Submitted:
08 September 2024
Posted:
09 September 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Types of Knee Joint Injuries
Anterior Cruciate Ligament (ACL) Injuries
Mechanisms of Injury
Cellular and Molecular Responses to ACL Injury
Inflammatory Response
Extracellular Matrix (ECM) Degradation and Remodeling
Fibroblast Activation and Proliferation
Mesenchymal Stem Cell (MSC) Recruitment and Differentiation
Angiogenesis
Mechanotransduction and Mechanical Loading
| Category | Details | Processes Involved | Key Molecular/Cellular Players |
| ACL Function and Vulnerability | ACL stabilizes the knee by limiting excessive anterior translation of the tibia relative to the femur and controlling rotational movements. | Stabilizes the knee during dynamic activities like pivoting, jumping, and sudden stops, which put high stress on the ACL. Sports with rapid directional changes like soccer, basketball, and skiing are high-risk. | ACL, tibia, femur, dynamic activities (pivoting, jumping, sudden stops), rotational forces, anterior translation |
| Risk Factors for ACL Injuries | Common in sports with sudden directional changes and high-impact landings. Women are at higher risk due to hormonal influences, neuromuscular control differences, and anatomical factors. | Hormonal fluctuations, neuromuscular control deficits, and biomechanics like greater Q-angle in women increase susceptibility. | Hormones (estrogen, relaxin), Q-angle, neuromuscular control, biomechanics, deceleration, valgus collapse |
| Mechanisms of Injury | ACL injuries are often non-contact, accounting for 70% of cases, particularly during rapid deceleration, pivoting, or improper landing from jumps. | High knee stress due to valgus stress, rotational forces, and rapid changes in movement, often without direct contact. | Valgus stress, rotational forces, knee mechanics, pivoting, deceleration, improper landing |
| Consequences of ACL Rupture | Immediate joint instability occurs with ACL rupture, leading to increased risk of secondary injuries to the meniscus and cartilage. Long-term consequences include osteoarthritis. | Altered knee mechanics after ACL injury lead to abnormal wear patterns in the joint, increasing the likelihood of degenerative joint disease like osteoarthritis. | Meniscus, articular cartilage, joint instability, osteoarthritis, abnormal joint kinematics |
| Inflammatory Response to Injury | The body initiates a biochemical cascade involving cytokine release, immune cell recruitment, and ECM degradation. This starts with an acute inflammatory phase. | Neutrophils and macrophages clear debris; cytokines (TNF-α, IL-1β, IL-6) promote inflammation, while MMPs degrade ECM, allowing tissue repair but potentially causing excessive damage. | Cytokines (TNF-α, IL-1β, IL-6), neutrophils, macrophages (M1 to M2), MMPs (MMP-1, MMP-3, MMP-13), ECM, fibroblasts |
| Tissue Repair and Remodeling | After inflammation, the repair phase begins with fibroblast activation and ECM synthesis, particularly collagen production to restore ligament strength. | Growth factors (TGF-β, FGF, PDGF) activate fibroblasts, leading to ECM synthesis. Mechanical forces influence collagen fiber alignment for proper tissue remodeling. | Fibroblasts, growth factors (TGF-β, FGF, PDGF), collagen (type I), mechanical forces, ECM, integrins, YAP/TAZ |
| Extracellular Matrix (ECM) Degradation and Remodeling | ECM, composed of collagen, elastin, and proteoglycans, is crucial for ACL structural integrity. Injury disrupts this balance, leading to both degradation and remodeling. | MMPs degrade ECM components like collagen, while growth factors like TGF-β promote ECM synthesis. TIMPs regulate MMP activity to balance degradation and repair. | MMPs (MMP-1, MMP-3, MMP-13), TIMPs (TIMP-1, TIMP-2), TGF-β, collagen (type I), ECM, fibroblasts, proteoglycans |
| Cellular Recruitment and MSC Involvement | Mesenchymal stem cells (MSCs) are recruited to the injury site, where they differentiate into fibroblasts and other cell types essential for tissue repair. | Chemotactic signals (SDF-1, VEGF) recruit MSCs, which differentiate into fibroblasts to aid in ECM production and remodeling. MSCs also release growth factors that modulate immune responses. | MSCs, chemotactic signals (SDF-1/CXCR4, VEGF), fibroblasts, immune cells, ECM, cytokines, paracrine signaling |
| Mechanical Loading and Tissue Repair | Mechanical forces play a key role in ACL healing, influencing cellular behaviors such as collagen alignment and fibroblast activity. Proper loading can improve tissue repair, while excessive loading can cause further damage. | Mechanical forces are sensed by integrins and mechanoreceptors like YAP/TAZ, which modulate gene expression related to ECM production, collagen fiber alignment, and cell proliferation. | Integrins, mechanoreceptors (Piezo1), YAP/TAZ, focal adhesion kinase (FAK), fibroblasts, ECM, collagen alignment |
| Role of Angiogenesis in ACL Healing | Angiogenesis is crucial for delivering nutrients and oxygen to the injured tissue. Growth factors like VEGF promote new blood vessel formation, which supports tissue regeneration. | Hypoxia after injury triggers HIF-1α, which upregulates VEGF and promotes endothelial cell proliferation and migration, leading to new blood vessel formation. | VEGF, HIF-1α, endothelial cells, angiogenesis, fibroblasts, PDGF, endothelial progenitor cells (EPCs) |
| Chronic Effects of Dysregulated Healing | If inflammation is not properly resolved, or if mechanical loading is inappropriate, ACL repair can be impaired, leading to chronic instability or the development of fibrosis. | Chronic inflammation or excessive ECM degradation due to unchecked MMP activity can impair tissue regeneration, increasing the risk of long-term complications like osteoarthritis. | Chronic inflammation, fibrosis, osteoarthritis, MMPs, TIMPs, cytokines, abnormal joint kinematics |
| Therapeutic Interventions and Potential Strategies | Therapies like platelet-rich plasma (PRP) and MSC-derived exosomes aim to enhance healing by modulating inflammation and promoting tissue regeneration. Rehabilitation strategies emphasize controlled mechanical loading. | Biologics (PRP, MSC exosomes) and targeted therapies modulate the healing environment by reducing inflammation, enhancing ECM production, and promoting proper mechanical loading for optimal healing. | PRP, MSC exosomes, cytokines, growth factors, rehabilitation, mechanical loading, integrins, YAP/TAZ |
Understanding Biological and Physiological Processes
| Inflammation phase | |
| Injury/ incident | Immediate vasoconstriction of the blood flow. Immobilization to reduce pain and swelling |
| 24–48 h post | Vasodilatation and proliferation of tissue. Inflammation. Icing is not recommended as it slows down healing by decreasing lymphatic flow, proliferation, and cell– cell-interactions. Same rules apply for anti-inflammatory drugs. Ice has numbing effects and should only be used for a few minutes for pain relief |
| Proliferation phase | |
| 5 days | Type III collagen is produced and will be transferred to type I over time. Reconstruction and orientation of the type III fibers depend on stress of movement and weight bearing. That is why exercises in full range of motion allowed and weight bearing are so important to guarantee good healing of tissue and scars |
| Remodeling/ maturation | |
| ~3 weeks | Type III collagen is transferred into type I. regaining range of motion, proprioceptive and contractile information to allow good healing. Regaining biomechanical qualities of the tissue. Formation of cross-links for greater stiffness. This process is supported by load and mobilization into the end of ROM in exercises. 300–500 days until tissue regains its former function |
Molecular Description of Ligament Healing Process.
- Injury incydent (0h).
- 24–48 h after ACL injury.
- 5 days after ACL injury.
- ~3 weeks post ACL injury.
Meniscal Injuries
Mechanisms of Injury
Cellular Responses to Meniscal Injury
Initial Inflammatory Response
Extracellular Matrix Degradation and Remodeling
Chondrocyte and Fibrochondrocyte Activation
Mesenchymal Stem Cell (MSC) Recruitment and Differentiation
Angiogenesis
Mechanotransduction and Mechanical Loading
| Category | Details | Processes Involved | Key Molecular/Cellular Players |
| Meniscus Structure and Function | The meniscus is a fibrocartilaginous structure located between the femoral condyles and tibial plateau in the knee joint. It plays a crucial role in load distribution, shock absorption, joint stability, and lubrication. | The meniscus spreads compressive forces, reducing stress on articular cartilage and maintaining knee joint function. | Meniscus, fibrocartilage, type I collagen, proteoglycans (aggrecan), fibrochondrocytes, ECM (extracellular matrix) |
| Meniscal Injury Types | Meniscal injuries can result from acute trauma (e.g., twisting motion) or degenerative changes (wear and tear over time). Acute injuries are common in athletes, while degenerative injuries are more prevalent in older adults. | Acute injuries lead to tears, while degenerative injuries cause fragmentation or fraying. Both result in knee pain, swelling, and mechanical symptoms (e.g., locking, instability). | Meniscal tears, acute trauma, degeneration, knee joint instability, swelling, pain, locking |
| Healing Capacity Based on Vascular Zones | The meniscus has different healing capacities depending on its vascular zones: the red-red (vascularized), red-white (partially vascularized), and white-white (avascular) zones. | The outer (red-red) zone has better healing potential due to vascular supply, while the inner (white-white) zone has limited healing ability due to avascularity. | Red-red zone, white-white zone, blood supply, vascularization, synovial fluid, meniscal healing |
| Cellular Response to Injury | Meniscal injury triggers fibrochondrocytes to produce enzymes (MMPs) that degrade ECM components, aiming to remodel damaged tissue. Inflammatory cytokines (e.g., IL-1β, TNF-α) are upregulated, promoting tissue breakdown. | Increased synthesis of proteolytic enzymes (MMPs) and cytokines drives ECM degradation. However, excessive degradation impairs tissue repair. | MMPs (MMP-1, MMP-3, MMP-13), inflammatory cytokines (IL-1β, TNF-α, IL-6), ECM, fibrochondrocytes |
| Extracellular Matrix (ECM) Changes | ECM composition shifts after injury, with a decrease in collagen and proteoglycan synthesis, leading to reduced biomechanical properties. This loss of elasticity compromises shock absorption and load distribution. | Reduced synthesis of collagen and proteoglycans results in diminished ability to retain water, further weakening the meniscus. | ECM, collagen (type I, type II), proteoglycans, elastin, glycosaminoglycans (GAGs), fibrotic tissue |
| Inflammatory Response and ECM Breakdown | Meniscal injuries trigger the release of DAMPs and the recruitment of immune cells (e.g., macrophages, neutrophils), which exacerbate ECM degradation. Chronic inflammation can lead to fibrosis and joint degeneration. | Immune cell infiltration and cytokine release activate inflammatory pathways (e.g., NF-κB), amplifying the degradation of the ECM. | DAMPs, macrophages, neutrophils, pro-inflammatory cytokines (TNF-α, IL-1β, IL-6), NF-κB, ECM |
| Chondrocyte and Fibrochondrocyte Role in Repair | Chondrocytes in articular cartilage respond to meniscal injury by producing ECM components (e.g., collagen X) but may also contribute to cartilage degeneration, particularly in osteoarthritis. | Hypertrophic chondrocytes produce collagen X and MMPs, while fibrochondrocytes in the meniscus increase collagen and ECM synthesis in response to injury. | Chondrocytes, fibrochondrocytes, collagen (type I, type II, type X), MMPs, vascular endothelial growth factor (VEGF) |
| Molecular Signaling Pathways in Healing | TGF-β signaling plays a dual role, promoting ECM synthesis and fibroblast proliferation but also driving fibrosis if overactivated. Hypoxia-induced factors (HIFs) promote limited neovascularization in the avascular zones. | TGF-β promotes collagen synthesis through SMAD signaling, while hypoxia activates HIFs, inducing VEGF and angiogenesis attempts. | TGF-β, SMAD, hypoxia-inducible factors (HIF-1α), VEGF, fibroblasts, collagen synthesis, fibrosis |
| Therapeutic Strategies for Meniscal Repair | Biological therapies (e.g., PRP, MSCs) aim to stimulate meniscal healing, especially in avascular zones, by enhancing ECM synthesis, reducing inflammation, and promoting chondrocyte activity. | Platelet-rich plasma (PRP) and mesenchymal stem cells (MSCs) support regeneration by providing growth factors that stimulate chondrocyte activity and ECM repair. | PRP, MSCs, growth factors (TGF-β, FGF, IGF-1), chondrocytes, ECM, biological scaffolds |
| Mechanisms of Meniscal Injury | Meniscal injuries are caused by rotational forces, direct impacts, and repetitive microtrauma, common in high-impact sports (e.g., football, basketball). Degenerative tears are more common in older adults. | Acute injuries result from sudden twisting or deceleration, while degenerative tears are due to cumulative microtrauma and aging-related tissue degradation. | Rotational forces, direct impact, meniscal tears, degenerative changes, high-impact sports, aging |
| Meniscal Healing and Surgical Interventions | Tears in the red-red zone are more likely to heal with conservative treatments or suturing. Tears in the white-white zone may require meniscectomy due to poor healing potential. | Surgical options like meniscal suturing and meniscectomy are determined by tear location and the meniscus’s vascular supply. | Meniscal suturing, meniscectomy, vascular supply, red-red zone, white-white zone, conservative treatment |
Articular Cartilage and Its Functionality with Molecular Biology Insights
Cartilage Injuries: Causes and Types
Cellular Responses to Cartilage Injury
Inflammatory Response to Cartilage Injury
Extracellular Matrix Remodeling and Repair
Chondrocyte Responses to Injury
Mesenchymal Stem Cell (MSC) Recruitment and Differentiation
Angiogenesis and Subchondral Bone Response
Mechanotransduction and Mechanical Loading
| Category | Details | Processes Involved | Key Molecular/Cellular Players |
| Articular Cartilage Structure | Articular cartilage is found on the surfaces of bones in synovial joints, providing frictionless movement and efficient load distribution. It consists of chondrocytes embedded in an extracellular matrix (ECM) composed of type II collagen and proteoglycans like aggrecan. The ECM’s unique properties enable the cartilage to resist mechanical forces such as compression and shear stress. | The ECM consists of type II collagen for tensile strength and proteoglycans, particularly aggrecan, to retain water for compressive resistance. Cartilage is stratified into superficial, middle, and deep zones, each with varying collagen fiber orientation and chondrocyte density, which contribute to the tissue’s mechanical functionality. | Chondrocytes maintain the ECM by synthesizing collagen and proteoglycans. Type II collagen provides tensile strength, while aggrecan retains water. Glycosaminoglycans (GAGs), like chondroitin sulfate, enable water retention. Superficial zone chondrocytes handle shear forces, while deep zone chondrocytes remodel the ECM. |
| Limited Regenerative Capacity | Articular cartilage has limited healing ability due to its avascularity, meaning it lacks a direct blood supply. Nutrient delivery and waste removal rely on diffusion from synovial fluid. Injuries lead to insufficient chondrocyte proliferation and slow synthesis of ECM components, often resulting in incomplete repair or progressive degeneration, such as in osteoarthritis (OA). | Cartilage injuries trigger a slow repair response due to limited availability of growth factors and progenitor cells. Matrix metalloproteinases (MMPs) degrade the ECM, particularly collagen and aggrecan, and if left unchecked, can lead to further breakdown. Inflammatory cytokines like IL-1β and TNF-α exacerbate ECM degradation and inhibit new matrix synthesis. | MMPs degrade ECM components, while tissue inhibitors of metalloproteinases (TIMPs) regulate this activity. IL-1β and TNF-α upregulate MMPs and inhibit matrix synthesis, worsening tissue degeneration. Chondrocytes have limited ability to proliferate and migrate, further limiting repair. Synovial fluid is critical for nutrient delivery in the absence of blood supply. |
