Submitted:
05 July 2024
Posted:
08 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Study Protocol
2.3. Hematology Blood Parameteres
2.4. Thrombin Generation
2.5. Platelet Aggregation
2.6. Statistical Analysis
3. Results
3.1. Study Population
3.2. Effects of Volanesorsen on Platelet Count
3.3. Thrombin Generation Analysis
3.4. Platelet Aggregation Analysis
4. Discussion
5. Conclusions
Data Availability Statement:. Not applicable.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Moulin, P.; Dufour, R.; Averna, M.; Arca, M.; Cefalu, A.B.; Noto, D.; D’Erasmo, L.; Di Costanzo, A.; Marcais, C.; Alvarez-Sala Walther, L.A. , et al. Identification and diagnosis of patients with familial chylomicronaemia syndrome (FCS): Expert panel recommendations and proposal of an “FCS score”. Atherosclerosis 2018, 275, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Ooi, E.M.; Barrett, P.H.; Chan, D.C.; Watts, G.F. Apolipoprotein C-III: understanding an emerging cardiovascular risk factor. Clinical science 2008, 114, 611–624. [Google Scholar] [CrossRef] [PubMed]
- Gouni-Berthold, I. The role of antisense oligonucleotide therapy against apolipoprotein-CIII in hypertriglyceridemia. Atherosclerosis. Supplements 2017, 30, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Warden, B.A.; Duell, P.B. Volanesorsen for treatment of patients with familial chylomicronemia syndrome. Drugs of today 2018, 54, 721–735. [Google Scholar] [CrossRef]
- Calcaterra, I.; Lupoli, R.; Di Minno, A.; Di Minno, M.N.D. Volanesorsen to treat severe hypertriglyceridaemia: A pooled analysis of randomized controlled trials. Eur J Clin Invest 2022, 52, e13841. [Google Scholar] [CrossRef]
- Gaudet, D.; Brisson, D.; Tremblay, K.; Alexander, V.J.; Singleton, W.; Hughes, S.G.; Geary, R.S.; Baker, B.F.; Graham, M.J.; Crooke, R.M. , et al. Targeting APOC3 in the familial chylomicronemia syndrome. The New England journal of medicine 2014, 371, 2200–2206. [Google Scholar] [CrossRef]
- L. D’Erasmo, S.D.F., A. Gelrud, A. