Submitted:
27 September 2024
Posted:
30 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Phage Library and Bacterial Strain
2.3. Immobilization of Copper Ions on Plates
2.4. Surface Panning Procedure
2.5. Binding Specificity Assay
2.6. Peptide Synthesis, Modification and Characterization
2.7. General Spectroscopy Measurements
3. Results and Discussion
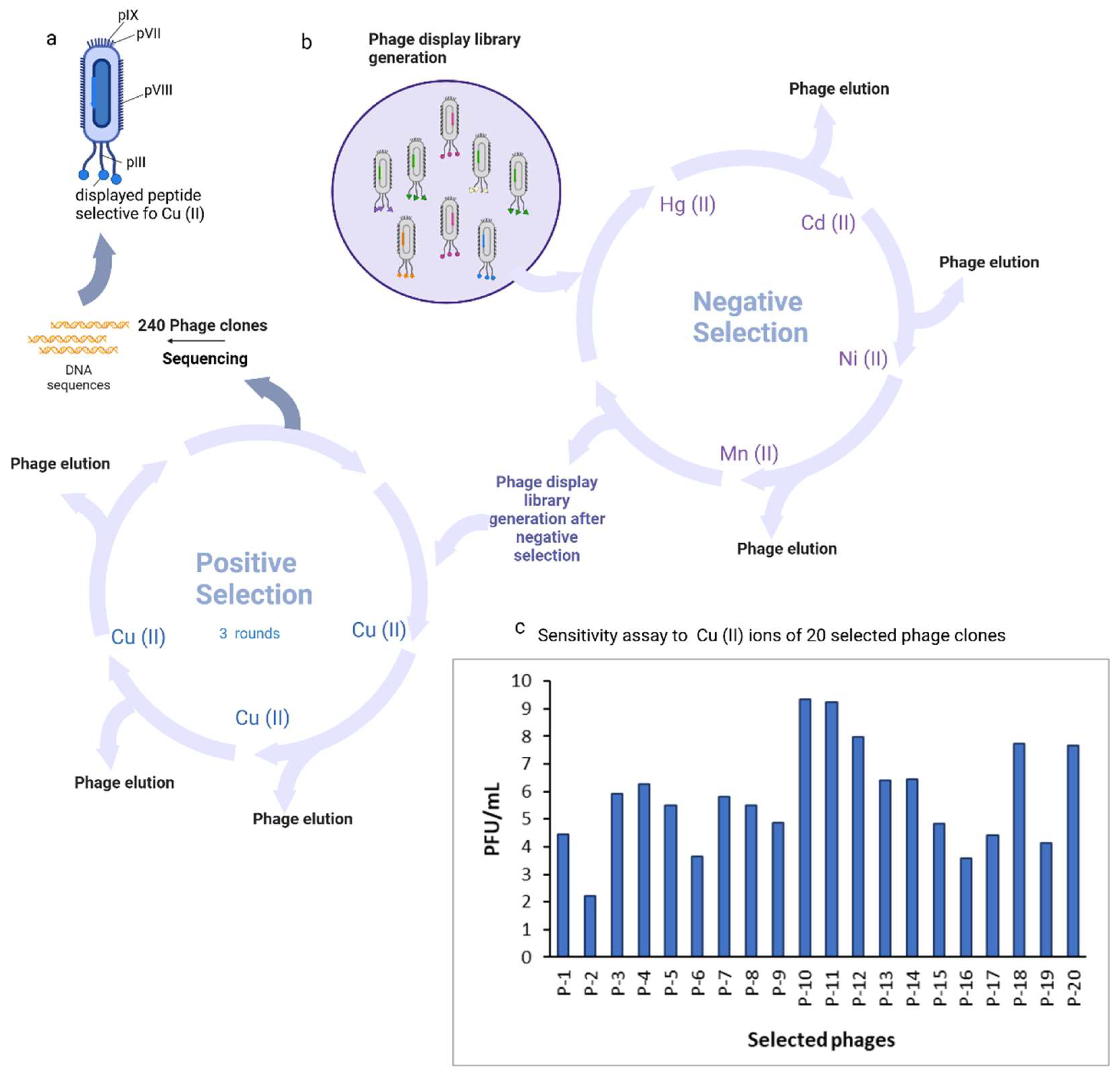
3.1. Phage Display Derived Unique Peptide for Cu2+ Ions Detection
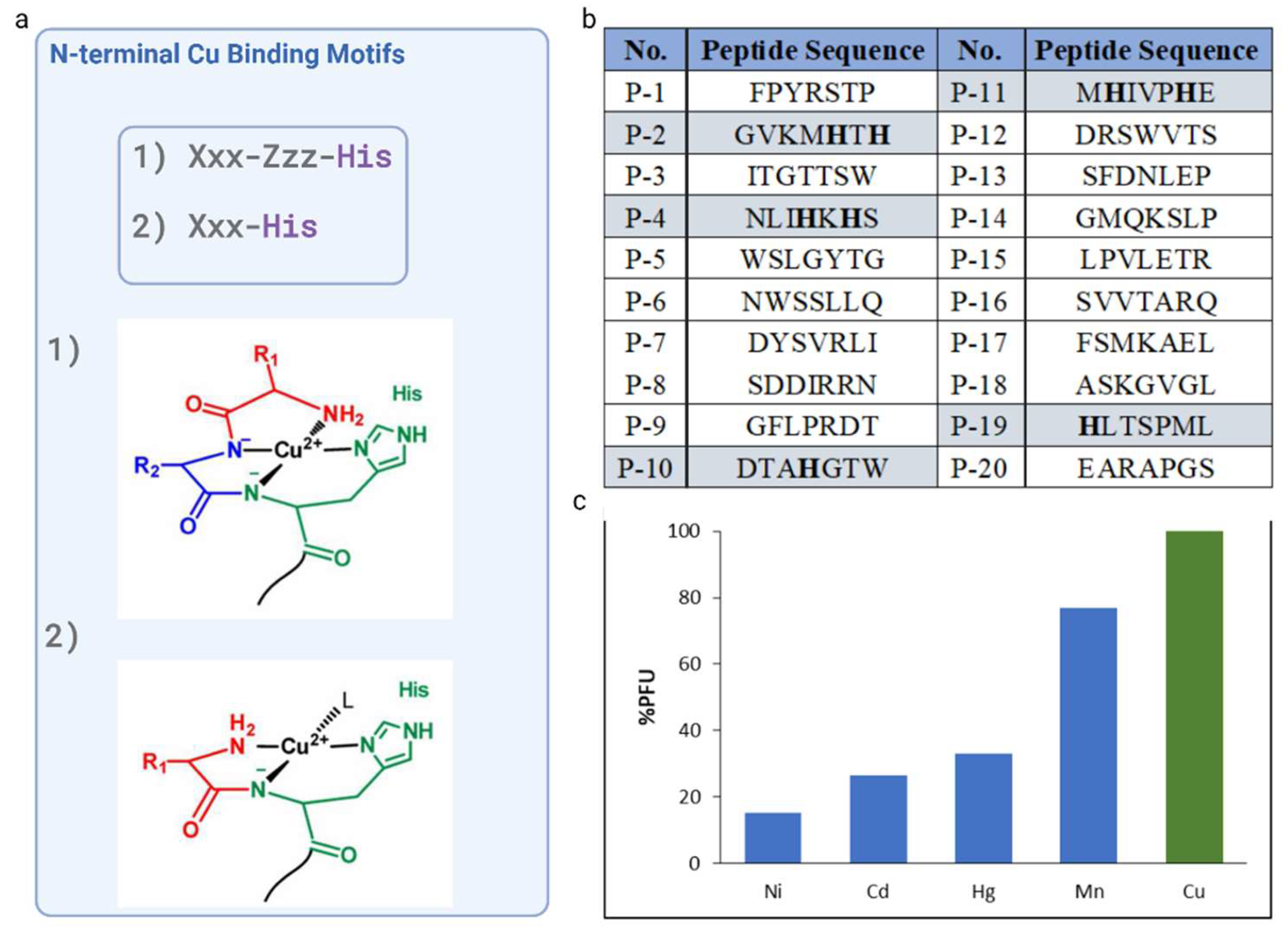
3.2. Characterization and Amio Acid Sequences Analysis of the Unique Phage Display Derive Peptides
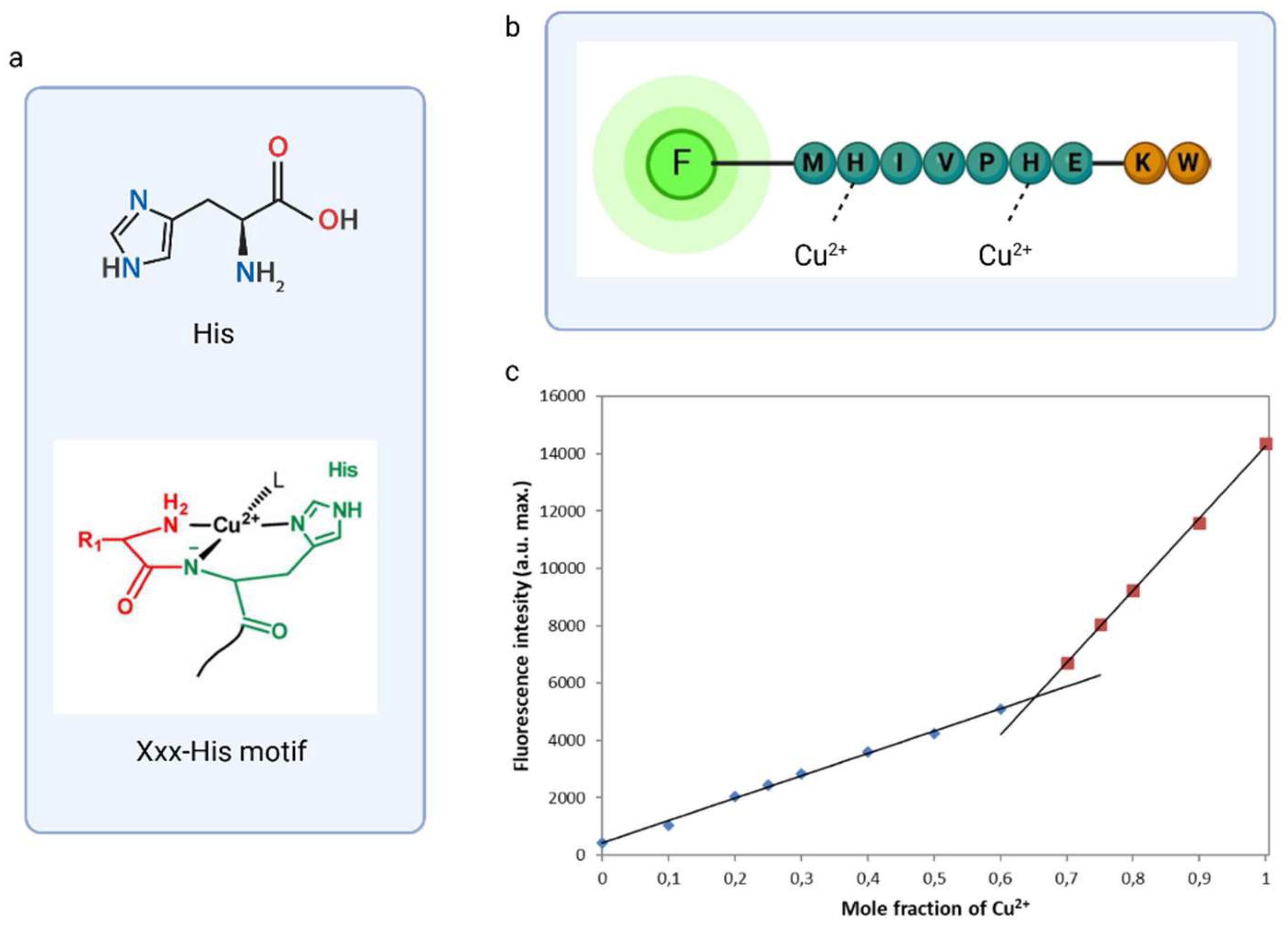