| Cartilage Injury Types | Cartilage injuries are either focal (localized, often caused by acute trauma) or diffuse (widespread degeneration due to conditions like osteoarthritis). Focal injuries are common in athletes and can evolve into broader degeneration if untreated. Diffuse injuries, associated with OA, result from chronic wear and tear, leading to progressive loss of cartilage and joint dysfunction. | Focal injuries arise from mechanical trauma and often lead to cartilage defects that expose subchondral bone, causing joint friction and pain. Diffuse injuries like OA involve pro-inflammatory cytokines (IL-1β, TNF-α) that promote the breakdown of ECM by MMPs and aggrecanases, leading to joint degeneration. Reactive oxygen species (ROS) contribute to chondrocyte apoptosis and matrix damage. | Pro-inflammatory cytokines (IL-1β, TNF-α) are key drivers of inflammation and cartilage degradation. MMPs and aggrecanases (ADAMTS-4, ADAMTS-5) degrade type II collagen and aggrecan. Reactive oxygen species (ROS) contribute to oxidative stress and further tissue damage. Chondrocytes experience apoptosis and reduced ability to maintain ECM in injured cartilage. |
| Inflammatory Response to Injury | Cartilage injury triggers an inflammatory response, with the release of cytokines (IL-1β, TNF-α) that initiate tissue repair but can exacerbate damage if prolonged. This inflammation activates catabolic pathways that degrade the ECM, especially type II collagen and aggrecan, leading to tissue degeneration in conditions like osteoarthritis. | Inflammatory cytokines activate signaling pathways such as NF-κB and MAPK, which upregulate the production of MMPs and aggrecanases, driving ECM degradation. ROS and nitric oxide (NO) further damage chondrocytes and impair ECM synthesis. Excessive degradation disrupts chondrocyte mechanotransduction, leading to a loss of mechanical cues that regulate ECM production and survival. | Cytokines (IL-1β, TNF-α) upregulate MMPs and aggrecanases, promoting matrix breakdown. NF-κB and MAPK pathways mediate the inflammatory response and ECM degradation. ROS and NO drive oxidative stress and chondrocyte apoptosis, exacerbating tissue damage. Chondrocytes lose mechanotransduction abilities, further impairing ECM repair. |
| Extracellular Matrix Remodeling | Cartilage repair involves balancing ECM degradation and synthesis. During injury, MMPs and aggrecanases degrade key ECM components, but this is counteracted by anabolic signaling pathways like TGF-β, IGF-1, and BMPs, which promote matrix synthesis. However, prolonged inflammation and oxidative stress tip this balance towards degradation, leading to progressive cartilage breakdown. | MMPs and aggrecanases degrade type II collagen and aggrecan during injury, while growth factors like TGF-β, IGF-1, and BMPs promote ECM synthesis. The Smad signaling pathway, activated by TGF-β, stimulates the production of cartilage ECM components. Oxidative stress and excessive inflammation can impair ECM synthesis, pushing cartilage toward degeneration. | MMPs and ADAMTS enzymes drive ECM degradation. TGF-β, IGF-1, and BMPs promote ECM repair through anabolic pathways like Smad signaling. ROS and NO impair chondrocyte function and matrix synthesis. Chondrocytes produce matrix components in response to growth factors but are limited by chronic inflammation and oxidative damage. |
| Therapeutic Strategies | Recent advances in cartilage repair focus on overcoming its limited regenerative capacity. Tissue engineering with scaffolds, stem cell therapies (particularly mesenchymal stem cells (MSCs)), and growth factor delivery (like TGF-β, IGF-1) are being explored to enhance cartilage regeneration and promote ECM synthesis. | Stem cell therapies aim to differentiate MSCs into chondrocytes that can synthesize ECM components. Scaffolds provide a framework for cells to regenerate tissue. Growth factors like TGF-β and IGF-1 stimulate chondrocyte activity, enhancing collagen and proteoglycan synthesis. MSC-derived exosomes and growth factors have paracrine effects that modulate inflammation and repair. | Mesenchymal stem cells (MSCs) differentiate into chondrocytes and secrete growth factors like TGF-β and IGF-1 to promote repair. Scaffolds provide structural support for tissue regeneration. MSCs also secrete exosomes containing anti-inflammatory molecules, aiding in cartilage healing. Growth factors enhance ECM synthesis and chondrocyte activity. |
| Mechanotransduction in Cartilage | Cartilage cells respond to mechanical loading through mechanotransduction pathways, where external forces are converted into biochemical signals that regulate ECM synthesis, repair, and cell survival. Integrins, ion channels, and mechanosensitive signaling pathways like FAK, MAPK, and YAP/TAZ play critical roles in directing cellular responses to mechanical stimuli. | Integrins activate FAK and PI3K/Akt signaling in response to mechanical stress, promoting cell survival and ECM synthesis. Ion channels like TRPV4 mediate calcium influx, triggering signaling pathways that regulate matrix production. MAPK and YAP/TAZ pathways also respond to mechanical cues, driving anabolic processes that maintain cartilage structure. Excessive mechanical stress can shift these pathways towards catabolic activities, promoting ECM degradation. | Integrins activate mechanotransduction pathways like FAK and PI3K/Akt, which regulate ECM synthesis and survival. TRPV4 and Piezo channels mediate calcium signaling in response to mechanical loading. MAPK and YAP/TAZ pathways influence chondrocyte activity in response to mechanical stress. Excessive loading can activate MMPs and drive cartilage degeneration. |
Cellular Responses to Knee Joint Injuries
Inflammation and Immune Response
Extracellular Matrix (ECM) Remodeling
Chondrocyte Responses
Synoviocyte Activation
Fibroblast Activation and Proliferation
Mesenchymal Stem Cell (MSC) Recruitment and Differentiation
Angiogenesis
| Cell Type/Process | Response to Knee Joint Injury | Key Molecular Pathways | Role in Tissue Repair |
| Chondrocytes | Shift from a quiescent to an active state after injury, producing ECM components like type II collagen and aggrecan. Also release catabolic enzymes (MMP-13, ADAMTS-5) that degrade the ECM. Hypoxia in cartilage activates HIF-1α, which influences both anabolic (repair) and catabolic (degradation) processes. | TGF-β/SMAD: Promotes ECM synthesis; HIF-1α: Regulates response to hypoxia; MMP-13, ADAMTS-5: Degrade ECM; VEGF: Promotes angiogenesis, influencing subchondral bone. | Repair ECM through collagen and proteoglycan production, but also contribute to degradation via MMP-13 and ADAMTS-5. Regulate balance between ECM synthesis and degradation. Participate in cartilage degeneration in osteoarthritis and other chronic conditions when not properly regulated. |
| Fibroblasts | Fibroblasts proliferate and migrate to injury sites, synthesizing ECM components like type I collagen and proteoglycans. They also align newly formed collagen fibers in response to mechanical cues. Produce MMPs to degrade damaged ECM and facilitate new tissue formation. | TGF-β/SMAD: Induces ECM production; PI3K/AKT, FAK: Regulate proliferation and migration; MAPK/ERK: Controls gene expression for ECM synthesis. | Produce collagen and other ECM components necessary for ligament and tendon repair. Regulate collagen fiber organization. Contribute to ECM remodeling by balancing matrix degradation and synthesis. Mechanotransduction pathways guide their alignment of collagen based on mechanical stress. |
| Macrophages | Initiate the inflammatory response by releasing pro-inflammatory cytokines (TNF-α, IL-1β) in their M1 phenotype. Transition to the M2 phenotype as healing progresses, secreting growth factors (TGF-β, PDGF) to promote tissue repair. Macrophages also clear debris via phagocytosis and promote fibroblast activity for ECM remodeling. | TGF-β, PDGF: Induce tissue repair; NF-κB, JAK/STAT: Regulate cytokine production; MMP-9: Break down ECM for repair. | Early-stage macrophages (M1) promote inflammation, while later-stage macrophages (M2) support repair and ECM synthesis. Macrophages regulate the inflammatory response, clear necrotic tissue, and stimulate fibroblasts and chondrocytes to produce collagen and other ECM proteins essential for healing. |
| Mesenchymal Stem Cells (MSCs) | MSCs are recruited to injury sites via chemotactic signals like SDF-1 and VEGF. They differentiate into repair-specific cell types such as chondrocytes, fibroblasts, and osteoblasts. MSCs also secrete paracrine factors (IL-10, TGF-β) and exosomes that modulate inflammation, reduce apoptosis, and enhance ECM production by resident cells. | SDF-1/CXCR4, VEGF: Recruit MSCs; TGF-β/SMAD, Wnt/β-catenin: Drive chondrogenic and osteogenic differentiation; PI3K/AKT, ERK/MAPK: Control cell survival and migration. | MSCs contribute to tissue repair by differentiating into key cell types (chondrocytes, fibroblasts). Their paracrine signaling modulates inflammation, enhances tissue regeneration, and promotes ECM synthesis. Their exosomes and microRNAs help regulate surrounding cells’ behavior to optimize tissue recovery. |
| Extracellular Matrix (ECM) Remodeling | Injured tissues undergo ECM degradation to clear damaged components, driven by matrix metalloproteinases (MMPs) like MMP-13 and aggrecanases (ADAMTS-5). Tissue inhibitors of metalloproteinases (TIMPs) regulate MMP activity to prevent excessive ECM breakdown. Growth factors stimulate the synthesis of new ECM components like collagen and proteoglycans. | MMP-13, ADAMTS: Degrade ECM; TIMPs: Inhibit MMPs to regulate ECM turnover; TGF-β, IGF-1, FGF: Promote new ECM synthesis. | Clear damaged ECM, enabling new matrix formation. TIMPs ensure a controlled balance between ECM degradation and synthesis. Growth factors stimulate fibroblasts and chondrocytes to synthesize new ECM, restoring tissue integrity and function after injury. Excessive MMP activity leads to tissue degeneration. |
| Inflammation and Immune Response | The initial inflammatory response involves neutrophils, which release reactive oxygen species (ROS) and cytokines (TNF-α, IL-1β), followed by macrophages that regulate inflammation and repair. Prolonged inflammation, if uncontrolled, leads to chronic joint damage. Lymphocytes, including T cells, regulate the immune response to limit excessive inflammation. | TNF-α, IL-1β: Drive inflammation; NF-κB, MAPK: Regulate inflammatory response; PI3K/AKT: Modulates cell survival; TGF-β, IL-10: Control inflammation and repair. | Orchestrates the body’s response to injury by recruiting immune cells and regulating cytokine production. Balances pro-inflammatory and anti-inflammatory signals to initiate repair while avoiding excessive tissue damage. Proper immune regulation ensures a transition from inflammation to tissue healing. |
| Angiogenesis | New blood vessels form in response to injury, driven by VEGF, FGF, and PDGF signaling. These blood vessels supply oxygen and nutrients to the repair site. Excessive angiogenesis can lead to pathological changes in cartilage and subchondral bone, exacerbating cartilage degeneration in chronic conditions. | VEGF/VEGFR: Induce endothelial cell migration and proliferation; PI3K/AKT, ERK/MAPK: Promote survival and migration; PDGF: Stabilize new blood vessels. | Provides necessary blood supply for healing, enhancing oxygen and nutrient delivery. Supports tissue regeneration, but excessive or poorly regulated angiogenesis can contribute to pathological changes, such as bone sclerosis and cartilage breakdown in conditions like osteoarthritis. |
Mechanical Loading and Cellular Mechanisms
Mechanotransduction Pathways
Integrin Signaling
Ion Channels and Calcium Signaling
MAPK Pathway
ECM Synthesis and Remodeling
Cell Proliferation and Differentiation
Inflammation and Immune Response
Angiogenesis
| Category | Molecular Components | Mechanism of Action | Cellular/Tissue Response |
| Integrin Signaling | Integrins (α and β subunits), Focal Adhesion Kinase (FAK), Src Kinases, PI3K/Akt, RhoA, MAPK, Paxillin, P130Cas | Mechanical loading induces integrin clustering at focal adhesions, activating FAK. FAK phosphorylates Src and recruits downstream effectors, including paxillin and p130Cas, which activate MAPK and PI3K/Akt pathways. This signaling cascade regulates cytoskeletal dynamics, cell adhesion, and migration, while promoting ECM synthesis. | Increased fibroblast adhesion, migration, and survival. Enhanced ECM production (collagen, fibronectin) and cytoskeletal reorganization contribute to stronger tissue integrity and wound healing. |
| Mechanosensitive Ion Channels | Piezo1, TRPV4, ASICs (Acid-Sensing Ion Channels), Calcium (Ca²⁺), Calmodulin, CaMK (Ca²⁺/calmodulin-dependent protein kinase), Calcineurin/NFAT | Mechanical stress opens ion channels (Piezo1, TRPV4), allowing Ca²⁺ influx. Ca²⁺ binds to calmodulin, activating CaMK, calcineurin, and subsequent dephosphorylation of NFAT. This regulates gene expression related to ECM production, cellular proliferation, and adaptation to mechanical forces. | Increased intracellular Ca²⁺ levels drive NFAT translocation to the nucleus, upregulating genes for ECM remodeling, chondrocyte activity, and mesenchymal stem cell (MSC) differentiation. |
| MAPK Pathway | ERK1/2, JNK, p38 MAPK, Raf, MEK, Elk-1, c-Fos, AP-1, MEF2, ATF2 | Integrin-mediated mechanical stress activates MAPK signaling, including ERK1/2, JNK, and p38 MAPK. ERK1/2 promotes cell proliferation and survival by phosphorylating transcription factors such as Elk-1 and c-Fos. JNK is activated by mechanical and oxidative stress, influencing AP-1 activity, while p38 MAPK phosphorylates transcription factors like ATF2 and MEF2, regulating stress responses and inflammation. | ERK1/2 promotes collagen synthesis, cell proliferation, and differentiation. JNK and p38 MAPK are activated in response to excessive mechanical loading, regulating inflammation, apoptosis, and tissue repair processes. |
| ECM Synthesis and Remodeling | Collagen Types I, III, and II, Proteoglycans (Aggrecan, Decorin), Lysyl Oxidase, Matrix Metalloproteinases (MMP-2, MMP-9, MMP-13), Tissue Inhibitors of Metalloproteinases (TIMPs), Smad2/3, TGF-β | Mechanical forces activate TGF-β/Smad2/3 and PI3K/Akt signaling, increasing collagen and proteoglycan synthesis. Lysyl oxidase catalyzes cross-linking of collagen fibers, enhancing ECM tensile strength. MMPs degrade damaged ECM, while TIMPs control MMP activity to prevent excessive breakdown. | Enhanced production of type I and III collagen strengthens ECM structure, while proteoglycans maintain compressive strength. Balanced MMP and TIMP activity allows controlled ECM remodeling during tissue repair. |
| Cell Proliferation & Differentiation | Fibroblasts, Mesenchymal Stem Cells (MSCs), YAP/TAZ, Wnt/β-catenin, Rho GTPases (RhoA, Rac1, Cdc42), TGF-β, PDGF, Smad2/3, NFAT | Mechanical loading activates YAP/TAZ and Wnt/β-catenin pathways, promoting MSC differentiation into fibroblasts, chondrocytes, and osteoblasts. Growth factors (TGF-β, PDGF) trigger Smad2/3 signaling, enhancing fibroblast proliferation and ECM production. Rho GTPases regulate cytoskeletal dynamics, influencing cell shape and migration. | Increased fibroblast proliferation enhances ECM production. MSC differentiation into chondrocytes and osteoblasts accelerates tissue regeneration, while cytoskeletal remodeling supports cell migration and wound healing. |
| Inflammatory Response | NF-κB, PI3K/Akt, IL-1β, TNF-α, IL-6, IL-10, TGF-β, M1/M2 Macrophages, Chemokines (CCL2, CXCL8), Reactive Oxygen Species (ROS), Calcineurin | Mechanical loading modulates cytokine production, balancing pro-inflammatory (IL-1β, TNF-α) and anti-inflammatory (TGF-β, IL-10) signals. NF-κB regulates pro-inflammatory gene transcription, while PI3K/Akt and calcineurin pathways support anti-inflammatory responses. Chemokines recruit immune cells, and mechanical signals polarize macrophages to the reparative M2 phenotype. | Enhanced immune cell recruitment (macrophages, neutrophils) supports tissue repair and controlled inflammation. M2 macrophages secrete growth factors like PDGF and VEGF, promoting angiogenesis and ECM remodeling. |
