Submitted:
24 September 2024
Posted:
25 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Literature Review
2.1. Rechargeable Battery Primer
2.1.1. Basic Operation
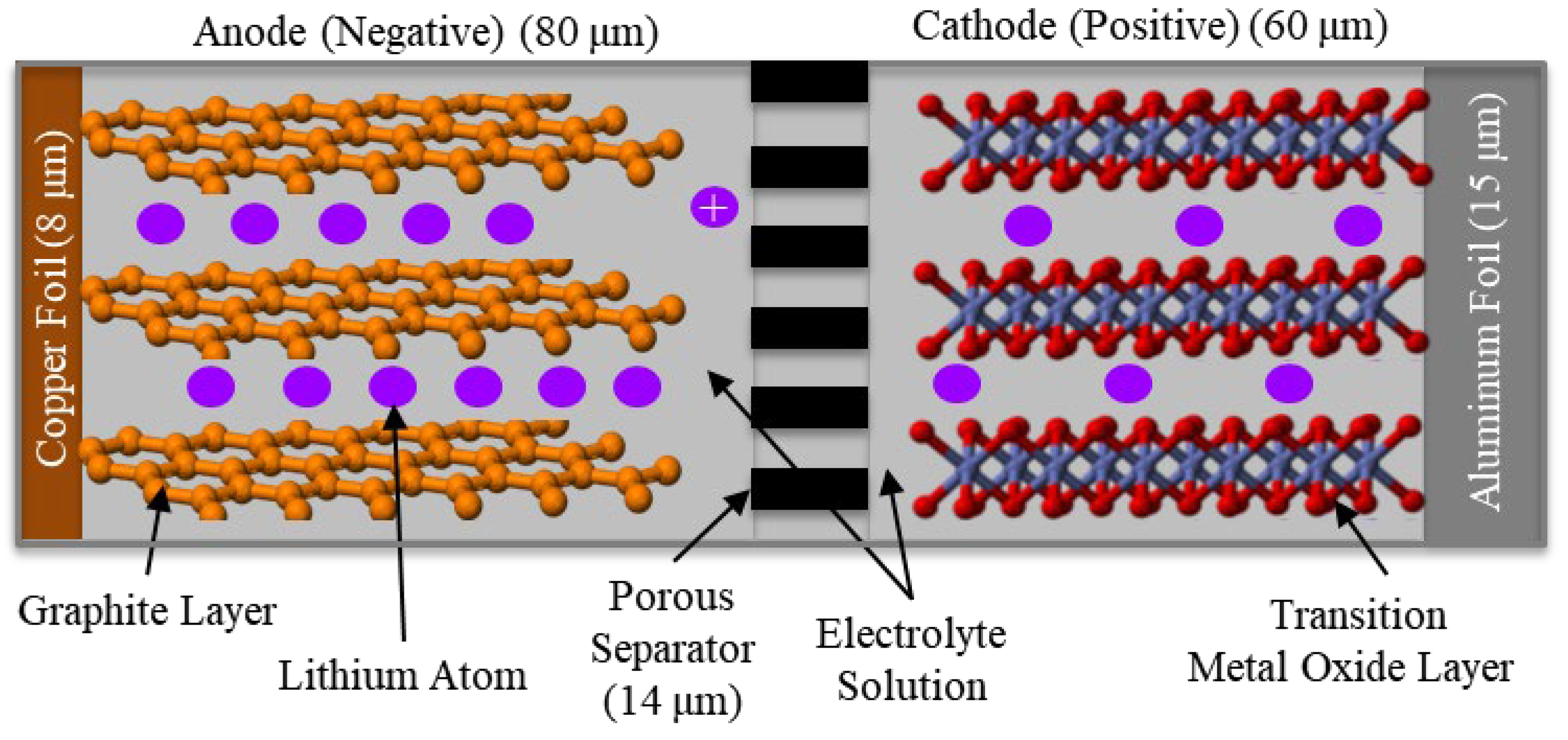
2.1.2. Anode Material
2.1.3. Cathode Material
2.1.4. Electrolyte Material
2.2. Solid State Designs
2.3. Active Research Areas
2.3.1. Electrolyte
2.3.2. Anode
2.3.3. Cathode
2.3.4. Interfacial Integrity
2.3.5. Battery Pack Development
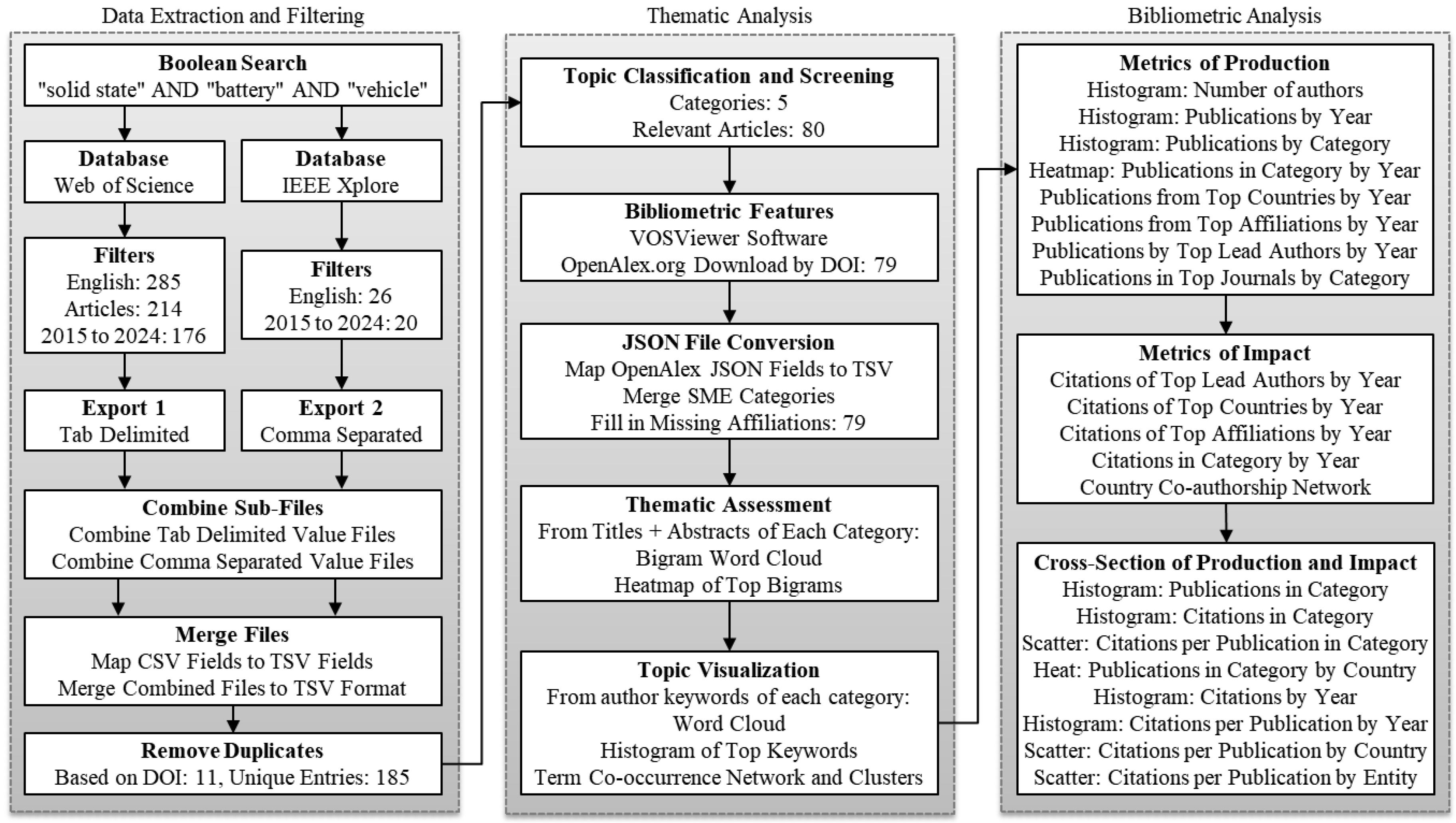
3. Methodology
4. Results
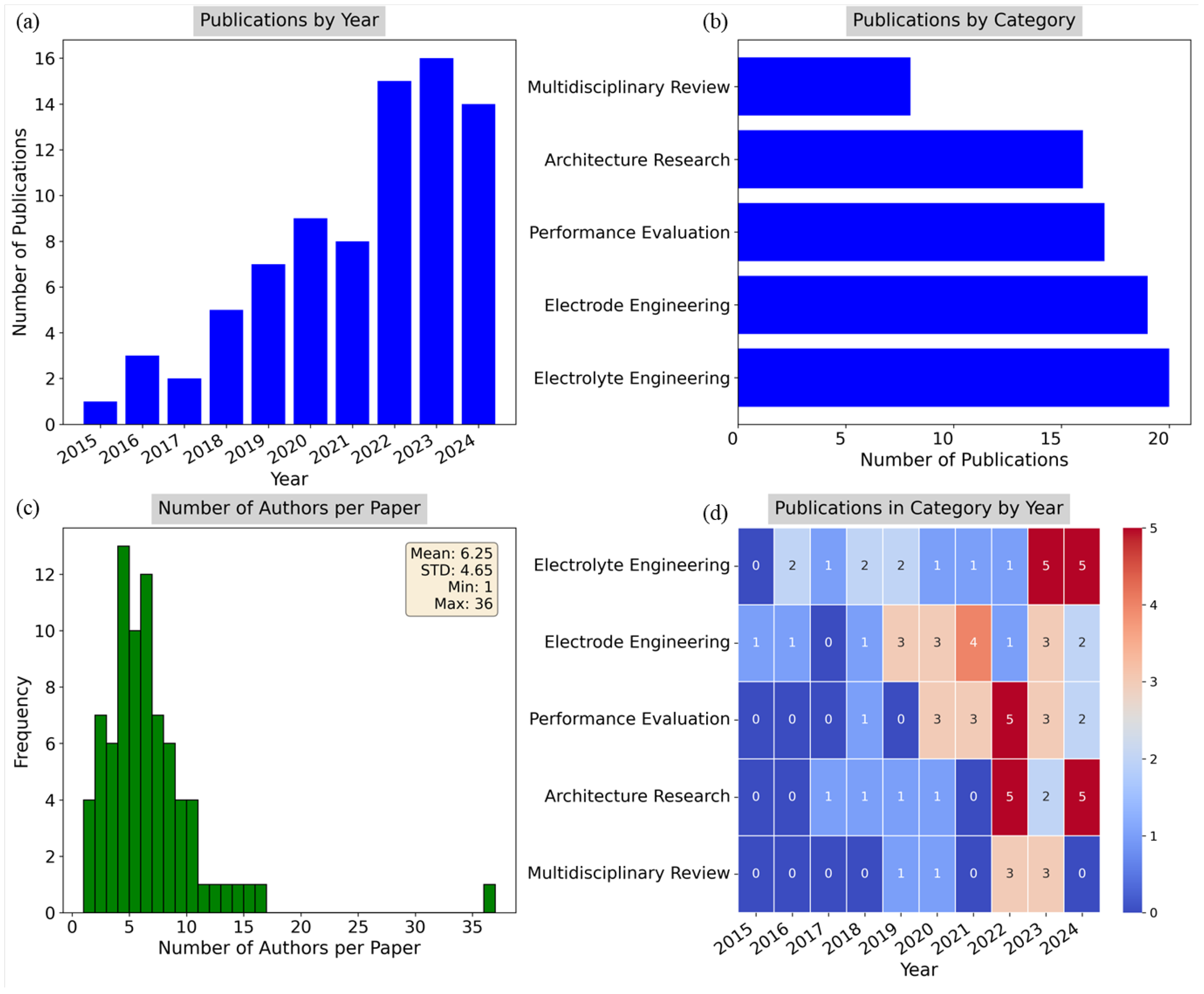
4.1. Thematic Analysis
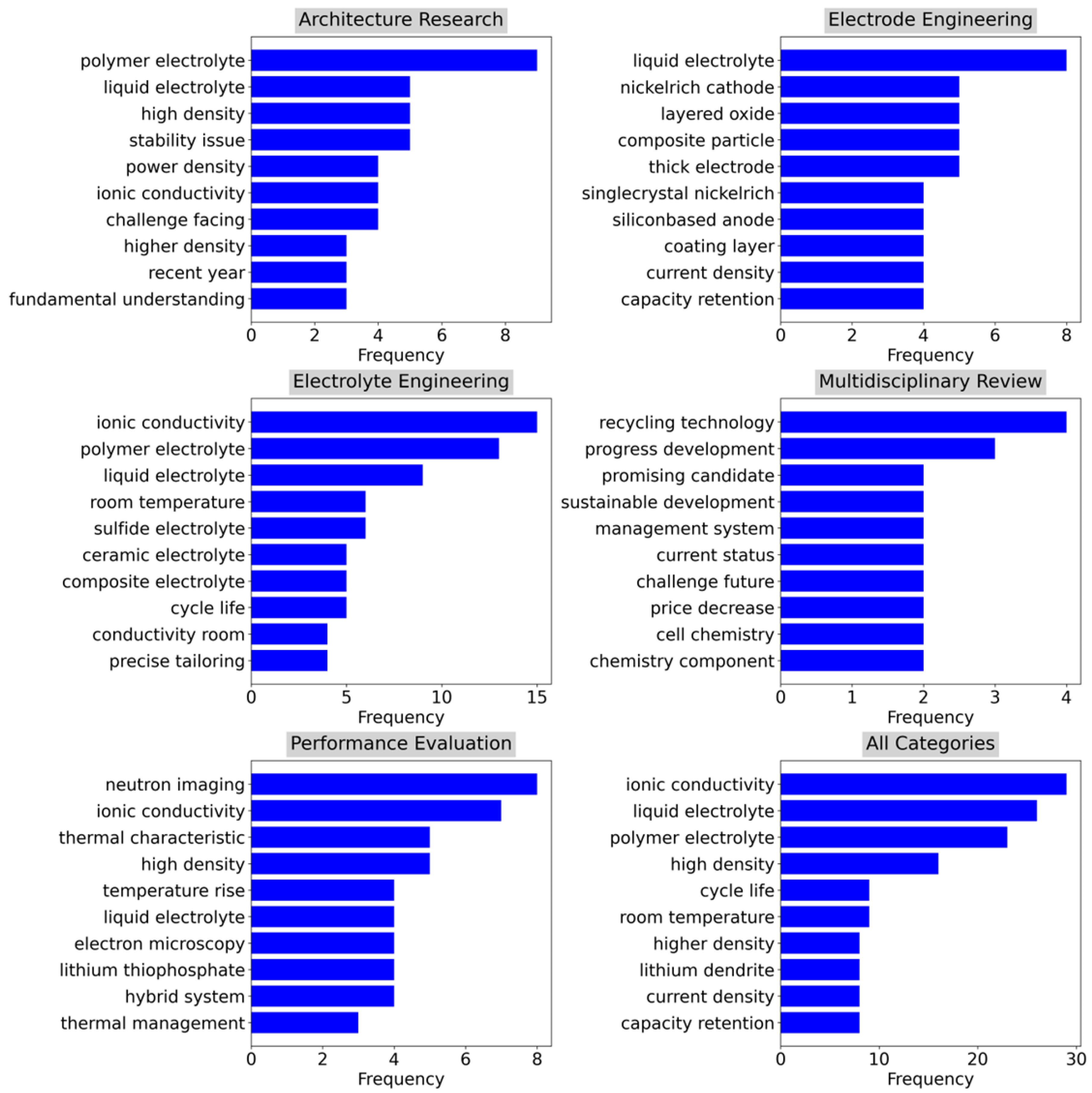

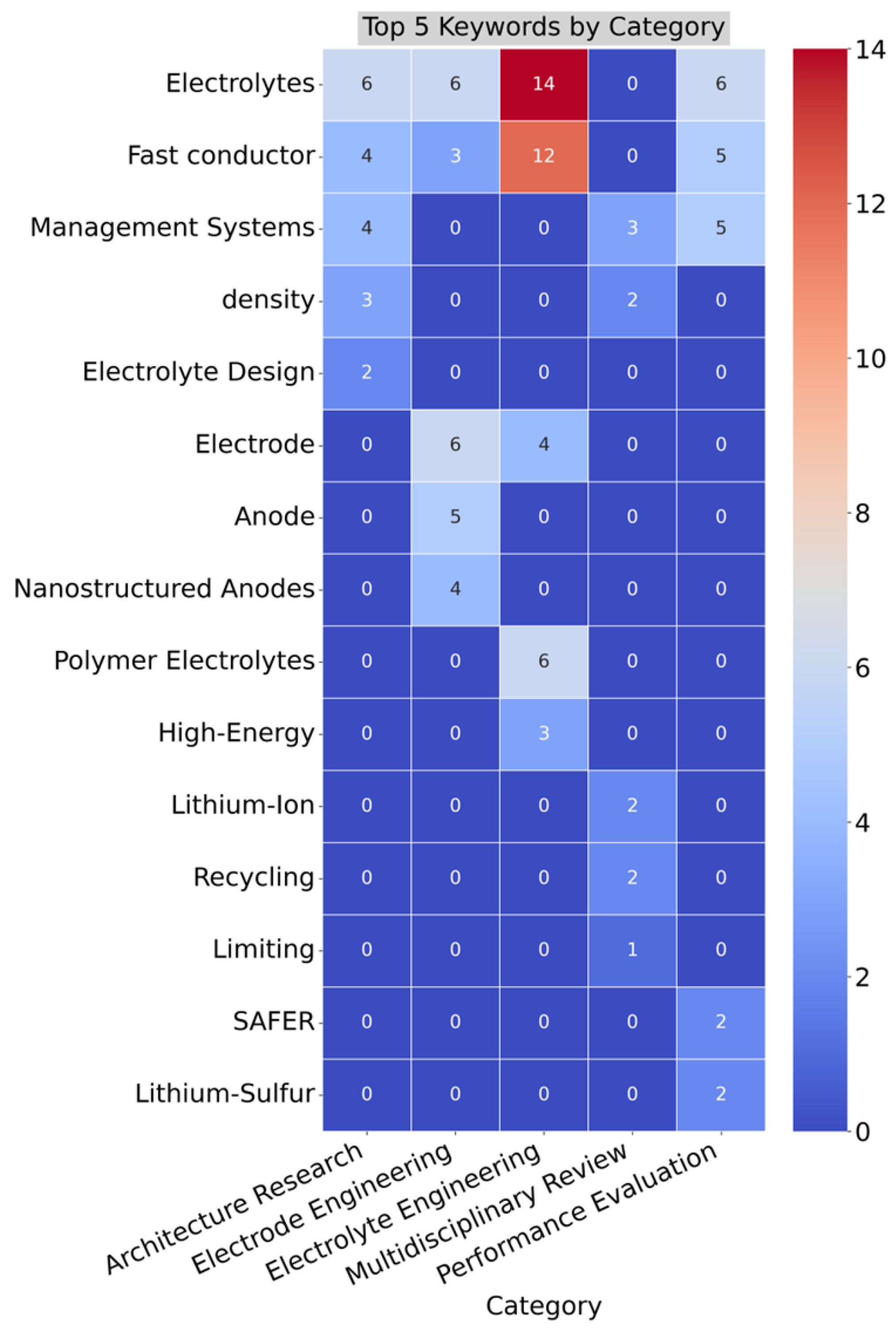
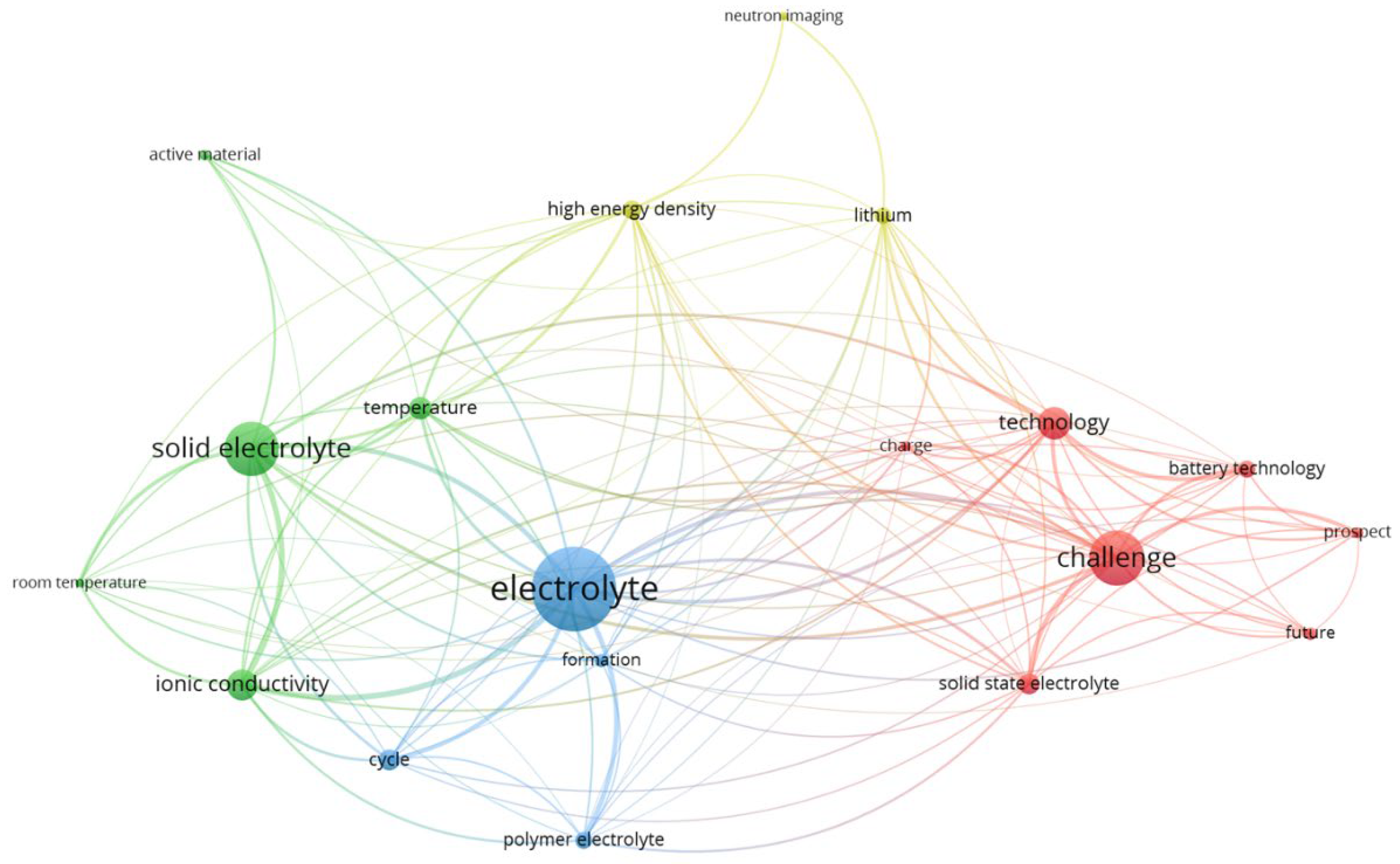
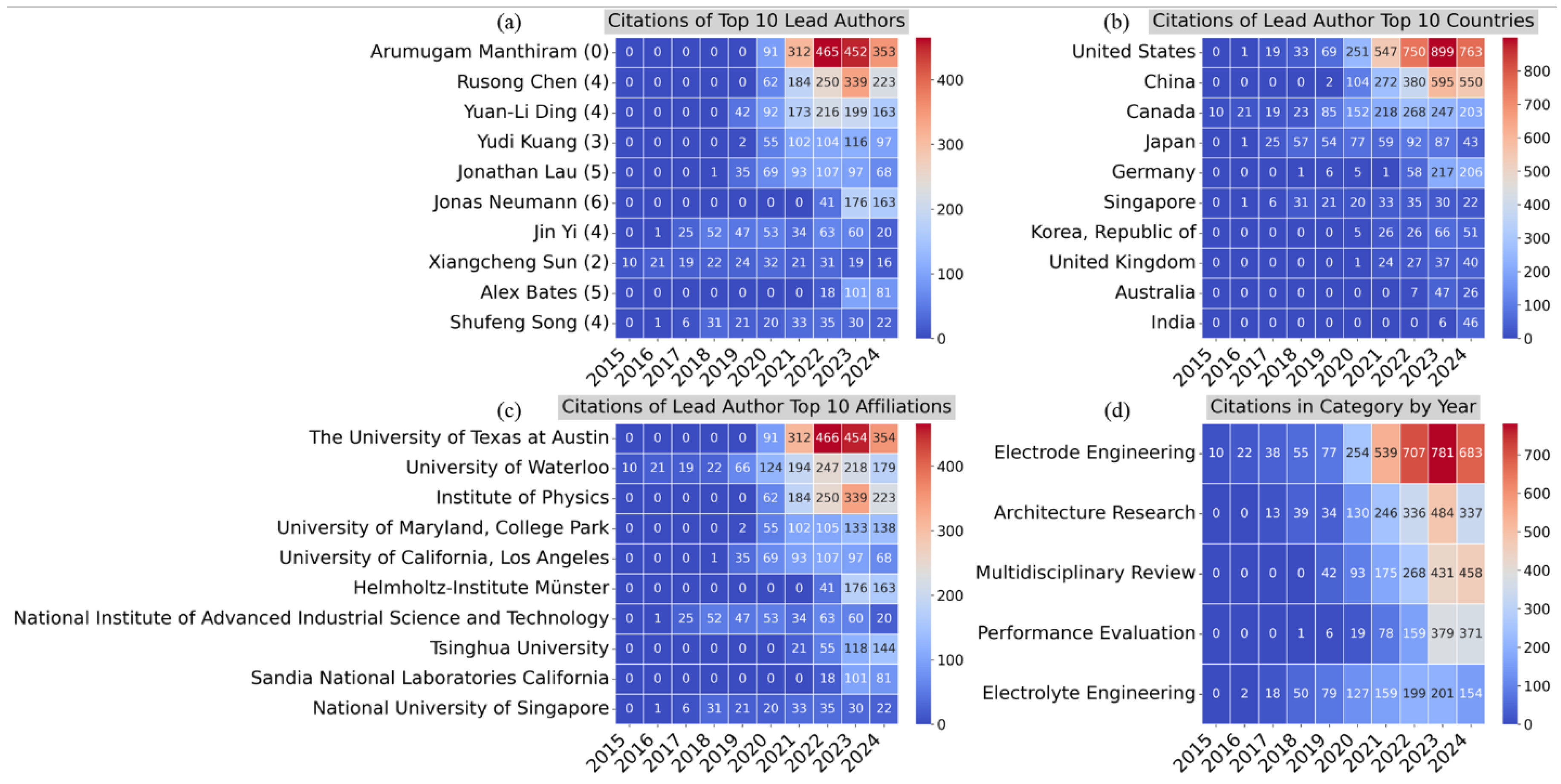
4.2. Bibliometric Analysis
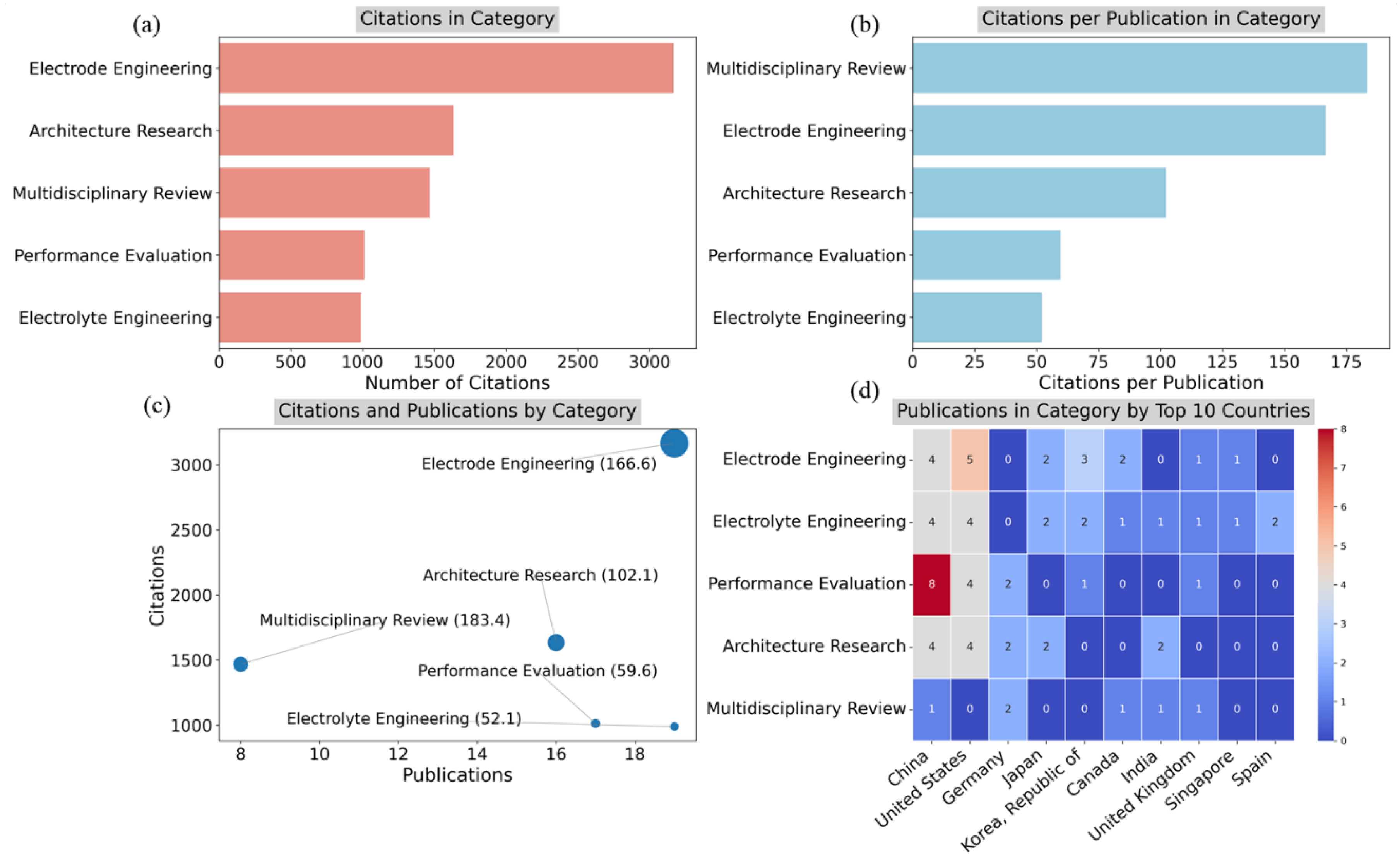
4.3. Specific Developments
4.3.1. Electrolyte Engineering
4.3.2. Electrode Engineering
4.3.3. Performance Evaluation
4.3.4. Multidisciplinary Reviews
5. Discussion
5.1. Electrolyte Engineering
5.2. Electrode Engineering
5.3. Architecture Research
5.4. Performance Evaluation
5.5. Multidisciplinary Review
5.6. Limitations and Future Research
6. Conclusions
Funding
Data Availability Statement
Conflicts of Interest
References
- Bajolle, H.; Lagadic, M.; Louvet, N. The Future of Lithium-Ion Batteries: Exploring Expert Conceptions, Market Trends, and Price Scenarios. Energy Res. Soc. Sci. 2022, 93, 102850. [Google Scholar] [CrossRef]
- Fichtner, M. Recent Research and Progress in Batteries for Electric Vehicles. Batter. Supercaps 2021, 5. [Google Scholar] [CrossRef]
- Yang, R.; Xie, Y.; Li, K.; Tran, M.-K.; Fowler, M.; Panchal, S.; Deng, Z.; Zhang, Y. Comparative Study on the Thermal Characteristics of Solid-State Lithium-Ion Batteries. IEEE Trans. Transp. Electrification 2023, 10, 1541–1557. [Google Scholar] [CrossRef]
- Cao, D.; Zhang, Y.; Ji, T.; Zhu, H. In operando neutron imaging characterizations of all-solid-state batteries. MRS Bull. 2023, 48, 1257–1268. [Google Scholar] [CrossRef]
- Wu, X.; Ji, G.; Wang, J.; Zhou, G.; Liang, Z. Toward Sustainable All Solid-State Li–Metal Batteries: Perspectives on Battery Technology and Recycling Processes. Adv. Mater. 2023, 35, e2301540. [Google Scholar] [CrossRef]
- Razali, A.A.; Norazli, S.N.; Sum, W.S.; Yeo, S.Y.; Dolfi, A.; Srinivasan, G. State-of-the-Art of Solid-State Electrolytes on the Road Map of Solid-State Lithium Metal Batteries for E-Mobility. ACS Sustain. Chem. Eng. 2023, 11, 7927–7964. [Google Scholar] [CrossRef]
- Alexander, G.V.; Shi, C.; O’neill, J.; Wachsman, E.D. Extreme lithium-metal cycling enabled by a mixed ion- and electron-conducting garnet three-dimensional architecture. Nat. Mater. 2023, 22, 1136–1143. [Google Scholar] [CrossRef]
- Hayakawa, E.; Nakamura, H.; Ohsaki, S.; Watano, S. Design of active-material/solid-electrolyte composite particles with conductive additives for all-solid-state lithium-ion batteries. J. Power Sources 2022, 555. [Google Scholar] [CrossRef]
- Lee, J.; Zhao, C.; Wang, C.; Chen, A.; Sun, X.; Amine, K.; Xu, G.-L. Bridging the gap between academic research and industrial development in advanced all-solid-state lithium–sulfur batteries. Chem. Soc. Rev. 2024, 53, 5264–5290. [Google Scholar] [CrossRef]
- Manthiram, A. A reflection on lithium-ion battery cathode chemistry. Nat. Commun. 2020, 11, 1–9. [Google Scholar] [CrossRef]
- Huang, W.; Feng, X.; Han, X.; Zhang, W.; Jiang, F. Questions and Answers Relating to Lithium-Ion Battery Safety Issues. Cell Rep. Phys. Sci. 2021, 2. [Google Scholar] [CrossRef]
- Bridgelall, R. Aircraft Innovation Trends Enabling Advanced Air Mobility. Inventions 2024, 9, 84. [Google Scholar] [CrossRef]
- Wu, B.; Wang, S.; Iv, W.J.E.; Deng, D.Z.; Yang, J.; Xiao, J. Interfacial behaviours between lithium ion conductors and electrode materials in various battery systems. J. Mater. Chem. A 2016, 4, 15266–15280. [Google Scholar] [CrossRef]
- Mills, A.; Kalnaus, S.; Tsai, W.-Y.; Su, Y.-F.; Williams, E.; Zheng, X.; Vaidyanathan, S.; Hallinan, D.T.; Nanda, J.; Yang, G. Elucidating Polymer Binder Entanglement in Freestanding Sulfide Solid-State Electrolyte Membranes. ACS Energy Lett. 2024, 9, 2677–2684. [Google Scholar] [CrossRef]
- York, M.; Larson, K.; Harris, K.C.; Carmona, E.; Albertus, P.; Sharma, R.; Noked, M.; Strauss, E.; Ragones, H.; Golodnitsky, D. Recent advances in solid-state beyond lithium batteries. J. Solid State Electrochem. 2022, 26, 1851–1869. [Google Scholar] [CrossRef]
- Kumar, P.; Channi, H.K.; Babbar, A.; Kumar, R.; Bhutto, J.K.; Khan, T.M.Y.; Bhowmik, A.; Razak, A.; Wodajo, A.W. A Systematic Review of Nanotechnology for Electric Vehicles Battery. Int. J. Low-Carbon Technol. 2024, 19, 747. [Google Scholar] [CrossRef]
