Submitted:
23 August 2024
Posted:
23 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
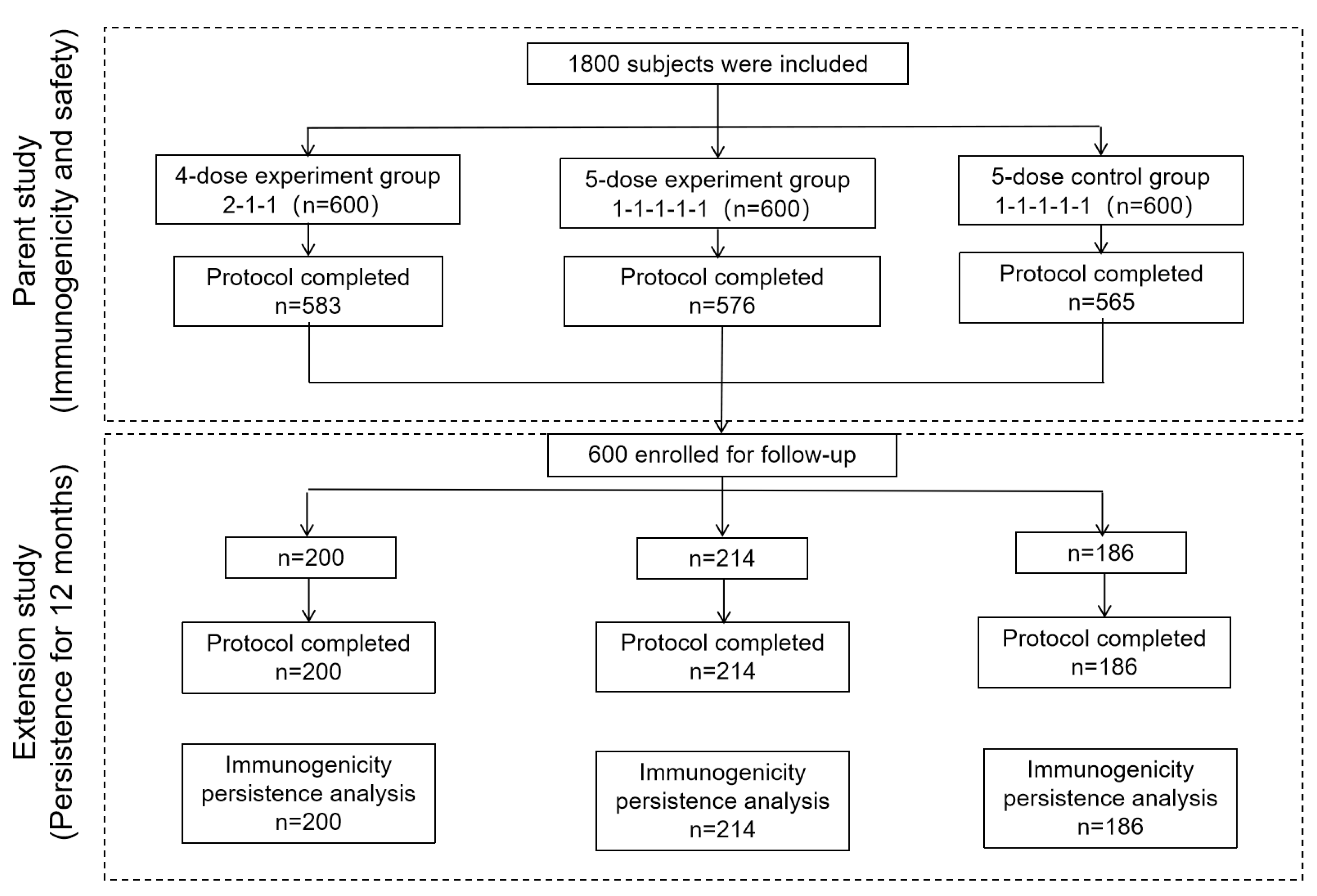
2.1. Patients
2.2. Intervention
2.3. Sample Testing
2.4. Evaluation of Immune Persistence
2.5. Statistical Analysis
2.6. Ethical Review
3. Results
3.1. Baseline Characteristics
3.2. Immunological Characteristics Analysis
3.3. Analysis of Immunogenic Persistence
3.4. Distribution of Antibody Levels at 12 Months Post-Immunization
3.5. Subgroup Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, L. Y.; Sun, M. P.; Zhang, X. C.; Suo, L. D.; Xu, R. H.; Zou, Y. J.; Zuo, L. B.; Qi, H. Safety and Immunogenicity of Two Freeze-Dried Vero Cell Rabies Vaccines for Human Use in Post-Exposure Prophylaxis. Vaccine 2011, 24, 2679–2681. [Google Scholar] [CrossRef] [PubMed]
- Shi, N.; Zhang, Y.; Zheng, H.; Zhu, Z.; Wang, D.; Li, S.; Li, Y.; Yang, L.; Zhang, J.; Bai, Y.; et al. Immunogenicity, Safety and Antibody Persistence of a Purified Vero Cell Cultured Rabies Vaccine (Speeda) Administered by the Zagreb Regimen or Essen Regimen in Post-Exposure Subjects. Hum Vaccin Immunother 2017, 13, 1–8. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Expert Consultation on Rabies. Second report. Technical report series no. 931. Available online: https://www.who.int/publications/i/item/who-trs-931 (accessed on 24 July 2024).
- Vodopija, I.; Sureau, P.; Smerdel, S.; Lafon, M.; Baklaić, Z.; Ljubicić, M.; Svjetlicić, M. Interaction of Rabies Vaccine with Human Rabies Immunoglobulin and Reliability of a 2-1-1 Schedule Application for Postexposure Treatment. Vaccine 1988, 6, 283–286. [Google Scholar] [CrossRef]
- Rupprecht, C. E.; Briggs, D.; Brown, C. M.; Franka, R.; Katz, S. L.; Kerr, H. D.; Lett, S. M.; Levis, R.; Meltzer, M. I.; Schaffner, W.; et al. Use of a Reduced (4-Dose) Vaccine Schedule for Postexposure Prophylaxis to Prevent Human Rabies: Recommendations of the Advisory Committee on Immunization Practices. MMWR Recomm Rep 2010, 59, 1–9. [Google Scholar] [PubMed]
