Submitted:
03 September 2024
Posted:
04 September 2024
You are already at the latest version
Abstract
Keywords:
1. Background
1.1. Sarcopenia and Frailty Negatively Impact the Longevity of the Mid-Life and Elderly Population
1.2. Hypothyroidism Is Potentially Associated with Sarcopenia and Frailty.
1.3. Low ALT Values Serve as a Bio-Marker for Sarcopenia and Frailty
1.4. Aim of the Current Study
2. Patients and Methods
2.1. Study Population
2.2. Statistical Analysis
3. Results
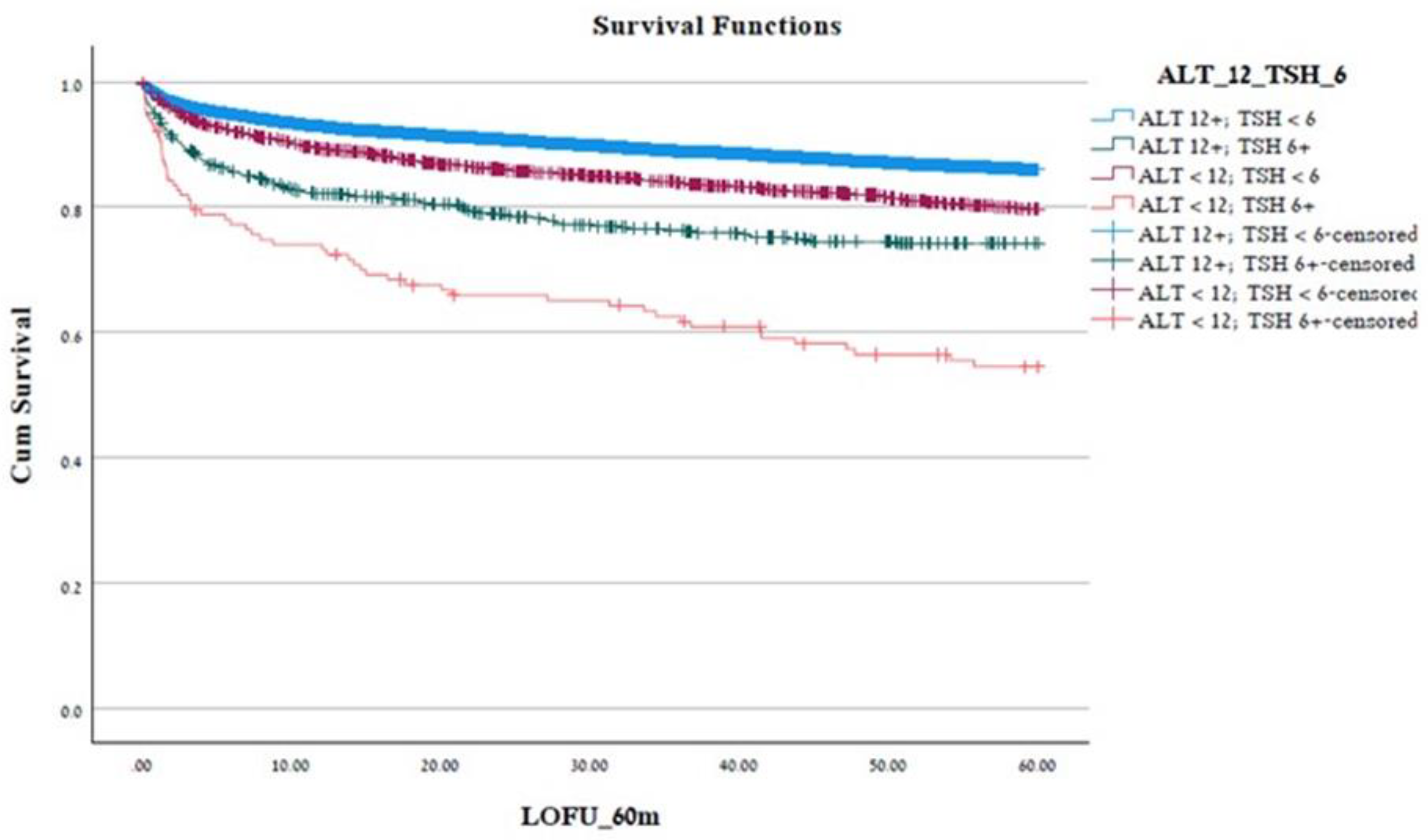
3.1. Univariate Analysis
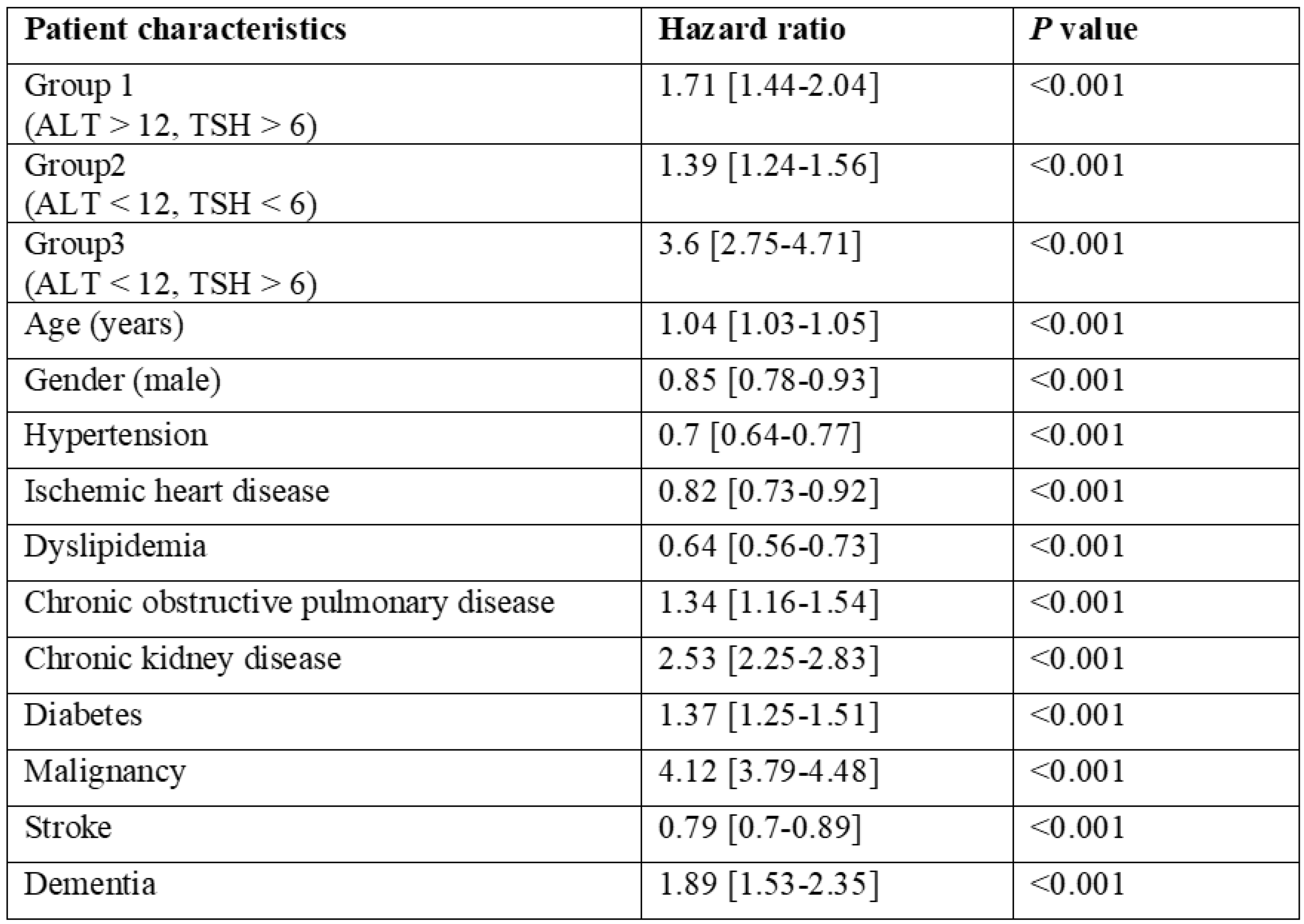
3.2. Multivariate Analysis
4. Discussion
5. Conclusions
Limitations
Conflicts of Interests’ declaration
References
- Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, et al. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci [Internet]. 2001 [cited 2024 Jan 29];56(3). Available from: https://pubmed.ncbi.nlm.nih.gov/11253156/. [CrossRef]
- Ajibawo T, Okunowo O. Higher Hospital Frailty Risk Score Is an Independent Predictor of In-Hospital Mortality in Hospitalized Older Adults with Obstructive Sleep Apnea. Geriatrics (Basel) [Internet]. 2022 Nov 14 [cited 2022 Dec 15];7(6):127. Available from: https://pubmed.ncbi.nlm.nih.gov/36412616/. [CrossRef]