- Pimlott, J.L.; Street, R.J.; Down, M.P.; Banks, C.E. Electrochemical Overview: A Summary of ACoxMnyNizO2 and Metal Oxides as Versatile Cathode Materials for Metal-Ion Batteries. Adv. Funct. Mater. 2021, 31, 2107761. [Google Scholar] [CrossRef]
- Kuang, Y.; Chen, C.; Kirsch, D.J.; Hu, L. Thick Electrode Batteries: Principles, Opportunities, and Challenges. Adv. Energy Mater. 2019, 9. [Google Scholar] [CrossRef]
- Liu, H.; Sun, Q.; Zhang, H.; Cheng, J.; Li, Y.; Zeng, Z.; Zhang, S.; Xu, X.; Ji, F.; Li, D.; et al. The application road of silicon-based anode in lithium-ion batteries: From liquid electrolyte to solid-state electrolyte. Energy Storage Mater. 2022, 55, 244–263. [Google Scholar] [CrossRef]
- Mageto, T.; Bhoyate, S.; Souza, F.M.D.; Mensah-Darkwa, K.; Kumar, A.; Gupta, R.K. Developing Practical Solid-State Rechargeable Li-Ion Batteries: Concepts, Challenges, and Improvement Strategies. J. Energy Storage 2022, 55, 105688. [Google Scholar] [CrossRef]
- Hu, Z.; Li, J.; Zhang, X.; Zhu, Y. Strategies to Improve the Performance of Li Metal Anode for Rechargeable Batteries. Front. Chem. 2020, 8, 409. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-J.; Park, J.-W.; Kim, B.G.; Lee, Y.-J.; Ha, Y.-C.; Lee, S.-M.; Baeg, K.-J. Facile fabrication of solution-processed solid-electrolytes for high-energy-density all-solid-state-batteries by enhanced interfacial contact. Sci. Rep. 2020, 10, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Xu, N.; Zhang, Y.; Wang, M.; Fan, X.; Zhang, T.; Peng, L.; Zhou, X.-D.; Qiao, J. High-performing rechargeable/flexible zinc-air batteries by coordinated hierarchical Bi-metallic electrocatalyst and heterostructure anion exchange membrane. Nano Energy 2019, 65, 104021. [Google Scholar] [CrossRef]
- Chen, Z.; Wang, K.; Pei, P.; Zuo, Y.; Wei, M.; Wang, H.; Zhang, P.; Shang, N. Advances in electrolyte safety and stability of ion batteries under extreme conditions. Nano Res. 2022, 16, 2311–2324. [Google Scholar] [CrossRef]
- Yu, D.; Min, J.; Lin, F.; Madsen, L.A. Mechanically and Thermally Robust Gel Electrolytes Built from A Charged Double Helical Polymer. Adv. Mater. 2024, 36, e2312513. [Google Scholar] [CrossRef]
- Shen, H.; Yi, E.; Cheng, L.; Amores, M.; Chen, G.; Sofie, S.W.; Doeff, M.M. Solid-state electrolyte considerations for electric vehicle batteries. Sustain. Energy Fuels 2019, 3, 1647–1659. [Google Scholar] [CrossRef]
- Lee, J.-H.; Lee, H.; Lee, J.W.; Kang, T.W.; Park, J.H.; Shin, J.-H.; Lee, H.; Majhi, D.; Lee, S.U.; Kim, J.-H. Multicomponent Covalent Organic Framework Solid Electrolyte Allowing Effective Li-Ion Dissociation and Diffusion for All-Solid-State Batteries. ACS Nano 2023, 17, 17372. [Google Scholar] [CrossRef]
- Singh, D.K.; Fuchs, T.; Krempaszky, C.; Mogwitz, B.; Janek, J. Non-Linear Kinetics of The Lithium Metal Anode on Li6PS5Cl at High Current Density: Dendrite Growth and the Role of Lithium Microstructure on Creep. Adv. Sci. 2023, 10, e2302521. [Google Scholar] [CrossRef]
- Sharma, V.; Singh, K.; Narayanan, K. A Review on Conventional to Bipolar Design of Anode Less All Solid-State Batteries. Energy Adv. 2024, 3, 1222. [Google Scholar] [CrossRef]
- Li, W.; Quirk, J.A.; Li, M.; Xia, W.; Morgan, L.M.; Yin, W.; Zheng, M.; Gallington, L.C.; Ren, Y.; Zhu, N.; King, G.; Feng, R.; Li, R.; Dawson, J.A.; Sham, T.-K.; Sun, X. Precise Tailoring of Lithium-Ion Transport for Ultralong-Cycling Dendrite-Free All-Solid-State Lithium Metal Batteries. Adv. Mater. 2023, 36. [Google Scholar] [CrossRef]
- Choi, H.J.; Kang, D.W.; Park, J.-W.; Park, J.-H.; Lee, Y.; Ha, Y.-C.; Lee, S.-M.; Yoon, S.Y.; Kim, B.G. In Situ Formed Ag-Li Intermetallic Layer for Stable Cycling of All-Solid-State Lithium Batteries. Adv. Sci. 2021, 9. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Wu, N. Ionic conductivity and ion transport mechanisms of solid-state lithium-ion battery electrolytes: A review. Energy Sci. Eng. 2022, 10, 1643–1671. [Google Scholar] [CrossRef]
- Chen, R.; Li, Q.; Yu, X.; Chen, L.; Li, H. Approaching Practically Accessible Solid-State Batteries: Stability Issues Related to Solid Electrolytes and Interfaces. Chem. Rev. 2019, 120, 6820–6877. [Google Scholar] [CrossRef] [PubMed]
- Swathi, P.; V., S.O.; Panneerselvam, T.; Murugan, R.; Ramaswamy, A.P. Cross-Linked Polymer Composite Electrolyte Incorporated with Waste Seashell Based Nanofiller for Lithium Metal Batteries. Energy Fuels 2023, 37, 6186–6196. [Google Scholar] [CrossRef]
- Zhao, G.; Wang, X.; Negnevitsky, M. Connecting battery technologies for electric vehicles from battery materials to management. iScience 2022, 25, 103744. [Google Scholar] [CrossRef]
- Ramasubramanian, B.; Dalapati, G.K.; Reddy, M.V.V.; Zaghib, K.; Chellappan, V.; Ramakrishna, S. Progress and Complexities in Metal–Air Battery Technology. Energy Technol. 2024. [Google Scholar] [CrossRef]
