Submitted:
17 July 2024
Posted:
18 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
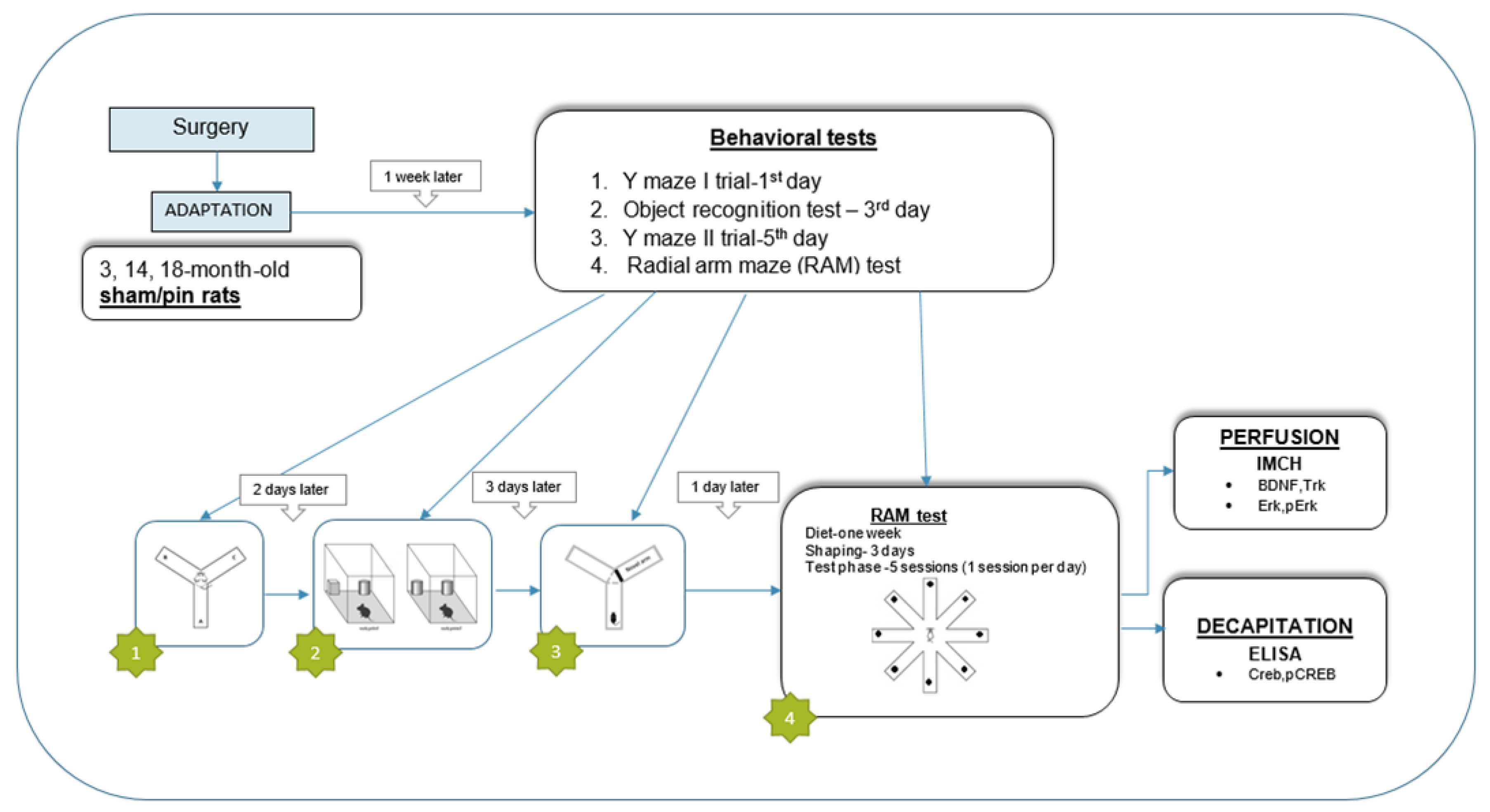
2.1. Experimental Design
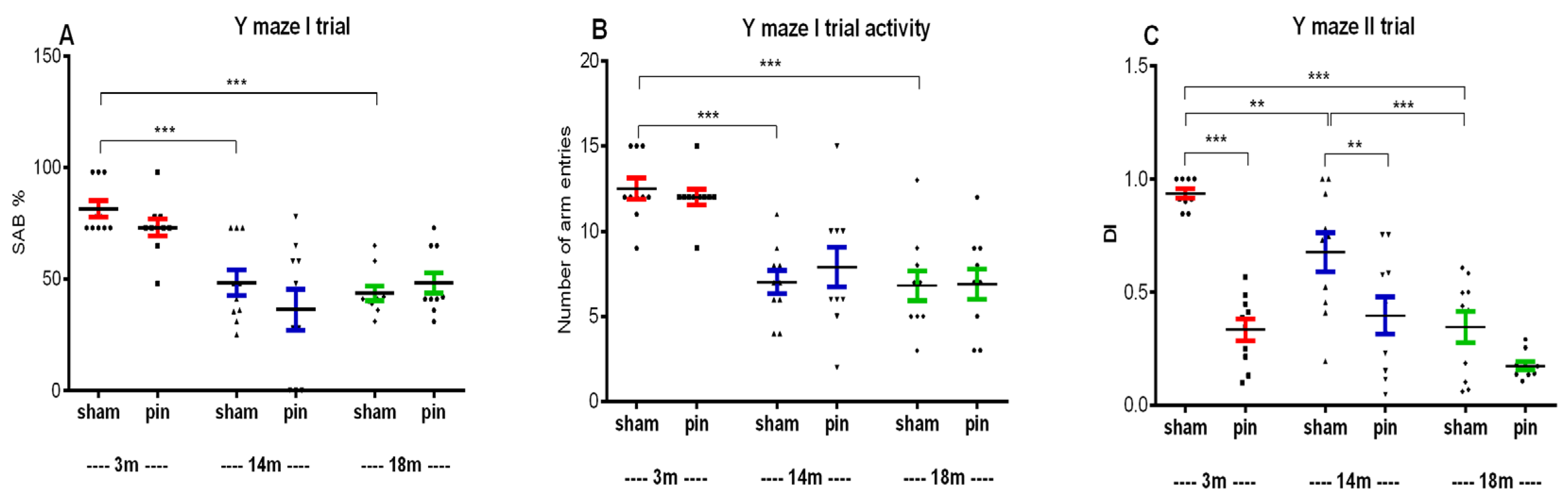
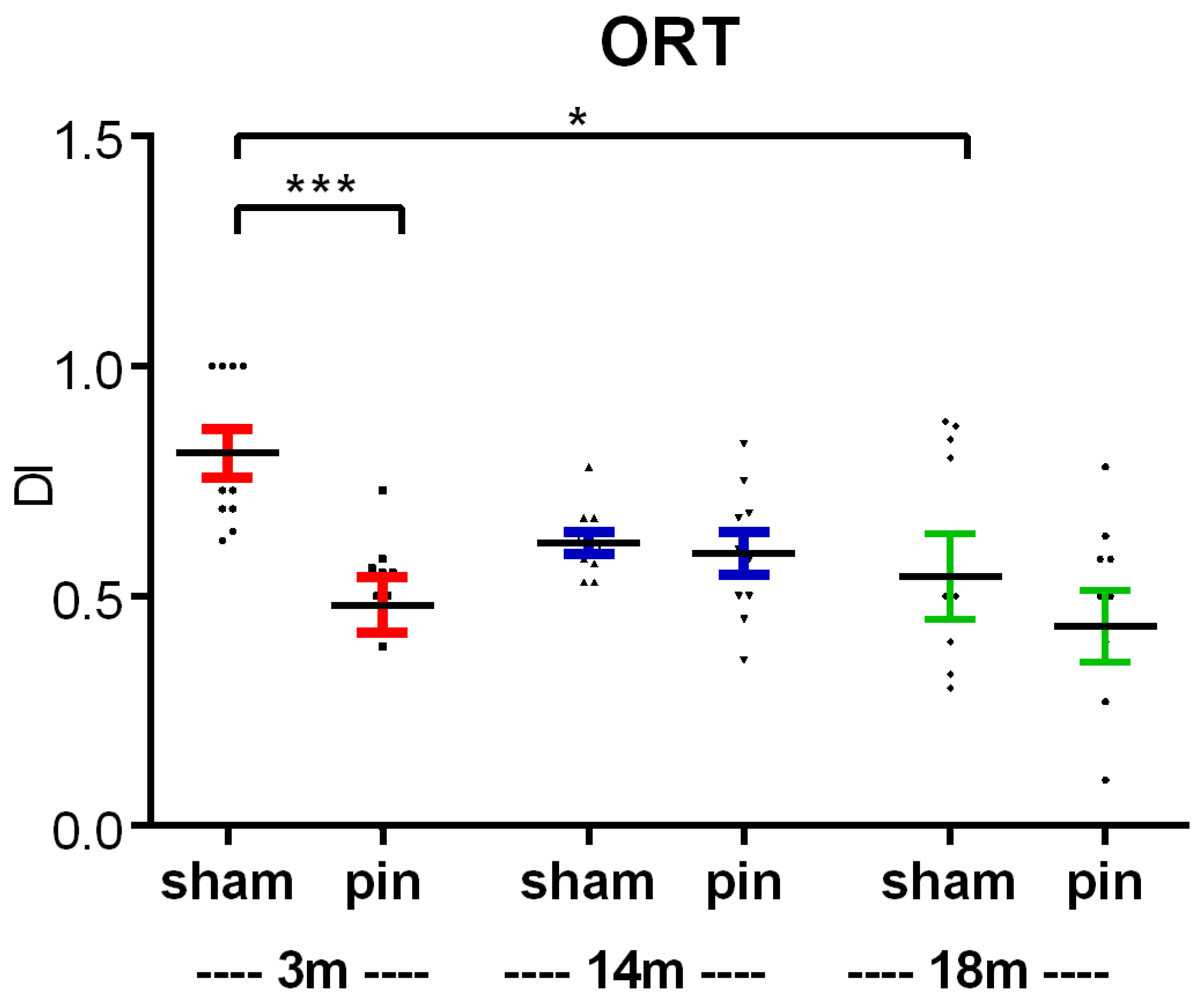
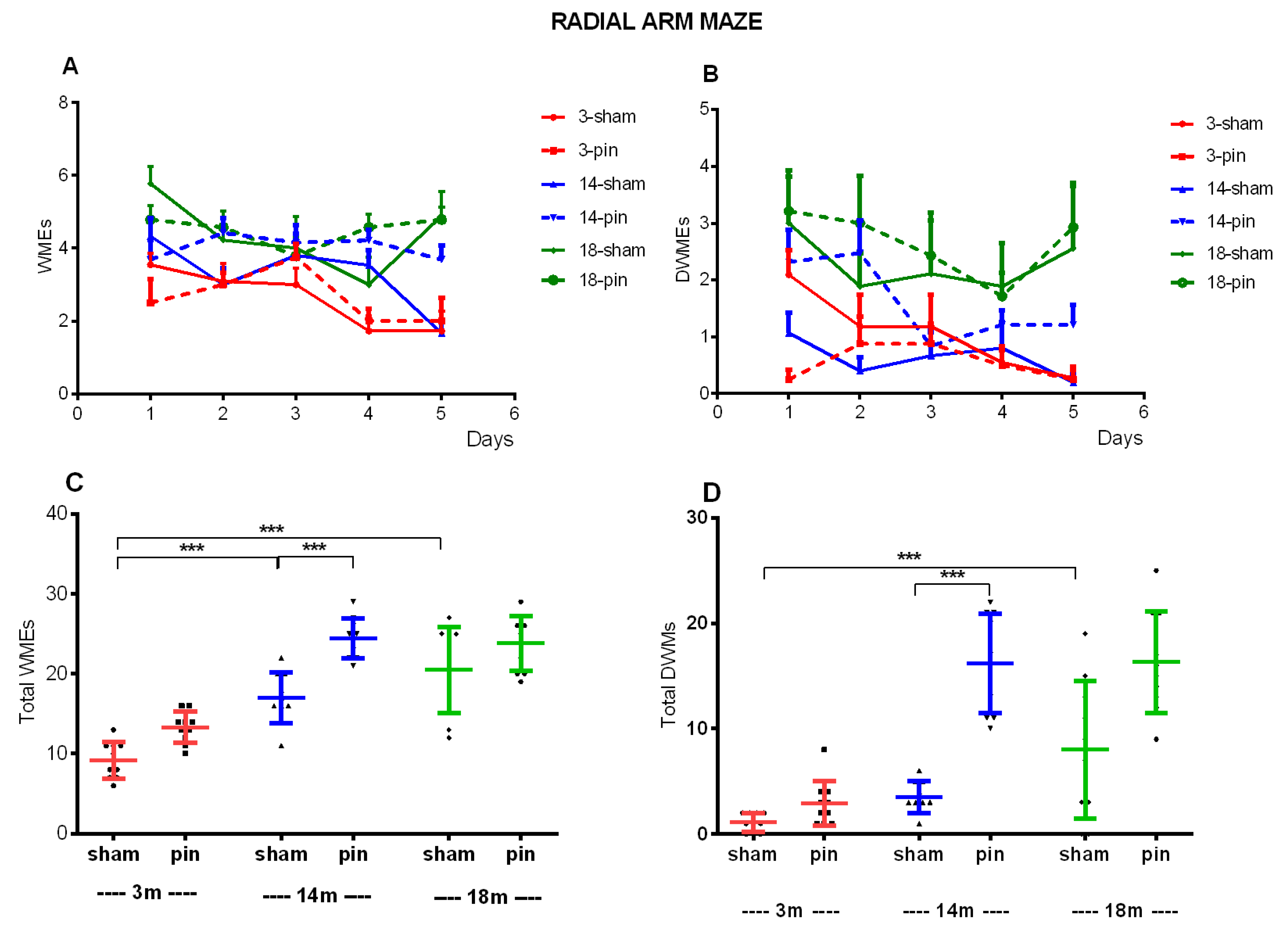
2.2. Melatonin Deficiеncy Associated with Pinealectomy Caused an Age-Dependent Impairment of Working and Short-Term Spatial and Recognition Memory, as Measured by the Y-Maze, ORT and RAM Test
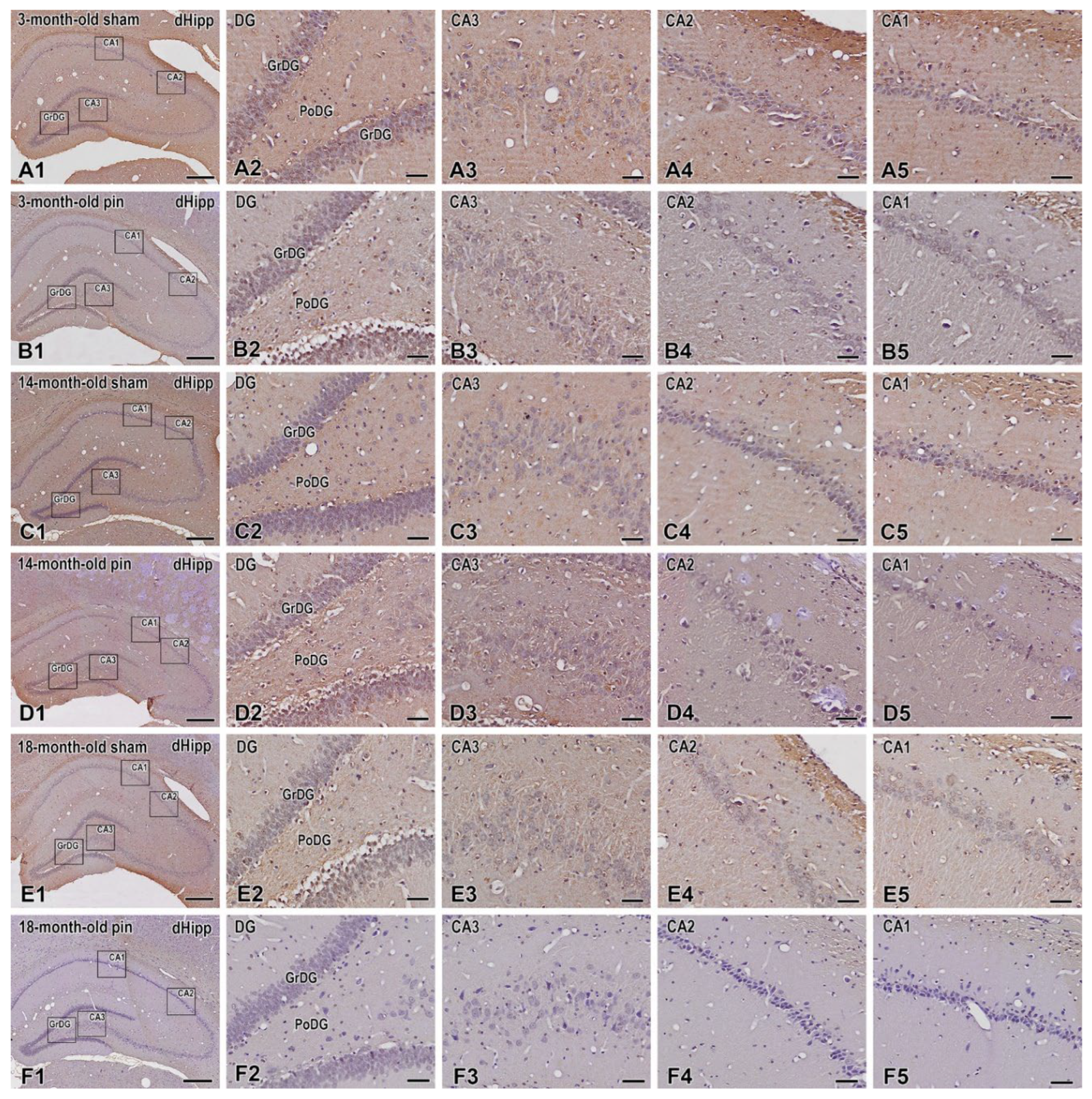
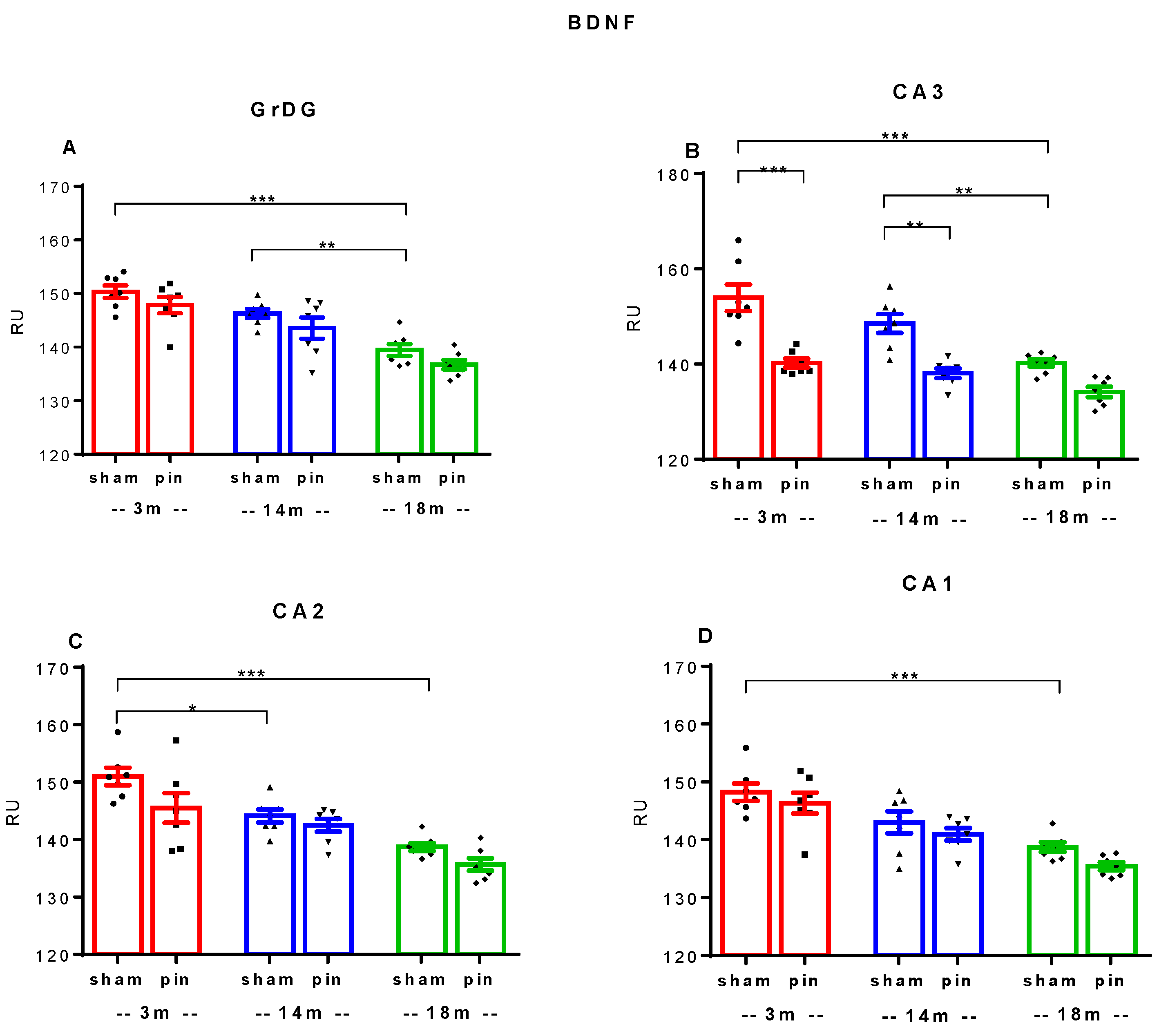
2.3. Melatonin Deficiency Induced by Pinealectomy Reduced BDNF in the CA3 Area of the Hippocampus in Young Adult and Middle-Aged Rats
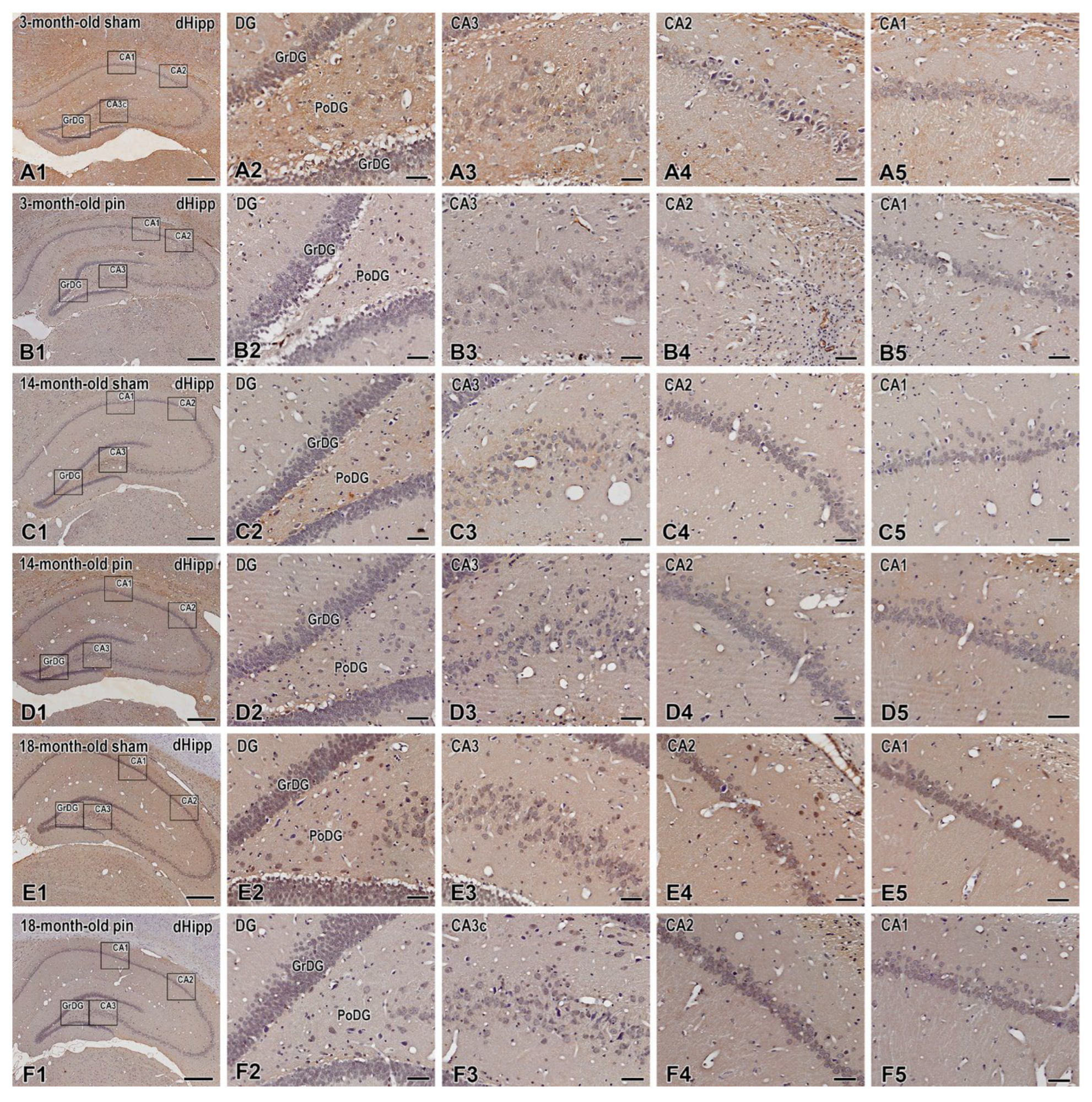
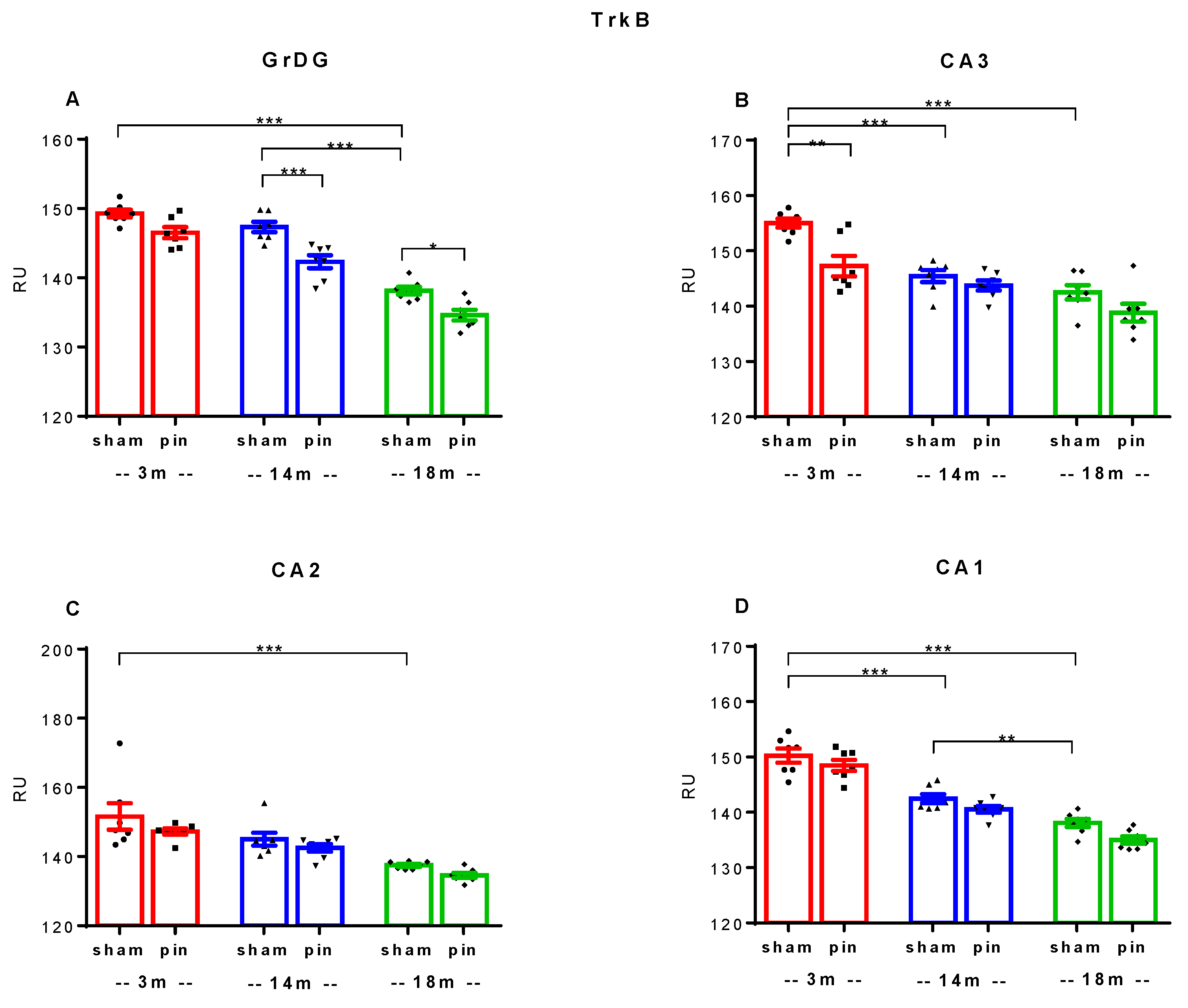
2.4. Melatonin Deficiency Induced by Pinealectomy Reduced TrkB Receptor Expression in Middle-Aged Rats in the GrDG and in Young Adult Rats in the CA3 Region of the Hippocampus
