Submitted:
08 August 2024
Posted:
12 August 2024
You are already at the latest version
Abstract
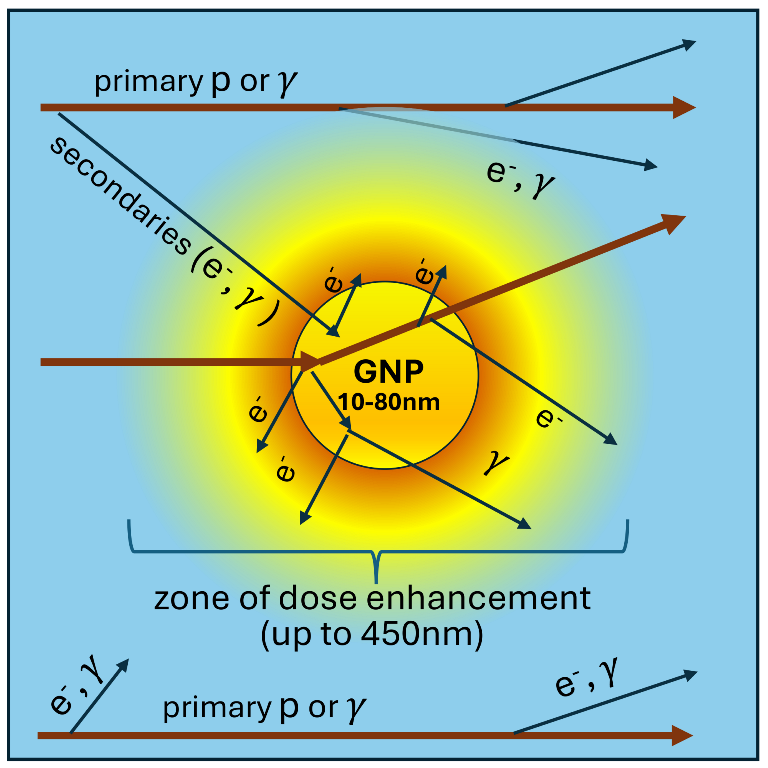
Keywords:
1. Introduction
2. Results
3. Methods and Models
3.1. Geometry of the Simulated System
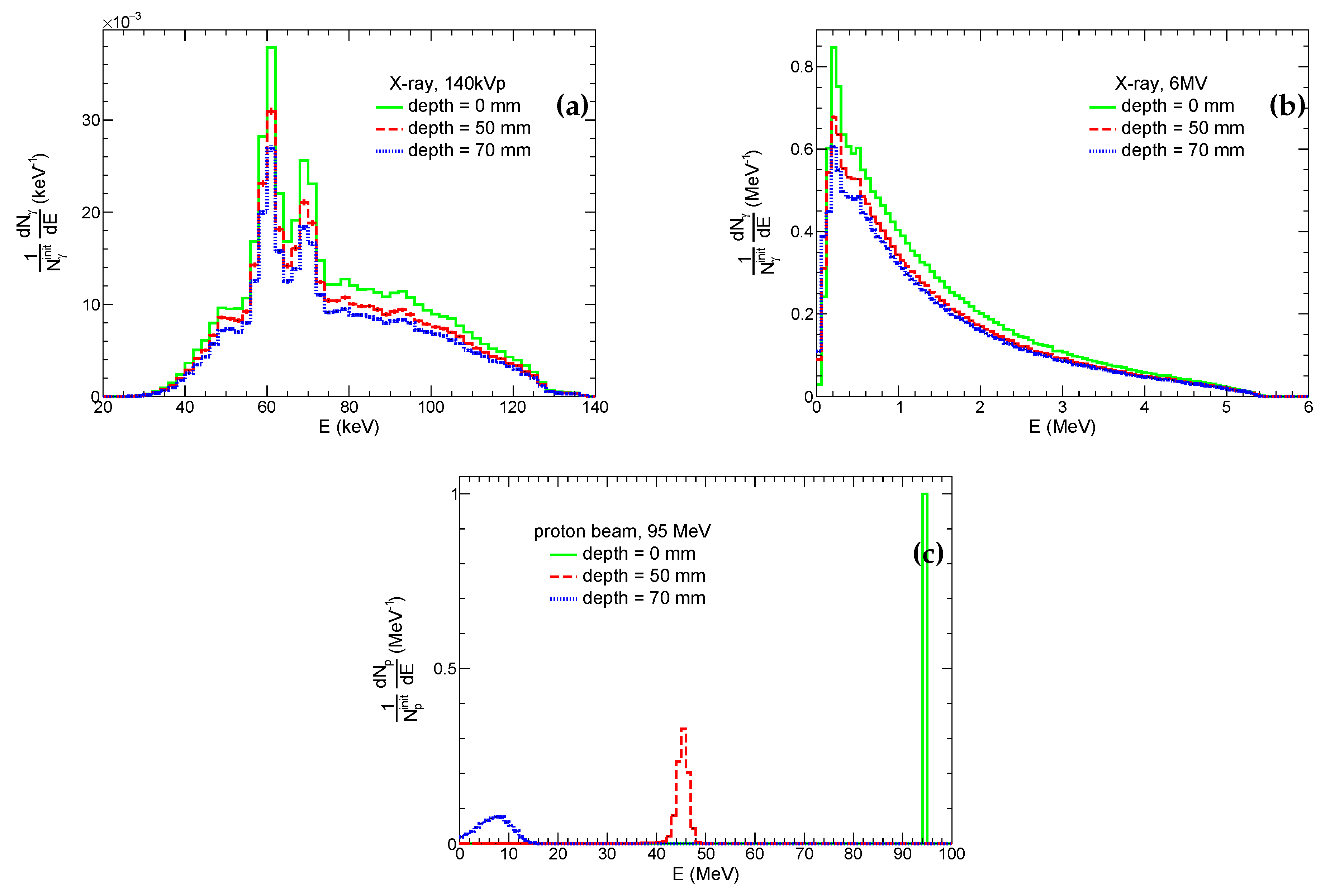
3.2. Radiation Fields
3.3. Physics Models
- -
- in water region, Geant4-DNA models are implemented
- -
- in GNP region, a new set of Geant4-DNA discrete models for electron interaction with gold [31];
- -
- in water region, Geant4-DNA models are implemented
- -
- in GNP region, standard Geant4 models are being used for lack of a better option.
4. Discussion
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of open access journals |
| TLA | Three letter acronym |
| LD | Linear dichroism |
References
- Gong, L.; Zhang, Y.; Liu, C.; Zhang, M.; Han, S. Application of radiosensitizers in cancer radiotherapy. Int. J. Nanomedicine 2021, 16, 1083–1102. [Google Scholar] [CrossRef]
- Moulder, J.E. Chemical radiosensitizers: the Journal history. International Journal of Radiation Biology 2019, 95, 940–944. [Google Scholar] [CrossRef] [PubMed]
- Kwatra, D.; Venugopal, A.; Anant, S. Nanoparticles in radiation therapy: a summary of various approaches to enhance radiosensitization in cancer. Translational Cancer Research 2013, 2. [Google Scholar] [CrossRef]
- Augustine, R.; Hasan, A.; Primavera, R.; Wilson, R.J.; Thakor, A.S.; Kevadiya, B.D. Cellular uptake and retention of nanoparticles: Insights on particle properties and interaction with cellular components. Materials Today Communications 2020, 25, 101692. [Google Scholar] [CrossRef]
- Kong, T.; Zeng, J.; Wang, X.; Yang, X.; Yang, J.; McQuarrie, S.; McEwan, A.; Roa, W.; Chen, J.; Xing, J.Z. Enhancement of Radiation Cytotoxicity in Breast-Cancer Cells by Localized Attachment of Gold Nanoparticles. Small 4, 1537–1543. [CrossRef]
- Zhang, X.D.; Wu, D.; Shen, X.; Chen, J.; Sun, Y.M.; Liu, P.X.; Liang, X.J. Size-dependent radiosensitization of PEG-coated gold nanoparticles for cancer radiation therapy. Biomaterials 2012, 33, 6408–6419. [Google Scholar] [CrossRef]
- Soares, S.; Faria, I.; Aires, F.; Monteiro, A.; Pinto, G.; Sales, M.G.; Correa-Duarte, M.A.; Guerreiro, S.G.; Fernandes, R. Application of Gold Nanoparticles as Radiosensitizer for Metastatic Prostate Cancer Cell Lines. International Journal of Molecular Sciences 2023, 24. [Google Scholar] [CrossRef]
- Hainfeld, J.F.; Slatkin, D.N.; Smilowitz, H.M. The use of gold nanoparticles to enhance radiotherapy in mice. Physics in Medicine Biology 2004, 49, N309. [Google Scholar] [CrossRef]
