Submitted:
19 July 2024
Posted:
22 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Patients and Surgical Specimens
2.2. Tissue Microarrays and Immunohistochemical Analysis
2.3. Statistical Analysis
3. Results
3.1. Study Population
3.2. Association of CDX2 Staining with Clinicopathological Features
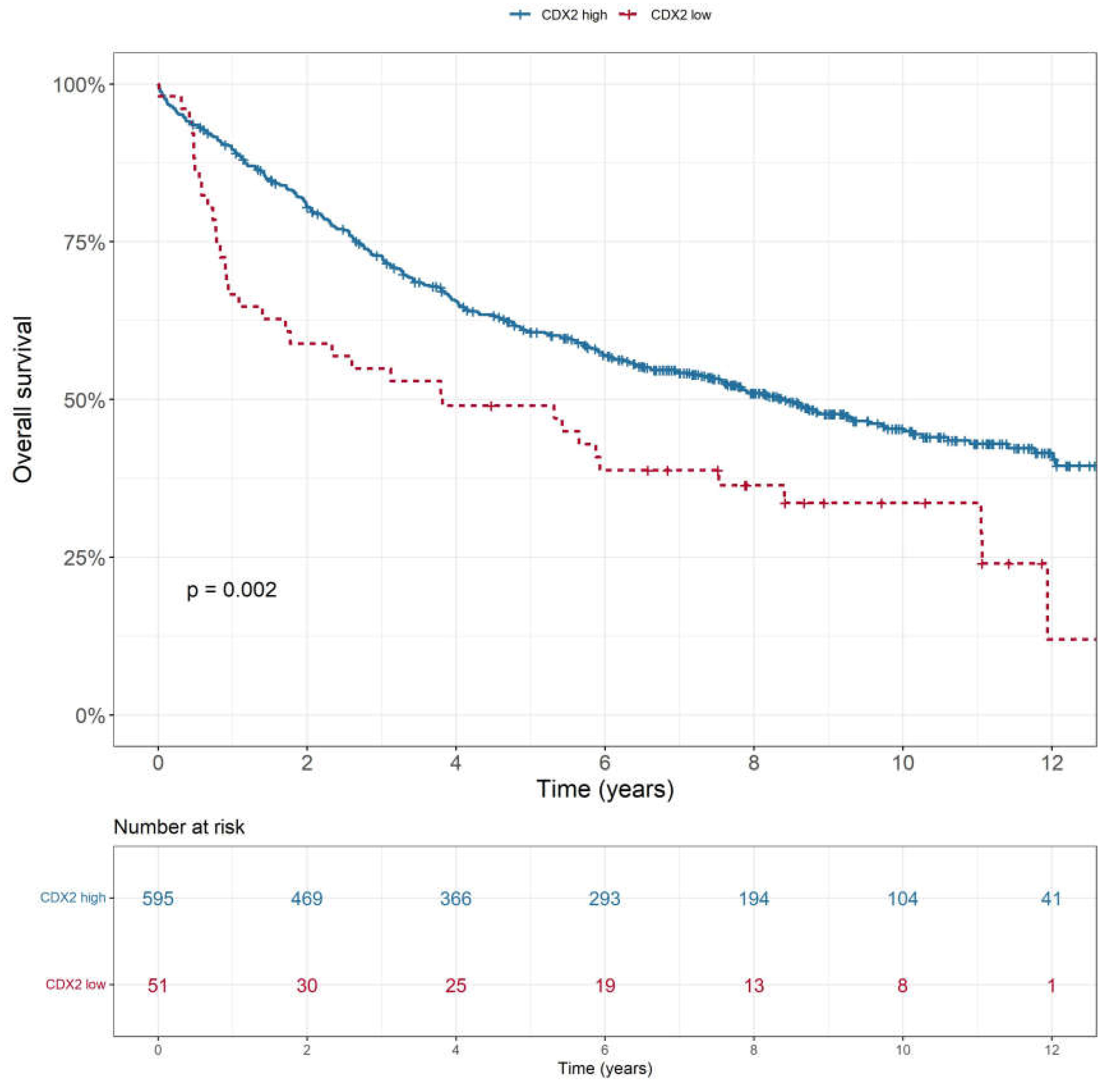
3.3. CDX2 Low Expression Correlates with Poorer Prognosis.
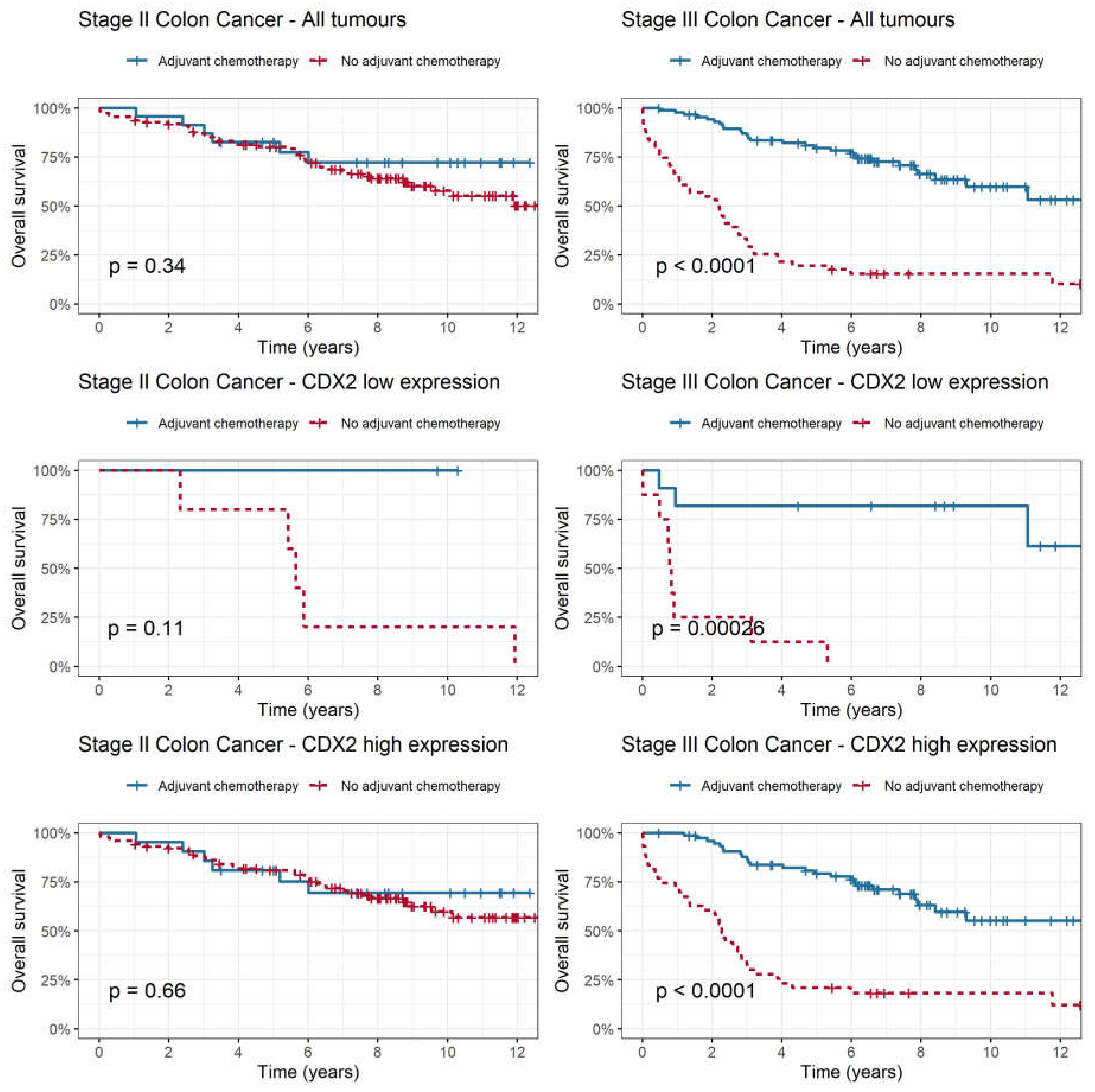
3.4. CDX2 as a Predictive Biomarker for Adjuvant Chemotherapy in Stage II and III Colon Cancer
