Submitted:
24 July 2024
Posted:
26 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Population
2.3. Randomization and Study Treatment
2.4. Study Treatment
2.5. Study Procedures
2.6. Study Outcomes
2.7. Statistical Considerations
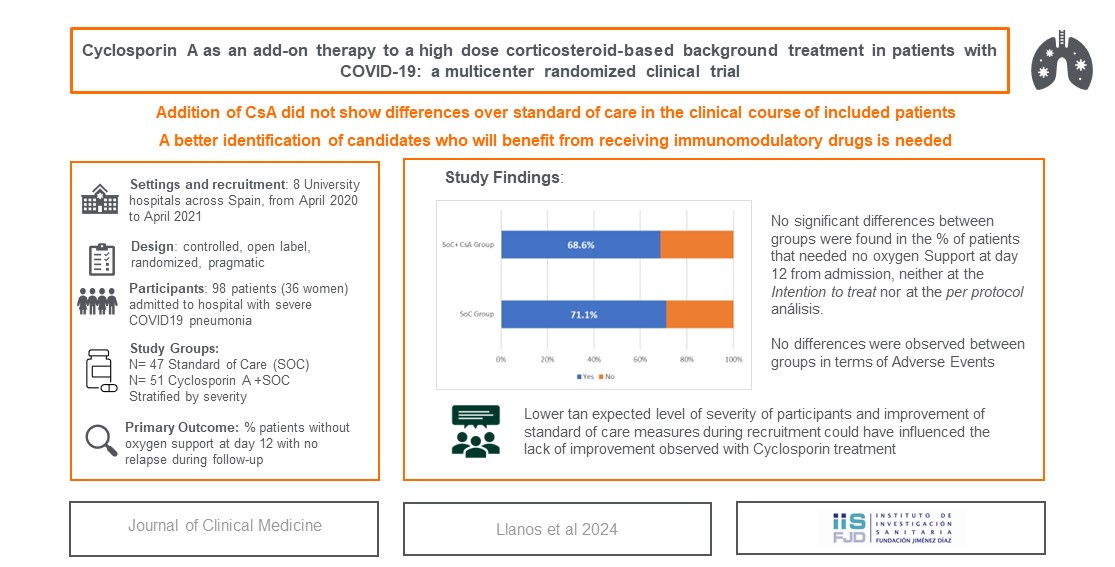
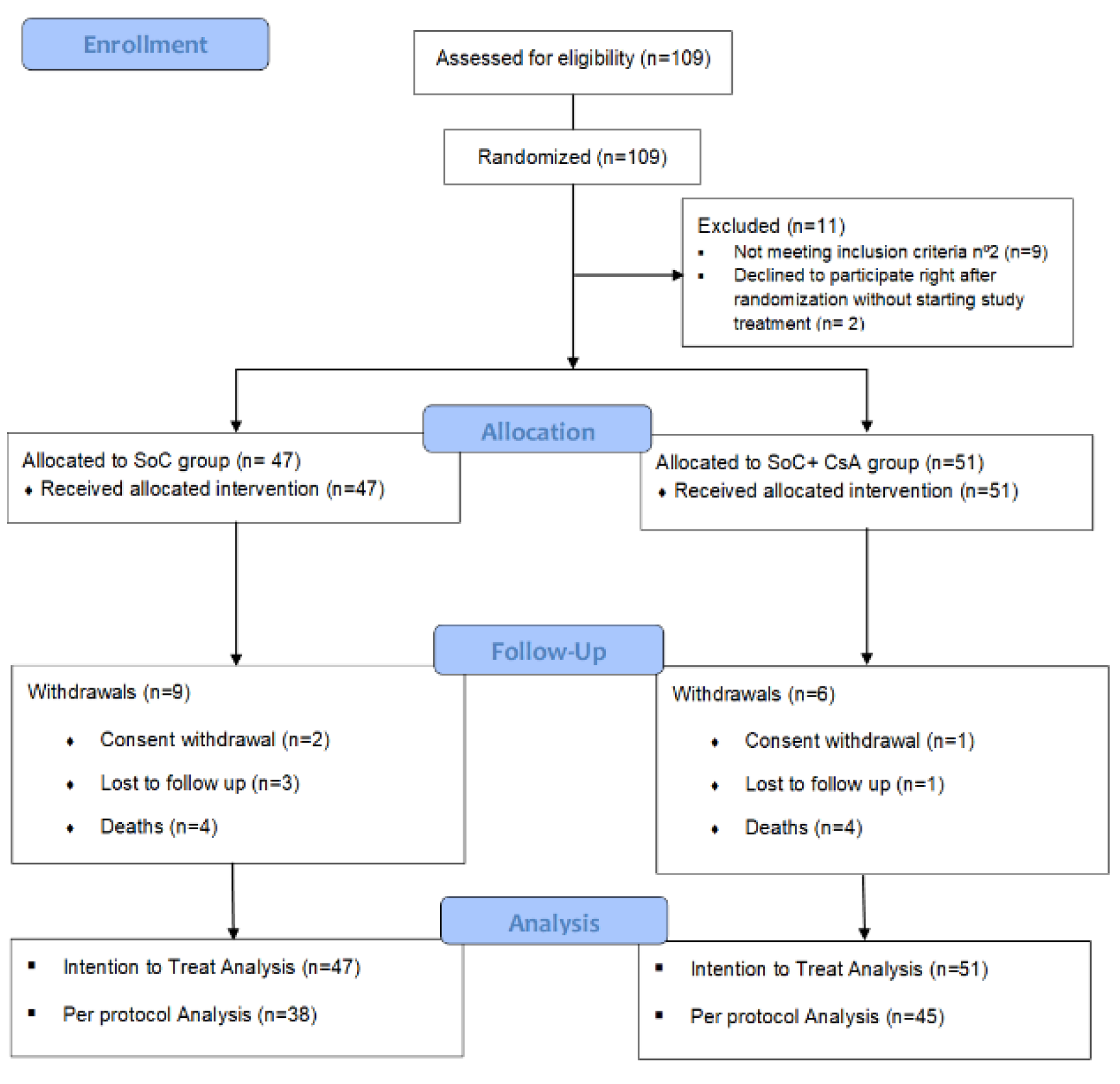
3. Results
3.1. Setting and Study Population
3.2. Study Intervention
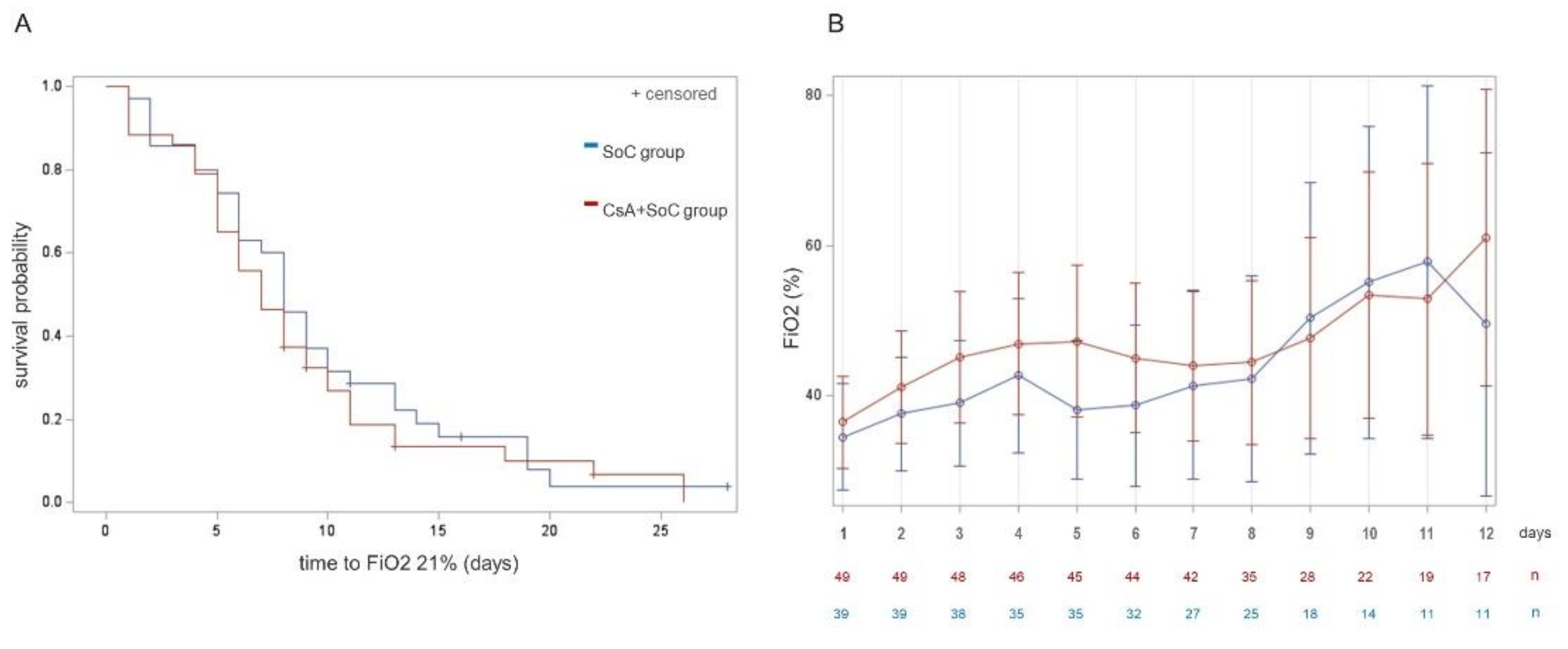
3.3. Primary Outcome
3.3. Secondary Outcomes
3.4. Safety Measures
3.5. Process Related Analytes
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Z. Xu, L. Shi, Y. Wang, J. Zhang, L. Huang, C .Zhang, et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med., 8 (2020), pp. 420-422. [CrossRef]
- C. Qin, L. Zhou, Z. Hu, S. Zhang, S. Yang, Y. Tao, et al. Dysregulation of Immune Response in Patients With Coronavirus 2019 (COVID-19) in Wuhan, China. Clin Infect Dis, 71 (2020), pp. 762-768. [CrossRef]
- P.S. Kim, S.W. Read, A.S. Fauci. Therapy for Early COVID-19: A Critical Need. JAMA, 324 (2020), pp. 2149–2150. [CrossRef]
- M. Merad, J.C. Martin. Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages. Nat Rev Immunol, 20 (2020), pp. 355-362. [CrossRef]
- O. Sanchez-Pernaute , F.I. Romero-Bueno, A. Selva-O'Callaghan. Why choose cyclosporin A as first-line therapy in COVID-19 pneumonia. Reumatol Clin (Engl Ed)., 17 (2021), pp. 556-557. [CrossRef]
- T. Koshiba. Mitochondrial-mediated antiviral immunity. Biochim Biophys Acta, 1833 (2013), pp. 225–232. [CrossRef]
- S.J. Kim, G.H. Syed, M. Khan, W.W. Chiu, M.A. Sohail, R.G. Gish, A. Siddiqui. Hepatitis C virus triggers mitochondrial fission and attenuates apoptosis to promote viral persistence. Proc Natl Acad Sci USA., 111 (2014), pp. 6413-6418. [CrossRef]
