Submitted:
06 October 2024
Posted:
08 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Basics of Extracellular Vesicles (EVs)
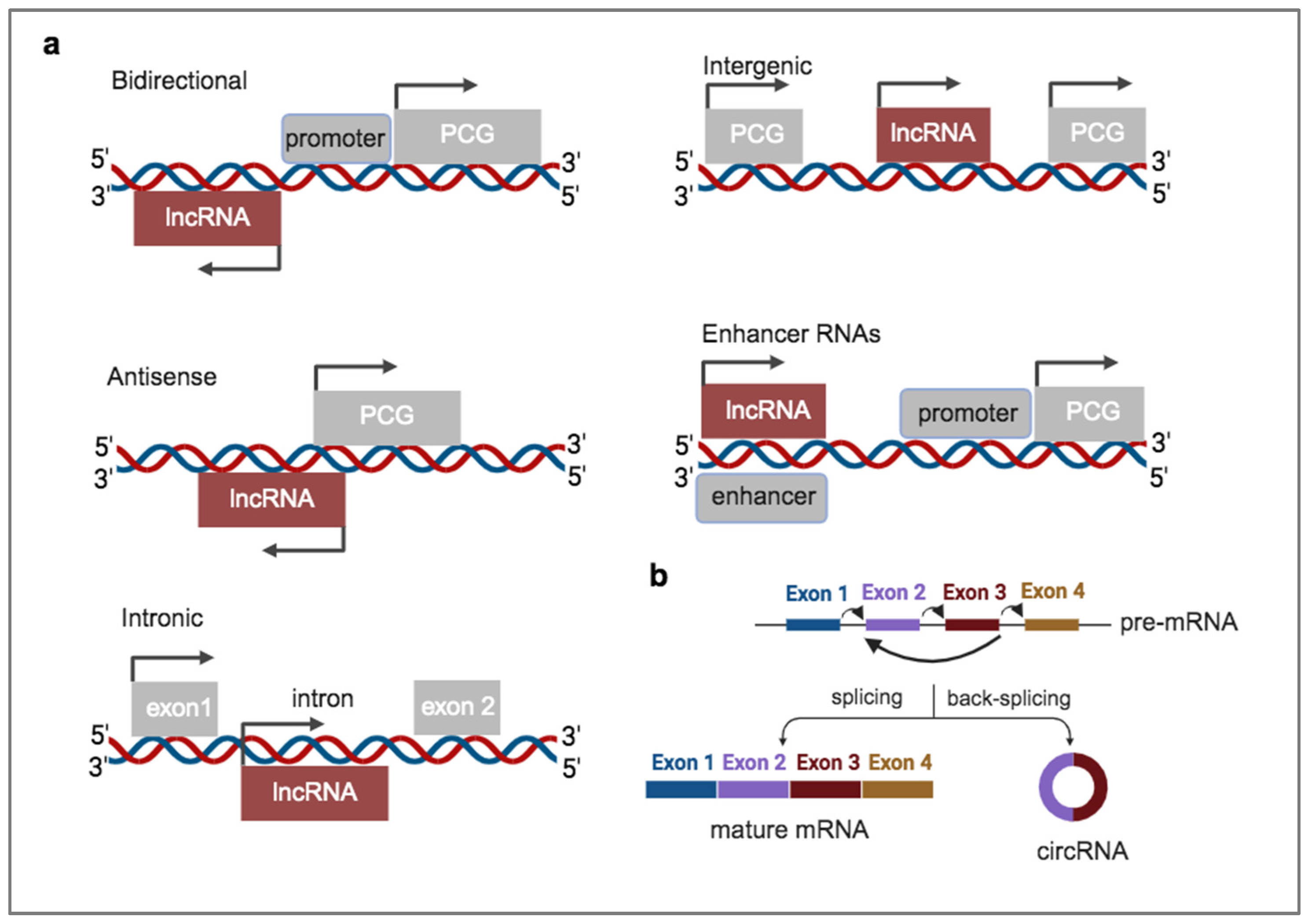
1.2. A plethora of Diverse RNA Biotypes Can Be Packaged into EVs
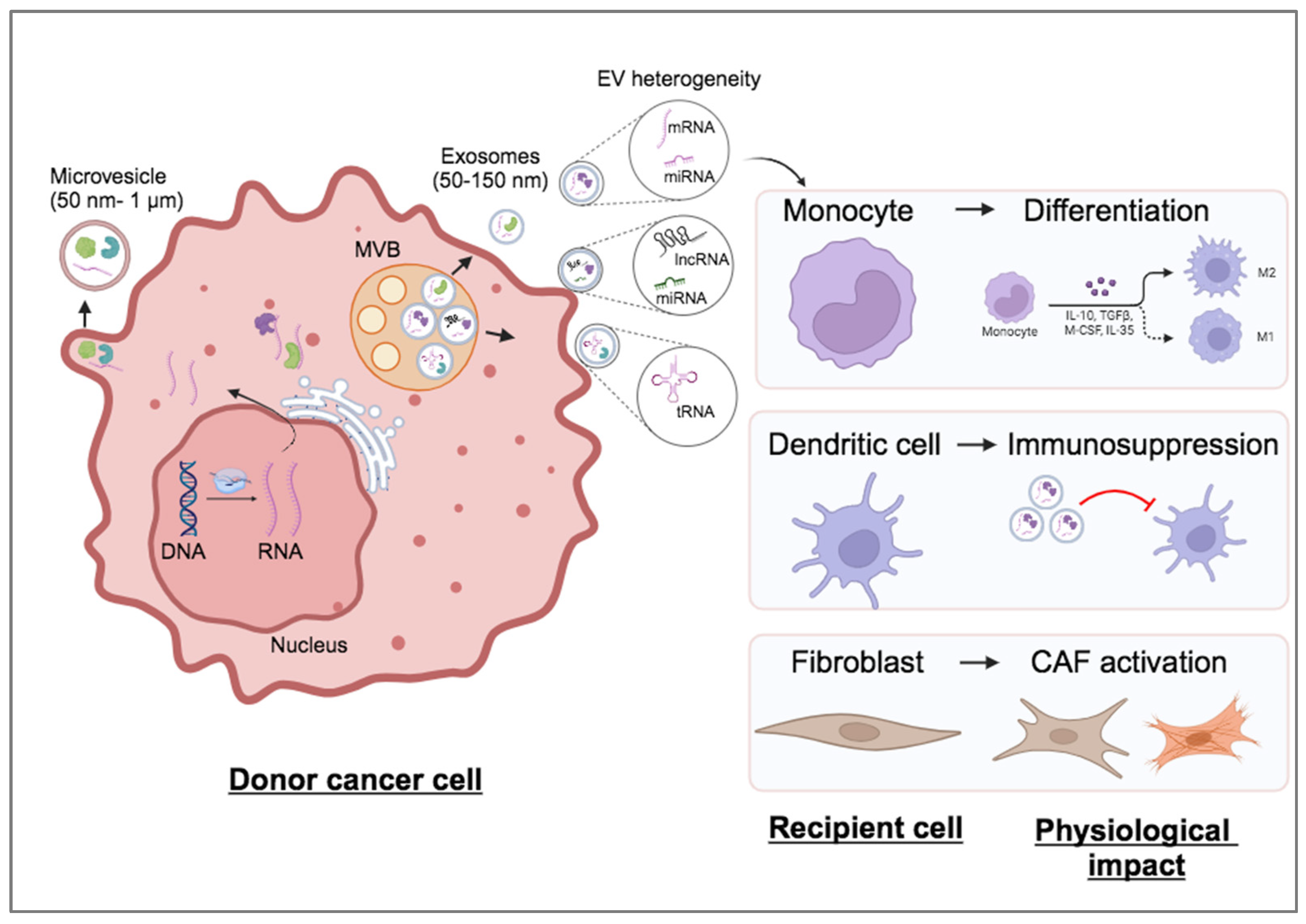
1.3. Sorting and Loading of RNAs into EVs: Active or Passive Process?
1.4. Functionality and Fate of Transferred EV RNA in Recipient Cells
1.5. EVs Possess Multiple Functional Roles during Cancer Progression
2. EV-ncRNAs with Functional Relevance in Cancer
2.1. EV-ncRNAs in Breast Cancer
2.2. EV-ncRNAs in Prostate Cancer
2.3. EV-ncRNAs in Liver Cancer
2.4. EV-ncRNAs in Pancreatic Cancer
3. EV ncRNA in Cancer Diagnosis and Therapeutics
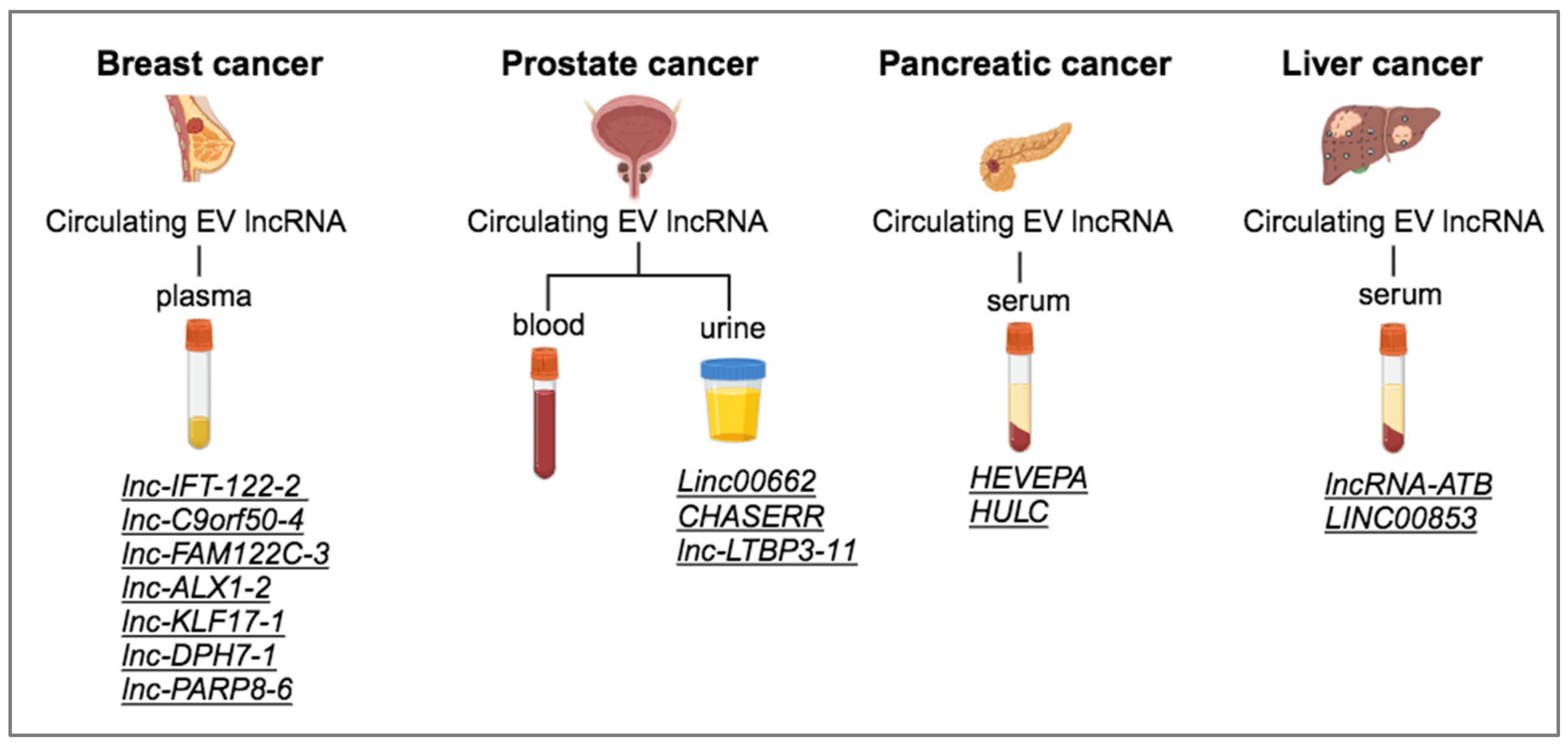
3.1. EV ncRNA as Biomarkers in Cancer
3.2. EVs as Delivery Tools for Therapeutic Purposes
4. Future Perspectives
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Welsh, J.A.; Goberdhan, D.C.I.; O’Driscoll, L.; Buzas, E.I.; Blenkiron, C.; Bussolati, B.; Cai, H.; Di Vizio, D.; Driedonks, T.A.P.; Erdbrügger, U.; et al. Minimal Information for Studies of Extracellular Vesicles (MISEV2023): From Basic to Advanced Approaches. J of Extracellular Vesicle 2024, 13, e12404. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.-Y.; Gonzalez-Kozlova, E.; Soleymani, T.; La Salvia, S.; Kyprianou, N.; Sahoo, S.; Tewari, A.K.; Cordon-Cardo, C.; Stolovitzky, G.; Dogra, N. Extracellular Vesicles Carry Distinct Proteo-Transcriptomic Signatures That Are Different from Their Cancer Cell of Origin. iScience 2022, 25, 104414. [Google Scholar] [CrossRef] [PubMed]
