Submitted:
15 August 2024
Posted:
16 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. TPE-Derived Exosomes Induce the Invasion of Lung Cancer Cells
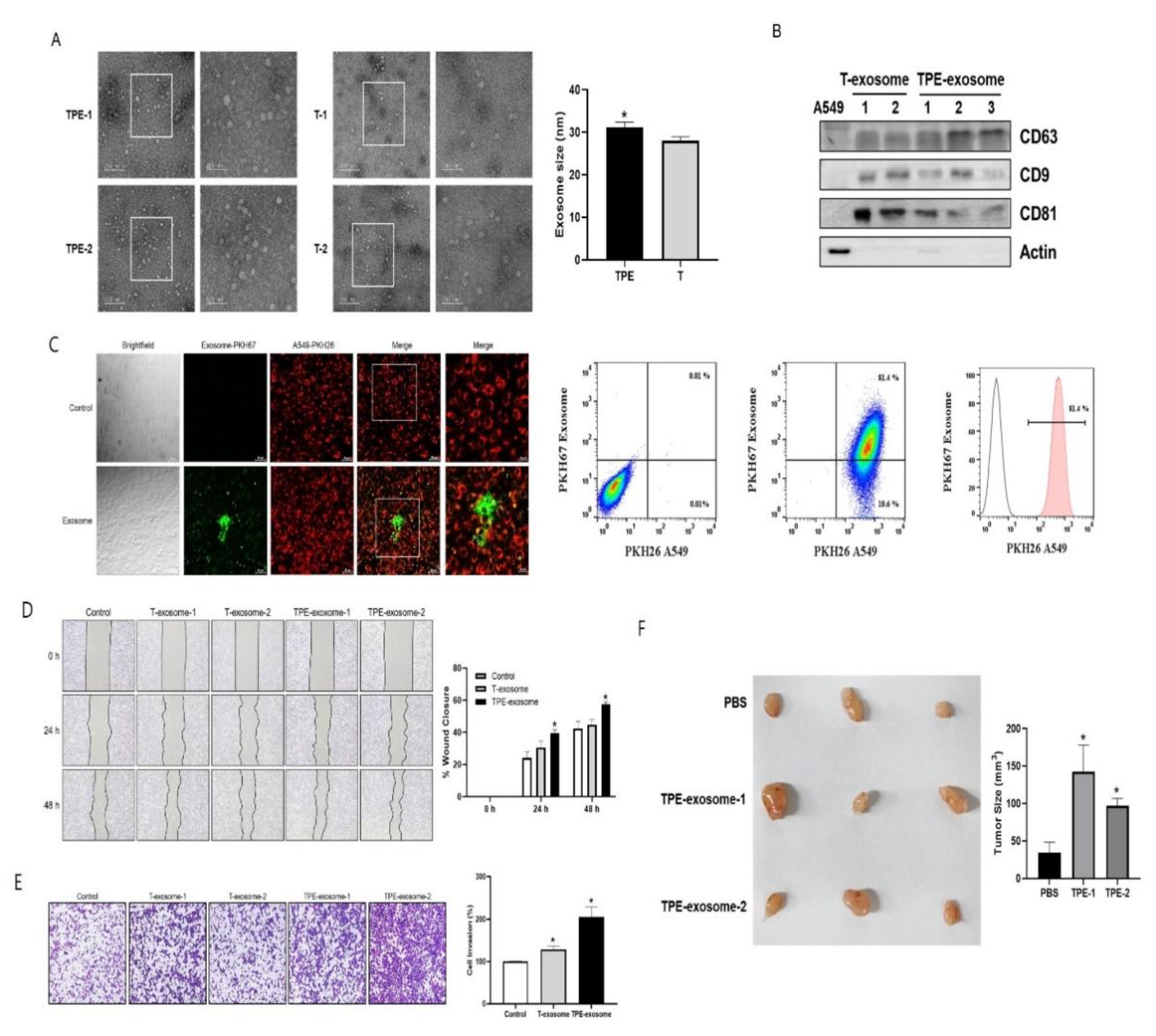
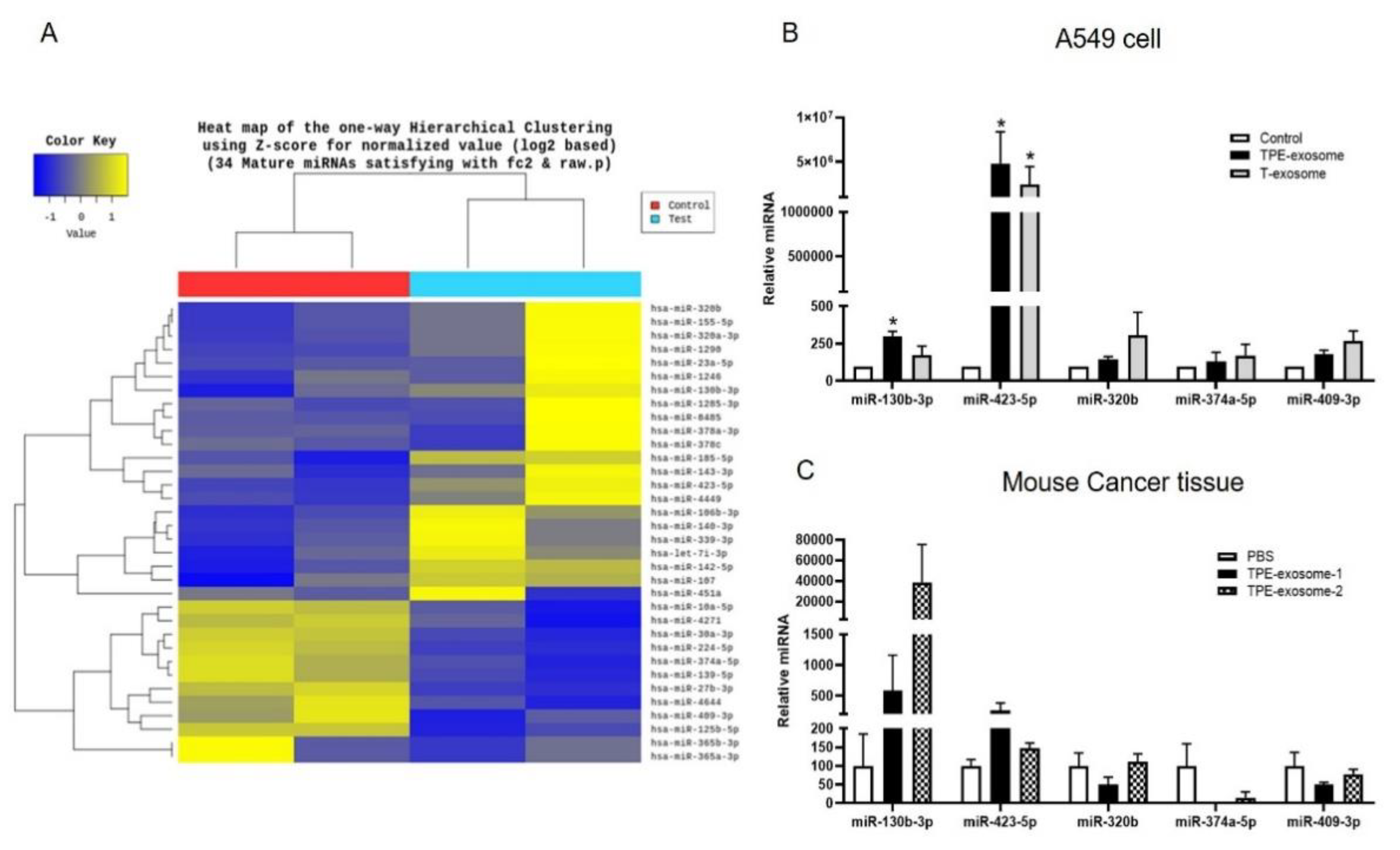
2.2. Identifying Exosomal miRNAs from TPE Involved in the Invasion of Lung Cancer Cells
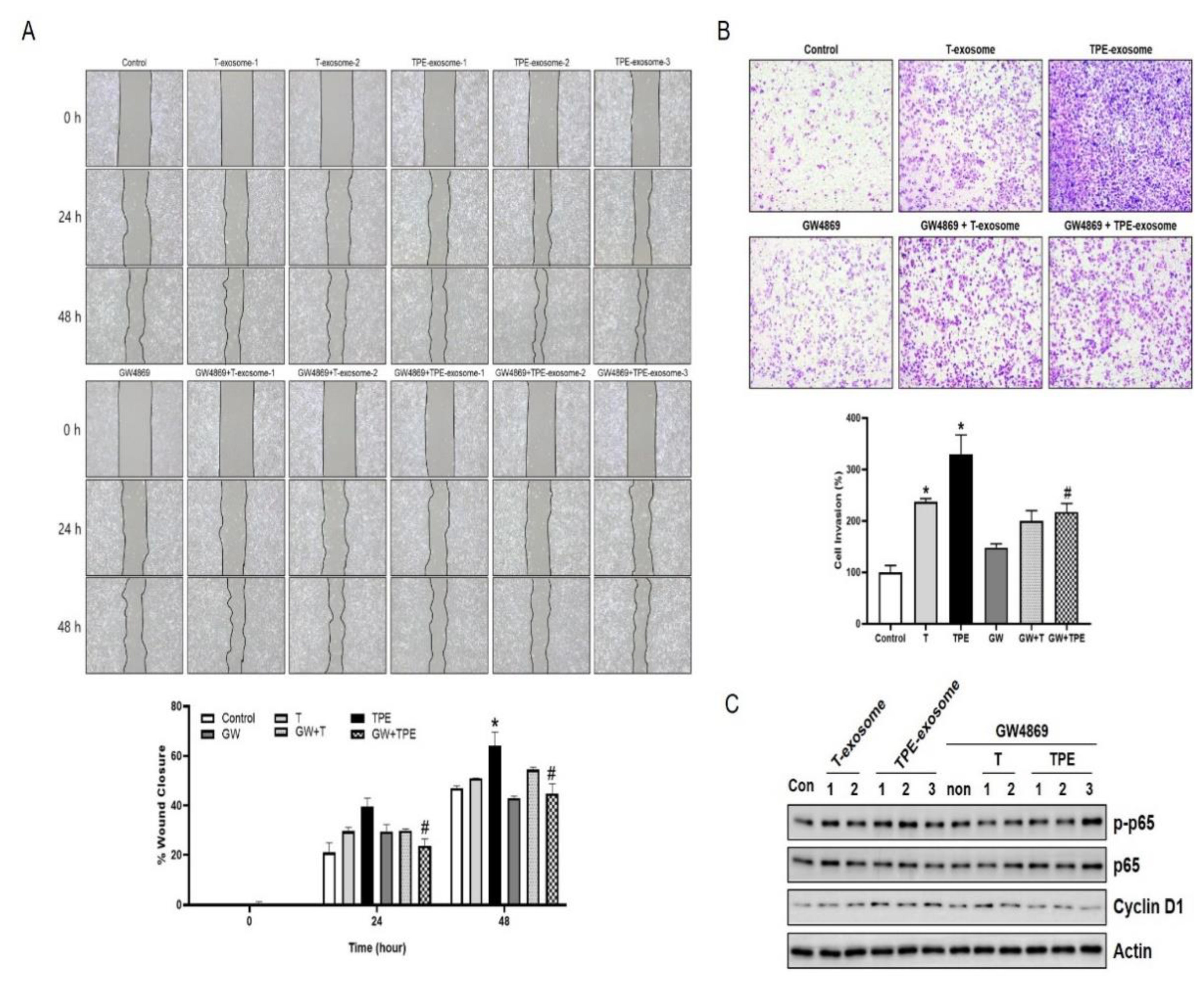
2.3. GW4869 Inhibits the Migration and Invasion of TPE-Derived Exosome-Stimulated A549 Cells
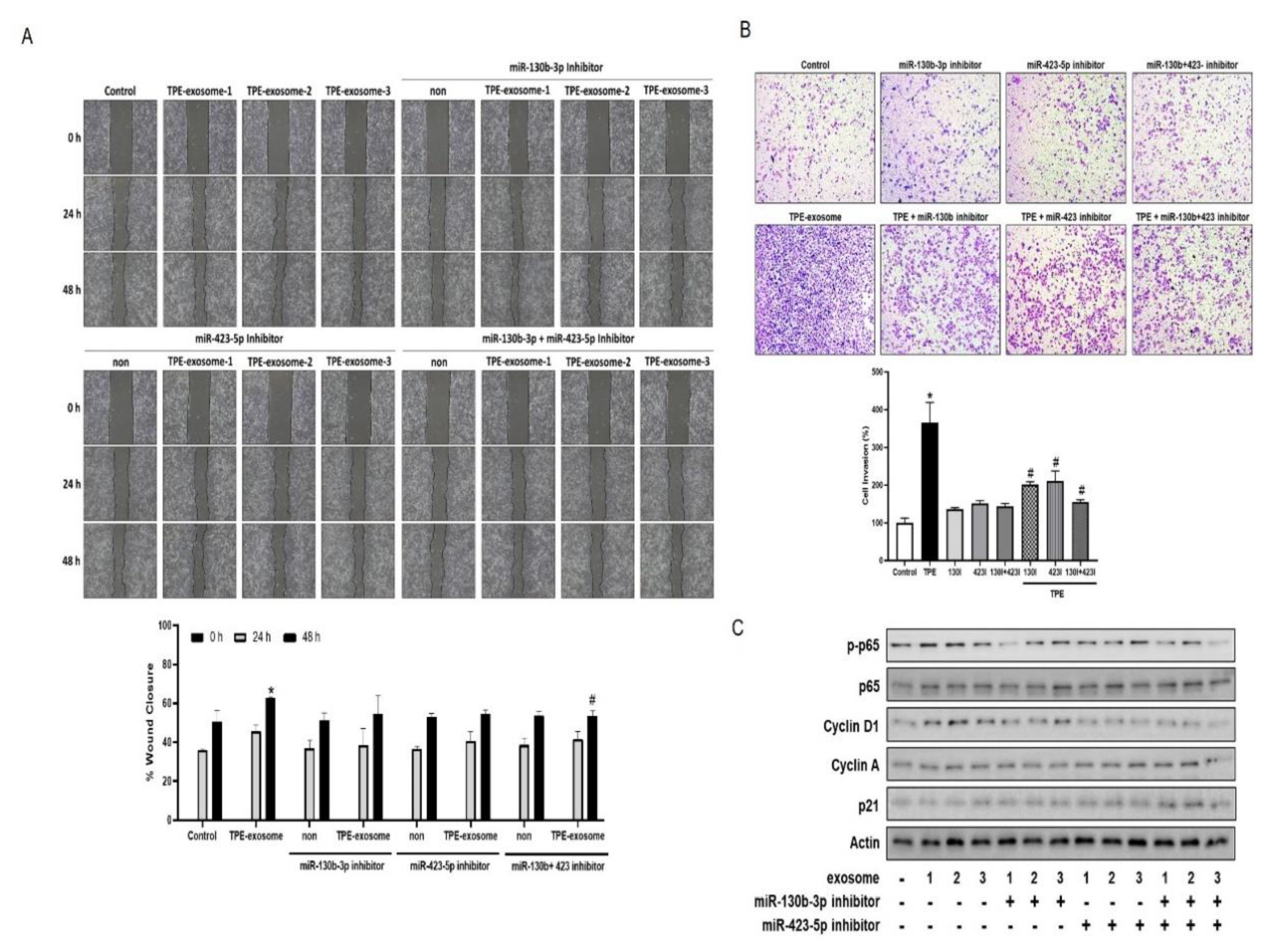
2.4. Exosome-Mediated Transfer miR-130 and miR-423 from TPE Promotes the Invasion of Cancer Cells via Cyclin-D
3. Discussion
4. Materials and Methods
4.1. Patients and Samples
4.2. Exosome Isolation and Quantification
4.3. Transmission Electron Microscopy
4.4. Small RNA Library Construction and miRNA Sequencing
4.5. Animal Experiments
4.6. Cell Culture and Reagent Treatment
4.7. Cellular Internalization of Exosomes
4.8. Cell Migration and Invasion Assays
4.9. Extraction of miRNA, Real-Time PCR, and Western Blotting
