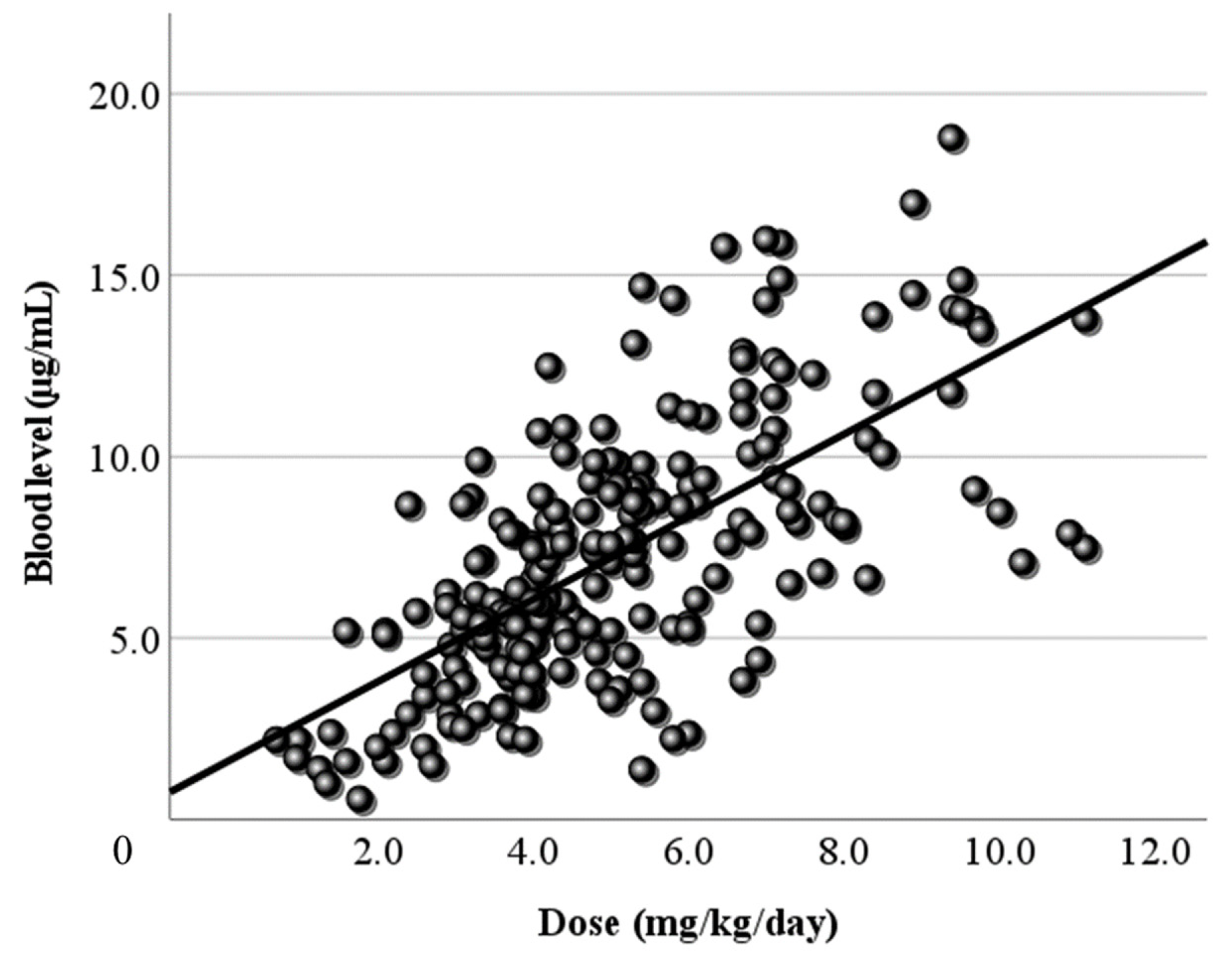
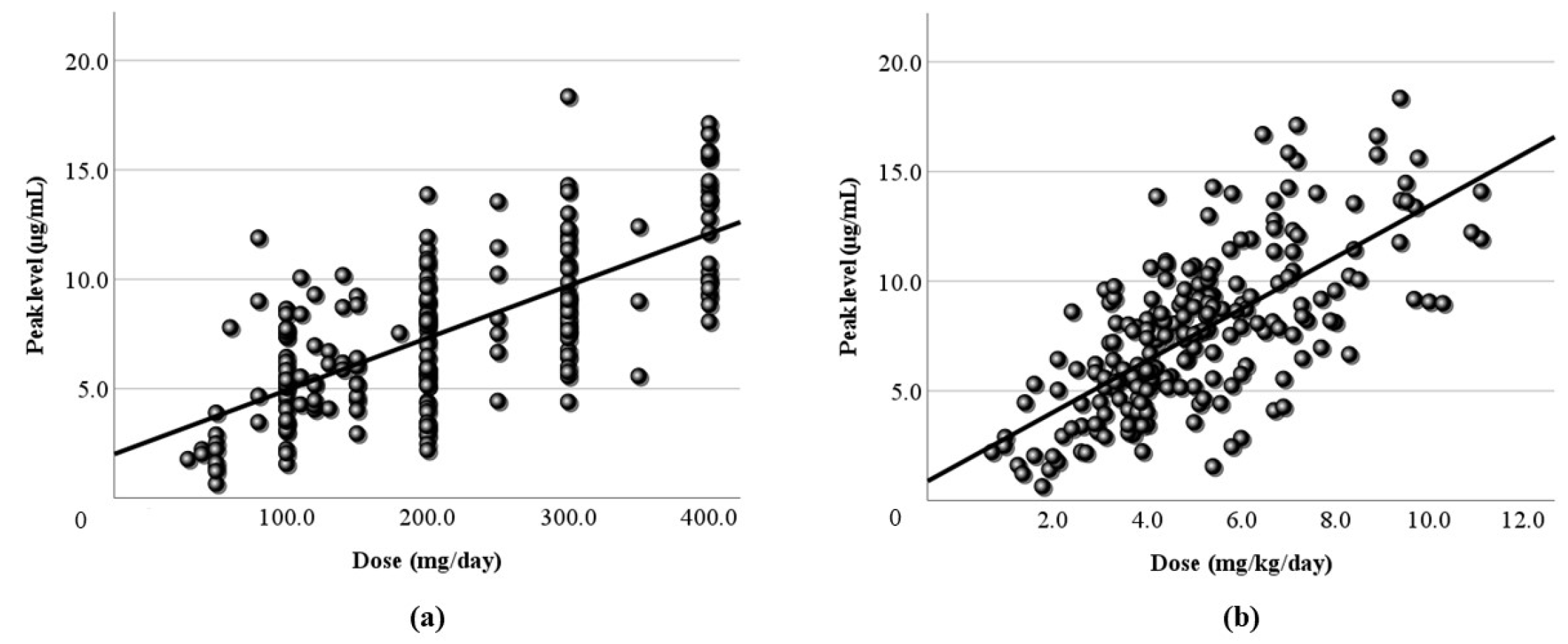
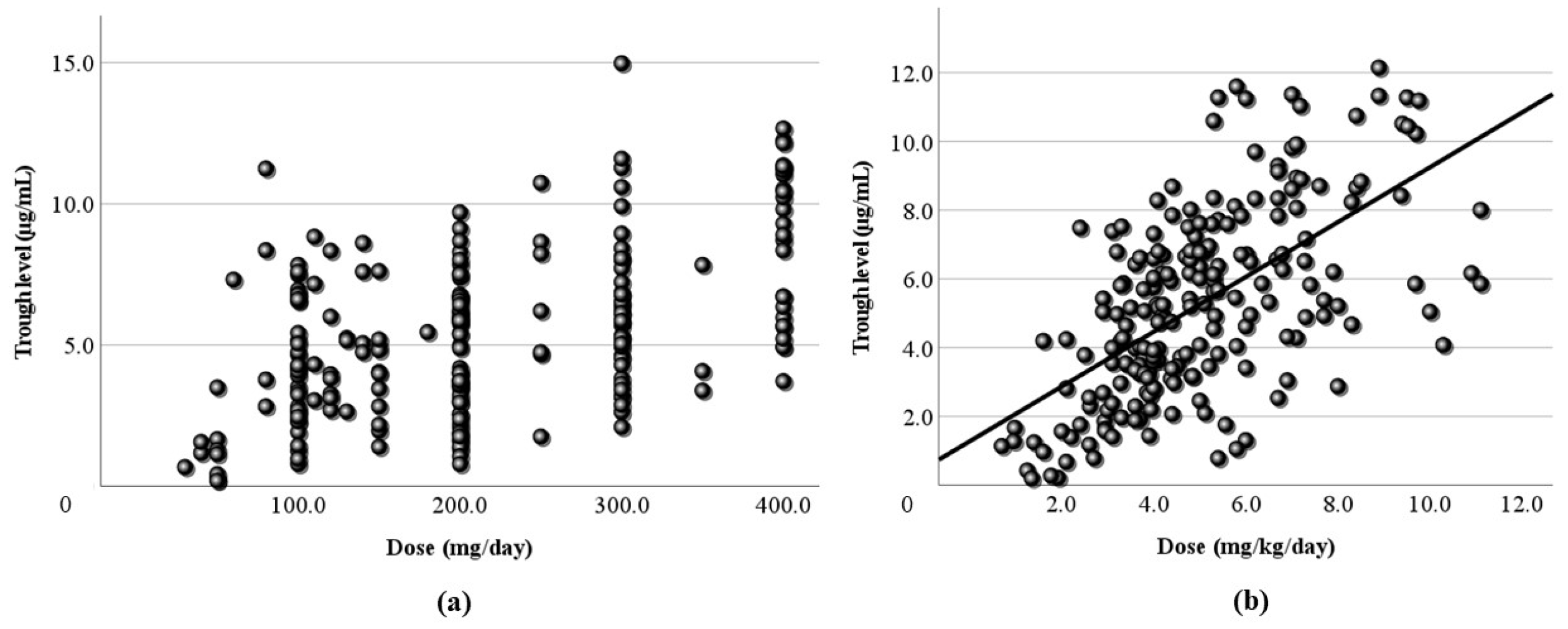
3.2. Relationship Between Dose and Blood Level
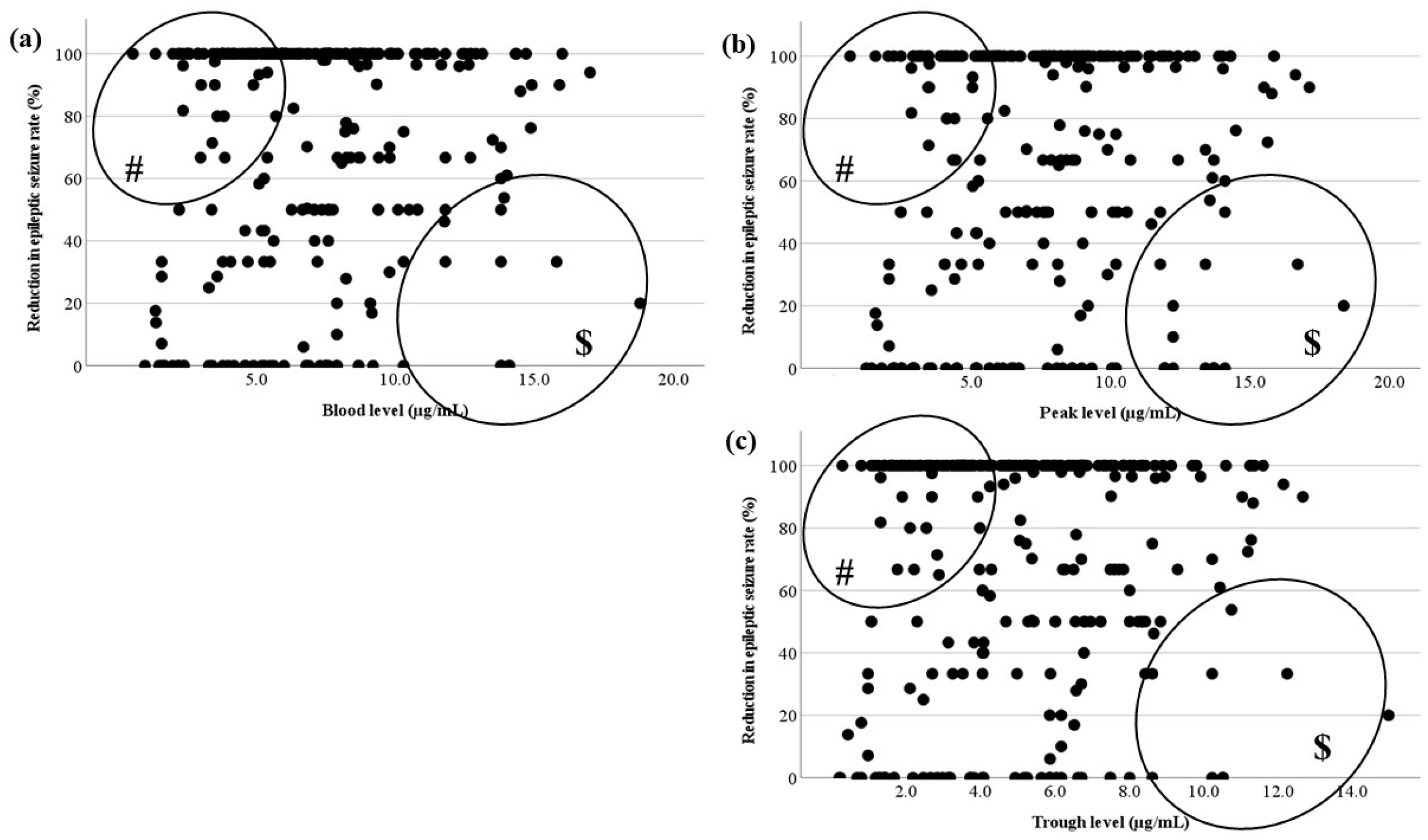
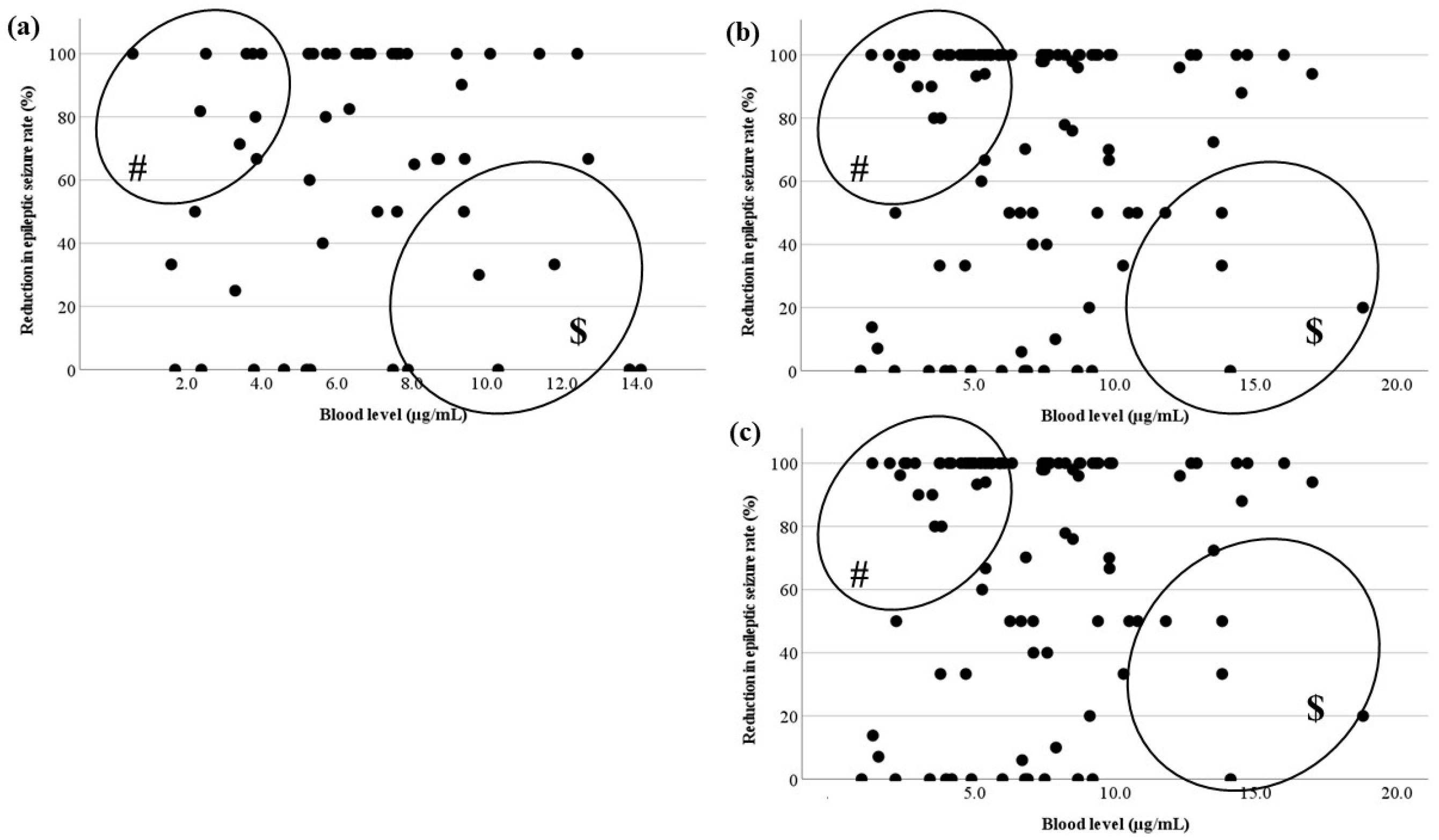
3.3. Relationship between the Blood Level and RR
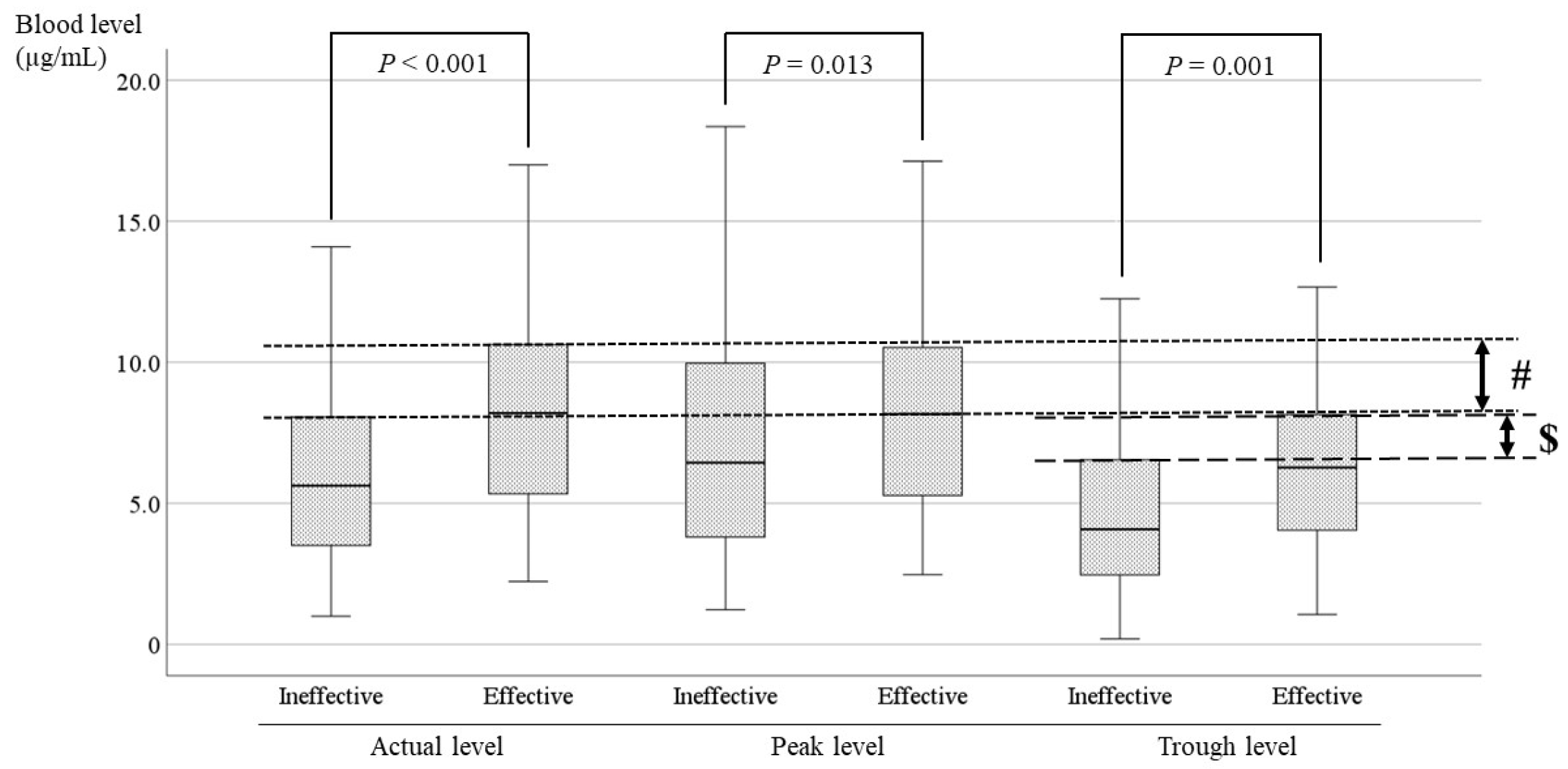
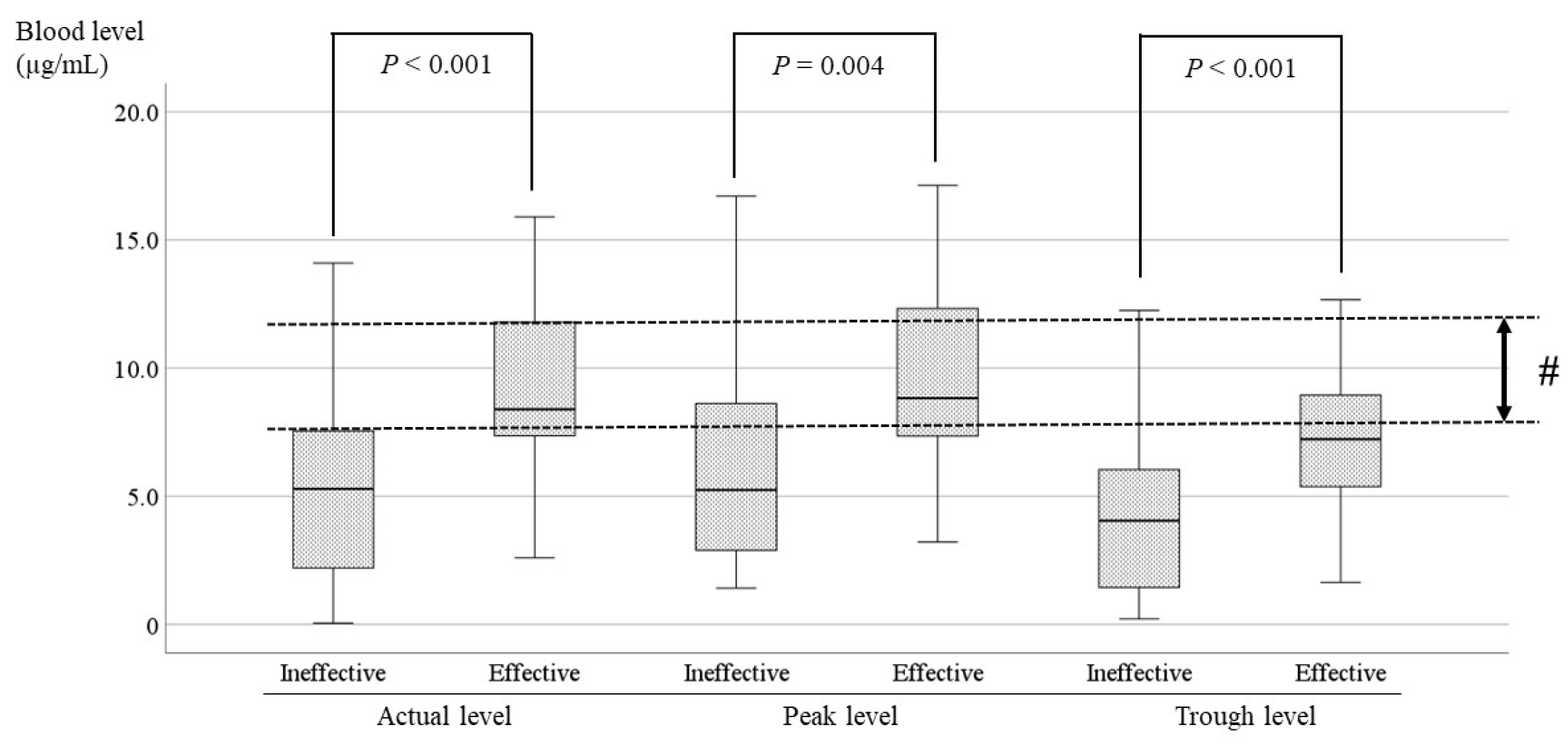
3.4. Comparisons of the LCM blood Level between the Effective and Ineffective Cases and Identification of the Optimal Range
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Errington, A.C.; Coyne, L.; Stöhr, T.; Selve, N.; Lees, G. Seeking a mechanism of action for the novel anticonvulsant lacosamide. Neuropharmacology 2006, 50(8), 1016-1029. [CrossRef]
- Errington, A.C.; Stöhr, T.; Heers, C.; Lees, G. The investigational anticonvulsant lacosamide selectively enhances slow inactivation of voltage-gated sodium channels. Mol Pharmacol 2008, 73(1), 157–169. [CrossRef]
