Submitted:
07 September 2024
Posted:
09 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Exclusion Criteria
2.3. Norepinephrine Equivalent (NEE)
2.4. Clinical Protocol
2.5. Sample Collection and Measurement
2.6. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shankar-Hari, M.; Phillips, G.S.; Levy, M.L.; Seymour, C.W.; Liu, V.X.; Deutschman, C.S.; Angus, D.C.; Rubenfeld, G.D.; Singer, M.; Sepsis Definitions Task Force. Developing a new definition and assessing new clinical criteria for septic shock: for the third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA 2016, 315, 775–787. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.D.; MacCallum, N.S.; Harris, S.; Brealey, D.A.; Palmer, E.; Hetherington, J.; Shi, S.; Perez-Suarez, D.; Ercole, A.; Watkinson, P.J.; et al. Descriptors of sepsis using the Sepsis-3 criteria: A cohort study in critical care units within the U.K. National Institute for Health Research critical care health informatics collaborative. Crit Care Med 2021, 49, 1883–1894. [Google Scholar] [CrossRef]
- Im, Y.; Kang, D.; Ko, R.E.; Lee, Y.J.; Lim, S.Y.; Park, S.; Na, S.J.; Chung, C.R.; Park, M.H.; Oh, D.K.; et al. Time-to-antibiotics and clinical outcomes in patients with sepsis and septic shock: a prospective nationwide multicenter cohort study. Crit Care 2022, 26, 19. [Google Scholar] [CrossRef] [PubMed]
- Levy, M.M.; Evans, L.E.; Rhodes, A. The Surviving Sepsis Campaign Bundle: 2018 update. Intensive Care Med 2018, 44, 925–928. [Google Scholar] [CrossRef]
- Tang, F.; Yuan, H.; Li, X.; Qiao, L. Effect of delayed antibiotic use on mortality outcomes in patients with sepsis or septic shock: A systematic review and meta-analysis. Int Immunopharmacol 2024, 129, 111616. [Google Scholar] [CrossRef]
- Vallés, J.; Rello, J.; Ochagavía, A.; Garnacho, J.; Alcalá, M.A. Community-acquired bloodstream infection in critically ill adult patients: impact of shock and inappropriate antibiotic therapy on survival. Chest 2003, 123, 1615–1624. [Google Scholar] [CrossRef]
- Micek, S.T.; Welch, E.C.; Khan, J.; Pervez, M.; Doherty, J.A.; Reichley, R.M.; Kollef, M.H. Empiric combination antibiotic therapy is associated with improved outcome against sepsis due to Gram-negative bacteria: a retrospective analysis. Antimicrob Agents Chemother 2010, 54, 1742–1748. [Google Scholar] [CrossRef] [PubMed]