- Huang, C.; Zheng, H.; Qin, N.; Wang, C.; Wang, L.; Lu, J. Single-Crystal Nickel-Rich Cathode Materials: Challenges and Strategies. Acta Phys. Chim. Sin. 2023, 40. [Google Scholar] [CrossRef]
- Ma, Y.; Shang, R.; Liu, Y.; Lake, R.; Ozkan, M.; Ozkan, C.S. Enabling fast-charging capability for all-solid-state lithium-ion batteries. J. Power Sources 2023, 559. [Google Scholar] [CrossRef]
- Wang, C.; Zhao, Y.; Sun, Q.; Li, X.; Liu, Y.; Liang, J.; Li, X.; Lin, X.; Li, R.; Adair, K.R.; et al. Stabilizing interface between Li10SnP2S12 and Li metal by molecular layer deposition. Nano Energy 2018, 53, 168–174. [Google Scholar] [CrossRef]
- Wang, Y.; Ni, R.; Jiang, X.; Yin, M.; Zhang, D.; Xie, Z. An electrochemical-mechanical coupled multi-scale modeling method and full-field stress distribution of lithium-ion battery. Appl. Energy 2023, 347. [Google Scholar] [CrossRef]
- Yang, R.; Xie, Y.; Li, K.; Li, W.; Hu, X.; Fan, Y.; Zhang, Y. Thermal characteristics of solid-state battery and its thermal management system based on flat heat pipe. Appl. Therm. Eng. 2024, 252. [Google Scholar] [CrossRef]
- Waseem, M.; Ahmad, M.; Parveen, A.; Suhaib, M. Battery Technologies and Functionality of Battery Management System for EVs: Current Status, Key Challenges, and Future Prospectives. J. Power Sources 2023, 580, 233349. [Google Scholar] [CrossRef]
- Iyer, V.; Petersen, J.; Geier, S.; Wierach, P. Development and Multifunctional Characterization of a Structural Sodium-Ion Battery Using a High-Tensile-Strength Poly(ethylene oxide)-Based Matrix Composite. ACS Appl. Energy Mater. 2024, 7, 3968–3982. [Google Scholar] [CrossRef]
- Bridgelall, R.; Corcoran, M. Conformal Body Capacitors Suitable for Vehicles. United States of America Patent 9,966,790, 8 May 2018.
- N. J. v. Eck and L. Waltman, "VOSviewer," Leiden University, 1 July 2024. [Online]. Available: vosviewer.com. [Accessed 30 June 2024].
- Pasta, M.; Armstrong, D.; Brown, Z.L.; Bu, J.; Castell, M.R.; Chen, P.; Cocks, A.; Corr, S.A.; Cussen, E.J.; Darnbrough, E.; et al. 2020 roadmap on solid-state batteries. J. Phys. Energy 2020, 2, 032008. [Google Scholar] [CrossRef]
- Choi, Y.J.; Hwang, Y.J.; Kim, S.-I.; Kim, T. Performance Improvement of Argyrodite Solid Electrolyte for All-Solid-State Battery Using Wet Process. ACS Appl. Energy Mater. 2024, 7, 4421–4428. [Google Scholar] [CrossRef]
- Zhu, Q.-C.; Ma, J.; Huang, J.-H.; Mao, D.-Y.; Wang, K.-X. Realizing long-cycling solid-state Li–CO2 batteries using Zn-doped LATP ceramic electrolytes. Chem. Eng. J. 2024, 482. [Google Scholar] [CrossRef]
- Thomas, C.M.; Zeng, D.; Huang, H.C.; Pham, T.; Torres-Castanedo, C.G.; Bedzyk, M.J.; Dravid, V.P.; Hersam, M.C. Earth-Abundant Kaolinite Nanoplatelet Gel Electrolytes for Solid-State Lithium Metal Batteries. ACS Appl. Mater. Interfaces 2024, 16, 34913–34922. [Google Scholar] [CrossRef]
- Zhang, H.; Shi, X.; Zeng, Z.; Zhang, Y.; Du, Y. Fast-charging batteries based on dual-halogen solid-state electrolytes. Mater. Chem. Front. 2023, 7, 4961–4970. [Google Scholar] [CrossRef]
- Ha, Y.-C. A Review on the Wet Chemical Synthesis of Sulfide Solid Electrolytes for All-Solid-State Li Batteries. J. Korean Electrochem. Soc. 2022, 25, 95–104. [Google Scholar]
- Martinez, A.I.P.; Aguesse, F.; Otaegui, L.; Schneider, M.; Roters, A.; Llordés, A.; Buannic, L. The Cathode Composition, A Key Player in the Success of Li-Metal Solid-State Batteries. J. Phys. Chem. C 2019, 123, 3270–3278. [Google Scholar] [CrossRef]
- Lau, J.; DeBlock, R.H.; Butts, D.M.; Ashby, D.S.; Choi, C.S.; Dunn, B.S. Sulfide Solid Electrolytes for Lithium Battery Applications. Adv. Energy Mater. 2018, 8. [Google Scholar] [CrossRef]
- Yi, J.; Liu, Y.; Qiao, Y.; He, P.; Zhou, H. Boosting the Cycle Life of Li-O2 Batteries At Elevated Temperature By Employing A Hybrid Polymer-Ceramic Solid Electrolyte. ACS Energy Lett. 2017, 2, 1378. [Google Scholar] [CrossRef]
- Song, S.; Duong, H.M.; Korsunsky, A.M.; Hu, N.; Lu, L. A Na+ Superionic Conductor for Room-Temperature Sodium Batteries. Sci. Rep. 2016, 6, 32330. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.K.; Wada, Y.; Hayashi, K.; Kawamura, G.; Muto, H.; Matsuda, A. Fabrication of An All-Solid-State Zn-Air Battery using Electroplated Zn on Carbon Paper and KOH-ZrO2 Solid Electrolyte. Appl. Surf. Sci. 2019, 487, 343. [Google Scholar] [CrossRef]
- Li, J.; Yang, M.; Huang, Z.; Zhao, B.; Zhang, G.; Li, S.; Cui, Y.; Dong, Z.; Liu, H. Nanoscale Operation of Ni-Rich Cathode Surface By Polycrystalline Solid Electrolytes Li3.2Zr0.4Si0.6O3.6 Coating. Chem. Eng. J. 2021, 417, 129217. [Google Scholar] [CrossRef]
- Wu, Y.; Liu, X.; Wang, L.; Feng, X.; Ren, D.; Li, Y.; Rui, X.; Wang, Y.; Han, X.; Xu, G.-L.; et al. Development of cathode-electrolyte-interphase for safer lithium batteries. Energy Storage Mater. 2021, 37, 77–86. [Google Scholar] [CrossRef]
