Submitted:
02 October 2024
Posted:
02 October 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Characteristics of the Studied Population
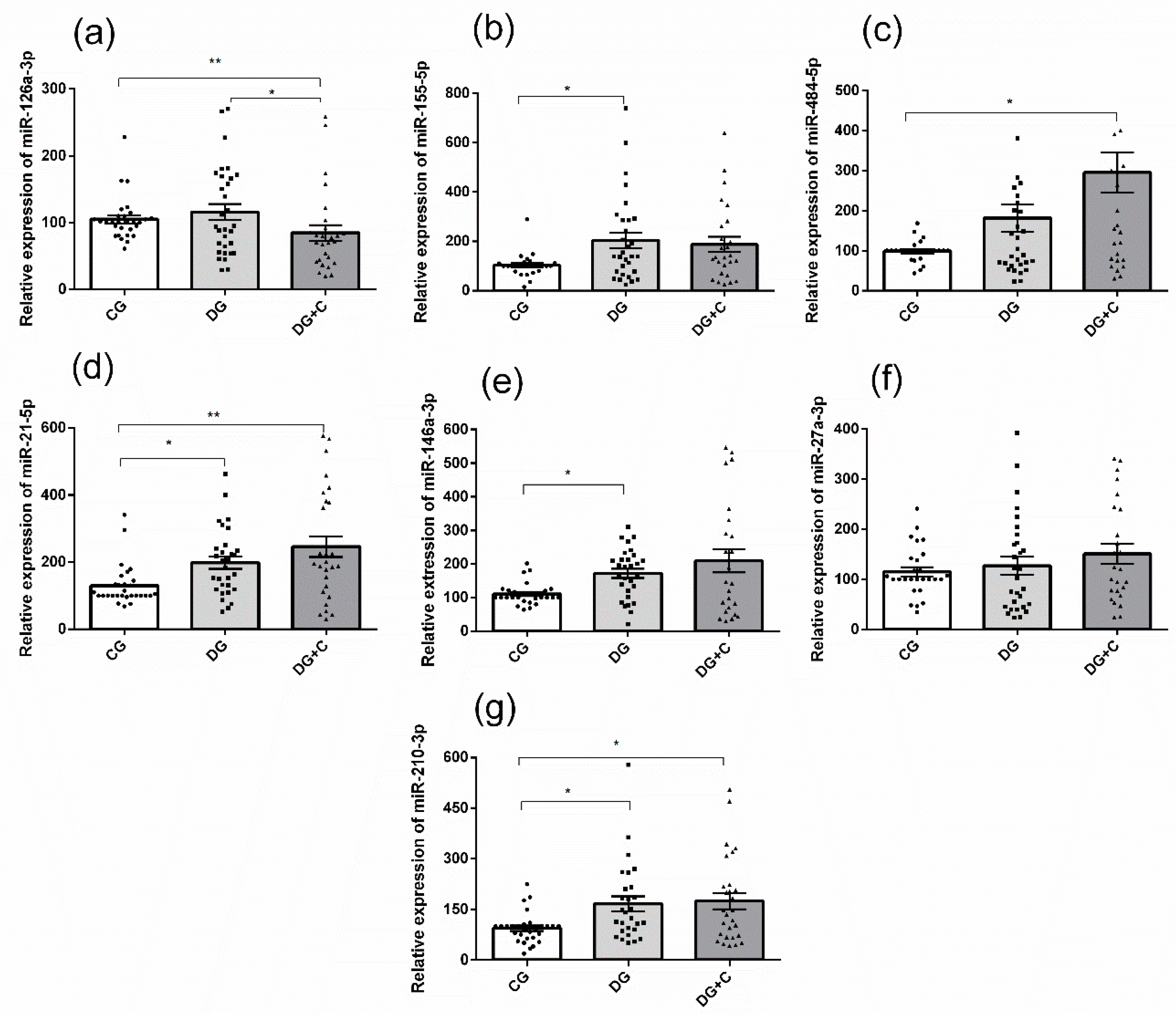
2.2. Differential Patterns of miRNA Expression in Studied Population
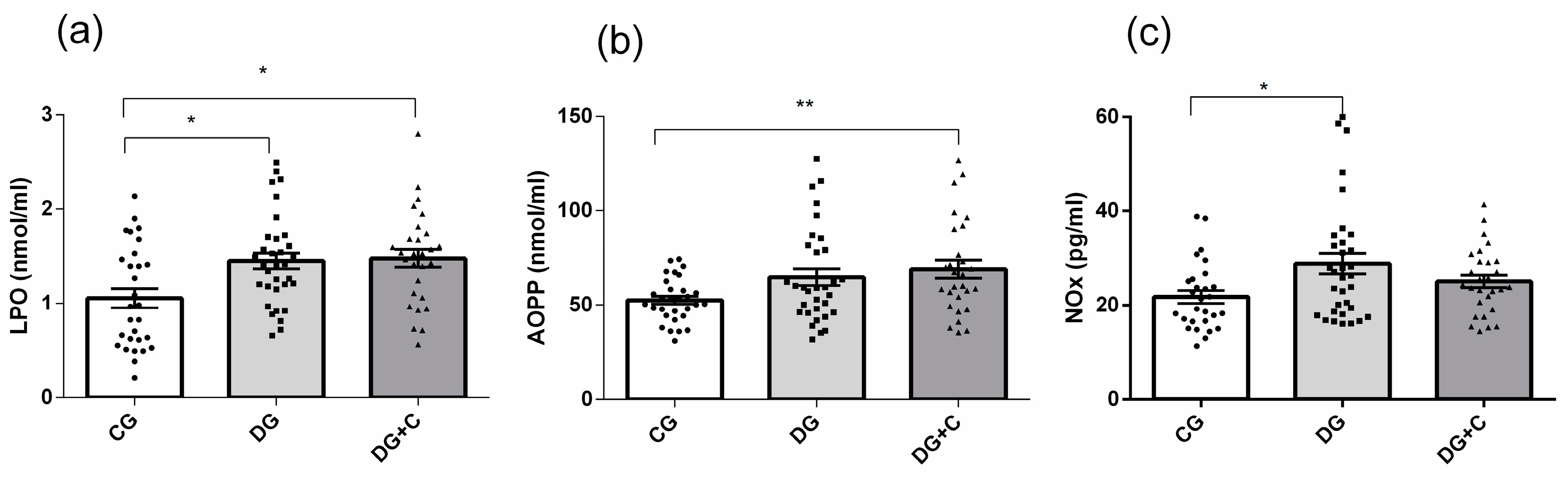
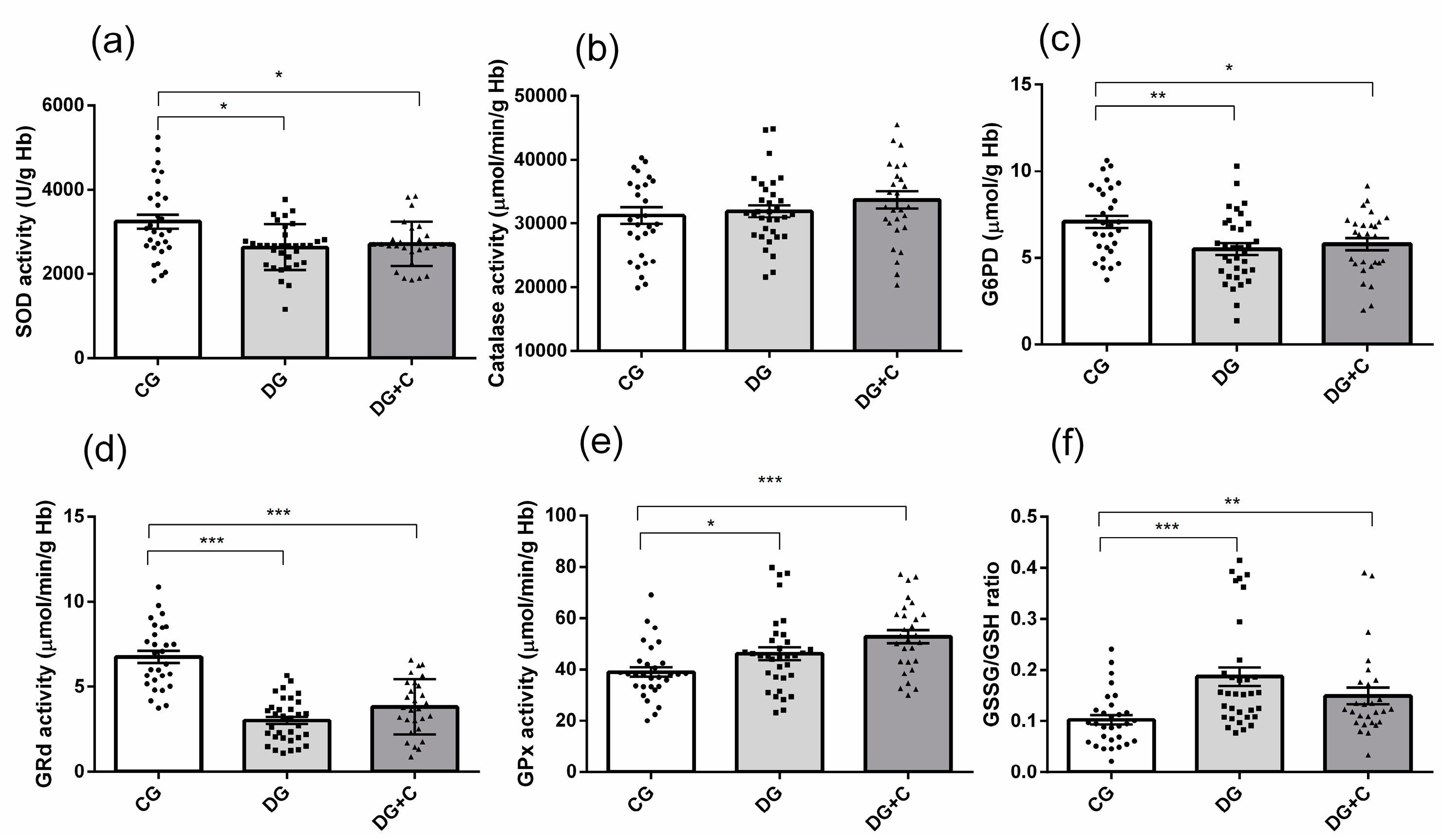
2.3. Oxidative Status Markers in Studied Samples
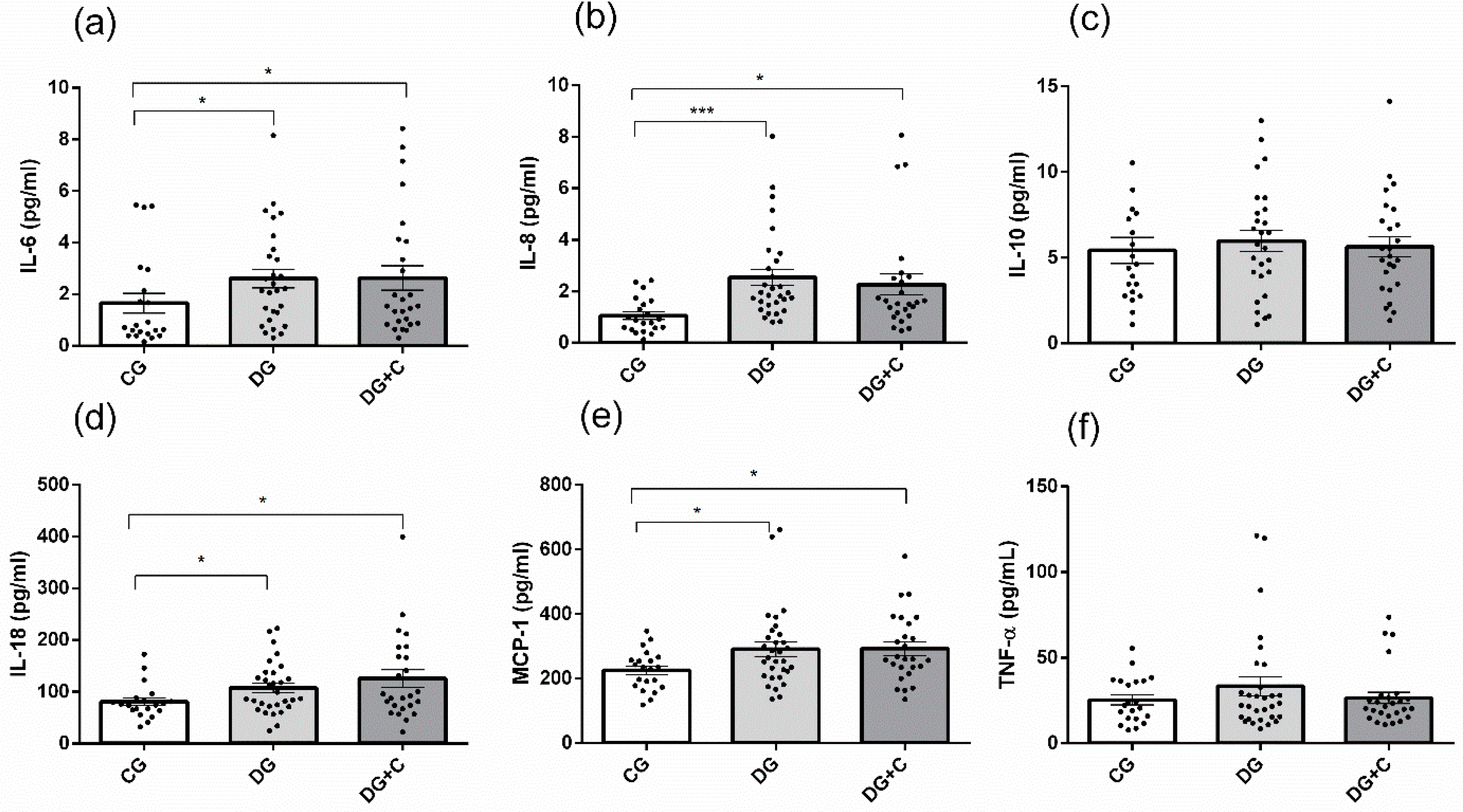
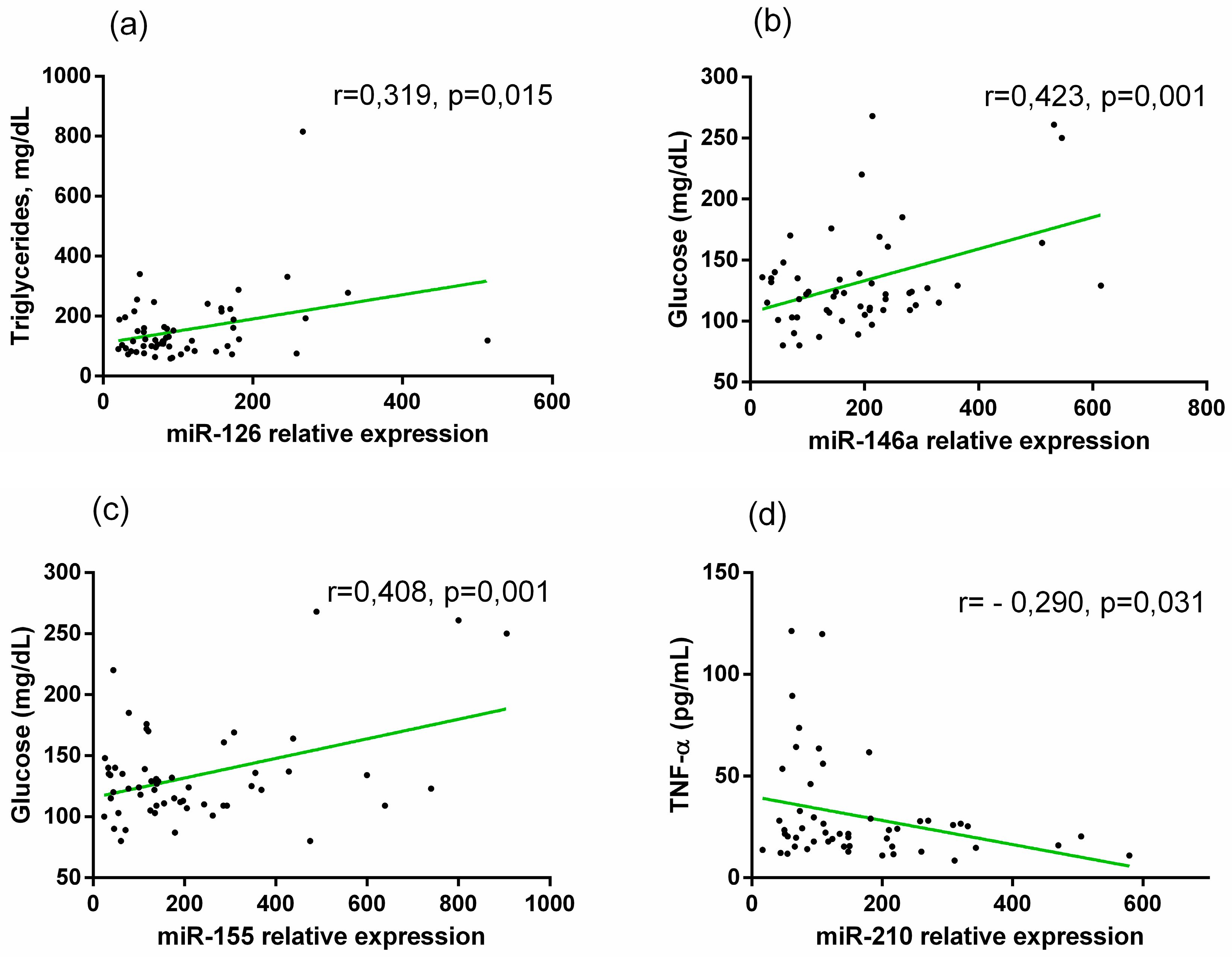
2.4. Inflammatory Status and Correlations between Studied Markers and Clinical Profile
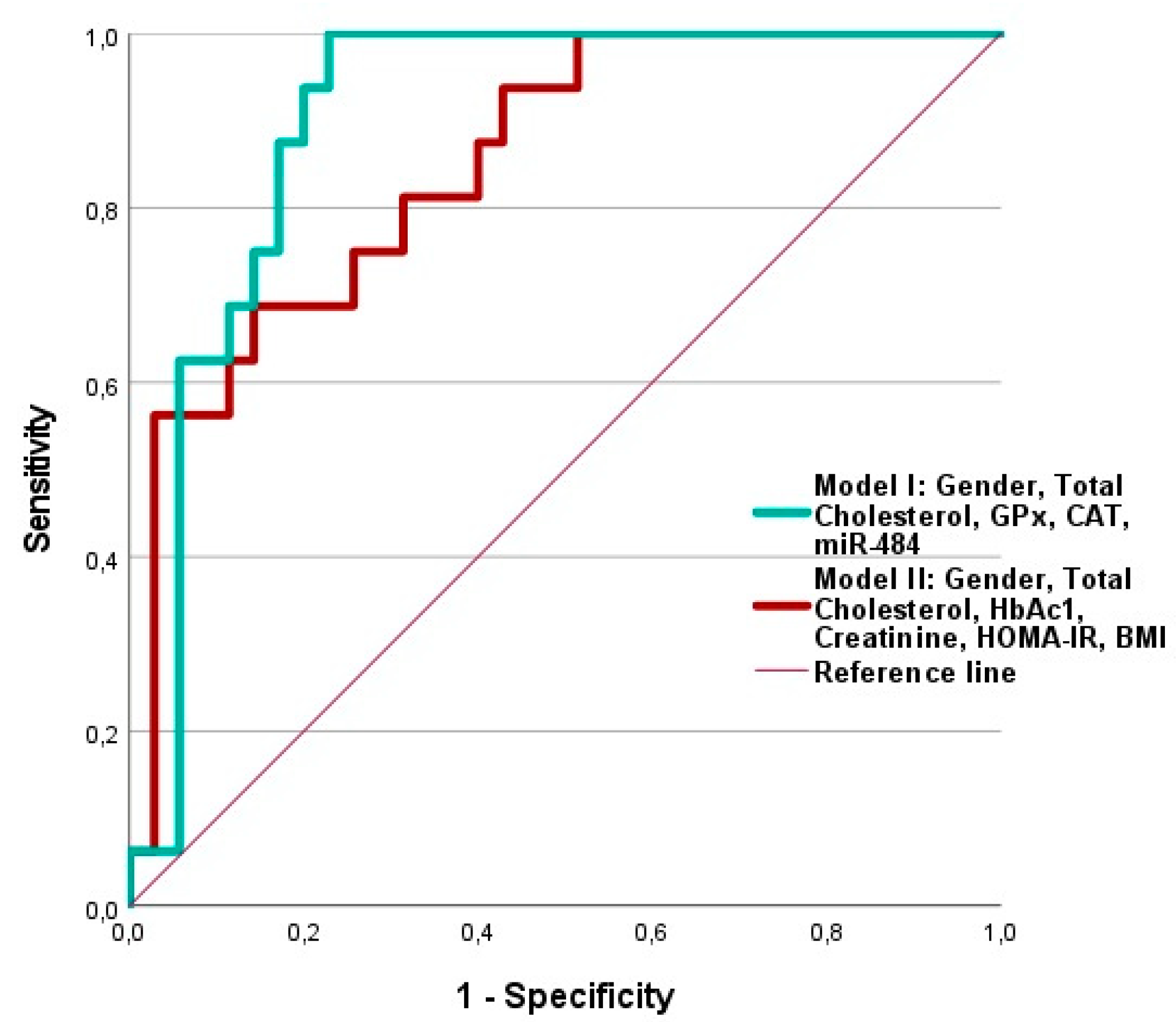
2.5. Evaluation of the Diagnostic Accuracy of Biomarkers Related to Diabetes and Macrovascular Complications in T2DM.
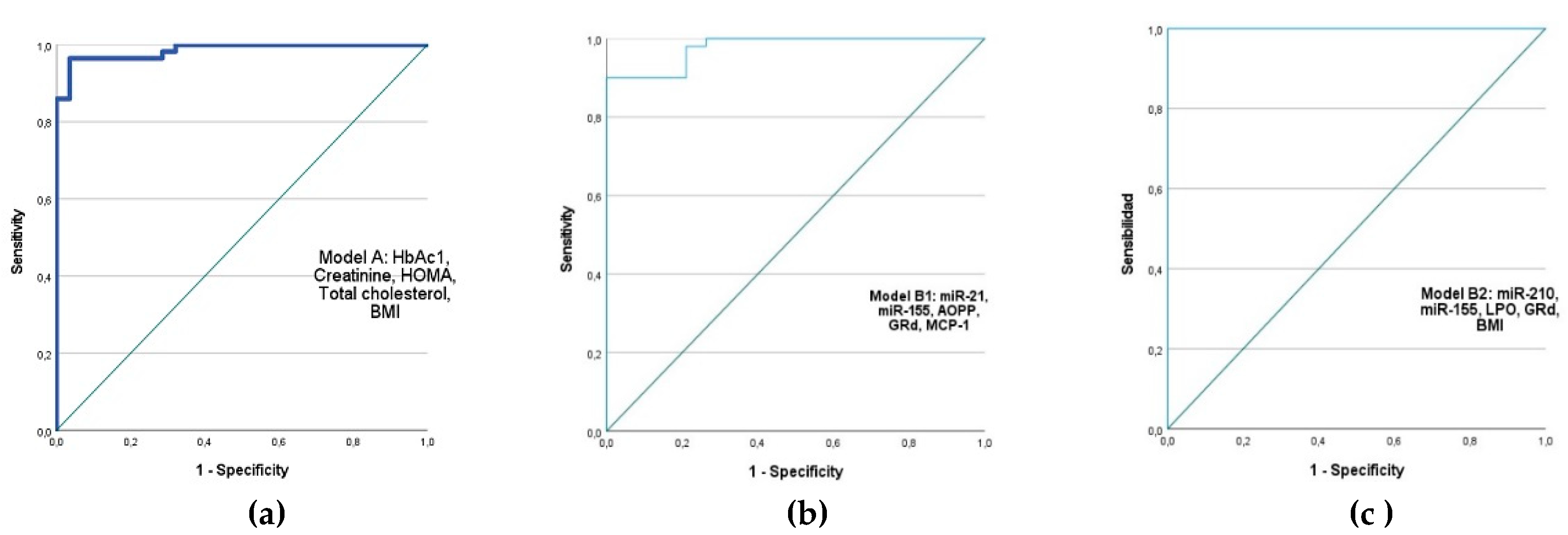
| Model | AUC (95%) | Exp (B) | φ2 | p | R2 (Cox y Snell) | R2 (Nagelkerke) |
|---|---|---|---|---|---|---|
| Model 1(Glucose, HbAc1, Creatinine, HOMA-IR, total cholesterol, TG, gender) | 0.845 | 0.727 | 25,724 | < 0.001 | 0.363 | 0.488 |
| Model 2(HbAc1, Creatinine, total cholesterol, LPO, GPx, SOD, miR-126, miR-484) | 0.913 | 0.926 | 34,093 | < 0.001 | 0.481 | 0.642 |
| Model 3(HbAc1, Creatinine, total cholesterol, IL-6, LPO, miR-126, miR-484) | 0.938 | 0.958 | 33,863 | < 0.001 | 0.513 | 0.685 |
| AUC, (95%) | Exp (B) = OR | Chi 2 | p | |
|---|---|---|---|---|
| HbAc1 | 0.488 | 0,929 (0,530-1,631) | 0,066 | 0,797 |
| Total cholesterol | 0.786 | 0,968 (0,949-0,987) | 15,101 | <0,001 |
| Creatinine | 0.461 | 0,658 (0,087-4,943) | 0,174 | 0,677 |
| HOMA-IR | 0.593 | 1,021 (0,893-1,167) | 0,092 | 0,762 |
| LPO | 0.491 | 0,999 (0,339-2,939) | 0,000 | 0,998 |
| AOPP | 0.524 | 1,000 (0,981-1,020) | 0,000 | 0,990 |
| CAT | 0.681 | 1,000 (1,000-1,000) | 4,554 | 0,033 |
| GPx | 0.711 | 1,051 (1,009-1,094) | 6,371 | 0,012 |
| GRd | 0.623 | 1,375 (0,949-1,992) | 2,971 | 0,085 |
| GSSG/GSH | 0.555 | 0,138 (0,000-46,142) | 0,465 | 0,495 |
| G6DH | 0.608 | 1,192 (0,917-1,550) | 1,787 | 0,181 |
| SOD | 0.603 | 1,001 (1,000-1,002) | 1,560 | 0,212 |
| NOX | 0.556 | 0,970 (0,921-1,021) | 1,688 | 0,194 |
| IL-6 | 0.529 | 1,113 (0,860-1,441) | 0,661 | 0,416 |
| IL-8 | 0633 | 0,750 (0,502-1,121) | 2,713 | 0,100 |
| IL-10 | 0.539 | 0,973 (0,808-1,171) | 0,084 | 0,771 |
| IL-18 | 0.558 | 1,006 (0,997-1,015) | 1,995 | 0,158 |
| MCP-1 | 0.485 | 1,000 (0,995-1,005) | 0,002 | 0,968 |
| TNF-a | 0.595 | 0,979 (0,949-1,010) | 2,397 | 0,122 |
| miR-21 | 0.600 | 1,003 (0,999-1,007) | 2,666 | 0,103 |
| miR-126 | 0.577 | 0,994 (0,985-1,003) | 1,807 | 0,179 |
| miR-146 | 0,516 | 1,002 (0,998-1,006) | 1,094 | 0,296 |
| miR-155 | 0.508 | 1,000 (0,997-1,003) | 0,001 | 0,980 |
| miR-484 | 0.674 | 1,004 (1,001-1,008) | 7,109 | 0,008 |
| miR-27a | 0.566 | 1,002 (0,997-1,008) | 0,533 | 0,465 |
| miR-210 | 0.597 | 1,003 (0,999-1,008) | 2,053 | 0,152 |
3. Discussion
4. Materials and Methods
4.2. Biochemical Analysis
4.3. MicroRNA Expression Analysis
4.4. Determination of Inflammatory Parameters
4.5. Measurement of LPO and AOPP Levels
4.6. Measurement of GSH and GSSG Levels:
4.7. Measurement of GPx, GRd, SOD, CAT, and G6PD Activities:
4.8. Nitrite Plus Nitrate Determination
4.9. Statistics
