Submitted:
18 July 2024
Posted:
19 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. The Evidence of N-arylpiperazine Derivatives in Carcinogenic Pathways
2.1. N-aryl Piperazine Derivatives in Prostate Cancer
2.2. N-aryl Piperazine Derivatives in Colorectal Cancer
2.3. N-aryl Piperazine Derivatives in Pancreatic Cancer
2.4. N-aryl Piperazine Derivatives in Breast Cancer
2.5. N-arylpiperazine Derivatives in Cervical Carcinoma
2.6. N-arylpiperazine Derivatives in Leukemia
2.7. N-arylpiperazine Derivatives in Melanoma
2.8. Other Examples of Arylpiperazines in Cancer
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ravilla, L.; Venkata Subba Naidu, N.; Nagarajan, K. An Efficient Scale up Process for Synthesis of N-Arylpiperazines. Tetrahedron Lett 2015, 56, 4541–4544. [Google Scholar] [CrossRef]
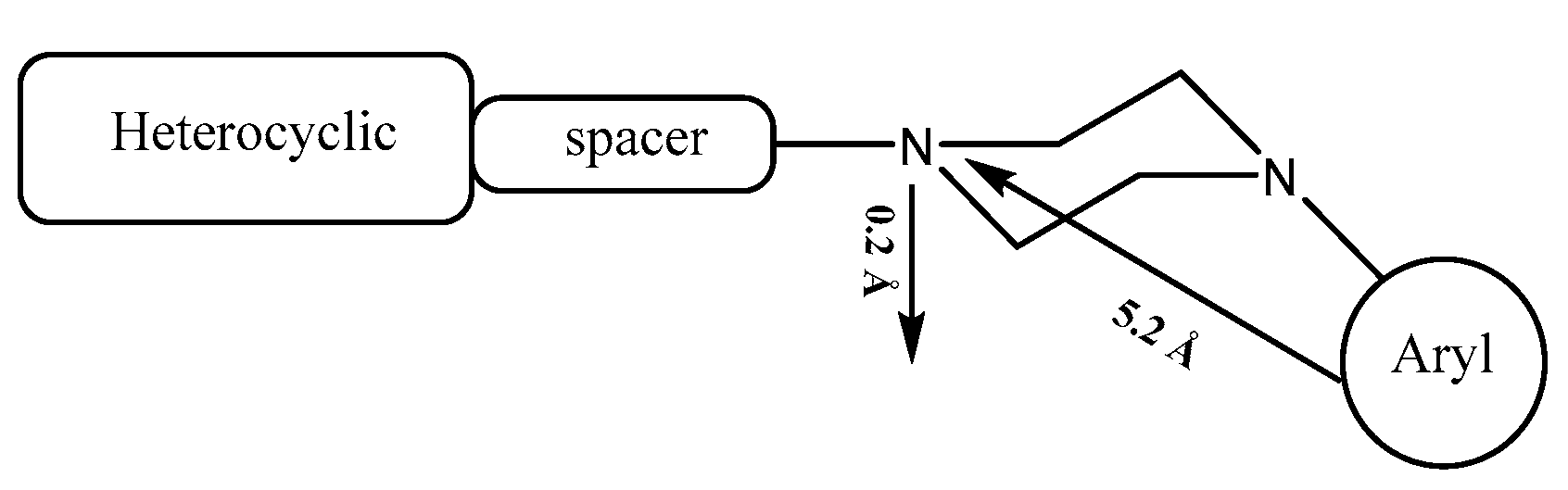
- Jaziri, E.; Louis, H.; Gharbi, C.; Lefebvre, F.; Kaminsky, W.; Agwamba, E.C.; Egemonye, T.C.; Unimuke, T.O.; Ikenyirimba, O.J.; Mathias, G.E.; Ben Nasr, C.; Khedhiri, L. Investigation of Crystal Structures, Spectral (FT-IR and NMR) Analysis, DFT, and Molecular Docking Studies of Novel Piperazine Derivatives as Antineurotic Drugs. J Mol Struct 2023, 1278, 134937. [Google Scholar] [CrossRef]
- Karolak-Wojciechowska, J.; Fruzinski, A.; Mokrosz, M.J. Structure and Conformational Analysis of New Arylpiperazines Containing N-Butyl Chain. Elsevier 2001, 542, 47–56. [Google Scholar] [CrossRef]
- Weber, K.C.; Salum, L.B.; Honorio, K.M.; Andricopulo, A.D.; da Silva, A.B.F. Pharmacophore-Based 3D QSAR Studies on a Series of High Affinity 5-HT1A Receptor Ligands. Eur J Med Chem 2010, 45, 1508–1514. [Google Scholar] [CrossRef] [PubMed]
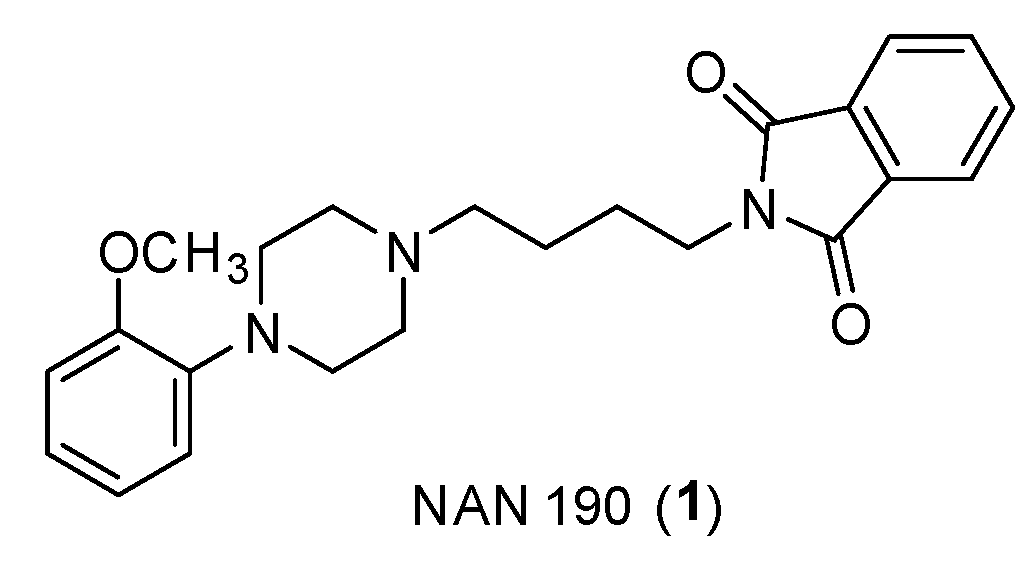
- Quaglia, W.; Cifani, C.; Del Bello, F.; Giannella, M.; Giorgioni, G.; Micioni di Bonaventura,m.v.; Piergentili, A. 4WD to Travel Inside the 5-HT1A Receptor World. In Serotonin - A Chemical Messenger Between All Types of Living Cells; InTech, 2017, pp. 67-108.
- Caliendo, G.; Santagada, V.; Perissutti, E.; Fiorino, F. Derivatives as 5HT 1A Receptor Ligands-Past and Present; Current Medicinal Chemistry, 2005, 12, 763-771.