- Hainfeld, J.F.; Smilowitz, H.M.; OâConnor, M.J.; Dilmanian, F.A.; Slatkin, D.N. Gold Nanoparticle Imaging and Radiotherapy of Brain Tumors in Mice. Nanomedicine 2013, 8, 1601–1609. [Google Scholar] [CrossRef]
- Cunningham, C.; de Kock, M.; Engelbrecht, M.; Miles, X.; Slabbert, J.; Vandevoorde, C. Radiosensitization Effect of Gold Nanoparticles in Proton Therapy. Front Public Health 2021, 9, 699822. [Google Scholar] [CrossRef]
- Porcel, E.; Liehn, S.; Remita, H.; Usami, N.; Kobayashi, K.; Furusawa, Y.; Sech, C.L.; Lacombe, S. Platinum nanoparticles: a promising material for future cancer therapy? Nanotechnology 2010, 21, 085103. [Google Scholar] [CrossRef]
- Briggs, A.; Corde, S.; Oktaria, S.; Brown, R.; Rosenfeld, A.; Lerch, M.; Konstantinov, K.; Tehei, M. Cerium oxide nanoparticles: influence of the high-Z component revealed on radioresistant 9L cell survival under X-ray irradiation. Nanomedicine: Nanotechnology, Biology and Medicine 2013, 9, 1098–1105. [Google Scholar] [CrossRef]
- Brown, R.; Tehei, M.; Oktaria, S.; Briggs, A.; Stewart, C.; Konstantinov, K.; Rosenfeld, A.; Corde, S.; Lerch, M. High-Z Nanostructured Ceramics in Radiotherapy: First Evidence of Ta2O5-Induced Dose Enhancement on Radioresistant Cancer Cells in an MV Photon Field. Particle & Particle Systems Characterization 31, 500–505. [CrossRef]
- Bláha, P.; Feoli, C.; Agosteo, S.; Calvaruso, M.; Cammarata, F.P.; Catalano, R.; Ciocca, M.; Cirrone, G.A.P.; Conte, V.; Cuttone, G.; et al. The Proton-Boron Reaction Increases the Radiobiological Effectiveness of Clinical Low- and High-Energy Proton Beams: Novel Experimental Evidence and Perspectives. Frontiers in Oncology 2021, 11. [Google Scholar] [CrossRef]
- Roy, I.; Krishnan, S.; Kabashin, A.V.; Zavestovskaya, I.N.; Prasad, P.N. Transforming Nuclear Medicine with Nanoradiopharmaceuticals. ACS Nano 2022, 16, 5036–5061. [Google Scholar] [CrossRef]
- Zavestovskaya, I.N.; Popov, A.L.; Kolmanovich, D.D.; Tikhonowski, G.V.; Pastukhov, A.I.; Savinov, M.S.; Shakhov, P.V.; Babkova, J.S.; Popov, A.A.; Zelepukin, I.V.; et al. Boron Nanoparticle-Enhanced Proton Therapy for Cancer Treatment. Nanomaterials 2023, 13. [Google Scholar] [CrossRef] [PubMed]
- Tabbakh, F.; Hosmane, N.S.; Tajudin, S.M.; Ghorashi, A.H.; Morshedian, N. Using 157Gd doped carbon and 157GdF4 nanoparticles in proton-targeted therapy for effectiveness enhancement and thermal neutron reduction: a simulation study. Scientific Reports 2022, 12, 17404. [Google Scholar] [CrossRef] [PubMed]
- Shahmohammadi Beni, M.; Islam, M.R.; Kim, K.M.; Krstic, D.; Nikezic, D.; Yu, K.N.; Watabe, H. On the effectiveness of proton boron fusion therapy (PBFT) at cellular level. Scientific Reports 2022, 12, 18098. [Google Scholar] [CrossRef] [PubMed]