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Afsharmanesh, M.R.; Mohammadi, Z.; Mansourian, A.R.; Jafari, S.M. A Review of micro RNAs changes in T2DM in animals and humans. J. Diabetes 2023, 15. [Google Scholar] [CrossRef]
- Medina-Leyte, D.J.; Zepeda-García, O.; Domínguez-Pérez, M.; González-Garrido, A.; Villarreal-Molina, T.; Jacobo-Albavera, L. Endothelial dysfunction, inflammation and coronary artery disease: Potential biomarkers and promising therapeutical approaches. Int. J. Mol. Sci. 2021, 22. [Google Scholar] [CrossRef]
- Yuan, J.; Chen, M.; Xu, Q.; Liang, J.; Chen, R.; Xiao, Y.; Fang, M.; Chen, L. Effect of the Diabetic Environment on the Expression of MiRNAs in Endothelial Cells: Mir-149-5p Restoration Ameliorates the High Glucose-Induced Expression of TNF-α and ER Stress Markers. Cell. Physiol. Biochem. 2017, 43. [Google Scholar] [CrossRef]
- He, Y.; Ding, Y.; Liang, B.; Lin, J.; Kim, T.K.; Yu, H.; Hang, H.; Wang, K. A systematic study of dysregulated MicroRNA in type 2 diabetes mellitus. Int. J. Mol. Sci. 2017, 18. [Google Scholar] [CrossRef]
- Castaño, C.; Kalko, S.; Novials, A.; Párrizas, M. Obesity-associated exosomal miRNAs modulate glucose and lipid metabolism in mice. Proc. Natl. Acad. Sci. U. S. A. 2018, 115, 12158–12163. [Google Scholar] [CrossRef]
- Li, X.; Xu, L.; Hou, X.; Geng, J.; Tian, J.; Liu, X.; Bai, X. Advanced Oxidation Protein Products Aggravate Tubulointerstitial Fibrosis Through Protein Kinase C-Dependent Mitochondrial Injury in Early Diabetic Nephropathy. Antioxidants Redox Signal. 2019, 30. [Google Scholar] [CrossRef]
- Liang, M.; Wang, J.; Xie, C.; Yang, Y.; Tian, J.W.; Xue, Y.M.; Hou, F.F. Increased plasma advanced oxidation protein products is an early marker of endothelial dysfunction in type 2 diabetes patients without albuminuria. J. Diabetes 2014, 6, 417–426. [Google Scholar] [CrossRef]
- Escames, G.; Khaldy, H.; León, J.; González, L.; Acuña-Castroviejo, D. Changes in iNOS activity, oxidative stress and melatonin levels in hypertensive patients treated with lacidipine. J. Hypertens. 2004, 22, 629–635. [Google Scholar] [CrossRef]
- López-Armas, G.C.; Yessenbekova, A.; González-Castañeda, R.E.; Arellano-Arteaga, K.J.; Guerra-Librero, A.; Ablaikhanova, N.; Florido, J.; Escames, G.; Acuña-Castroviejo, D.; Rusanova, I. Role of c-miR-21, c-miR-126, Redox Status, and Inflammatory Conditions as Potential Predictors of Vascular Damage in T2DM Patients. Antioxidants 2022, 11. [Google Scholar] [CrossRef]
- Jiménez-Osorio, A.S.; Picazo, A.; González-Reyes, S.; Barrera-Oviedo, D.; Rodríguez-Arellano, M.E.; Pedraza-Chaverri, J. Nrf2 and Redox Status in Prediabetic and Diabetic Patients. OPEN ACCESS Int. J. Mol. Sci 2014, 15, 15. [Google Scholar] [CrossRef]
- Andrade-Sierra, J.; Pazarín-Villaseñor, L.; Yanowsky-Escatell, F.G.; Díaz-de la Cruz, E.N.; García-Sánchez, A.; Cardona-Muñoz, E.G.; Munguía-Galaviz, F.J.; de Alba-Razo, A.; Miranda-Díaz, A.G. The Influence of the Severity of Early Chronic Kidney Disease on Oxidative Stress in Patients with and without Type 2 Diabetes Mellitus. Int. J. Mol. Sci. 2022, 23, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Paneque A., Fortus H., Zheng J., Werley G., J.E. The hexosamine biosynthesis pathway: regulation and function. 2023, 1–43.