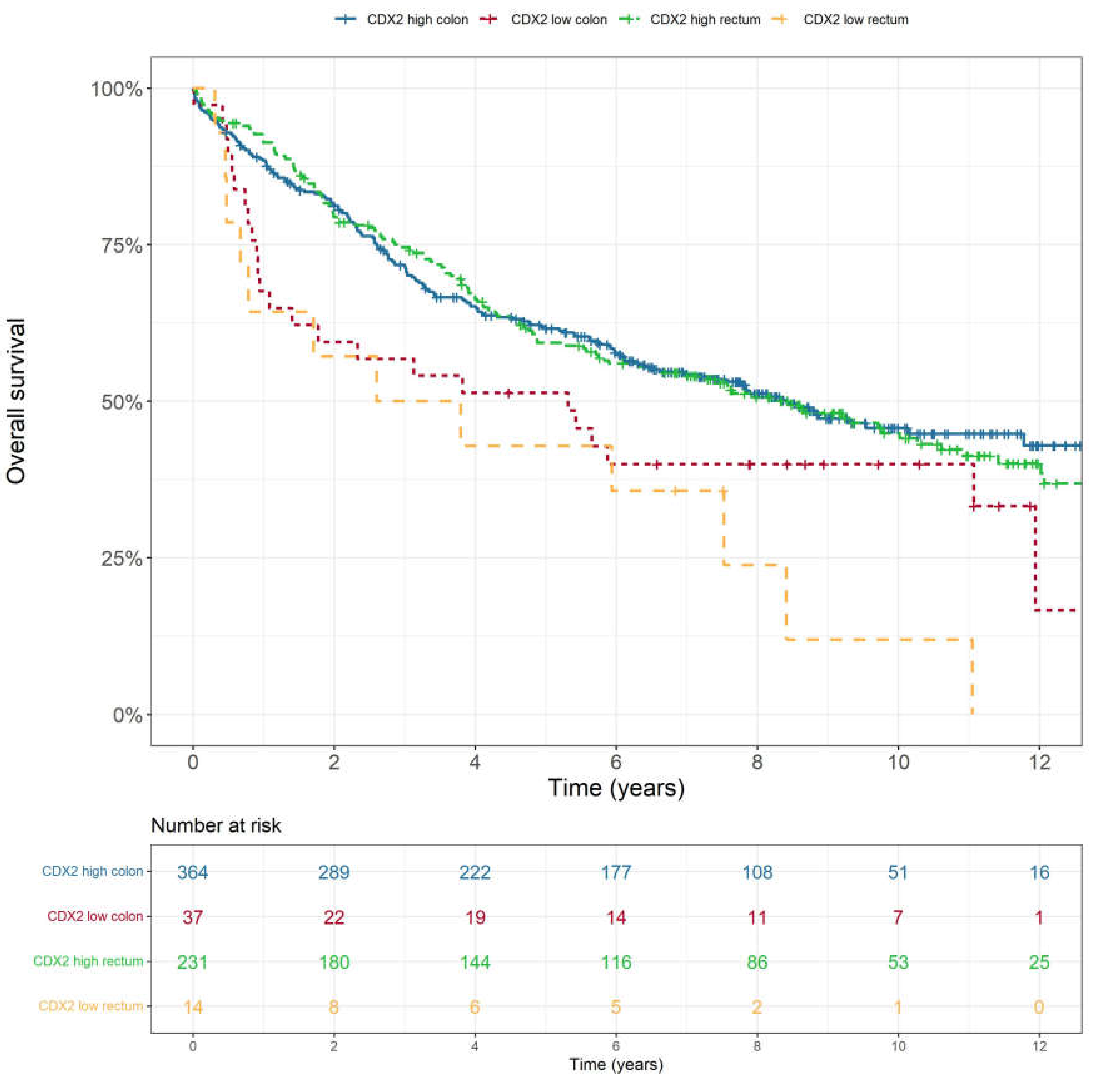
3.5. CDX2 as a Predictive Biomarker in Rectal Cancer
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Welfare AIoHa. Cancer data in Australia 2022 [Available from: https://www.aihw.gov.au/reports/cancer/cancer-data-in-australia].