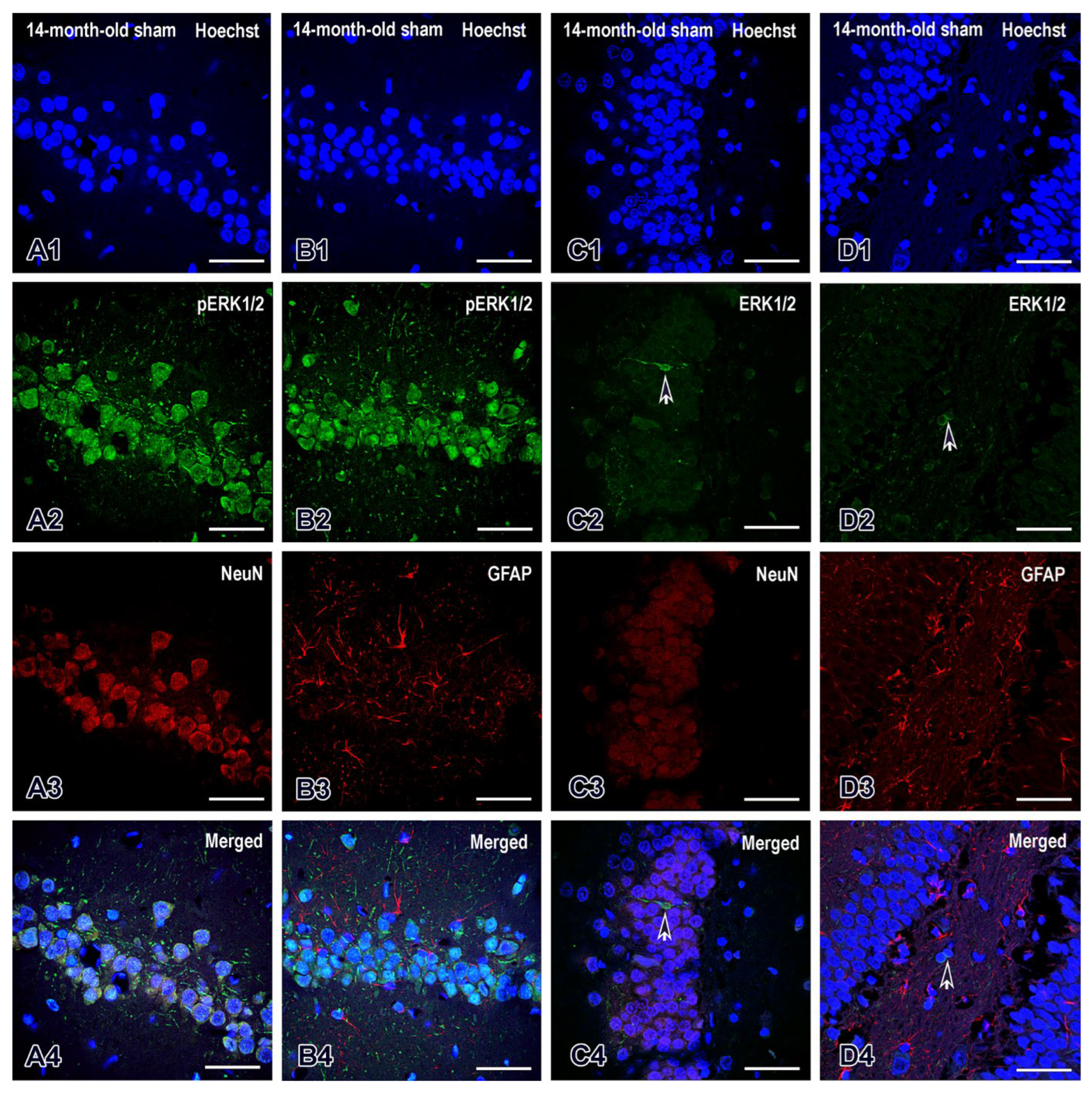
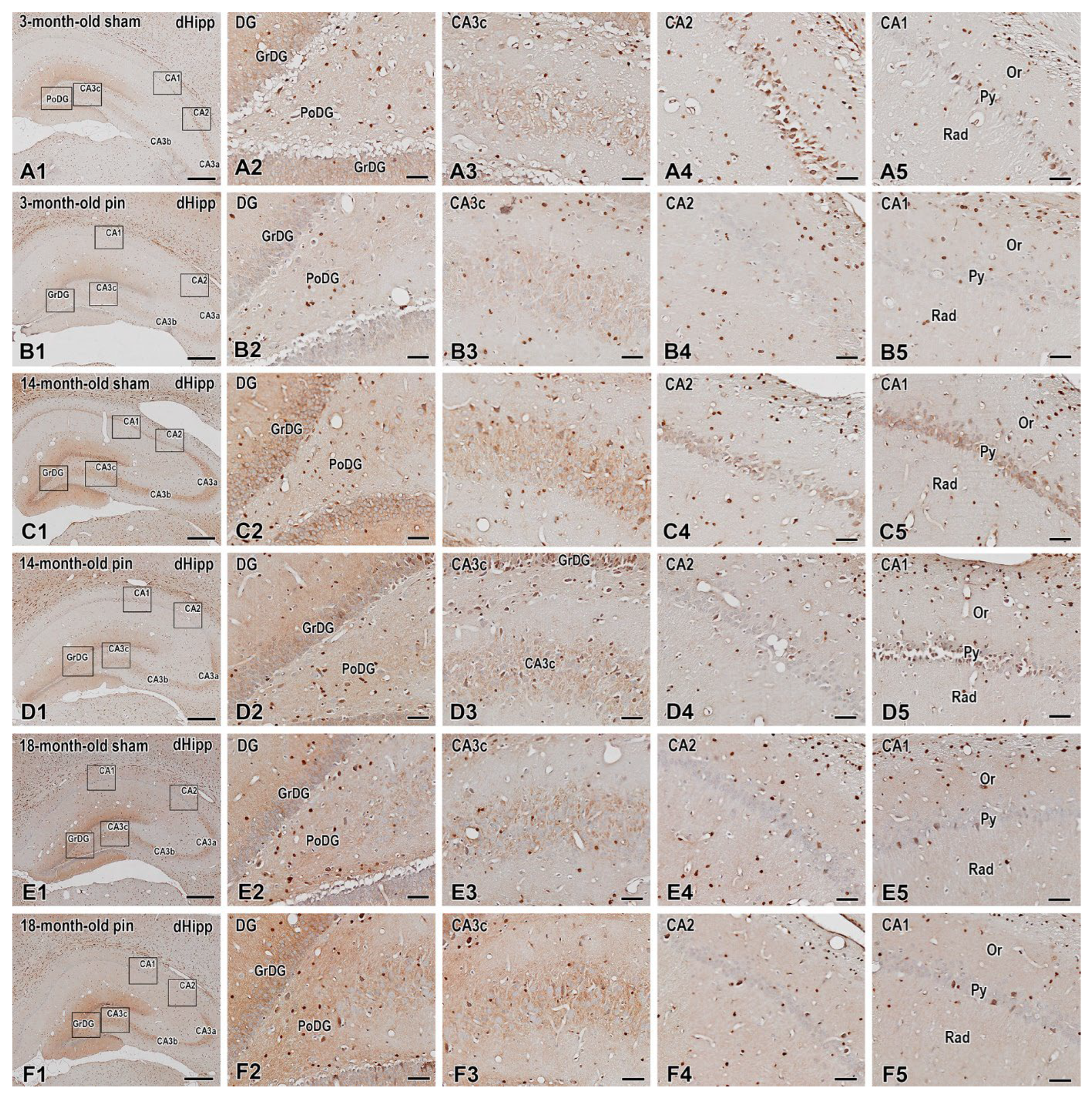
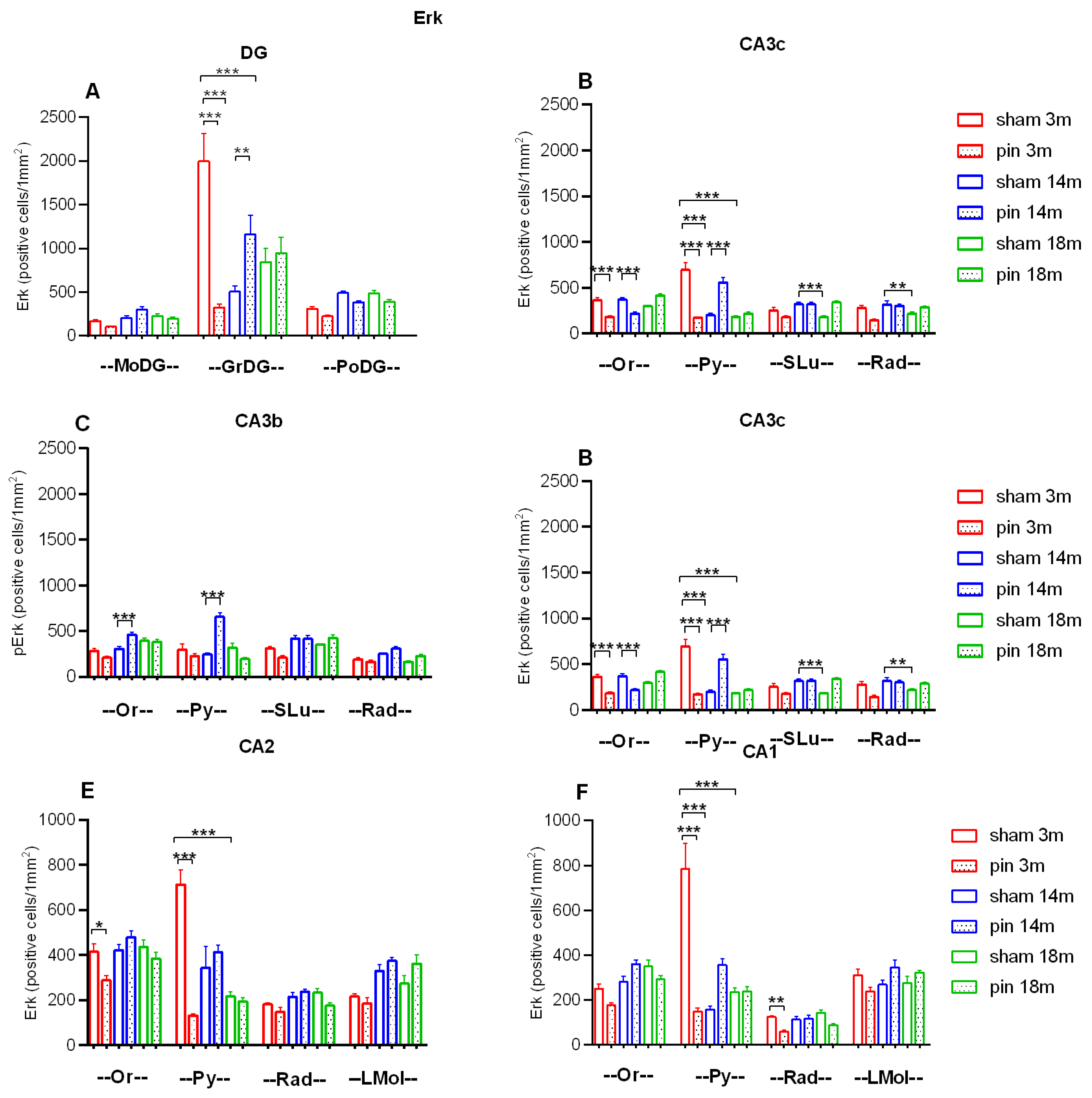
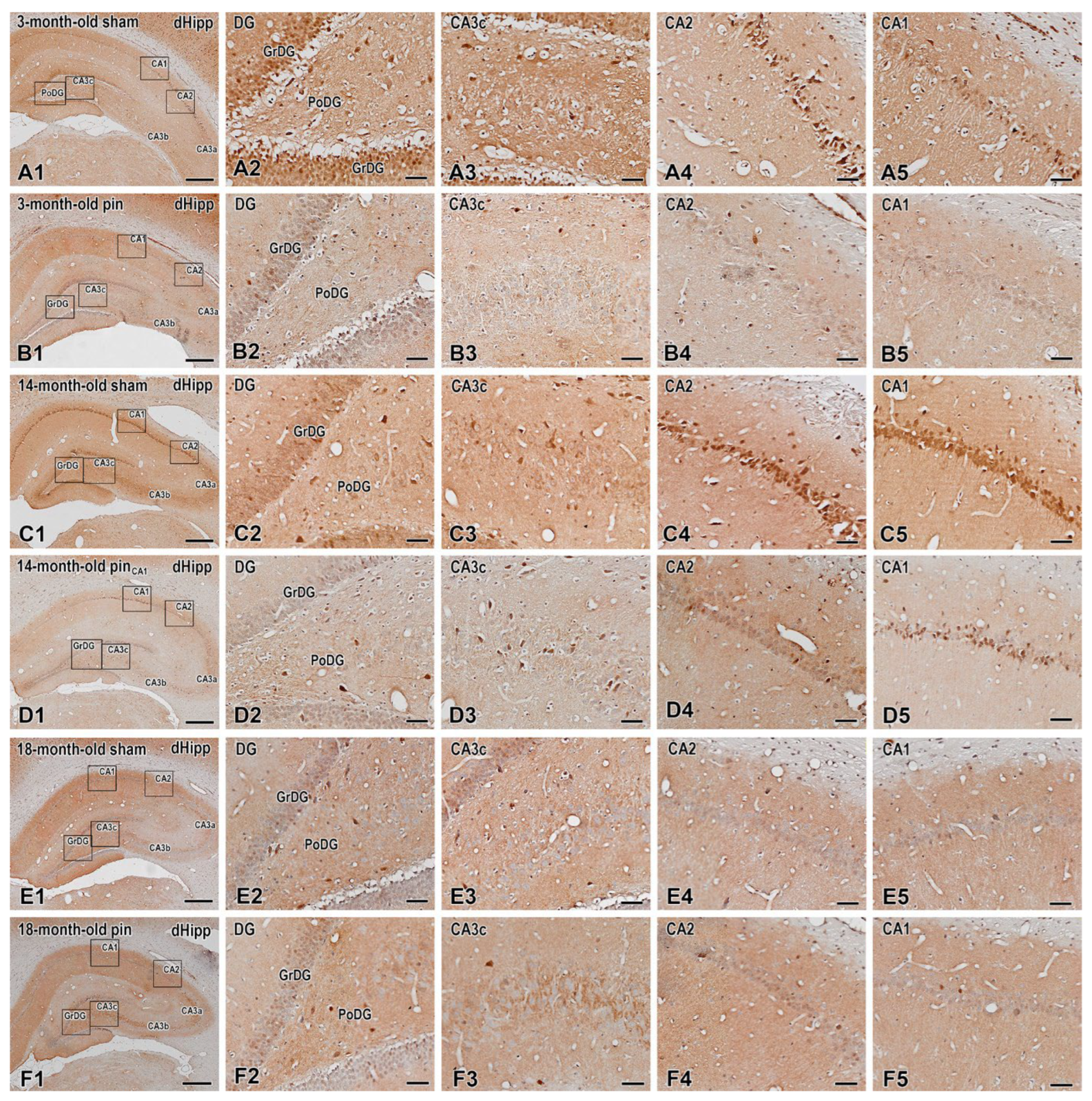
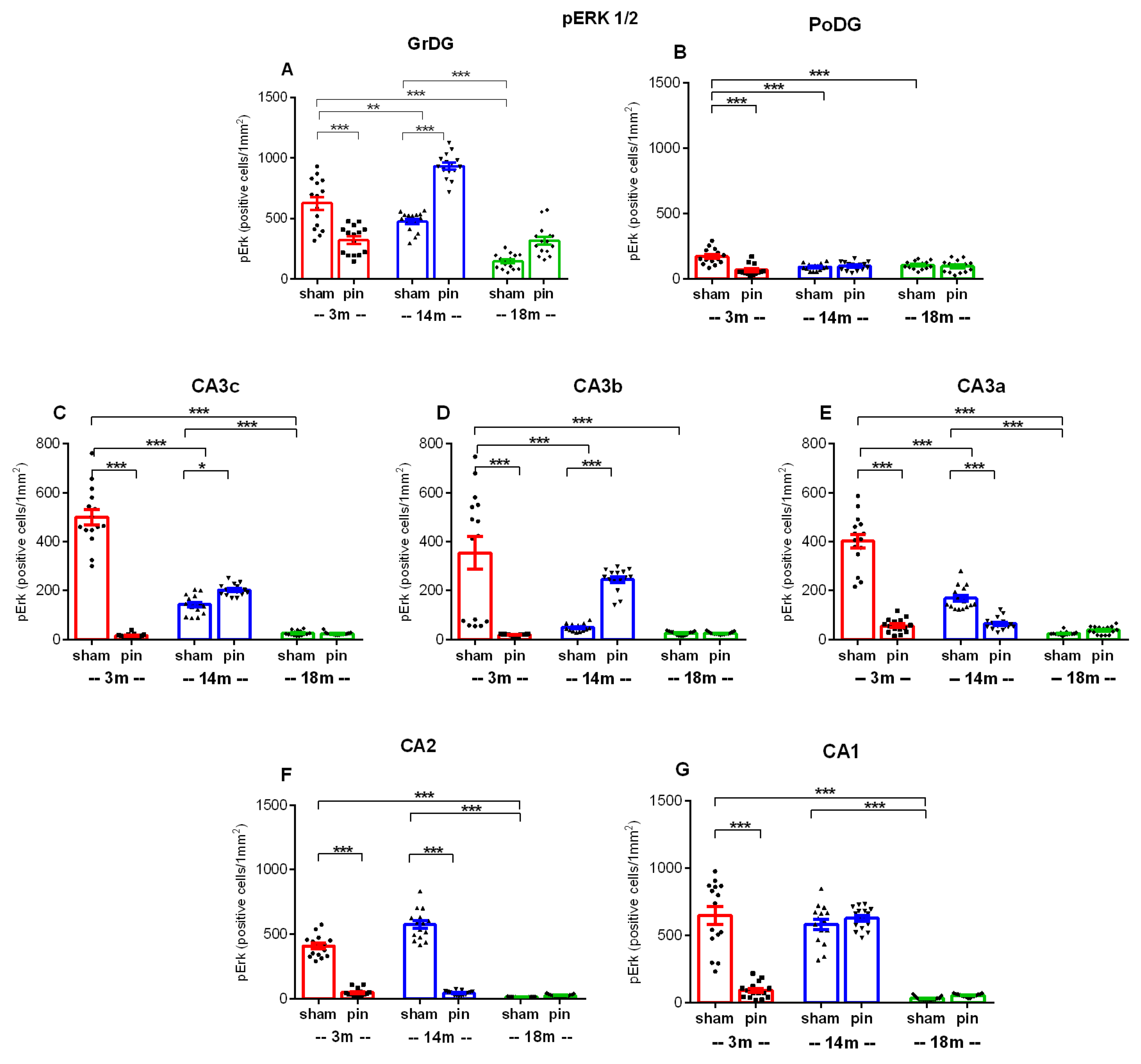
2.5. Melatonin Deficiency Induced by Pinealectomy Caused Age- and Region-Specific Changes in the Expression of ERK1/2 and Its Phosphorylated form (pERK1/2) in the DG and Dorsal Hippocampus
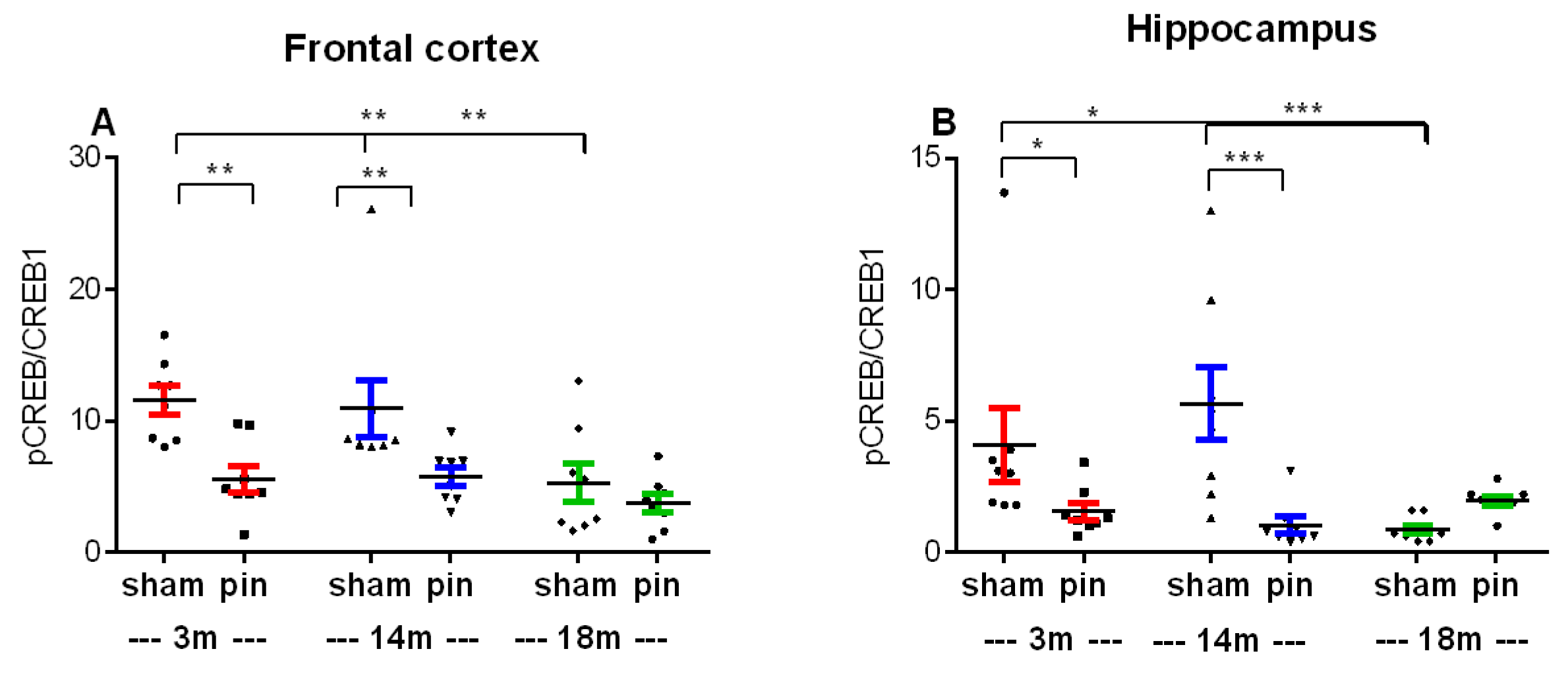
2.6. Melatonin Deficiency Associated with Pinealectomy Reduced the pCREB/CREB Ratio in the Frontal Cortex and Hippocampus in an Age-Dependent Manner
3. Discussion
3.1. The role of ERK/CREB/BDNF Pathway in the Hippocampus in Aging-Related Cognitive Decline
3.2. Effect of Pinealetomy on Aging-Related Changes in BDNF, TrkB, ERK, pERK, CREB and pCREB Expression in the Hippocampus