- Seo, J.A.; Jung, S.H.; Jeon, H.Y.; Lee, Y.J.; Lee, J.Y.; Han, E.T.; Park, W.S.; Hong, S.H.; Kim, Y.M.; Ha, K.S. Activity-expression profiling of glucose-6-phosphate dehydrogenase in tissues of normal and diabetic mice. Biochem. Biophys. Res. Commun. 2020, 524, 750–755. [Google Scholar] [CrossRef] [PubMed]
- Breeyear, J.H.; Hellwege, J.N.; Schroeder, P.H.; House, J.S.; Poisner, H.M.; Mitchell, S.L.; Charest, B.; Khakharia, A.; Basnet, T.B.; Halladay, C.W.; et al. Adaptive selection at G6PD and disparities in diabetes complications. Nat. Med. 2024. [Google Scholar] [CrossRef] [PubMed]
- Deshmane, S.L.; Kremlev, S.; Amini, S.; Sawaya, B.E. Monocyte chemoattractant protein-1 (MCP-1): An overview. J. Interf. Cytokine Res. 2009, 29, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.F.; Shabayek, M.I.; Abdel Ghany, M.E.; El-Hefnawy, M.H.; El-Mesallamy, H.O. Role of CTRP3, CTRP9 and MCP-1 for the evaluation of T2DM associated coronary artery disease in Egyptian postmenopausal females. PLoS One 2018, 13, 1–13. [Google Scholar] [CrossRef]
- Daniele, G.; Guardado Mendoza, R.; Winnier, D.; Fiorentino, T. V.; Pengou, Z.; Cornell, J.; Andreozzi, F.; Jenkinson, C.; Cersosimo, E.; Federici, M.; et al. The inflammatory status score including IL-6, TNF-α, osteopontin, fractalkine, MCP-1 and adiponectin underlies whole-body insulin resistance and hyperglycemia in type 2 diabetes mellitus. Acta Diabetol. 2014, 51, 123–131. [Google Scholar] [CrossRef]
- Olivieri, F.; Spazzafumo, L.; Bonafè, M.; Recchioni, R.; Prattichizzo, F.; Marcheselli, F.; Micolucci, L.; Mensà, E.; Giuliani, A.; Santini, G.; et al. MiR-21-5p and miR-126a-3p levels in plasma and circulating angiogenic cells: relationship with type 2 diabetes complications. Oncotarget 2015, 6. [Google Scholar] [CrossRef]
- Dehghani, M.; Aghaei Zarch, S.M.; Vahidi Mehrjardi, M.Y.; Nazari, M.; Babakhanzadeh, E.; Ghadimi, H.; Zeinali, F.; Talebi, M. Evaluation of miR-181b and miR-126-5p expression levels in T2DM patients compared to healthy individuals: Relationship with NF-κB gene expression. Endocrinol. Diabetes y Nutr. 2020, 67, 454–460. [Google Scholar] [CrossRef]
- Zhang N, Z.J.F.L. Mechanism of miR-126 in hypoxia-reoxygenation-induced cardiomyocyte pyroptosis by regulating HMGB1 and NLRP3 inflammasome. Immunopharmacol. Immunotoxicol. 2022, 44, 500–509. [Google Scholar] [CrossRef]
- Ginckels, P.; Holvoet, P. Oxidative Stress and Inflammation in Cardiovascular Diseases and Cancer: Role of Non-coding RNAs. Yale J. Biol. Med. 2022, 95, 129–152. [Google Scholar]
- Yang, W.Z.; Yang, J.; Xue, L.P.; Xiao, L.B.; Li, Y. MiR-126 overexpression inhibits high glucose-induced migration and tube formation of rhesus macaque choroid-retinal endothelial cells by obstructing VEGFA and PIK3R2. J. Diabetes Complications 2017, 31. [Google Scholar] [CrossRef] [PubMed]