- Kadri, S.S.; Lai, Y.L.; Warner, S.; Strich, J.R.; Babiker, A.; Ricotta, E.E.; Demirkale, C.Y.; Dekker, J.P.; Palmore, T.N.; Rhee, C.; et al. Inappropriate empirical antibiotic therapy for bloodstream infections based on discordant in-vitro susceptibilities: a retrospective cohort analysis of prevalence, predictors, and mortality risk in US hospitals. Lancet Infect Dis 2021, 21, 241–251. [Google Scholar] [CrossRef]
- Miller, M.B.; Atrzadeh, F.; Burnham, C.A.; Cavalieri, S.; Dunn, J.; Jones, S.; Mathews, C.; McNult, P.; Meduri, J.; Newhouse, C.; et al. Clinical utility of advanced microbiology testing tools. J Clin Microbiol 2019, 57, e00495–19. [Google Scholar] [CrossRef]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.D.; Coopersmith, C.M.; et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef]
- Kotani, Y.; Di Gioia, A.; Landoni, G.; Belletti, A.; Khanna, A.K. An updated “norepinephrine equivalent” score in intensive care as a marker of shock severity. Crit Care 2023, 27, 29. [Google Scholar] [CrossRef] [PubMed]
- Khanna, A.; English, S.W.; Wang, X.S.; Ham, K.; Tumlin, J.; Szerlip, H.; Busse, L.W.; Altaweel, L.; Albertson, T.E.; Mackey, C.; et al. Angiotensin II for the treatment of vasodilatory shock. N Engl J Med 2017, 377, 419–430. [Google Scholar] [CrossRef] [PubMed]
- Laterre, P.F.; Berry, S.M.; Blemings, A.; Carlsen, J.E.; François, B.; Graves, T.; Jacobsen, K.; Lewis, R.J.; Opal, S.M.; Perner, A.; et al. Effect of selepressin vs placebo on ventilator- and vasopressor-free days in patients with septic shock: the SEPSIS-ACT randomized clinical trial. JAMA 2019, 322, 1476–1485. [Google Scholar] [CrossRef]
- Goradia, S.; Sardaneh, A.A.; Narayan, S.W.; Penm, J.; Patanwala, A.E. Vasopressor dose equivalence: A scoping review and suggested formula. J Crit Care 2021, 61, 233–240. [Google Scholar] [CrossRef]
- Evans, L.; Rhodes, A.; Alhazzani, W.; Antonelli, M.; Coopersmith, C.M.; French, C.; Machado, F.R.; Mcintyre, L.; Ostermann, M.; Prescott, H.C.; et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med 2021, 47, 1181–1247. [Google Scholar] [CrossRef] [PubMed]