- Kamijo Y, Kanda E, Ishibashi Y, Yoshida M. Sarcopenia and Frailty in PD: Impact on Mortality, Malnutrition, and Inflammation. Perit Dial Int. 2018;38(6):447–54. [CrossRef]
- Tanaka T, Takahashi K, Hirano H, Kikutani T, Watanabe Y, Ohara Y, et al. Oral Frailty as a Risk Factor for Physical Frailty and Mortality in Community-Dwelling Elderly. J Gerontol A Biol Sci Med Sci [Internet]. 2018 Nov 10 [cited 2022 Dec 15];73(12):1661–7. Available from: https://pubmed.ncbi.nlm.nih.gov/29161342/. [CrossRef]
- Hanlon P, Nicholl BI, Jani BD, Lee D, McQueenie R, Mair FS. Frailty and pre-frailty in middle-aged and older adults and its association with multimorbidity and mortality: a prospective analysis of 493 737 UK Biobank participants. Lancet Public Health [Internet]. 2018 Jul 1 [cited 2022 Dec 15];3(7):e323–32. Available from: https://pubmed.ncbi.nlm.nih.gov/29908859/. [CrossRef]
- Dent E, Morley JE, Cruz-Jentoft AJ, Woodhouse L, Rodríguez-Mañas L, Fried LP, et al. Physical Frailty: ICFSR International Clinical Practice Guidelines for Identification and Management. J Nutr Health Aging [Internet]. 2019 Nov 1 [cited 2022 Dec 10];23(9):771. Available from:/pmc/articles/PMC6800406/. [CrossRef]