- López-Armas, G. C. , Yessenbekova, A., González-Castañeda, R. E., Arellano-Arteaga, K. J., Guerra-Librero, A., Ablaikhanova, N., Florido, J., Escames, G., Acuña-Castroviejo, D., & Rusanova, I. (2022 Role of c-miR-21, c-miR-126, Redox Status, and Inflammatory Conditions as Potential Predictors of Vascular Damage in T2DM Patients. Antioxidants 2022, 11. [Google Scholar]
- Chistiakov, D.A.; Orekhov, A.N.; Bobryshev, Y. V. The role of miR-126 in embryonic angiogenesis, adult vascular homeostasis, and vascular repair and its alterations in atherosclerotic disease. J. Mol. Cell. Cardiol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Catanzaro, G.; Conte, F.; Trocchianesi, S.; Splendiani, E.; Bimonte, V.M.; Mocini, E.; Filardi, T.; Po, A.; Besharat, Z.M.; Gentile, M.C.; et al. Network analysis identifies circulating miR-155 as predictive biomarker of type 2 diabetes mellitus development in obese patients: a pilot study. Sci. Rep. 2023, 13, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, A.; Vigorito, E.; Clare, S.; Warren, M. V.; Couttet, P.; Soond, D.R.; Van Dongen, S.; Grocock, R.J.; Das, P.P.; Miska, E.A.; et al. Requirement of bic/microRNA-155 for normal immune function. Science (80-. ). 2007, 316, 608–611. [Google Scholar] [CrossRef]
- Yin, R.; Zhu, X.; Wang, J.; Yang, S.; Ma, A.; Xiao, Q.; Song, J.; Pan, X. MicroRNA-155 promotes the ox-LDL-induced activation of NLRP3 inflammasomes via the ERK1/2 pathway in THP-1 macrophages and aggravates atherosclerosis in apoe−/− mice. Ann. Palliat. Med. 2019, 8, 676–689. [Google Scholar] [CrossRef]
- Jankauskas, S.S.; Gambardella, J.; Sardu, C.; Lombardi, A.; Santulli, G. Functional role of miR-155 in the pathogenesis of diabetes mellitus and its complications. Non-coding RNA 2021, 7. [Google Scholar] [CrossRef]
- Polina, E.R.; Oliveira, F.M.; Sbruzzi, R.C.; Crispim, D.; Canani, L.H.; Santos, K.G. Gene polymorphism and plasma levels of miR-155 in diabetic retinopathy. Endocr. Connect. 2019, 8, 1591–1599. [Google Scholar] [CrossRef]
- Papadopoulos, K.I.; Papadopoulou, A.; Aw, T.-C. MicroRNA-155 mediates endogenous angiotensin II type 1 receptor regulation: implications for innovative type 2 diabetes mellitus management. World J. Diabetes 2023, 14, 1334–1340. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, J.; Qin, Z.; Liu, N.; Zhang, Z.; Lu, Y.; Xu, Y.; Zhang, J.; Tang, J. Diagnostic and Predictive Values of Circulating Extracellular Vesicle-Carried microRNAs in Ischemic Heart Disease Patients With Type 2 Diabetes Mellitus. Front. Cardiovasc. Med. 2022, 9, 1–10. [Google Scholar] [CrossRef]
- Wang, J.; Wang, G.; Liang, Y.; Zhou, X. Expression profiling and clinical significance of plasma micrornas in diabetic nephropathy. J. Diabetes Res. 2019, 2019. [Google Scholar] [CrossRef] [PubMed]
- Bai, X.; Luo, Q.; Tan, K.; Guo, L. Diagnostic value of VDBP and miR-155-5p in diabetic nephropathy and the correlation with urinary microalbumin. Exp. Ther. Med. 2020, 20, 1–1. [Google Scholar] [CrossRef] [PubMed]