3.3. Limitation of the Study
Materials and Methods
4.1. Animals
4.2. Surgical Procedure
4.3. Memory Tests
4.3.1. Y-Maze Test
4.3.2. Object Recognition Test (ORT)
4.3.3. Radial Arm Maze (RAM)
4.4. Е. nzyme-Linked Immunosorbent Assay (ELISA)
4.5. Immunohistochemistry
4.5.1. BDNF Expression in the Hippocampus
Quantitative analysis of the intensity of immunohistochemical reactions
4.6.2. ERK1/2 and pERK1/2 Expression in the Hippocampus
Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Weaver Cargin, J.; Maruff, P.; Collie, A.; Masters, C. Mild Memory Impairment in Healthy Older Adults Is Distinct from Normal Aging. Brain and Cognition 2006, 60, 146–155. [CrossRef]
- Grundman, M. Mild Cognitive Impairment Can Be Distinguished From Alzheimer Disease and Normal Aging for Clinical Trials. Arch Neurol 2004, 61, 59. [CrossRef]
- Finger, C.E.; Moreno-Gonzalez, I.; Gutierrez, A.; Moruno-Manchon, J.F.; McCullough, L.D. Age-related immune alterations and cerebrovascular inflammation. Mol Psychiatry 2022, 27, 803-818. [CrossRef]
- Xu, J.; Wang, L.-L.; Dammer, E.B.; Li, C.-B.; Xu, G.; Chen, S.-D.; Wang, G. Melatonin for Sleep Disorders and Cognition in Dementia: A Meta-Analysis of Randomized Controlled Trials. Am J Alzheimers Dis Other Demen 2015, 30, 439–447. [CrossRef]
- Wu, Y.-H.; Feenstra, M.G.P.; Zhou, J.-N.; Liu, R.-Y.; Toranõ, J.S.; Van Kan, H.J.M.; Fischer, D.F.; Ravid, R.; Swaab, D.F. Molecular Changes Underlying Reduced Pineal Melatonin Levels in Alzheimer Disease: Alterations in Preclinical and Clinical Stages. The Journal of Clinical Endocrinology & Metabolism 2003, 88, 5898–5906. [CrossRef]
- Song, T.; Song, X.; Zhu, C.; Patrick, R.; Skurla, M.; Santangelo, I.; Green, M.; Harper, D.; Ren, B.; Forester, B.P.; et al. Mitochondrial Dysfunction, Oxidative