| Angiogenesis | VEGF, VEGFR-2, Hypoxia-Inducible Factor-1α (HIF-1α), Nitric Oxide (NO), eNOS, FAK, MMPs (MMP-2, MMP-9), Angiopoietins (Ang-1, Ang-2), PDGF, RhoA | Mechanical loading upregulates VEGF via HIF-1α stabilization and PI3K/Akt signaling. VEGF binds to VEGFR-2 on endothelial cells, promoting proliferation, migration, and capillary formation. FAK and MMP activity facilitate endothelial cell migration through ECM remodeling. Nitric oxide produced by eNOS enhances blood flow and vascular permeability. Angiopoietins regulate vessel stability. | Increased vascularization ensures adequate oxygen and nutrient delivery to regenerating tissues. PDGF recruits pericytes for vessel stabilization, while MMP-mediated ECM degradation allows new capillary formation. |
Rehabilitation Strategies Based on Musculoskeletal Healing Stages: Early Mechanical Loading
Inflammation Stage
Fibroblastic Stage
Remodeling Stage
| Healing stage | Cellular phase | Biophysical characteristics | Therapeutic intervention |
| Inflammation Stage | Vasodilation, invasion of platelets, and inflammatory cells (neutrophils, monocytes, and macrophages) are crucial processes in the body’s response to injury. These events are orchestrated by a complex interplay of chemical mediators, including histamine, bradykinin, and PGE2, each playing specific roles at the molecular level to injury, facilitating effective tissue repair and restoration of function. |
Swelling, erythema, warmth, pain | Cryotherapy, preferably with compression NSAIDs (unless contraindicated) Manual therapy |
| The strength of the scar depends on the temporary clot and stitches | Methods: electrical stimulation, laser therapy, ultrasound, PEMF, ESWT, isometric and BFR training. | ||
| Fibroblastic stage. |
Growth factors such as Transforming Growth Factor-beta 1 (TGF-β1), Bone Morphogenetic Proteins (BMP), and Connective Tissue Growth Factor (CTGF) play critical roles in wound healing by activating fibroblastic cells. Upon activation, these fibroblastic cells undergo proliferation and upregulate the synthesis of extracellular matrix (ECM) components including collagen, fibronectin, and proteoglycans. |
Expression of inflammatory markers | Manual therapy: passive range of motion, soft tissue mobilization, joint mobilization |
| The scar begins to gain tensile strength | Methods: electrical stimulation, laser therapy, ultrasound, PEMF, ESWT Therapeutic exercises: prescribed to achieve the goal of full weight bearing on the surgical limb while protecting the tissues (slow eccentric tempo) |
||
| Remodelling stage. |
The remodeling of the scar improves the organization and mechanical properties of the extracellular matrix (ECM) through a dynamic process involving the coordinated activity of various cells, enzymes, and signaling pathways. Fibroblasts and myofibroblasts play key roles in this process by synthesizing and remodeling collagen and other ECM components. |
The inflammation should subside; pain, if present, may be due to osteoarthritis, DOMS, re-damage to healing tissue | Manual therapy depending on needs, based on the patient’s assessment of the operated limb and the rest of the body; passive and active range of motion, soft tissue mobilization, including scar mobilization, joint mobilization |
| Methods: Typically discontinued at this stage unless patient assessment indicates special requirements for the surgical limb or rest of the body Therapeutic exercises: prescribed to increase active ROM and flexibility, build muscle strength and endurance, improve proprioception, motor control, and improve cardiovascular fitness | |||
| Abbreviations: BMP, bone morphogenetic protein; CTGF, connective tissue growth factor; DOMS, delayed onset muscle soreness; ECM, extracellular matrix; ESWT, extracorporeal shock wave therapy; NSAIDs, non-steroidal anti-inflammatory drugs; PEMF, pulsed electromagnetic field therapy; BFR, blood flow restriciton; PGE2, prostaglandin E2; ROM, range of motion; TGF-β1, transforming growth factor-β1. | |||
Early Mechanical Loading: Benefits and Risks
Benefits of Early Mechanical Loading
- 1. Enhanced Extracellular Matrix (ECM) Synthesis
- 2. Promotion of Cell Proliferation and Differentiation
- 3. Modulation of Inflammatory Responses
- 4. Enhanced Angiogenesis
- 5. Improved Functional Recovery
Risks and Considerations
- 1. Risk of Exacerbating Injury
- 2. Inflammation and Tissue Damage
- 3. Individual Variability
- 4. Monitoring and Adjustment
- 5. Rehabilitation Protocol Design
Clinical Guidelines and Recommendations
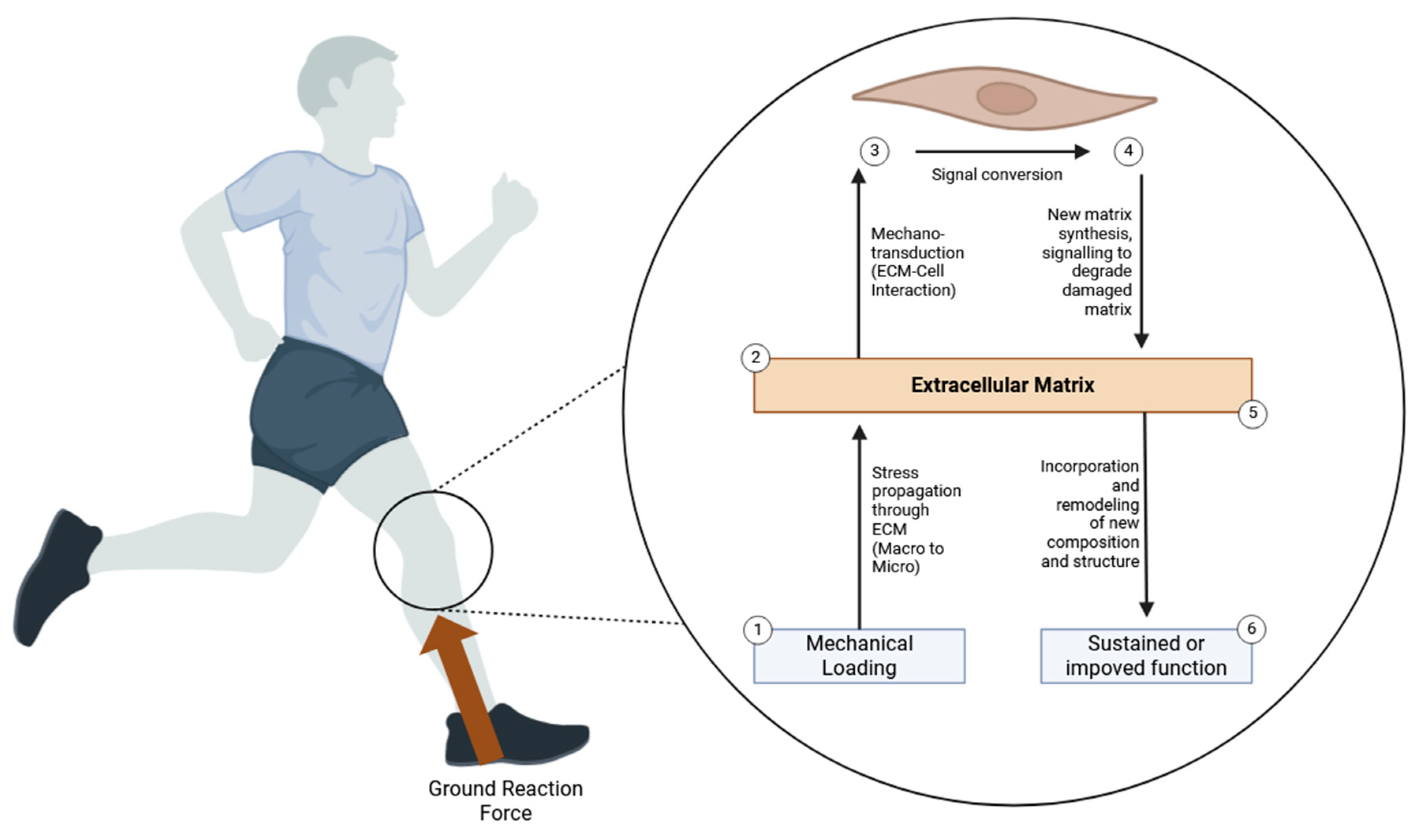
Signaling Pathways Involved in Mechanical Loading
Signaling Pathways Involved in Mechanical Loading
Integrin Signaling Pathway
Ion Channels and Calcium Signaling
Mitogen-Activated Protein Kinase (MAPK) Pathway
Wnt/β-Catenin Signaling Pathway
YAP/TAZ Signaling Pathway
Interactions among Signaling Pathways
| Signaling Pathway | Mechanism of Activation | Downstream Effects | Cellular Response |
| Integrin Signaling Pathway | Mechanical stress leads to integrin clustering at focal adhesion sites, initiating signaling via FAK and RhoA/ROCK pathways, directly connecting the ECM to intracellular structures. | FAK activation phosphorylates downstream kinases like Src, activating pathways such as PI3K/Akt, leading to enhanced cell survival, migration, and ECM production through cytoskeletal remodeling. | Promotes cell adhesion, migration, proliferation, and ECM remodeling, facilitating tissue repair and regeneration by enhancing cytoskeletal strength and structural integrity. |
| Ion Channels and Calcium Signaling | Mechanically gated ion channels, such as Piezo1, open in response to membrane deformation, allowing calcium influx, which serves as a rapid signal to trigger further cellular pathways. | Calcium influx through ion channels activates calcium-binding proteins like calmodulin, triggering the calcineurin/NFAT and CaMK pathways that regulate gene expression and cytoskeletal organization. | Calcium serves as a key secondary messenger, driving processes like gene expression, cytoskeletal reorganization, and ECM production, ensuring that cells can respond dynamically to mechanical stimuli. |
| Mitogen-Activated Protein Kinase (MAPK) Pathway | Mechanical stress activates MAPK pathways through integrin signaling and mechanosensitive receptors, such as FAK, leading to phosphorylation events that activate ERK1/2, JNK, and p38 MAPK. | ERK1/2 supports proliferation and differentiation, JNK governs stress responses including apoptosis, and p38 MAPK modulates inflammation, ECM remodeling, and cellular survival in response to mechanical signals. | Coordinates cellular responses such as proliferation, apoptosis, and ECM synthesis to help tissues adapt to mechanical stress and maintain tissue integrity, preventing overloading and damage. |
| Wnt/β-Catenin Signaling Pathway | Wnt ligands bind to Frizzled and LRP5/6 receptors, leading to the inhibition of the β-catenin destruction complex, stabilizing β-catenin, allowing it to accumulate in the cytoplasm and translocate to the nucleus. | Stabilized β-catenin interacts with TCF/LEF transcription factors in the nucleus to regulate genes critical for stem cell proliferation, differentiation, and ECM synthesis, crucial for tissue repair. | Directs mesenchymal stem cells toward osteogenic and chondrogenic lineages, ensuring efficient bone and cartilage formation, promoting tissue resilience and functional recovery. |
| YAP/TAZ Signaling Pathway | Mechanical loading inhibits the Hippo pathway, allowing dephosphorylated YAP/TAZ to enter the nucleus, where they regulate transcription in response to mechanical cues such as ECM stiffness. | YAP/TAZ interact with TEAD transcription factors to promote the expression of genes controlling cell cycle progression, apoptosis inhibition, and ECM production, adapting tissues to mechanical loading. | YAP/TAZ modulate the expression of genes involved in cell growth, differentiation, and survival, contributing to tissue regeneration, homeostasis, and adaptation to mechanical changes. |
Molecular and Cellular Biology Aspects of Differences Between Meniscus, Cartilage, Ligament, and Subchondral Bone in Rehabilitation and Injury
Meniscus
Cartilage
Ligament
Subchondral Bone
| Tissue Type | Molecular Composition | Response to Injury | Rehabilitation Strategies |
| Meniscus | Primarily Type I and Type II collagen, with proteoglycans like aggrecan. Fibrochondrocytes are responsible for ECM maintenance and adapting to mechanical stress. The ECM also contains glycosaminoglycans (GAGs) that help retain water for viscoelastic properties. | Injury triggers the release of cytokines (IL-1β, TNF-α) and MMPs, degrading collagen. Healing is limited in avascular zones, and surgical intervention may be necessary. Inflammation can exacerbate ECM breakdown and slow repair. | Controlled weight-bearing exercises to stimulate fibrochondrocytes and ECM synthesis. Proprioceptive training enhances joint stability, while biologic therapies (TGF-β, PDGF) promote matrix production. Surgical repair may be required for avascular regions. |
| Cartilage | Rich in Type II collagen and aggrecan, which bind water to resist compression. Chondrocytes are the sole cell type and produce ECM components. The ECM also contains hyaluronan and link proteins to stabilize the matrix. | Cartilage responds with an inflammatory cascade, releasing cytokines (IL-1, TNF-α) that activate enzymes like ADAMTS-4 and -5. This leads to aggrecan and collagen degradation, further weakening the ECM. Avascularity severely limits repair capacity. | Hydrotherapy and continuous passive motion (CPM) stimulate chondrocytes and prevent stiffness. Biologic therapies like autologous chondrocyte implantation (ACI) and mesenchymal stem cell (MSC) therapy introduce new cells for ECM repair. Scaffolds may be used to support cell growth. |
| Ligament | Dense Type I collagen fibers arranged in a crimped pattern, providing high tensile strength. Fibroblasts synthesize collagen and elastin, maintaining ECM structure. Proteoglycans and elastin contribute to the ligament’s viscoelastic properties. | Fibroblasts and immune cells release cytokines like IL-1β and TNF-α, initiating inflammation. Fibroblasts proliferate and synthesize collagen during the proliferative phase. Remodeling realigns collagen fibers to restore mechanical properties. | Early mobilization (isometric and isotonic exercises) promotes fibroblast activity and collagen realignment. Proprioceptive training helps restore neuromuscular control. Gradual loading and strengthening exercises prevent re-injury and promote ligament healing. |
| Subchondral Bone | Subchondral bone matrix is composed of Type I collagen, produced by osteoblasts. Osteocytes within the bone matrix regulate remodeling, while osteoclasts resorb bone. The ECM is mineralized with hydroxyapatite, giving strength to the bone. | Inflammatory cytokines (IL-1, IL-6, TNF-α) activate osteoclasts, increasing bone resorption. Disruption in osteoblast-osteoclast balance leads to weakened bone, subchondral sclerosis, or cyst formation, contributing to joint degeneration. | Gradual reintroduction of weight-bearing activities like resistance training to promote bone remodeling. Pharmacological interventions (bisphosphonates, anabolic agents) regulate bone turnover, while low-impact exercises like swimming maintain joint function without overloading the bone. |
Implications for Treatment and Rehabilitation
Rehabilitation Protocols
Pharmacological Interventions
Advanced Regenerative Medicine Approaches
Combination Therapies
Advanced Research and Future Directions
Novel Therapeutic Targets
Personalized Rehabilitation Strategies
Integration of Patient-Specific Data
- 1. Biomechanical Assessments
- 2. Imaging
- 3. Biomarker Profiles
- 4. Patient-Reported Outcomes
Predictive Models and Decision-Support Systems
- 1. Machine Learning
- 2. Decision-Support Systems
- 3. Personalized Rehabilitation Protocols
Advanced Technologies for Monitoring and Adjustment
- 1. Wearable Sensors
- 2. Telemedicine and Remote Monitoring
- 3. 3D Motion Analysis
Future Directions in Personalized Rehabilitation
- 1. Integration of Multi-Omics Data
- 2. Advanced Analytics and Machine Learning
- 3. Patient Engagement and Empowerment
Conclusion
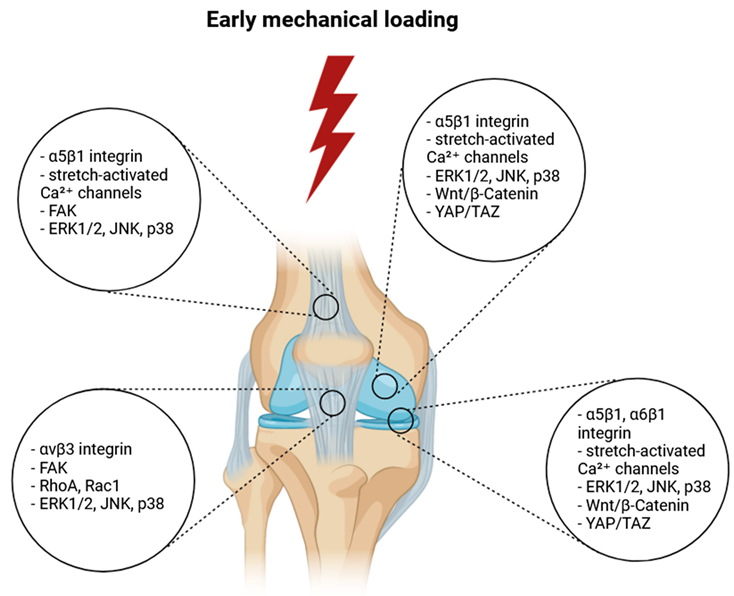
| Aspect | Cartilage | Ligaments | Tendons | Meniscus |
| Biophysical Stimulation | - Compression - Hydrostatic pressure - Shear stress - Dynamic loading to mimic joint movements |
- Tensile loading - Cyclic stretching - Controlled dynamic loading to prevent overstretching |
- Tensile loading - Cyclic stretching - Gradual progressive loading |
- Compression - Shear stress - Tensile loading - Cyclic and static loading for comprehensive stimulation |
| Mechanotransduction | - Integrin signaling (e.g., α5β1 integrin) - Ion channels (e.g., stretch-activated Ca²⁺ channels) - MAPK pathway (ERK1/2, JNK, p38) - Wnt/β-Catenin signaling - YAP/TAZ activation |
- Integrin signaling (e.g., αvβ3 integrin) - Focal Adhesion Kinase (FAK) activation - Rho family GTPases (RhoA, Rac1) - MAPK pathway (ERK1/2, JNK, p38) |
- Integrin signaling (e.g., α5β1 integrin) - Ion channels (e.g., stretch-activated Ca²⁺ channels) - FAK activation - MAPK pathway (ERK1/2, JNK, p38) |
- Integrin signaling (e.g., α5β1 and α6β1 integrins) - Ion channels (e.g., stretch-activated Ca²⁺ channels) - MAPK pathway (ERK1/2, JNK, p38) - Wnt/β-Catenin signaling - YAP/TAZ activation |
| Stress/Strain | - Moderate compressive stress (optimal to stimulate chondrocytes) - Cyclic loading to promote ECM production - Avoid excessive stress to prevent chondrocyte apoptosis |
- Tensile stress aligned with ligament fibers - Gradual increase in load to stimulate fibroblasts - Cyclic loading to enhance collagen synthesis |
- Tensile stress aligned with tendon fibers - Gradual increase in load to stimulate tenocytes - Cyclic loading to enhance collagen synthesis |
- Combination of compressive and tensile stress - Cyclic loading to stimulate chondrocytes and fibrochondrocytes - Avoid excessive stress to prevent further tearing |
| Stress-Relaxation | - Gradual application and release of load - Allows time for ECM adaptation - Prevents cell damage and apoptosis |
- Gradual relaxation phases - Reduces risk of re-injury - Enhances ligament compliance and function |
- Gradual relaxation phases - Reduces risk of tendinopathy - Enhances tendon compliance and function |
- Gradual relaxation phases - Allows time for ECM adaptation - Prevents further damage and promotes healing |
| Hysteresis | - Minimizes energy loss during loading/unloading - Maintains cartilage resilience and function - Promotes efficient load-bearing capacity |
- Reduces energy loss during cyclic loading - Enhances ligament elasticity and function - Promotes efficient load transfer and shock absorption |
- Reduces energy loss during cyclic loading - Enhances tendon elasticity and function - Promotes efficient force transmission and load-bearing capacity |
- Minimizes energy loss during loading/unloading - Maintains meniscal resilience and function - Promotes efficient load distribution and shock absorption |
| Cell Biology of Early Mechanical Loading | - Chondrocyte proliferation and differentiation - ECM synthesis (collagen II, aggrecan) - Autophagy activation for cell survival - Modulation of inflammatory response (decreased IL-1, TNF-α) - Enhanced synthesis of proteoglycans and glycosaminoglycans |
- Fibroblast proliferation and migration - Collagen synthesis (type I and III) - ECM remodeling and organization - Modulation of inflammatory response (decreased IL-6, MMPs) - Enhanced ligament strength and flexibility |
- Tenocyte proliferation and alignment - Collagen synthesis (type I) - ECM remodeling and organization - Modulation of inflammatory response (decreased MMPs, increased TIMPs) - Enhanced tendon strength and flexibility |
- Chondrocyte and fibrochondrocyte activity - ECM synthesis (collagen I and II, proteoglycans) - MSC recruitment and differentiation - Modulation of inflammatory response (decreased pro-inflammatory cytokines) - Enhanced meniscal function and integration |
 |
References
- Achenbach, L.; Bloch, H.; Klein, C.; Damm, T.; Obinger, M.; Rudert, M.; Krutsch, W.; Szymski, D. Four distinct patterns of anterior cruciate ligament injury in women’s professional football (soccer): A systematic video analysis of 37 match injuries. Br J Sports Med. 2024, 58, 709–716. [Google Scholar] [CrossRef] [PubMed]
