Submitted:
30 August 2024
Posted:
02 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Study Subjects
Study Protocol
- Test product: Brightseed® Bio Gut Fiber (BGF), (Brightseed Inc, South San Francisco, CA, USA), a proprietary hemp hull fiber with bioactives at a dose providing 2.8 g fiber/day.
- Comparator product: Inulin (Beneo, Mannheim, Germany), dose providing 2.8 g fiber/day. Maltodextrin was added to 4 g.
- Placebo: Maltodextrin (Grain Processing Corporation, Washington, IN, USA), 4 g dose to approximately match total test and comparator product administered
3. Results
3.1. Subject Characteristics and Retention
| Statistic | mITT | PP | ||||
|---|---|---|---|---|---|---|
| Bio Gut Fiber | Inulin | Placebo | Bio Gut Fiber | Inulin | Placebo | |
| N | 198 | 188 | 193 | 38 | 30 | 46 |
| Females, n (%) |
145 (73%) |
126 (67%) |
137 (71%) |
30 (79%) |
18 (60%) |
33 (72%) |
| White, n (%) |
163 (82%) |
145 (77%) | 165 (85%) |
32 (84%) |
26 (87%) |
36 (78%) |
| Mean Age (years) (min, max) |
44.4 (19, 77) |
43.7 (21, 72) |
44.2 (21, 78) |
46.7 (24, 71) |