3.3. Design of Modifications and Sold Phase Peptide Synthesis
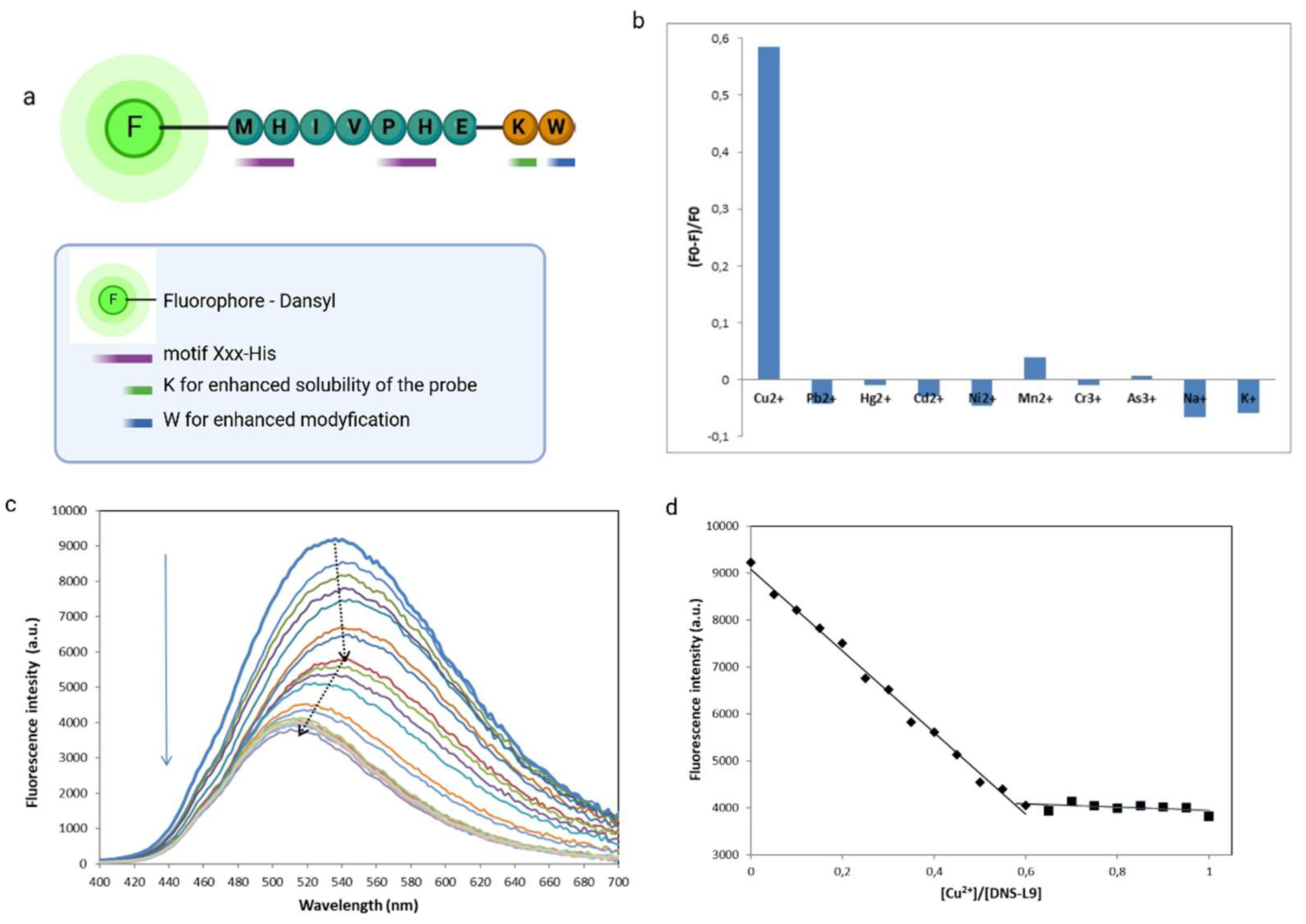
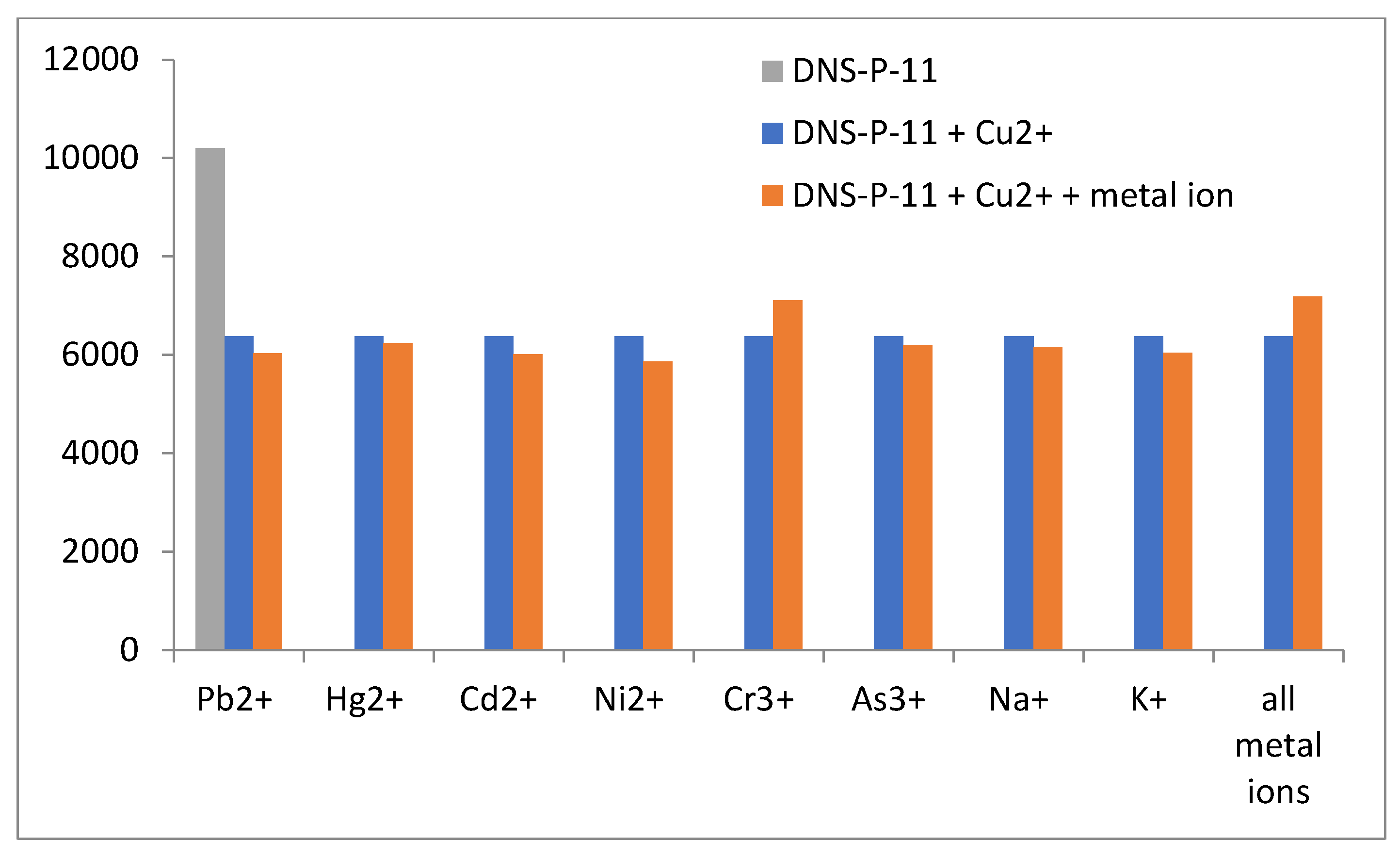
3.4. Metal Ions Selectivity Experiment
3.5. Characterization of The Sensing Process of Fluorescent Chemosensor based on Phage Display Derived Peptide for Cu2+ Detection
3.6. Analysis of Selectivity and Sensitivity of Fluorescent Chemosensor (DNS−P-11) based on Phage Display Derived Peptide for Cu2+ Detection
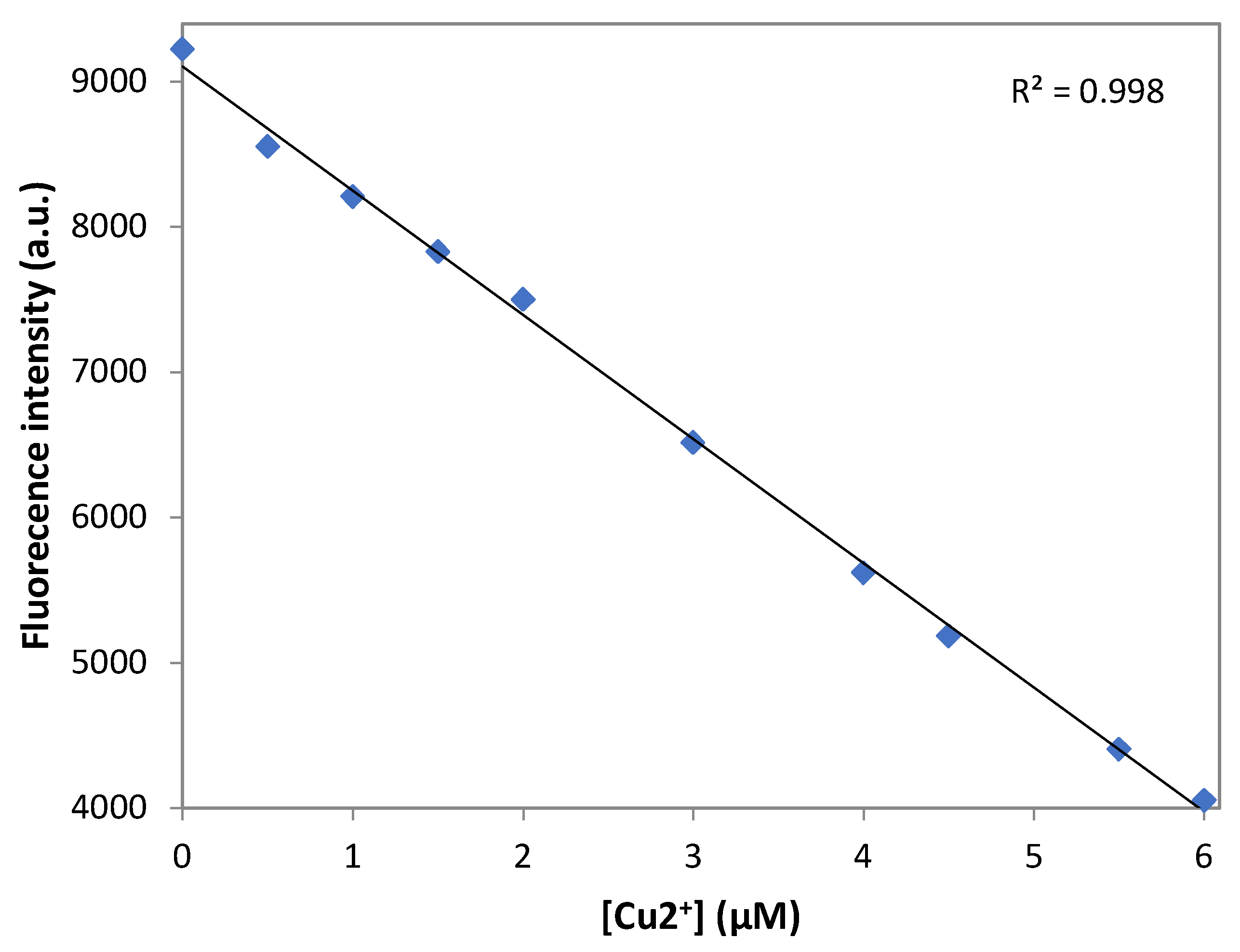
3.7. Detection Limit of DNS–P-11 for Cu2+ Ions
4. Conclusions
5. Patents