- Van Niel, G.; D’Angelo, G.; Raposo, G. Shedding Light on the Cell Biology of Extracellular Vesicles. Nat Rev Mol Cell Biol 2018, 19, 213–228. [Google Scholar] [CrossRef] [PubMed]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal Information for Studies of Extracellular Vesicles 2018 (MISEV2018): A Position Statement of the International Society for Extracellular Vesicles and Update of the MISEV2014 Guidelines. J of Extracellular Vesicle 2018, 7, 1535750. [Google Scholar] [CrossRef]
- Kowal, J.; Arras, G.; Colombo, M.; Jouve, M.; Morath, J.P.; Primdal-Bengtson, B.; Dingli, F.; Loew, D.; Tkach, M.; Théry, C. Proteomic Comparison Defines Novel Markers to Characterize Heterogeneous Populations of Extracellular Vesicle Subtypes. Proc. Natl. Acad. Sci. USA 2016, 113. [Google Scholar] [CrossRef]
- Wang, X.; Fan, H.; Wang, B.; Yuan, F. Research Progress on the Roles of lncRNAs in Plant Development and Stress Responses. Front. Plant Sci. 2023, 14, 1138901. [Google Scholar] [CrossRef]
- Mattick, J.S.; Amaral, P.P.; Carninci, P.; Carpenter, S.; Chang, H.Y.; Chen, L.-L.; Chen, R.; Dean, C.; Dinger, M.E.; Fitzgerald, K.A.; et al. Long Non-Coding RNAs: Definitions, Functions, Challenges and Recommendations. Nat Rev Mol Cell Biol 2023, 24, 430–447. [Google Scholar] [CrossRef]
- Statello, L.; Guo, C.-J.; Chen, L.-L.; Huarte, M. Gene Regulation by Long Non-Coding RNAs and Its Biological Functions. Nat Rev Mol Cell Biol 2021, 22, 96–118. [Google Scholar] [CrossRef]
- Papoutsoglou, P.; Moustakas, A. Long Non-coding RNAs and TGF-β Signaling in Cancer. Cancer Science 2020, 111, 2672–2681. [Google Scholar] [CrossRef]