- García-Díaz, D.F.; Pizarro, C.; Camacho-Guillén, P.; Codner, E.; Soto, N.; Pérez-Bravo, F. Expression of miR-155, miR-146a, and miR-326 in T1D patients from chile: Relationship with autoimmunity and inflammatory markers. Arch. Endocrinol. Metab. 2018, 62, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.H.; Ahn, J.; Choi, Y.M.; Son, H.J.; Choi, W.H.; Cho, H.J.; Yu, J.H.; Seo, J.A.; Jang, Y.J.; Jung, C.H.; et al. Differential circulating and visceral fat microRNA expression of non-obese and obese subjects. Clin. Nutr. 2020, 39, 910–916. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Wu, B.; Zhang, B.; Wang, K.; Wang, H. LncRNA CTBP1-AS2 alleviates high glucose-induced oxidative stress, ECM accumulation, and inflammation in diabetic nephropathy via miR-155-5p/FOXO1 axis. Biochem. Biophys. Res. Commun. 2020, 532, 308–314. [Google Scholar] [CrossRef]
- Gao, J.; Zhao, G.; Li, W.; Zhang, J.; Che, Y.; Song, M.; Gao, S.; Zeng, B.; Wang, Y. MiR-155 targets PTCH1 to mediate endothelial progenitor cell dysfunction caused by high glucose. Exp. Cell Res. 2018, 366, 55–62. [Google Scholar] [CrossRef]
- Li, X.; Jia, Z.; Zhao, X.; Xu, M.; Chen, M. Expression of miR-210 in the peripheral blood of patients with newly diagnosed type 2 diabetes mellitus and its effect on the number and function of endothelial progenitor cells. Microvasc. Res. 2020, 131. [Google Scholar] [CrossRef]
- Chen, X.; Tian, F.; Sun, Z.; Zeng, G.; Tang, P. Elevation of Circulating miR-210 Participates in the Occurrence and Development of Type 2 Diabetes Mellitus and Its Complications. J. Diabetes Res. 2022, 2022. [Google Scholar] [CrossRef]
- Li, T.; Cao, H.; Zhuang, J.; Wan, J.; Guan, M.; Yu, B.; Li, X.; Zhang, W. Identification of miR-130a, miR-27b and miR-210 as serum biomarkers for atherosclerosis obliterans. Clin. Chim. Acta 2011, 412, 66–70. [Google Scholar] [CrossRef]
- Chen, K.C.; Liao, Y.C.; Wang, J.Y.; Lin, Y.C.; Chen, C.H.; Juo, S.H.H. Oxidized low-density lipoprotein is a common risk factor for cardiovascular diseases and gastroenterological cancers via epigenomical regulation of microRNA-210. Oncotarget 2015, 6, 24105–24118. [Google Scholar] [CrossRef]
- Ismaeel, A.; FLETCHER, E.; MISERLIS, D.; WECHSLER, M.; PAPOUTSI, E.; HAYNATZKI, G.; SMITH, R.S.; BOHANNON, W.T.; KOUTAKIS, P. Skeletal muscle MiR-210 expression is associated with mitochondrial function in peripheral artery disease patients. Transl. Res. 2022, 246, 66–77. [Google Scholar] [CrossRef] [PubMed]
- Patra, D.; Roy, S.; Arora, L.; Kabeer, S.W.; Singh, S.; Dey, U.; Banerjee, D.; Sinha, A.; Dasgupta, S.; Tikoo, K.; et al. miR-210-3p Promotes Obesity-Induced Adipose Tissue Inflammation and Insulin Resistance by Targeting SOCS1-Mediated NF-kB Pathway. Diabetes 2023, 72, 375–388. [Google Scholar] [CrossRef] [PubMed]
- Taganov, K.D.; Boldin, M.P.; Chang, K.-J.; Baltimore, D. NF-kappaB-dependent induction of microRNA miR-146, an inhibitor targeted to signaling proteins of innate immune responses. Proc. Natl. Acad. Sci. U. S. A. 2006, 103, 12481–12486. [Google Scholar] [CrossRef] [PubMed]
- Ghaffari, M.; Razi, S.; Zalpoor, H.; Nabi-Afjadi, M.; Mohebichamkhorami, F.; Zali, H. Association of MicroRNA-146a with Type 1 and 2 Diabetes and their Related Complications. J. Diabetes Res. 2023, 2023. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; He, Y.; Wang, X.; Zhu, Y.; Huang, Y.; Cao, J.; Yan, R. LncRNA PTGS2 regulates islet β-cell function through the miR-146a-5p/RBP4 axis and its diagnostic value in type 2 diabetes mellitus. Am. J. Transl. Res. 2021, 13, 11316–11328. [Google Scholar]