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Taylor, A.A.; Tsuji, J.S.; Garry, M.R.; McArdle, M.E.; Goodfellow Jr, W.L; Adams, W.J.; Menzie, C.A. Critical Review of Exposure and Effects: Implications for Setting Regulatory Health Criteria for Ingested Copper. Environmental Management 2020, 65, 131–159. [Google Scholar] [CrossRef] [PubMed]
- Gaetke, L.M.; Chow, C.H. ; Copper toxicity, oxidative stress, and antioxidant nutrients, Toxicology 2003, 189, 1–2, 147-163. [CrossRef]
- Ejaz, H.W.; Wang, W.; Lang, M. Copper Toxicity Links to Pathogenesis of Alzheimer’s Disease and Therapeutics Approaches. Int. J. Mol. Sci. 2020, 21, 7660. [Google Scholar] [CrossRef] [PubMed]
- Strausak, D.; Mercer, J.F.B.; Dieter, H.H.; Stremmel, W.; Multhaup, G. Copper in disorders with neurological symptoms: Alzheimer’s, Menkes, and Wilson diseases, Brain Research Bulletin 2001, 55, 175-185. [CrossRef]
- Karpenko, M.N.; Muruzheva, Z.M.; Ilyechova, E.Y.; Babich, P.S.; Puchkova, L.V. Abnormalities in Copper Status Associated with an Elevated Risk of Parkinson’s Phenotype Development. Antioxidants 2023, 12, 1654. [Google Scholar] [CrossRef] [PubMed]
- Elkhatat, A.M.; Soliman, M.; Ismail, R.; Ahmed, S.; Abounahia, N.; Mubashir, S.; Fouladi, S.; Khraisheh, M. Recent trends of copper detection in water samples. Bull Natl Res Cent 2021, 45, 218. [Google Scholar] [CrossRef]