- Y. Lei, C.B. Moore, R.M. Liesman, B.P. O'Connor, D.T. Bergstralh, Z.J. Chen, et al. MAVS-mediated apoptosis and its inhibition by viral proteins. PLoS One, 4 (2009), pp. e5466. [CrossRef]
- K.K. Singh, G. Chaubey, J.Y. Chen, P. Suravajhala. Decoding SARS-CoV-2 hijacking of host mitochondria in COVID-19 pathogenesis. Am J Physiol Cell Physiol, 319 (2020), pp. C258-C267. [CrossRef]
- C. Bhowal, S. Ghosh, D. Ghatak, R. De. Pathophysiological involvement of host mitochondria in SARS-CoV-2 infection that causes COVID-19: a comprehensive evidential insight. Mol Cell Biochem, 478 (2023), pp.1325-1343. [CrossRef]
- A.P. Halestrap, S.J. Clarke, S.A. Javadov. Mitochondrial permeability transition pore opening during myocardial reperfusion--a target for cardioprotection. Cardiovasc Res, 61 (2004), pp.372-385. [CrossRef]
- M. Montero, C.D. Lobatón, S. Gutierrez-Fernández, A. Moreno, J. Alvarez. Calcineurin-independent inhibition of mitochondrial Ca2+ uptake by cyclosporin A. Br J Pharmacol, 141 (2004), pp.263-268. [CrossRef]
- Y. Tanaka, Y. Sato, T. Sasaki. Suppression of coronavirus replication by cyclophilin inhibitors. Viruses, 5 (2013), pp. 1250-1260. [CrossRef]
- S. Tavakolpour, T. Rakhshandehroo, E.X. Wei, M. Rashidian. Lymphopenia during the COVID-19 infection: What it shows and what can be learned. Immunol Lett, 225 (2020), pp. 31-32. [CrossRef]
- G.P. Rizzardi, A. Harari, B. Capiluppi, G. Tambussi, K. Ellefsen, D. Ciuffreda, et al. Treatment of primary HIV-1 infection with cyclosporin A coupled with highly active antiretroviral therapy. J Clin Invest, 109 (2002), pp. 681-688. [CrossRef]