- Lee, Y.-G.; Fujiki, S.; Jung, C.; Suzuki, N.; Yashiro, N.; Omoda, R.; Ko, D.-S.; Shiratsuchi, T.; Sugimoto, T.; Ryu, S.; et al. High-energy long-cycling all-solid-state lithium metal batteries enabled by silver–carbon composite anodes. Nat. Energy 2020, 5, 299–308. [Google Scholar] [CrossRef]
- Xu, B.; Li, Y. Designing electrolyte additives for lithium metal batteries through multi-factor principle. J. Energy Chem. 2021, 61, 147–148. [Google Scholar] [CrossRef]
- Keles, O.; Karahan, B.D.; Eryilmaz, L.; Amine, R.; Abouimrane, A.; Chen, Z.; Zuo, X.; Zhu, Z.; Al-Hallaj, S.; Amine, K. Superlattice-structured films by magnetron sputtering as new era electrodes for advanced lithium-ion batteries. Nano Energy 2020, 76, 105094. [Google Scholar] [CrossRef]
- Park, S.W.; Oh, G.; Park, J.; Ha, Y.; Lee, S.; Yoon, S.Y.; Kim, B.G. Graphitic Hollow Nanocarbon as a Promising Conducting Agent for Solid-State Lithium Batteries. Small 2019, 15, e1900235. [Google Scholar] [CrossRef]
- Sun, X.; Radovanovic, P.V.; Cui, B. Advances in Spinel Li4Ti5O12 Anode Materials for Lithium-Ion Batteries. New J. Chem. 2015, 39, 38. [Google Scholar] [CrossRef]
- Kazyak, E.; García-Méndez, R. Recent progress and challenges for manufacturing and operating solid-state batteries for electric vehicles. MRS Bull. 2024, 49, 717–729. [Google Scholar] [CrossRef]
- Chaudhary, R.; Xu, J.; Xia, Z.; Asp, L.E. Unveiling the Multifunctional Carbon Fiber Structural Battery. Adv. Mater. 2024, e2409725. [Google Scholar] [CrossRef] [PubMed]
- Whang, G.; Zeier, W.G. Transition Metal Sulfide Conversion: A Promising Approach to Solid-State Batteries. ACS Energy Lett. 2023, 8, 5264–5274. [Google Scholar] [CrossRef]
- Li, Z.; Lu, Y.; Su, Q.; Wu, M.; Que, X.; Liu, H. High-Power Bipolar Solid-State Batteries Enabled by In-Situ-Formed Ionogels for Vehicle Applications. ACS Appl. Mater. Interfaces 2022, 14, 5402–5413. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Chen, Y.; Yu, X.; Zhou, L. Fiber metal laminated structural batteries with multifunctional solid polymer electrolytes. Compos. Sci. Technol. 2022, 230. [Google Scholar] [CrossRef]
- Yamamoto, M.; Terauchi, Y.; Sakuda, A.; Takahashi, M. Binder-free sheet-type all-solid-state batteries with enhanced rate capabilities and high energy densities. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef]
- Yoon, K.; Kim, H.; Han, S.; Chan, T.-S.; Ko, K.-H.; Jo, S.; Park, J.; Kim, S.; Lee, S.; Noh, J.; et al. Detrimental effect of high-temperature storage on sulfide-based all-solid-state batteries. Appl. Phys. Rev. 2022, 9, 031403. [Google Scholar] [CrossRef]
- Guo, B.; Chen, J.; Wang, Z.; Su, Y.; Li, H.; Ye, H.; Zhang, L.; Tang, Y.; Huang, J. In situ TEM studies of electrochemistry of high temperature lithium-selenium all-solid-state batteries. Electrochimica Acta 2021, 404, 139773. [Google Scholar] [CrossRef]
- Zhang, J.; Zheng, C.; Li, L.; Xia, Y.; Huang, H.; Gan, Y.; Liang, C.; He, X.; Tao, X.; Zhang, W. Unraveling the Intra and Intercycle Interfacial Evolution of Li6PS5Cl-Based All-Solid-State Lithium Batteries. Adv. Energy Mater. 2019, 10. [Google Scholar] [CrossRef]
- Gutiérrez-Pardo, A.; Martinez, A.I.P.; Otaegui, L.; Schneider, M.; Roters, A.; Llordés, A.; Aguesse, F.; Buannic, L. Will the competitive future of solid state Li metal batteries rely on a ceramic or a composite electrolyte? Sustain. Energy Fuels 2018, 2, 2325–2334. [Google Scholar] [CrossRef]
- Boretti, A. Battery energy storage in electric vehicles by 2030. Energy Storage 2022, 5. [Google Scholar] [CrossRef]
- Neumann, J.; Petranikova, M.; Meeus, M.; Gamarra, J.D.; Younesi, R.; Winter, M.; Nowak, S. Recycling of Lithium-Ion Batteries—Current State of the Art, Circular Economy, and Next Generation Recycling. Adv. Energy Mater. 2022, 12. [Google Scholar] [CrossRef]
- Ball, S.; Clark, J.; Cookson, J. Battery Materials Technology Trends and Market Drivers for Automotive Applications. Johns. Matthey Technol. Rev. 2020, 64, 287–297. [Google Scholar] [CrossRef]
- Ding, Y.; Cano, Z.P.; Yu, A.; Lu, J.; Chen, Z. Automotive Li-Ion Batteries: Current Status and Future Perspectives. Electrochem. Energy Rev. 2019, 2, 1–28. [Google Scholar] [CrossRef]


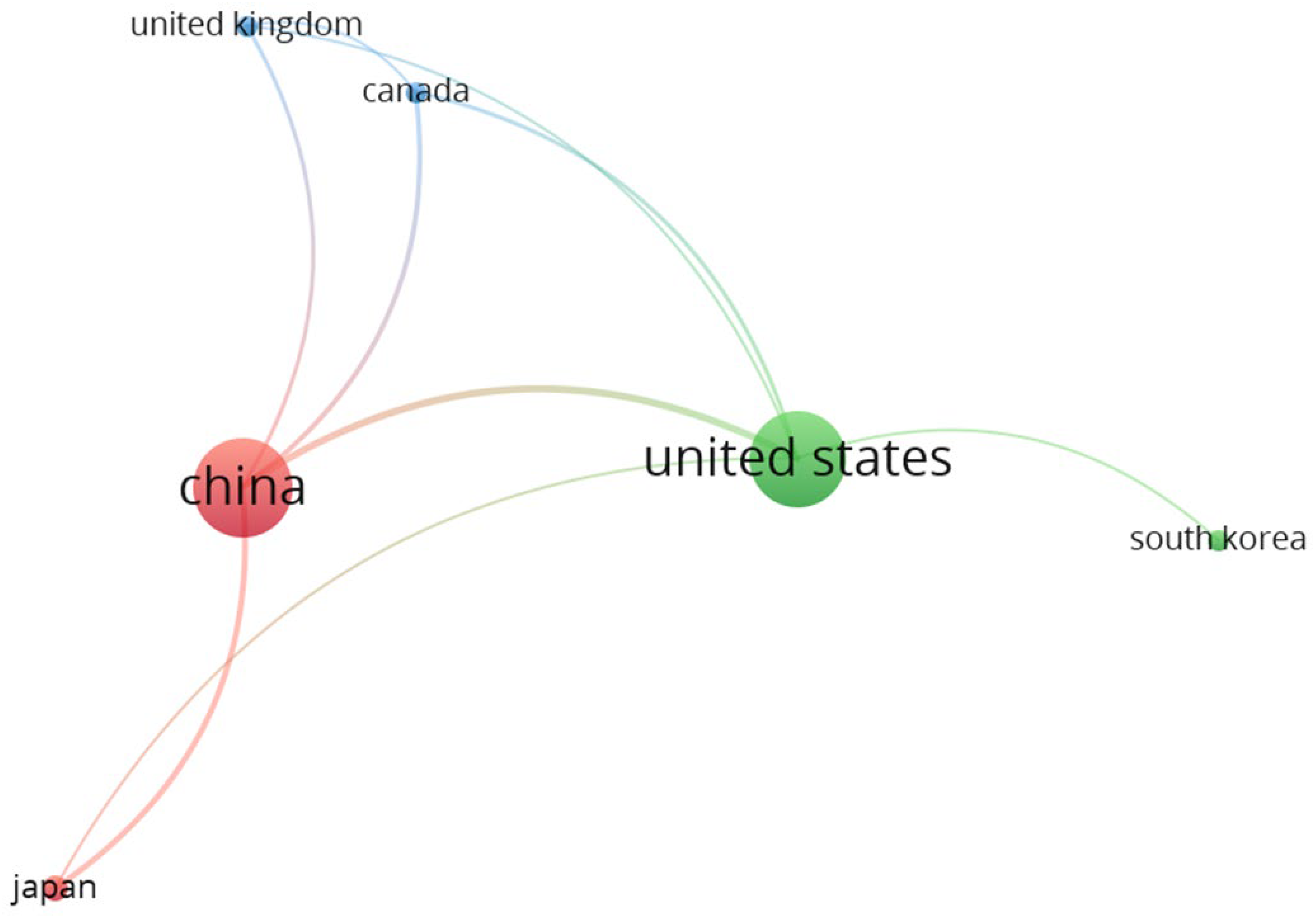
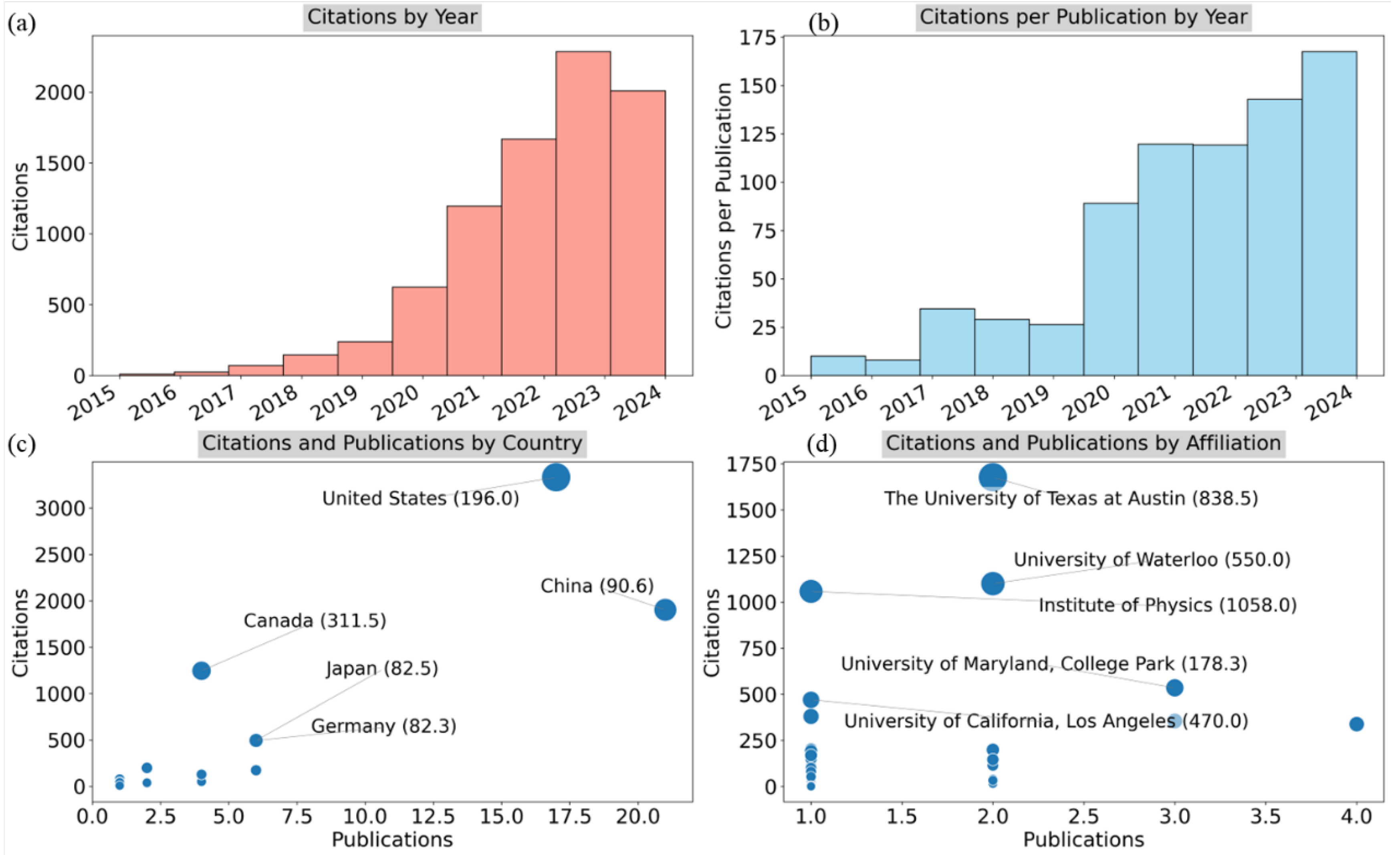
| Country | Documents | Citations | TLS |
|---|---|---|---|
| China | 27 | 2,617 | 15 |
| United States | 26 | 4,561 | 11 |
| Canada | 6 | 1,282 | 6 |
| Japan | 7 | 534 | 5 |
| United Kingdom | 6 | 333 | 4 |
| South Korea | 6 | 177 | 1 |
| Source | Key findings and/or contributions |
|---|---|
| Choi et al. (2024) | Synthesized solid electrolyte by separating the precipitate and removing the supernatant after high-energy ball milling to increase the ionic conductivity and compatibility with lithium metal at room temperature [47]. |