- Kateshiya, M.R. , Desai, M.L., Malek, N.I. et al. Advances in Ultra-small Fluorescence Nanoprobes for Detection of Metal Ions, Drugs, Pesticides and Biomarkers. J Fluoresc. 2023, 33, 775–798. [CrossRef]
- Shi, Y.; Zhang, W.; Xue, Y.; Zhang, J. Fluorescent Sensors for Detecting and Imaging Metal Ions in Biological Systems: Recent Advances and Future Perspectives. Chemosensors 2023, 11, 226. [Google Scholar] [CrossRef]
- Wang, P.; Zhou, D.; Chen, B. A fluorescent dansyl-based peptide probe for highly selective and sensitive detect Cd2+ ions and its application in living cell imaging. Spectrochim Acta A Mol Biomol Spectrosc 2019, 207, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Wei, P.; Xiao, L.; Gou, Y.; He, F.; Wang, P.; Yang, X. A novel peptide-based relay fluorescent probe with a large Stokes shift for detection of Hg2+ and S2− in 100 % aqueous medium and living cells: Visual detection via test strips and smartphone. Spectrochim Acta A Mol Biomol Spectrosc 2023, 285, 121836. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Wu, J.; Zhao, C. A water-soluble peptide fluorescent chemosensor for detection of cadmium (II) and copper (II) by two different response modes and its application in living LNcap cells, Spectrochim Acta A Mol Biomol Spectrosc 2020, 226, 117600. [CrossRef]
- Wang, P.; Wu, J. Highly selective and sensitive detection of Zn(II) and Cu(II) ions using a novel peptide fluorescent probe by two different mechanisms and its application in live cell imaging, Spectrochim Acta A Mol Biomol Spectrosc 2019, 208, 140-149. [CrossRef]
- Román-Azcona, M.S.; Trigüis, S.; Caballero, S.; Michajluck, J.; Sotelo, P.H. Generation of a cadmium-binding filamentous phage through cysteine-rich peptide display on PVIII. Indian J. Biotechnol. 2019, 18, 132-138.