- Niespodziany, I.; Leclère, N.; Vandenplas, C.; Foerch, P.; Wolff, C. Comparative study of lacosamide and classical sodium channel blocking antiepileptic drugs on sodium channel slow inactivation. J Neurosci Res, 2013, 91(3), 436-443. [CrossRef]
- Kohn, E.; Lezinger, M.; Daniel, S.; Masarwi, M;, Brandriss, N.; Bar-Chaim, A.: Berkovitch, M.; Heyman, E.; Komargodski, R. Therapeutic drug monitoring of lacosamide among children: is it helpful? Front Pharmacol 2023 Jul 6, 14, 1164902. [CrossRef]
- Steinhoff, B.J.; Hübers, E.; Kurth, C.; Jürges, Kehl-Kork.U. Plasma concentration and clinical effects of perampanel-The Kork experience. Seizure 2019 Apr, 67, 18-22. [CrossRef]
- Giroux, PC.; Salas-Prato, M.; Théorêt, Y.; Carmant, L. Levetiracetam in children with refractory epilepsy: lack of correlation between plasma concentration and efficacy. Seizure 2009 Oct, 18(8), 559-563. [CrossRef]
- Cawello, W. Clinical pharmacokinetic and pharmacodynamic profile of lacosamide. Clin Pharmacokinet. 2015 Sep, 54(9), 901-914. [CrossRef]