Stress, Neuroinflammation, and Metabolic Alterations in the Progression of Alzheimer’s Disease: A Meta-Analysis of in Vivo Magnetic Resonance Spectroscopy Studies. Ageing Research Reviews 2021, 72, 101503. [CrossRef]
- Pandi-Perumal, S.R.; BaHammam, A.S.; Brown, G.M.; Spence, D.W.; Bharti, V.K.; Kaur, C.; Hardeland, R.; Cardinali, D.P. Melatonin Antioxidative Defense: Therapeutical Implications for Aging and Neurodegenerative Processes. Neurotox Res 2013, 23, 267–300. [CrossRef]
- Cho, J.H.; Bhutani, S.; Kim, C.H.; Irwin, M.R. Anti-Inflammatory Effects of Melatonin: A Systematic Review and Meta-Analysis of Clinical Trials. Brain, Behavior, and Immunity 2021, 93, 245–253. [CrossRef]
- Zhou, J.; Liu, R.; Kamphorst, W.; Hofman, M.A.; Swaab, D.F. Early Neuropathological Alzheimer’s Changes in Aged Individuals Are Accompanied by Decreased Cerebrospinal Fluid Melatonin Levels. Journal of Pineal Research 2003, 35, 125–130. [CrossRef]
- Priyadarshini, R.; Raj, G.M.; Shewade, D.G. Pathophysiological and pharmacological modulation of melatonergic system. Int J Basic Clin Pharmacol. 2015, 4, 32–639. [CrossRef]
- Pierpaoli, W.; Bulian, D. The Pineal Aging and Death Program: Life Prolongation in Pre-aging Pinealectomized Mice. Annals of the New York Academy of Sciences 2005, 1057, 133–144. [CrossRef]
- Alonso, M.; Medina, J.H.; Pozzo-Miller, L. ERK1/2 Activation Is Necessary for BDNF to Increase Dendritic Spine Density in Hippocampal CA1 Pyramidal Neurons. Learn. Mem. 2004, 11, 172–178. [CrossRef]
- Tartaglia, N.; Du, J.; Tyler, W.J.; Neale, E.; Pozzo-Miller, L.; Lu, B. Protein Synthesis-Dependent and -Independent Regulation of Hippocampal Synapses by Brain-Derived Neurotrophic Factor. Journal of Biological Chemistry 2001, 276, 37585–37593. [CrossRef]
- Peng, S.; Zhang, Y.; Zhang, J.; Wang, H.; Ren, B. ERK in Learning and Memory: A Review of Recent Research. IJMS 2010, 11, 222–232. [CrossRef]
- Medina, J.H.; Viola, H. ERK1/2: A Key Cellular Component for the Formation, Retrieval, Reconsolidation and Persistence of Memory. Front. Mol. Neurosci. 2018, 11, 361. [CrossRef]
- Josiane; Tatiani; Francielle; Michelle; Alexandra The Involvement of BDNF, NGF and GDNF in Aging and Alzheimer’s Disease. Aging and disease 2015, 6, 331. [CrossRef]
- Albert-Gascó, H.; Ros-Bernal, F.; Castillo-Gómez, E.; Olucha-Bordonau, F.E. MAP/ERK Signaling in Developing Cognitive and Emotional Function and Its Effect on Pathological and Neurodegenerative Processes. IJMS 2020, 21, 4471. [CrossRef]
- Zakaria, R.; Ahmad, A.H.; Othman, Z. The Potential Role of Melatonin on Memory Function: Lessons from Rodent Studies. Folia Biol (Praha) 2016, 62, 181–187.