- Virgini VS, Rodondi N, Cawthon PM, Harrison SL, Hoffman AR, Orwoll ES, et al. Subclinical Thyroid Dysfunction and Frailty Among Older Men. J Clin Endocrinol Metab [Internet]. 2015 Dec 1 [cited 2022 Dec 10];100(12):4524–32. Available from: https://pubmed.ncbi.nlm.nih.gov/26495751/. [CrossRef]
- Abdel-Rahman EM, Mansour W, Holley JL. Hypothesis: Thyroid Hormone Abnormalities and Frailty in Elderly Patients with Chronic Kidney Disease: A Hypothesis. Semin Dial [Internet]. 2010 May 1 [cited 2022 Dec 10];23(3):317–23. Available from: https://onlinelibrary-wiley-com.sheba.idm.oclc.org/doi/full/10.1111/j.1525-139X.2010.00736.x. [CrossRef]
- Segev A, Itelman E, Avaky C, Negru L, Shenhav-Saltzman G, Grupper A, et al. Low ALT Levels Associated with Poor Outcomes in 8700 Hospitalized Heart Failure Patients. J Clin Med. 2020 Sep 30;9(10). [CrossRef]
- Vespasiani-Gentilucci U, de Vincentis A, Ferrucci L, Bandinelli S, Antonelli Incalzi R, Picardi A. Low Alanine Aminotransferase Levels in the Elderly Population: Frailty, Disability, Sarcopenia, and Reduced Survival. J Gerontol A Biol Sci Med Sci. 2018 Jun 14;73(7):925–30. [CrossRef]
- Ramati E, Israel A, Tal Kessler, Petz-Sinuani N, Sela BA, Idan Goren, et al. [Low ALT activity amongst patients hospitalized in internal medicine wards is a widespread phenomenon associated with low vitamin B6 levels in their blood]. Harefuah. 2015 Feb;154(2):89–93, 137.