| Zhu et al. (2024) | Demonstrated that doping a lithium-aluminum-titanium-phosphate (LATP) solid-state ceramic electrolyte with zinc strengthened its mechanical properties while providing an optimized network channel for lithium-ion transport at room temperature [48]. They achieved 180 cycles of battery life. |
| Thomas et al. (2024) | Proposed creating a nanocomposite electrolyte from kaolinite, an earth-abundant clay variety, by overcoming strong interlayer forces that have historically precluded their efficient exfoliation (separation) into thin sheets [49]. They used a special liquid process to break apart the kaolinite into very thin, tiny pieces (nanoplatelets) and combined those with a succinonitrile liquid electrolyte to form a nanocomposite gel electrolyte. They achieved a desired ionic conductivity at room temperature, high mechanical strength, a wide electrochemical stability window of 4.5 volts, and thermal stability beyond 100 degrees Celsius. |
| Yu et al. (2024) | Developed a gel polymer electrolyte by mixing lithium salt and poly (ethylene glycol) dimethyl ether [25]. This resulted in tiny fibers formed from a rigid double helical structure. Combined with a lithium anode and a LiFePO4 cathode, the battery cell exhibited strong mechanical properties, a wide operating temperature range up to 120 degrees Celsius, high charge cycles, and high-capacity retention. |
| Li et al. (2023) | Demonstrated the use of a halide solid electrolyte (Li3InCl6) to precisely tailor the movement of lithium-ion both within and between the grains of this material [30]. Their process led to the formation of unexpected supersonic conducting grain boundaries that lowered the energy required for ion hopping and suppressed lithium dendrite growth. They achieved 2,000 cycles of battery life and 93.7% capacity retention. |
| Lee et al. (2023) | Developed an organic solid electrolyte based on a diethylene glycol-modified pyridinium covalent organic framework that exhibited high ion conductivity (1.71×10-4 S/cm) at room temperature, 99% coulombic efficiency, and effective suppression of lithium dendrite formation [27]. |
| Swathi et al. (2023) | Incorporated a nanofiller made with calcium hydroxide derived from natural seashells into a polymer electrolyte (PEO + LiClO4 salt), yielding improved ionic conductivity of 4.12×10-5 S/cm at 25 degrees Celsius [34]. |
| Zhang et al. (2023) | Developed a dual-halogen solid-state electrolyte (Li3YCl6) that increased ionic conductivity by more than one order of magnitude and retained capacity at 80% after 1,000 cycles at 4 degrees Celsius [50]. |
| Ha et al. (2022) | Reviewed the literature on wet chemical synthesis of sulfide solid electrolytes, summarizing the reaction mechanism between the raw materials in the solvent [51]. |
| Martinez et al. (2019) | Created a hybrid solid electrolyte by combining both polymer and ceramic materials to improve ionic conductivity and mechanical stability [52]. Blending the solid electrolyte with the cathode material (LiFePO4) at 30% to 40% by volume yielded good interfacial contact between the components and efficient ionic transport throughout the cell. They calculated theoretical gravimetric and volumetric energy densities of 185 Wh/kg and 345 Wh/L, respectively. |
| Lau et al. (2018) | Reviewed the challenges and opportunities for sulfide-based solid electrolytes [53]. |
| Yi et al. (2017) | Developed a hybrid polymer-ceramic electrolyte for a lithium-air battery, achieving 3.2×10-4 S/cm ionic conductivity at room temperature, and 350 cycles at 50 degrees Celsius [54]. |
| Song et al. (2016) | Developed a solid electrolyte based on a sodium-zirconium-magnesium-silicon-phosphorus-oxide material and achieved ionic conductivity of 3.5×10-3 S/cm at room temperature [55]. The prototype used sodium and sulfur in the anode and cathode, respectively. |
| Source | Key findings and/or contributions |
|---|---|
| Mills et al. (2024) | Developed flexible and durable sheets of 30 micrometer-thick solid-state electrodes, bounded to a sulfide solid-state electrolyte [14]. They found that optimizing the molecular weight of the binder can achieve the best combination of structural integrity and grain boundary resistance. The prototype achieved an energy density of 500 Wh/kg. |
| Ramasubramanian et al. (2024) | Explored strategies for mitigating energy barriers of the electrodes for metal-air batteries [36]. Strategies included 3D electrodes, oxygen-selective membranes, solid-state electrolytes, hybrid polymer electrodes, conductive electrocatalysts, and artificial solid-electrolyte interfaces. |
| Hayakawa et al. (2023) | Coated the electrode materials with a conductive additive (acetylene black and vapor-grown carbon fiber), yielding improved contact with the solid electrolyte material and reduced internal resistance [8]. |