- Fiorino, F.; Severino, B.; Magli, E.; Ciano, A.; Caliendo, G.; Santagada, V.; Frecentese, F.; Perissutti, E. 5-HT1A Receptor: An Old Target as a New Attractive Tool in Drug Discovery from Central Nervous System to Cancer. J Med Chem 2014, 57, 4407–4426. [Google Scholar] [CrossRef] [PubMed]
- Ikwu, F.A.; Shallangwa, G.A.; Mamza, P.A. Ligand Based Design, ADMET and Molecular Docking Studies of Arylpiperazine Derivatives as Potent Anti-Proliferate Agents Against LNCAP Prostate Cancer Cell Lines. Chemistry Africa 2021, 4, 71–84. [Google Scholar] [CrossRef]
- Kaur, N.; Popli, P.; Tiwary, N.; Swami, R. Small Molecules as Cancer Targeting Ligands: Shifting the Paradigm. Journal of Controlled Release 2023, 355, 417–433. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Wu, J. : Liu, B. Therapeutic Strategies of Dual-Target Small Molecules to Overcome Drug Resistance in Cancer Therapy. Biochim Biophys Acta Rev Cancer 2023, 1878. [Google Scholar] [CrossRef] [PubMed]
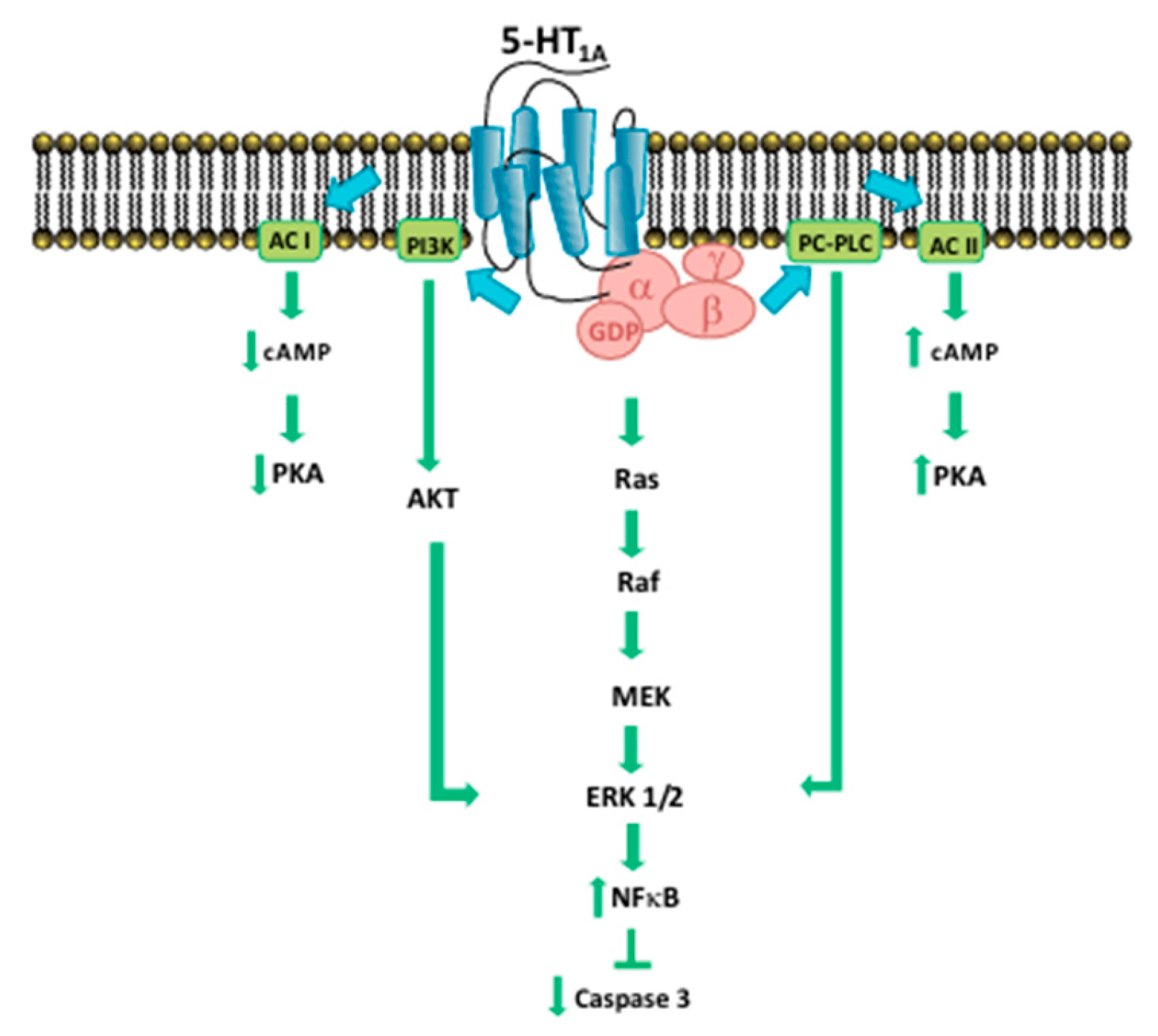
- Corvino, A.; Fiorino, F.; Severino, B.; Saccone, I.; Frecentese, F.; Perissutti, E.; Di Vaio, P.; Santagada, V.; Caliendo, G.; Magli, E. The Role of 5-HT1A Receptor in Cancer as a New Opportunity in Medicinal Chemistry. Curr Med Chem 2018, 25, 3214–3227. [Google Scholar] [CrossRef] [PubMed]
- Cowen, D.S.; Sowers, R.S.; Manning, D.R. Activation of a Mitogen-Activated Protein Kinase (ERK2) by the 5-Hydroxytryptamine 1A Receptor Is Sensitive Not Only to Inhibitors of Phosphatidylinositol 3-Kinase, but to an Inhibitor of Phosphatidylcholine Hydrolysis; JBC, 1996.
- Blesen, T.; Hawes, B.E.; Luttrell, D.K.; Krueger, K.M.; Touhara, K.; Porflrlt, E.; Sakauet, M.; Luttrell, L.M.; Lefkowitz, R.J. Receptor-tyrosine-kinase- and Gpy-mediated MAP kinase activation by a common signalling pathway LETTERS TO NATURE 1995, 376.
- Della Rocca, G.J.; Mukhin, Y. V.; Garnovskaya, M.N.; Daaka, Y.; Clark, G.J.; Luttrell, L.M.; Lefkowitz, R.J.; Raymond, J.R. Serotonin 5-HT1A Receptor-mediated Erk Activation Requires Calcium/Calmodulin-dependent Receptor Endocytosis. THE JOURNAL OF BIOLOGICAL CHEMISTRY, 1999, 4749-4753. [CrossRef]
- Shinka, T.; Onodera, D.; Tanaka, T.; Shoji, N.; Miyazaki, T.; Moriuchi, T.; Fukumoto, T. Serotonin Synthesis and Metabolism-Related Molecules in a Human Prostate Cancer Cell Line. Oncol Lett 2011, 2, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Tutton, P.J.M.; Steel, G.G. Influence of biogenic amines on the growth of xenografted human colorectal carcinomas; Br. J. Cancer, 1979; 40, 743-749.