- B.R. O'Driscoll, L.S. Howard, J. Earis, V Mak; British Thoracic Society Emergency Oxygen Guideline Group; BTS Emergency Oxygen Guideline Development Group. BTS guideline for oxygen use in adults in healthcare and emergency settings. Thorax, 72(Suppl1) (2017), pp. ii1-ii90. [CrossRef]
- Jones BE, Jones J, Bewick T, Lim WS, Aronsky D, Brown SM, Boersma WG, van der Eerden MM, Dean NC. CURB-65 pneumonia severity assessment adapted for electronic decision support. Chest. 2011 Jul;140(1):156-163. [CrossRef]
- P. Horby, W.S. Lim, J.R. Emberson, M. Mafham, J.L. Bell, L. Linsell, et al. Dexamethasone in hospitalized patients with covid-19. N Engl J Med, 384 (2021), pp. 693–704. [CrossRef]
- L. Ghosn, R. Assi, T. Evrenoglou, B.S. Buckley, N. Henschke, K. Probyn, et al. Interleukin-6 blocking agents for treating COVID-19: a living systematic review. Cochrane Database Syst Rev, 6 (2023), pp. CD013881. [CrossRef]
- M.K. Han, M. Antila, J.H. Ficker, I. Gordeev, A. Guerreros, A.L. Bernus, et al. Ruxolitinib in addition to standard of care for the treatment of patients admitted to hospital with COVID-19 (RUXCOVID): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Rheumatol, 4 (2022), pp. e351-e361. [CrossRef]
- I. Kralj-Hans, K. Li, A. Wesek, A. Lamorgese, F. Omar, K. Ranasinghe, et al. Leflunomide treatment for patients hospitalised with COVID-19: DEFEAT-COVID randomised controlled trial BMJ Open, 13 (2023), pp. e068179. [CrossRef]
