Submitted:
24 September 2024
Posted:
25 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
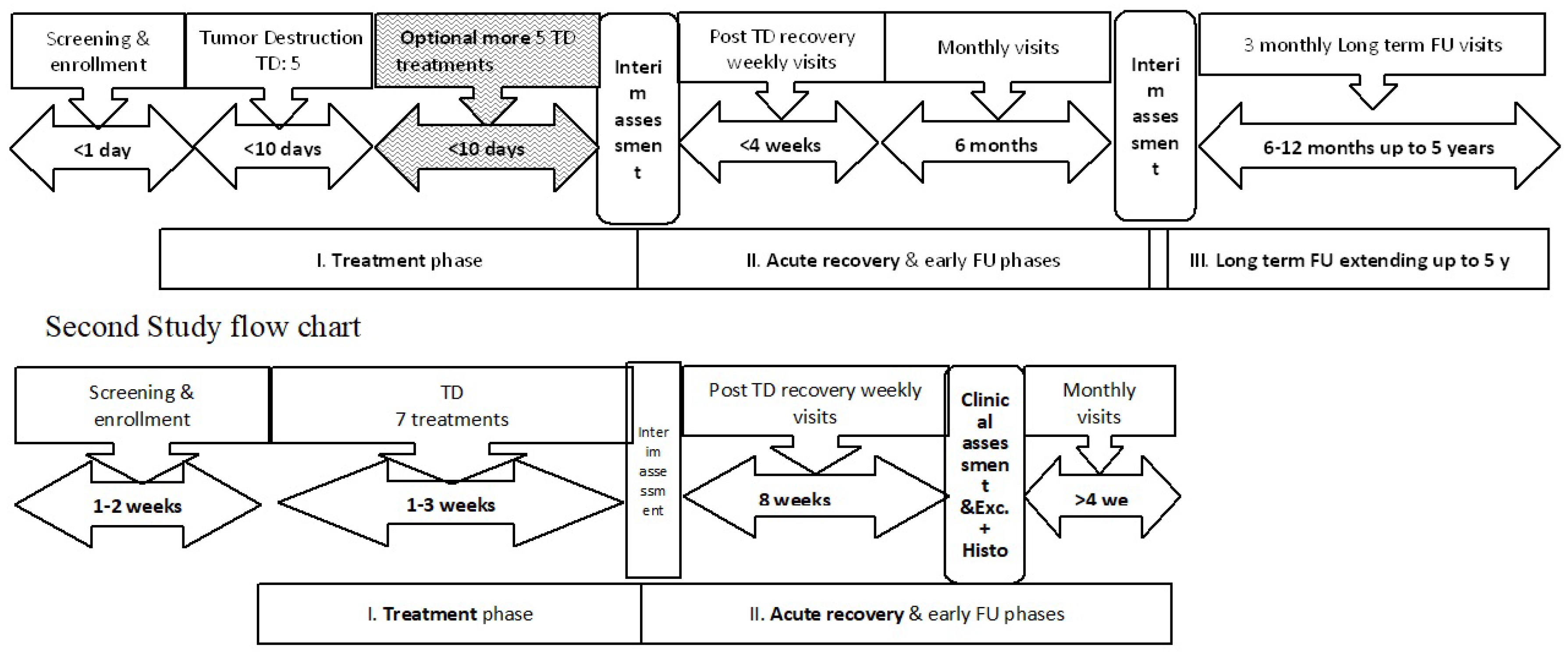
2. Materials and Methods
3. Results
Safety Results
4. Discussion
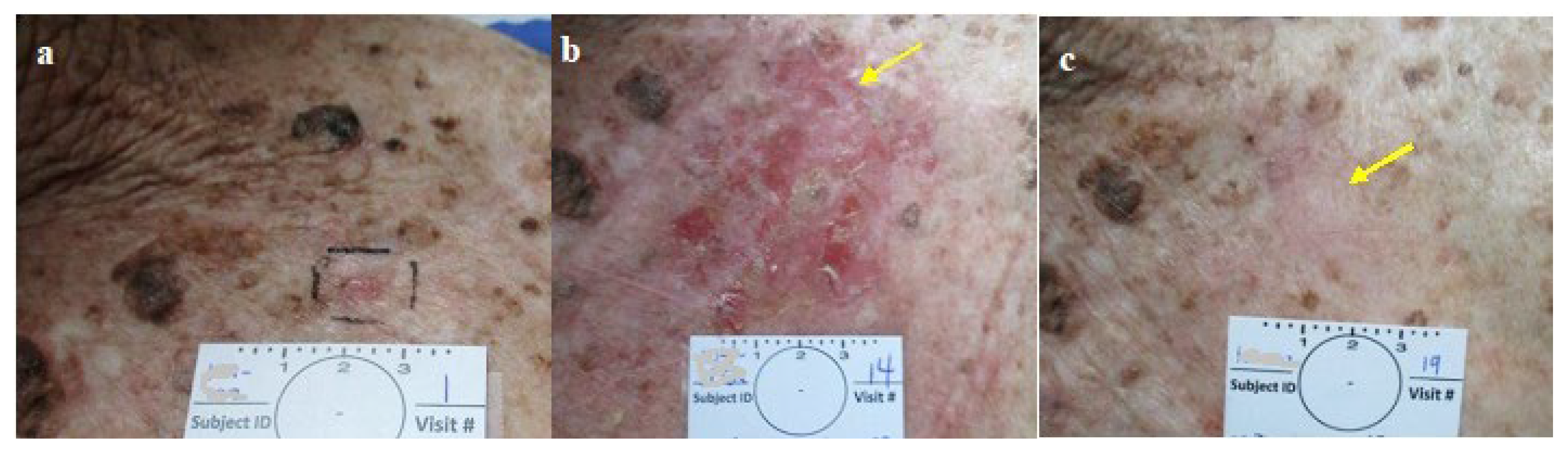
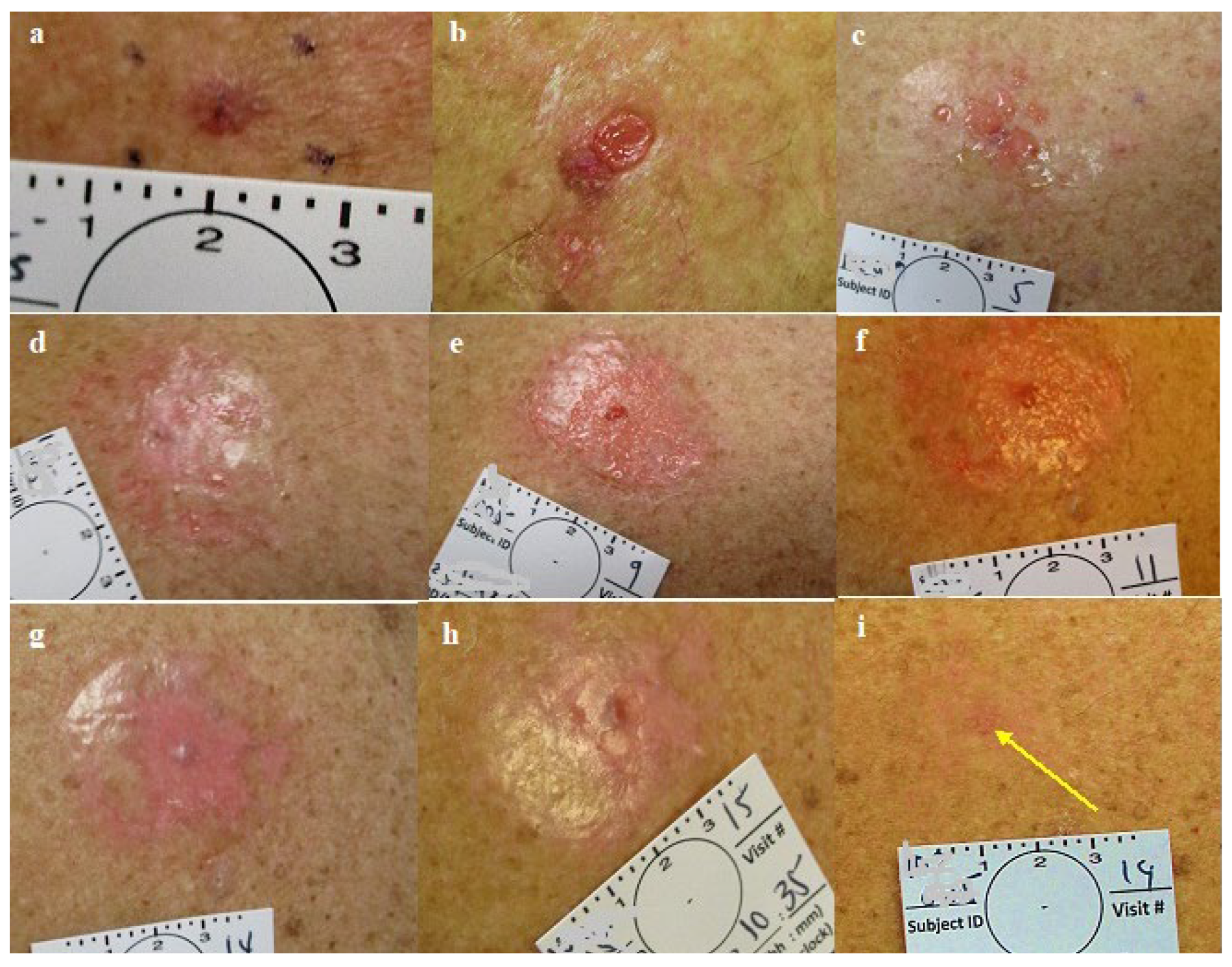
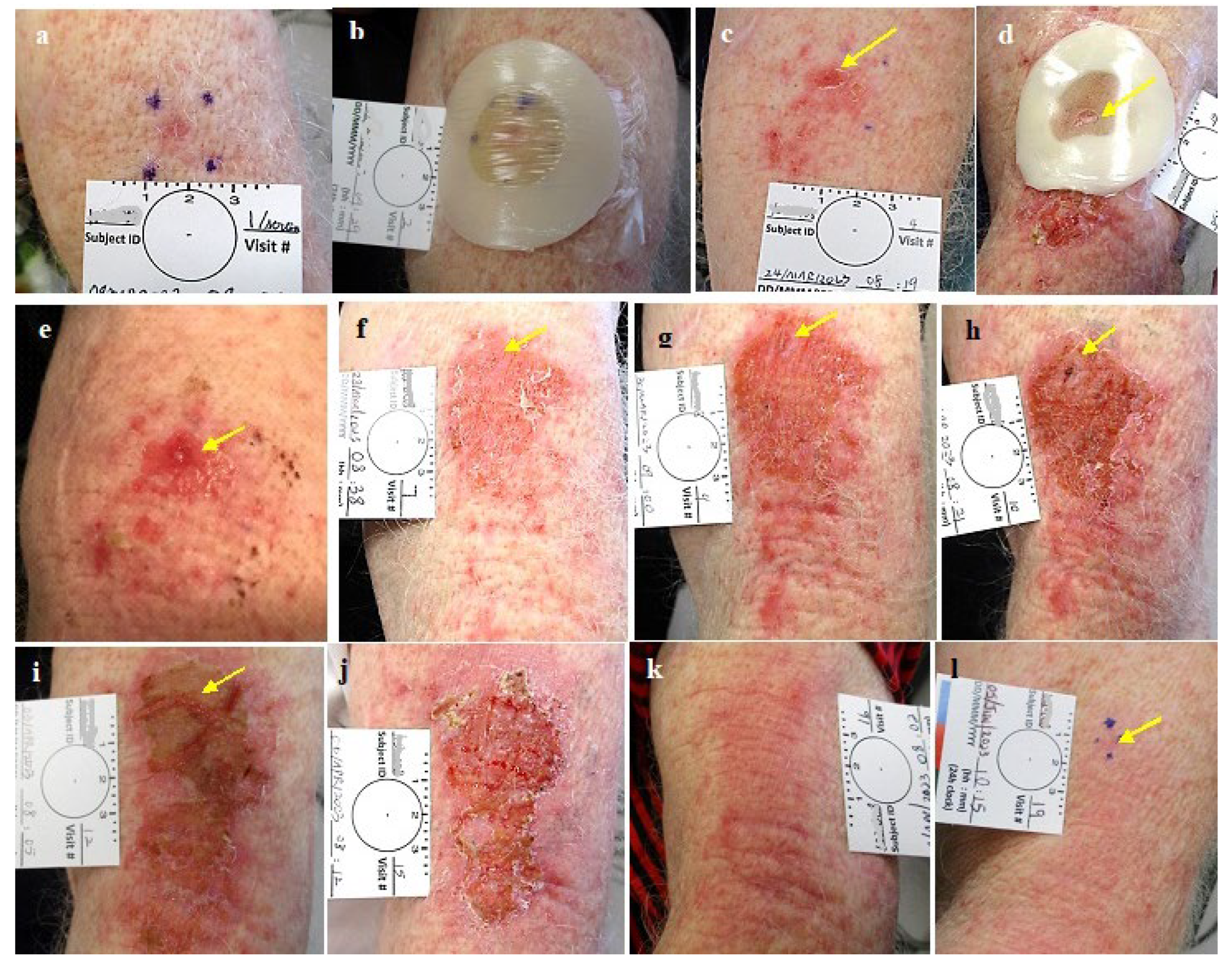
- Proteolysis of the different cutaneous structures according to their extra cellular matrix (ECM) density. This strong activity quickly destroys the tumors’ matrices that differ from the normal, denser and more resistant dermal collagenous extracellular matrix (ECM). This potent proteolysis explains the rapid (few days) eradication of these tumors that does not depend on the long anti-tumorigenic/proapoptotic activities that are related to the tumor’s cells growth cycle, which has a much slower effect. This proteolysis as seen in these studies seems to be dose/application related, also affecting the denser surrounding dermis but in a much weaker way, causing only a superficial erosion of the epidermis and the basal membrane complex underneath. This superficial erosion destroys the tumor’s horizontal edges as well as its vertical edges that penetrate the basal membrane. The immediate destruction of the tumor with its adjacent edges and the basal membrane is similar to surgical excision that removes the tumor with its surrounding tissues.