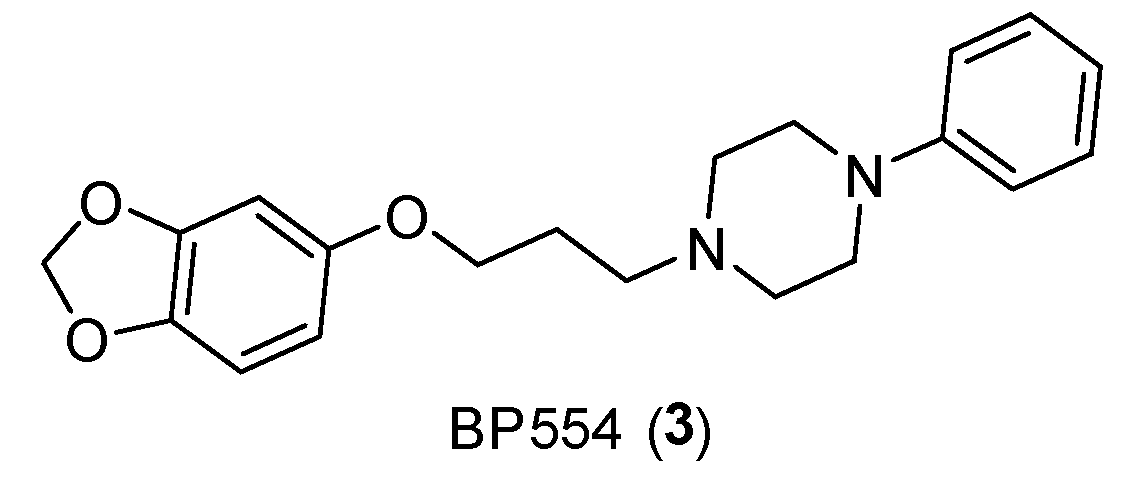
- Matsuda, T.; Seong, Y. H.; Aono, H.; Kanda, T.; Baba, A.; Saito, K.; Tobe, A.; Iwata, H. Agonist Activity of a Novel Compound, 1-[3-(3,4-Methylenedioxyphenoxy) Propyl]-4-Phenyl Piperazine (BP-554), at Central 5-HT1A Receptors. Eur J Pharmacol 1989, 170, 75–82. [Google Scholar] [CrossRef] [PubMed]
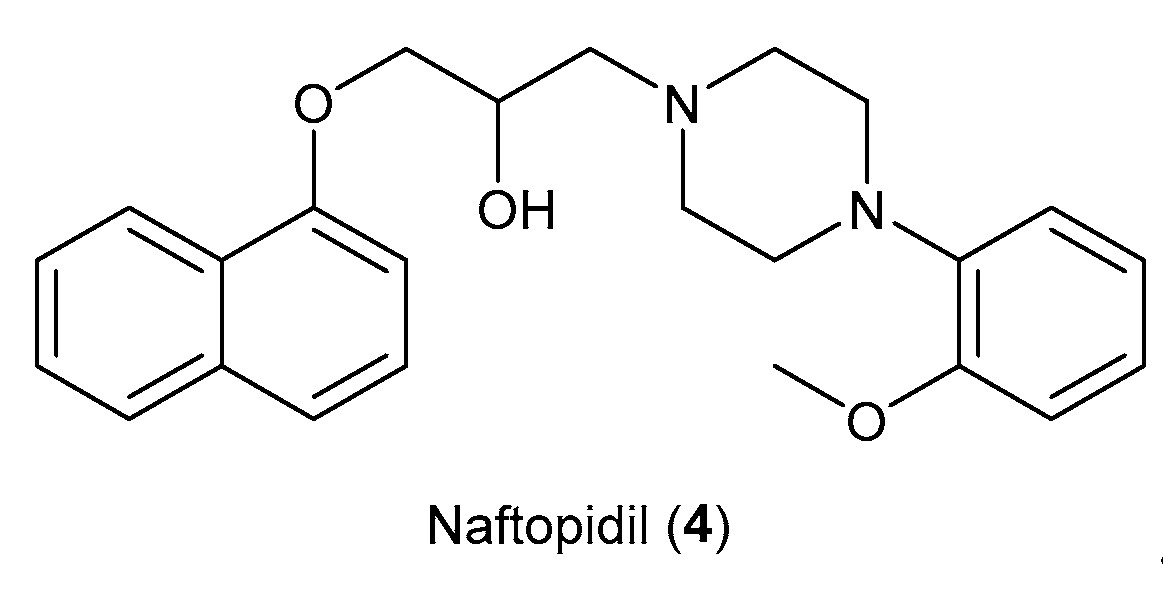
- Florent, R.; Poulain, L.; N’Diaye, M. Drug Repositioning of the A1-Adrenergic Receptor Antagonist Naftopidil: A Potential New Anti-Cancer Drug? Int J Mol Sci 2020, 21, 1–21. [Google Scholar] [CrossRef] [PubMed]
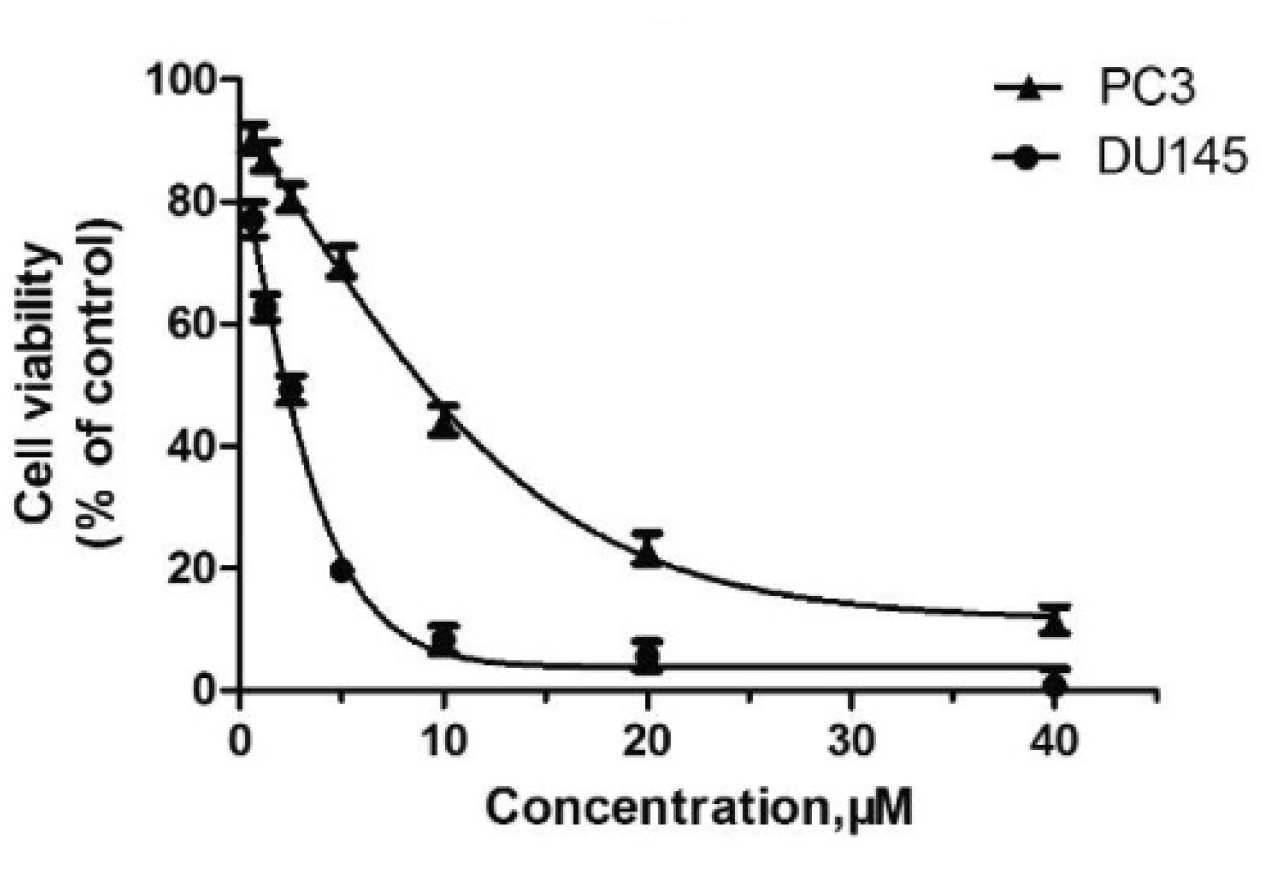
- Chen, H.; Liang, X.; Xu, F.; Xu, B.; He, X.; Huang, B.; Yuan, M. Synthesis and Cytotoxic Activity Evaluation of Novel Arylpiperazine Derivatives on Human Prostate Cancer Cell Lines. Molecules 2014, 19, 12048–12064. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Chen, H.; Xu, J.; Liang, X.; He, X.; Shao, B.; Sun, X.; Li, B.; Deng, X.; Yuan, M. Synthesis, Structure-Activity Relationship and Biological Evaluation of Novel Arylpiperzines as A1A/1D-AR Subselective Antagonists for BPH. Bioorg Med Chem 2015, 23, 7735–7742. [Google Scholar] [CrossRef] [PubMed]
- Hanušová, V.; Skálová, L.; Kralova, V.; Matouskova, P. Potential Anti-cancer Drugs Commonly Used for Other Indications. Current Cancer Drug Targets 2015, 15, 35-52 2015. [CrossRef] [PubMed]