- P. Fanlo, B.D.C. Gracia-Tello, E. Fonseca Aizpuru, J. Álvarez-Troncoso, A. Gonzalez, S. Prieto-González, et al. Efficacy and safety of anakinra plus standard of care for patients with severe COVID-19: A Randomized Phase 2/3 Clinical Trial. JAMA Netw Open, 6 (2023), pp. e237243. [CrossRef]
- E. Pairo-Castineira, K. Rawlik, A.D. Bretherick, T. Qi, Y. Wu, I. Nassiri, et al. GWAS and meta-analysis identifies 49 genetic variants underlying critical COVID-19. Nature, 617 (2023), pp. 764-768. [CrossRef]
- G. Chen, D. Wu, W Guo, Y. Cao, D. Huang, H. Wang, et al. Clinical and immunological features of severe and moderate coronavirus disease 2019. J Clin Invest, 130 (2020), pp. 2620-2629. [CrossRef]
| CsA+SoC group | SoC group | |
|---|---|---|
| Total N (women) | 51 | 47 |
| Female sex, n (%) | 22 (43.1) | 14 (29.8) |
| Age, mean ± SD [95% CI], median | 60.2 ± 11.7 [57.0, 63.5] 60.5 | 62.9 ± 13.0 [59.1, 66.7] 62.0 |
| Age ≥ 65, n (%) | 21 (44.7) | 21 (41.2) |
| Ethnicity, n (%) (caucasian, hispanic, others) | ||
| caucasian | 26 (51) | 32 (68) |
| hispanic | 15 (29.4) | 11 (23.4) |
| others | 10 (19.6) | 4 (8.6) |
| BMI, mean ± SD [CI95], median | 29.2 ± 5.7 [27.5, 30.9] 28.6 | 28.7 ± 4.1 [27.4, 30.0] 28.5 |
| BMI ≥ 30, n (%) | 16 (37.2) | 14 (34.1) |
| Smoking habit, n (%) | (3, 13, 35) | (2, 19, 26) |
| Active | 3 (5.9) | 2 (4.3) |
| Past | 13 (25.5) | 19 (40.4) |
| Never smoker | 35 (68.6) | 26 (55.3) |
| Comorbidities, n (%) a | 24 (47.1) | 29 (61.7) |
| Hypertension, n (%) | 19 (37.3) | 23 (48.9) |
| Diabetes, n (%) | 10 (19.6) | 5 (10.6) |
| Ischemic heart disease, n (%) | 1 (2.0) | 5 (10.6) |
| Active cancer, n (%) | 3 (5.9) | 1 (2.1) |
| Exposure to immunosuppresants, n (%) | 1 (2.0) | 3 (6.4) |
| COPD, n (%) | 3 (5.9) | 4 (8.5) |
