Submitted:
31 August 2024
Posted:
02 September 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
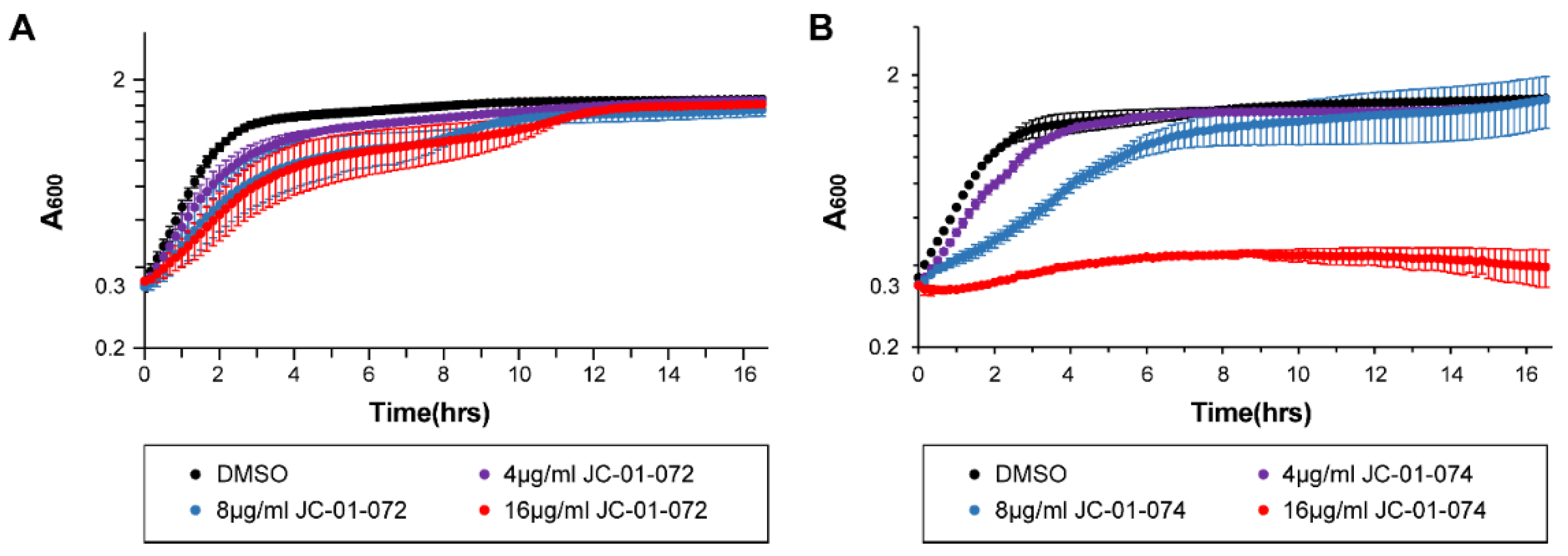
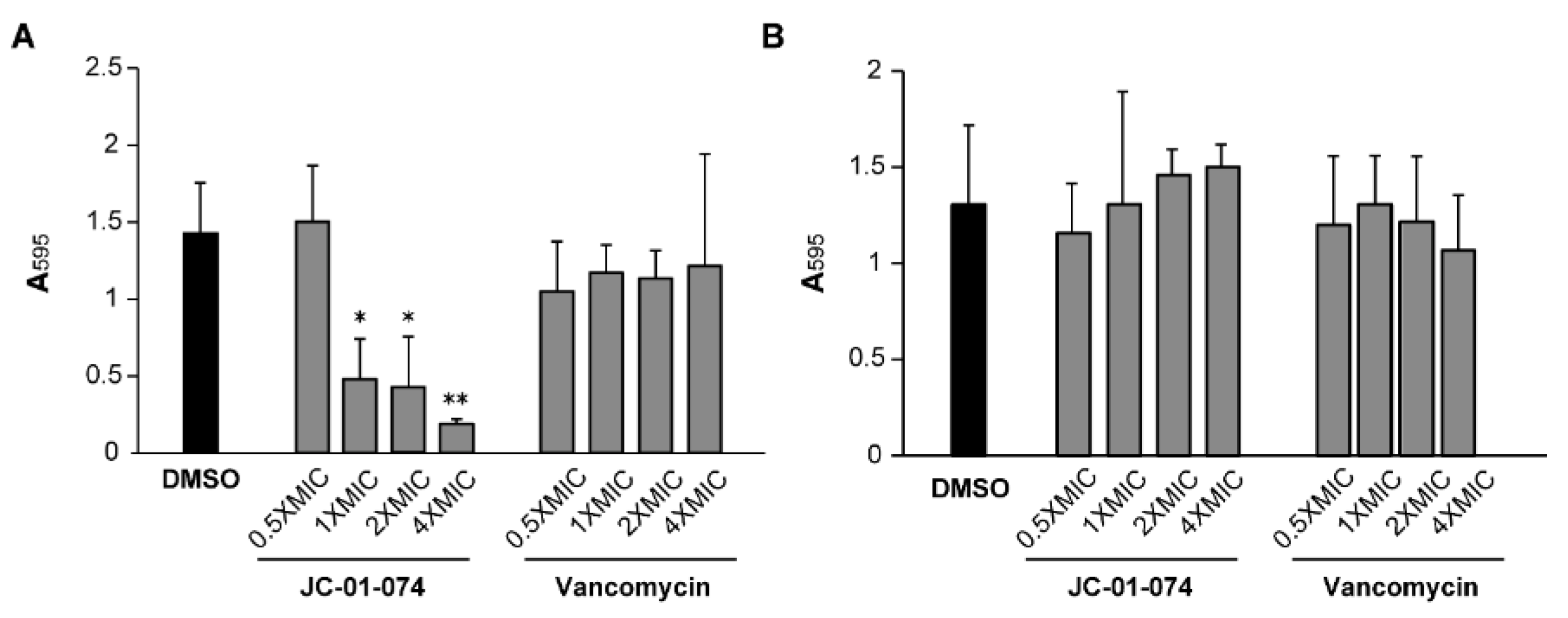
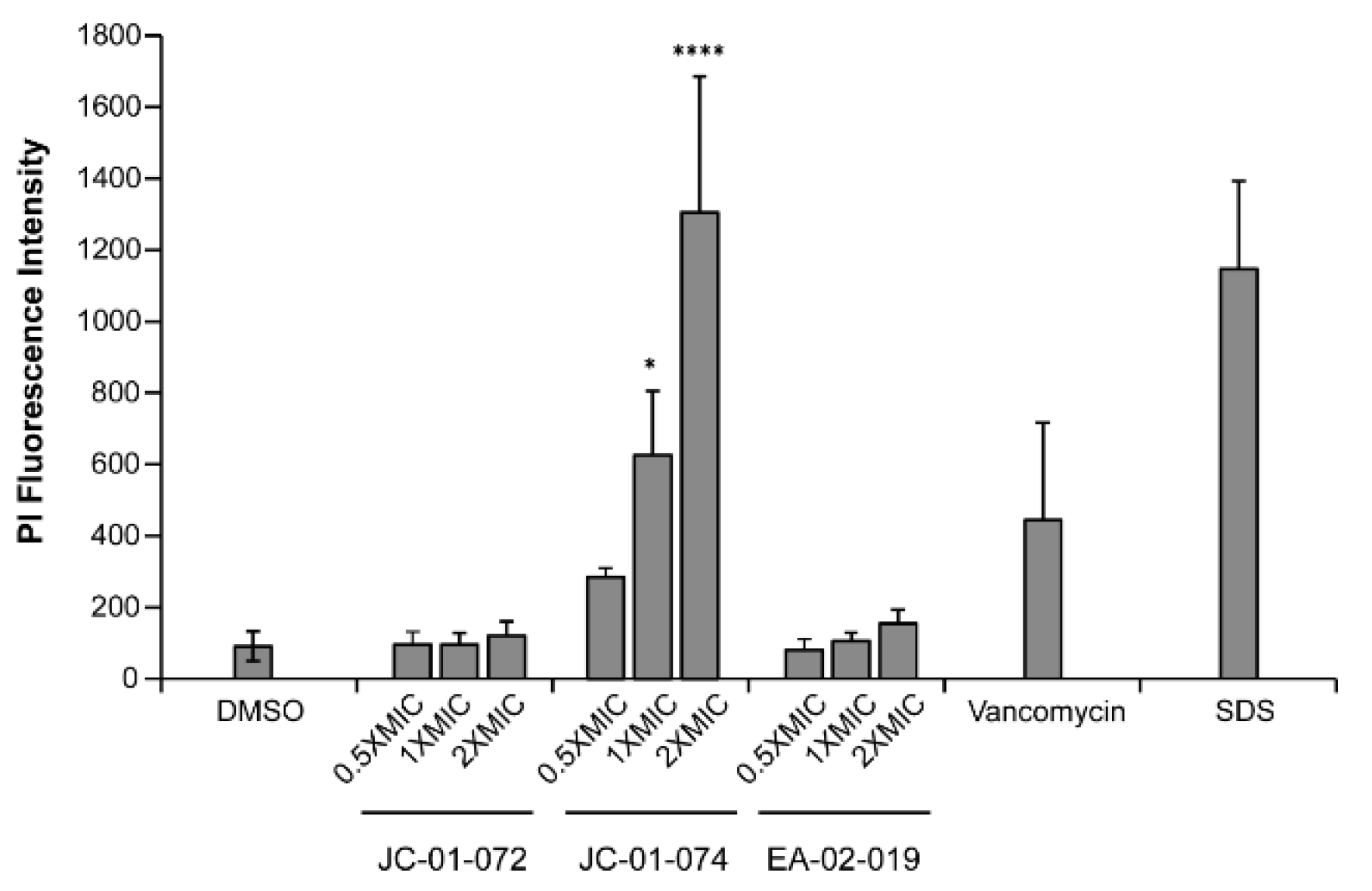
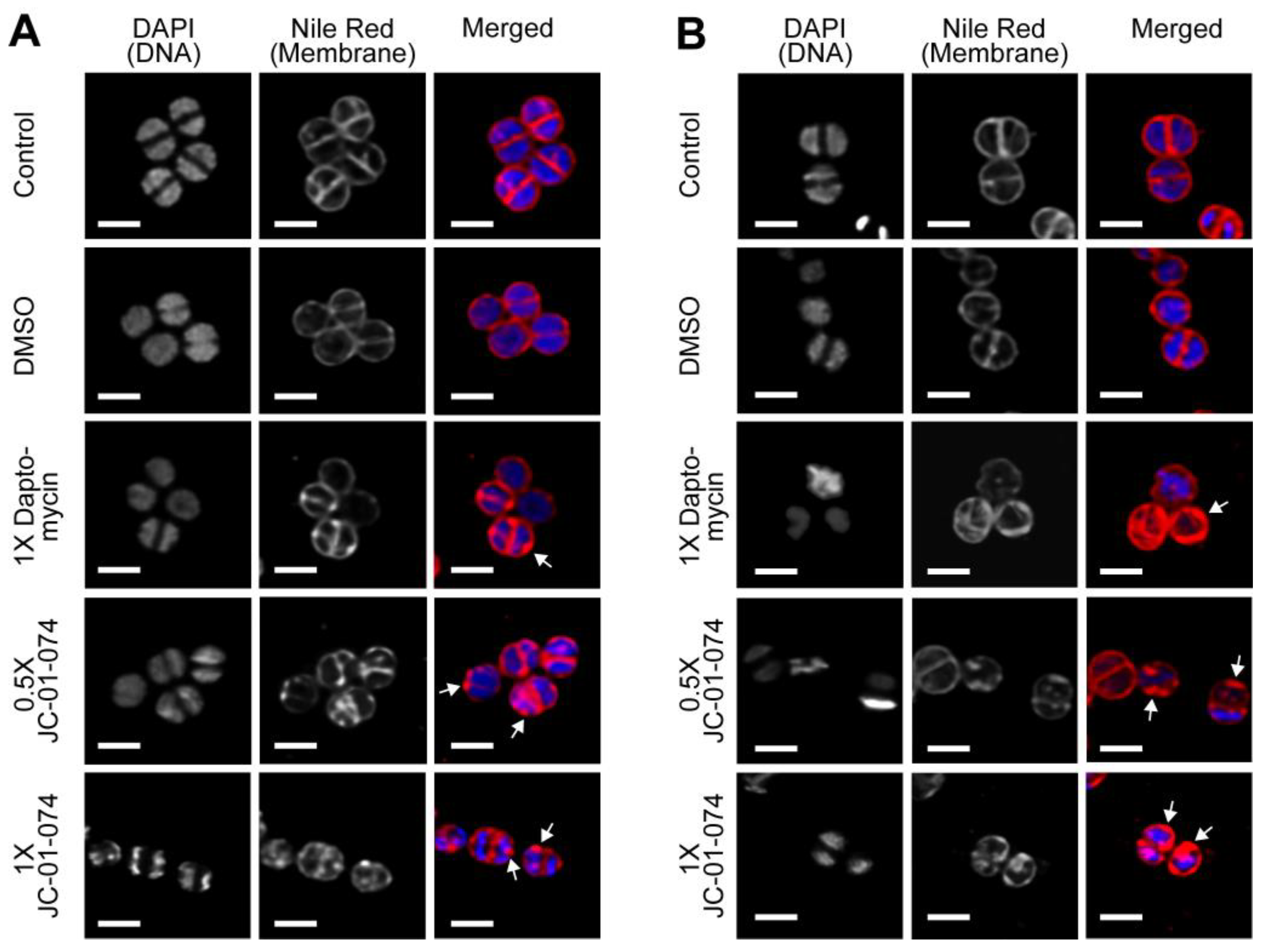
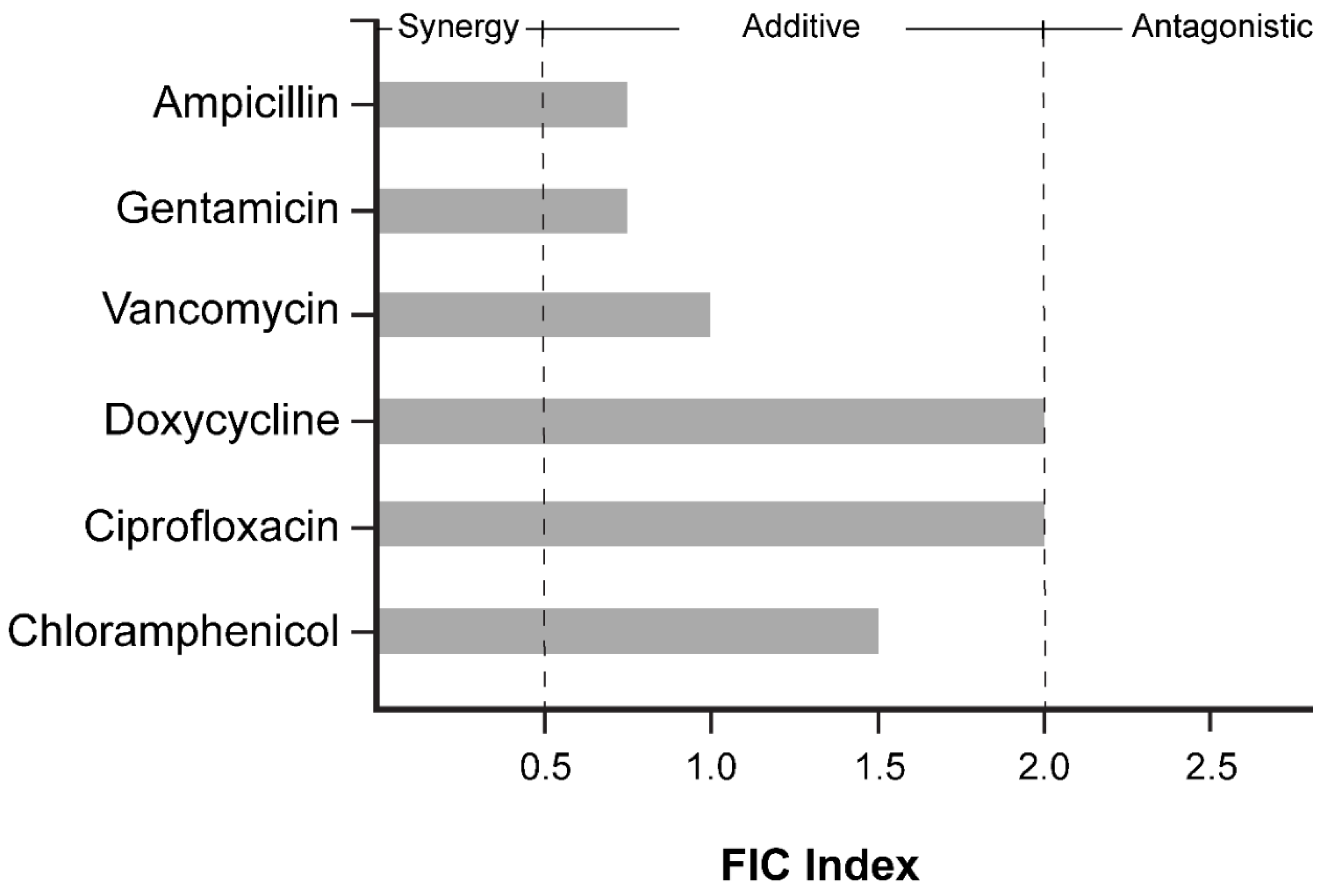
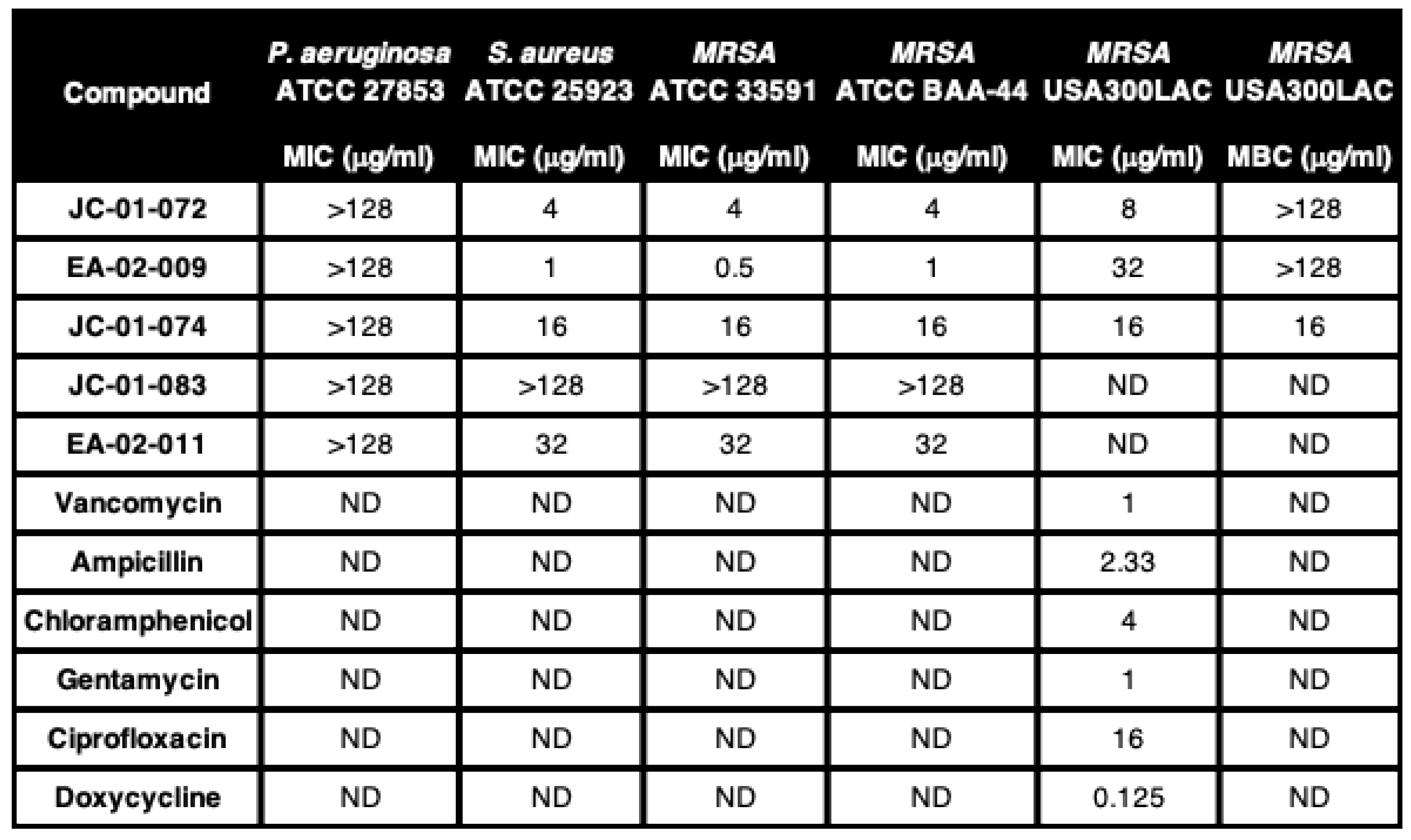
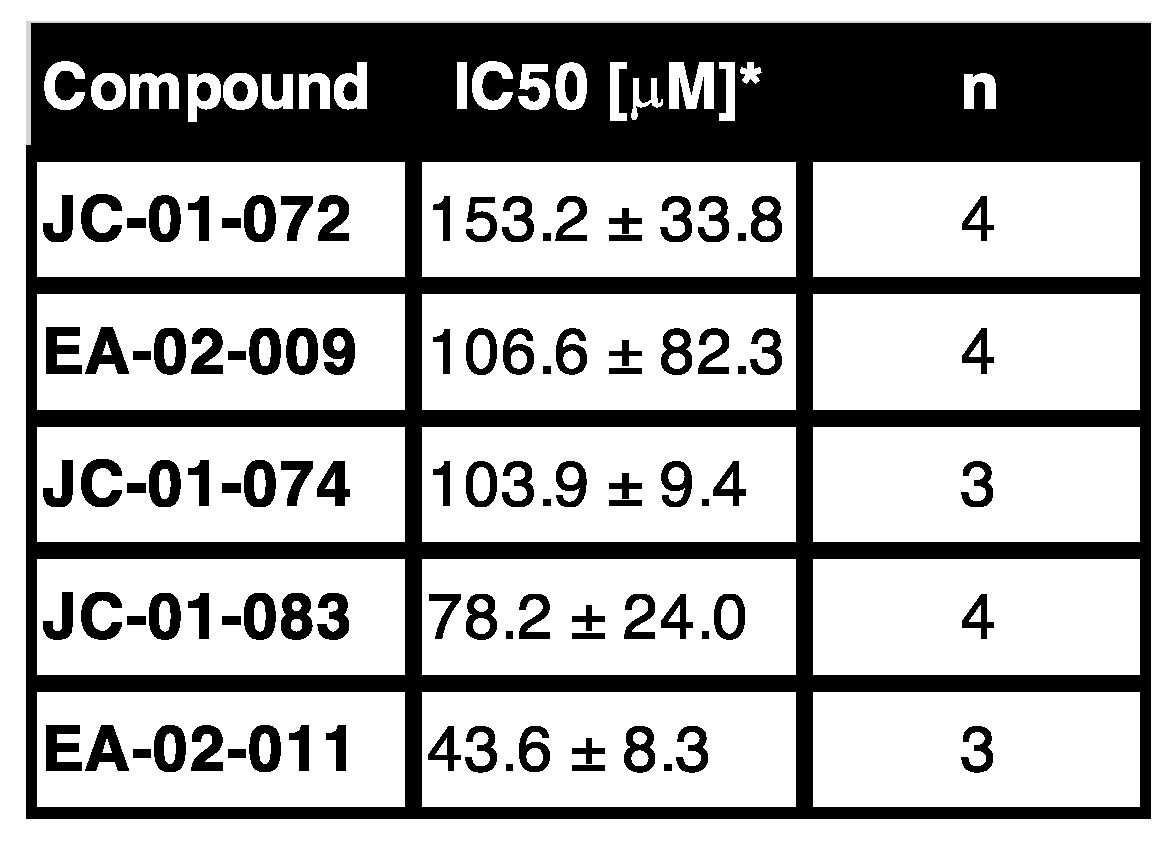
2. Results
3. Discussion
4. Materials and Methods
Supplementary Materials
Author Contributions
Funding
Acknowledgements
Conflicts of Interest
References
- Vaishampayan, A.; Grohmann, E. Antimicrobials Functioning through ROS-Mediated Mechanisms: Current Insights. Microorganisms 2022, 10, 61. [Google Scholar] [CrossRef] [PubMed]
- Shang, D.; Liu, Y.; Jiang, F.; Ji, F.; Wang, H.; Han, X. Synergistic Antibacterial Activity of Designed Trp-Containing Antibacterial Peptides in Combination With Antibiotics Against Multidrug-Resistant Staphylococcus Epidermidis. Front Microbiol 2019, 10, 2719. [Google Scholar] [CrossRef] [PubMed]