- Tchekalarova, J.; Hrischev, P.; Ivanova, P.; Boyadjiev, N.; Georgieva, K. Metabolic Footprint in Young, Middle-Aged and Elderly Rats with Melatonin Deficit. Physiology & Behavior 2022, 250, 113786. [CrossRef]
- Tchekalarova, J.; Nenchovska, Z.; Kortenska, L.; Uzunova, V.; Georgieva, I.; Tzoneva, R. Impact of Melatonin Deficit on Emotional Status and Oxidative Stress-Induced Changes in Sphingomyelin and Cholesterol Level in Young Adult, Mature, and Aged Rats. IJMS 2022, 23, 2809. [CrossRef]
- Prieur, E.; Jadavji, N. Assessing Spatial Working Memory Using the Spontaneous Alternation Y-Maze Test in Aged Male Mice. BIO-PROTOCOL 2019, 9. [CrossRef]
- Bettio, L.E.B.; Rajendran, L.; Gil-Mohapel, J. The Effects of Aging in the Hippocampus and Cognitive Decline. Neuroscience & Biobehavioral Reviews 2017, 79, 66–86. [CrossRef]
- Leal, S.L.; Yassa, M.A. Neurocognitive Aging and the Hippocampus across Species. Trends in Neurosciences 2015, 38, 800–812. [CrossRef]
- Foster, T.C. Dissecting the Age-Related Decline on Spatial Learning and Memory Tasks in Rodent Models: N-Methyl-D-Aspartate Receptors and Voltage-Dependent Ca2+ Channels in Senescent Synaptic Plasticity. Progress in Neurobiology 2012, 96, 283–303. [CrossRef]
- Gerrard, J.L.; Burke, S.N.; McNaughton, B.L.; Barnes, C.A. Sequence Reactivation in the Hippocampus Is Impaired in Aged Rats. J. Neurosci. 2008, 28, 7883–7890. [CrossRef]
- Smith, B.M.; Yao, X.; Chen, K.S.; Kirby, E.D. A Larger Social Network Enhances Novel Object Location Memory and Reduces Hippocampal Microgliosis in Aged Mice. Front. Aging Neurosci. 2018, 10, 142. [CrossRef]
- Scali, C.; Giovannini, M.G.; Prosperi, C.; Bartolini, L.; Pepeu, G. TACRINE ADMINISTRATION ENHANCES EXTRACELLULAR ACETYLCHOLINEIN VIVOAND RESTORES THE COGNITIVE IMPAIRMENT IN AGED RATS. Pharmacological Research 1997, 36, 463–469. [CrossRef]
- The American Association for Research into Nervous and Mental Diseases; Albert, M.S. The Ageing Brain: Normal and Abnormal Memory. Phil. Trans. R. Soc. Lond. B 1997, 352, 1703–1709. [CrossRef]
- Haider, S.; Saleem, S.; Perveen, T.; Tabassum, S.; Batool, Z.; Sadir, S.; Liaquat, L.; Madiha, S. Age-Related Learning and Memory Deficits in Rats: Role of Altered Brain Neurotransmitters, Acetylcholinesterase Activity and Changes in Antioxidant Defense System. AGE 2014, 36, 9653. [CrossRef]
- Stone, W.S.; Rudd, R.J.; Parsons, M.W.; Gold, P.E. Memory Scores in Middle-Aged Rats Predict Later Deficits in Memory, Paradoxical Sleep, and Blood Glucose Regulation in Old Age. Experimental Aging Research 1997, 23, 287–300. [CrossRef]
- Shekari, A.; Fahnestock, M. Retrograde Axonal Transport of BDNF and ProNGF Diminishes with Age in Basal Forebrain Cholinergic Neurons. Neurobiology of Aging 2019, 84, 131–140. [CrossRef]
- Countryman, R.A.; Gold, P.E. Rapid Forgetting of Social Transmission of Food Preferences in Aged Rats: Relationship to Hippocampal CREB Activation. Learn. Mem. 2007, 14, 350–358. [CrossRef]
- Morris, K.A.; Gold, P.E. Age-Related Impairments in Memory and in CREB and PCREB Expression in Hippocampus and Amygdala Following Inhibitory Avoidance Training. Mechanisms of Ageing and Development 2012, 133, 291–299. [CrossRef]
- Gallagher, M. Effects of Aging on the Hippocampal Formation in a Naturally Occurring Animal Model of Mild Cognitive Impairment. Experimental Gerontology 2003, 38, 71–77. [CrossRef]
- Rasmussen, T.; Schliemann, T.; Sørensen, J.C.; Zimmer, J.; West, M.J. Memory Impaired Aged Rats: No Loss of Principal Hippocampal and Subicular Neurons. Neurobiology of Aging 1996, 17, 143–147. [CrossRef]
- Tchekalarova, J.; Krushovlieva, D.; Ivanova, P.; Nenchovska, Z.; Toteva, G.; Atanasova, M. The Role of Melatonin Deficiency Induced by Pinealectomy on Motor Activity and Anxiety Responses in Young Adult, Middle-Aged and Old Rats. Behav Brain Funct 2024, 20, 3. [CrossRef]
- Pierpaoli, W. The Pineal Gland: A Circadian or Seasonal Aging Clock? Aging Clin Exp Res 1991, 3, 99–101. [CrossRef]
- Pierpaoli, W. The Pineal Gland as Ontogenetic Scanner of Reproduction, Immunity, and Aging The Aging Clock. Ann NY Acad Sci 1994, 741, 46–49. [CrossRef]
- Pierpaoli, W.; Bulian, D. The Pineal Aging and Death Program. I. Grafting of Old Pineals in Young Mice Accelerates Their Aging. Journal of Anti-Aging Medicine 2001, 4, 31–37. [CrossRef]
- Pierpaoli, W.; Bulian, D. The Pineal Aging and Death Program: Life Prolongation in Pre-aging Pinealectomized Mice. Annals of the New York Academy of Sciences 2005, 1057, 133–144. [CrossRef]
- Wurtman, R.J. Age-Related Decreases in Melatonin Secretion—Clinical Consequences. The Journal of Clinical Endocrinology & Metabolism 2000, 85, 2135–2136. [CrossRef]
- Cecon, E.; Oishi, A.; Jockers, R. Melatonin Receptors: Molecular Pharmacology and Signalling in the Context of System Bias. British J Pharmacology 2018, 175, 3263–3280. [CrossRef]
- De Butte, M.; Fortin, T.; Pappas, B.A. Pinealectomy: Behavioral and Neuropathological Consequences in a Chronic Cerebral Hypoperfusion Model. Neurobiology of Aging 2002, 23, 309–317. [CrossRef]
- Tzoneva, R.; Georgieva, I.; Ivanova, N.; Uzunova, V.; Nenchovska, Z.; Apostolova, S.;Stoyanova, T.; Tchekalarova, J. The Role of Melatonin on Behavioral Changes and Concomitant Oxidative Stress in icvA_1-42 Rat Model with Pinealectomy. Int. J. Mol. Sci. 2021, 22, 12763. [CrossRef]
- Karakaş, A.; Coşkun, H.; Kaya, A. The Effects of Pinealectomy, Melatonin Injections and Implants on the Spatial Memory Performance of Male Wistar Rats. Biological Rhythm Research 2011, 42, 457–472. [CrossRef]
- Tchekalarova, J.; Kortenska, L.; Ivanova, N.; Atanasova, M.; Marinov, P. Agomelatine Treatment Corrects Impaired Sleep-Wake Cycle and Sleep Architecture and Increases MT1 Receptor as Well as BDNF Expression in the Hippocampus during the Subjective Light Phase of Rats Exposed to Chronic Constant Light. Psychopharmacology 2020, 237, 503–518. [CrossRef]
- Shin, E.-J.; Chung, Y.H.; Le, H.-L.T.; Jeong, J.H.; Dang, D.-K.; Nam, Y.; Wie, M.B.; Nah, S.-Y.; Nabeshima, Y.-I.; Nabeshima, T.; et al. Melatonin Attenuates Memory Impairment Induced by Klotho Gene Deficiency Via Interactive Signaling Between MT2 Receptor, ERK, and Nrf2-Related Antioxidant Potential. International Journal of Neuropsychopharmacology 2015, 18. [CrossRef]
- Dash, P.K.; Karl, K.A.; Colicos, M.A.; Prywes, R.; Kandel, E.R. CAMP Response Element-Binding Protein Is Activated by Ca2+/Calmodulin- as Well as CAMP-Dependent Protein Kinase. Proc. Natl. Acad. Sci. U.S.A. 1991, 88, 5061–5065. [CrossRef]
- Mihanfar, A.; Yousefi, B.; Azizzadeh, B. et al. Interactions of melatonin with various signaling pathways: implications for cancer therapy. Cancer Cell Int 2022, 22, 420. [CrossRef]
- Amini, H.; Rezabakhsh, A.; Heidarzadeh, M.; Hassanpour, M.; Hashemzadeh, S.; Ghaderi, S.; Sokullu, E.; Rahbarghazi, R.; Reiter, R.J. An Examination of the Putative Role of Melatonin in Exosome Biogenesis. Front. Cell Dev. Biol. 2021, 9, 686551. [CrossRef]
- Smith. N.K.; Hackett, T.A.; Galli, A.; Flynn, C.R. GLP-1: Molecular mechanisms and outcomes of a complex signaling system. Neurochem Int. 2019 128, 94-105. [CrossRef]
- Kim, M.-S.; Kim, B.-Y.; Kim, J.I.; Lee, J.; Jeon, W.K. Mumefural Improves Recognition Memory and Alters ERK-CREB-BDNF Signaling in a Mouse Model of Chronic Cerebral Hypoperfusion. Nutrients 2023, 15, 3271. [CrossRef]
- Yan, L.; Xu, X.; He, Z.; Wang, S.; Zhao, L.; Qiu, J.; Wang, D.; Gong, Z.; Qiu, X.; Huang, H. Antidepressant-Like Effects and Cognitive Enhancement of Coadministration of Chaihu Shugan San and Fluoxetine: Dependent on the BDNF-ERK-CREB Signaling Pathway in the Hippocampus and Frontal Cortex. Biomed Res Int. 2020, 2020, 2794263. [CrossRef]
- Pak, M.E.; Yang, H.J.; Li, W.; Kim, J.K.; Go, Y. Yuk-Gunja-Tang attenuates neuronal death and memory impairment via ERK/CREB/BDNF signaling in the hippocampi of experimental Alzheimer’s disease model. Front. Pharmacol. 2022, 13, 1014840. [CrossRef]
- Hoffman, R.A., Reiter, R.J. Rapid pinealectomy in hamsters and other small rodents. Anat Rec. 1965, 153, 19-21. [CrossRef]
- Tchekalarova, J.; Nenchovska, Z.; Atanasova, D.; Atanasova, M.; Kortenska, L.; Stefanova, M.; Alova, L.; Lazarov, N. Consequences of Long-Term Treatment with Agomelatine on Depressive-like Behavior and Neurobiological Abnormalities in Pinealectomized Rats. Behavioural Brain Research 2016, 302, 11–28. [CrossRef]
- Shishmanova-Doseva, M.; Tchekalarova, J.; Nenchovska, Z.; Ivanova, N.; Georgieva, K.; Peychev, L. The Effect of Chronic Treatment with Lacosamide and Topiramate on Cognitive Functions and Impaired Emotional Responses in a Pilocarpine-induced Post-status Epilepticus Rat Model. Folia Medica 2020, 62, 723-9. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



