Submitted:
06 August 2024
Posted:
07 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
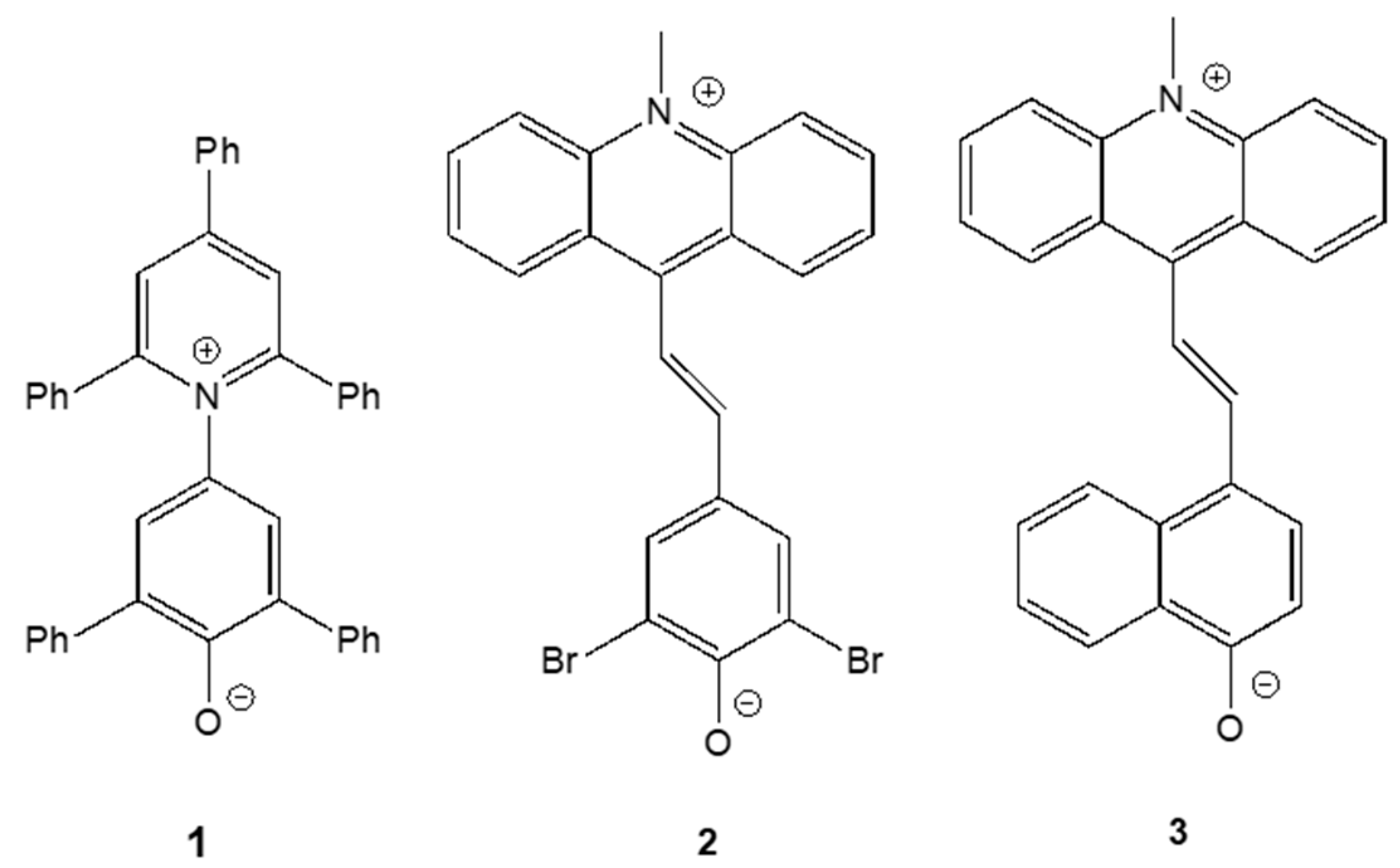
2. Results
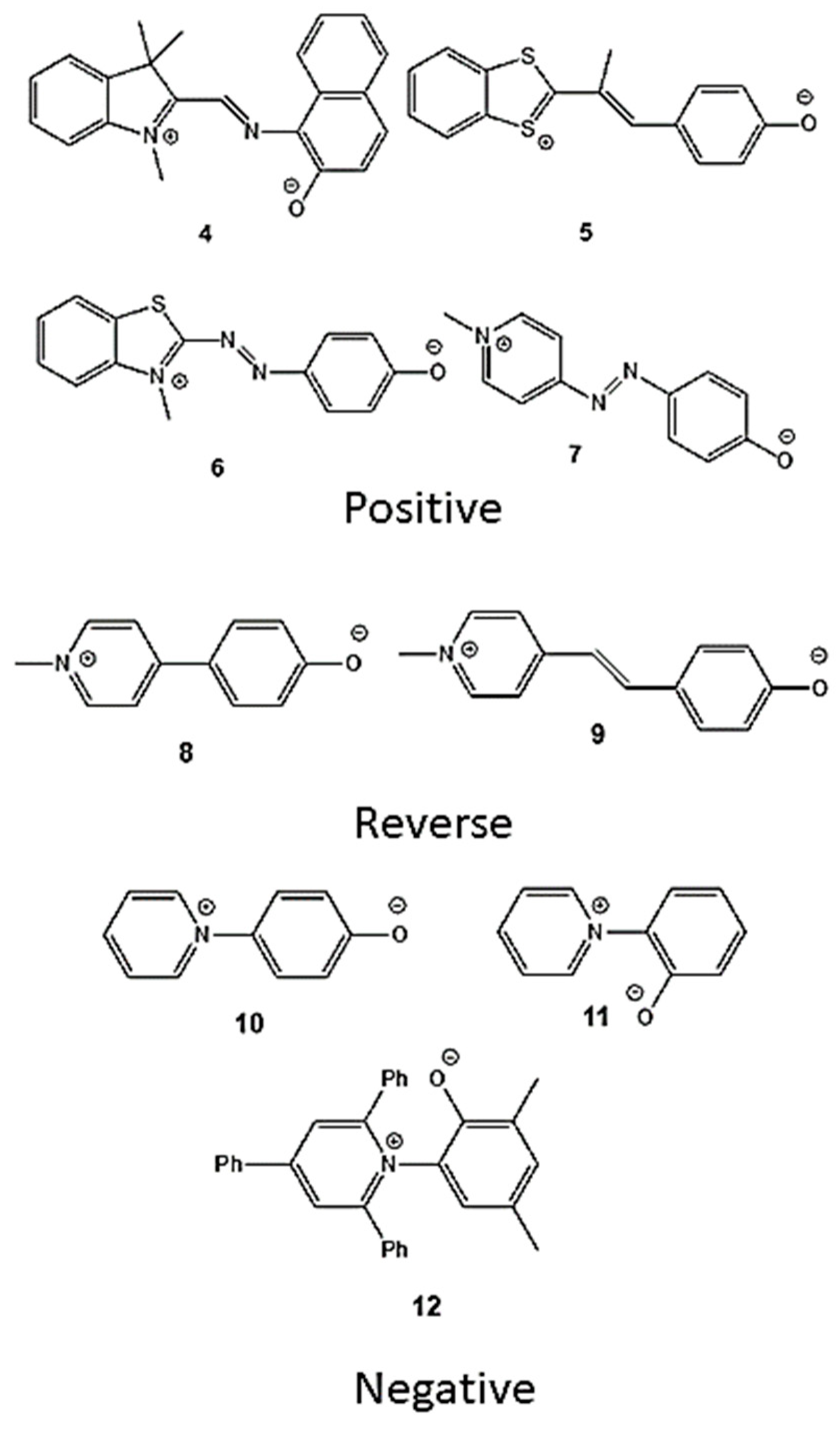
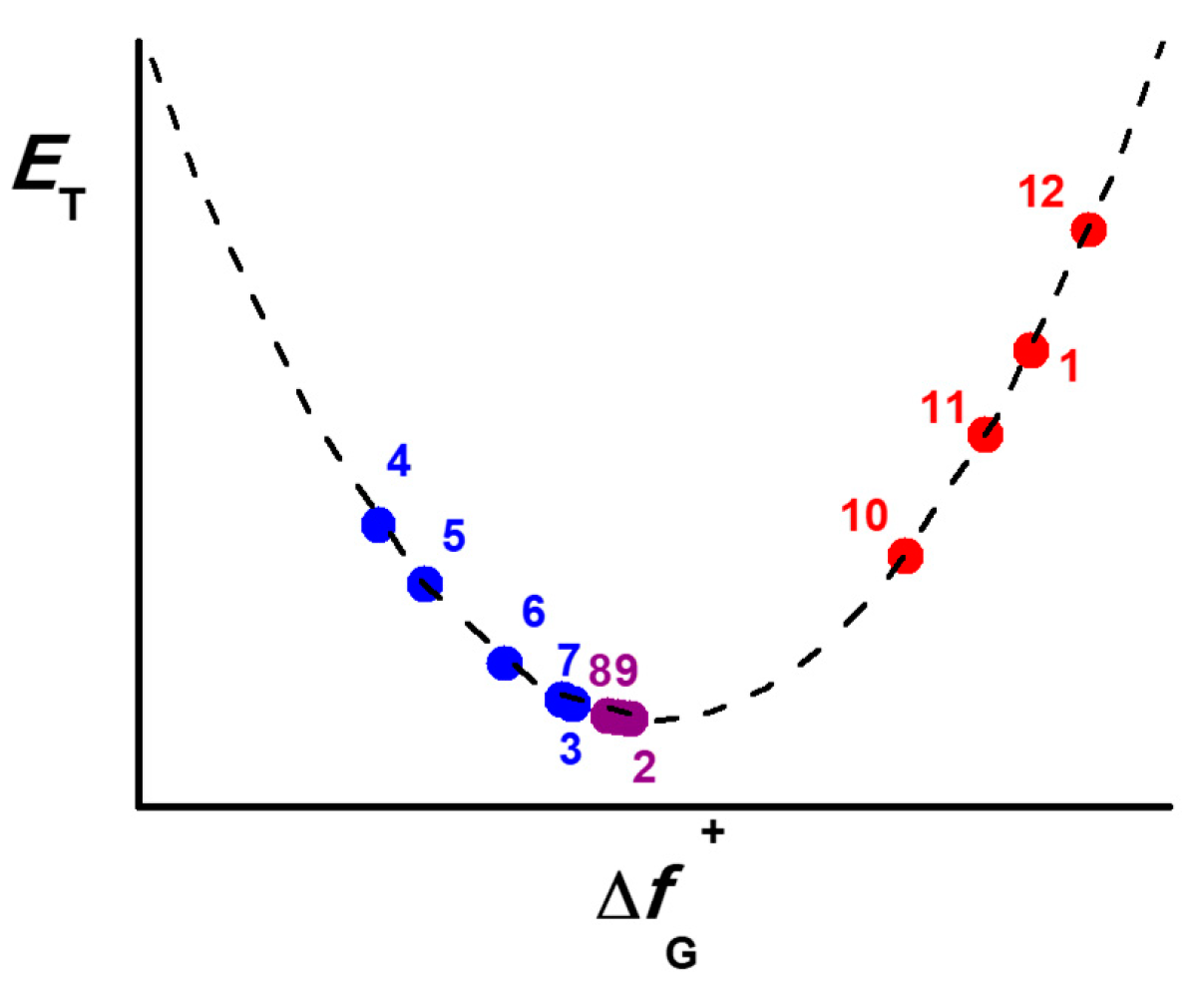
2.1. PCM Calculations of Betaines 1-3
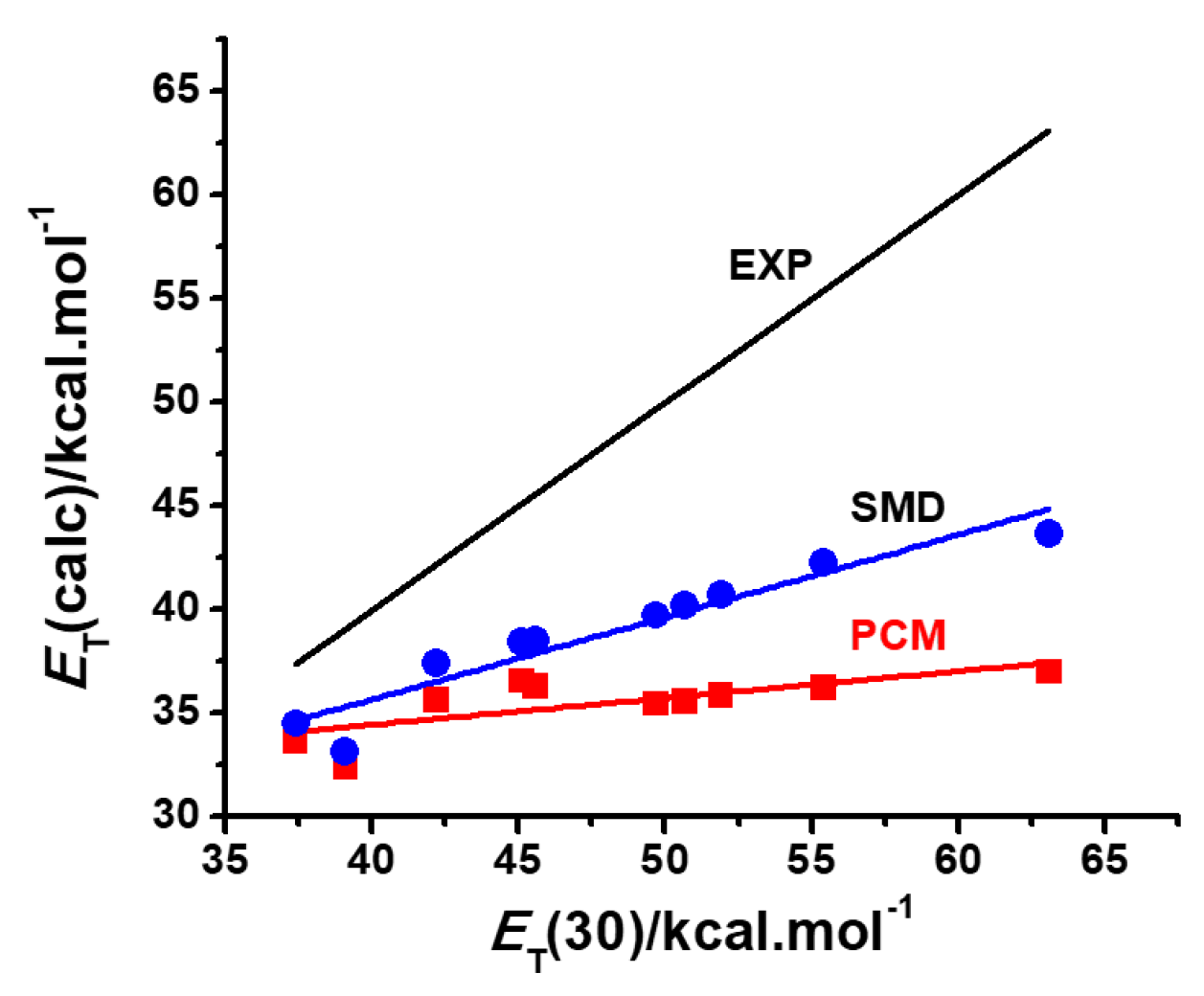
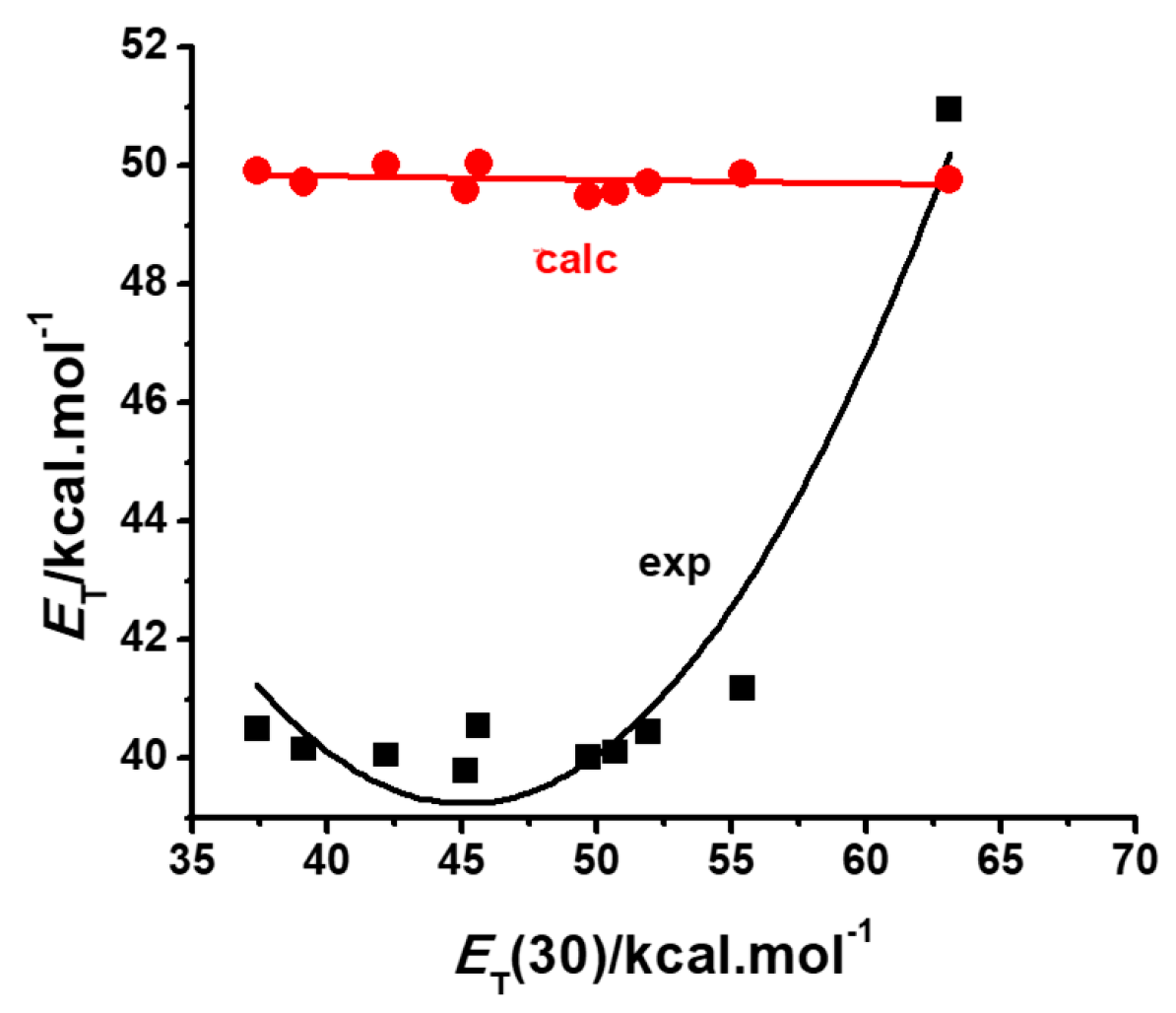
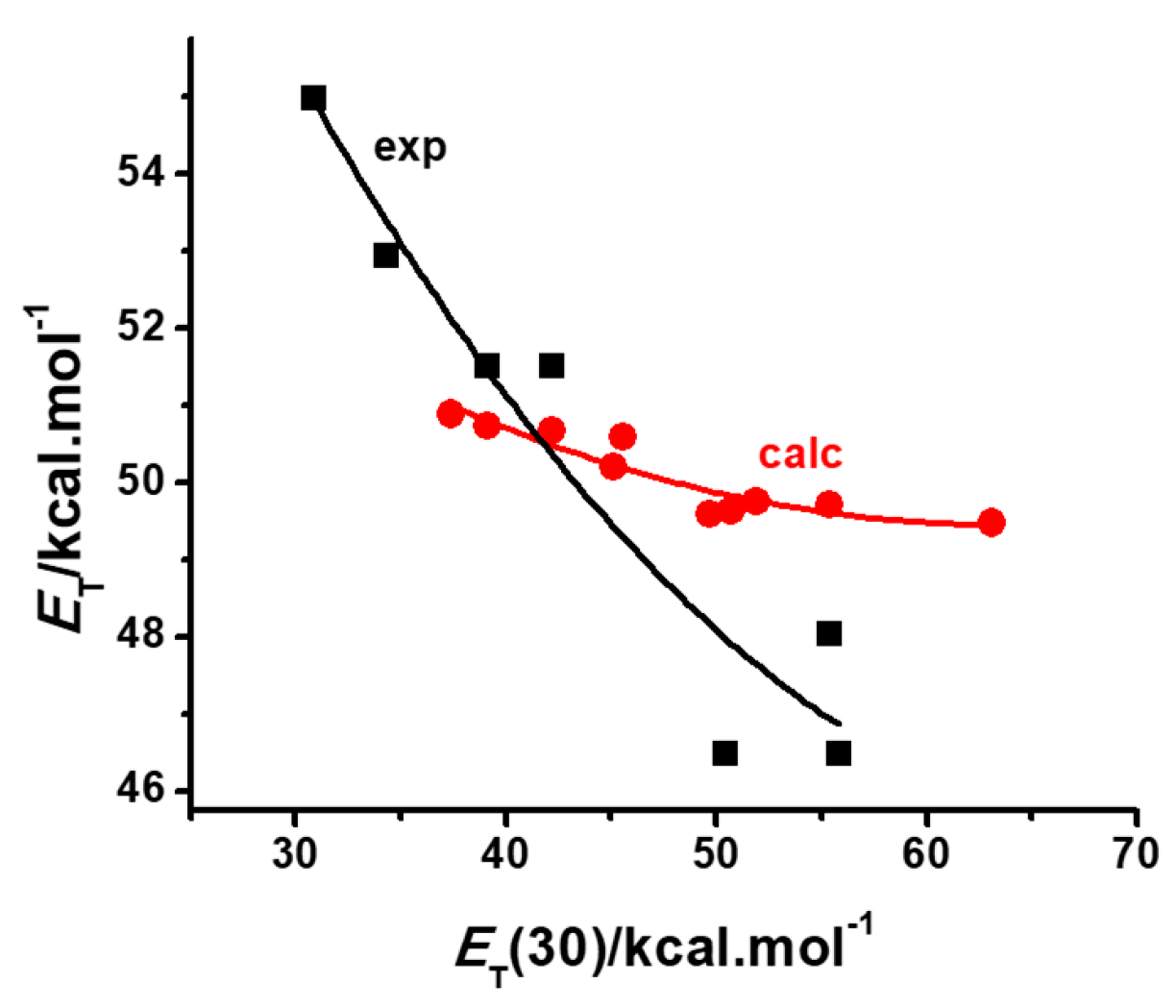
2.2. PCM and Model Validations
3. Materials and Methods
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Herbert, J. M. Dielectric continuum methods for quantum chemistry. WIREs Computational Molecular Science 2021, 11, 1–87. [Google Scholar] [CrossRef]
- Caricato, M.; Mennucci, B.; Tomasi, J. Solvent polarity scales revisited: a ZINDO-PCM study of the solvatochromism of betaine-30. Molecular Physics 2006, 104, 875. [Google Scholar] [CrossRef]
- Eilmes, A. Solvatochromic probe in molecular solvents: implicit versus explicit solvent model. Theoretical Chemistry Accounts 2014, 1538. [Google Scholar] [CrossRef]
- Rezende, M. C.; Domínguez, M. The solvatochromism of phenolate betaines: comparing different cavities of a polarized continuum model. Journal of Molecular Modeling 2015, 21, 212. [Google Scholar] [CrossRef] [PubMed]