- Abramowicz, A.; Story, M.D. The Long and Short of It: The Emerging Roles of Non-Coding RNA in Small Extracellular Vesicles. Cancers (Basel) 2020, 12, 1445. [Google Scholar] [CrossRef]
- Batagov, A.O.; Kurochkin, I.V. Exosomes Secreted by Human Cells Transport Largely mRNA Fragments That Are Enriched in the 3′-Untranslated Regions. Biol Direct 2013, 8, 12. [Google Scholar] [CrossRef]
- Padilla, J.-C.A.; Barutcu, S.; Malet, L.; Deschamps-Francoeur, G.; Calderon, V.; Kwon, E.; Lécuyer, E. Profiling the Polyadenylated Transcriptome of Extracellular Vesicles with Long-Read Nanopore Sequencing. BMC Genomics 2023, 24, 564. [Google Scholar] [CrossRef] [PubMed]
- Ransom, L.S.; Liu, C.S.; Dunsmore, E.; Palmer, C.R.; Nicodemus, J.; Ziomek, D.; Williams, N.; Chun, J. Human Brain Small Extracellular Vesicles Contain Selectively Packaged, Full-Length mRNA. Cell Rep 2024, 43, 114061. [Google Scholar] [CrossRef] [PubMed]
- O’Grady, T.; Njock, M.-S.; Lion, M.; Bruyr, J.; Mariavelle, E.; Galvan, B.; Boeckx, A.; Struman, I.; Dequiedt, F. Sorting and Packaging of RNA into Extracellular Vesicles Shape Intracellular Transcript Levels. BMC Biol 2022, 20, 72. [Google Scholar] [CrossRef] [PubMed]
- Almeida, A.; Gabriel, M.; Firlej, V.; Martin-Jaular, L.; Lejars, M.; Cipolla, R.; Petit, F.; Vogt, N.; San-Roman, M.; Dingli, F.; et al. Urinary Extracellular Vesicles Contain Mature Transcriptome Enriched in Circular and Long Noncoding RNAs with Functional Significance in Prostate Cancer. J of Extracellular Vesicle 2022, 11, e12210. [Google Scholar] [CrossRef] [PubMed]
- Dellar, E.R.; Hill, C.; Melling, G.E.; Carter, D.R.F.; Baena-Lopez, L.A. Unpacking Extracellular Vesicles: RNA Cargo Loading and Function. J Extracell Biol 2022, 1, e40. [Google Scholar] [CrossRef]