- 2.
- The reduced concentration from 10% to 5% compared to the first POC may have caused the efficacy to be more application technique sensitive.
- The application technique should be fine-tuned following the present study conclusions.
- Though histopathological diagnosis is considered the gold standard for tumor diagnosis, in clinical practice, the majority of low risk NMSC KC (BCC, SCC and SK) are clinically-visually diagnosed, with or without dermatoscopy. Based on this non invasive diagnosis, patients are treated (surgically and/or non-surgically) and followed for possible recurrency. Pretreatment histopathology is reserved for the more invasive cases and as a prerequisite for insurance coverage of the entire surgical treatment process that involves two procedures (pre and post treatment surgery) and histopathology. Using biopsies and diagnostic histopathology in trials of low risk lesions does confirm the nature of the lesion that is treated and the completness of eradication but adds a layer of costs, complexity and scarring. The patient with small, low-risk BCC that is aware of the non-surgical, conservative alternatives may be reluctant to join such a study that involves two surgical procedures and more scarring. Thus, designing a surgery-free trial that is based on pre and post treatment clinical/dermatoscopy diagnosis is challanging and will involve more prolonged follow-up (up to 5 years) but may be more appealing to patients.
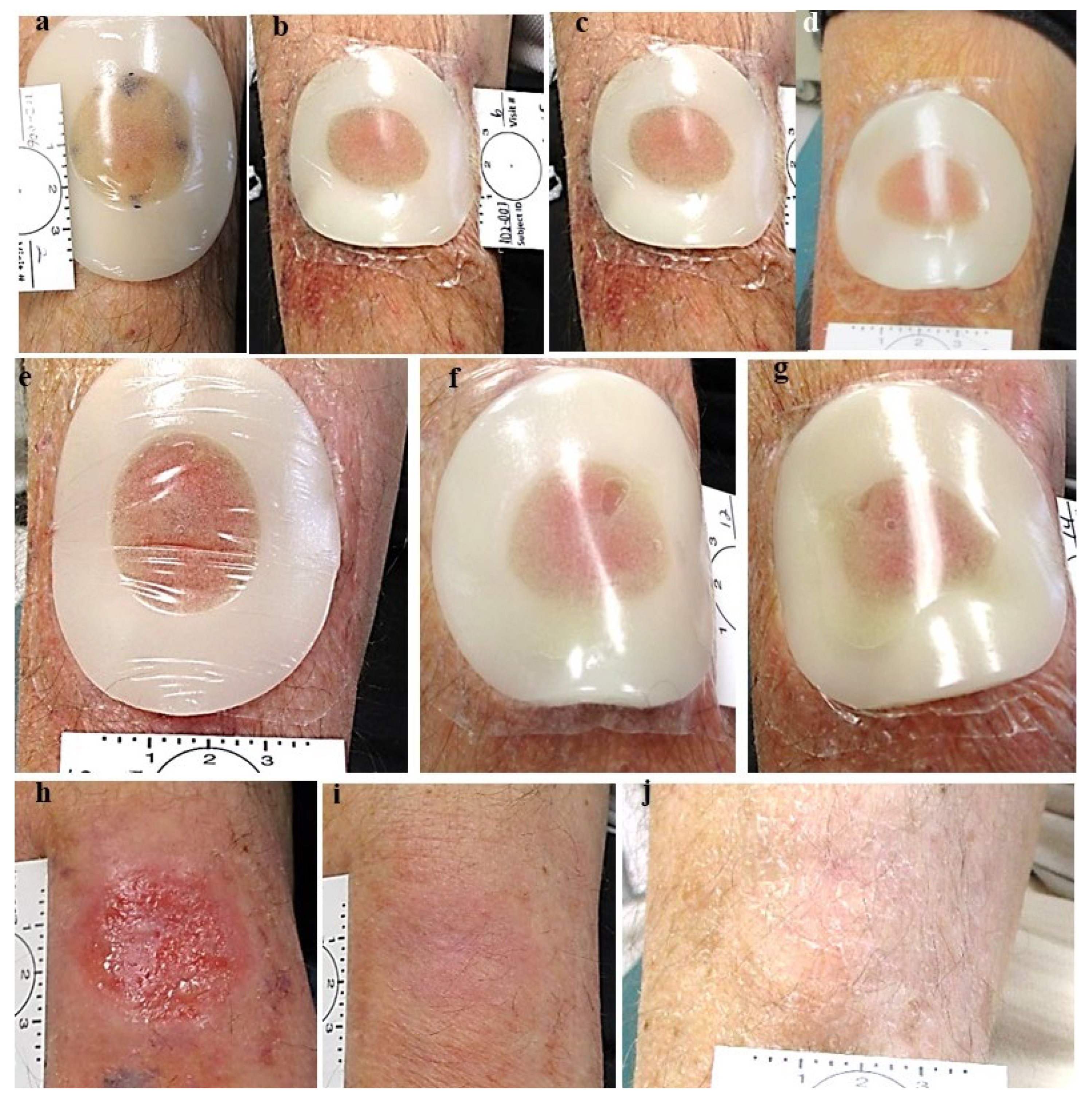
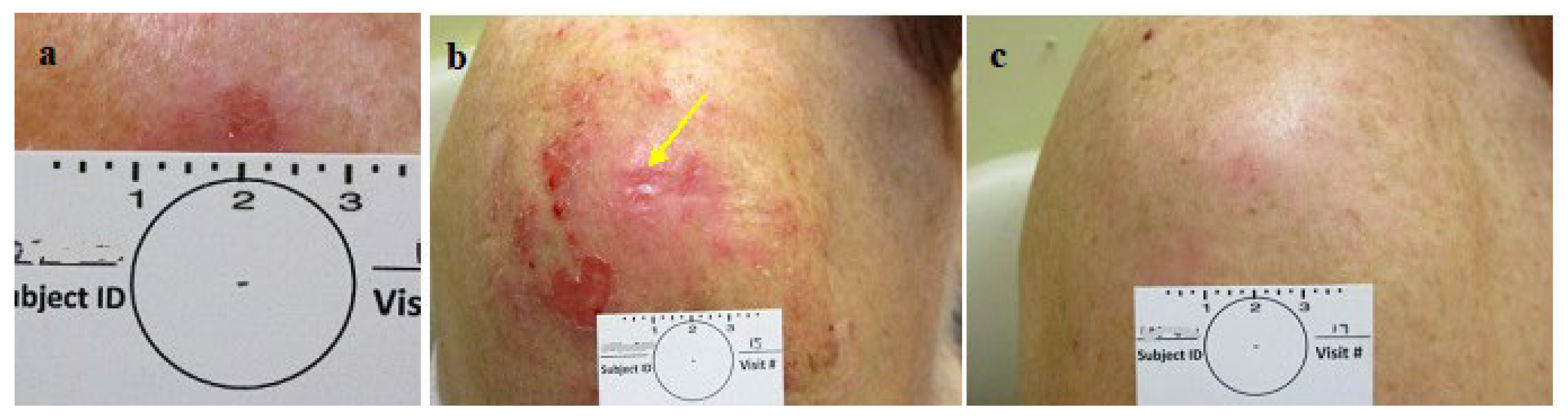