4.10. Transfection of miRNA Inhibitors
4.11. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mashouri, L.; Yousefi, H.; Aref, A. R.; Ahadi, A. M.; Molaei, F.; Alahari, S. K., Exosomes: composition, biogenesis, and mechanisms in cancer metastasis and drug resistance. Mol Cancer 2019, 18, (1), 75. [CrossRef]
- Boukouris, S.; Mathivanan, S., Exosomes in bodily fluids are a highly stable resource of disease biomarkers. Proteomics Clin Appl 2015, 9, (3-4), 358-67. [CrossRef]
- Lin, J.; Wang, Y.; Zou, Y. Q.; Chen, X.; Huang, B.; Liu, J.; Xu, Y. M.; Li, J.; Zhang, J.; Yang, W. M.; Min, Q. H.; Sun, F.; Li, S. Q.; Gao, Q. F.; Wang, X. Z., Differential miRNA expression in pleural effusions derived from extracellular vesicles of patients with lung cancer, pulmonary tuberculosis, or pneumonia. Tumour Biol 2016. [CrossRef]
- Kim, Y.; Shin, S.; Lee, K. A., Exosome-based detection of EGFR T790M in plasma and pleural fluid of prospectively enrolled non-small cell lung cancer patients after first-line tyrosine kinase inhibitor therapy. Cancer Cell Int 2021, 21, (1), 50. [CrossRef]