- Church, D.; Elsayed, S.; Reid, O.; Winston, B.; Lindsay, R. Burn Wound Infections. Clinical Microbiology Reviews 2006, 19, 403–434. [Google Scholar] [CrossRef]
- Santajit, S.; Indrawattana, N. Mechanisms of Antimicrobial Resistance in ESKAPE Pathogens. Biomed Res Int 2016, 2016, 2475067. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.; Zou, G.; Pan, W.; Fricke, N.; Faizi, H.A.; Kim, S.M.; Khader, R.; Li, S.; Lee, K.; Escorba, I.; et al. The Neutrally Charged Diarylurea Compound PQ401 Kills Antibiotic-Resistant and Antibiotic-Tolerant Staphylococcus Aureus. mBio 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Tong, S.Y.C.; Davis, J.S.; Eichenberger, E.; Holland, T.L.; Fowler, V.G. Staphylococcus Aureus Infections: Epidemiology, Pathophysiology, Clinical Manifestations, and Management. Clinical Microbiology Reviews 2015, 28, 603–661. [Google Scholar] [CrossRef]
- Gutiérrez, D.; Fernández, L.; Rodríguez, A.; García, P. Are Phage Lytic Proteins the Secret Weapon To Kill Staphylococcus Aureus? mBio 2018, 9. [Google Scholar] [CrossRef]
- Foster, T.J. Immune Evasion by Staphylococci. Nat Rev Microbiol 2005, 3, 948–958. [Google Scholar] [CrossRef]
