Submitted:
23 September 2024
Posted:
25 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
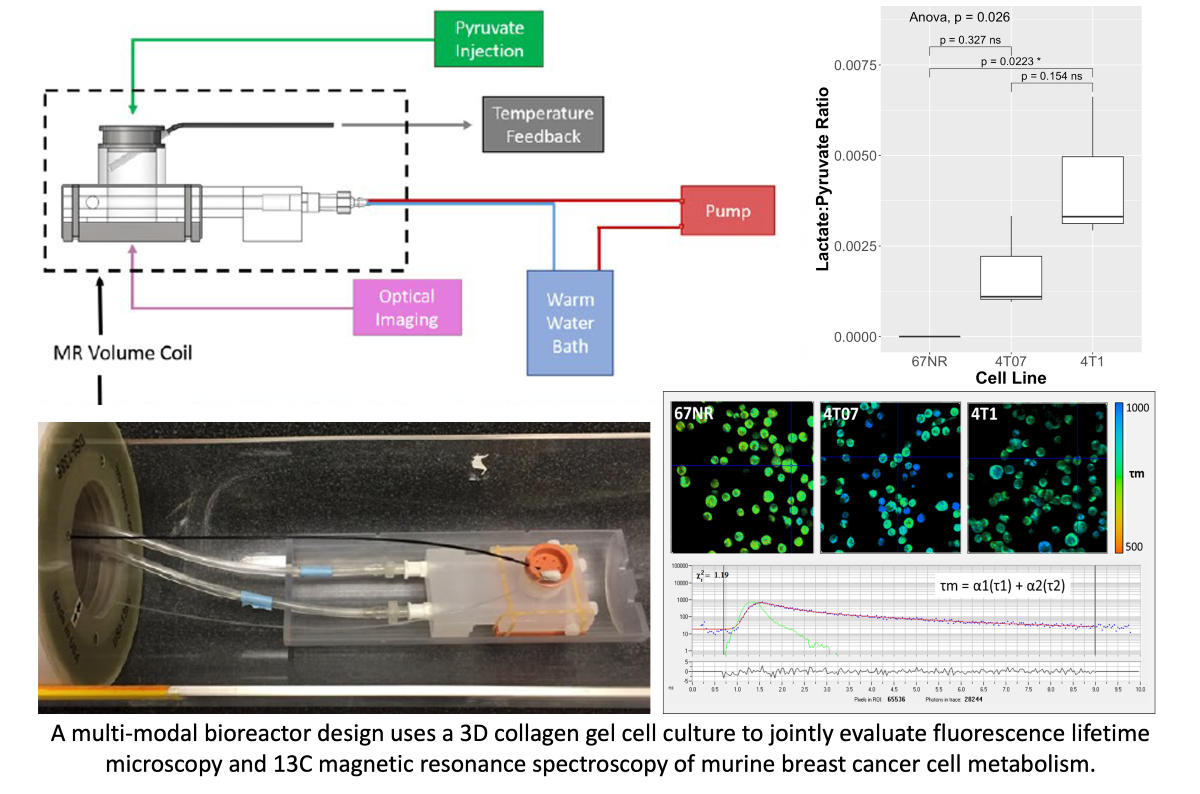
2.1. Cell Culture in the Bioreactor