- Rygård, S.L.; Butler, E.; Granholm, A.; Møller, M.H.; Cohen, J.; Finfer, S.; Perner, A.; Myburgh, J.; Venkatesh, B.; Delaney, A. Low-dose corticosteroids for adult patients with septic shock: a systematic review with meta-analysis and trial sequential analysis. Intensive Care Med 2018, 44, 1003–1016. [Google Scholar] [CrossRef]
- Vincent, J.L.; de Mendonça, A.; Cantraine, F.; Moreno, R.; Takala, J.; Suter, P.M.; Sprung, C.L.; Colardyn, F.; Blecher, S. Use of the SOFA score to assess the incidence of organ dysfunction/failure in intensive care units: results of a multicenter, prospective study. Working group on “sepsis-related problems” of the European Society of Intensive Care Medicine. Crit Care Med 1998, 26, 1793–1800. [Google Scholar] [CrossRef]
- Teja, B.; Bosch, N.A.; Walkey, A.J. How we escalate vasopressor and corticosteroid therapy in patients with septic shock. Chest 2023, 163, 567–574. [Google Scholar] [CrossRef]
- Ferrer, R.; Martin-Loeches, I.; Phillips, G.; Osborn, T.M.; Townsend, S.; Dellinger, R.P.; Artigas, A.; Schorr, C.; Levy, M.M. Empiric antibiotic treatment reduces mortality in severe sepsis and septic shock from the first hour: results from a guideline-based performance improvement program. Crit Care Med 2014, 42, 1749–1755. [Google Scholar] [CrossRef]
- Perez, K.K.; Olsen, R.J.; Musick, W.L.; Cernoch, P.L.; Davis, J.R.; Peterson, L.E.; Musser, J.M. Integrating rapid diagnostics and antimicrobial stewardship improves outcomes in patients with antibiotic-resistant Gram-negative bacteremia. J Infect 2014, 69, 216–225. [Google Scholar] [CrossRef]
- Okamoto, M.; Maejima, M.; Goto, T.; Mikawa, T.; Hosaka, K.; Nagakubo, Y.; Hirotsu, Y.; Amemiya, K.; Sueki, H.; Omata, M. Impact of the FilmArray rapid multiplex PCR assay on clinical outcomes of patients with bacteremia. Diagnostics (Basel) 2023, 13, 1935. [Google Scholar] [CrossRef] [PubMed]
- Rudd, K.E.; Johnson, S.C.; Agesa, K.M.; Shackelford, K.A.; Tsoi, D.; Kievlan, D.R.; Colombara, D.V.; Ikuta, K.S.; Kissoon, N.; Finfer, S.; et al. Global, regional, and national sepsis incidence and mortality, 1990–2017: analysis for the Global Burden of Disease Study. Lancet 2020, 395, 200–211. [Google Scholar] [CrossRef] [PubMed]

| Total | Susceptible Group |
Resistant group |
p value | ||
|---|---|---|---|---|---|