| History of thromboembolic disease, n (%) | 5 (9.8) | 4 (8.5) |
| Time (days) from first symptom, mean ± SD [CI95], median | 8.8 ± 6.9 [6.8, 10.7] 8.0 | 8.5 ± 6.4 [6.7, 10.4] 8.0 |
| Days of dyspnea, mean ± SD [CI95], median | 4.7 ± 5.2 [2.8, 6.7] 3.0 | 4.5 ± 3.9 [3.0, 6.0] 3.0 |
| Severity according to CURB-65, mean ± SD [CI95], median | 0.8 ± 0.9 (0.6-1.1) 1.0 | ± 0.8 (0.6-1.1) 1.0 |
| a Number of participants with at least one comorbidity; BMI: Body Mass Index; COPD: chronic obstructive pulmonary disease; ITT: intention to treat; CsA: Cyclosporine; SoC: standard of care; SD: standard deviation. | ||
| % patients without oxygen support at day 12 | All | CsA+SoC group | SoC group | Diference (95%CI) |
|---|---|---|---|---|
| ITT population, N | 96* | 51 | 45 | |
| n (%) | 67 (69.8) | 35 (68.6) | (71.1) | -2.5 (-20.9, 15.9, NS) |
| PP population, N | 83 | 45 | 38 | |
| n (%) | 63 (75.9) | 34 (75.6) | 29 (76.3) | -0.7 (-19.14, 17.74, NS) |
| * 2 patients withdrew consent before reaching day 12; ICU: Intensive Care Unit; : ITT: intention to treat; CsA: Cyclosporine; SoC: standard of care; NS: not statistically significant. | ||||
| All | CsA+SoC group | SoC group | p | |
|---|---|---|---|---|
| Total, N | 98 | 51 | 47 | |
| All Deaths, N (%) | 8 (8.2) | 4 (7.8) | 4 (8.5) | ns |
| Deaths Y1+Y2, n (%) | 5 (5.6) | 2 (4.3) | 3 (7.0) | ns |
| Deaths ≥ 65y, n (%) | 7 (16.6) | 3 (14.3) | 4 (19.0) | ns |
| Discharges, n (%) | 83 (84.7) | 43 (84.3) | 40 (85.1) | ns |
| LOS, mean ± SD [CI 95%], median | 9.9 ± 4.8 [8.8, 10.9] 9.0 | 10.3 ± 4.9 [8.8, 11.8] 9.0 | 9.4 ± 4.8 [7.9, 10.9] 9.0 | ns |
| LOS Y1+Y2, mean ± SD [CI 95%], median | 10.1 ± 4.9 [8.6, 11.6] 8.0 | 8.9 ± 4.3 [7.4, 10.5] 8.0 | ns | |
