Submitted:
03 October 2024
Posted:
04 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
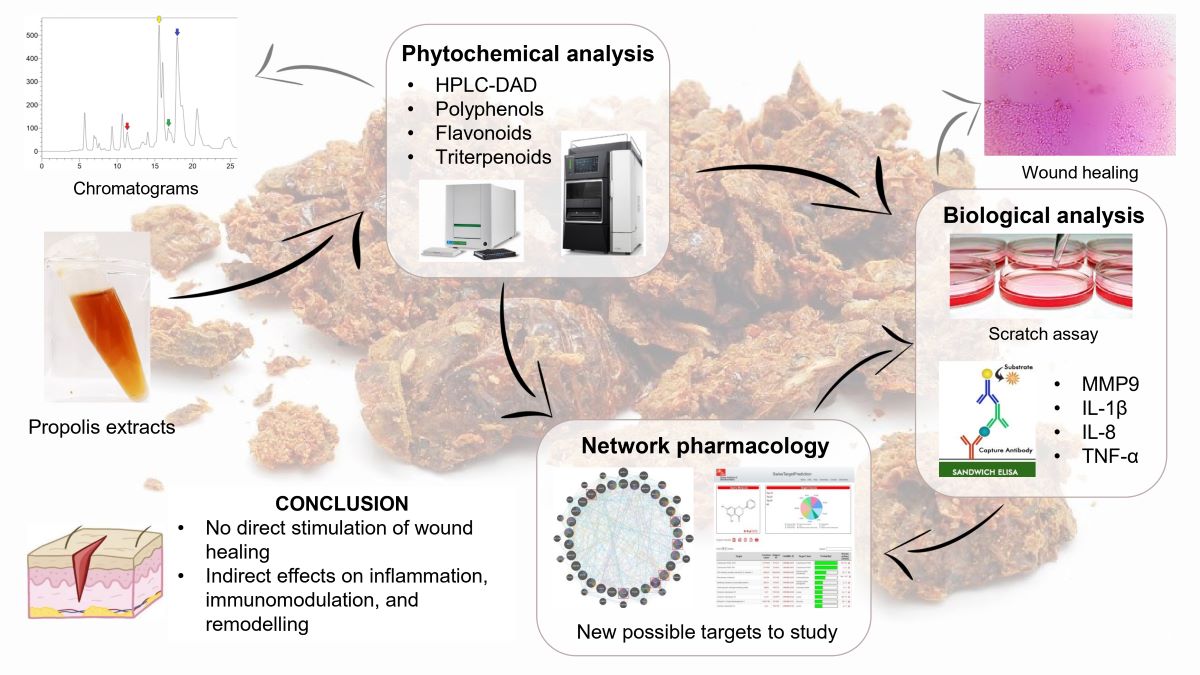
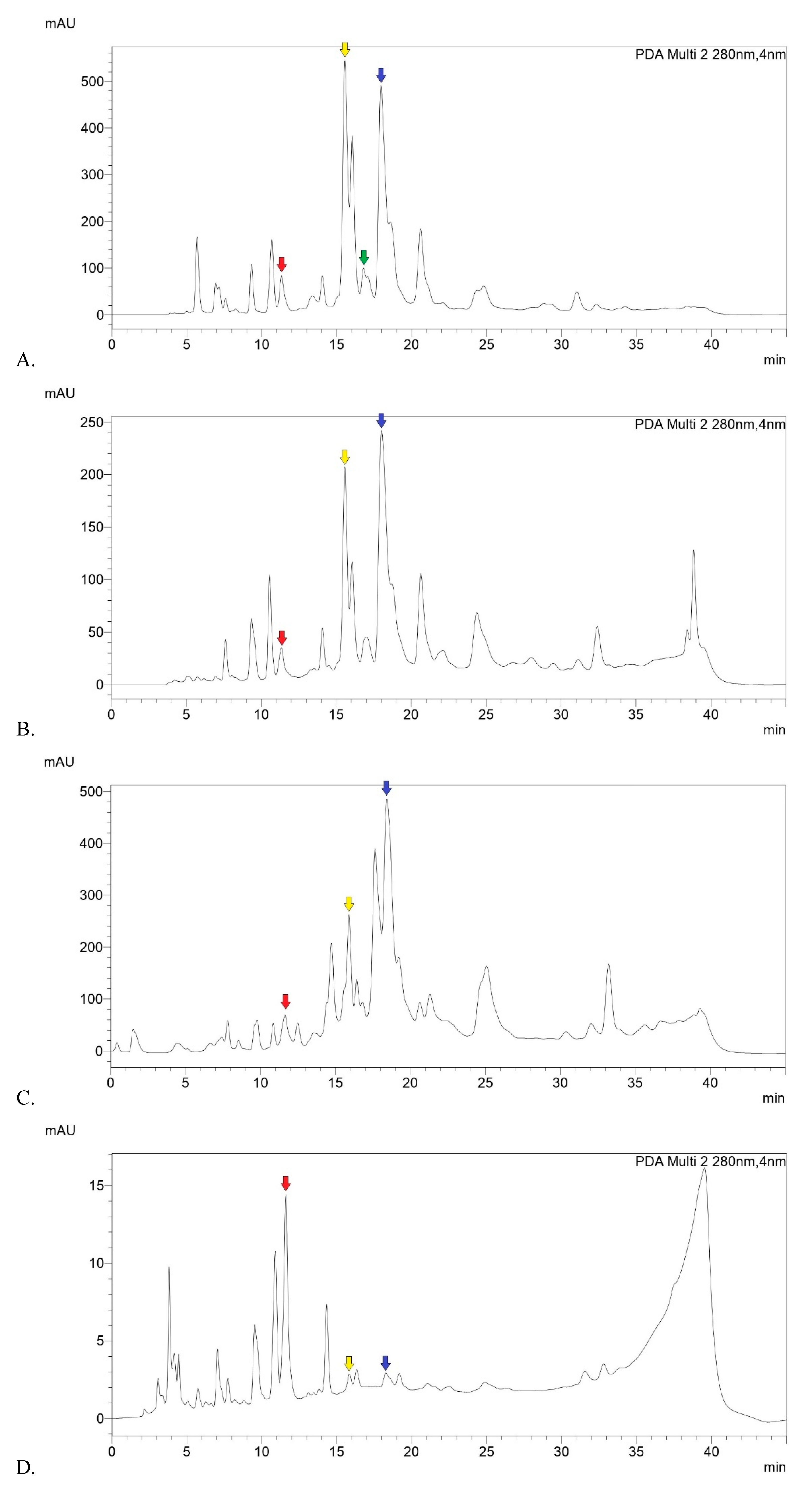
2.1. Phytochemical Analysis
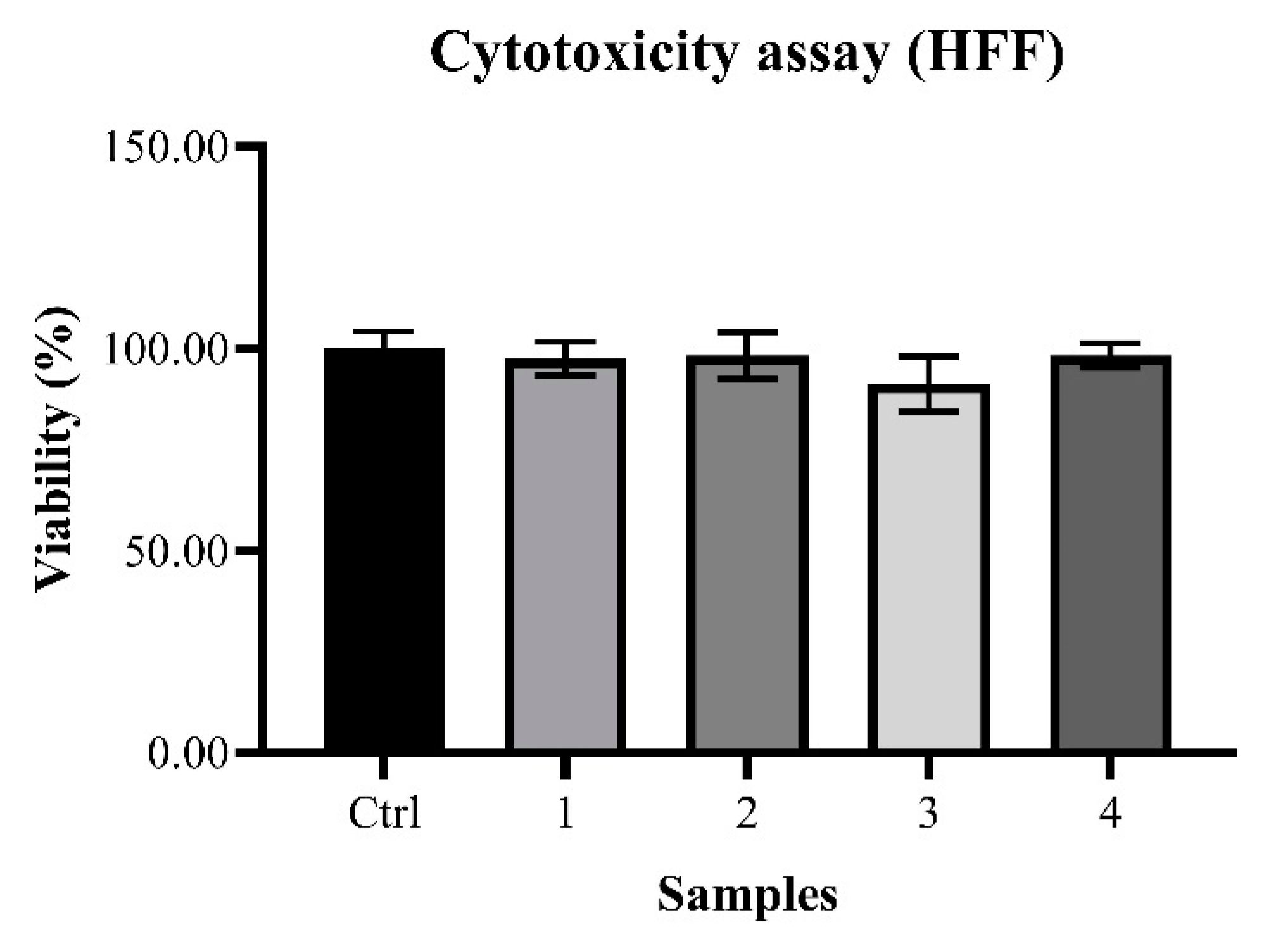
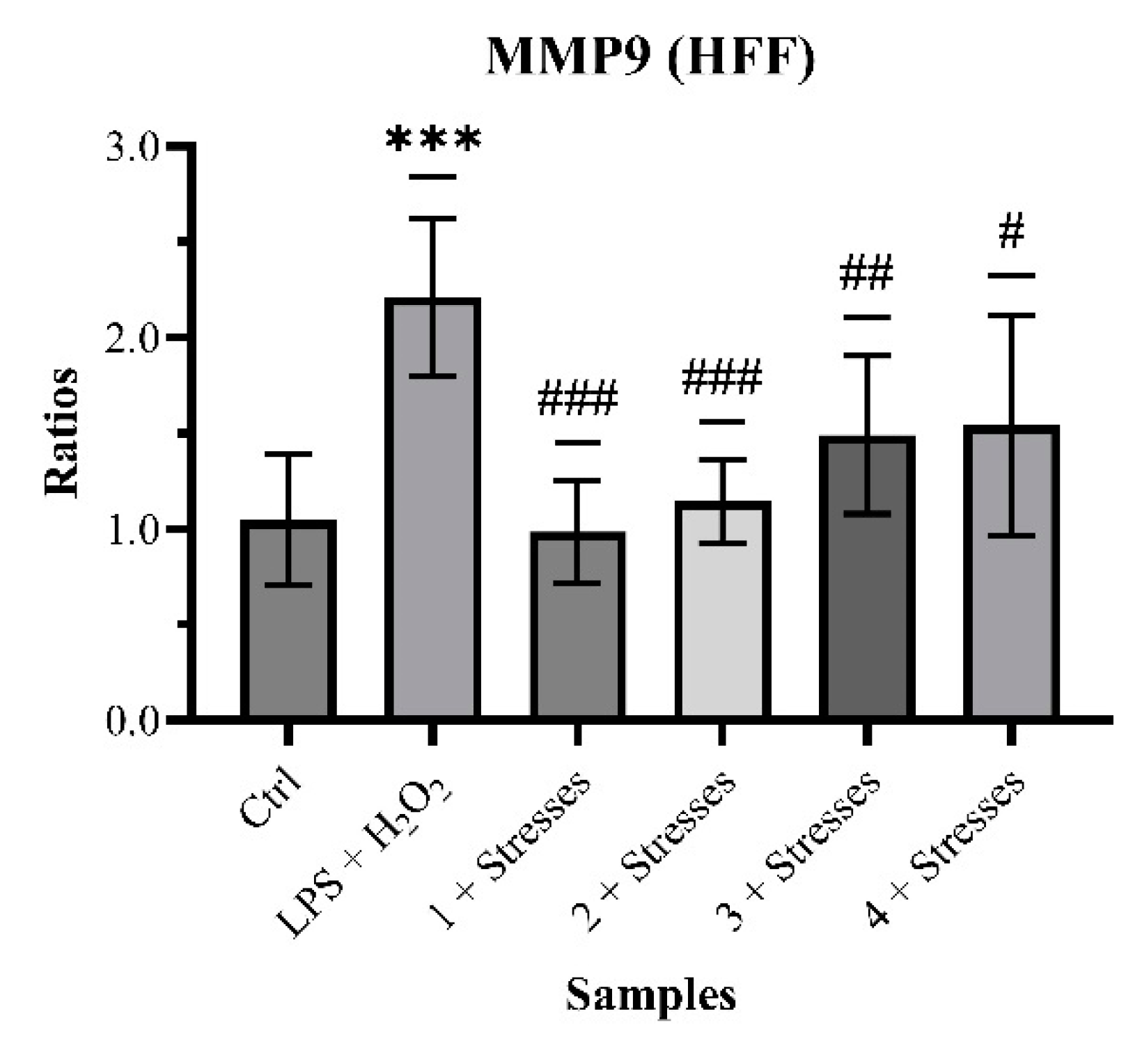
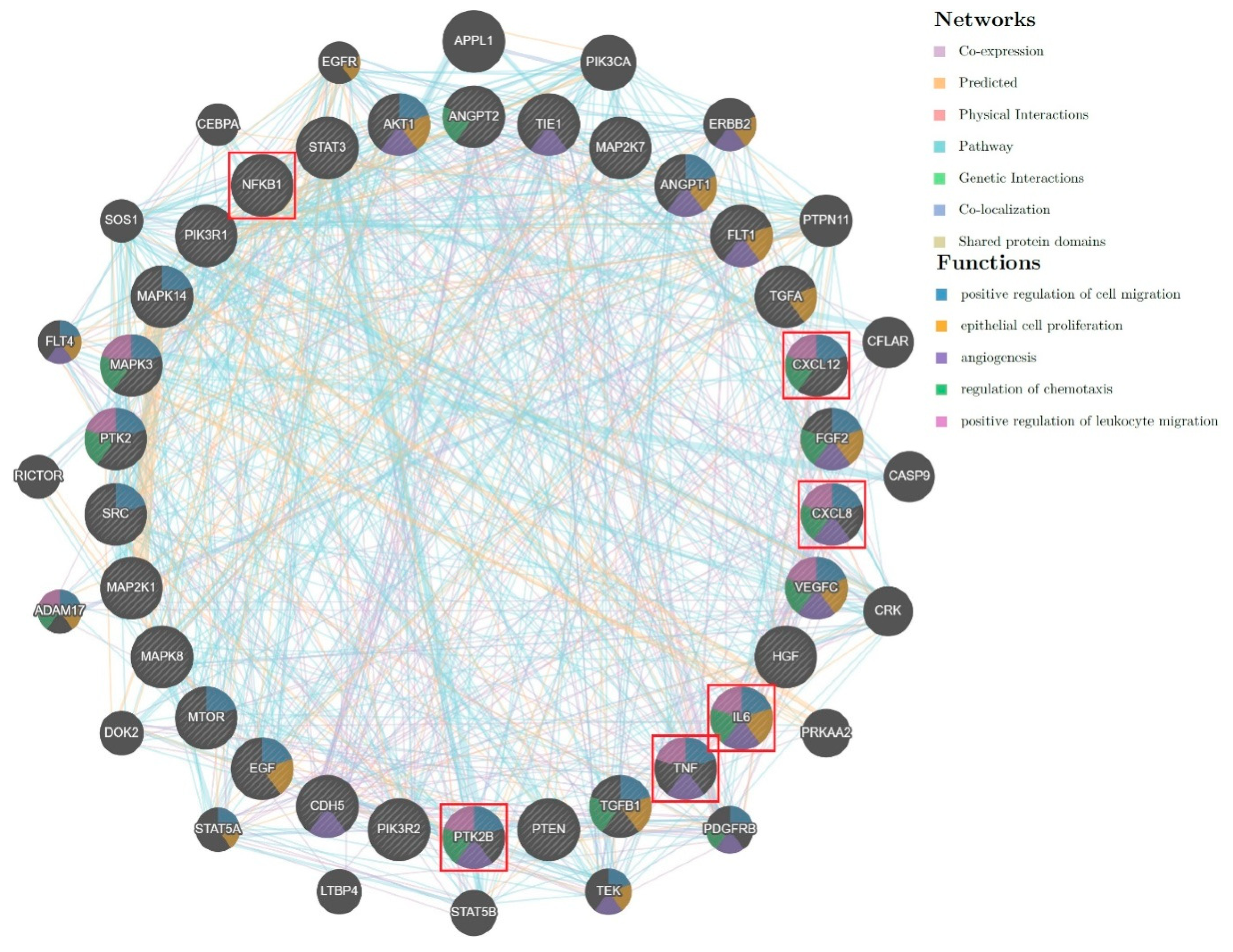
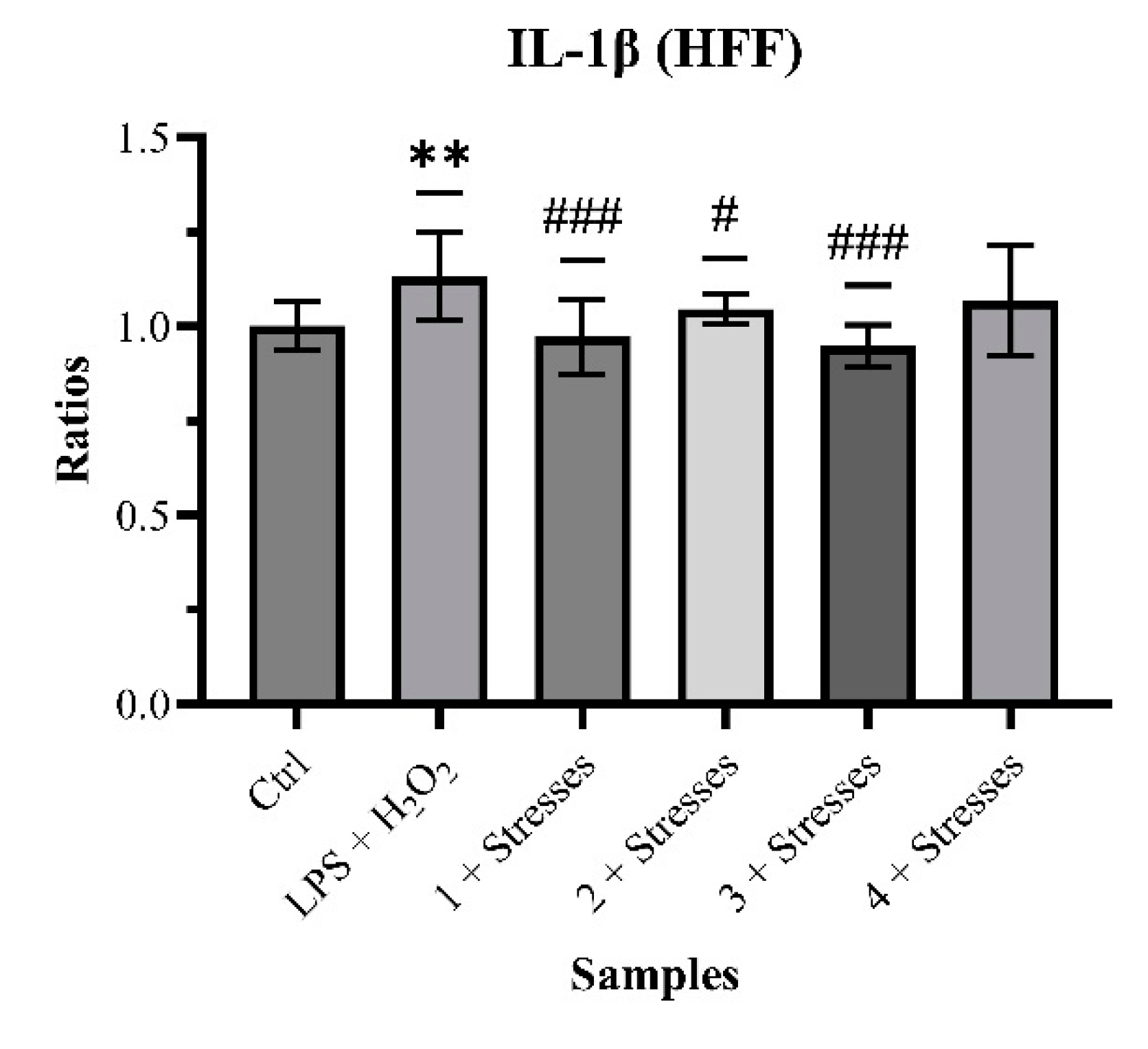
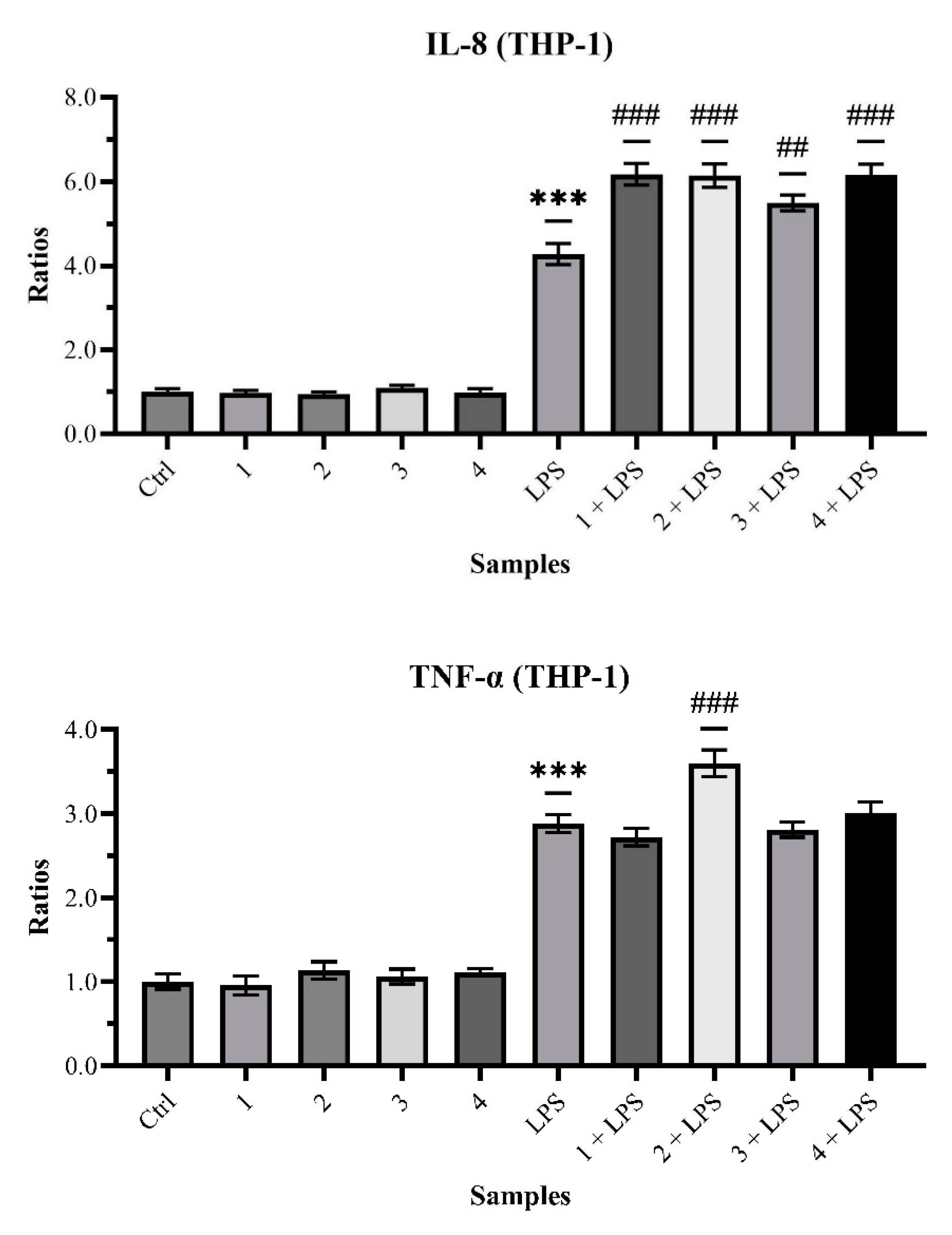
2.2. Biological Analysis and Network Pharmacology
3. Discussion
4. Materials and Methods
4.1. Samples
4.2. Phytochemical Analysis
4.2.1. Total Phenolic Content
4.2.2. Total Flavonoid Content
4.2.3. Triterpenoid Content
4.2.4. Chemical Characterization by HPLC-DAD
4.3. Biological Analysis
4.3.1. Scavenging Activity and Total Antioxidant Capacity
4.3.2. Cell Lines
4.3.3. Cytotoxicity Assay
4.3.4. Cell Migration and Proliferation Assay (Scratch Wound Healing Assay)
4.3.5. Quantifications through ELISAs
4.3.6. Inhibition of ECM-Degrading Enzymes
4.4. Statistical Analysis
4.5. In Silico Target Prediction