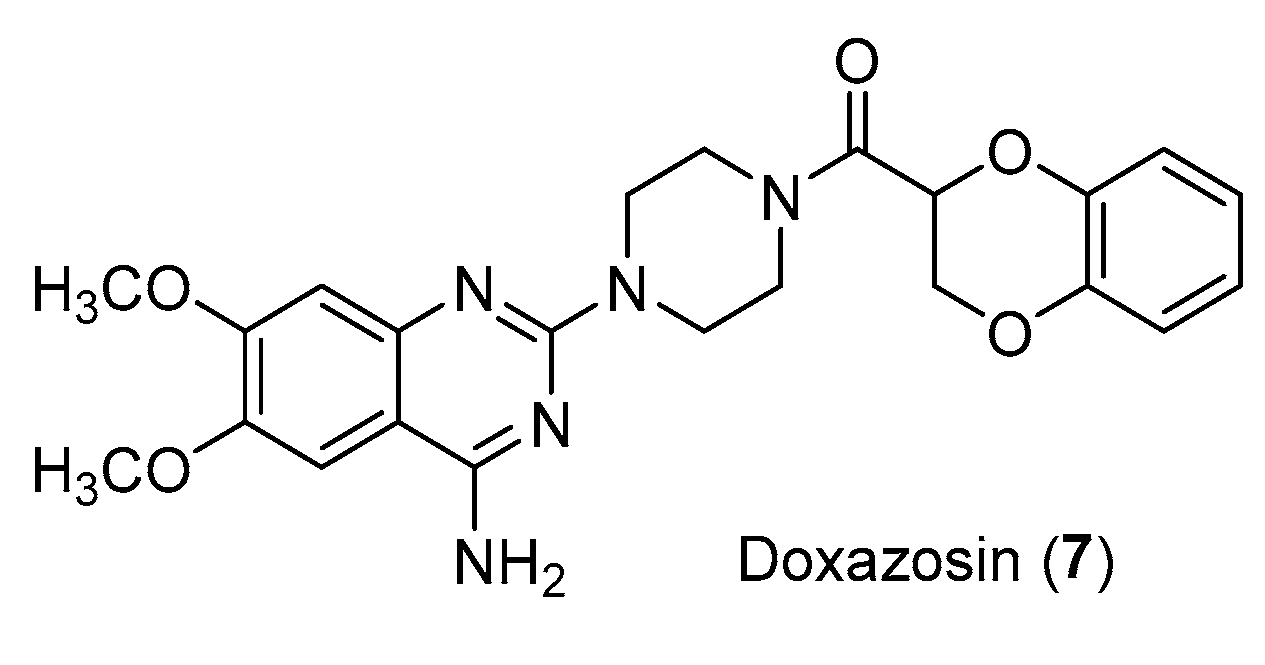
- Suzuki, S.; Yamamoto, M.; Sanomachi,T.; Togashi, K.; Sugai, A.; Seino, S.; Okada, M.; Yoshioka, T.; Kitanaka, C. Doxazosin, a Classic Alpha 1-Adrenoceptor Antagonist, Overcomes Osimertinib Resistance in Cancer Cells via the Upregulation of Autophagy as Drug Repurposing. Biomedicines, 2020; 8, 273. [CrossRef]
- Qi, Y.; Chen, H.; Chen, S.; Shen, J.; Li, J. Synthesis, Bioactivity, and Molecular Docking of Novel Arylpiperazine Derivatives as Potential AR Antagonists. Front Chem 2022, 10. [Google Scholar] [CrossRef] [PubMed]
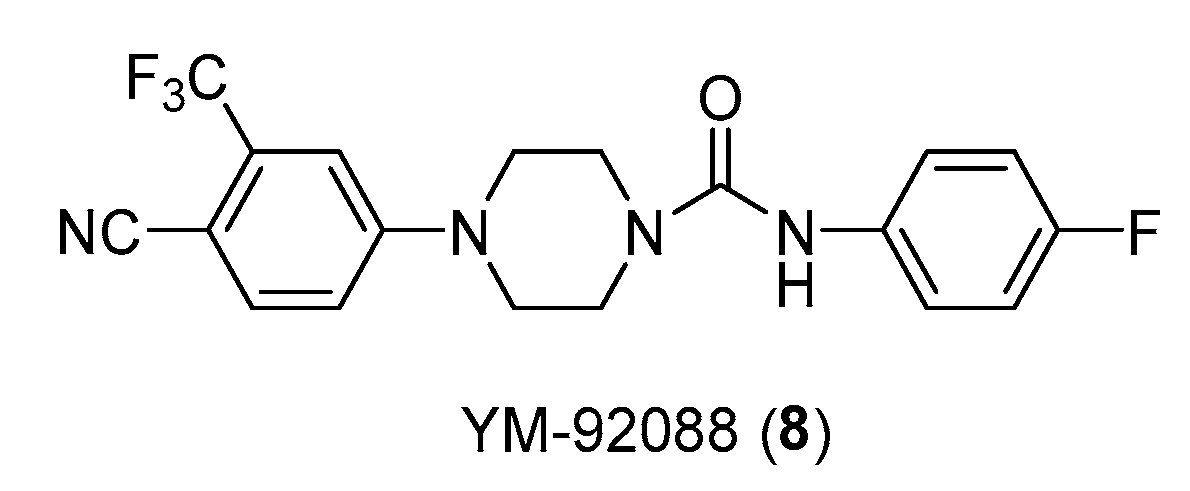
- Kinoyama, I.; Taniguchi, N.; Yoden, T.; Koutoku, H.; Furutani, T.; Kudoh, M.; Okada, M. Synthesis and Pharmacological Evaluation of Novel Arylpiperazine Derivatives as Nonsteroidal Androgen Receptor Antagonists. Chem Pharm Bull (Tokyo) 2004, 52, 1330–1333. [Google Scholar] [CrossRef]
- Sandhu, S.; Moore, C.M.; Chong, E.; Beltran, H.; Bristow, R.G.; Williams, S.G. Prostate Cancer. The Lancet 2021, 398, 1075–1090. [Google Scholar] [CrossRef] [PubMed]
- Attard, G.; Parker, C.; Eeles, R.A.; Schroder, F.; Tomlins, S.A.; Tannok,I.; Drake, C.G.; de Bono, J.S. Prostate Cancer. The Lancet 2016, 387, 70–82.