- Chambers, H.F.; DeLeo, F.R. Waves of Resistance: Staphylococcus Aureus in the Antibiotic Era. Nat Rev Microbiol 2009, 7, 629–641. [Google Scholar] [CrossRef]
- Boyle-Vavra, S.; Daum, R.S. Molecular Strategies of Staphylococcus Aureus for Resisting Antibiotics. In Staphylococcus: Genetics and Physiology; Caister Academic Press, 2016; pp. 249–300. ISBN 978-1-910190-49-4. [Google Scholar]
- Pantosti, A.; Venditti, M. What Is MRSA? European Respiratory Journal 2009, 34, 1190–1196. [Google Scholar] [CrossRef]
- Appelbaum, P.C. Microbiology of Antibiotic Resistance in Staphylococcus Aureus. Clinical Infectious Diseases 2007, 45, S165–S170. [Google Scholar] [CrossRef] [PubMed]
- Miller, W.R.; Bayer, A.S.; Arias, C.A. Mechanism of Action and Resistance to Daptomycin in Staphylococcus Aureus and Enterococci. Cold Spring Harb Perspect Med 2016, 6, a026997. [Google Scholar] [CrossRef] [PubMed]
- Alberge, F.; Espinosa, L.; Seduk, F.; Sylvi, L.; Toci, R.; Walburger, A.; Magalon, A. Dynamic Subcellular Localization of a Respiratory Complex Controls Bacterial Respiration. eLife 2015, 4, e05357. [Google Scholar] [CrossRef]