| Alexander et al. (2023) | Proposed a single-phase mixed ion- and electron-conducting garnet for both electrodes to address the issues of limited plate stripping rates and dendrite growth in lithium metal anodes [7]. They observed six times more energy transfer than state-of-the-art batteries that can potentially last for 3,700 charge cycles. |
| Kim et al. (2020) | Proposed two methods to ensure that the solid electrolyte material distributed evenly within the porous structure of the electrode [22]. Discovered that infiltration of the solid electrolyte into the electrode improved at higher processing temperatures, particularly above the boiling point of ethanol, due to the solvent vaporizing. Found that mixing active materials of different particle sizes helped fill the capillary pores in the electrode, resulting in a denser structure. Achieved initial discharge capacity of 177 mAh/g. |
| Tan et al. (2019) | Developed a solid-state zinc-air battery by electroplating a zinc anode onto carbon paper to improve electron flow [56]. The cathode was oxygen from air, which increased the theoretical energy density. The prototype achieved a 30 mAh/g discharge capacity. |
| Kuang et al. (2019) | Reviewed the principles, challenges, and potential of thick electrodes that can store more active materials and fewer inactive components to achieve higher energy density without fundamentally altering the battery chemistry [18]. |
| Wang et al. (2018) | Utilized a molecular layer deposition method to develop an inorganic-organic hybrid interlayer (alucone) at the interface between the Li metal anode and the solid electrolyte [39]. The alucone served as an artificial SEI to suppress unwanted reactions at the interface by blocking electron transport. The battery achieved a high initial capacity of 120 mAh/g and a retention capacity of 60 mAh/g after 150 cycles. |
| Wu et al. (2016) | Reviewed problems and solutions to the interfacial issues between electrodes and electrolytes of different types [13]. They concluded that despite the progress made in developing solid electrolytes, significant challenges remain in forming intimate contact at its anode and cathode interfaces. |
| Source | Key findings and/or contributions |
|---|---|
| Huang et al. (2024) | Reviewed common failure issues in both polycrystal and single-crystal cathode structures [37]. Outlined strategies for improving single-crystal nickel-rich layered cathode materials, including synthesis regulation, element doping, and surface-interface modification. Explored the connection between structural design and electrochemical performance. |
| Razali et al. (2023) | Reviewed developments in solid state electrolytes (ceramic, polymer, soft sulfur-based, and their hybrid combinations) and concluded that lithium dendrite growth remains the key bottleneck in SSB development [6]. |
| Li et al. (2021) | Tackled the problem that although nickel rich cathodes exhibit high energy storage capacity, they have low cycling stability. Found that coating the NMC cathode material with nanoscale polycrystalline particles of a solid electrolyte improved ionic conductivity, reduced unwanted reactions during cycling, and stabilized the interface with the solid electrolyte [57]. The battery retained 80% of its capacity after 400 cycles at a moderate charging rate. |
| Wu et al. (2021) | Reviewed advancements in cathode-electrolyte interface formation in situ by conventional carbonate-based, fluorinated, concentrated, and solid-state electrolytes to improve thermal stability [58]. |
| Pimlott et al. (2021) | Reviewed the state-of-the-art in lithium-based materials for cathode materials, highlighting their challenges and opportunities [17]. |
| Source | Key findings and/or contributions |
|---|---|
| Liu et al. (2023) | Surveyed the opportunities of silicon-based anode due to their abundance and ability to store much more lithium ions than carbon by forming alloys with lithium [19]. Found that silicon-based nanostructures or composite materials with higher structural stability can tackle the issue of anode failure due to silicon’s volume expansion during cycling. However, these approaches introduce a myriad of other challenges. |
| Choi et al. (2021) | Used a roll pressing method to yield the in-situ formation of an intermetallic layer of silver-lithium alloy that mitigated uneven lithium deposition [31]. This approach enhanced the stability of the electrolyte (Li6PS5Cl) interface with the anode. They achieved 94.3% capacity retention after 140 cycles of high current rates. They reported that cells without the silver layer short circuited after only 30 cycles. |
| Lee et al. (2020) | Utilized a silver-carbon nanocomposite layer to improve the uniformity of lithium deposition by forming a silver-lithium alloy during cycling [59]. This resulted in dense and uniform lithium deposits that enhanced stability over 1,000 cycles. The carbon component acted as a mechanical support and separator, suppressing lithium dendrites penetration into the solid electrolyte. |
| Xu et al. (2021) | Used an electrolyte additive (potassium perfluorohexyl sulfonate) to form a stable SEI layer that protects the battery during lithium plating and stripping at the anode [60]. |
| Keles et al. (2020) | Used a magnetron sputtering process to create a superlattice structured anode film comprised of alternating layers of silicon, molybdenum, and copper [61]. The alternating layers distributed the stress from silicon’s expansion evenly across the material, preventing damage. Achieved stable performance and maintained capacity over 150 cycles. |
| Park et al. (2019) | Found that chemical attachments to carbon (functional groups like hydroxyl creating oxygen and hydrogen bonds) can participate in unwanted reactions at the solid electrolyte interface, leading to insulating byproducts that increase internal resistance [62]. Developed a graphitized hollow nanocarbon with fewer functional groups that reduced side reactions. |
| Sun et al. (2015) | Explored the research directions for spinet (Li4Ti5O12) anodes based on their promise of good performance, ease of fabrication, and low cost [63]. |
| Source | Key findings and/or contributions |
|---|---|
| Kazyak et al. (2024) | Summarized recent progress and ongoing challenges in developing SSBs, emphasizing the need for scalable and sustainable processing of battery components [64]. |
| Sharma et al. (2024) | Reviewed anode-free arrangements through a bipolar architecture [29]. |
| Kumar et al. (2024) | Reviewed trends in using nanoscale technologies to make various electric vehicle parts, including battery materials that employ nanotubes, graphene, and metal oxides [16]. |
| Chaudhary et al. (2024) | Demonstrated a structural LIB with the electrodes made of carbon fiber material [65]. They coated the cathode with lithium iron phosphate (LiFePO4) and used lithium bis (trifluoro methane) sulfonamide (LiTFSI) dissolved in ethyl-methylimidazolium tetrafluoroborate for the solid electrolyte. The prototype exhibited 30 Wh/kg, cyclic stability up to 1,000 cycles with just under 100% Coulombic efficiency, and elastic modulus of 76 GPa. |
| Lee et al. (2024) | Reviewed the inherent challenges in cell chemistries within lithium-sulfur based SSBs, aiming to inspire the battery community to advance this technology toward practical applications [9]. |
| Iyer et al. (2024) | Developed a structural sodium-ion battery by sandwiching a glass fiber between thin solid-state poly(ethylene oxide)-based composite electrolyte layers [43]. This approach yielded a high-tensile-strength structural electrolyte. They formed the structural cathode by heat pressing carbon fibers against the structural electrolyte. The resulting structural battery had a tensile strength of just under 41 MPa. It provided a typical energy density of 23 Wh/kg, 4.5 volts, ionic conductivity of 1.02×10-4 S/cm at 60 degrees Celsius, 500 cycles of lifetime, and retained 80% capacity until 225 cycles. |
| Ma et al. (2023) | Reviewed issues that impede fast charging of SSBs [38]. These included low ionic and electronic conductivities in solid-state electrolytes (SSEs), electrode/electrolyte interfacial resistances, and mechanical instabilities at high currents. |
| Whang et al. (2023) | Explored the opportunity space for transition metal sulfide conversion chemistries for stationary energy storage applications [66]. |
| Li et al. (2022) | Proposed a bipolar stacking of two solid-state cells by forming a non-flammable gel (ionogel) between solid particles [67]. |
| Fu et al. (2022) | Proposed a structural battery based on modified fiber metal laminates with added energy storage capability [68]. Metal sheets supporting the battery core served the dual purpose of resisting impact and conducting electricity. Demonstrated a prototype car frame with 11 mAh capacity. |
| Xu et al. (2019) | Developed a catalyst and membrane that significantly improved the performance of rechargeable solid-state zinc-air batteries [23]. They used a hydrothermal process (water at high temperature and pressure) to create the hybrid material by combining cobalt oxide (Co3O4) anchored on manganese oxide (MnO2), mixed with carbon nanotubes. The hybrid structure and interaction between the cobalt and manganese oxides better catalyzed the chemical reactions with oxygen. |
| Yamamoto et al. (2018) | Proposed a binder-free sheet-type battery construction by using removable volatile poly(propylene carbonate)-based binders [69]. Achieved a 2.6-fold increase in energy density over previously reported sheet-type batteries. |