- The final appearance of the treated lesions after 2 months is typical of a fresh, healing wound that easily can be diagnosed as a persistant superficial lesion. Histologically, the inflammation with its atypical cells (which are present at 2 months after treatment) may challenge accurate diagnosis. Thus, the timing for the final, post treatment clearance assessment and biopsy should be done at the end of all healing and inflammatory processes (after 3-6 months).
- Following healing of the CPEEB treated sites, the skin looks intact and scarless, sometimes making the identication of the treated tumor’s exact location for assessment and diagnostic excision challenging. Thus, the tumor and its surrounding should be meticulously mapped initially on enrollment or even temporarily tatooed.
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 5FU | 5-fluorouracil |
| AE | Adverse Event |
| AK | Actinic (Solar) Keratosis |
| API | Active Pharmaceutical Ingredient |
| BCC | Basal Cell Carcinoma |
| CPEEB | Concentrate of Proteolytic Enzymes Enriched in Bromelain |
| DSMB | Data Safety Monitoring Board |
| IRB | Institutional Review Board |
| KC | Keratocyte Carcinoma |
| MBCC | Morphea type BCC |
| MOH | Ministry of Health |
| MOHS | Micrographic Surgery (developed by Fredric E. Mohs 1938) |
| NBCC | Nodular BCC |
| NMSC | Non-Melanoma Skin Cancer |
| PDT | Photo Dynamic Therapy |
| POC | Proof Of Concept |
| SAE | Serious Adverse Event |
| SBCC | Superficial BCC |
| SCC | Squamous Cell Carcinoma |
| TEAE | Treatment-Emergent Adverse Events |
| UV | Ultraviolet |
References
- American Cancer Society https://www.cancer.org/.
- Skin cancer foundation https://www.skincancer.org/.
- Cancer.Net https://www.cancer.net/.