- Toyofuku, M. Bacterial Communication through Membrane Vesicles. Bioscience, Biotechnology, and Biochemistry 2019, 83, 1599–1605. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.E. The ATP Synthase: The Understood, the Uncertain and the Unknown. Biochemical Society Transactions 2013, 41, 1–16. [Google Scholar] [CrossRef]
- Barbarek, S.C.; Shah, R.; Paul, S.; Alvarado, G.; Appala, K.; Phillips, C.; Henderson, E.C.; Strandquist, E.T.; Pokorny, A.; Singh, V.K.; et al. Lipidomics of Homeoviscous Adaptation to Low Temperatures in Staphylococcus Aureus Utilizing Exogenous Straight-Chain Unsaturated Fatty Acids. Journal of Bacteriology 2024, 206, e00187-24. [Google Scholar] [CrossRef]
- Pasupuleti, M.; Schmidtchen, A.; Malmsten, M. Antimicrobial Peptides: Key Components of the Innate Immune System. Crit Rev Biotechnol 2012, 32, 143–171. [Google Scholar] [CrossRef]
- Davies, J.; Davies, D. Origins and Evolution of Antibiotic Resistance. Microbiology and Molecular Biology Reviews 2010, 74, 417–433. [Google Scholar] [CrossRef]
- Harvey, A.L.; Edrada-Ebel, R.; Quinn, R.J. The Re-Emergence of Natural Products for Drug Discovery in the Genomics Era. Nat Rev Drug Discov 2015, 14, 111–129. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs from 1981 to 2014. J Nat Prod 2016, 79, 629–661. [Google Scholar] [CrossRef]
- Porras, G.; Chassagne, F.; Lyles, J.T.; Marquez, L.; Dettweiler, M.; Salam, A.M.; Samarakoon, T.; Shabih, S.; Farrokhi, D.R.; Quave, C.L. Ethnobotany and the Role of Plant Natural Products in Antibiotic Drug Discovery. Chem. Rev. 2021, 121, 3495–3560. [Google Scholar] [CrossRef] [PubMed]
- Atanasov, A.G.; Zotchev, S.B.; Dirsch, V.M.; Supuran, C.T. Natural Products in Drug Discovery: Advances and Opportunities. Nat Rev Drug Discov 2021, 20, 200–216. [Google Scholar] [CrossRef] [PubMed]
- Nicolaou, K.C.; Snyder, S.A. The Essence of Total Synthesis. Proceedings of the National Academy of Sciences 2004, 101, 11929–11936. [Google Scholar] [CrossRef]
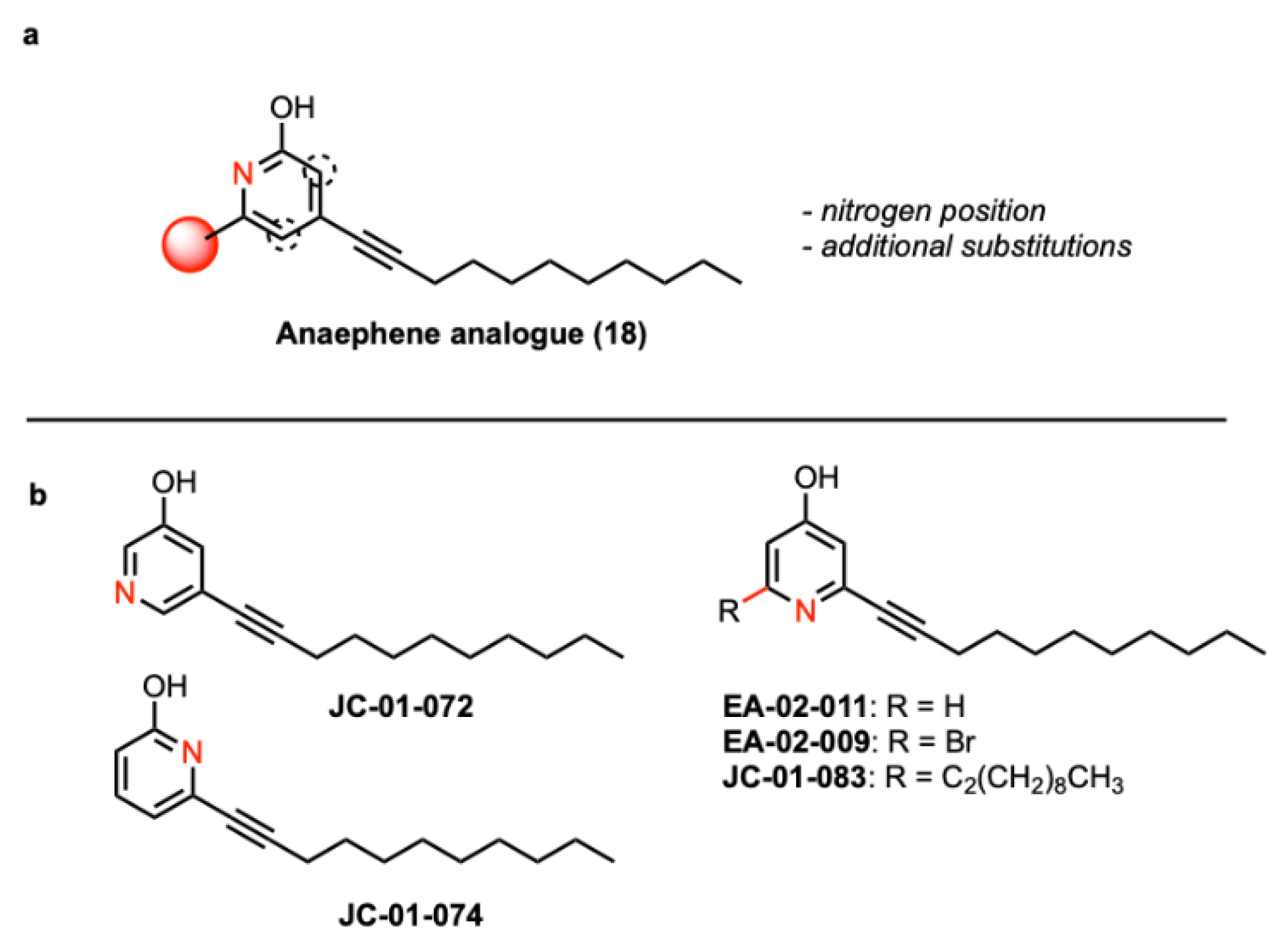
- Kukla, D.L.; Canchola, J.; Mills, J.J. Synthesis of the Cyanobacterial Antibiotics Anaephenes A and B. J Nat Prod 2020, 83, 2036–2040. [Google Scholar] [CrossRef]