- Gholipour Aghdam, G.M.; Alizadeh, M.H.; Minoonejad, H.; Shirzad, E.; Wilke, J. Knee Biomechanics During Neurocognitively Challenged Drop Landings in Male Elite Soccer Players with Anterior Cruciate Ligament Reconstruction. Sports Med Open. 2024, 10, 19. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Aizawa, J.; Hirohata, K.; Ohji, S.; Ohmi, T.; Mitomo, S.; Koga, H.; Yagishita, K. Cross-sectional study on relationships between physical function and psychological readiness to return to sport after anterior cruciate ligament reconstruction. BMC Sports Sci Med Rehabil. 2022, 14, 97. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ananías, J.; Vidal, C.; Ortiz-Muñoz, L.; Irarrázaval, S.; Besa, P. Use of electromyographic biofeedback in rehabilitation following anterior cruciate ligament reconstruction: A systematic review and meta-analysis. Physiotherapy. 2024, 123, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Andrushko, J.W.; Carr, J.C.; Farthing, J.P.; Lepley, L.K.; DeFreitas, J.M.; Goodall, S.; Hendy, A.M.; Howatson, G.; Grooms, D.R.; Zult, T.; Hortobagyi, T.; Harput, G.; Papandreou, M.; Nosaka, K.; Carson, R.G.; Manca, A.; Deriu, F.; Behm, D.G.; Kidgell, D.J.; Clark, N.C.; Boyd, L.A. Potential role of cross-education in early-stage rehabilitation after anterior cruciate ligament reconstruction. Br J Sports Med. 2023, 57, 1474–1475. [Google Scholar] [CrossRef] [PubMed]
- Armento, A.; Keeter, C.; Gagliardi, A.; Rossing, H.; Giachino, C.; VandenBerg, C.; Howell, D.; Albright, J. Association of Grit With Postoperative Knee Outcomes and Physical Function After ACL Reconstruction in Adolescent Athletes. Am J Sports Med. 2023, 51, 2900–2907. [Google Scholar] [CrossRef] [PubMed]
- Baez, S.; Harkey, M.; Birchmeier, T.; Triplett, A.; Collins, K.; Kuenze, C. Psychological Readiness, Injury-Related Fear, and Persistent Knee Symptoms After Anterior Cruciate Ligament Reconstruction. J Athl Train. 2023, 58, 998–1003. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bahr, R. Why screening tests to predict injury do not work-and probably never will…: A critical review. Br J Sports Med. 2016, 50, 776–780. [Google Scholar] [CrossRef] [PubMed]
- Bakhsh, H.R.; Metikala, S.; Billy, G.G.; Vairo, G.L. Association Between Self-Reported Kinesiophobia and Single-Leg Hop for Distance in Patients With ACL Reconstruction: A Systematic Review. Sports Health. 2022, 14, 674–680. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Balendra, G.; Jones, M.; Borque, K.A.; Willinger, L.; Pinheiro, V.H.; Williams, A. Factors affecting return to play and graft re-rupture after primary ACL reconstruction in professional footballers. Knee Surg Sports Traumatol Arthrosc. 2022, 30, 2200–2208. [Google Scholar] [CrossRef] [PubMed]
- Banios, K.; Raoulis, V.; Fyllos, A.; Chytas, D.; Mitrousias, V.; Zibis, A. Anterior and Posterior Cruciate Ligaments Mechanoreceptors: A Review of Basic Science. Diagnostics 2022, 12, 331. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Belcher, S.; Whatman, C.; Brughelli, M. A systematic video analysis of 21 anterior cruciate ligament injuries in elite netball players during games. Sports Biomech. 2022, 7, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Bencke, J.; Aagaard, P.; Zebis, M.K. Muscle Activation During ACL Injury Risk Movements in Young Female Athletes: A Narrative Review. Front Physiol. 2018, 9, 445. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Benjaminse, A.; Otten, B.; Gokeler, A.; Diercks, R.L.; Lemmink, K.A.P.M. Motor learning strategies in basketball players and its implications for ACL injury prevention: A randomized controlled trial. Knee Surg Sports Traumatol Arthrosc. 2017, 25, 2365–2376. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bertozzi, F.; Fischer, P.D.; Hutchison, K.A.; Zago, M.; Sforza, C.; Monfort, S.M. Associations Between Cognitive Function and ACL Injury-Related Biomechanics: A Systematic Review. Sports Health. 2023, 15, 855–866. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Betsch, M.; Darwich, A.; Chang, J.; Whelan, D.; Ogilvie-Harris, D.; Chahal, J.; Theodoropoulos, J. Wide Variability in Return-to-Sport Criteria used by Team Physicians After Anterior Cruciate Ligament Reconstruction in Elite Athletes-A Qualitative Study. Arthrosc Sports Med Rehabil. 2022, 4, e1759–e1766. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Beynnon, B.D.; Tourville, T.W.; Hollenbach, H.C.; Shultz, S.; Vacek, P. Intrinsic Risk Factors for First-Time Noncontact ACL Injury: A Prospective Study of College and High School Athletes. Sports Health. 2023, 15, 433–442. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Boden, B.P.; Sheehan, F.T. Mechanism of non-contact ACL injury: OREF Clinical Research Award 2021. J Orthop Res. 2022, 40, 531–540. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bodkin, S.G. Time to Reflect on Return to Sport Timing Following ACL Reconstruction. Sports Med. 2024. [Google Scholar] [CrossRef] [PubMed]
- Bolt, R.; Heuvelmans, P.; Benjaminse, A.; Robinson, M.A.; Gokeler, A. An ecological dynamics approach to ACL injury risk research: A current opinion. Sports Biomech. 2021, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Brophy, R.H.; Wojtys, E.M.; Mack, C.D.; Hawaldar, K.; Herzog, M.M.; Owens, B.D. Factors Associated With the Mechanism of ACL Tears in the National Football League: A Video-Based Analysis. Orthop J Sports Med. 2021, 9, 23259671211053301. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Buckthorpe, M.; Gokeler, A.; Herrington, L.; Hughes, M.; Grassi, A.; Wadey, R.; Patterson, S.; Compagnin, A.; La Rosa, G.; Della Villa, F. Optimising the Early-Stage Rehabilitation Process Post-ACL Reconstruction. Sports Med. 2024, 54, 49–72. [Google Scholar] [CrossRef] [PubMed]
- Byrne, S.; Lay, B.; Staynor, J.; Alderson, J.; Donnelly, C.J. The effect of planning time on penultimate and ultimate step kinematics and subsequent knee moments during sidestepping. Scand J Med Sci Sports. 2022, 32, 1366–1376. [Google Scholar] [CrossRef] [PubMed]
- Cacolice, P.A.; Starkey, B.E.; Carcia, C.R.; Higgins, P.E. Research Dominance Definitions May Not Identify Higher Risk Limb for Anterior Cruciate Ligament Injury in NCAA D3 Student-Athletes. Int J Sports Phys Ther. 2022, 17, 622–627. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Carlson, V.R.; Sheehan, F.T.; Boden, B.P. Video Analysis of Anterior Cruciate Ligament (ACL) Injuries: A Systematic Review. JBJS Rev. 2016, 4, e5. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chaaban, C.R.; Hearn, D.; Goerger, B.; Padua, D.A. Are Elite Collegiate Female Athletes PRIME for a Safe Return to Sport after ACLR? An Investigation of Physical Readiness and Integrated Movement Efficiency (PRIME). Int J Sports Phys Ther. 2022, 17, 445–455. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chaaban, C.R.; Turner, J.A.; Padua, D.A. Think outside the box: Incorporating secondary cognitive tasks into return to sport testing after ACL reconstruction. Front Sports Act Living. 2023, 4, 1089882. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chaput, M.; Onate, J.A.; Simon, J.E.; Criss, C.R.; Jamison, S.; McNally, M.; Grooms, D.R. Visual cognition associated with knee proprioception, time to stability, and sensory integration neural activity after ACL reconstruction. J Orthop Res. 2022, 40, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Larson, D.; Vu, V.; Ness, B.M.; Wellsandt, E.; Morrison, S. A Multi-Systems Approach to Human Movement after ACL Reconstruction: The Musculoskeletal System. Int J Sports Phys Ther. 2021, 17, 27–46. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, Z.; Li, Y.; Zhang, Y.; Zhang, Z.; Wang, J.; Deng, X.; Liu, C.; Chen, N.; Jiang, C.; Li, W.; Song, B. Analysis of Visual Risk Factors of Anterior Cruciate Ligament Injury of Knee Joint. J Clin Med. 2022, 11, 5602. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ciceklidag, M.; Kaya, I.; Ayanoglu, T.; Ayas, I.H.; Ozer, M.; Ataoglu, M.B.; Kanatli, U. Proprioception After Primary Repair of the Anterior Cruciate Ligament. Am J Sports Med. 2024, 52, 1199–1208, Erratum in Am J Sports Med. 2024, 3635465241253575. https://doi.org/10.1177/03635465241253575. [Google Scholar] [CrossRef] [PubMed]
- Colapietro, M.; Portnoff, B.; Miller, S.J.; Sebastianelli, W.; Vairo, G.L. Effects of Blood Flow Restriction Training on Clinical Outcomes for Patients With ACL Reconstruction: A Systematic Review. Sports Health. 2023, 15, 260–273. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Collings, T.J.; Diamond, L.E.; Barrett, R.S.; Timmins, R.G.; Hickey, J.T.; DUMoulin, W.S.; Williams, M.D.; Beerworth, K.A.; Bourne, M.N. Strength and Biomechanical Risk Factors for Noncontact ACL Injury in Elite Female Footballers: A Prospective Study. Med Sci Sports Exerc. 2022, 54, 1242–1251. [Google Scholar] [CrossRef] [PubMed]
- Crotti, M.; Heering, T.; Lander, N.; Fox, A.; Barnett, L.M.; Duncan, M.J. Extrinsic Risk Factors for Primary Noncontact Anterior Cruciate Ligament Injury in Adolescents Aged between 14 and 18 years: A Systematic Review. Sports Med. 2024, 54, 875–894. [Google Scholar] [CrossRef] [PubMed]
- Crotty, N.M.N.; Daniels, K.A.J.; McFadden, C.; Cafferkey, N.; King, E. Relationship Between Isokinetic Knee Strength and Single-Leg Drop Jump Performance 9 Months After ACL Reconstruction. Orthop J Sports Med. 2022, 10, 23259671211063800. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Culvenor, A.G.; Girdwood, M.A.; Juhl, C.B.; Patterson, B.E.; Haberfield, M.J.; Holm, P.M.; Bricca, A.; Whittaker, J.L.; Roos, E.M.; Crossley, K.M. Rehabilitation after anterior cruciate ligament and meniscal injuries: A best-evidence synthesis of systematic reviews for the OPTIKNEE consensus. Br J Sports Med. 2022, 56, 1445–1453. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cunha, J.; Solomon, D.J. ACL Prehabilitation Improves Postoperative Strength and Motion and Return to Sport in Athletes. Arthrosc Sports Med Rehabil. 2022, 4, e65–e69. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cuyul-Vásquez, I.; Álvarez, E.; Riquelme, A.; Zimmermann, R.; Araya-Quintanilla, F. Effectiveness of Unilateral Training of the Uninjured Limb on Muscle Strength and Knee Function of Patients With Anterior Cruciate Ligament Reconstruction: A Systematic Review and Meta-Analysis of Cross-Education. J Sport Rehabil. 2022, 31, 605–616. [Google Scholar] [CrossRef] [PubMed]
- D’Hooghe, P.; Grassi, A.; Villa, F.D.; Alkhelaifi, K.; Papakostas, E.; Rekik, R.; Marin, T.; Tosarelli, F.; Zaffagnini, S. The injury mechanism correlation between MRI and video-analysis in professional football players with an acute ACL knee injury reveals consistent bone bruise patterns. Knee Surg Sports Traumatol Arthrosc. 2023, 31, 121–132. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Daggett, M.C.; Witte, K.A.; Cabarkapa, D.; Cabarkapa, D.V.; Fry, A.C. Evidence-Based Data Models for Return-to-Play Criteria after Anterior Cruciate Ligament Reconstruction. Healthcare 2022, 10, 929. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Dalvandpour, N.; Zareei, M.; Abbasi, H.; Abdoli, B.; Mohammadian, M.A.; Rommers, N.; Rössler, R. Focus of Attention During ACL Injury Prevention Exercises Affects Improvements in Jump-Landing Kinematics in Soccer Players: A Randomized Controlled Trial. J Strength Cond Res. 2023, 37, 337–342. [Google Scholar] [CrossRef] [PubMed]
- D’Argenio, E.M.; Eckard, T.G.; Frank, B.S.; Prentice, W.E.; Padua, D.A. Examining the Dynamic Nature of Anterior Cruciate Ligament Injury Risk Factors in Women’s Collegiate Soccer. J Sport Rehabil. 2022, 31, 286–293. [Google Scholar] [CrossRef] [PubMed]
- Das, L.; Johri, A.S.; Abdusamad, V.; Schuh, A.; Goyal, T. Joint awareness and return to pre-injury level of activities after ACL reconstruction in athletes vs non-athletes. Eur J Orthop Surg Traumatol. 2023, 33, 819–827. [Google Scholar] [CrossRef] [PubMed]
- Dauty, M.; Crenn, V.; Louguet, B.; Grondin, J.; Menu, P.; Fouasson-Chailloux, A. Anatomical and Neuromuscular Factors Associated to Non-Contact Anterior Cruciate Ligament Injury. J Clin Med. 2022, 11, 1402. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Davies, W.T.; Ryu, J.H.; Graham-Smith, P.; Goodwin, J.E.; Cleather, D.J. Stronger Subjects Select a Movement Pattern That May Reduce Anterior Cruciate Ligament Loading During Cutting. J Strength Cond Res. 2022, 36, 1853–1859. [Google Scholar] [CrossRef] [PubMed]
- Della Villa, F.; Buckthorpe, M.; Grassi, A.; Nabiuzzi, A.; Tosarelli, F.; Zaffagnini, S.; Della Villa, S. Systematic video analysis of ACL injuries in professional male football (soccer): Injury mechanisms, situational patterns and biomechanics study on 134 consecutive cases. Br J Sports Med. 2020, 54, 1423–1432. [Google Scholar] [CrossRef] [PubMed]
- Detherage, J.P.; Divine, J.G.; Donaworth, M.A.; Palmer, T.G.; Hagen, J.A.; Hasselfeld, K.A.; Eifert-Mangine, M.; Mangine, R.E.; Clark, J.F.; Grawe, B.M. Physiological Monitoring Detected Changes During Women’s Soccer Anterior Cruciate Ligament Injury. Cureus. 2021, 13, e14838. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Devana, S.K.; Solorzano, C.A.; Vail, J.; Jackson, N.; Pham, D.; Jones, K.J. Outcomes of Blood Flow Restriction Training After ACL Reconstruction in NCAA Division I Athletes. Orthop J Sports Med. 2024, 12, 23259671241248589. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Alejandra Díaz, M.; Smeets, A.; Hagen, M.; Sankey, S.P.; Verschueren, S.; Vanrenterghem, J. Postural balance strategies during landing at the moment of return-to-sports after anterior cruciate ligament reconstruction. J Biomech. 2022, 145, 111381. [Google Scholar] [CrossRef] [PubMed]
- Di Paolo, S.; Bragonzoni, L.; Della Villa, F.; Grassi, A.; Zaffagnini, S. Do healthy athletes exhibit at-risk biomechanics for anterior cruciate ligament injury during pivoting movements? Sports Biomech. 2022, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Dischiavi, S.L.; Wright, A.A.; Heller, R.A.; Love, C.E.; Salzman, A.J.; Harris, C.A.; Bleakley, C.M. Do ACL Injury Risk Reduction Exercises Reflect Common Injury Mechanisms? A Scoping Review of Injury Prevention Programs. Sports Health. 2022, 14, 592–600. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Drole, K.; Paravlic, A.H. Interventions for increasing return to sport rates after an anterior cruciate ligament reconstruction surgery: A systematic review. Front Psychol. 2022, 13, 939209. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Dos’Santos, T.; McBurnie, A.; Donelon, T.; Thomas, C.; Comfort, P.; Jones, P.A. A qualitative screening tool to identify athletes with ‘high-risk’ movement mechanics during cutting: The cutting movement assessment score (CMAS). Phys Ther Sport. 2019, 38, 152–161. [Google Scholar] [CrossRef] [PubMed]
- Duthon, V.B.; Barea, C.; Abrassart, S.; Fasel, J.H.; Fritschy, D.; Ménétrey, J. Anatomy of the anterior cruciate ligament. Knee Surg Sports Traumatol Arthrosc. 2006, 14, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Emami, F.; Negahban, H.; Sinaei, E.; Mostafaee, N.; Shahtahmassebi, B.; Ebrahimzadeh, M.H.; Mehravar, M. The Effects of Various Cognitive Tasks Including Working Memory, Visuospatial, and Executive Function on Postural Control in Patients With Anterior Cruciate Ligament Injury. Motor Control. 2024, 28, 193–209. [Google Scholar] [CrossRef] [PubMed]
- Fältström, A.; Hägglund, M.; Hedevik, H.; Kvist, J. Poor Validity of Functional Performance Tests to Predict Knee Injury in Female Soccer Players With or Without Anterior Cruciate Ligament Reconstruction. Am J Sports Med. 2021, 49, 1441–1450. [Google Scholar] [CrossRef] [PubMed]
- Fausett, W.A.; Reid, D.A.; Larmer, P.J. Current perspectives of New Zealand physiotherapists on rehabilitation and return to sport following anterior cruciate ligament reconstruction: A survey. Phys Ther Sport. 2022, 53, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Figueroa, D.; Arce, G.; Espregueira-Mendes, J.; Maestu, R.; Mosquera, M.; Williams, A.; Parker, D.; Cohen, M.; Karahan, M.; Ochoa Perea, G.A.; Zaffagnini, S.; Neyret, P.; Karlsson, J.; Musahl, V.; Radice, F.; van der Merwe, W.M.; Landreau, P.; Imhoff, A.; Menetrey, J.; Ayeni, O.R.; Arliani, G.G.; Sherman, S.L.; Monllau, J.C.; D’Hooghe, P.; Pinczewski, L.; Feller, J.; Patnaik, S. Return to sport soccer after anterior cruciate ligament reconstruction: ISAKOS consensus. J ISAKOS. 2022, 7, 150–161. [Google Scholar] [CrossRef] [PubMed]