- Ahmadzada, T.; Kao, S.; Reid, G.; Clarke, S.; Grau, G. E.; Hosseini-Beheshti, E., Extracellular vesicles as biomarkers in malignant pleural mesothelioma: A review. Crit Rev Oncol Hematol 2020, 150, 102949. [CrossRef]
- Zhang, X.; Yuan, X.; Shi, H.; Wu, L.; Qian, H.; Xu, W., Exosomes in cancer: small particle, big player. J Hematol Oncol 2015, 8, 83. [CrossRef]
- Wang, L.; Chen, X.; Zhou, X.; Roizman, B.; Zhou, G. G., miRNAs Targeting ICP4 and Delivered to Susceptible Cells in Exosomes Block HSV-1 Replication in a Dose-Dependent Manner. Mol Ther 2018, 26, (4), 1032-1039. [CrossRef]
- Wang, J.; Ni, J.; Beretov, J.; Thompson, J.; Graham, P.; Li, Y., Exosomal microRNAs as liquid biopsy biomarkers in prostate cancer. Crit Rev Oncol Hematol 2020, 145, 102860. [CrossRef]
- Woodman, C.; Vundu, G.; George, A.; Wilson, C. M., Applications and strategies in nanodiagnosis and nanotherapy in lung cancer. Semin Cancer Biol 2021, 69, 349-364. [CrossRef]