- Marzuka, A.G. and S. E. Book, Basal cell carcinoma: pathogenesis, epidemiology, clinical features, diagnosis, histopathology, and management. Yale J Biol Med 2015, 88, 167–79. [Google Scholar]
- NCCN Guidelines; Cancer Treatment Reviews 64 (2018) 1-10; UpToDate (01/2021); Arch Dermatol. 1999 Oct;132(10):1177-83. J Invest Dermatol. 2017, 137, 614.
- Queirolo, P. Cinquini, M. Argenziano, G. Bassetto, F. Bossi, P. Boutros, A. Clemente, C. de Giorgi, V. Del Vecchio, M. Patuzzo, R. Peris, K. Quaglino, P. Reali, A. Zalaudek, I. and Spagnolo, F. Guidelines for the diagnosis and treatment of basal cell carcinoma: a GRADE approach for evidence evaluation and recommendations by the Italian Association of Medical Oncology, ESMO Open. 2023, 8, 102037. [Google Scholar]
- Product FDA Labels; UpToDate (01/2021); Arch Dermatol. Redbook (01.2021) 1999, 132, 1177–83.
- Bichakjian, *!!! REPLACE !!!*; et al. Guidelines of care for the management of basal cell carcinoma’. J Am Acad Dermatol. 2018, 79, e101. [Google Scholar] [CrossRef]
- Gupta_AK, Paquet_M, Villanueva_E, Brintnell_W. Interventions for actinic keratoses.Cochrane Database of Systematic Reviews 2012, Issue 12. Art. No.: CD004415. [CrossRef]
- B. Borgia F et al. Early and Late Onset Side Effects of Photodynamic Therapy Biomedicines 2018, 6, 12.
- Rosenberg L, et al. Basal Cell Carcinoma Destruction by a Concentrate of Proteolytic Enzymes Enriched in Bromelain: A Preliminary Report. 2021, 15.
- Shoham, Y. , et al. Bromelain-based enzymatic debridement of chronic wounds: A preliminary report. Int Wound J, 2018.
- Rosenberg L, Shoham Y, Krieger Y, et al. Minimally invasive burn care: a review of seven clinical studies of rapid and selective debridement using a bromelain-based debriding enzyme (Nexobrid®). Ann Burns Fire Disasters. 2015, 28, 264–274. [PubMed] [PubMed Central]
- Shoham, Y. , et al. Bromelain-based enzymatic debridement of chronic wounds: A preliminary report. Int Wound J, 2018.
- Rathnavelu, V. , et al. Potential role of bromelain in clinical and therapeutic applications. Biomed Rep. 2016, 5, 283–288. [Google Scholar]
- Chobotova, K., A. B. Vernallis, and F.A. Majid. Bromelain’s activity and potential as an anti-cancer agent: Current evidence and perspectives. Cancer Lett 2010, 290, 148–56. [Google Scholar]
- Chang, T.C. , et al. Bromelain inhibits the ability of colorectal cancer cells to proliferate via activation of ROS production and autophagy. PLoS One 2019, 14, e0210274. [Google Scholar]
- Bhui, K. , et al. Bromelain inhibits COX-2 expression by blocking the activation of MAPK regulated NF-kappa B against skin tumor-initiation triggering mitochondrial death pathway. Cancer Lett 2009, 282, 167–76. [Google Scholar]
- Kalra, N. , et al. Regulation of p53, nuclear factor kappaB and cyclooxygenase-2 expression by bromelain through targeting mitogen-activated protein kinase pathway in mouse skin. Toxicol Appl Pharmacol 2008, 226, 30–7. [Google Scholar]
- Bhui, K. , et al. Bromelain inhibits nuclear factor kappa-B translocation, driving human epidermoid carcinoma A431 and melanoma A375 cells through G(2)/M arrest to apoptosis. Mol Carcinog 2012, 51, 231–43. [Google Scholar]
- Maurer, H. R. Bromelain: Biochemistry, pharmacology and medical use. Cell Mol. Life Sci. 2001, 58, 1234. [Google Scholar] [CrossRef]
- Paroulek A F, J.M. , Rathinavelu A. The effects of the herbal enzyme bromelain against breast cancer cell line GI101A The FASEB Journal 2010.