- Indicators NCC. Cancer Incidence 8 Aug 2022 [Available from: https://ncci.canceraustralia.gov.au/diagnosis/cancer-incidence/cancer-incidence].
- Cancer.Net A. Colorectal Cancer: Statistics 2023 [Available from: https://www.cancer.net/cancer-types/colorectal-cancer/statistics].
- Argilés, G.; Tabernero, J.; Labianca, R.; Hochhauser, D.; Salazar, R.; Iveson, T.; Laurent-Puig, P.; Quirke, P.; Yoshino, T.; Taieb, J.; et al. Localised colon cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2020, 31, 1291–1305. [CrossRef]
- Rebuzzi, S.E.; Pesola, G.; Martelli, V.; Sobrero, A.F. Adjuvant Chemotherapy for Stage II Colon Cancer. Cancers 2020, 12, 2584. [CrossRef]
- Koncina, E.; Haan, S.; Rauh, S.; Letellier, E. Prognostic and Predictive Molecular Biomarkers for Colorectal Cancer: Updates and Challenges. Cancers 2020, 12, 319. [CrossRef]
- Alves Martins BA, de Bulhões GF, Cavalcanti IN, Martins MM, de Oliveira PG, Martins AMA. Biomarkers in Colorectal Cancer: The Role of Translational Proteomics Research. Frontiers in Oncology. 2019;9.
- Asgari-Karchekani, S.; Karimian, M.; Mazoochi, T.; Taheri, M.A.; Khamehchian, T. CDX2 Protein Expression in Colorectal Cancer and ItsCorrelation with Clinical and Pathological Characteristics, Prognosis, and Survival Rate of Patients. J. Gastrointest. Cancer 2019, 51, 844–849. [CrossRef]
- A Moskaluk, C.; Zhang, H.; Powell, S.M.; A Cerilli, L.; Hampton, G.M.; Frierson, H.F. Cdx2 Protein Expression in Normal and Malignant Human Tissues: An Immunohistochemical Survey Using Tissue Microarrays. Mod. Pathol. 2003, 16, 913–919. [CrossRef]
- A Moskaluk, C.; Zhang, H.; Powell, S.M.; A Cerilli, L.; Hampton, G.M.; Frierson, H.F. Cdx2 Protein Expression in Normal and Malignant Human Tissues: An Immunohistochemical Survey Using Tissue Microarrays. Mod. Pathol. 2003, 16, 913–919. [CrossRef]
- Werling RW, Yaziji H, Bacchi CE, Gown AM. CDX2, a highly sensitive and specific marker of adenocarcinomas of intestinal origin: an immunohistochemical survey of 476 primary and metastatic carcinomas. Am J Surg Pathol. 2003;27(3):303-10.
- Tomasello, G.; Barni, S.; Turati, L.; Ghidini, M.; Pezzica, E.; Passalacqua, R.; Petrelli, F. Association of CDX2 Expression With Survival in Early Colorectal Cancer: A Systematic Review and Meta-analysis. Clin. Color. Cancer 2018, 17, 97–103. [CrossRef]
- Baba, Y.; Nosho, K.; Shima, K.; Freed, E.; Irahara, N.; Philips, J.; Meyerhardt, J.A.; Hornick, J.L.; Shivdasani, R.A.; Fuchs, C.S.; et al. Relationship of CDX2 Loss with Molecular Features and Prognosis in Colorectal Cancer. Clin. Cancer Res. 2009, 15, 4665–4673. [CrossRef]
- Zhang, B.Y.; Jones, J.C.; Briggler, A.M.; Hubbard, J.M.; Kipp, B.R.; Sargent, D.J.; Dixon, J.G.; Grothey, A. Lack of Caudal-Type Homeobox Transcription Factor 2 Expression as a Prognostic Biomarker in Metastatic Colorectal Cancer. Clin. Color. Cancer 2016, 16, 124–128. [CrossRef]
- Dalerba, P.; Sahoo, D.; Paik, S.; Guo, X.; Yothers, G.; Song, N.; Wilcox-Fogel, N.; Forgó, E.; Rajendran, P.S.; Miranda, S.P.; et al. CDX2 as a Prognostic Biomarker in Stage II and Stage III Colon Cancer. New Engl. J. Med. 2016, 374, 211–222. [CrossRef]
- Ballman KV. Biomarker: Predictive or Prognostic? Journal of Clinical Oncology. 2015;33(33):3968-71.