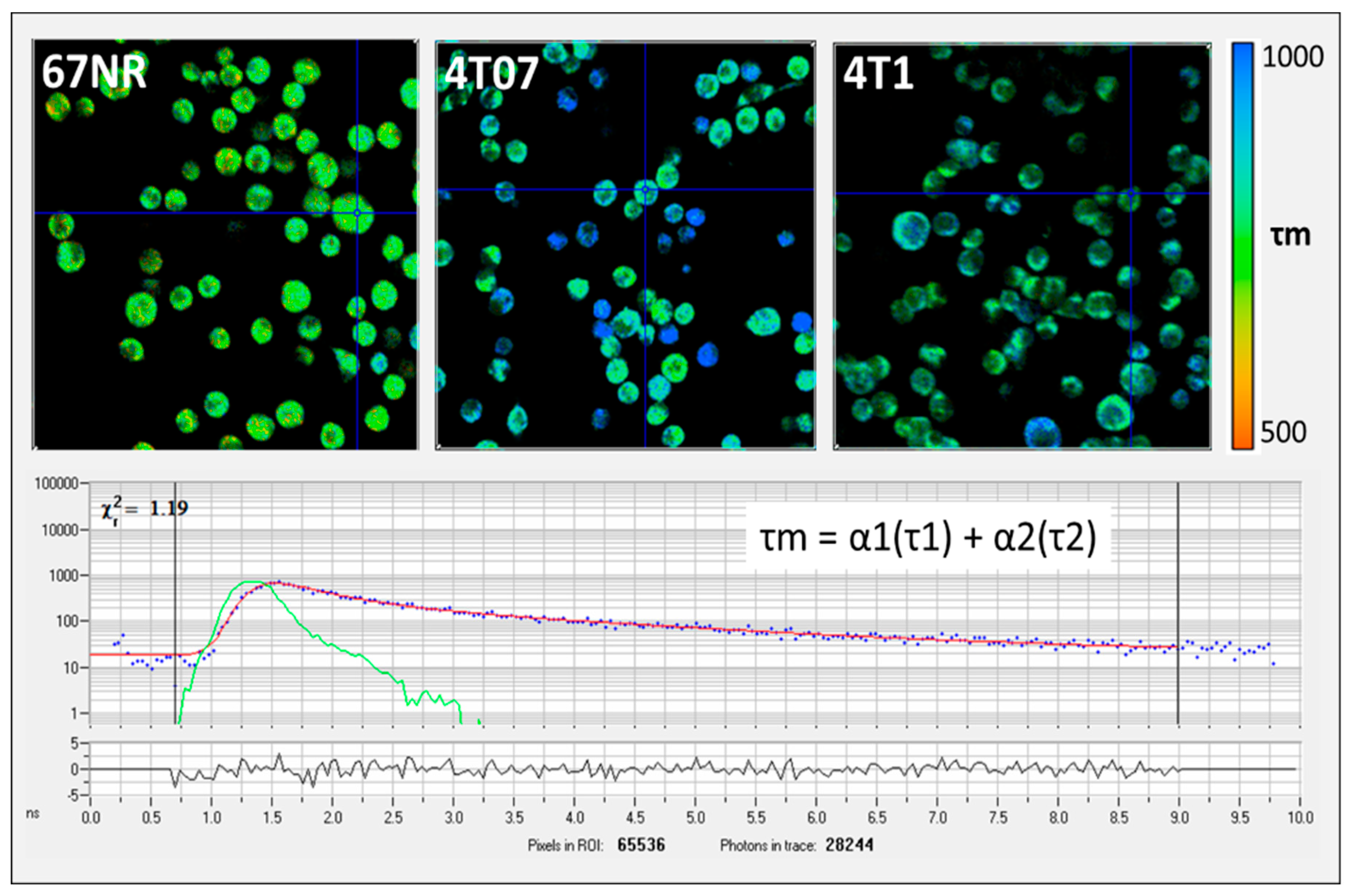
2.2. Fluorescence Lifetime Imaging Microscopy
2.3. Hyperpolarized Pyruvate MRS
2.4. Data and Statistical Analysis