- Garcia, J. I.; Mambuque, E.; Nguenha, D.; Vilanculo, F.; Sacoor, C.; Sequera, V. G.; Fernandez-Quevedo, M.; Pierre, M. L.; Chiconela, H.; Faife, L. A.; Respeito, D.; Saavedra, B.; Nhampossa, T.; Lopez-Varela, E.; Garcia-Basteiro, A. L., Mortality and risk of tuberculosis among people living with HIV in whom TB was initially ruled out. Sci Rep 2020, 10, (1), 15442. [CrossRef]
- Luczynski, P.; Poulin, P.; Romanowski, K.; Johnston, J. C., Tuberculosis and risk of cancer: A systematic review and meta-analysis. PLoS One 2022, 17, (12), e0278661. [CrossRef]
- Abdeahad, H.; Salehi, M.; Yaghoubi, A.; Aalami, A. H.; Aalami, F.; Soleimanpour, S., Previous pulmonary tuberculosis enhances the risk of lung cancer: systematic reviews and meta-analysis. Infect Dis (Lond) 2022, 54, (4), 255-268. [CrossRef]
- Liao, K. M.; Lee, C. S.; Wu, Y. C.; Shu, C. C.; Ho, C. H., Prior treated tuberculosis and mortality risk in lung cancer. Front Med (Lausanne) 2023, 10, 1121257. [CrossRef]