- Saad RS, Ghorab Z, Khalifa MA, Xu M. CDX2 as a marker for intestinal differentiation: Its utility and limitations. World J Gastrointest Surg. 2011;3(11):159-66.
- Delgado A, Guddati AK. Clinical endpoints in oncology - a primer. Am J Cancer Res. 2021;11(4):1121-31.
- Statisticsc ABo. Sydney South West 2021 Census All persons QuickStats 2021 [Available from: https://abs.gov.au/census/find-census-data/quickstats/2021/127].
- Bae, J.M.; Lee, T.H.; Cho, N.-Y.; Kim, T.-Y.; Kang, G.H. Loss of CDX2 expression is associated with poor prognosis in colorectal cancer patients. World J. Gastroenterol. 2015, 21, 1457–67. [CrossRef]
- André T, de Gramont A, Vernerey D, Chibaudel B, Bonnetain F, Tijeras-Raballand A, Scriva A, Hickish T, Tabernero J, Van Laethem JL, et al. Adjuvant Fluorouracil, Leucovorin, and Oxaliplatin in Stage II to III Colon Cancer: Updated 10-Year Survival and Outcomes According to BRAF Mutation and Mismatch Repair Status of the MOSAIC Study. J Clin Oncol. 2015;33(35):4176-87.
- Varghese, A. Chemotherapy for Stage II Colon Cancer. Clin. Colon Rectal Surg. 2015, 28, 256–261. [CrossRef]
- Bruun, J.; Sveen, A.; Barros, R.; Eide, P.W.; Eilertsen, I.; Kolberg, M.; Pellinen, T.; David, L.; Svindland, A.; Kallioniemi, O.; et al. Prognostic, predictive, and pharmacogenomic assessments of CDX2 refine stratification of colorectal cancer. Mol. Oncol. 2018, 12, 1639–1655. [CrossRef]
- Liu, S.; Jiang, T.; Xiao, L.; Yang, S.; Liu, Q.; Gao, Y.; Chen, G.; Xiao, W. Total Neoadjuvant Therapy (TNT) versus Standard Neoadjuvant Chemoradiotherapy for Locally Advanced Rectal Cancer: A Systematic Review and Meta-Analysis. Oncol. 2021, 26, e1555–e1566. [CrossRef]
- Valenzuela, G.; Canepa, J.; Simonetti, C.; de Zaldívar, L.S.; Marcelain, K.; González-Montero, J. Consensus molecular subtypes of colorectal cancer in clinical practice: A translational approach. World J. Clin. Oncol. 2021, 12, 1000–1008. [CrossRef]
- Pilati, C.; Taieb, J.; Balogoun, R.; Marisa, L.; de Reyniès, A.; Laurent-Puig, P. CDX2 prognostic value in stage II/III resected colon cancer is related to CMS classification. Ann. Oncol. 2017, 28, 1032–1035. [CrossRef]
- Li Y, Yao Q, Zhang L, Mo S, Cai S, Huang D, Peng J. IHC-Based Consensus Molecular Subtypes as A Prognostic and Predictive Biomarker for Adjuvant Chemotherapy in Patients with Stage II Colorectal Cancer. The Oncologist. 2020.