| Yi et al. (2017) | Reviewed the development of polymer electrolytes for lithium-air batteries [54]. |
| Source | Key findings and/or contributions |
|---|---|
| Yang et al. (2024) | The SSB evaluated has a higher internal resistance than LIB, causing it to generate more heat, thus requiring a more capable thermal management system [3]. |
| Yang et al. (2024) | Developed a micro heat pipe array with air cooling to improve thermal management at the SSB pack level [41]. |
| Cao et al. (2023) | Advocated for using a neuron imaging technique to visualize SSB operation and evaluate issues such as interfacial compatibility, reaction mechanisms, structural stability, ion transport constraints, and lithium dendrite inhibition [4]. |
| Singh et al. (2023) | Utilized in-situ electrochemical scanning electron microscopy to examine the degradation mechanism at the anode interface between lithium metal and a solid electrolyte (Li6PS5Cl) during battery operation. They found that a nanocrystalline structure of lithium can help create a stable interface by allowing for a specific type of deformation (Coble creep) and uniform removal of lithium [28]. Also, they found that flaw-free surfaces can support high currents of more than 150 mA per square-centimeter. |
| Yang et al. (2022) | Reviewed the literature on ionic conductivity in SSB and concluded that some strategies for improvement include doping, defect engineering, microstructure tuning, and interface modification [32]. |
| Yoon et al. (2022) | Evaluated the solid electrolyte Li6PS5Cl and found that it is vulnerable to elevated-temperature storage as low as 70 degrees Celsius [70]. |
| Li et al. (2022) | Summarized experimental observations and modeling efforts to understand the factors influencing LIB cell degradation due to mechanical origins, structural changes, and electrochemical changes to forecast the evolution of SSB performance [67]. |
| Guo et al. (2022) | Investigated the electrochemistry of lithium-selenium SSBs at different temperatures using in-situ transmission electron microscopy equipped with a microelectromechanical systems heating device [71]. They found that lithium-ion conductivity and not electronic conductivity dictates the performance of this battery type. |
| Pasta et al. (2020) | Highlighted that the main challenges in developing SSBs lie in the interfaces between electrolytes and electrodes, mechanical properties of the device, and scalability of processing, rather than in the ionic conductivity of solid electrolytes [46]. |
| Zhang et al. (2019) | Utilized in-situ electrochemical impedance and Raman spectroscopy to study the degradation mechanisms in SSBs [72]. They found that lithium-ion migration is a significant cause of interfacial reactions that deteriorate the interfacial structure and electrochemical performance. |
| Gutierrez-Pardo et al. (2018) | Compared the performance of ceramic and polymer-ceramic composite electrolytes without coating to improve interfacial contacts with the electrodes [73]. They found that the composite electrolyte performed better at 70 degrees Celsius due to its enhanced interfacial contact. |
| Source | Key findings and/or contributions |
|---|---|
| Wu et al. (2023) | Explored the challenges, technologies, and concepts for recycling SSBs relative to LIBs [5]. |
| Boretti (2022) | Reviewed the battery market and found that despite significant technological improvements, environmental concerns rather than economics have been driving the shift to battery electric vehicles [74]. |
| Neumann et al. (2022) | Explored current practices in recycling, including material collection, sorting, transportation, handling, regulations, and new battery directive demands to predict challenges in recycling future SSBs [75]. |
| Bajolle et al. (2022) | Forecasted LIB price trends for scenarios ranging from rapid price stabilization due to insufficient raw material supply to market disruption by the introduction of SSBs [1]. |
| Fichtner (2021) | Reviewed trends in LIB storage capacity and price reduction [2]. Found that only a fraction of the overall improvement was due to better chemical compositions. Predicted that further improvements will be due to innovations in cell-to-pack and cell-to-chassis designs, allowing for the replacement of less sustainable, albeit more energy dense, cobalt-based active materials with more earth abundant and lower-cost materials such as LiFePO4. |
| Ball et al. (2020) | Reviewed the changing requirements for active materials in automotive LIB applications, anticipating responses from science and industry [76]. |
| Ding et al. (2019) | Discussed recent advances and challenges of electric vehicles, focusing on resources of critical elements, EV market evolution, and LIB cost and performance [77]. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