- Kukla, D.L.; Canchola, J.; Rosenthal, J.D.; Mills, J.J. Design, Synthesis, and Structure-Activity Relationship Studies of the Anaephene Antibiotics. Chem Biol Drug Des 2021, 98, 295–304. [Google Scholar] [CrossRef]
- Kikuchi, H.; Ito, I.; Takahashi, K.; Ishigaki, H.; Iizumi, K.; Kubohara, Y.; Oshima, Y. Isolation, Synthesis, and Biological Activity of Chlorinated Alkylresorcinols from Dictyostelium Cellular Slime Molds. J. Nat. Prod. 2017, 80, 2716–2722. [Google Scholar] [CrossRef] [PubMed]
- Bissantz, C.; Kuhn, B.; Stahl, M. Corrections to A Medicinal Chemist’s Guide to Molecular Interactions. J. Med. Chem. 2010, 53, 6241–6241. [Google Scholar] [CrossRef]
- Hamada, Y. Role of Pyridines in Medicinal Chemistry and Design of BACE1 Inhibitors Possessing a Pyridine Scaffold. In Pyridine; IntechOpen, 2018; ISBN 978-1-78923-423-7. [Google Scholar]
- M07: Dilution AST for Aerobically Grown Bacteria - CLSI. Available online: https://clsi.org/standards/products/microbiology/documents/m07/ (accessed on 3 November 2023).