- Xu, W.; Zhu, Y.; Shen, W.; Ding, W.; Wu, T.; Guo, Y.; Chen, X.; Zhou, M.; Chen, Y.; Cui, L.; et al. Combination of CDX2 expression and T stage improves prognostic prediction of colorectal cancer. J. Int. Med Res. 2019, 47, 1829–1842. [CrossRef]
- Hestetun, K.E.; Aasebø, K.; Rosenlund, N.B.; Müller, Y.; Dahl, O.; Myklebust, M.P. Mismatch repair phenotype determines the implications of tumor grade and CDX2 expression in stage II–III colon cancer. Mod. Pathol. 2020, 34, 161–170. [CrossRef]
- Hansen, T.F.; Kjær-Frifeldt, S.; Eriksen, A.C.; Lindebjerg, J.; Jensen, L.H.; Sørensen, F.B.; Jakobsen, A. Prognostic impact of CDX2 in stage II colon cancer: results from two nationwide cohorts. Br. J. Cancer 2018, 119, 1367–1373. [CrossRef]
- Loughrey MB, Webster F, Arends MJ, Brown I, Burgart LJ, Cunningham C, Flejou JF, Kakar S, Kirsch R, Kojima M, et al. Dataset for Pathology Reporting of Colorectal Cancer: Recommendations From the International Collaboration on Cancer Reporting (ICCR). Ann Surg. 2022;275(3):e549-e61.
- Singh J, Rajesh N, Dubashi B, Maroju NK, Ganesan P, Matta KK, Charles I, Kayal S. Pattern of expression of CDX2 in colorectal cancer and its role in prognosis. Journal of Cancer Research and Therapeutics. 2022;18(Suppl 2):S420-S7.
- Ben-Aharon, I.; Goshen-Lago, T.; Sternschuss, M.; Morgenstern, S.; Geva, R.; Beny, A.; Dror, Y.; Steiner, M.; Hubert, A.; Idelevich, E.; et al. Sidedness Matters: Surrogate Biomarkers Prognosticate Colorectal Cancer upon Anatomic Location. Oncol. 2019, 24, e696–e701. [CrossRef]
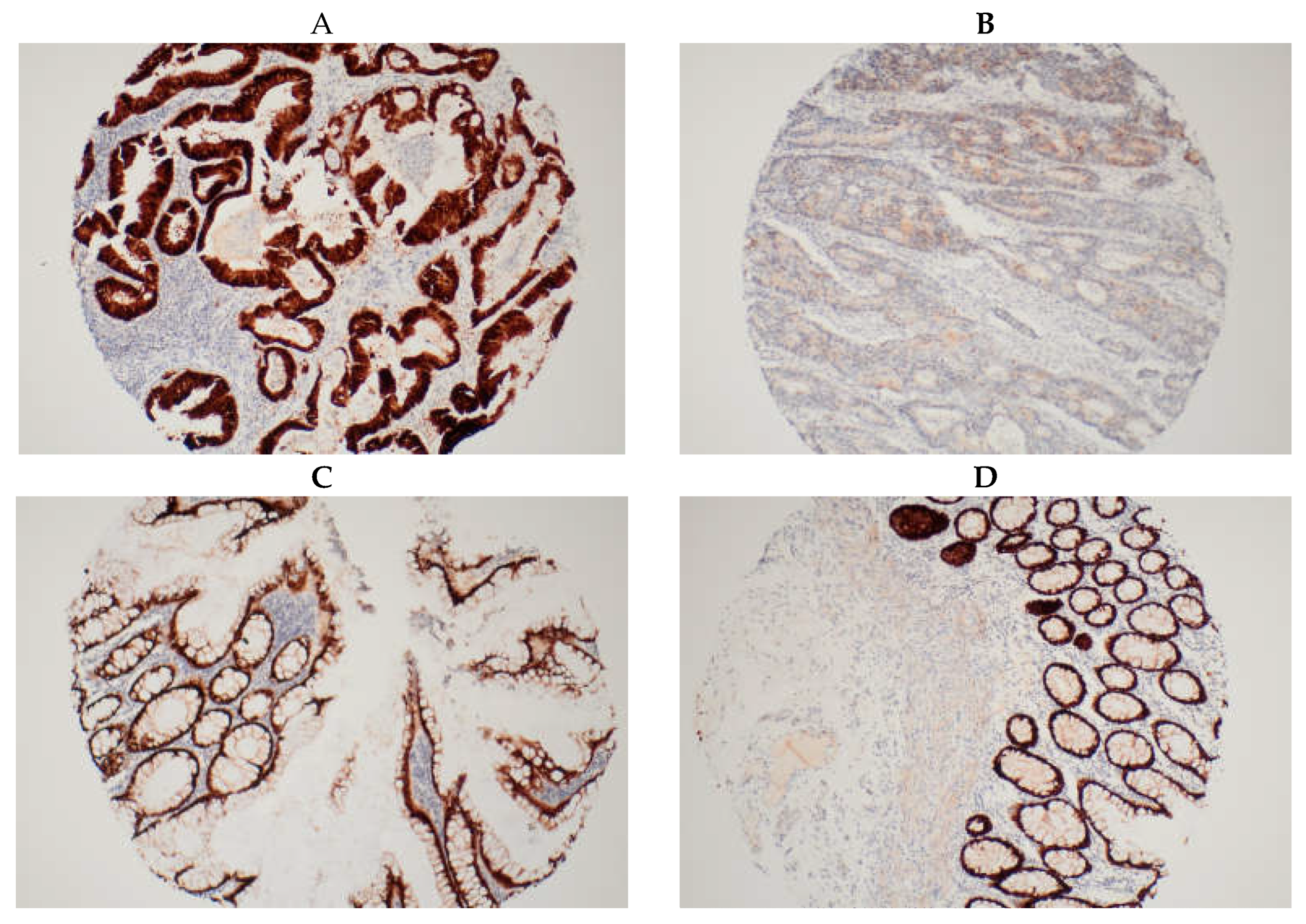
| Percentage of Deeply Stained Nuclei (%) | Score |
|---|---|
| Less than 5 | 1 |
| 5 – 24 | 2 |
| 25 – 49 | 3 |
| 50 – 74 | 4 |
| 75 – 100 | 5 |
| Variable | Class | Total (N = 668) | Percentage (%) | ||
|---|---|---|---|---|---|
| Sex | Male | 392 | 58.7 | ||
| Female | 276 | 41.3 | |||
| CDX2 expression | High (5) | 595 | 89.1 | ||
| Low (1-4) | 51 | 7.6 | |||