- Chiniforoush, T.A.; Hadadi, A.; Kasesaz, Y.; Sardjono, Y. Evaluation of effectiveness of equivalent dose during proton boron fusion therapy (PBFT) for brain cancer: A Monte Carlo study. Appl Radiat Isot 2021, 170, 109596. [Google Scholar] [CrossRef]
- Bagulya, A.V.; Grichine, V.M.; Zavestovskaya, I.N.; Ryabov, V.A. Geant4 Simulation of p + 11B → 3α reaction. BULLETIN OF THE LEBEDEV PHYISICS INSTITUTE 2023, 27–35. [Google Scholar] [CrossRef]
- Azarkin, M.; Kirakosyan, M.; Ryabov, V. Study of Nuclear Reactions in Therapy of Tumors with Proton Beams. International Journal of Molecular Sciences 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.J.; Wang, C.H.; Chen, S.T.; Chen, H.H.; Leng, W.H.; Chien, C.C.; Wang, C.L.; Kempson, I.M.; Hwu, Y.; Lai, T.C.; et al. Enhancement of cell radiation sensitivity by pegylated gold nanoparticles. Physics in Medicine and Biology 2010, 55, 931–945. [Google Scholar] [CrossRef] [PubMed]
- Polf, J.C.; Bronk, L.F.; Driessen, W.H.P.; Arap, W.; Pasqualini, R.; Gillin, M. Enhanced relative biological effectiveness of proton radiotherapy in tumor cells with internalized gold nanoparticles. Appl. Phys. Lett. 2011, 98, 193702. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Penninckx, S.; Karmani, L.; Heuskin, A.C.; Watillon, K.; Marega, R.; Zola, J.; Corvaglia, V.; Genard, G.; Gallez, B.; et al. LET-dependent radiosensitization effects of gold nanoparticles for proton irradiation. Nanotechnology 2016, 27, 455101. [Google Scholar] [CrossRef]
- Lo, C.Y.; Tsai, S.W.; Niu, H.; Chen, F.H.; Hwang, H.C.; Chao, T.C.; Hsiao, I.T.; Liaw, J.W. Gold-Nanoparticles-Enhanced Production of Reactive Oxygen Species in Cells at Spread-Out Bragg Peak under Proton Beam Radiation. ACS Omega 2023, 8, 17922–17931. [Google Scholar] [CrossRef]
- Kim, J.K.; Seo, S.J.; Kim, H.T.; Kim, K.H.; Chung, M.H.; Kim, K.R.; Ye, S.J. Enhanced proton treatment in mouse tumors through proton irradiated nanoradiator effects on metallic nanoparticles. Physics in Medicine and Biology 2012, 57, 8309. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, T.; Grant, J.; Wolfe, A.; Gillin, M.; Krishnan, S. WE-G-BRE-07: Proton Therapy Enhanced by Tumor-Targeting Gold Nanoparticles: A Pilot in Vivo Experiment at The Proton Therapy Center at MD Anderson Cancer Center. Medical Physics 41, 518–518. [CrossRef]
- McKinnon, S.; Guatelli, S.; Incerti, S.; Ivanchenko, V.; Konstantinov, K.; Corde, S.; Lerch, M.; Tehei, M.; Rosenfeld, A. Local dose enhancement of proton therapy by ceramic oxide nanoparticles investigated with Geant4 simulations. Physica Medica 2016, 32, 1584–1593. [Google Scholar] [CrossRef]