- Olivieri, F.; Prattichizzo, F.; Giuliani, A.; Matacchione, G.; Rippo, M.R.; Sabbatinelli, J.; Bonafè, M. miR-21 and miR-146a: The microRNAs of inflammaging and age-related diseases. Ageing Res. Rev. 2021, 70. [Google Scholar] [CrossRef]
- Zhou, Z.; Wan, J.; Hou, X.; Geng, J.; Li, X.; Bai, X. MicroRNA-27a promotes podocyte injury via PPARγ-mediated β-catenin activation in diabetic nephropathy. Cell Death Dis. 2017, 8, 1–13. [Google Scholar] [CrossRef]
- Hou, X.; Tian, J.; Geng, J.; Li, X.; Tang, X.; Zhang, J.; Bai, X. MicroRNA-27a promotes renal tubulointerstitial fibrosis via suppressing PPARγ pathway in diabetic nephropathy. Oncotarget 2016, 7, 47760–47776. [Google Scholar] [CrossRef]
- Song, J.; Zhang, H.; Sun, Y.; Guo, R.; Zhong, D.; Xu, R.; Song, M. Omentin-1 protects renal function of mice with type 2 diabetic nephropathy via regulating miR-27a-Nrf2/Keap1 axis. Biomed. Pharmacother. 2018, 107. [Google Scholar] [CrossRef]
- Li, J.M.; Li, X.; Chan, L.W.C.; Hu, R.; Zheng, T.; Li, H.; Yang, S. Lipotoxicity-polarised macrophage-derived exosomes regulate mitochondrial fitness through Miro1-mediated mitophagy inhibition and contribute to type 2 diabetes development in mice. Diabetologia 2023, 66, 2368–2386. [Google Scholar] [CrossRef]
- Akpınar, K.; Aslan, D.; Fenkçi, S.M.; Caner, V. miR-21-3p and miR-192-5p in patients with type 2 diabetic nephropathy. Diagnosis 2022, 9, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Phu, T.A.; Ng, M.; Vu, N.K.; Bouchareychas, L.; Raffai, R.L. IL-4 polarized human macrophage exosomes control cardiometabolic inflammation and diabetes in obesity. Mol. Ther. 2022, 30, 2274–2297. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Yin, T.; Chen, D.; Xu, S.; Ye, R.; Zhang, Y. Astragaloside IV Regulates Insulin Resistance and Inflammatory Response of Adipocytes via Modulating MIR-21/PTEN/PI3K/AKT Signaling. Endocrine, Metab. Immune Disord. - Drug Targets 2023, 23, 1538–1547. [Google Scholar] [CrossRef]
- Doghish, A.S.; Elsisi, A.M.; Amin, A.I.; Abulsoud, A.I. Circulating miR-148a-5p and miR-21-5p as Novel Diagnostic Biomarkers in Adult Egyptian Male Patients With Metabolic Syndrome. Can. J. Diabetes 2021, 45, 614–618. [Google Scholar] [CrossRef] [PubMed]
- Yazdanpanah, Z.; Kazemipour, N.; Kalantar, S.M.; Vahidi Mehrjardi, M.Y. Plasma miR-21 as a potential predictor in prediabetic individuals with a positive family history of type 2 diabetes mellitus. Physiol. Rep. 2022, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Helal, H.G.; Rashed, M.H.; Abdullah, O.A.; Salem, T.I.; Daifalla, A. MicroRNAs (−146a, −21 and −34a) are diagnostic and prognostic biomarkers for diabetic retinopathy. Biomed. J. 2021, 44, S242–S251. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Li, M.; Zheng, M.; Tang, Y.; Yang, Z.; Ma, G.; Zheng, Q.; Li, L.; Wang, Y.; Ma, F.; et al. Diagnostic value of galectin-3, fractalkine, IL-6, miR-21 and cardiac troponin I in human ischemic cardiomyopathy. Aging (Albany. NY). 2024, 16, 10539–10545. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Chen, T.; Li, X.; Zhu, Y.; Zhang, S.; He, P.; Peng, Y.; Fan, Q. The relevance of the non-invasive biomarkers lncRNA GAS5/miR-21 ceRNA regulatory network in the early identification of diabetes and diabetic nephropathy. Diabetol. Metab. Syndr. 2023, 15, 1–10. [Google Scholar] [CrossRef]