- Nalbandian, A.; Yan, B. S.; Pichugin, A.; Bronson, R. T.; Kramnik, I., Lung carcinogenesis induced by chronic tuberculosis infection: the experimental model and genetic control. Oncogene 2009, 28, (17), 1928-38. [CrossRef]
- Woo, S. J.; Kim, Y.; Jung, H.; Lee, J. J.; Hong, J. Y., Tuberculous Fibrosis Enhances Tumorigenic Potential via the NOX4-Autophagy Axis. Cancers (Basel) 2021, 13, (4). [CrossRef]
- Woo, S. J.; Kim, Y.; Kang, H. J.; Jung, H.; Youn, D. H.; Hong, Y.; Lee, J. J.; Hong, J. Y., Tuberculous pleural effusion-induced Arg-1(+) macrophage polarization contributes to lung cancer progression via autophagy signaling. Respir Res 2024, 25, (1), 198. [CrossRef]
- Karimian, A.; Ahmadi, Y.; Yousefi, B., Multiple functions of p21 in cell cycle, apoptosis and transcriptional regulation after DNA damage. DNA Repair (Amst) 2016, 42, 63-71. [CrossRef]
- Louloupi, A.; Ntini, E.; Liz, J.; Orom, U. A., Microprocessor dynamics shows co- and post-transcriptional processing of pri-miRNAs. RNA 2017, 23, (6), 892-898.