- Fleming, B.C. Fifty Years of ACL Biomechanics: What’s Next? Am J Sports Med. 2022, 50, 3745–3748. [Google Scholar] [CrossRef] [PubMed]
- García-Rodríguez, P.; Pecci, J.; Vázquez-González, S.; Pareja-Galeano, H. Acute and Chronic Effects of Blood Flow Restriction Training in Physically Active Patients With Anterior Cruciate Ligament Reconstruction: A Systematic Review. Sports Health. 2023, 19417381231208636. [Google Scholar] [CrossRef] [PubMed]
- Gholami, F.; Letafatkar, A.; Moghadas Tabrizi, Y.; Gokeler, A.; Rossettini, G.; Ghanati, H.A.; Schöllhorn, W.I. Comparing the Effects of Differential and Visuo-Motor Training on Functional Performance, Biomechanical, and Psychological Factors in Athletes after ACL Reconstruction: A Randomized Controlled Trial. J Clin Med. 2023, 12, 2845. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Giesche, F.; Vieluf, S.; Wilke, J.; Engeroff, T.; Niederer, D.; Banzer, W. Cortical Motor Planning and Biomechanical Stability During Unplanned Jump Landings in Men With Anterior Cruciate Ligament Reconstruction. J Athl Train. 2022, 57, 547–556. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gokeler, A.; Seil, R.; Kerkhoffs, G.; Verhagen, E. A novel approach to enhance ACL injury prevention programs. J Exp Orthop. 2018, 5, 22. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gokeler, A.; Neuhaus, D.; Benjaminse, A.; Grooms, D.R.; Baumeister, J. Principles of Motor Learning to Support Neuroplasticity After ACL Injury: Implications for Optimizing Performance and Reducing Risk of Second ACL Injury. Sports Med. 2019, 49, 853–865, Erratum in Sports Med. 2019, 49, 979. https://doi.org/10.1007/s40279-019-01078-w. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gokeler, A.; Dingenen, B.; Hewett, T.E. Rehabilitation and Return to Sport Testing After Anterior Cruciate Ligament Reconstruction: Where Are We in 2022? Arthrosc Sports Med Rehabil. 2022, 4, e77–e82. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gokeler, A.; Grassi, A.; Hoogeslag, R.; van Houten, A.; Lehman, T.; Bolling, C.; Buckthorpe, M.; Norte, G.; Benjaminse, A.; Heuvelmans, P.; Di Paolo, S.; Tak, I.; Villa, F.D. Return to sports after ACL injury 5 years from now: 10 things we must do. J Exp Orthop. 2022, 9, 73, Erratum in J Exp Orthop. 2022, 9, 111. https://doi.org/10.1186/s40634-022-00548-x. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gokeler, A.; Tosarelli, F.; Buckthorpe, M.; Della Villa, F. Neurocognitive Errors and Noncontact Anterior Cruciate Ligament Injuries in Professional Male Soccer Players. J Athl Train. 2024, 59, 262–269. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Golberg, E.; Sommerfeldt, M.; Pinkoski, A.; Dennett, L.; Beaupre, L. Anterior Cruciate Ligament Reconstruction Return-to-Sport Decision-Making: A Scoping Review. Sports Health. 2024, 16, 115–123. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gopinatth, V.; Smith, M.V.; Matava, M.J.; Brophy, R.H.; Knapik, D.M. Most Anterior Cruciate Ligament Injuries in Professional Athletes Occur Without Contact to the Injured Knee: A Systematic Review of Video Analysis Studies. Arthroscopy 2024. [Google Scholar] [CrossRef] [PubMed]
- Grooms, D.R.; Page, S.J.; Nichols-Larsen, D.S.; Chaudhari, A.M.; White, S.E.; Onate, J.A. Neuroplasticity Associated With Anterior Cruciate Ligament Reconstruction. J Orthop Sports Phys Ther. 2017, 47, 180–189. [Google Scholar] [CrossRef] [PubMed]
- Grooms, D.; Appelbaum, G.; Onate, J. Neuroplasticity following anterior cruciate ligament injury: A framework for visual-motor training approaches in rehabilitation. J Orthop Sports Phys Ther. 2015, 45, 381–393. [Google Scholar] [CrossRef] [PubMed]
- Grooms, D.R.; Chaput, M.; Simon, J.E.; Criss, C.R.; Myer, G.D.; Diekfuss, J.A. Combining Neurocognitive and Functional Tests to Improve Return-to-Sport Decisions Following ACL Reconstruction. J Orthop Sports Phys Ther. 2023, 53, 415–419. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gureck, A.E.; Crockett, Z.; Barsky, B.W.; Samuels, S.; Frank, J.S.; Storer, S.K.; Fazekas, M.L. Do Differences Exist in Impact Test Domains between Youth Athletes with and without an Anterior Cruciate Ligament Injury? Healthcare 2023, 11, 2764. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hamdan, M.; Haddad, B.; Alshrouf, M.A.; Azzam, M.I.; Isleem, U.; Hamasha, R.; Albtoush, O.M.; Alhusban, M.T.; Mubarak, N.; Alryalat, S.A. Can MRI knee joint measurements predict the population at risk of ACL injury? BMC Sports Sci Med Rehabil. 2022, 14, 98. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hamoongard, M.; Hadadnezhad, M.; Abbasi, A. Effect of combining eight weeks of neuromuscular training with dual cognitive tasks on landing mechanics in futsal players with knee ligament dominance defect: A randomized controlled trial. BMC Sports Sci Med Rehabil. 2022, 14, 196. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Harput, G.; Demirci, S.; Soylu, A.R.; Bayrakci Tunay, V. Association between quadriceps muscle thickness and knee function in anterior cruciate ligament reconstructed athletes: A cross-sectional study. Physiother Theory Pract. 2023, 39, 2171–2179. [Google Scholar] [CrossRef] [PubMed]
- Henderson, F.J.; Konishi, Y.; Shima, N.; Shimokochi, Y. Effects of 8-Week Exhausting Deep Knee Flexion Flywheel Training on Persistent Quadriceps Weakness in Well-Trained Athletes Following Anterior Cruciate Ligament Reconstruction. Int J Environ Res Public Health. 2022, 19, 13209. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hewett, T.E.; Myer, G.D.; Ford, K.R.; Paterno, M.V.; Quatman, C.E. Mechanisms, prediction, and prevention of ACL injuries: Cut risk with three sharpened and validated tools. J Orthop Res. 2016, 34, 1843–1855. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Homan, M.D.; Braaten, J.A.; Banovetz, M.T.; Monson, J.K.; Kennedy, N.I.; LaPrade, R.F. Principles for optimizing anterior cruciate ligament reconstruction outcomes in elite athletes: A review of current techniques. Ann Jt. 2024, 9, 19. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hoogeslag, R.A.G.; Huis In ‘t Veld, R.; Brouwer, R.W.; de Graaff, F.; Verdonschot, N. Acute Anterior Cruciate Ligament Rupture: Repair or Reconstruction? Five-Year Results of a Randomized Controlled Clinical Trial. Am J Sports Med. 2022, 50, 1779–1787. [Google Scholar] [CrossRef] [PubMed]
- Hurley, E.T.; Mojica, E.S.; Haskel, J.D.; Mannino, B.J.; Alaia, M.; Strauss, E.J.; Jazrawi, L.M.; Gonzlaez-Lomas, G. Return to play testing following anterior cruciate reconstruction—A systematic review & meta-analysis. Knee. 2022, 34, 134–140. [Google Scholar] [CrossRef] [PubMed]
- Imai, S.; Harato, K.; Morishige, Y.; Kobayashi, S.; Niki, Y.; Sato, K.; Nagura, T. Effects of Dual Task Interference on Biomechanics of The Entire Lower Extremity During the Drop Vertical Jump. J Hum Kinet. 2022, 81, 5–14. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jack, R.A., 2nd; Lambert, B.S.; Hedt, C.A.; Delgado, D.; Goble, H.; McCulloch, P.C. Blood Flow Restriction Therapy Preserves Lower Extremity Bone and Muscle Mass After ACL Reconstruction. Sports Health. 2023, 15, 361–371. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jenkins, S.M.; Guzman, A.; Gardner, B.B.; Bryant, S.A.; Del Sol, S.R.; McGahan, P.; Chen, J. Rehabilitation After Anterior Cruciate Ligament Injury: Review of Current Literature and Recommendations. Curr Rev Musculoskelet Med. 2022, 15, 170–179. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jiang, L.; Zhang, L.; Huang, W.; Zeng, Q.; Huang, G. The effect of proprioception training on knee kinematics after anterior cruciate ligament reconstruction: A randomized control trial. J Back Musculoskelet Rehabil. 2022, 35, 1085–1095. [Google Scholar] [CrossRef] [PubMed]
- Johnston, J.T.; Mandelbaum, B.R.; Schub, D.; Rodeo, S.A.; Matava, M.J.; Silvers-Granelli, H.J.; Cole, B.J.; ElAttrache, N.S.; McAdams, T.R.; Brophy, R.H. Video Analysis of Anterior Cruciate Ligament Tears in Professional American Football Athletes. Am J Sports Med. 2018, 46, 862–868. [Google Scholar] [CrossRef] [PubMed]
- Jones, H.S.R.; Moore, I.S.; King, E.; Stiles, V.H.; Laudani, L.; McCarthy-Ryan, M.; McFadden, C.; Daniels, K.A.J. Movement strategy correspondence across jumping and cutting tasks after anterior cruciate ligament reconstruction. Scand J Med Sci Sports. 2022, 32, 612–621. [Google Scholar] [CrossRef] [PubMed]
- Josse, C.M. Gym-Based Training Interventions for Anterior Cruciate Ligament Injury Reduction in American Football Players. HSS J. 2023, 19, 285–291. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kadlec, D.; Miller-Dicks, M.; Nimphius, S. Training for “Worst-Case” Scenarios in Sidestepping: Unifying Strength and Conditioning and Perception-Action Approaches. Sports Med Open. 2023, 9, 22. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kakavas, G.; Malliaropoulos, N.; Pruna, R.; Traster, D.; Bikos, G.; Maffulli, N. Neuroplasticity and Anterior Cruciate Ligament Injury. Indian J Orthop. 2020, 54, 275–280. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kakavas, G.; Forelli, F.; Malliaropoulos, N.; Hewett, T.E.; Tsaklis, P. Periodization in Anterior Cruciate Ligament Rehabilitation: New Framework Versus Old Model? A Clinical Commentary. Int J Sports Phys Ther. 2023, 18, 541–546. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kellis, E.; Sahinis, C.; Baltzopoulos, V. Is hamstrings-to-quadriceps torque ratio useful for predicting anterior cruciate ligament and hamstring injuries? A systematic and critical review. J Sport Health Sci. 2023, 12, 343–358. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- King, E.; Richter, C.; Daniels, K.A.J.; Franklyn-Miller, A.; Falvey, E.; Myer, G.D.; Jackson, M.; Moran, R.; Strike, S. Can Biomechanical Testing After Anterior Cruciate Ligament Reconstruction Identify Athletes at Risk for Subsequent ACL Injury to the Contralateral Uninjured Limb? Am J Sports Med. 2021, 49, 609–619. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Koc, B.B.; Truyens, A.; Heymans, M.J.L.F.; Jansen, E.J.P.; Schotanus, M.G.M. Effect of Low-Load Blood Flow Restriction Training After Anterior Cruciate Ligament Reconstruction: A Systematic Review. Int J Sports Phys Ther. 2022, 17, 334–346. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kotsifaki, R.; Korakakis, V.; King, E.; Barbosa, O.; Maree, D.; Pantouveris, M.; Bjerregaard, A.; Luomajoki, J.; Wilhelmsen, J.; Whiteley, R. Aspetar clinical practice guideline on rehabilitation after anterior cruciate ligament reconstruction. Br J Sports Med. 2023, 57, 500–514. [Google Scholar] [CrossRef] [PubMed]
- Kiani Haft Lang, M.; Mofateh, R.; Orakifar, N.; Goharpey, S. Differences in Neurocognitive Functions Between Healthy Controls and Anterior Cruciate Ligament-Reconstructed Male Athletes Who Passed or Failed Return to Sport Criteria: A Preliminary Study. J Sport Rehabil. 2023, 32, 645–654. [Google Scholar] [CrossRef] [PubMed]
- Larson, D.; Vu, V.; Ness, B.M.; Wellsandt, E.; Morrison, S. A Multi-Systems Approach to Human Movement after ACL Reconstruction: The Musculoskeletal System. Int J Sports Phys Ther. 2021, 17, 27–46. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Liew, B.X.W.; Feller, J.A.; Webster, K.E. Understanding the psychological mechanisms of return to sports readiness after anterior cruciate ligament reconstruction. PLoS ONE. 2022, 17, e0266029. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Louw, Q.; Gillion, N.; van Niekerk, S.M.; Morris, L.; Baumeister, J. The effect of vision on knee biomechanics during functional activities—A systematic review. J Sci Med Sport. 2015, 18, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Lucarno, S.; Zago, M.; Buckthorpe, M.; Grassi, A.; Tosarelli, F.; Smith, R.; Della Villa, F. Systematic Video Analysis of Anterior Cruciate Ligament Injuries in Professional Female Soccer Players. Am J Sports Med. 2021, 49, 1794–1802. [Google Scholar] [CrossRef] [PubMed]
- Maestroni, L.; Turner, A.; Papadopoulos, K.; Sideris, V.; Read, P. Total Score of Athleticism: Profiling Strength and Power Characteristics in Professional Soccer Players After Anterior Cruciate Ligament Reconstruction to Assess Readiness to Return to Sport. Am J Sports Med. 2023, 51, 3121–3130. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mangine, R.; Tersak, J.; Palmer, T.; Hill-Lindsay, A.; Patton, B.; Eifert-Mangine, M.; Jacobs, B.; Colosimo, A.J. The Longitudinal Neurophysiological Adaptation of a Division I Female Lacrosse Player Following Anterior Cruciate Rupture and Repair: A Case Report. Int J Sports Phys Ther. 2023, 18, 467–476. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Maniar, N.; Cole, M.H.; Bryant, A.L.; Opar, D.A. Muscle Force Contributions to Anterior Cruciate Ligament Loading. Sports Med. 2022, 52, 1737–1750. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Meierbachtol, A.; Obermeier, M.; Yungtum, W.; Bottoms, J.; Paur, E.; Nelson, B.J.; Tompkins, M.; Russell, H.C.; Chmielewski, T.L. Injury-Related Fears During the Return-to-Sport Phase of ACL Reconstruction Rehabilitation. Orthop J Sports Med. 2020, 8, 2325967120909385. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mok, A.C.; Fancher, A.J.; Vopat, M.L.; Baker, J.; Tarakemeh, A.; Mullen, S.; Schroeppel, J.P.; Templeton, K.; Mulcahey, M.K.; Vopat, B.G. Sex-Specific Outcomes After Anterior Cruciate Ligament Reconstruction: A Systematic Review and Meta-analysis. Orthop J Sports Med. 2022, 10, 23259671221076883. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Momaya, A.M.; Wood, A.S.; Benson, E.M.; Kwapisz, A.L. The Influence of Psychosocial Factors on Patients Undergoing Anterior Cruciate Ligament Reconstruction. Sports Health. 2024, 16, 230–238. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mørtvedt, A.I.; Krosshaug, T.; Bahr, R.; Petushek, E. I spy with my little eye … a knee about to go ‘pop’? Can coaches and sports medicine professionals predict who is at greater risk of ACL rupture? Br J Sports Med. 2020, 54, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Nae, J.Ä.; Cronström, A. Association between sensorimotor function and visual assessment of postural orientation in patients with ACL injury. Phys Ther Sport. 2022, 55, 160–167. [Google Scholar] [CrossRef] [PubMed]
- Nessler, T.; Denney, L.; Sampley, J. ACL Injury Prevention: What Does Research Tell Us? Curr Rev Musculoskelet Med. 2017, 10, 281–288. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ngatuvai, M.S.; Yang, J.; Kistamgari, S.; Collins, C.L.; Smith, G.A. Epidemiological Comparison of ACL Injuries on Different Playing Surfaces in High School Football and Soccer. Orthop J Sports Med. 2022, 10, 23259671221092321. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Nilstad, A.; Petushek, E.; Mok, K.M.; Bahr, R.; Krosshaug, T. Kiss goodbye to the ‘kissing knees’: No association between frontal plane inward knee motion and risk of future non-contact ACL injury in elite female athletes. Sports Biomech. 2023, 22, 65–79. [Google Scholar] [CrossRef] [PubMed]
- Oladeji, L.; Reynolds, G.; Gonzales, H.; DeFroda, S. Anterior Cruciate Ligament Return to Play: Where Are We Now? J Knee Surg. 2024, 37, 586–592. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi Orangi, B.; Dehghani, M.; Jones, P.A. Manipulation of task constraints is the most effective motor learning method for reducing risk factors for ACL injury during side-step cutting in both male and female athletes. Res Sports Med. 2024, 32, 631–647. [Google Scholar] [CrossRef] [PubMed]
- Parsons, J.L.; Coen, S.E.; Bekker, S. Anterior cruciate ligament injury: Towards a gendered environmental approach. Br J Sports Med. 2021, 55, 984–990. [Google Scholar] [CrossRef] [PubMed]
- Piskin, D.; Benjaminse, A.; Dimitrakis, P.; Gokeler, A. Neurocognitive and Neurophysiological Functions Related to ACL Injury: A Framework for Neurocognitive Approaches in Rehabilitation and Return-to-Sports Tests. Sports Health. 2022, 14, 549–555. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rambaud, A.J.; Neri, T.; Dingenen, B.; Parker, D.; Servien, E.; Gokeler, A.; Edouard, P. The modifying factors that help improve anterior cruciate ligament reconstruction rehabilitation: A narrative review. Ann Phys Rehabil Med. 2022, 65, 101601. [Google Scholar] [CrossRef] [PubMed]
- Read, P.J.; Pedley, J.S.; Eirug, I.; Sideris, V.; Oliver, J.L. Impaired Stretch-Shortening Cycle Function Persists Despite Improvements in Reactive Strength After Anterior Cruciate Ligament Reconstruction. J Strength Cond Res. 2022, 36, 1238–1244. [Google Scholar] [CrossRef] [PubMed]