- Gongol, B.; Marin, T.; Zhang, J.; Wang, S.C.; Sun, W.; He, M.; Chen, S.; Chen, L.; Li, J.; Liu, J.H.; et al. Shear stress regulation of miR-93 and miR-484 maturation through nucleolin. Proc. Natl. Acad. Sci. U. S. A. 2019, 116, 12974–12979. [Google Scholar] [CrossRef]
- Wang, K.; Long, B.; Jiao, J.Q.; Wang, J.X.; Liu, J.P.; Li, Q.; Li, P.F. MiR-484 regulates mitochondrial network through targeting Fis1. Nat. Commun. 2012, 3. [Google Scholar] [CrossRef]
- Jia, Y.Z.; Liu, J.; Wang, G.Q.; Song, Z.F. miR-484: A Potential Biomarker in Health and Disease. Front. Oncol. 2022, 12, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Caparosa, E.M.; Sedgewick, A.J.; Zenonos, G.; Zhao, Y.; Carlisle, D.L.; Stefaneanu, L.; Jankowitz, B.T.; Gardner, P.; Chang, Y.-F.; Lariviere, W.R.; et al. Regional Molecular Signature of the Symptomatic Atherosclerotic Carotid Plaque. Neurosurgery 2019, 85, E284–E293. [Google Scholar] [CrossRef] [PubMed]
- Macgrogan, D.; De La Pompa, J.L. DACH1-Driven Arterialization: Angiogenic Therapy for Ischemic Heart Disease? Circ. Res. 2021, 129, 717–719. [Google Scholar] [CrossRef] [PubMed]
- Raitoharju, E.; Seppälä, I.; Oksala, N.; Lyytikäinen, L.P.; Raitakari, O.; Viikari, J.; Ala-Korpela, M.; Soininen, P.; Kangas, A.J.; Waldenberger, M.; et al. Blood microRNA profile associates with the levels of serum lipids and metabolites associated with glucose metabolism and insulin resistance and pinpoints pathways underlying metabolic syndrome: The cardiovascular risk in Young Finns Study. Mol. Cell. Endocrinol. 2014, 391, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Witko-Sarsat, V.; Friedlander, M.; Capeillère-Blandin, C.; Nguyen-Khoa, T.; Nguyen, A.T.; Zingraff, J.; Jungers, P.; Descamps-Latscha, B. Advanced oxidation protein products as a novel marker of oxidative stress in uremia. Kidney Int. 1996. [Google Scholar] [CrossRef] [PubMed]
- Hissin, P.J.; Hilf, R. A fluorometric method for determination of oxidized and reduced glutathione in tissues. Anal. Biochem. 1976. [Google Scholar] [CrossRef]
- Jaskot, R.H.; Charlet, E.G.; Grose, E.C.; Grady, M.A.; Roycroft, J.H.; Roycroft, J.H. An Automated Analysis of Glutathione Peroxidase, S-Transferase, and Reductase Activity in Animal Tissue. J. Anal. Toxicol. 1983. [Google Scholar] [CrossRef]
- Misra, H.P.; Fridovich, I. The role of superoxide anion in the autoxidation of epinephrine and a simple assay for superoxide dismutase. J. Biol. Chem. 1972. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro assay methods. Methods Enzymol. 1984. [Google Scholar]
| Parameters | CG (n=30) |
DG (n=34) |
DG + C (n=28) |
p value |
|---|---|---|---|---|
| Age | 51.43 ± 1.77 | 63.47 ± 1.30a | 64.96 ± 1.68b | a,bp < 0.001 |
| Gender (female/men) | 18/12 | 18/16 | 5/23 | |