- World Health Organization. Rabies vaccines: WHO position paper–recommendations. Vaccine, 2010; 28, 7140–7142. [CrossRef]
- Ma, J.; Wang, H.; Li, J.; Chang, L.; Xie, Y.; Liu, Z.; Zhao, Y.; Malerczyk, C. A Randomized Open-Labeled Study to Demonstrate the Non-Inferiority of Purified Chick-Embryo Cell Rabies Vaccine Administered in the Zagreb Regimen (2-1-1) Compared with the Essen Regimen in Chinese Adults. Hum Vaccin Immunother 2014, 10, 2805–2812. [Google Scholar] [CrossRef] [PubMed]
- Mahendra, B. J.; Narayana, D. A.; Agarkhedkar, S.; Ravish, H. S.; Harish, B. R.; Agarkhedkar, S.; Madhusudana, S. N.; Belludi, A.; Ahmed, K.; Jonnalagedda, R.; et al. Comparative Study on the Immunogenicity and Safety of a Purified Chick Embryo Cell Rabies Vaccine (PCECV) Administered According to Two Different Simulated Post Exposure Intramuscular Regimens (Zagreb versus Essen). Hum Vaccin Immunother 2015, 11, 428–434. [Google Scholar] [CrossRef] [PubMed]
- Li, R.; Li, Y.; Wen, S.; Wen, H.; Nong, Y.; Mo, Z.; Xie, F.; Pellegrini, M. Immunogenicity and Safety of Purified Chick-Embryo Cell Rabies Vaccine Under Zagreb 2-1-1 or 5-Dose Essen Regimen in Chinese Children 6 to 17 Years Old and Adults Over 50 Years: A Randomized Open-Label Study. Hum Vaccin Immunother 2015, 11, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Zhang, J.; Meng, S.; Ge, L.; You, Y.; Xu, Q.; Wang, H.; Yang, J.; Wang, S.; Wu, H. Safety and Immunogenicity of Human Rabies Vaccine for the Chinese Population After PEP: A Systematic Review and Meta-Analysis. Vaccine 2022, 40, 4371–4379. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Li, J.; Zhou, L.; Chen, J.; Jin, Z.; Meng, Q.; Chai, J.; Gao, H.; Wang, Y.; Zhao, D.; et al. A Randomized, Double-Blind, Controlled Phase III Clinical Trial to Evaluate the Immunogenicity and Safety of a Lyophilized Human Rabies Vaccine (Vero Cells) in Healthy Participants Aged 10-60 Years Following Essen and Zagreb Vaccination Procedures. Vaccines (Basel) 2023, 11, 1311. [Google Scholar] [CrossRef]