| Number | 108 | 94 | 14 | ||
| Age (years) | 78 (70−86) | 77 (70−85) | 86 (74−90) | 0.0608 | |
| Male | 64 (59%) | 53 (56%) | 11 (79%) | 0.1498 | |
| BMI, (kg/m2) | 21 (18-25) | 22 (18-25) | 20 (17-26) | 0.6706 | |
| Weight (kg) | 53 (43-65) | 54 (44-65) | 51 (40-55) | 0.2625 | |
| Outcome | Survived in ICU | 77 (71%) | 69 (73%) | 8 (57%) | 0.2196 |
| ICU stay (days) | 6 (3−11.5) | 7 (3−13) | 3.5 (1−5) | 0.0137 | |
| Patient Profile | SOFA score | 11.5 (9−13) | 12 (9−13) | 11 (9−13) | 0.5191 |
| Respiration | 2 (1−2) | 2 (1−2) | 2 (2−3) | 0.3060 | |
| Coagulation | 1 (0−2) | 1 (0−2) | 0 (0−1) | 0.0178 | |
| Hepatobiliary | 0 (0−1) | 0 (0−1) | 0 (0−1) | 0.4969 | |
| Cardiovascular | 4 (4−4) | 4 (4−4) | 4 (4−4) | 0.2934 | |
| Central nervous | 2 (1−3) | 2 (1−3) | 2 (1−2) | 0.4846 | |
| Renal | 2 (1−4) | 2 (1−4) | 3 (1−4) | 0.6333 | |
| Initial lactate (mmol/L) | 5.4 (3.4−7.3) | 5.7 (3.6−7.8) | 3.8 (2.0−5.8) | 0.0197 | |
| Initial pH | 7.35 (7.28-7.44) | 7.35 (7.27-7.44) | 7.39 (7.34-7.45) | 0.2510 | |
| Initial HCO3- initial (mmol/L) | 17.5 (13.1-20.3) | 17.0 (12.0-20.1) | 19.4 (16.2-23.0) | 0.1200 | |
| WBC (104/µl) | 11 (6−18) | 11 (6−18) | 12 (8−15) | 0.7076 | |
| Neutro/Lympho ratio1 | 17 (9-34) | 17 (9-35) | 19 (9-21) | 0.6657 | |
| CRP (mg/dl) | 13 (5−25) | 13 (5−26) | 9 (4−20) | 0.3652 | |
| Medical history | Diabetes | 47 (44%) | 43 (46%) | 4 (29%) | 0.2621 |
| Steroid use | 11 (10%) | 10 (11%) | 1 (7%) | 1.0000 | |
| Treatment | Ventilator use | 63 (58%) | 56 (60%) | 7 (50%) | 0.5673 |
| Haemodialysis | 48 (44%) | 41 (44%) | 7 (50%) | 0.7753 | |
| Low-dose steroid | 92 (85%) | 81 (86%) | 11 (79%) | 0.4328 | |
| Invasive source control | 31 (29%) | 27 (29%) | 4 (29%) | 1.0000 | |
| Total infusion volume in first 24 hours, ml | 6515 (5325−8412) | 6526 (5299−8659) | 5831 (5451−7299) | 0.5280 |
| Total | Susceptible group |
Resistant group |
p value | ||
|---|---|---|---|---|---|
| Number | 108 | 94 | 14 | ||
| Infectious source1 | UTI | 40 (37%) | 36 (38%) | 4 (29%) | 0.7665 |
| Pneumonia | 18 (17%) | 14 (15%) | 4 (29%) | 0.2651 | |
| STI | 16 (15%) | 14 (15%) | 2 (14%) | 0.9524 | |
| biliary tract | 15 (14%) | 14 (15%) | 1 (7%) | 0.4340 | |
| Intra−abdominal | 14 (13%) | 12 (13%) | 2 (14%) | 0.8745 | |