- Scheffer, I.E.; Berkovic, S.; Capovilla, G.; Connolly, MB.; French, J.; Guilhoto, L.; Hirsch, E.; Jain, S.; Mathern, G.W.; Moshé, S.L.; Nordli, D.R.; Perucca, E.; Tomson, T.; Wiebe, S.; Zhang, Y-H.; Zuberi, S.M. ILAE classification of the epilepsies: Position paper of the ILAE Commission for Classification and Terminology. Epilepsia. 2017 Apr, 58(4), 512-521. [CrossRef]
- Fisher, R.S.; Cross, J.H.; French, J.A.; Higurashi, N.; Hirsch, E.; Jansen, F.E.; Lagae, L.; Moshé, S.L.; Peltola, J.; Perez, E.R.; Scheffer, I.E.; Zuberi, S.M. Operational classification of seizure types by the International League Against Epilepsy: Position Paper of the ILAE Commission for Classification and Terminology. Epilepsia. 2017 Apr, 58(4), 522-530. [CrossRef]
- Casas-Fernández, C.; Martínez-Bermejo, A.; Rufo-Campos, M.; Smeyers-Durá, P.; Herranz-Fernández, J.L.; Ibáñez-Micó, S.; Campistol-Plana, J.; Alarcón-Martínez, H.; Campos-Castelló, J. Efficacy and tolerability of lacosamide in the concomitant treatment of 130 patients under 16 years of age with refractory epilepsy: a prospective, open-label, observational, multicenter study in Spain. Drugs R D. 2012 Dec 1, 12(4), 187-197. [CrossRef]