- Baryshnikov, G. V.; Bondarchuk, S. V.; Minaeva, V. A.; Ågren, H.; Minaev, B. F. Solvatochromic effect in absorption and emission spectra of star-shaped bipolar derivatives of 1,3,5-triazine and carbazole. A time-dependent density functional study. Journal of Molecular Modeling 2017, 23, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Rezende, M. C.; Dominguez, M.; Aracena, A.; Millán, D. Solvatochromism and electrophilicity. Chemical Physics Letters 2011, 514, 267–273. [Google Scholar] [CrossRef]
- Rezende, M. C.; Aracena, A. Electrophilicity and solvatochromic reversal of pyridinium phenolate betaine dyes. Chemical Physics Letters 2012, 542, 147–152. [Google Scholar] [CrossRef]
- Rezende, M. C. A generalized reversal model for the solvatochromism of merocyanines. Journal of Physical Organic Chemistry 2016, 29, 460–467. [Google Scholar] [CrossRef]
- Aracena, A.; Domínguez, M. Computational Quantification of the Zwitterionic/Quinoid Ratio of Phenolate Dyes for Their Solvatochromic Prediction. Molecules 2022, 27, 9023. [Google Scholar] [CrossRef]
- Mera-Adasme, R.; Moraga, D.; Medina, R.; Domínguez, M. Mapping the solute–solvent interactions for the interpretation of the three types of solvatochromism exhibited by phenolate-based dyes. Journal of Molecular Liquids 2022, 359, 119302. [Google Scholar] [CrossRef]
- Catalán, J.; Díaz, C.; López, V.; Pérez, P.; de Paz, J.-L.G.; Rodriguez, J.G. A generalized solvent basicity scale: The solvatochromism of 5-nitroindoline and its homomorph 1-methyl-5-nitroindoline. Liebigs Ann. 1996, 11, 1785–1794. [Google Scholar] [CrossRef]
- Catalán, J.; Díaz, C. A generalized solvent acidity scale: The solvatochromism of o-tert-butylstilbazolium betaine dye and its homomorph o,o ′ -di-tert-butylstilbazolium betaine dye. Liebigs Ann. 1997, 9, 1941–1949. [Google Scholar] [CrossRef]
- Kamlet, M.J.; Abboud, J.L.; Taft, R.W. The solvatochromic comparison method. The π* scale of solvent polarities. J. Am. Chem. Soc 1977, 99, 6027–6038. [Google Scholar] [CrossRef]
- Catalán, J.; López, V.; Pérez, P.; Martín-Villamil, R.; Rodriguez, J.G. Progress towards a generalized solvent polarity scale: The solvatochromism of 2-(dimethylamino)-7-nitrofluorene and its homomorph 2-fluoro-7-nitrofluorene. Liebigs Ann. 1995, 2, 241–252. [Google Scholar] [CrossRef]