46.3 (21, 72) |
46.7 (26, 72) |
| BMI Category Underweight Normal Weight Overweight Obese |
4 (2%) 52 (26%) 63 (32%) 79 (40%) |
2 (1%) 52 (28%) 56 (30%) 78 (41%) |
3 (2%) 52 (28%) 57 (30%) 79 (41%) |
1 (3%) 9 (24%) 11 (29%) 17 (45%) |
2 (7%) 8 (27%) 7 (23%) 13 (43%) |
0 17 (37%) 14 (30%) 15 (33%) |
3.2. Statistical Analysis
3.3. Survey Results
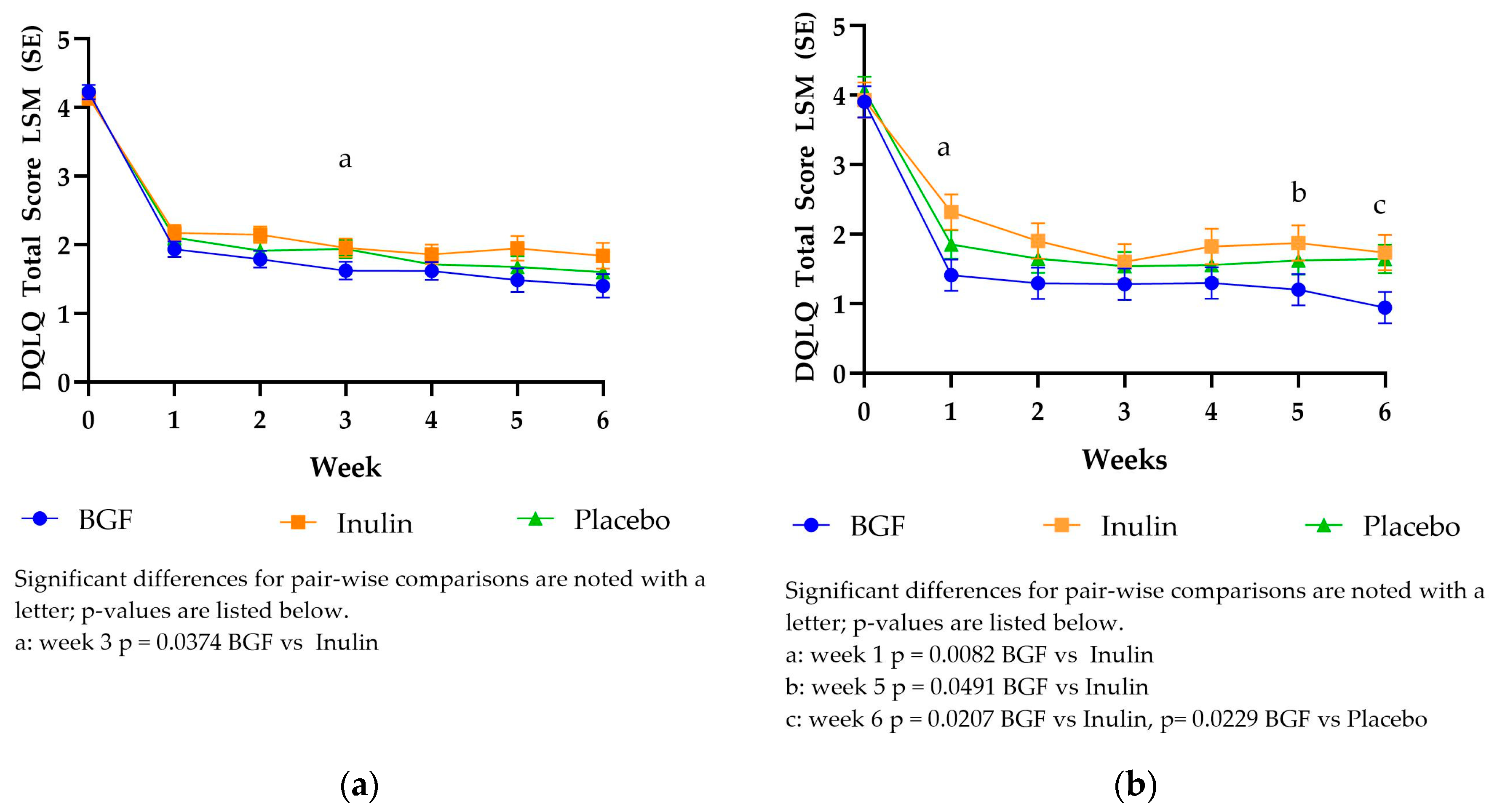
3.3.1. Digestion Associated Quality of Life Questionnaire (DQLQ) Scores
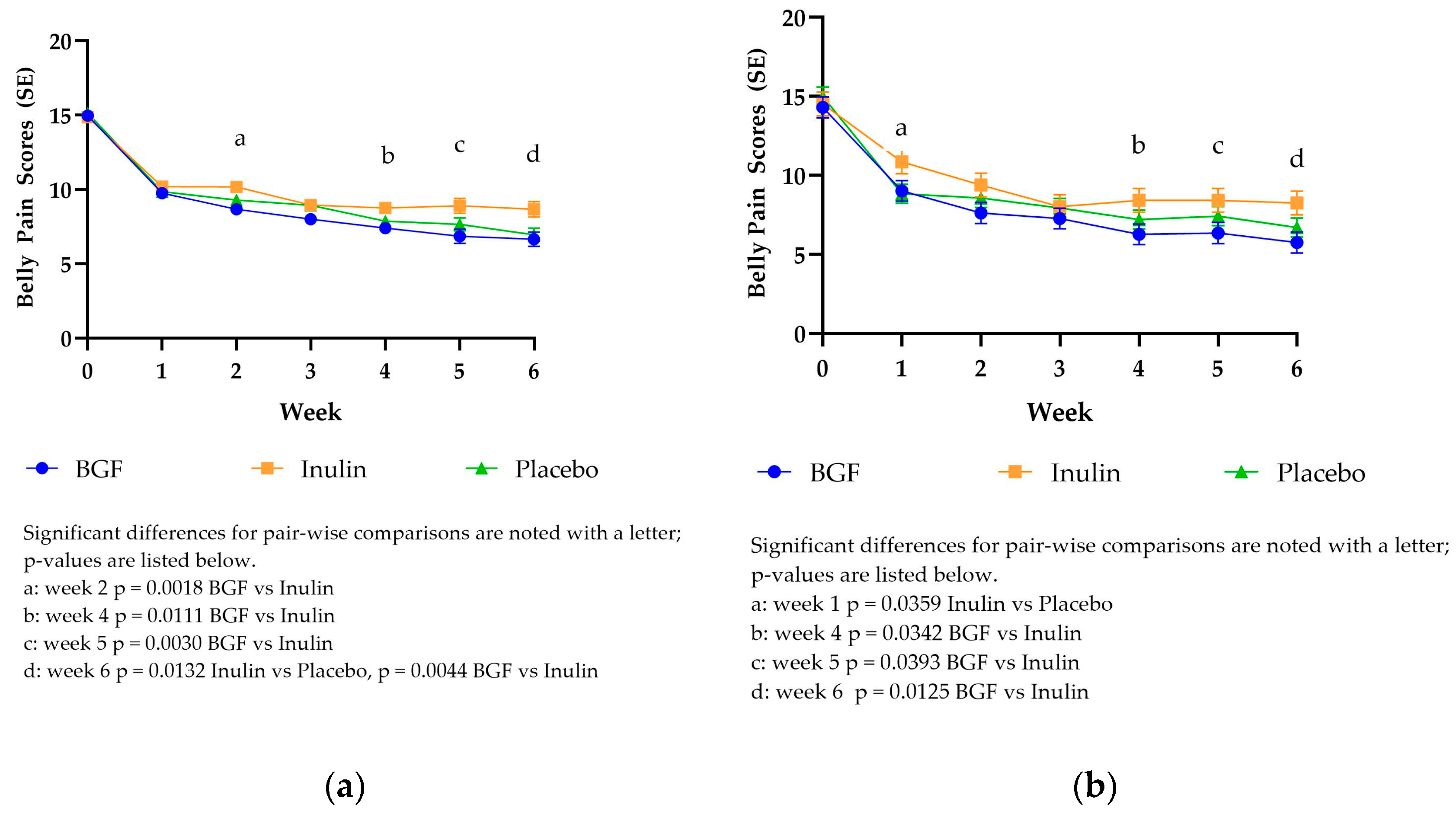
3.3.2. PROMIS Belly Pain Scores
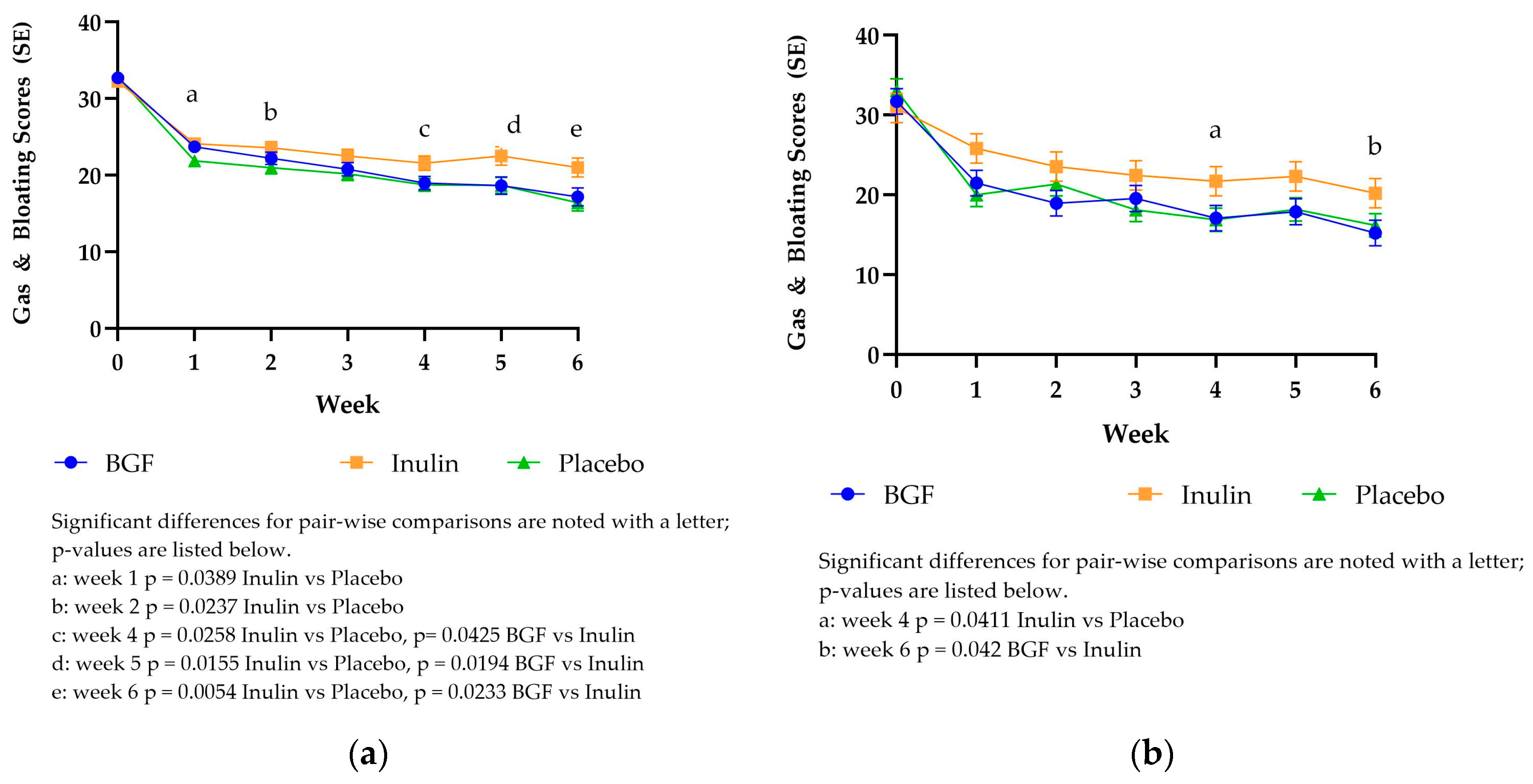
3.3.3. PROMIS Gas & Bloating Scores
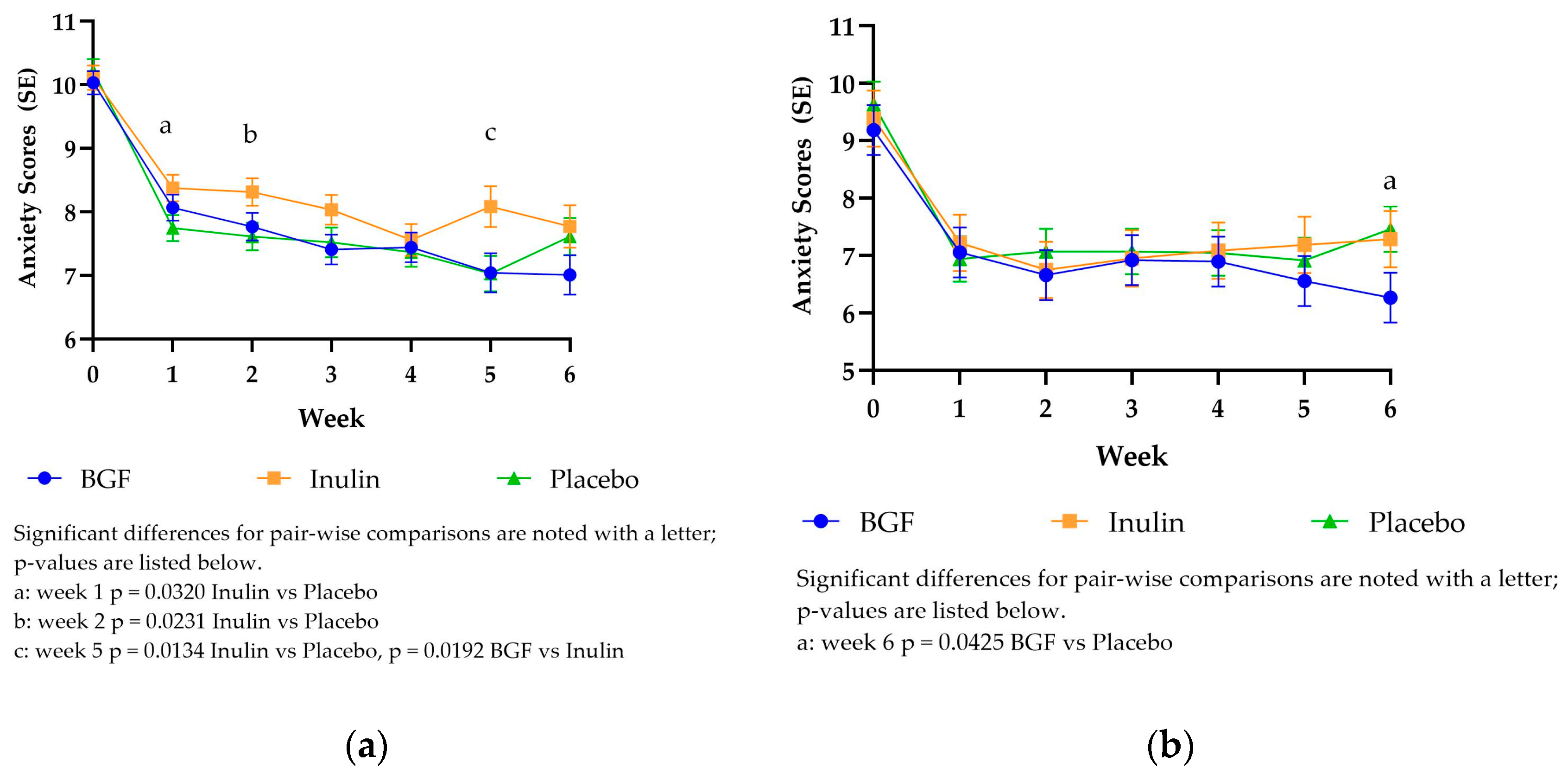
3.3.4. PROMIS Anxiety Scores
3.3.5. Side Effects
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Timm, M.; Offringa, L.C.; Van Klinken, B.J.-W.; Slavin, J. Beyond Insoluble Dietary Fiber: Bioactive Compounds in Plant Foods. Nutrients 2023, 15. [Google Scholar] [CrossRef] [PubMed]
- Lattimer, J.M.; Haub, M.D. Effects of Dietary Fiber and Its Components on Metabolic Health. Nutrients 2010, 2, 1266–1289. [Google Scholar] [CrossRef]