- Gavatha, M.; Ioannou, I.; Papavasiliou, A.S. Efficacy and tolerability of oral lacosamide as adjunctive therapy in pediatric patients with pharmacoresistant focal epilepsyy. Epilepsy Behav. 2011 Apr, 20(4), 691-693. [CrossRef]
- Yorns, W.R. Jr.; Khurana, D.S.; Carvalho, K.S.; Hardison, H.H.; Legido, A.; Valencia, I. Efficacy of lacosamide as adjunctive therapy in children with refractory epilepsy. J Child Neurol. 2014 Jan, 29(1), 23-27. [CrossRef]
- Cawello, W.; Nickel, B.; Eggert-Formella, A. No pharmacokinetic interaction between lacosamide and carbamazepine in healthy volunteers. J Clin Pharmacol. 2010 Apr, 50(4), 459-471. [CrossRef]
- Cawello, W.; Bonn, R. No pharmacokinetic interaction between lacosamide and valproic acid in healthy volunteers. J Clin Pharmacol. 2012 Nov, 52(11), 1739-48. [CrossRef]
- Ahn, S.J.; Oh, J.; Kim, D.Y.; Son, H.; Hwang, S.; Shin, H.R.; Kim, E.Y.; Lee, H.S.; Lee, W.J.; Moon, J.; Lee, S.T.; Jung, K.H.; Park, K.I.; Jung, K.Y.; Lee, S.; Yu, K.S.; Chu, K.; Lee, S.K. Effects of CYP2C19 genetic polymorphisms on the pharmacokinetics of lacosamide in Korean patients with epilepsy. Epilepsia. 