- Ormazábal-Toledo, R.; Rezende, M. C.; Vidal, M. The Simulation of Solvent Polarizabilities and Dipolarities with Polarizable Continuum Model. The Journal of Physical Chemistry B 2024, 128, 7208–7218. [Google Scholar] [CrossRef] [PubMed]
- Dimroth, K.; Reichardt, C.; Siepmann, T.; Bohlmann, F. Über Pyridinium-N-phenol-betaine und ihre Verwendung zur Charakterisierung der Polarität von Lösungsmitteln. Justus Liebigs Annalen Der Chemie 1963, 661, 1–37. [Google Scholar] [CrossRef]
- Reichardt, C. Solvatochromic Dyes as Solvent Polarity Indicators. Chem. Rev. 1994, 94, 2319–2358. [Google Scholar] [CrossRef]
- Machado, V.G.; Stock, R.I.; Reichardt, C. Pyridinium N-Phenolate Betaine Dyes. Chem. Rev. 2014, 114, 10429–10475. [Google Scholar] [CrossRef]
- Martins, C.T.; El Seoud, O.A. Thermo-Solvatochromism of Merocyanine Polarity Probes—What Are the Consequences of Increasing Probe Lipophilicity through Annelation? Eur. J. Org. Chem. 2008, 2008, 1165–1180. [Google Scholar] [CrossRef]