- Vanni, I.; Alama, A.; Grossi, F.; Dal Bello, M. G.; Coco, S., Exosomes: a new horizon in lung cancer. Drug Discov Today 2017, 22, (6), 927-936. [CrossRef]
- Goldie, B. J.; Dun, M. D.; Lin, M.; Smith, N. D.; Verrills, N. M.; Dayas, C. V.; Cairns, M. J., Activity-associated miRNA are packaged in Map1b-enriched exosomes released from depolarized neurons. Nucleic Acids Res 2014, 42, (14), 9195-208. [CrossRef]
- Li, C.; Zhou, T.; Chen, J.; Li, R.; Chen, H.; Luo, S.; Chen, D.; Cai, C.; Li, W., The role of Exosomal miRNAs in cancer. J Transl Med 2022, 20, (1), 6. [CrossRef]
- Buscail, E.; Alix-Panabieres, C.; Quincy, P.; Cauvin, T.; Chauvet, A.; Degrandi, O.; Caumont, C.; Verdon, S.; Lamrissi, I.; Moranvillier, I.; Buscail, C.; Marty, M.; Laurent, C.; Vendrely, V.; Moreau-Gaudry, F.; Bedel, A.; Dabernat, S.; Chiche, L., High Clinical Value of Liquid Biopsy to Detect Circulating Tumor Cells and Tumor Exosomes in Pancreatic Ductal Adenocarcinoma Patients Eligible for Up-Front Surgery. Cancers (Basel) 2019, 11, (11). [CrossRef]
- Vitiello, P. P.; De Falco, V.; Giunta, E. F.; Ciardiello, D.; Cardone, C.; Vitale, P.; Zanaletti, N.; Borrelli, C.; Poliero, L.; Terminiello, M.; Arrichiello, G.; Caputo, V.; Famiglietti, V.; Mattera Iacono, V.; Marrone, F.; Di Liello, A.; Martini, G.; Napolitano, S.; Caraglia, M.; Lombardi, A.; Franco, R.; De Vita, F.; Morgillo, F.; Troiani, T.; Ciardiello, F.; Martinelli, E., Clinical Practice Use of Liquid Biopsy to Identify RAS/BRAF Mutations in Patients with Metastatic Colorectal Cancer (mCRC): A Single Institution Experience. Cancers (Basel) 2019, 11, (10). [CrossRef]
- Holla, S.; Ghorpade, D. S.; Singh, V.; Bansal, K.; Balaji, K. N., Mycobacterium bovis BCG promotes tumor cell survival from tumor necrosis factor-alpha-induced apoptosis. Mol Cancer 2014, 13, 210. [CrossRef]
- Gupta, P. K.; Tripathi, D.; Kulkarni, S.; Rajan, M. G., Mycobacterium tuberculosis H37Rv infected THP-1 cells induce epithelial mesenchymal transition (EMT) in lung adenocarcinoma epithelial cell line (A549). Cell Immunol 2016, 300, 33-40. [CrossRef]
- Antony, V. B., Immunological mechanisms in pleural disease. Eur Respir J 2003, 21, (3), 539-44. [CrossRef]
- O’Brien, K.; Breyne, K.; Ughetto, S.; Laurent, L. C.; Breakefield, X. O., RNA delivery by extracellular vesicles in mammalian cells and its applications. Nat Rev Mol Cell Biol 2020, 21, (10), 585-606. [CrossRef]
- Zhao, L.; Ye, Y.; Gu, L.; Jian, Z.; Stary, C. M.; Xiong, X., Extracellular vesicle-derived miRNA as a novel regulatory system for bi-directional communication in gut-brain-microbiota axis. J Transl Med 2021, 19, (1), 202. [CrossRef]
- Liu, Y. J.; Wang, C., A review of the regulatory mechanisms of extracellular vesicles-mediated intercellular communication. Cell Commun Signal 2023, 21, (1), 77. [CrossRef]
- Zhang, Q.; Zhang, B.; Sun, L.; Yan, Q.; Zhang, Y.; Zhang, Z.; Su, Y.; Wang, C., MicroRNA-130b targets PTEN to induce resistance to cisplatin in lung cancer cells by activating Wnt/beta-catenin pathway. Cell Biochem Funct 2018, 36, (4), 194-202.