- Dahl, W.J.; Stewart, M.L. Position of the Academy of Nutrition and Dietetics: Health Implications of Dietary Fiber. J Acad Nutr Diet 2015, 115, 1861–1870. [Google Scholar] [CrossRef] [PubMed]
- van Klinken, B.J.-W.; Stewart, M.L.; Kalgaonkar, S.; Chae, L. Health-Promoting Opportunities of Hemp Hull: The Potential of Bioactive Compounds. J Diet Suppl 2024, 1–15. [Google Scholar] [CrossRef]
- Bolster, D.; Chae, L.; van Klinken, J.-W.; Kalgaonkar, S. Impact of Selected Novel Plant Bioactives on Improvement of Impaired Gut Barrier Function Using Human Primary Cell Intestinal Epithelium. Journal of Food Bioactives 2022, 20, 11–16. [Google Scholar] [CrossRef]
- Flores Martinez, K.E.; Bloszies, C.S.; Bolino, M.J.; Henrick, B.M.; Frese, S.A. Hemp Hull Fiber and Two Constituent Compounds, N-Trans-Caffeoyltyramine and N-Trans-Feruloyltyramine, Shape the Human Gut Microbiome in Vitro. Food Chemistry: X 2024, 23, 101611. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.-H.; Veeriah, V.; Levine, F. Liver Fat Storage Is Controlled by HNF4α through Induction of Lipophagy and Is Reversed by a Potent HNF4α Agonist. Cell Death Dis 2021, 12, 603. [Google Scholar] [CrossRef] [PubMed]