- Hünig, S.; Rosenthal, O. Farbe und Konstitution II: Phenolbetainfarbstoffe. Justus Liebigs Annalen Der Chemie 1955, 592, 161–179. [Google Scholar] [CrossRef]
- Yang, Weitao.; Mortier, W. J. The use of global and local molecular parameters for the analysis of the gas-phase basicity of amines. Journal of the American Chemical Society 1986, 108, 5708–5711. [CrossRef] [PubMed]
- Domínguez, M.; Rezende, M. C. Towards a unified view of the solvatochromism of phenolate betaine dyes. Journal of Physical Organic Chemistry 2009, 23, 156–170. [Google Scholar] [CrossRef]
- Pinheiro, J. M. F.; de Melo, C. P. Ab Initio Study of the Anomalous Solvatochromic Behavior of Large Betaines. The Journal of Physical Chemistry A 2011, 115, 7994–8002. [Google Scholar] [CrossRef]
- Rezende, M. C.; Aracena, A. A general framework for the solvatochromism of pyridinium phenolate betaine dyes. Chemical Physics Letters 2013, 558, 77–81. [Google Scholar] [CrossRef]
- Keum, S.-R.; Hur, M.-S.; Kazmaier, P. M.; Buncel, E. Thermo- and photochromic dyes: indolino-benzospiropyrans. Part 1. UV–VIS spectroscopic studies of 1,3,3-spiro(2H-1-benzopyran-2,2′-indolines) and the open-chain merocyanine forms; solvatochromism and medium effects on spiro ring formation. Canadian Journal of Chemistry 1991, 69, 1940–1947. [Google Scholar] [CrossRef]
- Le Beuze, A.; Samat, A.; Botrel, A.; Appriou, P.; Guglielmetti, R. Détermination expérimentale du moment dipolaire de mérocyanines hétérocycliques. Journal de Chimie Physique 1978, 75, 46–52. [Google Scholar] [CrossRef]
- Rajagopal, S.; Buncel, E. Synthesis and Electronic Spectral Characteristics of Some New Azo Merocyanine Dyes. Dyes Pigment 1991, 17, 303–321. [Google Scholar] [CrossRef]
- Diemer, V.; Chaumeil, H.; Defoin, A.; Jacques, P.; Carré, C. Synthesis of 4-[N-methyl-4-pyridinio]-phenolate (POMP) and negative solvatochromism of this model molecule in view of nonlinear optical applications. Tetrahedron Letters 2005, 46, 4737–4740. [Google Scholar] [CrossRef]
- Jacques, P. On the Relative Contributions of Nonspecific and Specific Interactions to the Unusual Solvatochromism of a Typical Merocyanine Dye. J. Phys. Chem. 1986, 90, 5535–5539. [Google Scholar] [CrossRef]