- Sekino, Y.; Sakamoto, N.; Sentani, K.; Oue, N.; Teishima, J.; Matsubara, A.; Yasui, W., miR-130b Promotes Sunitinib Resistance through Regulation of PTEN in Renal Cell Carcinoma. Oncology 2019, 97, (3), 164-172. [CrossRef]
- Miao, Y.; Zheng, W.; Li, N.; Su, Z.; Zhao, L.; Zhou, H.; Jia, L., MicroRNA-130b targets PTEN to mediate drug resistance and proliferation of breast cancer cells via the PI3K/Akt signaling pathway. Sci Rep 2017, 7, 41942. [CrossRef]
- Jia, L.; Lei, B.; Gao, H.; Jia, L.; Luo, D.; Han, J.; Jia, B., miR-130b suppresses the invasion and migration of prostate cancer via inhibiting DLL1 and regulating the PI3K/Akt pathways. Exp Ther Med 2022, 23, (1), 98. [CrossRef]
- Sharova, E.; Grassi, A.; Marcer, A.; Ruggero, K.; Pinto, F.; Bassi, P.; Zanovello, P.; Zattoni, F.; D’Agostino, D. M.; Iafrate, M.; Ciminale, V., A circulating miRNA assay as a first-line test for prostate cancer screening. Br J Cancer 2016, 114, (12), 1362-6. [CrossRef]
- Colangelo, T.; Fucci, A.; Votino, C.; Sabatino, L.; Pancione, M.; Laudanna, C.; Binaschi, M.; Bigioni, M.; Maggi, C. A.; Parente, D.; Forte, N.; Colantuoni, V., MicroRNA-130b promotes tumor development and is associated with poor prognosis in colorectal cancer. Neoplasia 2013, 15, (9), 1086-99.
- Jia, W.; Yu, T.; An, Q.; Cao, X.; Pan, H., MicroRNA-423-5p inhibits colon cancer growth by promoting caspase-dependent apoptosis. Exp Ther Med 2018, 16, (2), 1225-1231. [CrossRef]
- Huang, Y.; Feng, G., MiR-423-5p aggravates lung adenocarcinoma via targeting CADM1. Thorac Cancer 2021, 12, (2), 210-217.
- Jorge, K. T. O. S.; Souza, R. P.; Assis, M. T. A.; Arújo, M. G.; Locati, M.; Jesus, A. M. R.; Baptista, I. M. F. D.; Lima, C. X.; Teixeira, A. L.; Teixeira, M. M.; Soriani, F. M., Characterization of MicroRNA Expression Profiles and Identification of Potential Biomarkers in Leprosy. J Clin Microbiol 2017, 55, (5), 1516-1525. [CrossRef]
- Zhang, X.; Sai, B.; Wang, F.; Wang, L.; Wang, Y.; Zheng, L.; Li, G.; Tang, J.; Xiang, J., Hypoxic BMSC-derived exosomal miRNAs promote metastasis of lung cancer cells via STAT3-induced EMT. Mol Cancer 2019, 18, (1), 40. [CrossRef]
- Musgrove, E. A.; Caldon, C. E.; Barraclough, J.; Stone, A.; Sutherland, R. L., Cyclin D as a therapeutic target in cancer. Nat Rev Cancer 2011, 11, (8), 558-72. [CrossRef]
- Bandi, N.; Zbinden, S.; Gugger, M.; Arnold, M.; Kocher, V.; Hasan, L.; Kappeler, A.; Brunner, T.; Vassella, E., miR-15a and miR-16 are implicated in cell cycle regulation in a Rb-dependent manner and are frequently deleted or down-regulated in non-small cell lung cancer. Cancer Res 2009, 69, (13), 5553-9. [CrossRef]
- Bonci, D.; Coppola, V.; Musumeci, M.; Addario, A.; Giuffrida, R.; Memeo, L.; D’Urso, L.; Pagliuca, A.; Biffoni, M.; Labbaye, C.; Bartucci, M.; Muto, G.; Peschle, C.; De Maria, R., The miR-15a-miR-16-1 cluster controls prostate cancer by targeting multiple oncogenic activities. Nat Med 2008, 14, (11), 1271-7. [CrossRef]
- Hinz, M.; Krappmann, D.; Eichten, A.; Heder, A.; Scheidereit, C.; Strauss, M., NF-kappaB function in growth control: regulation of cyclin D1 expression and G0/G1-to-S-phase transition. Mol Cell Biol 1999, 19, (4), 2690-8.