- Read, P.J.; Davies, W.T.; Bishop, C.; McAuliffe, S.; Wilson, M.G.; Turner, A.N. Residual Deficits in Reactive Strength After Anterior Cruciate Ligament Reconstruction in Soccer Players. J Athl Train. 2023, 58, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Reiche, E.; Collins, K.; Genoese, F.; Walaszek, M.; Triplett, A.; Kuenze, C.; Harkey, M.; Baez, S. Lower Extremity Reaction Time in Individuals With Contact Versus Noncontact Anterior Cruciate Ligament Injuries After Reconstruction. J Athl Train. 2024, 59, 66–72. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Reyes, M.A.; Probasco, M.O.; Worby, T.N.; Loertscher, D.E.; Soderbeck, L.K.; Huddleston, W.E. Lower Kinetic Chain, Meet the Thinking Brain: A Scoping Review of Cognitive Function and Lower Extremity Injury Risk. Int J Sports Phys Ther. 2022, 17, 787–815. [Google Scholar] [PubMed] [PubMed Central]
- Robey, N.J.; Buchholz, K.O.; Murphy, S.P.; Smith, J.D.; Heise, G.D. Do Athletes With a Reconstructed Anterior Cruciate Ligament Respond Differently Than Controls to Visual Challenges When Dynamic Postural Stability is Assessed? J Appl Biomech. 2021, 37, 611–618. [Google Scholar] [CrossRef] [PubMed]
- Rolley, T.; Gill, S.D.; Keast, M.; Reade, T.; Page, R.; Bonacci, J.; Stella, J.; Johnson, B.; Fox, A. Anticipatory effects on side-step cutting biomechanics in Women’s Australian Football League players. BMJ Open Sport Exerc Med. 2023, 9, e001587. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Romanchuk, N.J.; Livock, H.; Lukas, K.J.; Del Bel, M.J.; Benoit, D.L.; Carsen, S. Criteria Used to Determine Unrestricted Return to Activity After ACL Reconstruction in Pediatric and Adolescent Patients: A Systematic Review. Orthop J Sports Med. 2023, 11, 23259671231154540. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Saadat, S.; Stephenson, M.L.; Gillette, J.C. Entry angle during jump landing changes biomechanical risk factors for ACL injury. Sports Biomech. 2022, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Saadat, S.; Bricarell, K.M.; Gillette, J.C. Dual tasking increases kinematic and kinetic risk factors of ACL injury. Sports Biomech. 2023, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Schilaty, N.D.; McPherson, A.L.; Nagai, T.; Bates, N.A. Differences in psychological readiness for return to sport after anterior cruciate ligament injury is evident in thigh musculature motor unit characteristics. BMJ Open Sport Exerc Med. 2023, 9, e001609. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Schweizer, N.; Strutzenberger, G.; Franchi, M.V.; Farshad, M.; Scherr, J.; Spörri, J. Screening Tests for Assessing Athletes at Risk of ACL Injury or Reinjury-A Scoping Review. Int J Environ Res Public Health. 2022, 19, 2864. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Schwery, N.A.; Kiely, M.T.; Larson, C.M.; Wulf, C.A.; Heikes, C.S.; Hess, R.W.; Giveans, M.R.; Solie, B.S.; Doney, C.P. Quadriceps Strength following Anterior Cruciate Ligament Reconstruction: Normative Values based on Sex, Graft Type and Meniscal Status at 3, 6 & 9 Months. Int J Sports Phys Ther. 2022, 17, 434–444. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sheean, A.J.; DeFoor, M.T.; Spindler, K.P.; Arner, J.W.; Athiviraham, A.; Bedi, A.; DeFroda, S.; Ernat, J.J.; Frangiamore, S.J.; Nuelle, C.W.; Sheean, A.J.; Spindler, K.P.; Bedi, A. The Psychology of ACL Injury, Treatment, and Recovery: Current Concepts and Future Directions. Sports Health. 2024, 19417381241226896. [Google Scholar] [CrossRef] [PubMed]
- Shekhar, A.; Pilar, A.; Ponnanna, K.M.; Tapasvi, S. ACL repair for athletes? J Orthop. 2022, 31, 61–66. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sherman, D.A.; Baumeister, J.; Stock, M.S.; Murray, A.M.; Bazett-Jones, D.M.; Norte, G.E. Inhibition of Motor Planning and Response Selection after Anterior Cruciate Ligament Reconstruction. Med Sci Sports Exerc. 2023, 55, 440–449. [Google Scholar] [CrossRef] [PubMed]
- Shibata, S.; Takemura, M.; Miyakawa, S. Kinematics, Kinetics and Muscle Activity Analysis during Single-leg Drop-jump Landing Followed by an Unanticipated Task: Focusing on Differences in Neurocognitive Function. Int J Sports Phys Ther. 2023, 18, 1085–1093. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sim, K.; Rahardja, R.; Zhu, M.; Young, S.W. Optimal Graft Choice in Athletic Patients with Anterior Cruciate Ligament Injuries: Review and Clinical Insights. Open Access J Sports Med. 2022, 13, 55–67. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Slater, L.V.; Hart, J.M. Quantifying the relationship between quadriceps strength and aerobic fitness following anterior cruciate ligament reconstruction. Phys Ther Sport. 2022, 55, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Smeets, A.; Willems, M.; Gilson, L.; Verschueren, S.; Staes, F.; Vandenneucker, H.; Claes, S.; Vanrenterghem, J. Neuromuscular and biomechanical landing alterations persist in athletes returning to sport after anterior cruciate ligament reconstruction. Knee. 2021, 33, 305–317. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Li, L.; Hughes, G.; Dai, B. Trunk motion and anterior cruciate ligament injuries: A narrative review of injury videos and controlled jump-landing and cutting tasks. Sports Biomech. 2023, 22, 46–64. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Li, L.; Layer, J.; Fairbanks, R.; Jenkins, M.; Hughes, G.; Smith, D.; Wilson, M.; Zhu, Q.; Dai, B. Indirect contact matters: Mid-flight external trunk perturbation increased unilateral anterior cruciate ligament loading variables during jump-landings. J Sport Health Sci. 2023, 12, 534–543. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Stearns, K.M.; Powers, C.M. Improvements in hip muscle performance result in increased use of the hip extensors and abductors during a landing task. Am J Sports Med. 2014, 42, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Stojanović, E.; Terrence Scanlan, A.; Radovanović, D.; Jakovljević, V.; Faude, O. A multicomponent neuromuscular warm-up program reduces lower-extremity injuries in trained basketball players: A cluster randomized controlled trial. Phys Sportsmed. 2023, 51, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Straub, R.K.; Powers, C.M. Is muscular strength a predictor for primary or secondary ACL injury? A scoping review of prospective studies. Phys Ther Sport. 2023, 61, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Sugarman, B.S.; Sullivan, Z.B.; Le, D.; Killelea, C.; Faherty, M.S.; Diehl, L.H.; Wittstein, J.R.; Riboh, J.C.; Toth, A.P.; Amendola, A.; Taylor, D.C.; Sell, T.C. Isometric Knee Strength is Greater in Individuals Who Score Higher on Psychological Readiness to Return to Sport After Primary Anterior Cruciate Ligament Reconstruction. Int J Sports Phys Ther. 2022, 17, 1330–1339. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Taberner, M.; van Dyk, N.; Allen, T.; Jain, N.; Richter, C.; Drust, B.; Betancur, E.; Cohen, D.D. Physical preparation and return to performance of an elite female football player following ACL reconstruction: A journey to the FIFA Women’s World Cup. BMJ Open Sport Exerc Med. 2020, 6, e000843. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Romain, T.; Francois, F. The Role of Foot-Ankle Complex in Rehabilitation After ACLR—Make Miracles Happen. 2023, 12, 310–316.
- Van Cant, J.; Pairot de Fontenay, B.; Douaihy, C.; Rambaud, A. Characteristics of return to running programs following an anterior cruciate ligament reconstruction: A scoping review of 64 studies with clinical perspectives. Phys Ther Sport. 2022, 57, 61–70. [Google Scholar] [CrossRef] [PubMed]
- van Melick, N.; Pronk, Y.; Nijhuis-van der Sanden, M.; Rutten, S.; van Tienen, T.; Hoogeboom, T. Meeting movement quantity or quality return to sport criteria is associated with reduced second ACL injury rate. J Orthop Res. 2022, 40, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Vitharana, T.N.; King, E.; Moran, K. Sensorimotor Dysfunction Following Anterior Cruciate Ligament Reconstruction- an Afferent Perspective: A Scoping Review. Int J Sports Phys Ther. 2024, 19, 1410–1437. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Weingart, A.; Rynecki, N.; Pereira, D. A Review of Neuromuscular Training and Biomechanical Risk Factor Screening for ACL Injury Prevention Among Female Soccer Players. Bull Hosp Jt Dis (2013). 2022, 80, 253–259. [Google Scholar] [PubMed]
- Weir, G. Anterior cruciate ligament injury prevention in sport: Biomechanically informed approaches. Sports Biomech. 2021, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Welling, W.; Frik, L. On-Field Tests for Patients After Anterior Cruciate Ligament Reconstruction: A Scoping Review. Orthop J Sports Med. 2022, 10, 23259671211055481. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Welling, W. Return to sports after an ACL reconstruction in 2024—A glass half full? A narrative review. Phys Ther Sport. 2024, 67, 141–148. [Google Scholar] [CrossRef] [PubMed]
- Wilk, K.; Thomas, Z.M.; Arrigo, C.A.; Davies, G.J. The Need To Change Return to Play Testing in Athletes Following ACL Injury: A Theoretical Model. Int J Sports Phys Ther. 2023, 18, 272–281. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wohl, T.R.; Criss, C.R.; Grooms, D.R. Visual Perturbation to Enhance Return to Sport Rehabilitation after Anterior Cruciate Ligament Injury: A Clinical Commentary. Int J Sports Phys Ther. 2021, 16, 552–564. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- An, Y.W.; Kim, K.M.; DiTrani Lobacz, A.; Baumeister, J.; Higginson, J.S.; Rosen, J.; Swanik, C.B. Cognitive Training Improves Joint Stiffness Regulation and Function in ACLR Patients Compared to Healthy Controls. Healthcare 2023, 11, 1875. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zago, M.; Esposito, F.; Stillavato, S.; Zaffagnini, S.; Frigo, C.A.; Della Villa, F. 3-Dimensional Biomechanics of Noncontact Anterior Cruciate Ligament Injuries in Male Professional Soccer Players. Am J Sports Med. 2024, 52, 1794–1803. [Google Scholar] [CrossRef] [PubMed]
- Zebis, M.K.; Aagaard, P.; Andersen, L.L.; Hölmich, P.; Clausen, M.B.; Brandt, M.; Husted, R.S.; Lauridsen, H.B.; Curtis, D.J.; Bencke, J. First-time anterior cruciate ligament injury in adolescent female elite athletes: A prospective cohort study to identify modifiable risk factors. Knee Surg Sports Traumatol Arthrosc. 2022, 30, 1341–1351. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhang, L.; Hacke, J.D.; Garrett, W.E.; Liu, H.; Yu, B. Bone Bruises Associated with Anterior Cruciate Ligament Injury as Indicators of Injury Mechanism: A Systematic Review. Sports Med. 2019, 49, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Kacprzak, B.; Rosińska, K.; Siuba-Jarosz, N. Hyalofast Cartilage Repair Surgery with a Full Load-Bearing Rehabilitation Program One Day after Operation Reduces the Time for Professional Athletes to Return to Play. Medicina. 2023, 59, 804. [Google Scholar] [CrossRef]
- Loeser, R.F.; Goldring, S.R.; Scanzello, C.R.; Gldring, M.B. Osteoarthritis: A Disease of the Joint as an Organ. Arthritis Rheum 2012, 64, 1697–1707. [Google Scholar] [CrossRef]
- Osteoarthritis Society International—OA Definition. Available online: https://oarsi.org/ (accessed on 4 February 2023).
- Zheng, L.; Zhang, Z.; Sheng, P.; Mobasheri, A. The role of metabolism in chondrocyte dysfunction and the progression of osteoarthritis. Ageing Res. Rev. 2021, 66, 101249. [Google Scholar] [CrossRef]
- Fenwick, S.A.; Gregg, P.J.; Rooney, P. Osteoarthritic cartilage loses its ability to remain avascular. Osteoarthr. Cart. 1999, 7, 441–452. [Google Scholar] [CrossRef]
- Fahy, N.; de Vries-van, M.M.I.; Lehmann, W.; Grotenhuis, N.; Farrel, E.; van der Kraan, P.M.; Murphy, J.M.; Bastiaansen-Jenniskens, Y.M.; van Osch, G.J.V.M. Human osteoarthritic synovium impacts chondrogenic differentiation of mesenchymal stem cells via macrophage polarization state. Osteoarthr. Cart. 2014, 22, 1167–1175. [Google Scholar] [CrossRef]
- Gómez-Aristizábal, A.; Gadhi, R.; Mahomed, N.N.; Marshall, K.W.; Viswanathan, S. Synovial fluid monocyte/macrophage subsets and their correlation to patient-reported outcomes in osteoarthritic patients: A cohort study. Arthritis Res. Ther. 2019, 21, 26. [Google Scholar] [CrossRef] [PubMed]
- Kemble, S.; Croft, A.P. Critical Role of Synovial Tissue–Resident Macrophage and Fibroblast Subsets in the Persistence of Joint Inflammation. Front. Immunol. 2021, 12, 715894. [Google Scholar] [CrossRef] [PubMed]
- Kawanishi, M.; Kami, K.; Nishimura, Y.; Minami, K.; Senba, E.; Umemoto, Y.; Kinoshita, T.; Tajima, F. Exercise-induced increase in M2 macrophages accelerates wound healing in young mice. Physiol. Rep. 2022, 10, e15447. [Google Scholar] [CrossRef] [PubMed]
- Jang, J.S.; Lee, K.; Ju, J.H. Recent Updates of Diagnosis, Pathophysiology, and Treatment on Osteoarthritis of the Knee. Int. J. Mol. Sci. 2021, 22, 2619. [Google Scholar] [CrossRef]
- Katsuola, G.; Kreitmaier, P.; Zeggini, E. Insights into the molecular landscape of osteoarthritis in human tissues. Curr. Opin. Rheumatol. 2022, 34, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.; Jeong, S.; Kim, H.; Kang, D.; Lee, J.; Kang, S.-J.; Kim, J.-H. Disease-modifying therapeutic strategies in osteoarthritis: Current status and future directions. Exp. Mol. Med. 2021, 53, 1689–1696. [Google Scholar] [CrossRef] [PubMed]
- Yu, P.; Zhang, X.; Liu, N.; Tang, L.; Peng, C.; Chen, X. Pyroptosis: Mechanisms and diseases. Signal Transduct. Target. Ther. 2021, 6, 128. [Google Scholar] [CrossRef]
- Jiang, W.; Wang, X.; Su, S.; Du, S.S.H. Identifying the shared genes and KEGG pathways of Resolvin D1-targeted network and osteoarthritis using bioinformatics. Bioengineered 2022, 13, 9839–9854. [Google Scholar] [CrossRef]
- Franceschi, C.; Bonafè, M.; Valensin, S.; Olivieri, F.; De Luca, M.; Ottaviani, E.; De Benedictis, G. Inflamm-aging. An evolutionary perspective on immunosenescence. Ann. N.Y. Acad. Sci. 2000, 908, 244–254. [Google Scholar] [CrossRef]
- Rausch Osthoff, A.-K.; Niedermann, K.; Braun, J.; Adams, J.; Brodin, N.; Dagfinrud, H.; Duruoz, T.; Esbensen, B.A.; Günther, K.-P.; Hurkmans, E.; et al. 2018 EULAR recommendations for physical activity in people with inflammatory arthritis and osteoarthritis. Ann. Rheum. Dis. 2018, 77, 1251–1260. [Google Scholar] [CrossRef]
- Dhillon, R.J.-S.; Hasni, S. Pathogenesis and Management of Sarcopenia. Clin. Geriatr. Med. 2017, 33, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Elagizi, A.; Kachur, S.; Carbone, S.; Lavie, C.J.; Blair, S.N. A Review of Obesity, Physical Activity and Cardiovascular Disease. Curr. Obes. Rep. 2020, 9, 571–581. [Google Scholar] [CrossRef] [PubMed]
- Boutron, I.; Tubach, F.; Giradeau, B.; Ravaud, P. Blinding was judged more difficult to achieve and maintain in nonpharmacologic than pharmacologic trials. J. Clin. Epidemiol. 2004, 57, 543–550. [Google Scholar] [CrossRef] [PubMed]
- Fregni, F.; Imamura, M.; Chien, H.F.; Lew, H.L.; Boggio, P.; Kaptchuk, T.J.; Riberto, M.; Hsing, W.T.; Battistella, L.R.; Furlan, A. Challenges and Recommendations for Placebo Controls in Randomized Trials in Physical and Rehabilitation Medicine: A Report of the International Placebo Symposium Working Group. Am. J. Phys. Med. Rehabil. 2010, 89, 160–172. [Google Scholar] [CrossRef]
- Villamar, M.F.; Contreras, V.S.; Kuntz, R.E.; Fregni, F. The Reporting of Blinding in Physical Medicine and Rehabilitation Randomized Controlled Trials: A Systematic Review. J. Rehabil. Med. 2013, 45, 6–13. [Google Scholar] [CrossRef]
- Lv, Z.; Yang, Y.X.; Li, J.; Fei, Y.; Guo, H.; Sun, Z.; Lu, J.; Xu, X.; Jiang, Q.; Ikegawa, S.; et al. Molecular Classification of Knee Osteoarthritis Front. Cell Dev. Biol. 2021, 9, 725568. [Google Scholar] [CrossRef]
- Vassao, P.G.; de Souza, A.C.F.; da Silveira Campos, R.M.; Garcia, L.A.; Tucci, H.T.; Renno, A.C.M. Effects of photobiomodulation and a physical exercise program on the expression of inflammatory and cartilage degradation biomarkers and functional capacity in women with knee osteoarthritis: A randomized blinded study. Adv. Rheumatol. 2021, 61. [Google Scholar] [CrossRef]
- Kim, J.-R.; Yoo, J.J.; Kim, H.A. Therapeutics in Osteoarthritis Based on an Understanding of Its Molecular Pathogenesis. Int. J. Mol. Sci. 2018, 19, 674. [Google Scholar] [CrossRef]
- American Kinesiotherapy Association. Available online: https://akta.org/about/history (accessed on 4 February 2023).