- Minkovska, S.; Jeliazkova, B.; Borisova, E.; Avramov, L.; Deligeorgiev, T. Substituent and solvent effect on the photochromic properties of a series of spiroindolinonaphthooxazines. Journal of Photochemistry and Photobiology A: Chemistry 2004, 163, 121–126. [Google Scholar] [CrossRef]
- Paley, M. S.; Meehan, E. J.; Smith, C. D.; Rosenberger, F. E.; Howard, S. C.; Harris, J. M. Synthesis and characterization of a novel betaine dye: 2,4-dimethyl-6-(2,4,6-triphenyl-N-pyridinio)phenolate. The Journal of Organic Chemistry 1989, 54, 3432–3436. [Google Scholar] [CrossRef]
- Frisch, M. J.; Trucks, G. W.; Schlegel, H. B.; Scuseria, G. E.; Robb, M. A.; Cheeseman, J. R.; Scalmani, G.; Barone, V.; Petersson, G. A.; Nakatsuji, H.; et al. Gaussian 16, Revision C.01. Gaussian, Inc.: Wallingford, CT, USA, 2016. [Google Scholar]
- Manzoni, V.; Coutinho, K.; Canuto, S. An insightful approach for understanding solvatochromic reversal. Chemical Physics Letters 2016, 655–656, 30–34. [Google Scholar] [CrossRef]
| Solvent | Solvent ET(30) value | Dye 1 | Dye 2a | Dye 3b | |
|---|---|---|---|---|---|
| PCM | SMD | SMD | SMD | ||
| Cyclohexane | 30.9 | - | - | - | (54.98) |