2022 Nov, 63(11), 2958-2969. [CrossRef]
- Beyreuther, B.K.; Freitag, J.; Heers, C.; Krebsfänger, N.; Scharfenecker, U.; Stöhr, T. Lacosamide: a review of preclinical properties. CNS Drug Rev. 2007 Spring, 13(1), 21-42. [CrossRef]
- Wolff, C.; Carrington, B.; Varrin-Doyer, M.; Vandendriessche, A.; Van der Perren, C.; Famelart, M.; Gillard, M.; Foerch, P.; Rogemond, V.; Honnorat, J.; Lawson, A.; Miller, K. Drug binding assays do not reveal specific binding of lacosamide to collapsin response mediator protein 2 (CRMP-2). CNS Neurosci Ther. 2012 Jun, 18(6), 493-500. [CrossRef]
- Shiloh-Malawsky, Y.; Fan, Z.; Greenwood, R.; Tennison, M. Successful treatment of childhood prolonged refractory status epilepticus with lacosamide. Seizure. 2011 Sep, 20(7), 586-588. [CrossRef]
- Ishikawa, N.; Eguchi, Y.; Izumo, H.; Tateishi, Y.; Tani, H.; Kobayashi, Y.; Okada, S. Clinical impact of the dose and blood concentration of lacosamide in Japanese pediatric patients with epilepsy: A cohort study. Epilepsy Behav. 2022 Apr, 129, 108614. [CrossRef]
- Zhao, T.; Yu, L.H.; Zhang, HL.; Yu, J.; Feng, J.; Wang, T.T.; Sun, Y.; Li, H.J. Long-term effectiveness and safety of lacosamide as adjunctive therapy in children and adolescents with refractory epilepsy: a real-world study. BMC Pediatr. 2023 May 20, 23(1), 249. [CrossRef]
- Hiemke, C.; Baumann, P.; Bergemann, N.; Conca, A.; Dietmaier, O.; Egberts, K.; Fric, M.; Gerlach, M.; Greiner, C.; Gründer, G.; Haen, E.; Havemann-Reinecke, U.; Jaquenoud Sirot, E.; Kirchherr, H.; Laux, G.; Lutz, U.C.; Messer, T.; Müller, M.J.; Pfuhlmann, B.; Rambeck, B.; Riederer, P.; Schoppek, B.; Stingl, J.; Uhr, M.; Ulrich, S.; Waschgler, R.; Zernig, G. AGNP consensus guidelines for therapeutic drug monitoring in psychiatry: Update 2011. Pharmacopsychiatry. 2011 Sep, 44(6), 195-235.