- Oka, Y.; Tanaka, K.; Kawasaki, Y. A Novel Sorting Signal for RNA Packaging into Small Extracellular Vesicles. Sci Rep 2023, 13, 17436. [Google Scholar] [CrossRef]
- Corrado, C.; Barreca, M.M.; Zichittella, C.; Alessandro, R.; Conigliaro, A. Molecular Mediators of RNA Loading into Extracellular Vesicles. Cells 2021, 10, 3355. [Google Scholar] [CrossRef]
- Liu, X.-M.; Ma, L.; Schekman, R. Selective Sorting of microRNAs into Exosomes by Phase-Separated YBX1 Condensates. Elife 2021, 10, e71982. [Google Scholar] [CrossRef]
- Wozniak, A.L.; Adams, A.; King, K.E.; Dunn, W.; Christenson, L.K.; Hung, W.-T.; Weinman, S.A. The RNA Binding Protein FMR1 Controls Selective Exosomal miRNA Cargo Loading during Inflammation. J Cell Biol 2020, 219, e201912074. [Google Scholar] [CrossRef]
- Martins-Marques, T.; Costa, M.C.; Catarino, S.; Simoes, I.; Aasen, T.; Enguita, F.J.; Girao, H. Cx43-Mediated Sorting of miRNAs into Extracellular Vesicles. EMBO Rep 2022, 23, e54312. [Google Scholar] [CrossRef] [PubMed]
- Fabbiano, F.; Corsi, J.; Gurrieri, E.; Trevisan, C.; Notarangelo, M.; D’Agostino, V.G. RNA Packaging into Extracellular Vesicles: An Orchestra of RNA-binding Proteins? J of Extracellular Vesicle 2020, 10, e12043. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Singh, J.; Schekman, R. Two RNA-Binding Proteins Mediate the Sorting of miR223 from Mitochondria into Exosomes. Elife 2023, 12, e85878. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, K.; Breyne, K.; Ughetto, S.; Laurent, L.C.; Breakefield, X.O. RNA Delivery by Extracellular Vesicles in Mammalian Cells and Its Applications. Nat Rev Mol Cell Biol 2020, 21, 585–606. [Google Scholar] [CrossRef]