- Chen, H.; Wang, C.L.; Sun, T.; Zhou, Z.; Niu, J.; Tian, X.; Yuan, M. Synthesis, Biological Evaluation and SAR of Naftopidil-Based Arylpiperazine Derivatives. Bioorg Med Chem Lett 2018, 28, 1534–1539. [Google Scholar] [CrossRef] [PubMed]
- Kinoyama, I.; Taniguchi.; Kawaminami, E.; Nozawa, E.; Koutoku, H.; Furutani, T.; Kudoh, M.; Okada, M. N-Arylpiperazine-1-Carboxamide Derivatives: A Novel Series of Orally Active Nonsteroidal Androgen Receptor Antagonists. Chem. Pharm. Bull. 2005, 53, 402-409.
- Torre, L.A.; Bray, F.; Siegel, R. L.; Ferlay, J.; Lortet-Tieulent, J.; Jemal, A. CA Cancer J Clin 2015, 65, 87–108.
- Szczęśniak-Sięga, B.M.; Mogilski, S.; Wigluszc, R.J.; Janczak, J.; Maniewska, J.; Malinka, W.; Filipek, B. Synthesis and Pharmacological Evaluation of Novel Arylpiperazine Oxicams Derivatives as Potent Analgesics without Ulcerogenicity. Bioorg Med Chem 2019, 27, 1619–1628. [Google Scholar] [CrossRef] [PubMed]
- Szczuka, I.; Wierzbicki, J.; Serek, P.; Szczesniak-Siega, B.M.; Krzystek-Korpacka, M. Heat Shock Proteins Hspa1 and Hsp90aa1 Are Upregulated in Colorectal Polyps and Can Be Targeted in Cancer Cells by Anti-Inflammatory Oxicams with Arylpiperazine Pharmacophore and Benzoyl Moiety Substitutions at Thiazine Ring. Biomolecules 2021, 11, 1588. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Chu, M.; Song, G.; Zuo, Z.; Han, Z.; Chen, C.; Li, Y.; Wang, Z. Emerging Roles of Long Noncoding RNAs in Chemoresistance of Pancreatic Cancer. Semin Cancer Biol 2022, 83, 303–318. [Google Scholar] [CrossRef] [PubMed]
- Mizrahi, J.D.; Surana, R.; Valle, J.W.; Shroff, R.T. Pancreatic Cancer; 2008, 395, 2008-20.
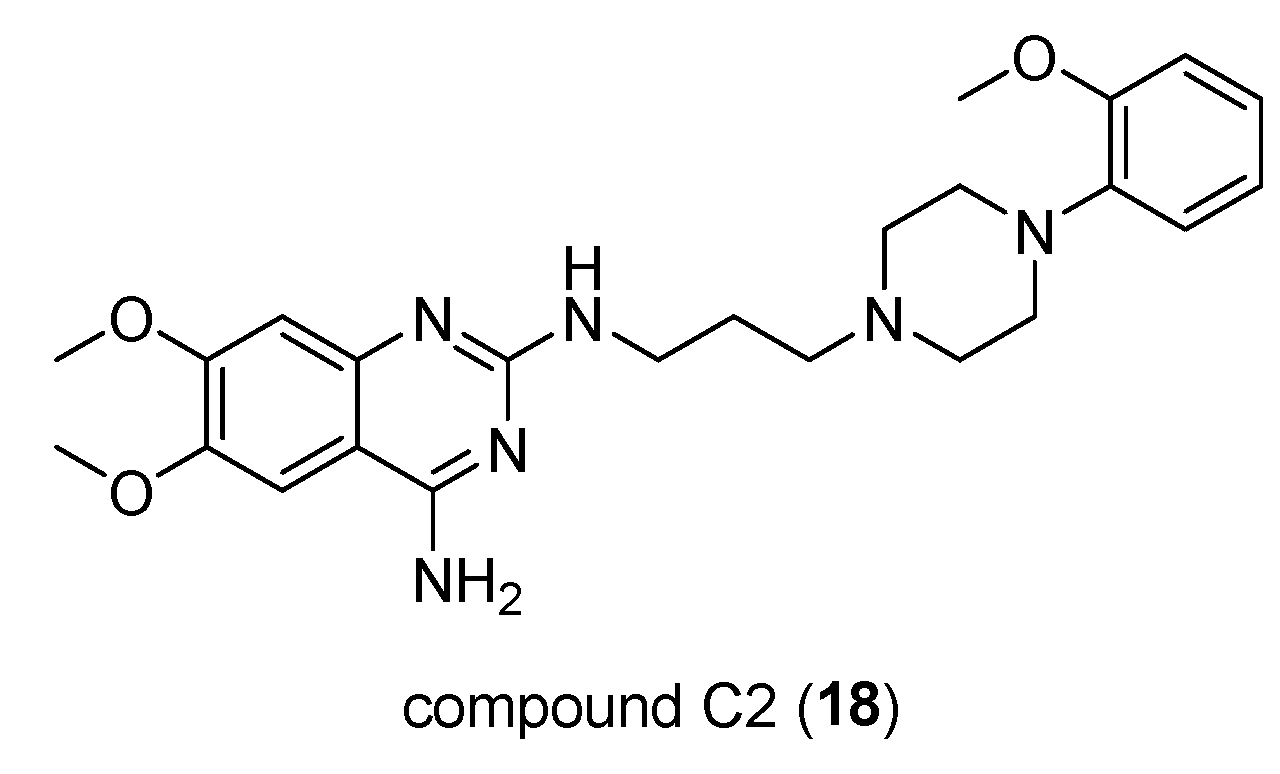
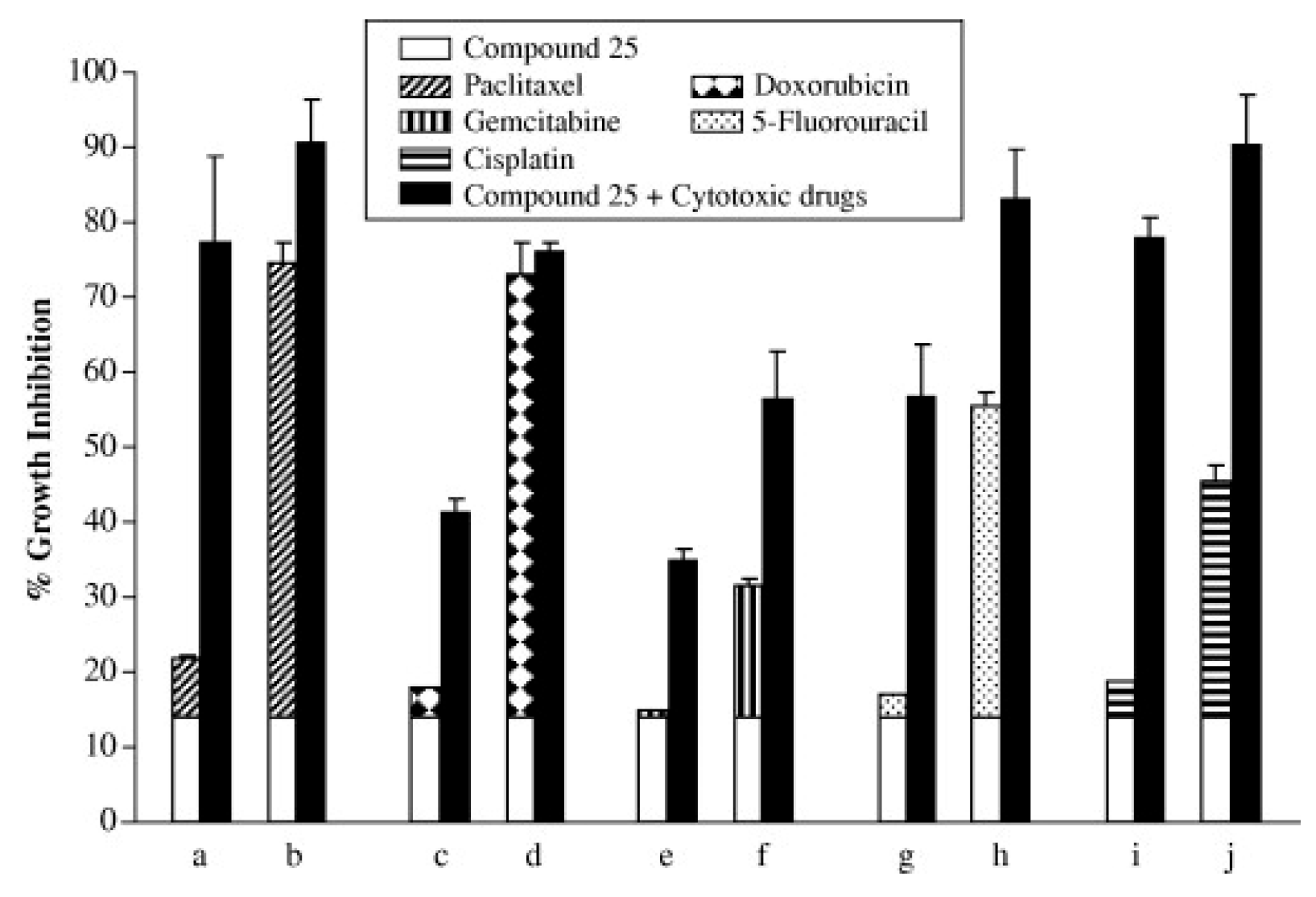
- Su, H.; Xue, Z.; Feng, Y.; Xie, Y.; Deng, B.; Yao, Y.; Tian, X.; An, Q.; Yang, L.; Yao, Q.; Xue, J.; Chen, G.; Hao, C.; Zhou,T. N-Arylpiperazine-Containing Compound (C2): An Enhancer of Sunitinib in the Treatment of Pancreatic Cancer, Involving D1DR Activation. Toxicol Appl Pharmacol 2019, 384, 114789. [CrossRef]
- Wang, C.; Niu, M.; Zhou, Z.; Zheng, X.; Zhang, L.; Tian, Y.; Yu, X.; Bu, G.; Xu, H.; Ma, Q.; Zhang, Y. VPS35 Regulates Cell Surface Recycling and Signaling of Dopamine Receptor D1. Neurobiol Aging 2016, 46, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Hao, F.; Wang, S.; Zhu1, X.; Xue, J.; Li, J.; Wang, L.; Li, J.; Lu, W.; Zhou, T. Pharmacokinetic-Pharmacodynamic Modeling of the Anti-Tumor Effect of Sunitinib Combined with Dopamine in the Human Non-Small Cell Lung Cancer Xenograft. Pharm Res 2017, 34, 408–418. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Wang, S.; Ren, Y.; Li, J.; Guo, T.; Lu, W.; Zhou, T. Antitumor Effect of Axitinib Combined with Dopamine and PK-PD Modeling in the Treatment of Human Breast Cancer Xenograft. Acta Pharmacol Sin 2019, 40, 243–256. [Google Scholar] [CrossRef] [PubMed]