| Weight (kg) | 70.83 ± 2.18 | 78.81 ± 2.69 | 86.66 ± 2.22b | bp < 0.001 |
| BMI (kg/m2) | 25.18 ± 0.61 | 29.84 ± 0.90a | 30.62 ± 0.86b | a,bp < 0.001 |
| Years of diabetes | no | 13.33 ± 1.85 | 17.43 ± 2.02 | |
| HbA1c (%) | 5.37 ± 0.07 | 6.85 ± 0.16a | 7.11 ± 0.19b | a,bp < 0.001 |
| Glucose (mg/dL) | 90.33 ± 2.68 | 123.5 ± 4.84a | 141.9 ± 9.34b | a,bp < 0.001 |
| Insulin (mU/l) | 6.76 ± 0.74 | 11.51 ± 1.68 a | 11.67 ± 2.02 b | a,bp < 0.001 |
| HOMA-IR Index | 1.50 ± 0.23 | 3.66 ± 0.63a | 3.99 ± 0.66b |
ap < 0.01 bp < 0.001 |
| Creatinine (mg/dL) | 0.80 ± 0.16 | 0.84 ± 0.18 | 0.98 ± 0.35 | bp = 0.019 |
| Urea (mg/dL) | 35.41 ± 2.34 | 42.45 ± 2.47 | 45.00 ± 4.82 | ap = 0.035 |
| Total cholesterol (mg/dL) | 198.8 ± 5.79 | 168.4 ± 6.30a | 136.3 ± 7.50b, c |
ap = 0.003 bp < 0.001 cp = 0.002 |
| TG (mg/dL) | 90.33 ± 5.73 | 165.3 ± 23.20a | 140.3 ± 14.29b |
ap = 0.01 bp = 0.05 |
| HDL (mg/dL) | 62.13 ± 2.41 | 52.65 ± 2.50a | 44.96 ± 2.16b |
ap = 0.01 bp < 0.001 |
| LDL (mg/dL) | 119.5 ± 5.21 | 85.97 ± 5.72a | 66.63 ± 6.32b | a,bp < 0.001 |
| Drug Therapy | Microangiopathies (n = 6) | CVD (n = 22) |
|---|---|---|
| 21.4% | 78.6% | |
| Glucose-lowering medication: | ||
| Metformin | 66.7% (4/6) | 68.2% (15/22) |
| Antihyperglycemic agents * | 83.3% (5/6) | 90.9% (20/22) |
| Insulin sensitive therapy** | 66.7% (4/6) | 80.0% (16/22) |
| Cholesterol-lowering therapy (statins) | 66.7% (4/6) | 50.0% (11/22) |
| Anticonvulsants | 0.0% | 22.7% (5/22) |
| Glucose | HbA1c | HOMA-IR | Total cholesterol | LDL | Creatinine | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| r p | r p | r p | r p | r p | r | p | ||||||
| GPx | -0.068 | 0.638 | -0.134 | 0.352 | -0.297 | 0.036 | -0.323 | 0.022 | -0.234 | 0.102 | 0.066 | 0.651 |
| GRd | 0.324 | 0.022 | 0.095 | 0.514 | -0.047 | 0.745 | -0.101 | 0.485 | -0.100 | 0.490 | 0.087 | 0.548 |
| Catalase | -0.044 | 0.764 | 0.020 | 0.892 | 0.127 | 0.258 | -0.410 | 0.003 | -0.404 | 0.004 | -0.209 | 0.146 |
| G6PD | 0.074 | 0.608 | 0.001 | 0.997 | -0.115 | 0.566 | -0.297 | 0.036 | -0.325 | 0.021 | -0.051 | 0.725 |
| GSH | -0.163 | 0.257 | -0.185 | 0.200 | 0.030 | 0.836 | -0.302 | 0.033 | -0.288 | 0.042 | 0.144 | 0.319 |
| IL-8 | 0.130 | 0.424 | 0.104 | 0.521 | -0.010 | 0.953 | 0.330 | 0.022 | 0.336 | 0.061 | -0.068 | 0.677 |
| IL-10 | -0.167 | 0.302 | -0.177 | 0.274 | -0.365 | 0.020 | 0.037 | 0.819 | 0.062 | 0.697 | 0.062 | 0.703 |
| MCP-1 | -0.215 | 0.183 | -0.171 | 0.290 | -0.087 | 0.595 | -0.047 | 0.771 | -0.058 | 0.714 | 0.366 | 0.020 |
| miRNA | Oxidative stress parameters |
r-value | p-value |
|---|---|---|---|
| Diabetic patients | |||
| miR-21 | SOD | -0.346 | 0.009 |
| LPO | 0.266 | 0.048 | |
| miR-126 | LPO | 0.424 | < 0.001 |
| miR-27a | LPO | 0.321 | 0.017 |
| miR-210 | CAT | 0.413 | 0.001 |
| Diabetic patients were adjusted for age, gender, and BMI. | |||
| miR-21 | SOD | -0.289 | 0,036 |
| miR-126 | LPO | 0.447 | 0.001 |
| AOPP | 0.281 | 0.046 | |
| miR-27a | LPO | 0.425 | 0.003 |
| AOPP | 0.288 | 0.047 | |
| miR-210 | CAT | 0.312 | 0.029 |
| LPO | 0.292 | 0.042 | |
| miR-484 | CAT | 0.360 | 0.039 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