- Veeriah, V.; Lee, S.-H.; Levine, F. Long-Term Oral Administration of an HNF4α Agonist Prevents Weight Gain and Hepatic Steatosis by Promoting Increased Mitochondrial Mass and Function. Cell Death Dis 2022, 13, 89. [Google Scholar] [CrossRef]
- Carlson, J.L.; Erickson, J.M.; Lloyd, B.B.; Slavin, J.L. Health Effects and Sources of Prebiotic Dietary Fiber. Curr Dev Nutr 2018, 2, nzy005. [Google Scholar] [CrossRef]
- Mysonhimer, A.R.; Holscher, H.D. Gastrointestinal Effects and Tolerance of Nondigestible Carbohydrate Consumption. Adv Nutr 2022, 13, 2237–2276. [Google Scholar] [CrossRef]
- Grabitske, H.A.; Slavin, J.L. Low-Digestible Carbohydrates in Practice. J Am Diet Assoc 2008, 108, 1677–1681. [Google Scholar] [CrossRef] [PubMed]
- Gibson, P.R.; Shepherd, S.J. Evidence-Based Dietary Management of Functional Gastrointestinal Symptoms: The FODMAP Approach. J Gastroenterol Hepatol 2010, 25, 252–258. [Google Scholar] [CrossRef]