- Vossler, DG.; Knake, S.; O'Brien, T.J.; Watanabe, M.; Brock, M.; Steiniger-Brach, B.; Williams, P.; Roebling, R.; SP0982 co-investigators. Efficacy and safety of adjunctive lacosamide in the treatment of primary generalised tonic-clonic seizures: a double-blind, randomised, placebo-controlled trial. J Neurol Neurosurg Psychiatry. 2020 Oct, 91(10), 1067-1075. [CrossRef]
- Li, Y.; Guo, HL.; Zhang, Y.Y.; Dong, N.; Hu, Y.H.; Chen, J.; Lu, XP.; Chen, F. Plasma lacosamide monitoring in children with epilepsy: Focus on reference therapeutic range and influencing factors. Front Pediatr. 2022 Sep 7, 10, 949783. [CrossRef]
- Terada, K.; Yamamoto, T.; Yokoyama, T.; Ikeda, K.; Hoshii, N. Long-term Efficacy and Safety of Conversion to Lacosamide Monotherapy in Japanese Adults with Partial-onset Seizures: A Multicenter, Open-label, Phase 3 Trial. Shinryo to Shinyaku. 2019, 56(1), 1-10. (in Japanese).
- Inoue, Y.; Osakabe, T.; Hirano, K.; Shimizu, S. Tolerability of adjunctive therapy with lacosamide, a novel antiepileptic drug, in adult epilepsy patiens: Secondary analysis of data from a double-blind comparative trial and an ongoing long-term open trial. Jpn. J. Clin. Psychophaemacol. 2017, 20(4), 439-453. (in Japanese).