- Weber Y, Epstein D, Miller A, Segal G, Berger G. Association of Low Alanine Aminotransferase Values with Extubation Failure in Adult Critically Ill Patients: A Retrospective Cohort Study. J Clin Med. 2021 Jul 25;10(15). [CrossRef]
- Portal D, Melamed G, Segal G, Itelman E. Sarcopenia as Manifested by L3SMI Is Associated with Increased Long-Term Mortality amongst Internal Medicine Patients-A Prospective Cohort Study. J Clin Med. 2022 Jun 17;11(12). [CrossRef]
- Irina G, Refaela C, Adi B, Avia D, Liron H, Chen A, et al. Low Blood ALT Activity and High FRAIL Questionnaire Scores Correlate with Increased Mortality and with Each Other. A Prospective Study in the Internal Medicine Department. J Clin Med. 2018 Oct 25;7(11). [CrossRef]
- Anani S, Goldhaber G, Brom A, Lasman N, Turpashvili N, Shenhav-Saltzman G, et al. Frailty and Sarcopenia Assessment upon HospitalAdmission to Internal Medicine Predicts Length ofHospital Stay and Re-Admission: A ProspectiveStudy of 980 Patients. J Clin Med. 2020 Aug 17;9(8).
- Lasman N, Shalom M, Turpashvili N, Goldhaber G, Lifshitz Y, Leibowitz E, et al. Baseline low ALT activity is associated with increased long-term mortality after COPD exacerbations. BMC Pulm Med. 2020 May 11;20(1):133. [CrossRef]
- Segev A, Itelman E, Beigel R, Segal G, Chernomordik F, Matetzky S, et al. Low ALT levels are associated with poor outcomes in acute coronary syndrome patients in the intensive cardiac care unit. J Cardiol. 2022 Mar;79(3):385–90. [CrossRef]
- Segev A, Itelman E, Avaky C, Negru L, Shenhav-Saltzman G, Grupper A, et al. Low ALT Levels Associated with Poor Outcomes in 8700 Hospitalized Heart Failure Patients. J Clin Med. 2020 Sep 30;9(10). [CrossRef]
- Saito Y, Okumura Y, Nagashima K, Fukamachi D, Yokoyama K, Matsumoto N, et al. Low alanine aminotransferase levels are independently associated with mortality risk in patients with atrial fibrillation. Sci Rep. 2022 Jul 16;12(1):12183. [CrossRef]
- Mendoza A, Hollenberg AN. New Insights into Thyroid Hormone Action. Pharmacol Ther [Internet]. 2017 May 1 [cited 2024 Feb 3];173:135. Available from:/pmc/articles/PMC5407910/. [CrossRef]
- Peeters, RP. Thyroid Function and Longevity: New Insights into an Old Dilemma. 2009 [cited 2024 Feb 3]; Available from: https://academic.oup.com/jcem/article/94/12/4658/2596319. [CrossRef]
- Rodondi N, den Elzen WPJ, Bauer DC, Cappola AR, Razvi S, Walsh JP, et al. Subclinical hypothyroidism and the risk of coronary heart disease and mortality. JAMA. 2010 Sep 22;304(12):1365–74. [CrossRef]
- Inoue K, Ritz B, Brent GA, Ebrahimi R, Rhee CM, Leung AM. Association of Subclinical Hypothyroidism and Cardiovascular Disease With Mortality. JAMA Netw Open. 2020 Feb 5;3(2):e1920745. [CrossRef]
- Kalra S, Unnikrishnan A, Sahay R. The global burden of thyroid disease. Thyroid Research and Practice [Internet]. 2013 [cited 2024 Jan 29];10(3):89. Available from: https://journals.lww.com/trap/fulltext/2013/10030/the_global_burden_of_thyroid_disease.1.aspx25.
- Ettleson MD, Papaleontiou M. Evaluating health outcomes in the treatment of hypothyroidism. Front Endocrinol (Lausanne) [Internet]. 2022 Oct 18 [cited 2024 Jan 29];13. Available from: https://pubmed.ncbi.nlm.nih.gov/36329885/. [CrossRef]
- Lee YJ, Kim MH, Lim DJ, Lee JM, Chang SA, Lee J. Exploring the Association between Thyroid Function and Frailty: Insights from Representative Korean Data. Endocrinol Metab (Seoul) [Internet]. 2023 Dec 31 [cited 2024 Jan 29];38(6):729–38. Available from: https://pubmed.ncbi.nlm.nih.gov/37915301/. [CrossRef]
- Vespasiani-Gentilucci U, De Vincentis A, Ferrucci L, Bandinelli S, Antonelli Incalzi R, Picardi A. Low Alanine Aminotransferase Levels in the Elderly Population: Frailty, Disability, Sarcopenia, and Reduced Survival. J Gerontol A Biol Sci Med Sci [Internet]. 2018 Jun 14 [cited 2023 Apr 2];73(7):925–30. Available from: https://pubmed.ncbi.nlm.nih.gov/28633440/. [CrossRef]
- Uliel N, Segal G, Perri A, Turpashvili N, Kassif Lerner R, Itelman E. Low ALT, a marker of sarcopenia and frailty, is associated with shortened survival amongst myelodysplastic syndrome patients: A retrospective study. Medicine. 2023 Apr 25;102(17):e33659. [CrossRef]
- Chung SM, Moon JS, Yoon JS, Won KC, Lee HW. Low alanine aminotransferase levels predict low muscle strength in older patients with diabetes: A nationwide cross-sectional study in Korea. Geriatr Gerontol Int. 2020 Apr;20(4):271–6. [CrossRef]
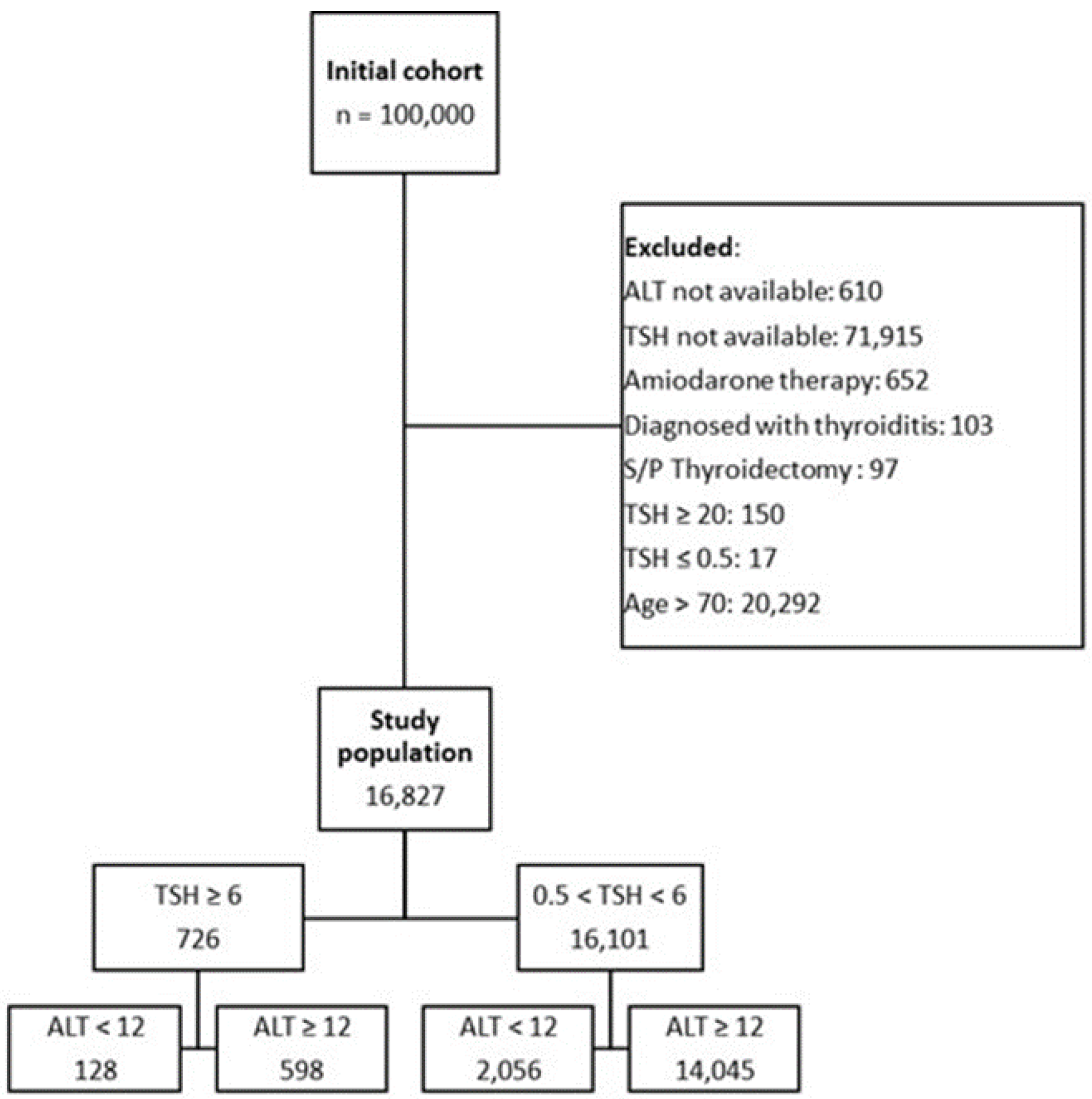
| Total cohort N = 16,827 |
ALT ≥ 12, TSH < 6 N = 14,045 |
ALT < 12, TSH < 6 N = 2,056 |
ALT ≥ 12, TSH ≥ 6 N = 598 |
ALT < 12, TSH ≥ 6 N = 128 |
P value | |
|---|---|---|---|---|---|---|