- Mulcahy, L.A.; Pink, R.C.; Carter, D.R.F. Routes and Mechanisms of Extracellular Vesicle Uptake. J Extracell Vesicles 2014, 3. [Google Scholar] [CrossRef]
- Bebelman, M.P.; Bun, P.; Huveneers, S.; van Niel, G.; Pegtel, D.M.; Verweij, F.J. Real-Time Imaging of Multivesicular Body-Plasma Membrane Fusion to Quantify Exosome Release from Single Cells. Nat Protoc 2020, 15, 102–121. [Google Scholar] [CrossRef]
- Choi, W.; Park, D.J.; Eliceiri, B.P. Defining Tropism and Activity of Natural and Engineered Extracellular Vesicles. Front Immunol 2024, 15, 1363185. [Google Scholar] [CrossRef]
- Bonsergent, E.; Grisard, E.; Buchrieser, J.; Schwartz, O.; Théry, C.; Lavieu, G. Quantitative Characterization of Extracellular Vesicle Uptake and Content Delivery within Mammalian Cells. Nat Commun 2021, 12, 1864. [Google Scholar] [CrossRef]
- Lai, C.P.; Kim, E.Y.; Badr, C.E.; Weissleder, R.; Mempel, T.R.; Tannous, B.A.; Breakefield, X.O. Visualization and Tracking of Tumour Extracellular Vesicle Delivery and RNA Translation Using Multiplexed Reporters. Nat Commun 2015, 6, 7029. [Google Scholar] [CrossRef]
- Rufino-Ramos, D.; Leandro, K.; Perdigão, P.R.L.; O’Brien, K.; Pinto, M.M.; Santana, M.M.; van Solinge, T.S.; Mahjoum, S.; Breakefield, X.O.; Breyne, K.; et al. Extracellular Communication between Brain Cells through Functional Transfer of Cre mRNA Mediated by Extracellular Vesicles. Mol Ther 2023, 31, 2220–2239. [Google Scholar] [CrossRef]
- Somiya, M.; Kuroda, S. Verification of Extracellular Vesicle-Mediated Functional mRNA Delivery via RNA Editing 2022.