- Zubair, M.; Wang, S.; Ali, N. Advanced Approaches to Breast Cancer Classification and Diagnosis. Front Pharmacol 2021, 11. [Google Scholar] [CrossRef] [PubMed]
- Ambrosio, M.R.; Magli, E.; Caliendo, G.; Sparaco, R.; Massarelli, P.; D’Esposito, V.; Migliaccio, T.; Mosca, G.; Fiorino, F.; Formisano, P. Serotoninergic Receptor Ligands Improve Tamoxifen Effectiveness on Breast Cancer Cells. BMC Cancer 2022, 22, 171. [Google Scholar] [CrossRef] [PubMed]
- Fiorino, F.; Perissutti, E.; Severino, B.; Santagada, V.; Cirillo, D.; Terracciano, S.; Massarelli, P.; Bruni, G.; Collavoli, E.; Renner, C.; Caliendo, G. New 5-Hydroxytryptamine1A Receptor Ligands Containing a Norbornene Nucleus: Synthesis and in Vitro Pharmacological Evaluation. J Med Chem 2005, 48, 5495–5503. [Google Scholar] [CrossRef] [PubMed]
- Fiorino, F.; Magli, E.; Ke˛dzierska, E.; Ciano, A.; Corvino, A.: Severino, B.; Perissutti, E.; Frecentese, F.; Di Vaio, P.; Saccone, I.; Izzo, A. A.; Capasso, R.; Massarelli, P.; Rossi, I.; Orzelska-Gòrkac, J.; Kotlin’ska, J.HJ; Santagada, V.; Caliendo, G. New 5-HT1A, 5HT2A and 5HT2C Receptor Ligands Containing a Picolinic Nucleus: Synthesis, in Vitro and in Vivo Pharmacological Evaluation. Bioorg Med Chem 2017, 25, 5820–5837.
- Andreozzi, G.; Ambrosio, M.R.; Magli, E.; Maneli, G.; Severino, B.; Corvino, A.; Sparaco, R.; Perissutti,E.; Le´ sniak, A.; Frecentese, F.; Santagada,V.; Bujalska-Zadrozny,M.; Caliendo, G.; Formisano, P.; Fiorino, F. Design, Synthesis and Biological Evaluation of Novel N-Arylpiperazines Containing a 4,5-Dihydrothiazole Ring. Pharmaceuticals 2023, 16, 1483.
- Ferrall, L.; Lin, K.Y.; Roden, R.B.S.; Hung, C.-F.; Wu, T.-C. Cervical Cancer Immunotherapy: Facts and Hopes. Clinical Cancer Research 2021, 27, 4953–4973. [Google Scholar] [CrossRef] [PubMed]
- Small, W.; Bacon, M.A.; Bajaj,A.; Chuang, L.T.; Fisher, B.J.; Harkenrider, M.M.; Jhingran,A.; Kitchener, H.C.; Mileshkin, L.R.; Viswanathan, A.N.; Gaffney, D.K. Cervical Cancer: A Global Health Crisis. Cancer 2017, 123, 2404–2412. [CrossRef]
- Mao, Z.W.; Zheng, X.; Lin, Y.-P.; Hu, C.-Y.; Wang, X.-L.; Wan, C.-P.; Rao, G.-X. Design, Synthesis and Anticancer Activity of Novel Hybrid Compounds between Benzofuran and N-Aryl Piperazine. Bioorg Med Chem Lett 2016, 26, 3421–3424. [Google Scholar] [CrossRef] [PubMed]
- Snyder, R. Leukemia and Benzene. Int J Environ Res Public Health 2012, 9, 2875–2893. [Google Scholar] [CrossRef] [PubMed]
- Choi, M.J.; No, E.S.; Thorat, D.A.; Jang, J.W.; Yang,H.; Lee,J.; Choo,H.; Kim,S.J.; Lee,C.S.; Ko,S.Y.; Lee,J.; Nam,G.; Pae, A.N. Synthesis and Biological Evaluation of Aryloxazole Derivatives as Antimitotic and Vascular-Disrupting Agents for Cancer Therapy. J Med Chem 2013, 56, 9008–9018. [CrossRef]
- Saginala, K.; Barsouk, A.; Aluru, J.S.; Rawla,P.; Barsouk,A. Epidemiology of Melanoma. Med Sci (Basel) 2021, 9, 63.
- Hayward, N.K. Genetics of Melanoma Predisposition. Oncogene 2003, 22, 3053–3062. [Google Scholar] [CrossRef] [PubMed]
- Romagnoli, R.; Oliva, P.; Prencipe, F.; Manfredini, S.; Germano, M.P.; De Luca, L.; Ricci,F.; Corallo, D.; Aveic,S.; Mariotto, E.; Viola, G.;Bortolozzi, R. Cinnamic Acid Derivatives Linked to Arylpiperazines as Novel Potent Inhibitors of Tyrosinase Activity and Melanin Synthesis. Eur J Med Chem 2022, 231. [CrossRef]
- Lee, Y.B.; Gong, Y.D.; Yoon, H.; Ahn,C.H.; Jeon, M.K.; Kong,J.Y. Synthesis and Anticancer Activity of New 1-[(5 or 6-Substituted 2-Alkoxyquinoxalin-3-Yl)Aminocarbonyl]-4-(Hetero)Arylpiperazine Derivatives. Bioorg Med Chem 2010, 18, 7966–7974. [CrossRef]
- Bai, J.W.; Qiu, S.Q. & Zhang, G.J. Molecular and functional imaging in cancer-targeted therapy: current applications and future directions. Sig Transduct Target Ther 2023, 8, 89. [CrossRef]
| Type of Cancer |
Cell Lines Expressing 5-HT1AR |
Drugs | Effects |
|---|---|---|---|
| Prostate Cancer | PC3, DU145, LNCap |
NAN-190 Pindobind |
5HT1A antagonists that inhibit cell growth in vitro, inducing apoptosis. |
| 6-nitroquipazine Zimelidine Fluoxetine |
5HT uptake inhibitors that cause dose- dependent inhibition of cells proliferation. | ||
| Bladder Carcinoma | SHT1376 | NAN 190 SB224289 |
5HT1A (NAN-190) and 5HT1B (SB224289) antagonists that show an inhibitory effect on the serotonin induced growth cells. |
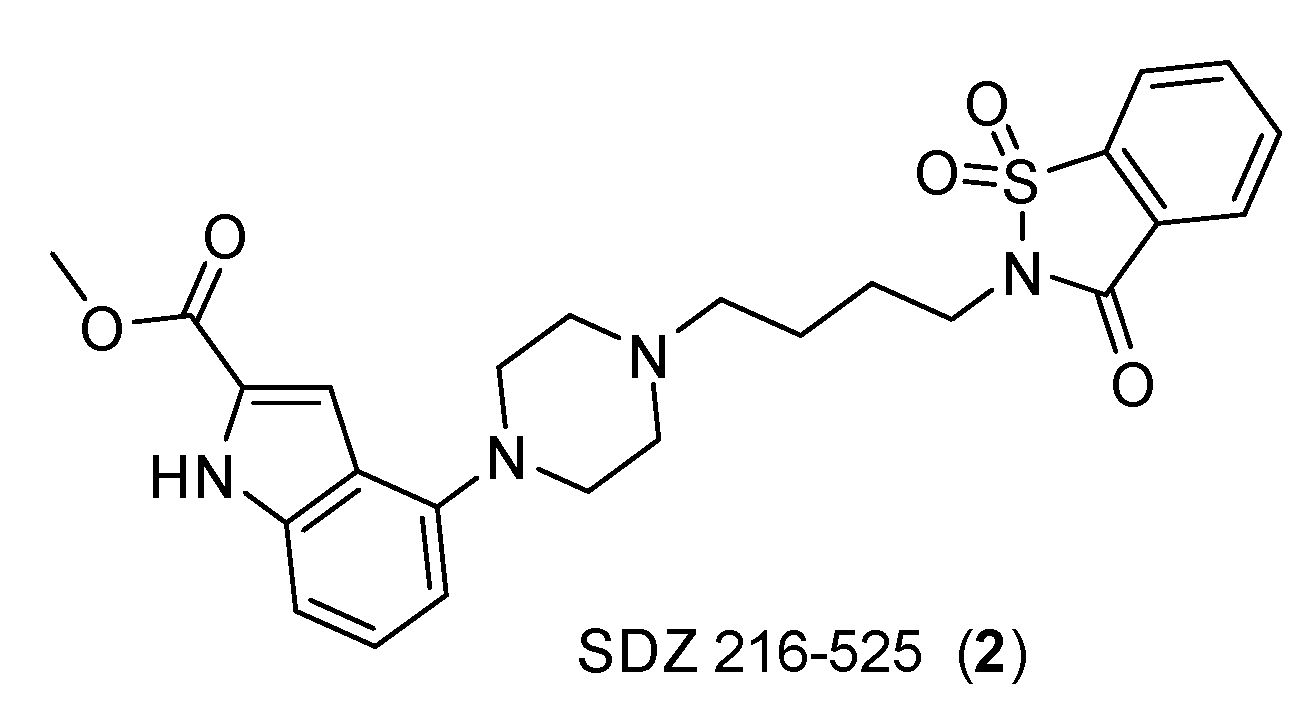
| Small Cell Lung Carcinoma | GLC8 | Spiperone SDZ 216-525 |
5-HT1A (spiperone) and 5-HT7 (SDZ 216-525) antagonists that inhibit 8-OH-DPAT-induced mitogenic effect. |
| Colonrectal Carcinoma | HT29 | BW501C Citalopram Fluoxetine |
Serotoninergic antagonists (BW501C) and SSRIs (Citalopram and Fluoxetine) that retard the tumor growth. |
| NAN 190 SB224289 |
5HT1A (NAN-190) and 5HT1B (SB224289) antagonists that reduce cell growth acting as antiproliferative agents. |