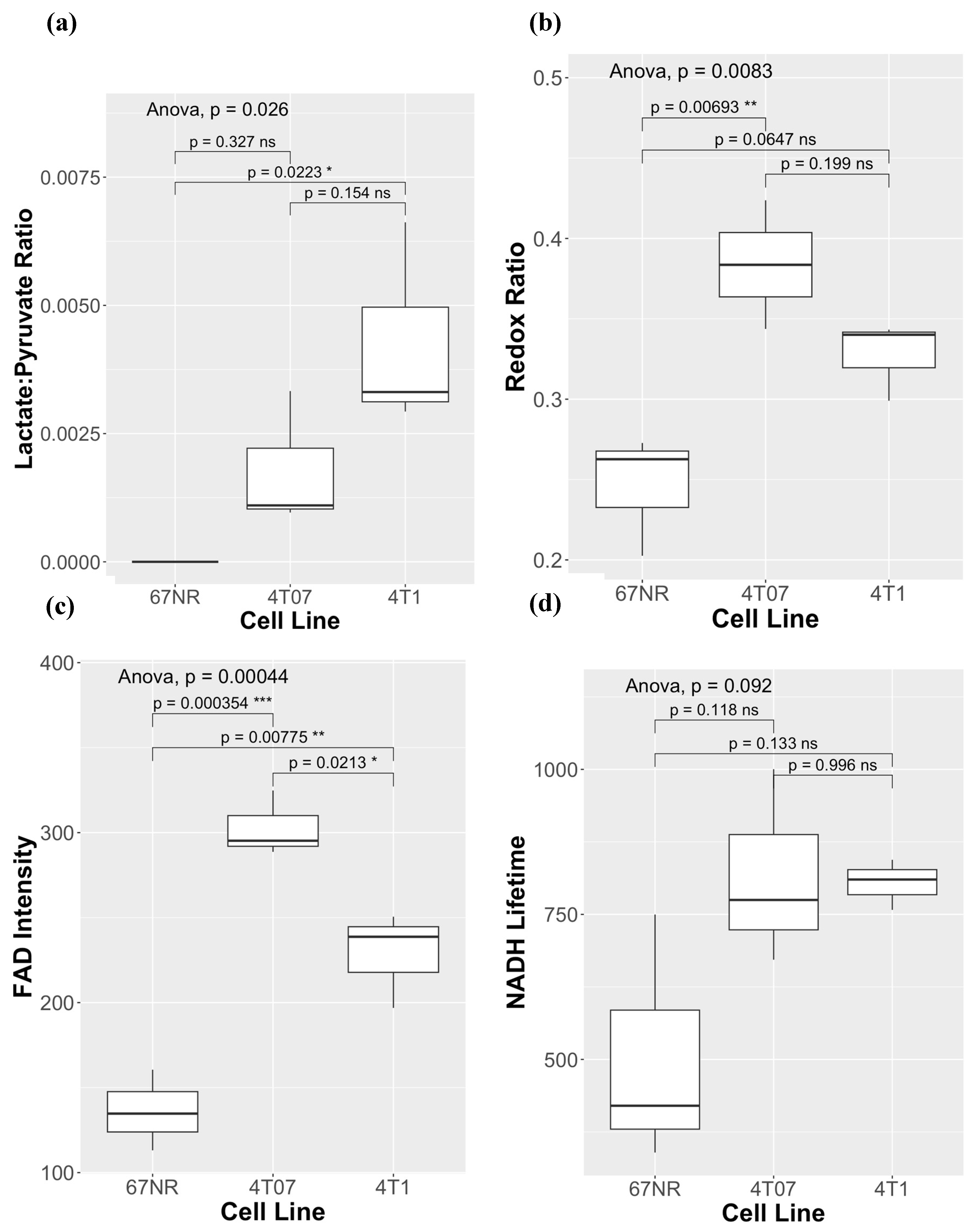
3. Results
4. Discussion
5. Conclusion
Acknowledgments
References
- Breast Cancer Facts and Figures 2017-2018. American Cancer Society. www.cancer.org.