| Missing | 22 | 3.3 | |||
| Ethnicity | Caucasian | 577 | 86.4 | ||
| Asian | 76 | 11.4 | |||
| Pacific Islander | 8 | 1.2 | |||
| African | 3 | 0.4 | |||
| Aboriginal / Torres Strait Islander | 4 | 0.6 | |||
| Mean age at diagnosis | 68.6 (23 – 96) | ||||
| Age at diagnosis | ≤ 70 years | 340 | 50.9 | ||
| > 70 years | 328 | 49.1 | |||
| Cancer stage | Colon | Rectum | Total | ||
| I | 77 | 39 | 116 | 17.4 | |
| II | 133 | 74 | 207 | 31.0 | |
| III | 138 | 112 | 250 | 37.4 | |
| IV | 58 | 37 | 95 | 14.2 | |
| Pathology | Adenocarcinoma | 657 | 98.4 | ||
| Mucinous | 9 | 1.3 | |||
| Signet ring | 2 | 0.3 | |||
| Cancer side | Right | 217 | 32.5 | ||
| Left | 451 | 67.5 | |||
| Colon vs Rectum | Colon | 406 | 60.8 | ||
| Rectum | 262 | 39.2 | |||
| Tumour differentiation | Poor | 98 | 14.7 | ||
| Well / moderate | 570 | 85.3 | |||
| Lymphovascular or perineural invasion | Absent | 438 | 65.8 | ||
| Present | 228 | 34.2 | |||
| Colon | |||||
| Stage II adjuvant chemotherapy | No | 109 | 82.6 | ||
| Yes | 23 | 17.4 | |||
| Stage III adjuvant chemotherapy | None | 51 | 37.5 | ||
| Single agent | 36 | 26.5 | |||
| Doublet chemo | 49 | 36.0 | |||
| Stage IV palliative chemotherapy | No | 20 | 34.5 | ||
| Yes | 38 | 65.5 | |||
| Rectum | |||||
| Neoadjuvant therapy | No | 200 | 76.3 | ||
| Yes | 62 | 23.7 | |||
| Adjuvant chemotherapy | No | 123 | 54.2 | ||
| Yes | 104 | 45.8 | |||
| Variable | Class | CDX2 Expression | ||
|---|---|---|---|---|
| High, N = 595 (%) | Low, N = 51 (%) | P-value | ||
| Sex | Male | 356 (94.4) | 21 (5.6) | 0.012 |
| Female | 239 (88.8) | 30 (11.2) | ||
| Ethnicity | Caucasian | 511 (91.9) | 45 (8.1) | 0.373 |
| Asian | 71 (94.7) | 4 (5.3) | ||
| Pacific Islander | 7 (87.5) | 1 (12.5) | ||
| African | 3 (100.0) | 0 (0.0) | ||
| Aboriginal / Torres Strait Islander | 3 (75.0) | 1 (25.1) | ||
| Age at diagnosis | ≤ 70 years | 288 (92.3) | 24 (7.7) | 0.885 |