- Wang, L. Y.; Sun, M. P.; Zhang, X. C.; Suo, L. D.; Xu, R. H.; Zou, Y. J.; Zuo, L. B.; Qi, H. Safety and Immunogenicity of Two Freeze-Dried Vero Cell Rabies Vaccines for Human Use in Post-Exposure Prophylaxis. Vaccine 2011, 29, 2679–2681. [Google Scholar] [CrossRef] [PubMed]
- Center For Drug Evaluation, China. Guidelines for Clinical Research on Human Rabies Vaccines. Available online: https://www.cde.org.cn/main/news/viewInfoCommon/c670820fd316d8977dc72c4fa7aaa749 (accessed on 12 July 2024).
- Lim, P. L.; Barkham, T. M. Serologic Response to Rabies Pre-Exposure Vaccination in Persons with Potential Occupational Exposure in Singapore. Int J Infect Dis 2010, 14, e511–e513. [Google Scholar] [CrossRef] [PubMed]
- Cramer, J. P.; Jelinek, T.; Paulke-Korinek, M.; Reisinger, E. C.; Dieckmann, S.; Alberer, M.; Bühler, S.; Bosse, D.; Meyer, S.; Fragapane, E.; et al. One-year Immunogenicity Kinetics and Safety of a Purified Chick Embryo Cell Rabies Vaccine and an Inactivated Vero Cell-Derived Japanese Encephalitis Vaccine Administered Concomitantly According to a New, 1-Week, Accelerated Primary Series. J Travel Med 2016, 23, taw011. [Google Scholar] [CrossRef] [PubMed]
- Overduin, L. A.; van Dongen, J. J. M.; Visser, L. G. The Cellular Immune Response to Rabies Vaccination: A Systematic Review. Vaccines (Basel) 2019, 7, 110. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Zhu, Z.; Wang, C. Persistence of Rabies Antibody 5 Years After Postexposure Prophylaxis with Vero Cell Antirabies Vaccine and Antibody Response to a Single Booster Dose. Clin Vaccine Immunol 2011, 18, 1477–1479. [Google Scholar] [CrossRef] [PubMed]
- National People’s Congress of, P.R.C. The China vaccine administration law. Available online: http://www.npc.gov.cn/npc/c30834/201907/11447c85e05840b9b12c62b5b645fe9d.shtml (accessed on 12 July 2024).