- Tao, S.-C.; Guo, S.-C. Role of Extracellular Vesicles in Tumour Microenvironment. Cell Commun Signal 2020, 18, 163. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R.; McAndrews, K.M. The Role of Extracellular Vesicles in Cancer. Cell 2023, 186, 1610–1626. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Lu, C.; Yixi, L.; Hong, J.; Dong, F.; Ruan, S.; Hu, T.; Zhao, X. Exosomal Linc00969 Induces Trastuzumab Resistance in Breast Cancer by Increasing HER-2 Protein Expression and mRNA Stability by Binding to HUR. Breast Cancer Res 2023, 25, 124. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhou, Y.; Chen, J.; Yang, J.; Yuan, Y.; Wu, W. Exosomal lncRNA SNHG12 Promotes Angiogenesis and Breast Cancer Progression. Breast Cancer 2024, 31, 607–620. [Google Scholar] [CrossRef]
- Chen, F.; Chen, J.; Yang, L.; Liu, J.; Zhang, X.; Zhang, Y.; Tu, Q.; Yin, D.; Lin, D.; Wong, P.-P.; et al. Extracellular Vesicle-Packaged HIF-1α-Stabilizing lncRNA from Tumour-Associated Macrophages Regulates Aerobic Glycolysis of Breast Cancer Cells. Nat Cell Biol 2019, 21, 498–510. [Google Scholar] [CrossRef]
- Tao, S.; Wang, J.; Li, F.; Shi, B.; Ren, Q.; Zhuang, Y.; Qian, X. Extracellular Vesicles Released by Hypoxia-Induced Tumor-Associated Fibroblasts Impart Chemoresistance to Breast Cancer Cells via Long Noncoding RNA H19 Delivery. FASEB J 2024, 38, e23165. [Google Scholar] [CrossRef]
- Ahadi, A.; Brennan, S.; Kennedy, P.J.; Hutvagner, G.; Tran, N. Long Non-Coding RNAs Harboring miRNA Seed Regions Are Enriched in Prostate Cancer Exosomes. Sci Rep 2016, 6, 24922. [Google Scholar] [CrossRef]
- Li, Q.; Hu, J.; Shi, Y.; Xiao, M.; Bi, T.; Wang, C.; Yan, L.; Li, X. Exosomal lncAY927529 Enhances Prostate Cancer Cell Proliferation and Invasion through Regulating Bone Microenvironment. Cell Cycle 2021, 20, 2531–2546. [Google Scholar] [CrossRef]
- Hu, C.-Y.; Chen, J.; Qin, X.-H.; You, P.; Ma, J.; Zhang, J.; Zhang, H.; Xu, J.-D. Long Non-Coding RNA NORAD Promotes the Prostate Cancer Cell Extracellular Vesicle Release via microRNA-541-3p-Regulated PKM2 to Induce Bone Metastasis of Prostate Cancer. J Exp Clin Cancer Res 2021, 40, 98. [Google Scholar] [CrossRef]
- Kato, T.; Kawakami, K.; Mizutani, K.; Ando, T.; Sakai, Y.; Sakurai, K.; Toyota, S.; Ehara, H.; Ito, H.; Ito, M. H19 in Serum Extracellular Vesicles Reflects Resistance to AR Axis-Targeted Therapy Among CRPC Patients. Cancer Genomics Proteomics 2023, 20, 456–468. [Google Scholar] [CrossRef]
- Rodríguez, M.; Bajo-Santos, C.; Hessvik, N.P.; Lorenz, S.; Fromm, B.; Berge, V.; Sandvig, K.; Linē, A.; Llorente, A. Identification of Non-Invasive miRNAs Biomarkers for Prostate Cancer by Deep Sequencing Analysis of Urinary Exosomes. Mol Cancer 2017, 16, 156. [Google Scholar] [CrossRef] [PubMed]