| Benzene | 34.3 | - | - | - | (52.94) |
| Tetrahydrofuran | 37.4 | 33.66 | 34.51 | 49.93 (40.50) | 50.89 |
| Chloroform | 39.1 | 32.42 | 33.13 | 49.73 (40.16) | 50.73 (51.51) |
| Acetone | 42.2 | 35.63 | 37.41 | 50.02 (40.06) | 50.68 (51.51) |
| Dimethylsulfoxide | 45.1 | 36.57 | 38.42 | 49.60 (39.78) | 50.21 |
| Acetonitrile | 45.6 | 36.30 | 38.51 | 50.05 (40.54) | 50.60 |
| 1-Butanol | 49.7 | 35.46 | 39.73 | 49.48 (40.02) | 49.60 |
| Benzyl alcohol | 50.4 | - | - | - | (46.49) |
| 1-Propanol | 50.7 | 35.60 | 40.24 | 49.56 (40.11) | 49.63 |
| Ethanol | 51.9 | 35.88 | 40.74 | 49.72 (40.44) | 49.75 |
| Methanol | 55.4 | 36.23 | 42.27 | 49.86 (41.18) | 49.71 (46.49) |
| Formamide | 55.8 | - | - | - | (46.49) |
| Water | 63.1 | 37.00 | 43.64 | 49.78 (50.96) | 49.48 |
| Compound | fG+ (phenolate) | fG+ (heterocyclic) | fG+ |
|---|---|---|---|
|
1b 2b 3b 4c 5c 6c 7c 8d 9d 10c 11c 12c |
0.17 0.54 0.57 0.74 0.70 0.63 0.58 0.48 0.53 0.28 0.21 0.12 |
0.83 0.46 0.43 0.26 0.30 0.37 0.42 0.52 0.47 0.72 0.79 0.88 |
0.66 -0.08 -0.14 -0.48 -0.40 -0.26 -0.16 -0.04 -0.06 0.44 0.58 0.76 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