| Others3 | 6 (6%) | 4 (4%) | 2 (14%) | 0.1264 | |
| Unknown | 11 (10%) | 10 (11%) | 1 (7%) | 0.6866 | |
| Gram stain classification of causative bacteria2 | GPC | 45 (42%) | 35 (37%) | 10 (71%) | 0.0206 |
| GPR | 15 (14%) | 14 (15%) | 1 (7%) | 0.6870 | |
| GNC | 0 | 0 | 0 | ||
| GNR | 64 (59%) | 59 (63%) | 5 (36%) | 0.0793 | |
| Initial antibiotics4 | ABPC/SBT | 6 | 4 | 2 | |
| TAZ/PIPC | 42 | 37 | 5 | ||
| CTX | 2 | 1 | 1 | ||
| CTRX | 18 | 16 | 2 | ||
| CFPM | 13 | 11 | 2 | ||
| MEPM | 10 | 9 | 1 | ||
| DRPM | 9 | 9 | 0 | ||
| LVFX | 2 | 1 | 1 | ||
| TEIC | 24 | 22 | 2 | ||
| VCM | 3 | 3 | 0 | ||
| Others | 8 | 7 | 1 | ||
| Time from diagnosis to initial antibiotic administration (min) | 75.5 (45−121) | 78 (45−125) | 62.5 (48−109.5) | 0.4643 | |
| Additional antibiotics | Cases | 26 (24%) | 14 (15%) | 12 (86%) | 0.0555 |
| days from initial to addition (day) | 2 (2−3) | 2 (2−3) | 2 (2−3) | 0.8656 | |
| De-escalation | 53 (49%) | 53 (71%) | 0 (0%) | <0.0001 | |
| Total days antibiotics administered (day) | 5 (12−16) | 12 (7−16) | 5 (3−13.5) | 0.0654 | |
| Total | Susceptible Group |
Resistant Group |
p value | ||
|---|---|---|---|---|---|
| Number | 108 | 94 | 14 | ||
| NEE1 | Max NEE | 0.39 (0.26−0.46) | 0.39 (0.26−0.45) | 0.41 (0.27−0.52) | 0.4209 |
| Max NE2 | 0.33 (0.24−0.40) | 0.32 (0.24−0.40) | 0.38 (0.24−0.44) | 0.2846 | |
| Max VA3 | 0 (0–0.33) | 0 (0–0.33) | 0 (0–0.33) | 0.7966 | |
| Max DoA4 | 0 (0–0) | 0 (0–0) | 0 (0–0) | 0.6504 | |
| Max Epi5 | 0 (0–0) | 0 (0–0) | 0 (0–0) | 0.3793 | |
| NEE time (h) | Max from initial | 9 (4–16) (n=108) | 9 (3–14) (n=94) | 11 (6–22) (n=14) | 0.1530 |
| Max last from initial | 14 (7–23) (n=101) | 14 (7–22) (n=91) | 19 (10–48) (n=10) | 0.1499 |
| Total | Susceptible group |
Resistant group |
p value | ||
|---|---|---|---|---|---|
| Number | 87 | 80 | 7 | ||
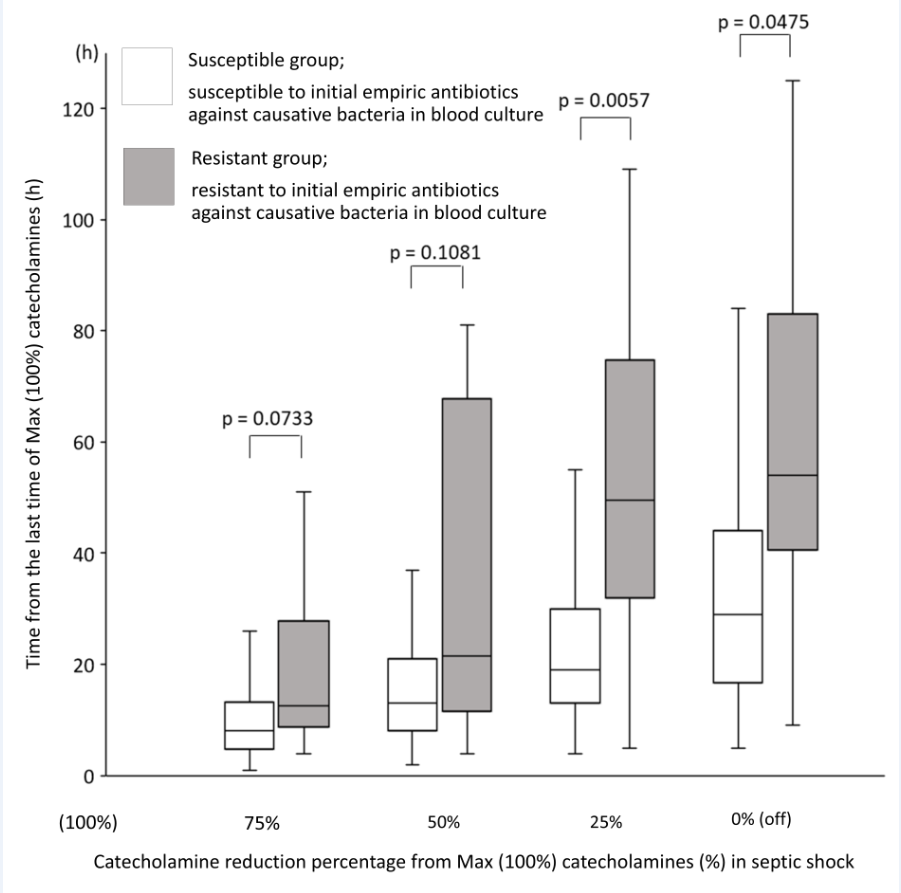
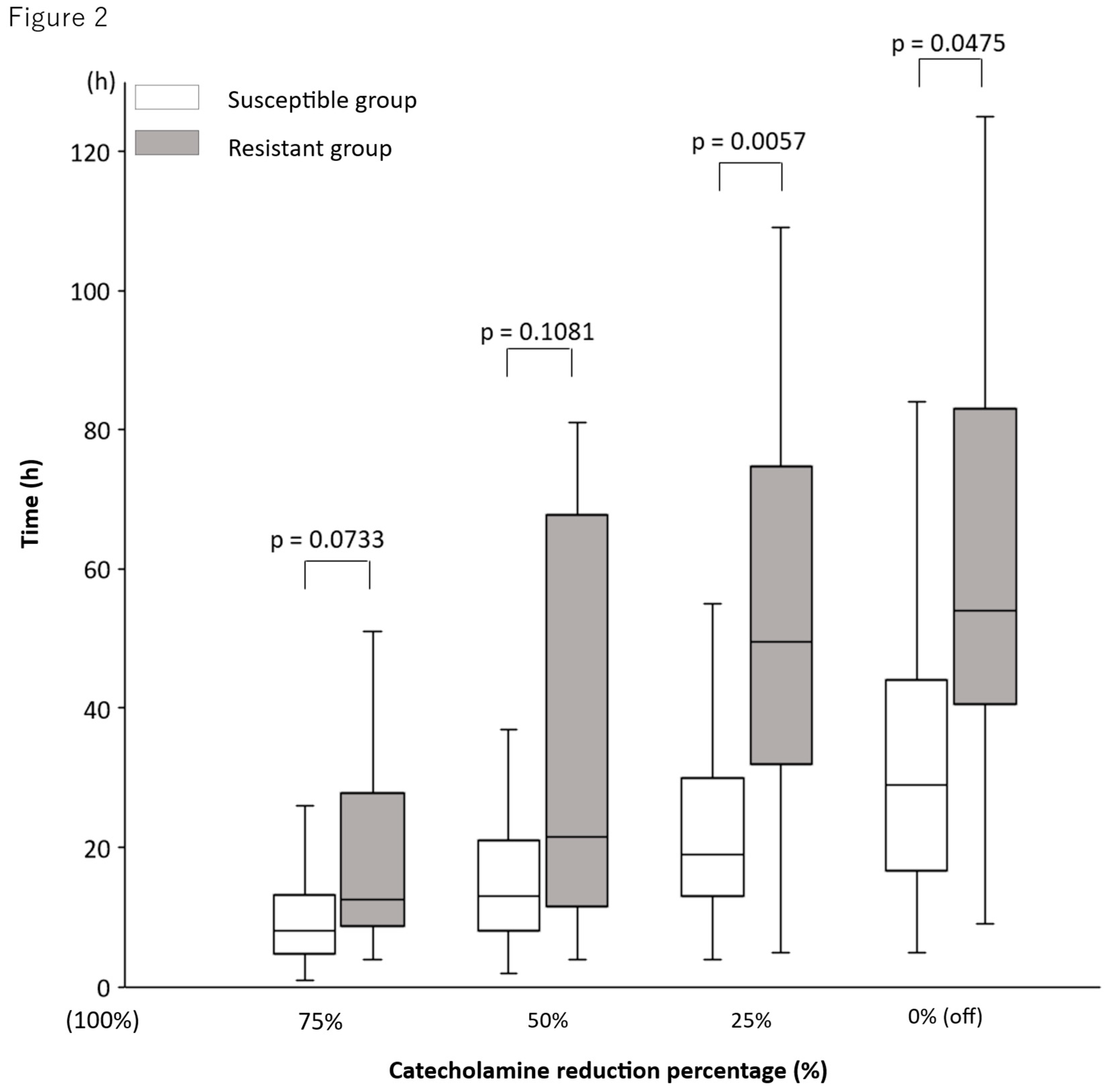
| Time to catecholamines reduction from 100% 1 (h) | |||||
| 75% | 9 (5–15) | 8 (5–14) | 10 (8.5–17.5) | 0.2348 | |
| 50% | 13 (8.5–21) | 13 (8–20) | 17 (11–51) | 0.2541 | |
| 25% | 20 (13–31.5) | 19 (13–30) | 46 (31–72.5) | 0.0152 | |
| 0% | 31 (17–47) | 29 (17–44) | 54 (40.5–83) | 0.0475 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