- Lv, S.; Wang, J.; Li, L. Extracellular Vesicular lncRNA FAL1 Promotes Hepatocellular Carcinoma Cell Proliferation and Invasion by Inducing Macrophage M2 Polarization. J Physiol Biochem 2023, 79, 669–682. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Yan, I.K.; Kogure, T.; Haga, H.; Patel, T. Extracellular Vesicle-Mediated Transfer of Long Non-Coding RNA ROR Modulates Chemosensitivity in Human Hepatocellular Cancer. FEBS Open Bio 2014, 4, 458–467. [Google Scholar] [CrossRef]
- Yao, H.; Huang, C.; Zou, J.; Liang, W.; Zhao, Y.; Yang, K.; Zhong, Z.; Zhou, S.; Li, J.; Li, Y.; et al. Extracellular Vesicle-Packaged lncRNA from Cancer-Associated Fibroblasts Promotes Immune Evasion by Downregulating HLA-A in Pancreatic Cancer. J Extracell Vesicles 2024, 13, e12484. [Google Scholar] [CrossRef]
- Zhang, P.; Wang, Q.; Lu, W.; Zhang, F.; Wu, D.; Sun, J. NNT-AS1 in CAFs-Derived Exosomes Promotes Progression and Glucose Metabolism through miR-889-3p/HIF-1α in Pancreatic Adenocarcinoma. Sci Rep 2024, 14, 6979. [Google Scholar] [CrossRef]
- Cheng, K.; Pan, J.; Liu, Q.; Ji, Y.; Liu, L.; Guo, X.; Wang, Q.; Li, S.; Sun, J.; Gong, M.; et al. Exosomal lncRNA XIST Promotes Perineural Invasion of Pancreatic Cancer Cells via miR-211-5p/GDNF. Oncogene 2024, 43, 1341–1352. [Google Scholar] [CrossRef]
- Liu, Y.; Tang, T.; Yang, X.; Qin, P.; Wang, P.; Zhang, H.; Bai, M.; Wu, R.; Li, F. Tumor-Derived Exosomal Long Noncoding RNA LINC01133, Regulated by Periostin, Contributes to Pancreatic Ductal Adenocarcinoma Epithelial-Mesenchymal Transition through the Wnt/β-Catenin Pathway by Silencing AXIN2. Oncogene 2021, 40, 3164–3179. [Google Scholar] [CrossRef]
- Zayakin, P.; Sadovska, L.; Eglītis, K.; Romanchikova, N.; Radoviča-Spalviņa, I.; Endzeliņš, E.; Liepniece-Karele, I.; Eglītis, J.; Linē, A. Extracellular Vesicles-A Source of RNA Biomarkers for the Detection of Breast Cancer in Liquid Biopsies. Cancers (Basel) 2023, 15, 4329. [Google Scholar] [CrossRef]
- Sadovska, L.; Zayakin, P.; Eglītis, K.; Endzeliņš, E.; Radoviča-Spalviņa, I.; Avotiņa, E.; Auders, J.; Keiša, L.; Liepniece-Karele, I.; Leja, M.; et al. Comprehensive Characterization of RNA Cargo of Extracellular Vesicles in Breast Cancer Patients Undergoing Neoadjuvant Chemotherapy. Front Oncol 2022, 12, 1005812. [Google Scholar] [CrossRef]
- Bajo-Santos, C.; Brokāne, A.; Zayakin, P.; Endzeliņš, E.; Soboļevska, K.; Belovs, A.; Jansons, J.; Sperga, M.; Llorente, A.; Radoviča-Spalviņa, I.; et al. Plasma and Urinary Extracellular Vesicles as a Source of RNA Biomarkers for Prostate Cancer in Liquid Biopsies. Front. Mol. Biosci. 2023, 10, 980433. [Google Scholar] [CrossRef]