- Meltzer A. Dormancy and breast cancer. J Surg Oncol. 1990 Mar;43(3):181-8. [CrossRef]
- Páez D, Labonte MJ, Bohanes P, Zhang W, Benhanim L, Ning Y, Wakatsuki T, Loupakis F, Lenz HJ. Cancer dormancy: a model of early dissemination and late cancer recurrence. Clin Cancer Res. 2012 Feb 1;18(3):645-53. [CrossRef]
- Brackstone M, Townson JL, Chambers AF. Tumour dormancy in breast cancer: an update. Breast Cancer Res. 2007;9(3):208. [CrossRef]
- Cox BL, Erickson-Bhatt S, Szulczewski JM, Squirrell JM, Ludwig KD, Macdonald EB, Swader R, Ponik SM, Eliceiri KW, Fain SB. A novel bioreactor for combined magnetic resonance spectroscopy and optical imaging of metabolism in 3D cell cultures. Magn Reson Med. 2019 May;81(5):3379-3391. [CrossRef]
- Warburg O. On the origin of cancer cells. Science. 1956 Feb 24;123(3191):309-14. [CrossRef]
- Vander Heiden MG, Cantley LC, Thompson CB. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science. 2009 May 22;324(5930):1029-33. [CrossRef]
- Dafni H, Ronen SM. Dynamic nuclear polarization in metabolic imaging of metastasis: common sense, hypersense and compressed sensing. Cancer Biomark. 2010;7(4):189-99. [CrossRef]
- Brindle KM. Imaging metabolism with hyperpolarized (13)C-labeled cell substrates. J Am Chem Soc. 2015 May 27;137(20):6418-27. [CrossRef]
- Christensen CE, Karlsson M, Winther JR, Jensen PR, Lerche MH. Non-invasive in-cell determination of free cytosolic [NAD+]/[NADH] ratios using hyperpolarized glucose show large variations in metabolic phenotypes. J Biol Chem. 2014 Jan 24;289(4):2344-52. [CrossRef]
- Witney T.H., Kettunen M.I., Hu D.E., Gallagher F.A., Bohndiek S.E., Napolitano R., Brindle K.M. Detecting Treatment Response in a Model of Human Breast Adenocarcinoma Using Hyperpolarised [1-13C]Pyruvate and [1,4-13C2]Fumarate. Br. J. Cancer. 2010;103:1400–1406. [CrossRef]
- Harris T., Eliyahu G., Frydman L., Degani H. Kinetics of Hyperpolarized 13C1-Pyruvate Transport and Metabolism in Living Human Breast Cancer Cells. Proc. Natl. Acad. Sci. USA. 2009;106:18131–18136. [CrossRef]
- Macdonald E.B., Begovatz P., Barton G.P., Erickson-Bhatt S., Inman D.R., Cox B.L., Eliceiri K.W., Strigel R.M., Ponik S.M., Fain S.B. Hyperpolarized 13C Magnetic Resonance Spectroscopic Imaging of Pyruvate Metabolism in Murine Breast Cancer Models of Different Metastatic Potential. Metabolites. 2021;11:274. [CrossRef]
- Grashei M., Biechl P., Schilling F., Otto A.M. Conversion of Hyperpolarized [1-13 C]Pyruvate in Breast Cancer Cells Depends on Their Malignancy, Metabolic Program and Nutrient Microenvironment. Cancers. 2022;14:1845. Sidani M1, Wyckoff J, Xue C, Segall JE, Condeelis J. Probing the microenvironment of mammary tumors using multiphoton microscopy. J Mammary Gland Biol Neoplasia. 2006 Apr;11(2):151-63. [CrossRef]
- Sidani M1, Wyckoff J, Xue C, Segall JE, Condeelis J. Probing the microenvironment of mammary tumors using multiphoton microscopy. J Mammary Gland Biol Neoplasia. 2006 Apr;11(2):151-63. [CrossRef]