| LOS < 65y, mean ± SD [CI 95%], median | 10.4 ± 5.5 [8.2, 12.6] 8.5 | 8.7 ± 3.3 [7.3, 10.0] 8.5 | ns | |
| LOS > 65y, mean ± SD [CI 95%], median | 10.1 ± 4. [8.0, 12.1] 9.0 | 10.8 ± 6.7 [6.9, 14.7] 9.5 | ns | |
| Patients achieving FiO2 21%, n (%) | 71 (72.4) | 39 (76.5) | 32 (68.1) | ns |
| LOS until FiO2 21%, mean ± SD [CI 95%], median | 7.4 ± 5.4 [5.7-9.2] | 7.9 ± 6.5 [6.1, 9.8] | ns | |
| SBP at day 16*, mean ± SD [CI 95%], median | 2.6 ± 22.5 [-18.2, 23.3] 6.0 | -25.6 ± 15.2 [-44.5, -6.7] -32.0 | 0.036 | |
| LOS: length of stay; SBP: Systolic Blood Pressure; ITT: intention to treat; CsA: Cyclosporine; SoC: standard of care; SD: standard deviation; * difference from levels at ER; NS: not statistically significant. | ||||
| Number of Participants with AE | Total | CsA+SoC group | SoC group | Comparison |
|---|---|---|---|---|
| All, N | 98 | 51 | 47 | |
| Related, n (%) | 34 (34,7) | 25 (49) | 9 (19,1) | p 0.003 |
| SAE, n (%) | 7 (7,1) | 3 (5,9) | 4 (8,5) | p>0.05 |
| TOTAL, n (%) | 54 (55,1) | 34 (66,7) | 20 (42,6) | p 0.025 |
| AE: adverse event; SAE: serious adverse event; SoC: standard of care; CsA: cyclosporine | ||||
| Positive/tested (%) | |||||
|---|---|---|---|---|---|
| Isotype | day 1 | day 8 | day 15 | day 22 | |
| CsA+SoC group | IgG | 15/29 (52%) | 25/31 (81%) | 4/5 (80%) | 0 |
| IgM | 17/29 (59%) | 24/31 (77%) | 4/5 (80%) | 0 | |
| SoC group | IgG | 10/22 (45%) | 22/23 (96%) | 6/6 (100%) | 1/1 (100%) |
| IgM | 10/22 (45%) | 21/23 (91%) | 4/6 (67%) | 1/1 (100%) | |
| SoC: standard of care; CsA: cyclosporine | |||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