- Yan, X. Comparison of Compliance and Economic Cost of Two Different Immunization Procedures for Rabies Vaccine. Contemporary Medicine 2020, 26, 44–46. (In Chinese) [Google Scholar]
| four-dose experimental group (n=200) | five-dose control group (n=186) | five-dose experimental group (n=214) | P value | |
| Age, mean±SD (median) | 36.55±15.44 (40.67) | 39.67±13.83 (43.93) | 38.70±13.60 (41.89) | 0.089 |
| Gender (male:female) | 83:117 | 67:119 | 73:141 | 0.277 |
| Items | four-dose experimental group (n=200) | five-dose control group (n=186) | five-dose experimental group (n=214) | P value |
| Pre-immune | ||||
| Antibody positivity rates, % | 9.00 (5.42–13.85) | 5.91 (2.99–10.34) | 8.41(5.06–12.97) | 0.490 |
| GMC (IU/mL) | 0.10 (0.09–0.12) | 0.09 (0.08–0.10) | 0.10 (0.08–0.11) | 0.708 |
| Day 7 post-first dose | ||||
| Antibody positivity rate, % | 79.00 (69.71–86.51) | 46.07 (35.44–56.96) | 59.46 (49.73–68.68) | <0.001 |
| GMC (IU/mL) | 1.34 (0.94–1.90) | 0.40 (0.30–0.54) | 0.55 (0.37–0.80) | <0.001 |
| Day 14 post-first dose | ||||
| Antibody positivity rate, % | 100.00 (96.38–100.0) | 100.00 (96.27–100.0) | 100.00 (96.48–100.0) | >0.999 |
| GMC (IU/mL) | 80.02 (66.02–96.99) | 85.49 (69.58–105.05) | 78.19 (64.42–94.90) | 0.807 |
| Overall 14 days post-vaccination | >0.999 | |||
| Antibody positivity rate, % | 100.00 (98.17–100.00) | 100.00 (98.04–100.00) | 100.00 (98.29–100.00) | |
| GMC (IU/mL) | 49.28 (42.73–56.83) | 44.56 (38.35–51.78) | 46.37 (40.47–53.12) | 0.621 |
| four-dose experimental group (n=200) | five-dose control group (n=186) | five-dose experimental group (n=214) | P value | |
| Number of positivity | 194 | 174 | 203 | |
| Positivity rate (95% CI) | 97.00 (93.58–98.89) | 93.55 (89.00–96.62) | 94.86 (90.99–97.41) | 0.277 |
| GMC (IU/mL) | 2.50 (2.12–2.94) | 2.05 (1.69–2.49) | 2.04 (1.73–2.42) | 0.184 |
| GMC increase time | 0.05 (0.04–0.06) | 0.05 (0.04–0.05) | 0.04 (0.04–0.05) | 0.421 |
| Neutralizing antibody Concentration (IU/mL) |
four-dose experimental group (n=200) | five-dose control group (n=186) | five-dose experimental group (n=214) | P | |||
| Number | % | Number | % | Number | % | ||
| ≥0.5 | 194 | 97.00 | 174 | 93.55 | 203 | 94.86 | 0.2774 |
| ≥1 | 165 | 82.50 | 139 | 74.73 | 160 | 74.77 | 0.1018 |
| ≥2 | 92 | 46.00 | 64 | 34.41 | 84 | 39.25 | 0.0648 |
| ≥4 | 74 | 37.00 | 49 | 26.34 | 58 | 27.10 | 0.0355 |
| ≥8 | 35 | 17.50 | 29 | 15.59 | 28 | 13.08 | 0.4569 |
| ≥16 | 10 | 5.00 | 16 | 8.60 | 14 | 6.54 | 0.3646 |
| ≥32 | 6 | 3.00 | 9 | 4.84 | 9 | 4.21 | 0.6424 |
| ≥64 | 0 | 0.00 | 4 | 2.15 | 2 | 0.93 | 0.0861 |
| ≥128 | 0 | 0.00 | 2 | 1.08 | 0 | 0.93 | 0.4707 |
| ≥256 | 0 | 0.00 | 0 | 0.00 | 0 | 0.00 | 1.0000 |
| four-dose experimental group (n=200) | five-dose control group (n=186) | five-dose experimental group (n=214) | P | |
| Number of pre-immune positive participants , n | 18 | 11 | 18 | |
| Pre-immune GMC (IU/mL) | 1.32 | 1.27 | 1.45 | 0.933 |
| 12 months post-immunization GMC (IU/mL) | 5.71 | 8.24 | 6.15 | 0.771 |
| 12 months post-immunization positivity rate, % | 18 (9.00%) | 11 (5.91%) | 17 (7.94%) | 0.439 |
| Number of pre-immune negative participants, n | 182 | 175 | 196 | |
| Pre-immune GMC (IU/mL) | 0.08 | 0.08 | 0.07 | 0.861 |
| 12 months post-immunization GMC (IU/mL) | 2.30 | 1.88 | 1.85 | 0.148 |
| 12 months post-immunization positivity rate, % | 176 (88.00%) | 163 (87.63%) | 186 (86.92%) | 0.308 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