- Provenzano PP1, Rueden CT, Trier SM, Yan L, Ponik SM, Inman DR, Keely PJ, Eliceiri KW. Nonlinear optical imaging and spectral-lifetime computational analysis of endogenous and exogenous fluorophores in breast cancer. J Biomed Opt. 2008 May-Jun;13(3):031220. [CrossRef]
- Conklin MW, Provenzano PP, Eliceiri KW, Sullivan R, Keely PJ. Fluorescence lifetime imaging of endogenous fluorophores in histopathology sections reveals differences between normal and tumor epithelium in carcinoma in situ of the breast. Cell Biochem Biophys. 2009;53(3):145-57. [CrossRef]
- Li LZ, Xu HN, Ranji M, Nioka S, Chance B. Mitochondrial redox imaging for cancer diagnostic and therapeutic studies. J Innov Opt Health Sci. 2009 Oct;2(4):325-341. [CrossRef]
- Ostrander JH, McMahon CM, Lem S, Millon SR, Brown JQ, Seewaldt VL, Ramanujam N. Optical redox ratio differentiates breast cancer cell lines based on estrogen receptor status. Cancer Res. 2010 Jun 1;70(11):4759-66. [CrossRef]
- Walsh A, Cook RS, Rexer B, Arteaga CL, Skala MC. Optical imaging of metabolism in HER2 overexpressing breast cancer cells. Biomed Opt Express. 2012 Jan 1;3(1):75-85. [CrossRef]
- Xu HN, Zheng G, Tchou J, Nioka S, Li LZ. Characterizing the metabolic heterogeneity in human breast cancer xenografts by 3D high resolution fluorescence imaging. Springerplus. 2013 Dec;2(1):73. [CrossRef]
- Walsh AJ, Cook RS, Sanders ME, Aurisicchio L, Ciliberto G, Arteaga CL, Skala MC. Quantitative optical imaging of primary tumor organoid metabolism predicts drug response in breast cancer. Cancer Res. 2014 Sep 15;74(18):5184-94. [CrossRef]
- Cannon TM, Shah AT, Walsh AJ, Skala MC. High-throughput measurements of the optical redox ratio using a commercial microplate reader. J Biomed Opt. 2015 Jan;20(1):010503. [CrossRef]
- Sun N, Xu HN, Luo Q, Li LZ. Potential Indexing of the Invasiveness of Breast Cancer Cells by Mitochondrial Redox Ratios. Adv Exp Med Biol. 2016;923:121-127. [CrossRef]
- Alhallak K, Rebello LG, Muldoon TJ, Quinn KP, Rajaram N. Optical redox ratio identifies metastatic potential-dependent changes in breast cancer cell metabolism. Biomed Opt Express. 2016 Oct 3;7(11):4364-4374. [CrossRef]
- Cannon TM, Shah AT, Skala MC. Autofluorescence imaging captures heterogeneous drug response differences between 2D and 3D breast cancer cultures. Biomed Opt Express. 2017 Feb 28;8(3):1911-1925. [CrossRef]
- Hou J, Williams J, Botvinick EL, Potma EO, Tromberg BJ. Visualization of Breast Cancer Metabolism Using Multimodal Nonlinear Optical Microscopy of Cellular Lipids and Redox State. Cancer Res. 2018 May 15;78(10):2503-2512. [CrossRef]
- Aslakson CJ, Miller FR. Selective events in the metastatic process defined by analysis of the sequential dissemination of subpopulations of a mouse mammary tumor. Cancer Res. 1992 Mar 15;52(6):1399-405.