- Dai, T.; Zhao, X.; Li, Y.; Yu, L.; Li, Y.; Zhou, X.; Gong, Q., miR-423 Promotes Breast Cancer Invasion by Activating NF-kappaB Signaling. Onco Targets Ther 2020, 13, 5467-5478.
- Cui, X.; Kong, C.; Zhu, Y.; Zeng, Y.; Zhang, Z.; Liu, X.; Zhan, B.; Piao, C.; Jiang, Z., miR-130b, an onco-miRNA in bladder cancer, is directly regulated by NF-kappaB and sustains NF-kappaB activation by decreasing Cylindromatosis expression. Oncotarget 2016, 7, (30), 48547-48561. [CrossRef]
- Brock, M.; Haider, T. J.; Vogel, J.; Gassmann, M.; Speich, R.; Trenkmann, M.; Ulrich, S.; Kohler, M.; Huber, L. C., The hypoxia-induced microRNA-130a controls pulmonary smooth muscle cell proliferation by directly targeting CDKN1A. Int J Biochem Cell Biol 2015, 61, 129-37.
- Lin, J.; Huang, S.; Wu, S.; Ding, J.; Zhao, Y.; Liang, L.; Tian, Q.; Zha, R.; Zhan, R.; He, X., MicroRNA-423 promotes cell growth and regulates G(1)/S transition by targeting p21Cip1/Waf1 in hepatocellular carcinoma. Carcinogenesis 2011, 32, (11), 1641-7. [CrossRef]
- Light, R. W.; Macgregor, M. I.; Luchsinger, P. C.; Ball, W. C., Jr., Pleural effusions: the diagnostic separation of transudates and exudates. Ann Intern Med 1972, 77, (4), 507-13. [CrossRef]
- Workman, P.; Aboagye, E. O.; Balkwill, F.; Balmain, A.; Bruder, G.; Chaplin, D. J.; Double, J. A.; Everitt, J.; Farningham, D. A.; Glennie, M. J.; Kelland, L. R.; Robinson, V.; Stratford, I. J.; Tozer, G. M.; Watson, S.; Wedge, S. R.; Eccles, S. A.; Committee of the National Cancer Research, I., Guidelines for the welfare and use of animals in cancer research. Br J Cancer 2010, 102, (11), 1555-77. [CrossRef]
| Characteristics | TPE1 | TPE 2 | T1 | T2 |
| Age | 23 | 77 | 51 | 51 |
| Sex | Male | Male | Male | Male |
| Comorbidity | none | HTN, CAOD | Cirrhosis, CKD | Wernicke’s encephalopathy |
| Pleural fluid analysis | ||||
| Leukocyte (/mm3) | 288 | 1530 | 576 | 46 |
| Total protein (g/dL) | 5.4 | 4.7 | 3 | <2.0 |
| Albumin | 3.6 | 2.5 | 1.8 | 1.2 |
| LDH (IU/L) | 1613 | 178 | 76 | 61 |
| CRP (mg/L) | 6.7 | 6.2 | <4.0 | 4.8 |
| ADA (IU/L) | 102.5 | 77.5 | 29 | 6 |
| Neutrophil (%) | 20 | 20 | 60 | 5 |
| Lymphocyte (%) | 80 | 80 | 40 | 95 |
| Serum | ||||
| Total protein (g/dL) | 7.1 | 6.4 | 6.5 | 5.2 |
| Albumin (g/dL) | 4.6 | 3.4 | 3.6 | 3.1 |
| LDH (IU/L) | 225 | 229 | 176 | 177 |
| Light criteria | ||||
| Pleural protein/serum protein>0.5 | Yes | Yes | No | No |
| Pleural LDH/serum LDH>0.6 | Yes | Yes | No | No |
| Pleural LDH>2/3 of serum LDH | Yes | Yes | No | No |
| Positive pleural fluid TB culture | Yes | Yes | No | No |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