- Martà nez-Rovira, I.; Prezado, Y. Evaluation of the local dose enhancement in the combination of proton therapy and nanoparticles. Medical Physics 42, 6703–6710. [CrossRef]
- Tran, H.; Karamitros, M.; Ivanchenko, V.; Guatelli, S.; McKinnon, S.; Murakami, K.; Sasaki, T.; Okada, S.; Bordage, M.; Francis, Z.; et al. Geant4 Monte Carlo simulation of absorbed dose and radiolysis yields enhancement from a gold nanoparticle under MeV proton irradiation. Nuclear Instruments and Methods in Physics Research Section B: Beam Interactions with Materials and Atoms 2016, 373, 126–139. [Google Scholar] [CrossRef]
- Sakata, D.; Kyriakou, I.; Okada, S.; Tran, H.N.; Lampe, N.; Guatelli, S.; Bordage, M.C.; Ivanchenko, V.; Murakami, K.; Sasaki, T.; et al. Geant4-DNA track-structure simulations for gold nanoparticles: The importance of electron discrete models in nanometer volumes. Physica medica 2018, 45, 2230–2242. [Google Scholar] [CrossRef] [PubMed]
- Sakata, D.; Kyriakou, I.; Tran, H.N.; Bordage, M.C.; Rosenfeld, A.; Ivanchenko, V.; Incerti, S.; Emfietzoglou, D.; Guatelli, S. Electron track structure simulations in a gold nanoparticle using Geant4-DNA. Physica medica 2019, 63, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Peukert, D.; Kempson, I.; Douglass, M.; Bezak, E. Gold Nanoparticle Enhanced Proton Therapy: Monte Carlo Modeling of Reactive Speciesâ Distributions Around a Gold Nanoparticle and the Effects of Nanoparticle Proximity and Clustering. International Journal of Molecular Sciences 2019, 20. [Google Scholar] [CrossRef] [PubMed]
- KonefaÅ, A.; Lniak, W.; Rostocka, J.; Orlef, A.; SokóÅ, M.; Kasperczyk, J.; JarzÄ bek, P.; WroÅska, A.; Rusiecka, K. Influence of a shape of gold nanoparticles on the dose enhancement in the wide range of gold mass concentration for high-energy X-ray beams from a medical linac. Reports of Practical Oncology & Radiotherapy 2020, 25, 579–585. [Google Scholar] [CrossRef]
- Agostinelli, S.; Allison, J.; Amako, K.; Apostolakis, J.; Araujo, H.; Arce, P.; Asai, M.; Axen, D.; Banerjee, S.; Barrand, G.; et al. Geant4âa simulation toolkit. Nuclear Instruments and Methods in Physics Research Section A: Accelerators, Spectrometers, Detectors and Associated Equipment 2003, 506, 250–303. [Google Scholar] [CrossRef]
- Incerti, S.; Baldacchino, G.; Bernal, M.; Capra, R.; Champion, C.; Fransis, Z.; Guèye, P.; Mantero, A.; Mascialino, B.; Moretto, P.; et al. The Geant4-DNA project. International Journal of Modeling, Simulation, and Scientific Computing 2010, 01, 157–178. [Google Scholar] [CrossRef]
- Geant4-DNA collaboration. Geant4-DNA : Extending the Geant4 Monte Carlo simulation toolkit for radiobiology. Available online: http://geant4-dna.org.
- Chen, Y.; Yang, J.; Fu, S.; Wu, J. Gold nanoparticles as radiosensitizers in cancer radiotherapy. International Journal of Nanomedicine 2020, 15, 9407–9430. [Google Scholar] [CrossRef]
- Piccolo, O.; Lincoln, J.D.; Melong, N.; Orr, B.C.; Fernandez, N.R.; Borsavage, J.; Berman, J.N.; Robar, J.; Ha, M.N. Radiation dose enhancement using gold nanoparticles with a diamond linear accelerator target: a multiple cell type analysis. Scientific Reports 2022, 12, 1559. [Google Scholar] [CrossRef]
- Duan, X.; Wang, J.; Yu, L.; Leng, S.; McCollough, C.H. CT scanner x-ray spectrum estimation from transmission measurements. Medical Physics 2011, 38, 993–997. [Google Scholar] [CrossRef]
- Brualla, L.; Rodriguez, M.; Sempau, J.; Andreo, P. PENELOPE/PRIMO-calculated photon and electron spectra from clinical accelerators. Radiation Oncology 2019, 14, 6. [Google Scholar] [CrossRef]
- Morita, K.; Nishimura, Y.; Nakamura, S.; Arai, Y.; Numako, C.; Sato, K.; Nakayama, M.; Akasaka, H.; Sasaki, R.; Ogino, C.; et al. Titanium oxide nano-radiosensitizers for hydrogen peroxide delivery into cancer cells. Colloids and Surfaces B: Biointerfaces 2021, 198, 111451. [Google Scholar] [CrossRef] [PubMed]
- Meyer, T.J.; Scherzad, A.; Moratin, H.; Gehrke, T.E.; Killisperger, J.; Hagen, R.; Wohlleben, G.; Polat, B.; Dembski, S.; Kleinsasser, N.; et al. The Radiosensitizing Effect of Zinc Oxide Nanoparticles in Sub-Cytotoxic Dosing Is Associated with Oxidative Stress In Vitro. Materials (Basel) 2019, 12. [Google Scholar] [CrossRef]