- Burkel B, Morris BA, Ponik SM, Riching KM, Eliceiri KW, Keely PJ. Preparation of 3D Collagen Gels and Microchannels for the Study of 3D Interactions In Vivo. J Vis Exp. 2016 May 9;(111). [CrossRef]
- Becker W. Fluorescence lifetime imaging--techniques and applications. J Microsc. 2012 Aug;247(2):119-36. [CrossRef]
- Ardenkjaer-Larsen JH, Fridlund B, Gram A, Hansson G, Hansson L, Lerche MH, Servin R, Thaning M, Golman K. Proc Natl Acad Sci U S A. 2003 Sep 2;100(18):10158-63. [CrossRef]
- Rowland IJ, Peterson ET, Gordon JW, Fain SB. Curr Pharm Biotechnol. 2010 Sep 1;11(6):709-19. Review. [CrossRef]
- Naressi A, Couturier C, Devos JM, Janssen M, Mangeat C, de Beer R, Graveron-Demilly D. Java-based graphical user interface for the MRUI quantitation package. MAGMA. 2001 May;12(2-3):141-52. [CrossRef]
- Alhallak K, Rebello LG, Muldoon TJ, Quinn KP, Rajaram N. Optical redox ratio identifies metastatic potential-dependent changes in breast cancer cell metabolism. Biomed Opt Express. 2016 Oct 3;7(11):4364-4374. [CrossRef]
- Morris BA, Burkel B, Ponik SM, Fan J, Condeelis JS, Aguirre-Ghiso JA, Castracane J, Denu JM, Keely PJ. Collagen Matrix Density Drives the Metabolic Shift in Breast Cancer Cells. EBioMedicine. 2016 Nov;13:146-156. [CrossRef]
- Xing, J., Qi, L., Liu, X. et al. Roles of mitochondrial fusion and fission in breast cancer progression: a systematic review. World J Surg Onc 20, 331 (2022). [CrossRef]
- Zakic T, Kalezic A, Drvendzija Z, Udicki M, Ivkovic Kapicl T, Srdic Galic B, Korac A, Jankovic A, Korac B. Breast Cancer: Mitochondria-Centered Metabolic Alterations in Tumor and Associated Adipose Tissue. Cells. 2024 Jan 15;13(2):155. [CrossRef]

| NADH | FAD | |||||
|---|---|---|---|---|---|---|
| Cell Line | Lac/Pyr Ratio | Redox Ratio | Intensity |
Lifetime (Tau Mean, ps) |
Intensity |
Lifetime (Tau Mean, ps) |
| 4T1 | 0.00293 | 0.299 | 613.58 | 757.8 | 250.56 | 751.8 |
| 4T1 | 0.00331 | 0.343 | 347.40 | 810.1 | 196.88 | 835.2 |
| 4T1 | 0.00662 | 0.340 | 416.14 | 844.2 | 238.73 | 975.8 |
| Mean (SD) | 0.00429 (0.0020) | 0.327 (0.02) | 459.04 (138.2) | 804.0 (43.5) | 228.7 (28.2) | 854.3 (113.2) |
| 4T07 | 0.00333 | 0.424 | 432.05 | 1000.3 | 324.82 | 964.1 |
| 4T07 | 0.0011 | 0.344 | 520.70 | 671.9 | 288.71 | 910.8 |
| 4T07 | 0.00096 | 0.384 | 438.34 | 774.9 | 295.24 | 971.7 |
| Mean (SD) | 0.00180 (0.0013) | 0.384 (0.04) | 463.70 (49.5) | 815.7 (168.0) | 302.9 (19.2) | 948.9 (33.2) |
| 67NR | 0 | 0.273 | 464.36 | 749.8 | 134.70 | 813.2 |
| 67NR | 0 | 0.263 | 414.45 | 339.5 | 160.55 | 396.9 |
| 67NR | 0 | 0.203 | 448.20 | 420.1 | 113.11 | 935.1 |
| Mean (SD) | 0 (0) | 0.246 (0.04) | 442.33 (25.5) | 503.1 (217.4) | 136.1 (23.8) | 715.1 (282.2) |
| Comparison HP-MRS Lac/Pyr Ratio vs. |
Spearman Correlation Coefficient | p-value |
|---|---|---|
| Redox Ratio | 0.63 | 0.071+ |
| NADH Intensity | -0.25 | 0.51 |
| NADH Lifetime | 0.86 | 0.0026* |
| FAD intensity | 0.59 | 0.092 |
| FAD Lifetime | 0.49 | 0.18 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