- Aícua-Rapún, I.; André, P.; Rossetti, A.O.; Ryvlin, P.; Hottinger, AF.; Decosterd, L.A.; Buclin, T.; Novy, J. Therapeutic Drug Monitoring of Newer Antiepileptic Drugs: A Randomized Trial for Dosage Adjustment. Ann Neurol. 2020 Jan, 87(1), 22-29. [CrossRef]
- Patsalos, P.N.; Spencer, E.P.; Berry, D.J. Therapeutic Drug Monitoring of Antiepileptic Drugs in Epilepsy: A 2018 Update. Ther Drug Monit. 2018 Oct, 40(5), 526-548. [CrossRef]
- Jacob, S.; Nair, AB.; An Updated Overview on Therapeutic Drug Monitoring of Recent Antiepileptic Drugs. Drugs R D. 2016 Dec, 16(4), 303-316. [CrossRef]
- Asadi-Pooya AA, Farazdaghi M. Idiopathic generalized epilepsies: Which seizure type is more difficult to control? J Clin Neurosci. 2023 Aug; 114: 93-96. [CrossRef]
- Duy PQ, Krauss GL, Crone NE, Ma M, Johnson EL. Antiepileptic drug withdrawal and seizure severity in the epilepsy monitoring unit. Epilepsy Behav. 2020 Aug: 109: 107128. [CrossRef]
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