- Bláha, P.; Feoli, C.; Agosteo, S.; Calvaruso, M.; Cammarata, F.P.; Catalano, R.; Ciocca, M.; Cirrone, G.A.P.; Conte, V.; Cuttone, G.; et al. The Proton-Boron Reaction Increases the Radiobiological Effectiveness of Clinical Low- and High-Energy Proton Beams: Novel Experimental Evidence and Perspectives. Front Oncol 2021, 11, 682647. [Google Scholar] [CrossRef] [PubMed]
- Bano, A.; Dawood, A.; Saira, F.; Malik, A.; Alkholief, M.; Ahmad, H.; Khan, M.A.; Ahmad, Z.; Bazighifan, O. Enhancing catalytic activity of gold nanoparticles in a standard redox reaction by investigating the impact of AuNPs size, temperature and reductant concentrations. Scientific Reports 2023, 13, 12359. [Google Scholar] [CrossRef] [PubMed]
- Penninckx, S.; Heuskin, A.C.; Michiels, C.; Lucas, S. Gold Nanoparticles as a Potent Radiosensitizer: A Transdisciplinary Approach from Physics to Patient. Cancers 2020, 12. [Google Scholar] [CrossRef]
- Kessler, A.; Hedberg, J.; Blomberg, E.; Odnevall, I. Reactive Oxygen Species Formed by Metal and Metal Oxide Nanoparticles in Physiological MediaâA Review of Reactions of Importance to Nanotoxicity and Proposal for Categorization. Nanomaterials 2022, 12. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Jiang, C.; Wu, L.; Bai, X.; Zhai, S. Cytotoxicity-Related Bioeffects Induced by Nanoparticles: The Role of Surface Chemistry. Frontiers in Bioengineering and Biotechnology 2019, 7. [Google Scholar] [CrossRef] [PubMed]
- Verkhovtsev, A.V.; Korol, A.V.; Solov’yov, A.V. Revealing the Mechanism of the Low-Energy Electron Yield Enhancement from Sensitizing Nanoparticles. Phys. Rev. Lett. 2015, 114, 063401. [Google Scholar] [CrossRef] [PubMed]
- Zygmanski, P.; Sajo, E.; Brivio, D. Nanoparticle-based radiotherapy: Is dose all that matters? Z Med Phys 2023, 33, 119–122. [Google Scholar] [CrossRef]
- Her, S.; Jaffray, D.A.; Allen, C. Gold nanoparticles for applications in cancer radiotherapy: Mechanisms and recent advancements. Advanced Drug Delivery Reviews 2017, 109, 84–101. [Google Scholar] [CrossRef]
- Schaeublin, N.M.; Braydich-Stolle, L.K.; Schrand, A.M.; Miller, J.M.; Hutchison, J.; Schlager, J.J.; Hussain, S.M. Surface charge of gold nanoparticles mediates mechanism of toxicity. Nanoscale 2011, 3, 410–420. [Google Scholar] [CrossRef] [PubMed]
- May, S.; Hirsch, C.; Rippl, A.; Bohmer, N.; Kaiser, J.P.; Diener, L.; Wichser, A.; Bürkle, A.; Wick, P. Transient DNA damage following exposure to gold nanoparticles. Nanoscale 2018, 10, 15723–15735. [Google Scholar] [CrossRef] [PubMed]
- Adabi, M.; Naghibzadeh, M.; Adabi, M.; Zarrinfard, M.A.; Esnaashari, S.S.; Seifalian, A.M.; Faridi-Majidi, R.; Tanimowo Aiyelabegan, H.; Ghanbari, H. Biocompatibility and nanostructured materials: applications in nanomedicine. Artif Cells Nanomed Biotechnol 2016, 45, 833–842. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Guo, L.H.; Zhang, H.; Zhao, L. UV irradiation induced transformation of TiO2 nanoparticles in water: aggregation and photoreactivity. Environmental Science & Technology 2014, 48, 11962–11968. [Google Scholar] [CrossRef]
- Kose, O.; Tomatis, M.; Turci, F.; Belblidia, N.B.; Hochepied, J.F.; Pourchez, J.; Forest, V. Short Preirradiation of TiO(2) Nanoparticles Increases Cytotoxicity on Human Lung Coculture System. Chemical Research in Toxicology 2021, 34, 733–742. [Google Scholar] [CrossRef] [PubMed]
- Petković, J.; Küzma, T.; Rade, K.; Novak, S.; Filipič, M. Pre-irradiation of anatase TiO2 particles with UV enhances their cytotoxic and genotoxic potential in human hepatoma HepG2 cells. Journal of Hazardous Materials 2011, 196, 145–152. [Google Scholar] [CrossRef]
- Rosa, S.; Connolly, C.; Schettino, G.; Butterworth, K.T.; Prise, K.M. Biological mechanisms of gold nanoparticle radiosensitization. Cancer Nanotechnology 2017, 8, 2. [Google Scholar] [CrossRef]
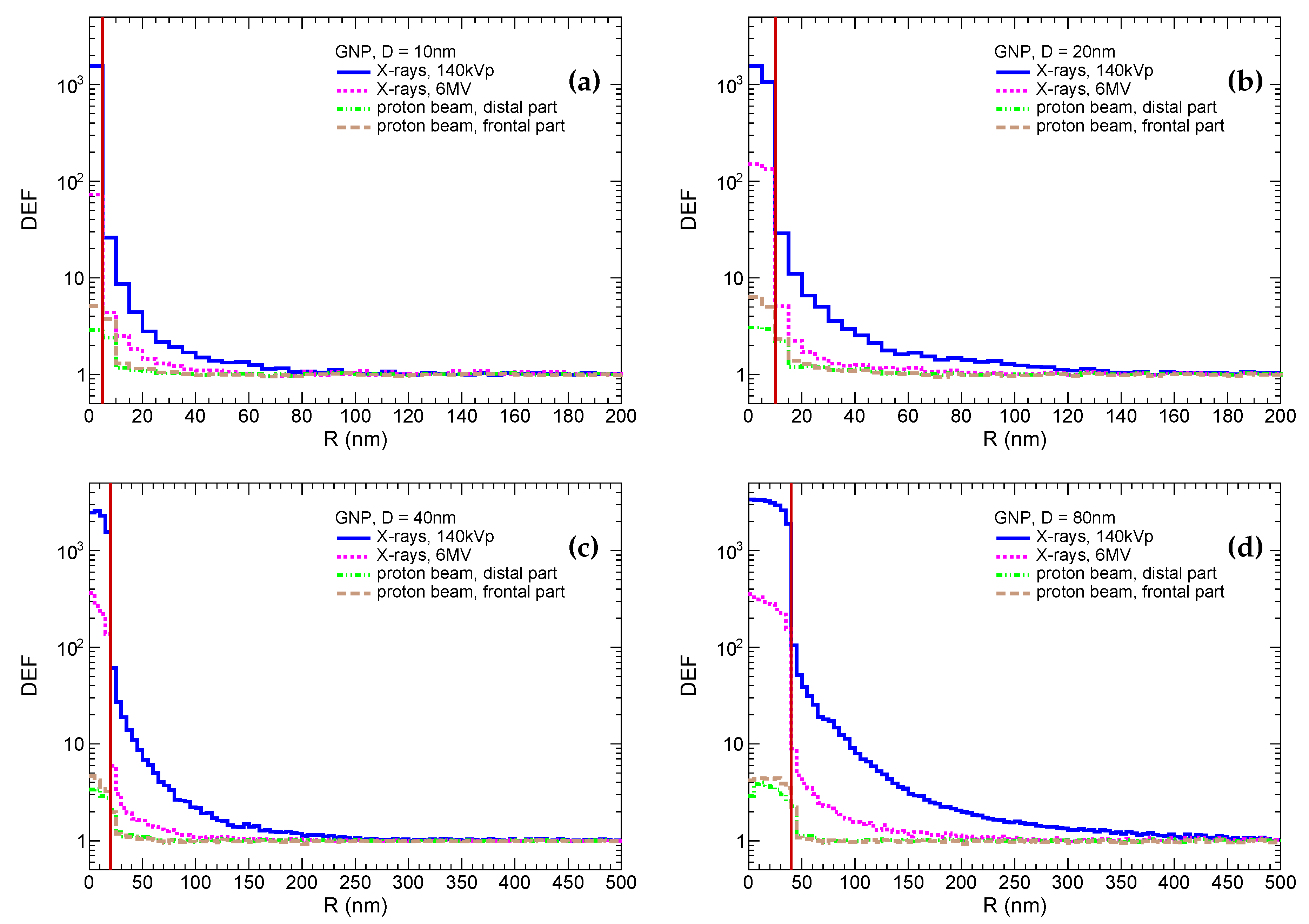
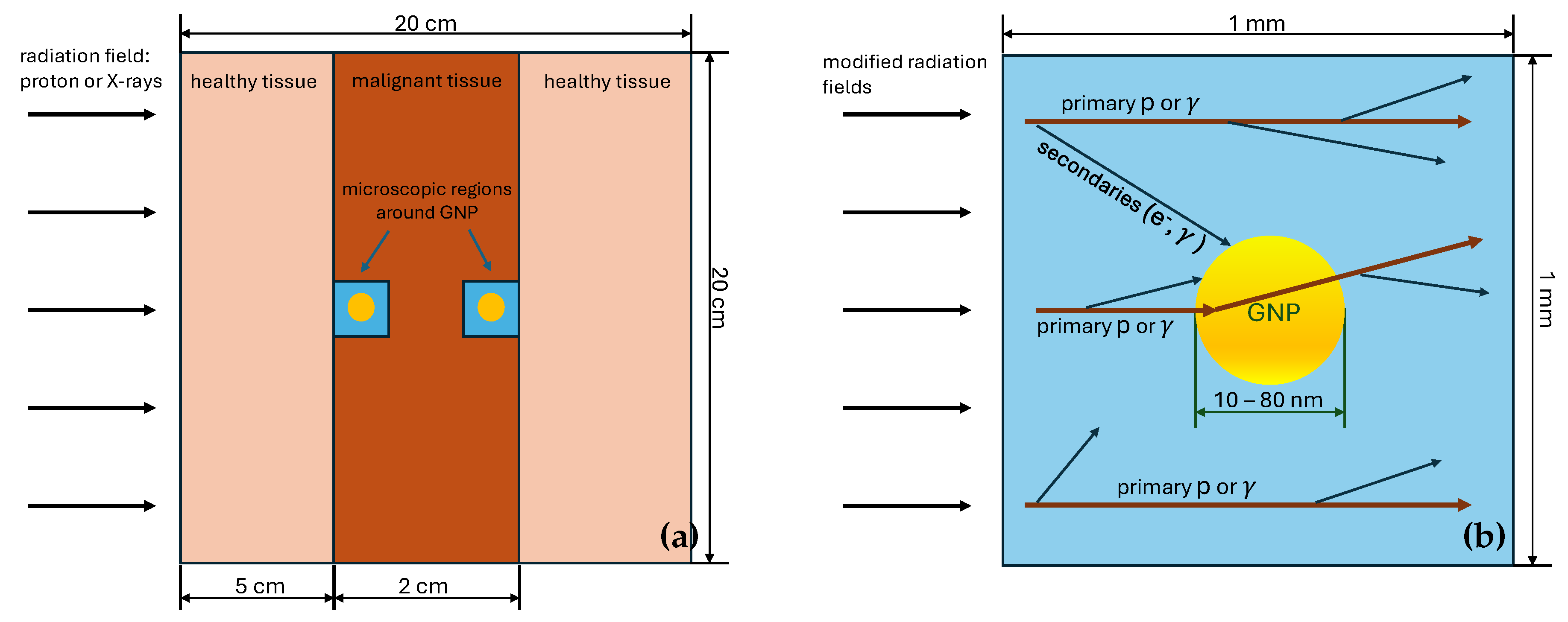
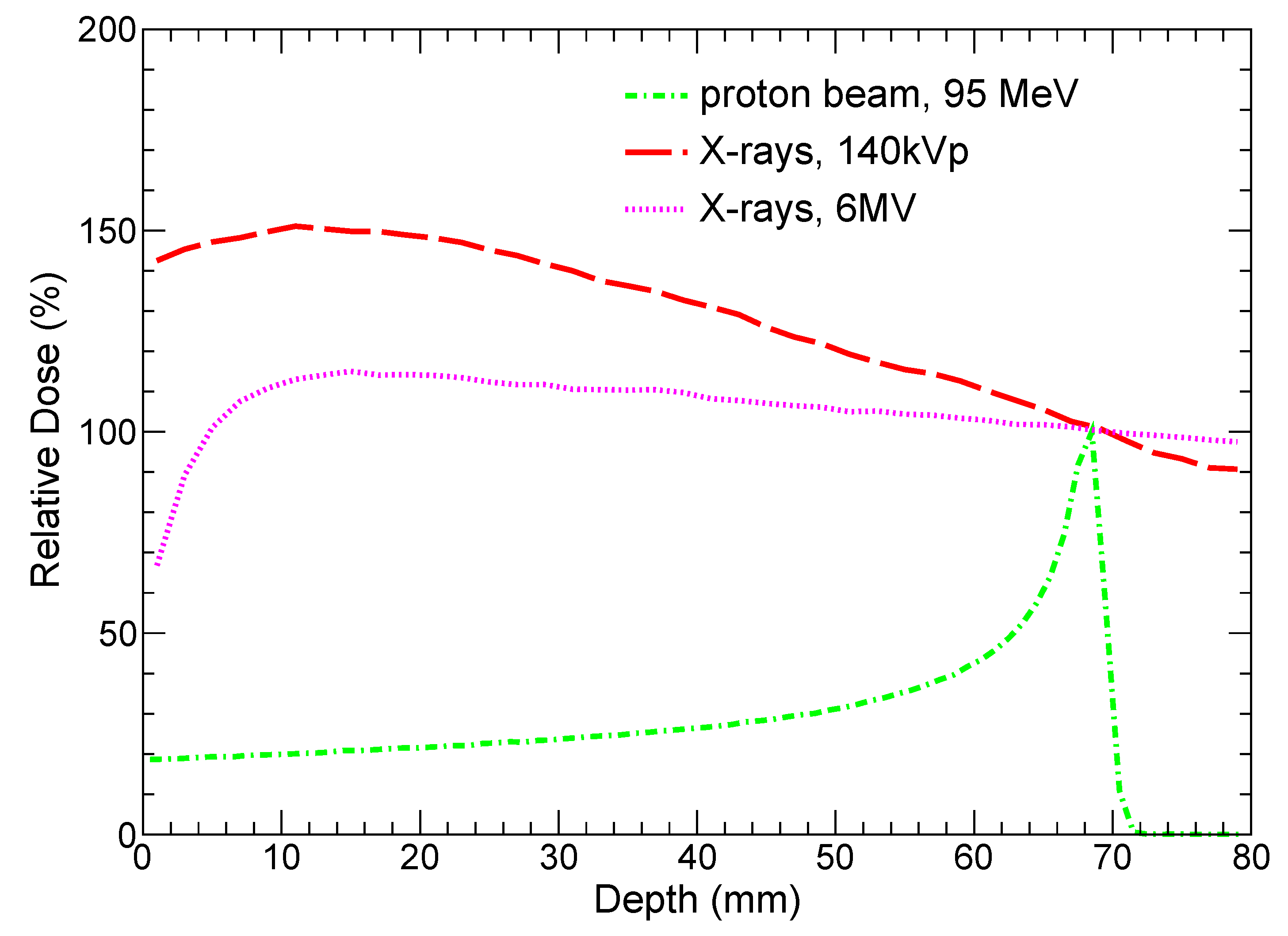
| Radiation Type | GNP Size | |||
|---|---|---|---|---|
| 10 nm | 20 nm | 40 nm | 80 nm | |
| X-rays, 140kVp | ||||
| X-rays, 6MV | ||||
| proton beam | not applicable | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