- Atzler, J.J.; Sahin, A.W.; Gallagher, E.; Zannini, E.; Arendt, E.K. Characteristics and Properties of Fibres Suitable for a Low FODMAP Diet- an Overview. Trends in Food Science & Technology 2021, 112, 823–836. [Google Scholar] [CrossRef]
- Dietary Reference Intakes: The Essential Guide to Nutrient Requirements |The National Academies Press. Available online: https://nap.nationalacademies.org/download/11537 (accessed on 15 May 2024).
- Wong, J.M.W.; de Souza, R.; Kendall, C.W.C.; Emam, A.; Jenkins, D.J.A. Colonic Health: Fermentation and Short Chain Fatty Acids. J Clin Gastroenterol 2006, 40, 235–243. [Google Scholar] [CrossRef]
- Saleska, J.L.; Bryant, C.; Kolobaric, A.; D’Adamo, C.R.; Colwell, C.S.; Loewy, D.; Chen, J.; Pauli, E.K. The Safety and Comparative Effectiveness of Non-Psychoactive Cannabinoid Formulations for the Improvement of Sleep: A Double-Blinded, Randomized Controlled Trial. J Am Nutr Assoc 2024, 43, 1–11. [Google Scholar] [CrossRef]
- Kolobaric, A.; Hewlings, S.J.; Bryant, C.; Colwell, C.S.; R. D’Adamo, C.; Rosner, B.; Chen, J.; Pauli, E.K. A Randomized, Double-Blind, Placebo-Controlled Decentralized Trial to Assess Sleep, Health Outcomes, and Overall Well-Being in Healthy Adults Reporting Disturbed Sleep, Taking a Melatonin-Free Supplement. Nutrients 2023, 15, 3788. [Google Scholar] [CrossRef] [PubMed]
- Stewart, M.L.; Timm, D.A.; Slavin, J.L. Fructooligosaccharides Exhibit More Rapid Fermentation than Long-Chain Inulin in an in Vitro Fermentation System. Nutr Res 2008, 28, 329–334. [Google Scholar] [CrossRef]
- Grabitske, H.A.; Slavin, J.L. Gastrointestinal Effects of Low-Digestible Carbohydrates. Crit Rev Food Sci Nutr 2009, 49, 327–360. [Google Scholar] [CrossRef]
- Mutuyemungu, E.; Singh, M.; Liu, S.; Rose, D.J. Intestinal Gas Production by the Gut Microbiota: A Review. Journal of Functional Foods 2023, 100, 105367. [Google Scholar] [CrossRef]
- Dietary Guidelines Advisory Committee Scientific Report of the 2020 Dietary Guidelines Advisory Committee: Advisory Report to the Secretary of Agriculture and the Secretary of Health and Human Services; U.S. Department of Agriculture, Agricultural Research Service, 2020; p. 835.