| > 70 years | 307 (91.9) | 27 (8.1) | ||
| Cancer stage | I | 110 (96.5) | 4 (3.5) | 0.015 |
| II | 189 (95.0) | 10 (5.0) | ||
| III | 216 (89.3) | 26 (10.7) | ||
| IV | 80 (87.9) | 11 (12.1) | ||
| Pathology | Adenocarcinoma | 587 (92.2) | 50 (7.8) | 0.525 |
| Mucinous | 7 (87.5) | 1 (12.5) | ||
| Signet ring | 1 (100.0) | 0 (0.0) | ||
| Cancer side | Right | 189 (88.3) | 25 (11.7) | 0.019 |
| Left | 406 (94.0) | 26 (6.0) | ||
| Colon vs rectum | Colon | 364 (90.8) | 37 (9.2) | 0.132 |
| Rectum | 231 (94.3) | 14 (5.7) | ||
| Tumour differentiation | Poor | 74 (77.1) | 22 (22.9) | <0.001 |
| Well / moderate | 521 (94.7) | 29 (5.3) | ||
| Lymphovascular or perineural invasion | Absent | 399 (94.5) | 23 (5.5) | 0.002 |
| Present | 194 (87.4) | 28 (12.6) | ||
| Colon stage II | Received adjuvant chemotherapy | 21 (91.3) | 2 (8.7) | 0.607 |
| Did not receive adjuvant chemotherapy | 102 (95.3) | 5 (4.7) | ||
| Colon stage III | Received adjuvant chemotherapy | 76 (87.4) | 11 (12.6) | 0.618 |
| Did not receive adjuvant chemotherapy | 43 (84.3) | 8 (15.7) | ||
| Subgroup | Univariate Analysis | Multivariable Analysis | ||
|---|---|---|---|---|
| Hazard ratio (95% CI) | P value | Hazard ratio (95% CI) | P value | |
| All patients (n = 668) | ||||
| CDX2-negative | 0.584 (0.413 – 0.825) | 0.002 | 0.602 (0.421 – 0.86) | 0.005 |
| Tumour stage | ||||
| Stage I vs stage IV | 0.117 (0.080 – 0.172) | <0.001 | 0.118 (0.08 – 0.173) | <0.001 |
| Stage II vs stage IV | 0.125 (0.091 – 0.172) | <0.001 | 0.123 (0.089 – 0.17) | <0.001 |
| Stage III vs stage IV | 0.182 (0.136 – 0.244) | <0.001 | 0.175 (0.13 – 0.235) | <0.001 |
| Male vs female sex | 0.947 (0.763 – 1.177) | 0.624 | 0.906 (0.726 – 1.131) | 0.384 |
| Right vs left | 1.119 (0.891 – 1.404) | 0.332 | 1.116 (0.841 – 1.482) | 0.447 |
| Colon vs rectum | 0.963 (0.775 – 1.198) | 0.736 | 0.937 (0.715 – 1.228) | 0.637 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