- Li, H.; Dong, W.; Liu, Y.; Zhang, H.; Wang, G. Enhanced Biosorption of Nickel Ions on Immobilized Surface-Engineered Yeast Using Nickel-Binding Peptides. Front Microbiol. 2019, 26, 1254. [Google Scholar] [CrossRef]
- Matys, S.; Schönberger, N.; Lederer, F.L.; Pollmann, K. Characterization of specifically metal-binding phage clones for selective recovery of cobalt and nickel. J. Environ. Chem. Eng. 2020, 2, 103606. [Google Scholar] [CrossRef]
- Korkmaz, N. , Kim, M. Phage display selection of a Pb(II) specific peptide and its application as a biorecognition unit for colorimetric detection of Pb(II) ions. Biotechnol. J. 2024, 19, 2300482. [CrossRef]
- Korkmaz, N.; Hwang, C.; Kessler, K.K.; Silina, Y.E.; Müller, L.; Park, J. A novel copper (II) binding peptide for a colorimetric biosensor system design. Talanta 2021, 232, 122439. [Google Scholar] [CrossRef]
- Yang, T.; Zhang, X.Y.; Zhang, X.X.; Chen, M.L.; Wang, J.H. Chromium(III) Binding Phage Screening for the Selective Adsorption of Cr(III) and Chromium Speciation. ACS Appl Mater Interfaces 2015, 30, 21287–21294. [Google Scholar] [CrossRef]
- . Henryk Kozłowski, H,; Bal, W.; Dyba, M.; Kowalik-Jankowska, T. Specific structure–stability relations in metallopeptide. Coord. Chem. Rev. 1999, 184,1, 319-346. [CrossRef]
- Gonzalez, P.; Vileno, B.; Bossak, K.; Khoury, Y.E.; Hellwig, P.; Bal, W.; Hureau, C.; Faller, P. Cu(II) Binding to the Peptide Ala-His-His, a Chimera of the Canonical Cu(II)-Binding Motifs Xxx-His and Xxx-Zzz-His. Inorg. Chem. 2017, 24, 14870–14879. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, P.; Bossak, K. ; Stefaniak,E. ; Hureau, C.; Raibaut, L.; Bal, W.; Falle, P. N-Terminal Cu-Binding Motifs (Xxx-Zzz-His, Xxx-His) and Their Derivatives: Chemistry, Biology and Medicinal Applications. Chem. Eur. J. 2018, 32, 8029–8041. [Google Scholar] [CrossRef]
- Noormägi, A.; Golubeva, T.; Berntsson, E.; Wärmlänr, S.K.T.S.; Tõugu, V.; Palumaa, P. Direct Competition of ATCUN Peptides with Human Serum Albumin for Copper(II) Ions Determined by LC-ICP MS. ACS Omega 2023, 8, 37, 33912–33919. [Google Scholar] [CrossRef]
- . Maiti, B.K.; Govil, N.; Kundu, T.; Moura, J.J.G. Designed Metal-ATCUN Derivatives: Redox- and Non-redox-Based Applications Relevant for Chemistry, Biology, and Medicine. iScience 2020, 23, 101792. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, S.L.; Tofteng, A.P.; Malik, L.; Jensen, K.J. Microwave heating in solid-phase peptide synthesis. Chem. Soc. Rev. 2012, 41, 1826–1844. [Google Scholar] [CrossRef] [PubMed]
- Pang, X.; Dong, J.; Gao, L.; Wang, L. ; Yu,S. ; Kong, J.; Li, L.Dansyl-peptide dual-functional fluorescent chemosensor for Hg2+ and biothiols. Dyes Pigm. 2020, 173, 107888. [Google Scholar] [CrossRef]
- Wang, P.; Zhou, D.; Chen, B. A fluorescent dansyl-based peptide probe for highly selective and sensitive detect Cd2+ ions and its application in living cell imaging. Spectrochim Acta A Mol Biomol Spectrosc. 2019, 207, 276–283. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