- Eswaran, S.; Muir, J.; Chey, W.D. Fiber and Functional Gastrointestinal Disorders. Am J Gastroenterol 2013, 108, 718–727. [Google Scholar] [CrossRef]
- Barrett, J.S. Extending Our Knowledge of Fermentable, Short-Chain Carbohydrates for Managing Gastrointestinal Symptoms. Nutr Clin Pract 2013, 28, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Musial, F.; Klosterhalfen, S.; Enck, P. Placebo Responses in Patients with Gastrointestinal Disorders. World J Gastroenterol 2007, 13, 3425–3429. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.-L.; Chang, F.-Y. Placebo Effect in Patients with Irritable Bowel Syndrome. J Gastroenterol Hepatol 2011, 26 Suppl 3, 116–118. [Google Scholar] [CrossRef]
- Dahne, J.; Hawk, L.W.J. Health Equity and Decentralized Trials. JAMA 2023, 329, 2013–2014. [Google Scholar] [CrossRef] [PubMed]
| Inclusion Criteria | Exclusion Criteria |
|---|---|
| At least 21 years of age Any ethnicity, race, or gender/gender identity Resides in United States Currently experiences bloating or indigestion and seeks relief from either or both Expresses willingness to consume a blinded study product for the duration of the study |
Pregnant, trying to become pregnant, breastfeeding Unable to provide valid US phone number and shipping address Did not meet health screening criteria Reported liver or kidney disease Current or recent (last 3 months) chemotherapy or immunotherapy Currently taking medications that could potentially interact with study products, including anticoagulants, corticosteroids, oral anti-infectives, antipsychotics, or monoamine oxidase inhibitors Reported diagnosis of heart disease Consumes more than 3 alcoholic drinks per day Unable to read or understand English Concurrent enrollment in a clinical trial |
| Measure | Description | Scoring interpretation |
|---|---|---|
| Digestion Associated Quality of Life Questionnaire (DQLQ) | 9-item measure assessing digestion-associated quality of life (i.e how bothersome are GI-related symptoms to the individual) | Scoring from 0 to 9, with higher scores indicating more bothersome GI symptoms/lower digestion-associated quality of life |
| PROMIS Belly Pain 5a | 5-item measure assessing abdominal pain in the past 7 days | Scoring from 5 to 25 with higher scores translating to worse abdominal pain |
| PROMIS GI Gas and Bloating 13a | 13- item measure assessing gas/bloating in the past 7 days | Scoring from 13 to 45, with higher scores translating to worse gas/bloating |
| PROMIS Anxiety 4a | 4-item measure assessing frequency of depression symptoms in the past 7 days | Scoring from 4 to 20, with higher scores translating to greater anxiety |
| Statistic | BGF | Inulin | Placebo |
|---|---|---|---|
| N | 316 | 317 | 315 |
| Females, n (%) |
212 (67%) |
215 (68%) |
211 (67%) |
| White, n (%) |
260 (82%) |
240 (76%) |
262 (83%) |
| Mean Age (years) (min, max) |
43.7 (19, 77) |
42.8 (20, 72) |
42.6 (20, 78) |
| BMI Category, n (%) Underweight Normal Weight Overweight Obese |
7 (2%) 73 (23%) 98 (31%) 138 (44%) |
3 (1%) 86 (27%) 93 (29%) 135 (43%) |
3 (1%) 83 (26%) 99 (32%) 130 (41%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