- Alves, M.D.d.J.; Silva, D.d.S.; Pereira, E.V.M.; Pereira, D.D.; de Sousa Fernandes, M.S.; Santos, D.F.C.; Oliveira, D.P.M.; Vieira-Souza, L.M.; Aidar, F.J.; de Souza, R.F. Changes in Cytokines Concentration Following Long-Distance Running: A Systematic Review and Meta-Analysis. Front. Physiol. 2022, 13, 838069. [Google Scholar] [CrossRef]
- Balan, E.; De Groote, E.; Bouillon, M.; Vieconte, N.; Mahieu, M.N.; Aslain, D.; Nielens, H.; Decottignies, A.; Deldicque, L. No effect of the endurance training status on senescence despite reduced inflammation in skeletal muscle of older individuals. Am. J. Physiol. Endocrinol. Metab. 2020, 319, E447–E454. [Google Scholar] [CrossRef]
- Lehner, C.; et al. Tenophages: A novel macrophage-like tendon cell population expressing CX3CL1 and CX3CR1. DMM Dis Model Mech. 2019, 12. [Google Scholar] [CrossRef] [PubMed]
- Bi, Y.; Ehirchiou, D.; Kilts, T.M.; Inkson, C.A.; Embree, M.C.; Sonoyama, W.; Li, L.; Leet, A.I.; Seo, B.-M.; Zhang, L.; Shi, S.; Young, M.F. Identification of tendon stem/progenitor cells and the role of the extracellular matrix in their niche. Nat Med. 2007, 13, 1219–1227. [Google Scholar] [CrossRef] [PubMed]
- Rui, Y.F.; Lui, P.P.Y.; Ni, M.; Chan, L.S.; Lee, Y.W.; Chan, K.M. Mechanical loading increased BMP-2 expression which promoted osteogenic differentiation of tendon-derived stem cells. J Orthop Res. 2011, 29, 390–396. [Google Scholar] [CrossRef]
- Spiesz, E.; Thorpe, C.T.; Chaudhry, S.; Riley, G.P.; Birch, H.L.; Clegg, P.D.; Screen, H.R.C. Tendon extracellular matrix damage, degradation and inflammation in response to in vitro overload exercise. J Orthop Res. 2015, 33, 889–897. [Google Scholar] [CrossRef] [PubMed]
- Cadby, J.A.; Buehler, E.; Godbout, C.; Van Weeren, P.R.; Snedeker, J.G.; Lionetti, V. Differences between the cell populations from the peritenon and the tendon core with regard to their potential implication in tendon repair. PLoS ONE. 2014, 9, e92474. [Google Scholar] [CrossRef] [PubMed]
- Dyment, N.A.; Hagiwara, Y.; Matthews, B.G.; Li, Y.; Kalajzic, I.; Rowe, D.W. Lineage tracing of resident tendon progenitor cells during growth and natural healing. PLoS ONE. 2014, 9, e96113. [Google Scholar] [CrossRef]
- Yoshida, R.; Alaee, F.; Dyrna, F.; Kronenberg, M.S.; Maye, P.; Kalajzic, I.; Rowe, D.W.; Mazzocca, A.D.; Dyment, N.A. Murine supraspinatus tendon injury model to identify the cellular origins of rotator cuff healing. Connect Tissue Res. 2016, 57, 507–515. [Google Scholar] [CrossRef] [PubMed]
- Carroll, C.C.; Dickinson, J.M.; Haus, J.M.; Lee, G.A.; Hollon, C.J.; Aagaard, P.; Magnusson, S.P.; Trappe, T.A. Influence of aging on the in vivo properties of human patellar tendon. J Appl Physiol. 2008, 105, 1907–1915. [Google Scholar] [CrossRef]
- Light, N.D.; Bailey, A.J. Changes in crosslinking during aging in bovine tendon collagen. FEBS letters 1979, 97, 183–188. [Google Scholar] [CrossRef]
- Li, Y.; Fessel, G.; Georgiadis, M.; Snedeker, J.G. Advanced glycation end-products diminish tendon collagen fiber sliding. Matrix Biol. 2013, 32, 169–177. [Google Scholar] [CrossRef]
- Thorpe, C.T.; Riley, G.P.; Birch, H.L.; Clegg, P.D.; Screen, H.R.C. Fascicles from energy-storing tendons show an age-specific response to cyclic fatigue loading. J R Soc Interface. 2014, 11, 20131058. [Google Scholar] [CrossRef] [PubMed]
- Zuskov, A.; et al. Tendon biomechanics and crimp properties following fatigue loading are influenced by tendon type and age in mice. J Orthop Res. 2020, 54, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Thorpe, C.T.; McDermott, B.T.; Goodship, A.E.; Clegg, P.D.; Birch, H.L. Ageing does not result in a decline in cell synthetic activity in an injury prone tendon. Scand J Med Sci Sports. 2016, 26, 684–693. [Google Scholar] [CrossRef] [PubMed]
- Connizzo, B.K.; Piet, J.M.; Shefelbine, S.J.; Grodzinsky, A.J. Age-associated changes in the response of tendon explants to stress deprivation is sex-dependent. Connect Tissue Res. 2020, 61, 48–62. [Google Scholar] [CrossRef]
- Zhou, Z.; Akinbiyi, T.; Xu, L.; Ramcharan, M.; Leong, D.J.; Ros, S.J.; Colvin, A.C.; Schaffler, M.B.; Majeska, R.J.; Flatow, E.L.; Sun, H.B. Tendon-derived stem/progenitor cell aging: Defective self-renewal and altered fate. Aging Cell. 2010, 9, 911–915. [Google Scholar] [CrossRef]
- Veres, S.P.; Harrison, J.M.; Lee, J.M. Repeated subrupture overload causes progression of nanoscaled discrete plasticity damage in tendon collagen fibrils. J Orthop Res. 2013, 31, 731–737. [Google Scholar] [CrossRef]
- Egerbacher, M.; Arnoczky, S.P.; Caballero, O.; Lavagnino, M.; Gardner, K.L. Loss of homeostatic tension induces apoptosis in tendon cells: An in vitro study. Clin Orthop Relat Res. 2008, 466, 1562–1568. [Google Scholar] [CrossRef]
- Thorpe, C.T.; et al. Tendon overload results in alterations in cell shape and increased markers of inflammation and matrix degradation. Scand J Med Sci Sports. 2015, 25, e381–e391. [Google Scholar] [CrossRef]
- Lavagnino, M.; Arnoczky, S.P.; Gardner, K.L. In situ deflection of tendon cell-cilia in response to tensile loading: An in vitro study. J Orthop Res. 2011, 29, 925–930. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, E.; Ascenzi, M.-G.; Farnum, C. Primary cilia are highly oriented with respect to collagen direction and long axis of extensor tendon. J Orthop Res. 2009, 28. [Google Scholar] [CrossRef]
- Gardner, K.; Arnoczky, S.P.; Lavagnino, M. Effect of in vitro stress-deprivation and cyclic loading on the length of tendon cell cilia in situ. J Orthop Res. 2011, 29, 582–587. [Google Scholar] [CrossRef] [PubMed]
- Rowson, D.T.; Shelton, J.C.; Screen, H.R.C.; Knight, M.M. Mechanical loading induces primary cilia disassembly in tendon cells via TGFβ and HDAC6. Sci Rep. 2018, 8, 11107. [Google Scholar] [CrossRef] [PubMed]
- Rowson, D.; Knight, M.M.; Screen, H.R.C. Zonal variation in primary cilia elongation correlates with localized biomechanical degradation in stress deprived tendon. J Orthop Res. 2016, 34, 2146–2153. [Google Scholar] [CrossRef] [PubMed]
- Fang, F.; Schwartz, A.G.; Moore, E.R.; Sup, M.E.; Thomopoulos, S. Primary cilia as the nexus of biophysical and hedgehog signaling at the tendon enthesis. Sci Adv. 2020, 6, eabc1799. [Google Scholar] [CrossRef]
- Cohen, M.; Kam, Z.; Addadi, L.; Geiger, B. Dynamic study of the transition from hyaluronan- to integrin-mediated adhesion in chondrocytes. EMBO J. 2006, 25, 302–311. [Google Scholar] [CrossRef]
- Chopra, A.; Murray, M.E.; Byfield, F.J.; Mendez, M.G.; Halleluyan, R.; Restle, D.J.; Raz-Ben Aroush, D.; Galie, P.A.; Pogoda, K.; Bucki, R.; et al. Augmentation of integrin-mediated mechanotransduction by hyaluronic acid. Biomaterials. 2014, 35, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Smith, S.M.; Thomas, C.E.; Birk, D.E. Pericellular proteins of the developing mouse tendon: A proteomic analysis. Connect Tissue Res. 2012, 53, 2–13. [Google Scholar] [CrossRef]
- Docheva, D.; Popov, C.; Alberton, P.; Aszodi, A. Integrin signaling in skeletal development and function. Birth Defects Res Part C Embryo Today Rev. 2014, 102, 13–36. [Google Scholar] [CrossRef]
- Wang, D.; Pun, C.C.M.; Huang, S.; Tang, T.C.M.; Ho, K.K.W.; Rothrauff, B.B.; Yung, P.S.H.; Blocki, A.M.; Ker, E.D.F.; Tuan, R.S. Tendon-derived extracellular matrix induces mesenchymal stem cell tenogenesis via an integrin/transforming growth factor-β crosstalk-mediated mechanism. FASEB J. 2020, 34, 8172–8186. [Google Scholar] [CrossRef]
- Mousavizadeh, R.; Hojabrpour, P.; Eltit, F.; McDonald, P.C.; Dedhar, S.; McCormack, R.G.; Duronio, V.; Jafarnejad, S.M.; Scott, A. β1 integrin, ILK and mTOR regulate collagen synthesis in mechanically loaded tendon cells. Sci Rep. 2020, 10. [Google Scholar] [CrossRef]
- Popov, C.; Burggraf, M.; Kreja, L.; Ignatius, A.; Schieker, M.; Docheva, D. Mechanical stimulation of human tendon stem/progenitor cells results in upregulation of matrix proteins, integrins and MMPs, and activation of p38 and ERK1/2 kinases. BMC Mol Biol. 2015, 16, 6. [Google Scholar] [CrossRef]
- Hannafin, J.A.; Attia, E.A.; Henshaw, R.; Warren, R.F.; Bhargava, M.M. Effect of cyclic strain and plating matrix on cell proliferation and integrin expression by ligament fibroblasts. J Orthop Res. 2006, 24, 149–158. [Google Scholar] [CrossRef]
- Kaneko, D.; Sasazaki, Y.; Kikuchi, T.; Ono, T.; Nemoto, K.; Matsumoto, H.; Toyama, Y. Temporal effects of cyclic stretching on distribution and gene expression of integrin and cytoskeleton by ligament fibroblasts in vitro. Connect Tissue Res. 2009, 50, 263–269. [Google Scholar] [CrossRef]
- Bayer, M.L.; Schjerling, P.; Herchenhan, A.; Zeltz, C.; Heinemeier, K.M.; Christensen, L.; Krogsgaard, M.; Gullberg, D.; Kjaer, M. Release of tensile strain on engineered human tendon tissue disturbs cell adhesions, changes matrix architecture, and induces an inflammatory phenotype. PLoS ONE. 2014, 9, e86078. [Google Scholar] [CrossRef]
- Sun, H.B.; Andarawis-Puri, N.; Li, Y.; Fung, D.T.; Lee, J.Y.; Wang, V.M.; Basta-Pljakic, J.; Leong, D.J.; Sereysky, J.B.; Ros, S.J.; Klug, R.A.; Braman, J.; Schaffler, M.B.; Jepsen, K.J.; Flatow, E.L. Cycle-dependent matrix remodeling gene expression response in fatigue-loaded rat patellar tendons. J Orthop Res. 2010, 28, 1380–1386. [Google Scholar] [CrossRef]
- McNeilly, C.M.; Banes, A.J.; Benjamin, M.; Ralphs, J.R. Tendon cells in vivo form a three-dimensional network of cell processes linked by gap junctions. J Anat. 1996, 189, 593–600. [Google Scholar] [CrossRef]
- Waggett, A.D.; Benjamin, M.; Ralphs, J.R. Connexin 32 and 43 gap junctions differentially modulate tenocyte response to cyclic mechanical load. Eur J Cell Biol. 2006, 85, 1145–1154. [Google Scholar] [CrossRef]
- Shen, H.; Schwartz, A.G.; Civitelli, R.; Thomopoulos, S. Connexin 43 is necessary for murine tendon enthesis formation and response to loading. J Bone Miner Res. 2020, 35, 4018. [Google Scholar] [CrossRef]
- Foolen, J.; Wunderli, S.L.; Loerakker, S.; Snedeker, J.G. Tissue alignment enhances remodeling potential of tendon-derived cells—Lessons from a novel microtissue model of tendon scarring. Matrix Biol. 2018, 65, 14–29. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Tytell, J.D.; Ingber, D.E. Mechanotransduction at a distance: Mechanically coupling the extracellular matrix with the nucleus. Nat Rev Mol Cell Biol. 2009, 10, 75–82. [Google Scholar] [CrossRef]
- Lavagnino, M.; Arnoczky, S.P. In vitro alterations in cytoskeletal tensional homeostasis control gene expression in tendon cells. J Orthop Res. 2005, 23, 1211–1218. [Google Scholar] [CrossRef] [PubMed]
- Lavagnino, M.; Gardner, K.L.; Arnoczky, S.P. High magnitude, in vitro, biaxial, cyclic tensile strain induces actin depolymerization in tendon cells. Muscles Ligaments Tendons J. 2015, 5, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Lavagnino, M.; Brooks, A.E.; Oslapas, A.N.; Gardner, K.L.; Arnoczky, S.P. Crimp length decreases in lax tendons due to cytoskeletal tension, but is restored with tensional homeostasis. J Orthop Res. 2017, 35, 573–579. [Google Scholar] [CrossRef]
- Lavagnino, M.; Gardner, K.; Arnoczky, S.P. Age-related changes in the cellular, mechanical, and contractile properties of rat tail tendons. Connect Tissue Res. 2013, 54, 70–75. [Google Scholar] [CrossRef]
- Lavagnino, M.; Bedi, A.; Walsh, C.P.; Sibilsky Enselman, E.R.; Sheibani-Rad, S.; Arnoczky, S.P. Tendon contraction after cyclic elongation is an age-dependent phenomenon: In vitro and in vivo comparisons. Am J Sports Med. 2014, 42, 1471–1477. [Google Scholar] [CrossRef]
- Arnoczky, S.P.; Lavagnino, M.; Whallon, J.H.; Hoonjan, A. In situ cell nucleus deformation in tendons under tensile load; a morphological analysis using confocal laser microscopy. J Orthop Res. 2002, 20, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Henderson, J.T.; Shannon, G.; Veress, A.I.; Neu, C.P. Direct measurement of intranuclear strain distributions and RNA synthesis in single cells embedded within native tissue. Biophys J. 2013, 105, 2252–2261. [Google Scholar] [CrossRef] [PubMed]
- Long, H.; Liu, Q.; Yin, H.; Wang, K.; Diao, N.; Zhang, Y.; et al. Prevalence trends of site-specific osteoarthritis from 1990 to 2019: Findings from the Global Burden of Disease Study 2019. Arthritis Rheumatol. 2022, 74, 1172–1183. [Google Scholar] [CrossRef] [PubMed]
- Cieza, A.; Causey, K.; Kamenov, K.; Hanson, S.W.; Chatterji, S.; Vos, T. Global estimates of the need for rehabilitation based on the Global Burden of Disease Study 2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2021, 396, 2006–2017. [Google Scholar] [CrossRef]
- Safiri, S.; Kolahi, A.A.; Smith, E.; Hill, C.; Bettampadi, D.; Mansournia, M.A.; et al. Global, regional and national burden of osteoarthritis 1990-2017: A systematic analysis of the Global Burden of Disease Study 2017. Ann. Rheum. Dis. 2020, 79, 819–828. [Google Scholar] [CrossRef]
- Poole, A.R. Osteoarthritis as a whole joint disease. HSS J. 2012, 8, 4–6. [Google Scholar] [CrossRef]
- Blanco, F.J. Osteoarthritis and atherosclerosis in joint disease. Reumatol. Clin. (Engl Ed) 2018, 14, 251–253. [Google Scholar] [CrossRef] [PubMed]
- Uhlen, M.; Fagerberg, L.; Hallstrom, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; et al. Tissue-based map of the human proteome. Science 2015, 347, 1260419. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, M.; Zhang, C.; Méar, L.; Zhong, W.; Digre, A.; Katona, B.; et al. A single-cell type transcriptomics map of human tissues. Sci. Adv. 2021, 7, eabh2169. [Google Scholar] [CrossRef] [PubMed]
- Loeser, R.F.; Goldring, S.R.; Scanzello, C.R.; Goldring, M.B. Osteoarthritis: A disease of the joint as an organ. Arthritis Rheum. 2012, 64, 1697–1707. [Google Scholar] [CrossRef] [PubMed]
- Aebersold, R.; Bader, G.D.; Edwards, A.M.; van Eyk, J.E.; Kussmann, M.; Qin, J.; et al. The biology/disease-driven human proteome project (B/D-HPP): Enabling protein research for the life sciences community. J. Proteome Res. 2013, 12, 23–27. [Google Scholar] [CrossRef]
- Han, Y.; Wennersten, S.A.; Lam, M.P.Y. Working the literature harder: What can text mining and bibliometric analysis reveal? Expert Rev. Proteomics 2019, 16, 871–873. [Google Scholar] [CrossRef]
- Ruiz-Romero, C.; Lam, M.P.Y.; Nilsson, P.; Önnerfjord, P.; Utz, P.J.; Van Eyk, J.E.; et al. Mining the proteome associated with rheumatic and autoimmune diseases. J. Proteome Res. 2019, 18, 4231–4239. [Google Scholar] [CrossRef]
- Kraus, V.B.; Blanco, F.J.; Englund, M.; Henrotin, Y.; Lohmander, L.S.; Losina, E.; et al. OARSI clinical trials recommendations: Soluble biomarker assessments in clinical trials in osteoarthritis. Osteoarthritis Cartilage 2015, 23, 686–697. [Google Scholar] [CrossRef]
- Lau, E.; Venkatraman, V.; Thomas, C.T.; Wu, J.C.; Van Eyk, J.E.; Lam, M.P.Y. Identifying high-priority proteins across the human diseasome using semantic similarity. J. Proteome Res. 2018, 17, 4267–4278. [Google Scholar] [CrossRef] [PubMed]
- Szklarczyk, D.; Gable, A.L.; Nastou, K.C.; Lyon, D.; Kirsch, R.; Pyysalo, S.; et al. The STRING database in 2021: Customizable protein-protein networks, and functional characterization of user-uploaded gene/measurement sets. Nucleic Acids Res. 2021, 49, D605–D612. [Google Scholar] [CrossRef] [PubMed]
- Supek, F.; Bošnjak, M.; Škunca, N.; Šmuc, T. REVIGO summarizes and visualizes long lists of gene ontology terms. PLoS ONE 2011, 6, e21800. [Google Scholar] [CrossRef] [PubMed]
- Thysen, S.; Luyten, F.P.; Lories, R.J. Loss of Frzb and Sfrp1 differentially affects joint homeostasis in instability-induced osteoarthritis. Osteoarthritis Cartilage 2015, 23, 275–279. [Google Scholar] [CrossRef]