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| CAPE | Caffeic Acid Phenethyl Ester |
| DMEM | Dulbecco’s Modified Eagle’s Medium |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| ECM | Extracellular matrix |
| ELISA | Enzyme-Linked Immunosorbent Assay |
| FBS | Fetal Bovine Serum |
| FGF-7 | Keratinocytes growth factor |
| GAE | Gallic Acid Equivalent |
| GF | Growth factor |
| HPLC-DAD | High-Performance Liquid Chromatography with Diode-Array Detection |
| IL | Interleukin |
| LAP | Latency Associated Peptide |
| LPS | Lipopolysaccharides |
| MMP | Matrix metalloprotease |
| NP | Network Pharmacology |
| ORAC | Oxygen Radical Absorbance Capacity |
| PBS | Phosphate Buffered Saline |
| RPMI | Roswell Park Memorial Institute |
| RT | Retention time |
| TAC | Total Antioxidant Capacity |
| TEAC | Trolox Equivalent Antioxidant Capacity |
| TFC | Total Flavonoid Content |
| TGF | Transforming Growth Factor |
| TNF | Tumor Necrosis Factor |
| TPC | Total Phenolic Content |
References
- Tottoli, E.M.; Dorati, R.; Genta, I.; Chiesa, E.; Pisani, S.; Conti, B. Skin Wound Healing Process and New Emerging Technologies for Skin Wound Care and Regeneration. Pharmaceutics 2020, 12, 735. [Google Scholar] [CrossRef]
- Cioce, A.; Cavani, A.; Cattani, C.; Scopelliti, F. Role of the Skin Immune System in Wound Healing. Cells 2024, 13, 624. [Google Scholar] [CrossRef]
- Governa, P.; Carullo, G.; Biagi, M.; Rago, V.; Aiello, F. Evaluation of the In Vitro Wound-Healing Activity of Calabrian Honeys. Antioxidants 2019, 8, 36. [Google Scholar] [CrossRef]
- El-Sherbeni, S.A.; Negm, W.A. The wound healing effect of botanicals and pure natural substances used in in vivo models. Inflammopharmacol, 2023; 31, 755–772. [Google Scholar] [CrossRef]
- Albahri, G.; Badran, A.; Hijazi, A.; Daou, A.; Baydoun, E.; Nasser, M.; Merah, O. The Therapeutic Wound Healing Bioactivities of Various Medicinal Plants. Life 2023, 13, 317. [Google Scholar] [CrossRef]
- Criollo-Mendoza, M.S.; Contreras-Angulo, L.A.; Leyva-López, N.; Gutiérrez-Grijalva, E.P.; Jiménez-Ortega, L.A.; Heredia, J.B. Wound Healing Properties of Natural Products: Mechanisms of Action. Molecules 2023, 28, 598. [Google Scholar] [CrossRef]
- Fernandes, A.; Rodrigues, P.M.; Pintado, M.; Tavaria, F.K. A systematic review of natural products for skin applications: Targeting inflammation, wound healing, and photo-aging. Phytomedicine 2023, 115, 154824. [Google Scholar] [CrossRef]
- Monavarian, M; Kader, S; Moeinzadeh, S; Jabbari, E. Regenerative Scar-Free Skin Wound Healing. Tissue Eng Part B Rev 2019, 25, 294–311. [CrossRef]
- Moses, R.L.; Prescott, T.A.K.; Mas-Claret, E.; Steadman, R.; Moseley, R.; Sloan, A.J. Evidence for Natural Products as Alternative Wound-Healing Therapies. Biomolecules 2023, 13, 444. [Google Scholar] [CrossRef]
- Yang, Juan, Pi, Anjuan, Yan, Lele, Li, Juan, Nan, Sha, Zhang, Jing, Hao, Yuhui, Research Progress on Therapeutic Effect and Mechanism of Propolis on Wound Healing, Evidence-Based Complementary and Alternative Medicine, 2022, 5798941, 15 pages. [CrossRef]
- Daina, A.; Michielin, O.; Zoete, V. SwissTargetPrediction: updated data and new features for efficient prediction of protein targets of small molecules. Nucleic Acids Research 2019, 47, W357–W364. [Google Scholar] [CrossRef]
- Kasote, D.; Bankova, V.; Viljoen, A.M. Propolis: chemical diversity and challenges in quality control. Phytochem Rev, 2022; 21, 1887–1911. [Google Scholar] [CrossRef]
- Anjum, S.I.; Ullah, A.; Khan, K.A.; Attaullah, M.; Khan, H.; Ali, H.; Bashir, M.A.; Tahir, M.; Ansari, M.J.; Ghramh, H.A.; Adgaba, N.; Dash, C.K. Composition and functional properties of propolis (bee glue): A review. Saudi Journal of Biological Sciences 2019, 26, 1695–1703. [Google Scholar] [CrossRef]
- Hossain, R.; Quispe, C.; Khan, R.A.; et al. Propolis: An update on its chemistry and pharmacological applications. Chin Med 2022, 17, 100. [Google Scholar] [CrossRef]
- Dezmirean, D.S.; Paşca, C.; Moise, A.R.; Bobiş, O. Plant Sources Responsible for the Chemical Composition and Main Bioactive Properties of Poplar-Type Propolis. Plants 2021, 10, 22. [Google Scholar] [CrossRef]
- Finetti, F.; Biagi, M.; Ercoli, J.; Macrì, G.; Miraldi, E.; Trabalzini, L. Phaseolus vulgaris L. var. Venanzio Grown in Tuscany: Chemical Composition and In Vitro Investigation of Potential Effects on Colorectal Cancer. Antioxidants 2020, 9, 1181. [Google Scholar] [CrossRef]
- Biagi, M.; Manca, D.; Barlozzini, B.; Miraldi, E.; Giachetti, D. Optimization of extraction: Of drugs containing polyphenols using an innovative technique. Agro Food Industry Hi Tech. 2014, 25, 60–65. [Google Scholar]
- Sberna, G.; Biagi, M.; Marafini, G.; Nardacci, R.; Biava, M.; Colavita, F.; Piselli, P.; Miraldi, E.; D'Offizi, G.; Capobianchi, M.R.; Amendola, A. In vitro Evaluation of Antiviral Efficacy of a Standardized Hydroalcoholic Extract of Poplar Type Propolis Against SARS-CoV-2. Front Microbiol. 2022, 13, 799546. [Google Scholar] [CrossRef]
- Lamponi, S.; Baratto, M.C.; Miraldi, E.; Baini, G.; Biagi, M. Chemical Profile, Antioxidant, Anti-Proliferative, Anticoagulant and Mutagenic Effects of a Hydroalcoholic Extract of Tuscan Rosmarinus officinalis. Plants 2021, 10, 97. [Google Scholar] [CrossRef]
- Bonetti, A.; Faraloni, C.; Venturini, S.; Baini, G.; Miraldi, E.; Biagi, M. Characterization of phenolic profile and antioxidant activity of the leaves of the forgotten medicinal plant Balsamita major grown in Tuscany, Italy, during the growth cycle. Plant Biosystems - An International Journal Dealing with All Aspects of Plant Biology, 2020, 155, 908–913. [Google Scholar] [CrossRef]
- Cappellucci, G.; Baini, G.; Miraldi, E.; Pauletto, L.; De Togni, H.; Raso, F.; Biagi, M. Investigation on the Efficacy of Two Food Supplements Containing a Fixed Combination of Selected Probiotics and β-Glucans or Elderberry Extract for the Immune System: Modulation on Cytokines Expression in Human THP-1 and PBMC. Foods 2024, 13, 458. [Google Scholar] [CrossRef]
- Cappellucci, G.; Paganelli, A.; Ceccarelli, P.L.; Miraldi, E.; Biagi, M. Insights on the In Vitro Wound Healing Effects of Sedum telephium L. Leaf Juice. Cosmetics 2024, 11, 131. [Google Scholar] [CrossRef]
- https://www.theimagingsource.com/en-us/support/download/icmeasure-2.0.0.286/.
- Governa, P.; Cusi, M.G.; Borgonetti, V.; Sforcin, J.M.; Terrosi, C.; Baini, G.; Miraldi, E.; Biagi, M. Beyond the Biological Effect of a Chemically Characterized Poplar Propolis: Antibacterial and Antiviral Activity and Comparison with Flurbiprofen in Cytokines Release by LPS-Stimulated Human Mononuclear Cells. Biomedicines 2019, 7, 73. [Google Scholar] [CrossRef]
- Pressi, G.; Rigillo, G.; Governa, P.; Borgonetti, V.; Baini, G.; Rizzi, R.; Guarnerio, C.; Bertaiola, O.; Frigo, M.; Merlin, M.; et al. A Novel Perilla frutescens (L.) Britton Cell-Derived Phytocomplex Regulates Keratinocytes Inflammatory Cascade and Barrier Function and Preserves Vaginal Mucosal Integrity In Vivo. Pharmaceutics 2023, 15, 240. [Google Scholar] [CrossRef]
- Rigillo, G.; Cappellucci, G.; Baini, G.; Vaccaro, F.; Miraldi, E.; Pani, L.; Tascedda, F.; Bruni, R.; Biagi, M. Comprehensive Analysis of Berberis aristata DC. Bark Extracts: In Vitro and In Silico Evaluation of Bioaccessibility and Safety. Nutrients 2024, 16, 2953. [Google Scholar] [CrossRef]
- https://www.generecommender.com/.
- http://www.genemania.org/.
- Brambilla, D.; Giacomini, D.M.; Muscarnera, L.; Mazzoleni, A. DeepProphet2 – a deep learning gene recommendation engine. arXiv (Cornell University). [CrossRef]
- Hu, LT.; Deng, WJ.; Chu, ZS.; et al. Comprehensive analysis of CXCR family members in lung adenocarcinoma with prognostic values. BMC Pulm Med 2022, 22, 259. [Google Scholar] [CrossRef]


| Sample | GAE (% w/w ± SD) |
|---|---|
| 1 | 21.13 ± 1.17 |
| 2 | 14.25 ± 0.57 |
| 3 | 18.50 ± 0.90 |
| 4 | 0.50 ± 0.06 |
| Sample | Galangin Equivalents (% w/w ± SD) |
|---|---|
| 1 | 19.68 ± 0.36 |
| 2 | 9.55 ± 0.23 |
| 3 | 13.09 ± 0.46 |
| 4 | 0.25 ± 0.20 |
| Sample | β-sitosterol Equivalents (% w/w ± SD) |
|---|---|
| 1 | 61.19 ± 0.60 |
| 2 | 43.15 ± 0.31 |
| 3 | 65.00 ± 0.64 |
| 4 | 2.74 ± 0.14 |
| Sample | Pinobanksin (% w/w± SD) |
Pinocembrin (% w/w± SD) |
CAPE (% w/w± SD) |
Galangin + chrysin (% w/w± SD) |
|---|---|---|---|---|
| 1 | 1.46 ± 0.27 | 8.14 ± 1.40 | 1.93 ± 0.34 | 13.22 ± 2.25 |
| 2 | 0.66 ± 0.08 | 2.88 ± 0.42 | < 0.01 | 8.60 ± 2.76 |
| 3 | 1.29 ± 0.05 | 3.68 ± 0.81 | < 0.01 | 12.68 ± 2.23 |
| 4 | 0.31 | 0.03 | < 0.01 | 0.02 |
| Sample | IC50 (μg/mL ± SD) |
|---|---|
| 1 | 26.09 ± 1.30 |
| 2 | 54.38 ± 2.72 |
| 3 | 45.50 ± 2.28 |
| 4 | > 1 000 |
| Sample | Trolox Equivalents (mM/g ± SD) |
|---|---|
| 1 | 8.89 ± 1.50 |
| 2 | 4.93 ± 2.21 |
| 3 | 6.96 ± 2.05 |
| 4 | 3.01 ± 1.79 |
| MMPs | N° of matches with query molecules |
|---|---|
| MMP2 | 4 |
| MMP9 | 4 |
| MMP12 | 4 |
| MMP13 | 3 |
| Sample | IC50 (μg/mL ± SD) |
|---|---|
| 1 | 41.61 ± 2.08 |
| 2 | 113.96 ± 5.70 |
| 3 | 51.57 ± 2.58 |
| 4 | > 200 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