- Gutierrez, A.; Jain, S.; Bhargava, P.; Hamblin, M.; Lobritz, M.A.; Collins, J.J. Understanding and Sensitizing Density-Dependent Persistence to Quinolone Antibiotics. Molecular Cell 2017, 68, 1147–1154.e3. [Google Scholar] [CrossRef]
- McCall, I.C.; Shah, N.; Govindan, A.; Baquero, F.; Levin, B.R. Antibiotic Killing of Diversely Generated Populations of Nonreplicating Bacteria. Antimicrobial Agents and Chemotherapy 2019, 63. [Google Scholar] [CrossRef]
- Lobritz, M.A.; Belenky, P.; Porter, C.B.M.; Gutierrez, A.; Yang, J.H.; Schwarz, E.G.; Dwyer, D.J.; Khalil, A.S.; Collins, J.J. Antibiotic Efficacy Is Linked to Bacterial Cellular Respiration. Proc. Natl. Acad. Sci. U.S.A. 2015, 112, 8173–8180. [Google Scholar] [CrossRef]
- Guilhen, C.; Forestier, C.; Balestrino, D. Biofilm Dispersal: Multiple Elaborate Strategies for Dissemination of Bacteria with Unique Properties. Molecular Microbiology 2017, 105, 188–210. [Google Scholar] [CrossRef] [PubMed]
- Schilcher, K.; Horswill, A.R. Staphylococcal Biofilm Development: Structure, Regulation, and Treatment Strategies. Microbiology and Molecular Biology Reviews 2020, 84. [Google Scholar] [CrossRef]
- Arciola, C.R.; An, Y.H.; Campoccia, D.; Donati, M.E.; Montanaro, L. Etiology of Implant Orthopedic Infections: A Survey on 1027 Clinical Isolates. Int J Artif Organs 2005, 28, 1091–1100. [Google Scholar] [CrossRef] [PubMed]
- Otto, M. Staphylococcal Biofilms. Curr Top Microbiol Immunol 2008, 322, 207–228. [Google Scholar] [PubMed]
- MacNair, C.R.; Brown, E.D. Outer Membrane Disruption Overcomes Intrinsic, Acquired, and Spontaneous Antibiotic Resistance. mBio 2020, 11. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, C.W.; Sim, J.H.; Shah, K.R.; Kolesnikova-Kaplan, A.; Shi, W.; Eckert, R. Selective Membrane Disruption: Mode of Action of C16G2, a Specifically Targeted Antimicrobial Peptide ▿. Antimicrob Agents Chemother 2011, 55, 3446–3452. [Google Scholar] [CrossRef]
- Castaneda, P.; McLaren, A.; Tavaziva, G.; Overstreet, D. Biofilm Antimicrobial Susceptibility Increases With Antimicrobial Exposure Time. Clin Orthop Relat Res 2016, 474, 1659–1664. [Google Scholar] [CrossRef]
- Chen, X.; Thomsen, T.R.; Winkler, H.; Xu, Y. Influence of Biofilm Growth Age, Media, Antibiotic Concentration and Exposure Time on Staphylococcus Aureus and Pseudomonas Aeruginosa Biofilm Removal in Vitro. BMC Microbiol 2020, 20, 264. [Google Scholar] [CrossRef]
- Sorrell, T.C.; Packham, D.R.; Shanker, S.; Foldes, M.; Munro, R. Vancomycin Therapy for Methicillin-Resistant Staphylococcus Aureus. Ann Intern Med 1982, 97, 344–350. [Google Scholar] [CrossRef]
- Zhong, W.; Shi, Z.; Mahadevegowda, S.H.; Liu, B.; Zhang, K.; Koh, C.H.; Ruan, L.; Chen, Y.; Zeden, M.S.; Pee, C.J.E.; et al. Designer Broad-Spectrum Polyimidazolium Antibiotics. Proceedings of the National Academy of Sciences 2020, 117, 31376–31385. [Google Scholar] [CrossRef]
- Riss, T.L.; Moravec, R.A.; Niles, A.L.; Duellman, S.; Benink, H.A.; Worzella, T.J.; Minor, L. Cell Viability Assays. In Assay Guidance Manual; Markossian, S., Grossman, A., Arkin, M., Auld, D., Austin, C., Baell, J., Brimacombe, K., Chung, T.D.Y., Coussens, N.P., Dahlin, J.L., Devanarayan, V., Foley, T.L., Glicksman, M., Gorshkov, K., Haas, J.V., Hall, M.D., Hoare, S., Inglese, J., Iversen, P.W., Lal-Nag, M., Li, Z., Manro, J.R., McGee, J., McManus, O., Pearson, M., Riss, T., Saradjian, P., Sittampalam, G.S., Tarselli, M., Trask, O.J., Weidner, J.R., Wildey, M.J., Wilson, K., Xia, M., Xu, X., Eds.; Eli Lilly & Company and the National Center for Advancing Translational Sciences: Bethesda (MD), 2004. [Google Scholar]
- Crowley, L.C.; Scott, A.P.; Marfell, B.J.; Boughaba, J.A.; Chojnowski, G.; Waterhouse, N.J. Measuring Cell Death by Propidium Iodide Uptake and Flow Cytometry. Cold Spring Harb Protoc 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Propidium Iodide. Available online: https://www.thermofisher.com/order/catalog/product/P1304MP (accessed on 30 June 2023).