- Cai, S.; Zou, Y.; Zhao, H.; Lin, H.; Zheng, D.; Xu, L.; et al. Mechanical stress reduces secreted frizzled-related protein expression and promotes temporomandibular joint osteoarthritis via Wnt/β-catenin signaling. Bone 2022, 161, 116445. [Google Scholar] [CrossRef]
- Claudel, M.; Jouzeau, J.Y.; Cailotto, F. Secreted Frizzled-related proteins (sFRPs) in osteo-articular diseases: Much more than simple antagonists of Wnt signaling? FEBS J. 2019, 286, 4832–4851. [Google Scholar] [CrossRef]
- Wang, R.; Zhu, J.; Dong, X.; Shi, M.; Lu, C.; Springer, T.A. GARP regulates the bioavailability and activation of TGFβ. Mol. Biol. Cell 2012, 23, 1129–1139. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.J.; Chang, W.A.; Wu, L.Y.; Hsu, Y.L.; Chen, C.H.; Kuo, P.L. Systematic analysis of transcriptomic profile of chondrocytes in osteoarthritic knee using next-generation sequencing and bioinformatics. J. Clin. Med. 2018, 7, 535. [Google Scholar] [CrossRef]
- Jayaseelan, V.P.; Arumugam, P. A computational data mining strategy to identify the common genetic markers of temporomandibular joint disorders and osteoarthritis. Glob. Med. Genet. 2022, 9, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Lourido, L.; Calamia, V.; Mateos, J.; Fernández-Puente, P.; Fernández-Tajes, J.; Blanco, F.J.; et al. Quantitative proteomic profiling of human articular cartilage degradation in osteoarthritis. J. Proteome Res. 2014, 13, 6096–6106. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.; Choi, J.; Hwang, N.S. Regulation of lubricin for functional cartilage tissue regeneration: A review. Biomater. Res. 2018, 22, 9. [Google Scholar] [CrossRef]
- Carballo, C.B.; Nakagawa, Y.; Sekiya, I.; Rodeo, S.A. Basic science of articular cartilage. Clin. Sports Med. 2017, 36, 413–425. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Puente, P.; González-Rodríguez, L.; Calamia, V.; Picchi, F.; Lourido, L.; Camacho-Encina, M.; et al. Analysis of endogenous peptides released from osteoarthritic cartilage unravels novel pathogenic markers. Mol. Cell. Proteomics 2019, 18, 2018–2028. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.J.; Cheng, J.H.; Chou, W.Y.; Hsu, S.L.; Chen, J.H.; Huang, C.Y. Changes of articular cartilage and subchondral bone after extracorporeal shockwave therapy in osteoarthritis of the knee. Int. J. Med. Sci. 2017, 14, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Goldring, S.R.; Goldring, M.B. Changes in the osteochondral unit during osteoarthritis: Structure, function and cartilage-bone crosstalk. Nat. Rev. Rheumatol. 2016, 12, 632–644. [Google Scholar] [CrossRef]
- Funck-Brentano, T.; Cohen-Solal, M. Subchondral bone and osteoarthritis. Curr. Opin. Rheumatol. 2015, 27, 420–426. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, C.; Gabay, C.; Salvat, C.; Henrotin, Y.E.; Berenbaum, F. Mechanical loading highly increases IL-6 production and decreases OPG expression by osteoblasts. Osteoarthritis Cartilage 2009, 17, 473–481. [Google Scholar] [CrossRef]
- Sanchez, C.; Deberg, M.A.; Piccardi, N.; Msika, P.; Reginster, J.Y.; Henrotin, Y.E. Subchondral bone osteoblasts induce phenotypic changes in human osteoarthritic chondrocytes. Osteoarthritis Cartilage 2005, 13, 988–997. [Google Scholar] [CrossRef]
- Yu, D.; Xu, J.; Liu, F.; Wang, X.; Mao, Y.; Zhu, Z. Subchondral bone changes and the impacts on joint pain and articular cartilage degeneration in osteoarthritis. Clin. Exp. Rheumatol. 2016, 34, 929–934. [Google Scholar] [PubMed]
- Chan, P.M.B.; Zhu, L.; Wen, C.Y.; Chiu, K.Y. Subchondral bone proteomics in osteoarthritis: Current status and perspectives. J. Orthop. Translat. 2015, 3, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, C.; Pesesse, L.; Gabay, C.; Delcour, J.P.; Msika, P.; Baudouin, C.; et al. Regulation of subchondral bone osteoblast metabolism by cyclic compression. Arthritis Rheum. 2012, 64, 1193–1203. [Google Scholar] [CrossRef]
- Sanchez, C.; Deberg, M.A.; Piccardi, N.; Msika, P.; Reginster, J.Y.; Henrotin, Y.E. Osteoblasts from the sclerotic subchondral bone downregulate aggrecan but upregulate metalloproteinases expression by chondrocytes. This effect is mimicked by interleukin-6, -1beta and oncostatin M pre-treated non-sclerotic osteoblasts. Osteoarthritis Cartilage 2005, 13, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Chim, S.M.; Tickner, J.; Chow, S.T.; Kuek, V.; Guo, B.; Zhang, G.; et al. Angiogenic factors in bone local environment. Cytokine Growth Factor Rev. 2013, 24, 297–310. [Google Scholar] [CrossRef] [PubMed]
- Creecy, J.G.; Damrath, J.M.; Wallace, J.M. Control of bone matrix properties by osteocytes. Front. Endocrinol. 2020, 11, 578477. [Google Scholar] [CrossRef] [PubMed]
- Sakao, K.; Takahashi, K.A.; Arai, Y.; Saito, M.; Honjyo, K.; Hiraoka, N.; et al. Asporin and transforming growth factor-beta gene expression in osteoblasts from subchondral bone and osteophytes in osteoarthritis. J. Orthop. Sci. 2009, 14, 738–747. [Google Scholar] [CrossRef]
- MacDonald, I.J.; Liu, S.C.; Su, C.M.; Wang, Y.H.; Tsai, C.H.; Tang, C.H. Implications of angiogenesis involvement in arthritis. Int. J. Mol. Sci. 2018, 19, 2012. [Google Scholar] [CrossRef] [PubMed]
- Mishra, A.; Awasthi, S.; Raj, S.; Mishra, P.; Srivastava, R.N. Identifying the role of ASPN and COMP genes in knee osteoarthritis development. J. Orthop. Surg. Res. 2019, 14, 337. [Google Scholar] [CrossRef] [PubMed]
- Mathiessen, A.; Conaghan, P.G. Synovitis in osteoarthritis: Current understanding with therapeutic implications. Arthritis Res. Ther. 2017, 19, 18. [Google Scholar] [CrossRef]
- Orr, C.; Vieira-Sousa, E.; Boyle, D.L.; Buch, M.H.; Buckley, C.D.; Cañete, J.D.; et al. Synovial tissue research: A state-of-the-art review. Nat. Rev. Rheumatol. 2017, 13, 463–475. [Google Scholar] [CrossRef]
- Kemble, S.; Croft, A.P. Critical role of synovial tissue-resident macrophage and fibroblast subsets in the persistence of joint inflammation. Front. Immunol. 2021, 12, 715894. [Google Scholar] [CrossRef]
- Wehling, P.; Evans, C.; Wehling, J.; Maixner, W. Effectiveness of intra-articular therapies in osteoarthritis: A literature review. Ther. Adv. Musculoskelet. Dis. 2017, 9, 183–196. [Google Scholar] [CrossRef]
- Lambert, C.; Dubuc, J.E.; Montell, E.; Vergés, J.; Munaut, C.; Noël, A.; et al. Gene expression pattern of cells from inflamed and normal areas of osteoarthritis synovial membrane. Arthritis Rheumatol. 2014, 66, 960–968. [Google Scholar] [CrossRef] [PubMed]
- Plsikova Matejova, J.; Spakova, T.; Harvanova, D.; Lacko, M.; Filip, V.; Sepitka, R.; et al. A preliminary study of combined detection of COMP, TIMP-1, and MMP-3 in synovial fluid: Potential indicators of osteoarthritis progression. Cartilage 2020, 13, 1421S–1430S. [Google Scholar] [CrossRef] [PubMed]
- Ravalli, S.; Szychlinska, M.A.; Lauretta, G.; Di Rosa, M.; Musumeci, G. Investigating lubricin and known cartilage-based biomarkers of osteoarthritis. Expert Rev. Mol. Diagn. 2020, 20, 443–452. [Google Scholar] [CrossRef]
- Qadri, M.; Jay, G.D.; Zhang, L.X.; Schmidt, T.A.; Totonchy, K.A.; Elsaid, K.A. Proteoglycan-4 is an essential regulator of synovial macrophage polarization and inflammatory macrophage joint infiltration. Arthritis Res. Ther. 2021, 23, 241. [Google Scholar] [CrossRef] [PubMed]
- Mucke, J.; Hoyer, A.; Brinks, R.; Bleck, E.; Pauly, T.; Schneider, M.; et al. Inhomogeneity of immune cell composition in the synovial sublining: Linear mixed modelling indicates differences in distribution and spatial decline of CD68+ macrophages in osteoarthritis and rheumatoid arthritis. Arthritis Res. Ther. 2016, 18, 170. [Google Scholar] [CrossRef]
- Tamer, T.M. Hyaluronan and synovial joint: Function, distribution and healing. Interdiscip. Toxicol. 2013, 6, 111–125. [Google Scholar] [CrossRef] [PubMed]
- Posey, K.L.; Coustry, F.; Hecht, J.T. Cartilage oligomeric matrix protein: COMPopathies and beyond. Matrix Biol. 2018, 71–72, 161–173. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, D.J. Serpins in cartilage and osteoarthritis: What do we know? Biochem. Soc. Trans. 2021, 49, 1013–1026. [Google Scholar] [CrossRef]
- Wilkinson, D.J.; Falconer, A.M.D.; Wright, H.L.; Lin, H.; Yamamoto, K.; Cheung, K.; et al. Matrix metalloproteinase-13 is fully activated by neutrophil elastase and inactivates its serpin inhibitor, alpha-1 antitrypsin: Implications for osteoarthritis. FEBS J. 2021, 289, 121–139. [Google Scholar] [CrossRef]
- Heit, C.; Jackson, B.C.; McAndrews, M.; Wright, M.W.; Thompson, D.C.; Silverman, G.A.; et al. Update of the human and mouse SERPIN gene superfamily. Hum. Genomics 2013, 7, 22. [Google Scholar] [CrossRef]
- Fox, A.J.; Bedi, A.; Rodeo, S.A. The basic science of human knee menisci: Structure, composition, and function. Sports Health 2012, 4, 340–351. [Google Scholar] [CrossRef] [PubMed]
- Englund, M.; Roemer, F.W.; Hayashi, D.; Crema, M.D.; Guermazi, A. Meniscus pathology, osteoarthritis and the treatment controversy. Nat. Rev. Rheumatol. 2012, 8, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Folkesson, E.; Turkiewicz, A.; Ali, N.; Rydén, M.; Hughes, H.V.; Tjörnstrand, J.; et al. Proteomic comparison of osteoarthritic and reference human menisci using data-independent acquisition mass spectrometry. Osteoarthritis Cartilage 2020, 28, 1092–1101. [Google Scholar] [CrossRef]
- Sun, Y.; Mauerhan, D.R. Meniscal calcification, pathogenesis and implications. Curr. Opin. Rheumatol. 2012, 24, 152–157. [Google Scholar] [CrossRef]
- Kiraly, A.J.; Roberts, A.; Cox, M.; Mauerhan, D.; Hanley, E.; Sun, Y. Comparison of meniscal cell-mediated and chondrocyte-mediated calcification. Open Orthop. J. 2017, 11, 225–233. [Google Scholar] [CrossRef]
- Schulze-Tanzil, G. Intraarticular ligament degeneration is interrelated with cartilage and bone destruction in osteoarthritis. Cells 2019, 8, 990. [Google Scholar] [CrossRef] [PubMed]
- Harkey, M.S.; Luc, B.A.; Golightly, Y.M.; Thomas, A.C.; Driban, J.B.; Hackney, A.C.; et al. Osteoarthritis-related biomarkers following anterior cruciate ligament injury and reconstruction: A systematic review. Osteoarthritis Cartilage 2015, 23, 1–12. [Google Scholar] [CrossRef]
- Yoshida, H.; Kojima, T.; Kurokouchi, S.; Takahashi, S.; Hanamura, H.; Kojima, M.; et al. Relationship between pre-radiographic cartilage damage following anterior cruciate ligament injury and biomarkers of cartilage turnover in clinical practice: A cross-sectional observational study. Osteoarthritis Cartilage 2013, 21, 831–838. [Google Scholar] [CrossRef]
- Tang, N.; Zhang, W.; George, D.M.; Su, Y.; Huang, T. The Top 100 most cited articles on anterior cruciate ligament reconstruction: A bibliometric analysis. Orthop. J. Sports Med. 2021, 9, 2325967120976372. [Google Scholar] [CrossRef]
- Zhang, D.; Cheriyan, T.; Martin, S.D.; Gomoll, A.H.; Schmid, T.M.; Spector, M. Lubricin distribution in the torn human anterior cruciate ligament and meniscus. J. Orthop. Res. 2011, 29, 1916–1922. [Google Scholar] [CrossRef] [PubMed]
- Jay, G.D.; Fleming, B.C.; Watkins, B.A.; McHugh, K.A.; Anderson, S.C.; Zhang, L.X.; et al. Prevention of cartilage degeneration and restoration of chondroprotection by lubricin tribosupplementation in the rat following anterior cruciate ligament transection. Arthritis Rheum. 2010, 62, 2382–2391. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, W. The role of HOPX in normal tissues and tumor progression. Biosci. Rep. 2020, 40, BSR20191953. [Google Scholar] [CrossRef] [PubMed]
- Tarka, M.C.; Davey, A.; Lonza, G.C.; O’Brien, C.M.; Delaney, J.P.; Endres, N.K. Alpine ski racing injuries. Sports Health 2019, 11, 265–271. [Google Scholar] [CrossRef]
- Chin, B.Z.; Wee, I.J.Y.; Syn, N.L.; Krishna, L. Arthroscopic anterior cruciate ligament reconstruction: A meta-analysis comparing semitendinosus alone and semitendinosus with gracilis tendon autografts. J. Knee Surg. 2019, 32, 796–803. [Google Scholar] [CrossRef] [PubMed]
- Lotz, M.; Martel-Pelletier, J.; Christiansen, C.; Brandi, M.L.; Bruyère, O.; Chapurlat, R.; et al. Value of biomarkers in osteoarthritis: Current status and perspectives. Ann. Rheum. Dis. 2013, 72, 1756–1763. [Google Scholar] [CrossRef] [PubMed]
- Styrkarsdottir, U.; Lund, S.H.; Thorleifsson, S.; Saevarsdottir, S.; Gudbjartsson, D.F.; Thorsteinsdottir, U.; et al. The CRTAC1 protein in plasma associates with prevalent osteoarthritis and predicts future risk as well as progression to joint replacements—Results from the UK Biobank resource. Arthritis Rheumatol. 2022, 75, 544–552. [Google Scholar] [CrossRef]
- Valdes, A.M.; Loughlin, J.; Oene, M.V.; Chapman, K.; Surdulescu, G.L.; Doherty, M.; et al. Sex and ethnic differences in the association of ASPN, CALM1, COL2A1, COMP, and FRZB with genetic susceptibility to osteoarthritis of the knee. Arthritis Rheum. 2007, 56, 137–146. [Google Scholar] [CrossRef] [PubMed]
- King, K.B.; Lindsey, C.T.; Dunn, T.C.; Ries, M.D.; Steinbach, L.S.; Majumdar, S. A study of the relationship between molecular biomarkers of joint degeneration and the magnetic resonance-measured characteristics of cartilage in 16 symptomatic knees. Magn. Reson. Imaging 2004, 22, 1117–1123. [Google Scholar] [CrossRef] [PubMed]
- Liem, Y.; Judge, A.; Kirwan, J.; Ourradi, K.; Li, Y.; Sharif, M. Multivariable logistic and linear regression models for identification of clinically useful biomarkers for osteoarthritis. Sci. Rep. 2020, 10, 11328. [Google Scholar] [CrossRef] [PubMed]
- Arden, N.; Richette, P.; Cooper, C.; Bruyère, O.; Abadie, E.; Branco, J.; et al. Can we identify patients with high risk of osteoarthritis progression who will respond to treatment? A focus on biomarkers and frailty. Drugs Aging 2015, 32, 525–535. [Google Scholar] [CrossRef]
- Kraus, V.B.; McDaniel, G.; Huebner, J.L.; Stabler, T.V.; Pieper, C.F.; Shipes, S.W.; et al. Direct in vivo evidence of activated macrophages in human osteoarthritis. Osteoarthritis Cartilage 2016, 24, 1613–1621. [Google Scholar] [CrossRef] [PubMed]
- Yao, Q.; Wu, X.; Tao, C.; Gong, W.; Chen, M.; Qu, M.; et al. Osteoarthritis: Pathogenic signaling pathways and therapeutic targets. Signal Transduct. Target. Ther. 2023, 8, 56. [Google Scholar] [CrossRef] [PubMed]
- Teeple, E.; Elsaid, K.A.; Jay, G.D.; Zhang, L.; Badger, G.J.; Akelman, M.; et al. Effects of supplemental intra-articular lubricin and hyaluronic acid on the progression of posttraumatic arthritis in the anterior cruciate ligament-deficient rat knee. Am. J. Sports Med. 2011, 39, 164–172. [Google Scholar] [CrossRef] [PubMed]
- Watkins, A.R.; Reesink, H.L. Lubricin in experimental and naturally occurring osteoarthritis: A systematic review. Osteoarthritis Cartilage 2020, 28, 1303–1315. [Google Scholar] [CrossRef] [PubMed]
- Teerlink, C.C.; Jurynec, M.J.; Hernandez, R.; Stevens, J.; Hughes, D.C.; Brunker, C.P.; et al. A role for the MEGF6 gene in predisposition to osteoporosis. Ann. Hum. Genet. 2021, 85, 58–72. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Tan, L.J.; Wang, P.; Chen, X.D.; Zhu, L.H.; Zeng, Q.; et al. Functional relevance for associations between osteoporosis and genetic variants. PLoS ONE 2017, 12, e0169926. [Google Scholar] [CrossRef]
- Glyn-Jones, S.; Palmer, A.J.R.; Agricola, R.; Price, A.J.; Vincent, T.L.; Weinans, H.; et al. Osteoarthritis. Lancet 2015, 386, 376–387. [Google Scholar] [CrossRef]
- Abramson, S.B.; Attur, M. Developments in the scientific understanding of osteoarthritis. Osteoarthritis Cartilage 2009, 17, 1610–1615. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