| Demographics | ||||||
| Age (years) (IQR) | 56.9 (46.5-64) |
57 (47-63.9) |
55 (41.8-63.8) |
59 (50.3-64.8) |
56.2 (45.5-64.3) |
<0.001 |
| Male, N (%) | 9,507 (56.5) | 8,394 (59.8) |
821 (39) |
249 (41.6) |
43 (33.6) |
<0.001 |
| Comorbidities | ||||||
| Hypertension, N (%) | 5,479 (32.6) | 4,687 (33.4) |
586 (28.5) |
172 (28.8) |
34 (26.6) |
<0.001 |
|
IHD, N (%) |
3,325 (19.8) | 2,933 (20.9) |
272 (13.2) |
108 (18.1) |
12 (9.4) |
<0.001 |
| Dyslipidemia, N (%) | 2,417 (14.4) | 2,125 (15.1) |
208 (10.1) |
73 (12.2) |
11 (8.6) |
<0.001 |
|
COPD, N (%) |
831 (4.9) |
675 (4.8) |
114 (5.5) |
30 (5) |
12 (9.4) |
0.057 |
|
CKD, N (%) |
1,051 (6.4) |
775 (5.6) |
194 (9.7) |
55 (9.4) |
27 (21.4) |
<0.001 |
| Diabetes, N (%) | 3,589 (21.3) | 3,004 (21.4) |
433 (21.1) |
124 (20.7) |
28 (21.9) |
0.96 |
| Malignancy, N (%) | 2,966 (17.6) | 2,386 (17) |
405 (19.7) |
139 (23.2) |
36 (28.1) |
<0.001 |
|
Stroke, N (%) |
2,639 (15.7) | 2,233 (15.9) |
313 (15.2) |
80 (13.4) |
13 (10.2) |
0.1 |
|
Dementia, N (%) |
255 (1.5) |
202 (1.4) |
44 (2.1) |
6 (1) |
3 (2.3) |
0.056 |
| Laboratory parameters | ||||||
|
Albumin (g/dl); (Mean ± SD) |
3.8 ± 0.5 | 3.8 ± 0.5 | 3.6 ± 0.5 | 3.6 ± 0.6 | 3.4 ± 0.6 | <0.001 |
| Creatinine (mg/dl); (Mean ± SD) | 0.9 ± 0.6 | 0.94 ± 0.5 | 1 ± 1 | 1 ± 0.7 | 1.3 ± 1.1 | <0.001 |
| Patients grouping | Months | P value |
|---|---|---|
| Reference group(ALT > 12, TSH < 6) | 54.19 ± 0.14 | < 0.001 |
| Group 1(ALT > 12, TSH > 6) | 47.35 ± 0.94 | |
| Group 2(ALT < 12, TSH < 6) | 51.48 ± 0.42 | |
| Group 3(ALT < 12, TSH > 6) | 39.32 ± 2.26 | |
| Whole cohort | 53.5 ± 0.13 |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