- Lee, Y.R.; Kim, G.; Tak, W.Y.; Jang, S.Y.; Kweon, Y.O.; Park, J.G.; Lee, H.W.; Han, Y.S.; Chun, J.M.; Park, S.Y.; et al. Circulating Exosomal Noncoding RNAs as Prognostic Biomarkers in Human Hepatocellular Carcinoma. Int J Cancer 2019, 144, 1444–1452. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.S.; Baek, G.O.; Ahn, H.R.; Sung, S.; Seo, C.W.; Cho, H.J.; Nam, S.W.; Cheong, J.Y.; Eun, J.W. Serum Small Extracellular Vesicle-Derived LINC00853 as a Novel Diagnostic Marker for Early Hepatocellular Carcinoma. Mol Oncol 2020, 14, 2646–2659. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Inuzuka, T.; Shimizu, Y.; Sawamoto, K.; Taniue, K.; Ono, Y.; Asai, F.; Koyama, K.; Sato, H.; Kawabata, H.; et al. Liquid Biopsy for Pancreatic Cancer by Serum Extracellular Vesicle-Encapsulated Long Noncoding RNA HEVEPA. Pancreas 2024, 53, e395–e404. [Google Scholar] [CrossRef]
- Takahashi, K.; Ota, Y.; Kogure, T.; Suzuki, Y.; Iwamoto, H.; Yamakita, K.; Kitano, Y.; Fujii, S.; Haneda, M.; Patel, T.; et al. Circulating Extracellular Vesicle-Encapsulated HULC Is a Potential Biomarker for Human Pancreatic Cancer. Cancer Sci 2020, 111, 98–111. [Google Scholar] [CrossRef]
- Wang, J.; Yin, B.; Lian, J.; Wang, X. Extracellular Vesicles as Drug Delivery System for Cancer Therapy. Pharmaceutics 2024, 16, 1029. [Google Scholar] [CrossRef]
- Payandeh, Z.; Tangruksa, B.; Synnergren, J.; Heydarkhan-Hagvall, S.; Nordin, J.Z.; Andaloussi, S.E.; Borén, J.; Wiseman, J.; Bohlooly, -Y. M.; Lindfors, L.; et al. Extracellular Vesicles Transport RNA between Cells: Unraveling Their Dual Role in Diagnostics and Therapeutics. Molecular Aspects of Medicine 2024, 99, 101302. [Google Scholar] [CrossRef]
- Kumar, M.A.; Baba, S.K.; Sadida, H.Q.; Marzooqi, S.A.; Jerobin, J.; Altemani, F.H.; Algehainy, N.; Alanazi, M.A.; Abou-Samra, A.-B.; Kumar, R.; et al. Extracellular Vesicles as Tools and Targets in Therapy for Diseases. Sig Transduct Target Ther 2024, 9, 27. [Google Scholar] [CrossRef]
- Tang, M.; Chen, Y.; Li, B.; Sugimoto, H.; Yang, S.; Yang, C.; LeBleu, V.S.; McAndrews, K.M.; Kalluri, R. Therapeutic Targeting of STAT3 with Small Interference RNAs and Antisense Oligonucleotides Embedded Exosomes in Liver Fibrosis. FASEB J 2021, 35, e21557. [Google Scholar] [CrossRef]
- Qiu, L.; Liu, X.; Zhu, L.; Luo, L.; Sun, N.; Pei, R. Current Advances in Technologies for Single Extracellular Vesicle Analysis and Its Clinical Applications in Cancer Diagnosis. Biosensors 2023, 13, 129. [Google Scholar] [CrossRef]
- Yousafzai, N.A.; El Khalki, L.; Wang, W.; Szpendyk, J.; Sossey-Alaoui, K. Advances in 3D Culture Models to Study Exosomes in Triple-Negative Breast Cancer. Cancers 2024, 16, 883. [Google Scholar] [CrossRef]
- Hulstaert, E.; Morlion, A.; Avila Cobos, F.; Verniers, K.; Nuytens, J.; Vanden Eynde, E.; Yigit, N.; Anckaert, J.; Geerts, A.; Hindryckx, P.; et al. Charting Extracellular Transcriptomes in The Human Biofluid RNA Atlas. Cell Reports 2020, 33, 108552. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