- Dombach, J.L.; Quintana, J.L.J.; Detweiler, C.S. Staphylococcal Bacterial Persister Cells, Biofilms, and Intracellular Infection Are Disrupted by JD1, a Membrane-Damaging Small Molecule. mBio 12, e01801-21. [CrossRef]
- Bellio, P.; Fagnani, L.; Nazzicone, L.; Celenza, G. New and Simplified Method for Drug Combination Studies by Checkerboard Assay. MethodsX 2021, 8, 101543. [Google Scholar] [CrossRef]
- Stephens, C.; Lucena, M.I.; Andrade, R.J. Idiosyncratic Drug-Induced Liver Injury: Mechanisms and Susceptibility Factors☆. In Comprehensive Toxicology (Third Edition); McQueen, C.A., Ed.; Elsevier: Oxford, 2018; pp. 625–650. ISBN 978-0-08-100601-6. [Google Scholar]
- Gomtsyan, A. Heterocycles in Drugs and Drug Discovery. Chem Heterocycl Comp 2012, 48, 7–10. [Google Scholar] [CrossRef]
- Hamlin, T.A.; Swart, M.; Bickelhaupt, F.M. Nucleophilic Substitution (SN2): Dependence on Nucleophile, Leaving Group, Central Atom, Substituents, and Solvent. ChemPhysChem 2018, 19, 1315–1330. [Google Scholar] [CrossRef]
- Boucher, H.W.; Corey, G.R. Epidemiology of Methicillin-Resistant Staphylococcus Aureus. CLIN INFECT DIS 2008, 46, S344–S349. [Google Scholar] [CrossRef] [PubMed]
- MLP Reddy JPhCPhB C Photochemistry Review. 2018. [CrossRef]
- Moormeier, D.E.; Bayles, K.W. Staphylococcus Aureus Biofilm: A Complex Developmental Organism. Mol Microbiol 2017, 104, 365–376. [Google Scholar] [CrossRef]
- Shree, P.; Singh, C.K.; Sodhi, K.K.; Surya, J.N.; Singh, D.K. Biofilms: Understanding the Structure and Contribution towards Bacterial Resistance in Antibiotics. Medicine in Microecology 2023, 16, 100084. [Google Scholar] [CrossRef]
- Høiby, N.; Ciofu, O.; Johansen, H.K.; Song, Z.; Moser, C.; Jensen, P.Ø.; Molin, S.; Givskov, M.; Tolker-Nielsen, T.; Bjarnsholt, T. The Clinical Impact of Bacterial Biofilms. Int J Oral Sci 2011, 3, 55–65. [Google Scholar] [CrossRef] [PubMed]
- Bich-Loan, N.T.; Kien, K.T.; Thanh, N.L.; Kim-Thanh, N.T.; Huy, N.Q.; The-Hai, P.; Muller, M.; Nachtergael, A.; Duez, P.; Thang, N.D. Toxicity and Anti-Proliferative Properties of Anisomeles Indica Ethanol Extract on Cervical Cancer HeLa Cells and Zebrafish Embryos. Life 2021, 11, 257. [Google Scholar] [CrossRef]
- Tamargo, J.; Le Heuzey, J.-Y.; Mabo, P. Narrow Therapeutic Index Drugs: A Clinical Pharmacological Consideration to Flecainide. Eur J Clin Pharmacol 2015, 71, 549–567. [Google Scholar] [CrossRef]
- Bennion, B.J.; Be, N.A.; McNerney, M.W.; Lao, V.; Carlson, E.M.; Valdez, C.A.; Malfatti, M.A.; Enright, H.A.; Nguyen, T.H.; Lightstone, F.C.; et al. Predicting a Drug’s Membrane Permeability: A Computational Model Validated With in Vitro Permeability Assay Data. J Phys Chem B 2017, 121, 5228–5237. [Google Scholar] [CrossRef] [PubMed]
- Saldívar-González, F.I.; Lenci, E.; Trabocchi, A.; Medina-Franco, J.L. Exploring the Chemical Space and the Bioactivity Profile of Lactams: A Chemoinformatic Study. RSC Adv. 2019, 9, 27105–27116. [Google Scholar] [CrossRef] [PubMed]
- Donkor, G.Y.; Anderson, G.M.; Stadler, M.; Tawiah, P.O.; Orellano, C.D.; Edwards, K.A.; Dahl, J.-U. A Novel Ruthenium-Silver Based Antimicrobial Potentiates Aminoglycoside Activity against Pseudomonas Aeruginosa. mSphere 2023, 0, e00190-23. [Google Scholar] [CrossRef] [PubMed]
- Shah, R.; Jankiewicz, O.; Johnson, C.; Livingston, B.; Dahl, J.-U. Pseudomonas Aeruginosa Kills Staphylococcus Aureus in a Polyphosphate-Dependent Manner. bioRxiv 2023, 2023.12.05.570291. [Google Scholar] [CrossRef]
 |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