- Lee, J.-H. The potential use of bromelain as a natural oral medicine having anticarcinogenic activities. Food Sci Nutr 2019, 7, 1656–1667. [Google Scholar] [CrossRef]
- Gaspani, L. , Limiroli, E. , Ferrario, P., and Bianchi, M. In vivo and in vitro effects of bromelain on PGE2 and SP concentrations in the inflammatory exudate in rats. Pharmacology 2002, 65, 83. [Google Scholar]
- Hale, L. P. , Greer, P. K., Trinh, C. T, et al. Treatment with oral bromelain decreases colonic inflammation in the IL-10-deficient murine model of inflammatory bowel disease. Clin. Immunol. 2005, 116, 135. [Google Scholar]
- ER Secor Jr Bromelain exerts anti-inflammatory effects in an ovalbumin-induced murine model of allergic airway disease. Cell Immunol. 2005, 237, 68–75. [CrossRef]
- Pilvi, Riihilä; et al. Matrix metalloproteinases in keratinocyte carcinomas. Experimental Dermatology. 2021, 30, 50–61. [Google Scholar]
- Kavasi, R-M; et al. Matrix Effectors in the Pathogenesis of Keratinocyte-Derived Carcinomas. Front. Med. 2022, 9, 879500. [Google Scholar] [CrossRef]
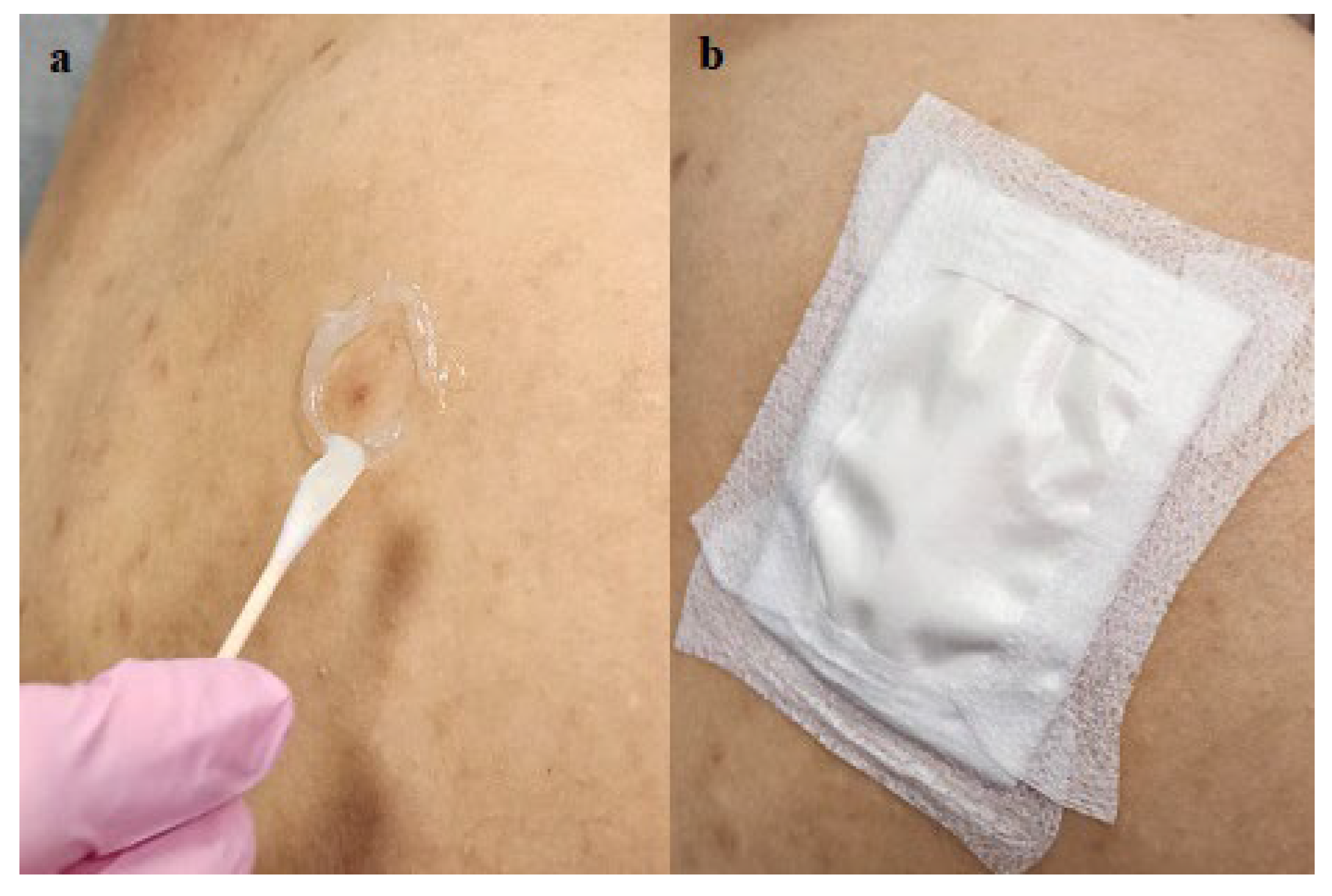
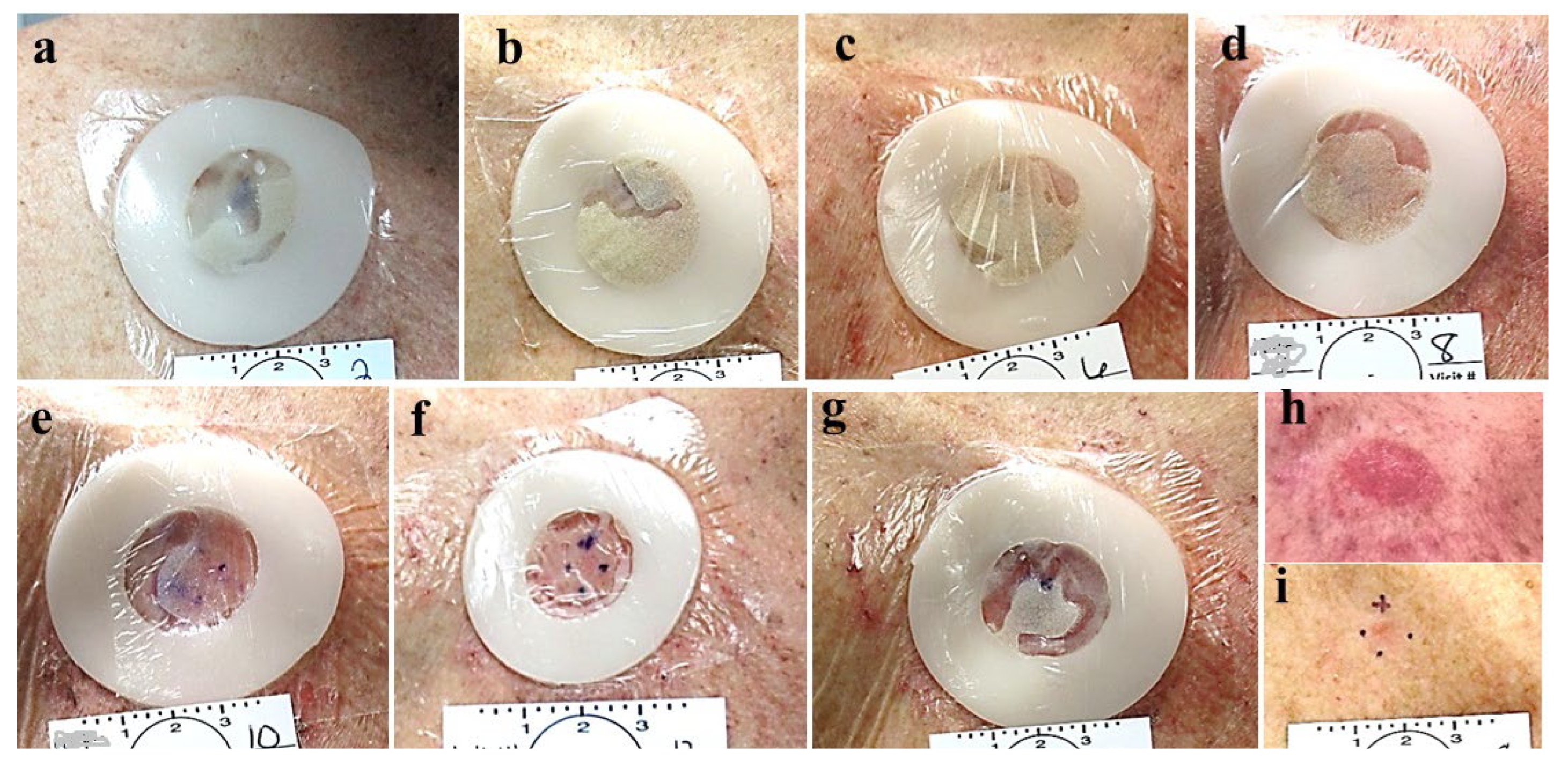
| Group % | Barrier | Occlusive film | Water tight | Leaking dressing | BCCs S/ N/ M | Clinical clearance | Histologic clearance |
|---|---|---|---|---|---|---|---|
| POC 10 % |
Thick ZnOx | x2 Opsite | Y | N | 6 3/ 2/ 1 |
Y all | Y 6/6 |
| 1 5% | Thick Aquaphor | Telfa+ Hypafix | Y | N | 5 2/3 |
Y all | Y 5/5 |
| 2 5% | Thin Aquaphor | Flexigrid+ DuoDerm | N | Y | 6 1/ 5 |
Y 3, N 3 | N 6/6 |
| 3 5% | 1 ring | Tegaderm | N | Y | 4 1/ 3 |
Y 1, N3 | Y 1 N 3 |
| 4 5% | 2 rings | x2 Tegaderm | Y | N | 1 0/ 1 |
Y | Y 1/1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