||
| Cholangiocarcinoma | Mz-chA1, HuH28, HUVV-T1, CCLP-1, SG231, TFK1. |
- | - |
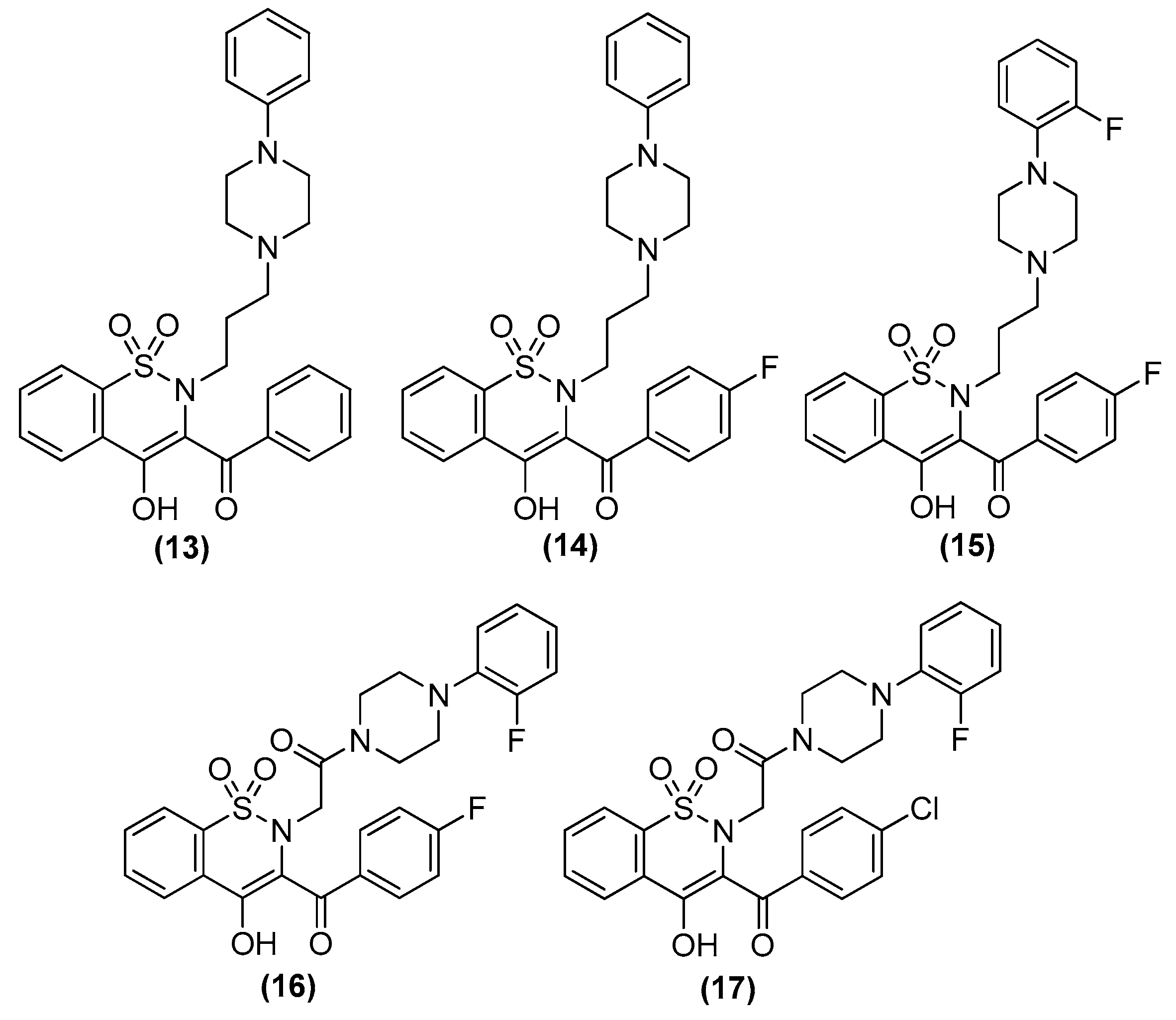
| Compd. | Structure | Selectivity ratio | IC50 values (DU145) |
|---|---|---|---|
| 13 (9) | 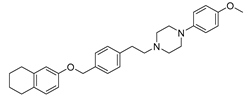 |
α1B/α1A ratio = 16.7 |
0.93 ± 0.19 |
| 17 (10) | 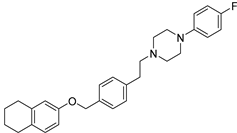 |
α1B/α1D ratio = 10.9 |
0.90 ± 0.20 |
| Compd. | Structure | IC50 (μM)a | % inhibitionb |
|---|---|---|---|
|
5 YM-92088 (11) |
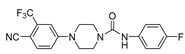 |
0.47 | - |
|
18g (12) |
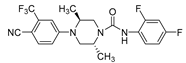 |
0.20 | 85%** ED50=1.1mg/kg |
| Compound | Structure | MCF-7 | MDA-MB231 |
|---|---|---|---|
| 2a (21) | 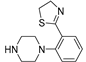 |
14,7±1,9 | 31,37±5,1 |
| 2b (22) | 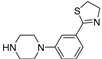 |
15,93±1,8 | 39,96±9,8 |
| 2c (23) | 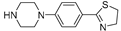 |
19,47±2,3 | 36,32±7,7 |
| 3a (24) | 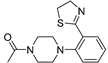 |
- |
23,27±3,4 |
| 3b (25) | 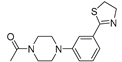 |
- |
34,6±5,4 |
| 3c (26) | 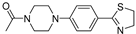 |
- | 47,15±6,7 |
| Compound 13 (27) | 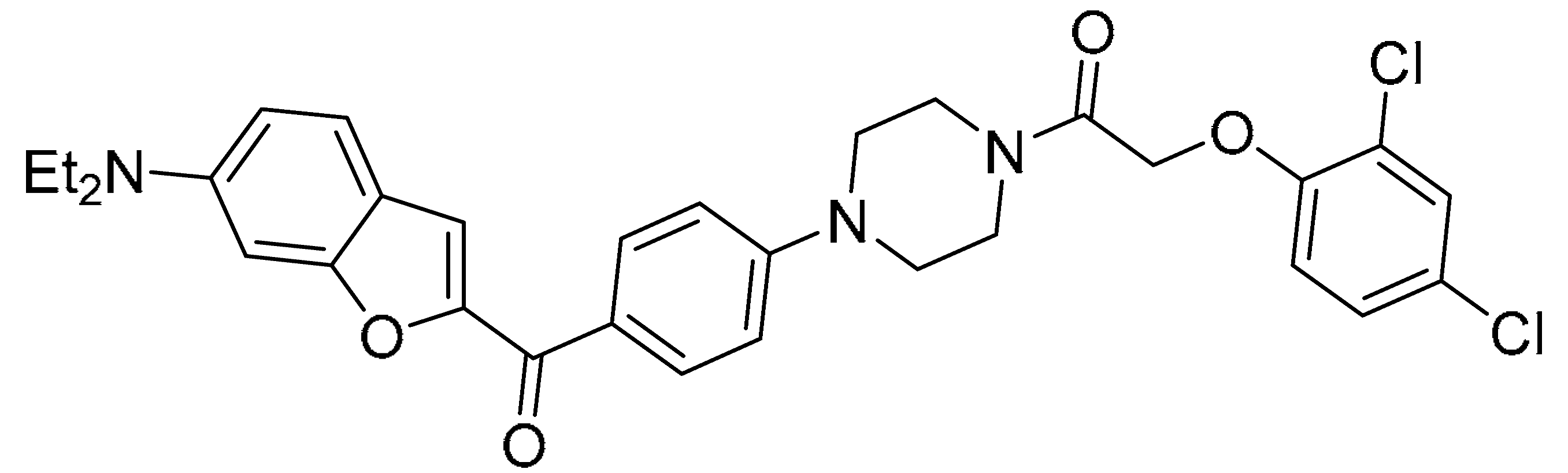 |
||
| Cell lines (IC50 μM) | |||
| A549 | Hela | MCF-7 | SGC7901 |
| 5.73±1.22 | 0.03±0.04 | 12.38±3.62 | 6.17±1.62 |
| Compound | Structure | IC50 (nM) |
|---|---|---|
| 6-48 (28) | 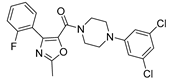 |
60.2 |
| Compound | Structure | IC50 (μM) |
|---|---|---|
| 19r (29) | 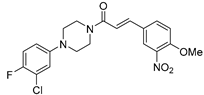 |
0.51±0.10 |
| MDA-MB-231 | 0.012 |
| Caki-1 | 0.011 |
| UMRC2 | 0.013 |
| PANC-1 | 0.021 |
| A549 | 0.021 |
| MKN-45 | 0.020 |
| HepG2 | 0.019 |
| HCT116 | 0.020 |
| HT29 | 0.021 |
| PC-3 | 0.021 |
| U251 | 0.015 |
| HeLa | 0.021 |
| SK-MEL-28 | 0.020 |
| OVCAR-3 | 0.012 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




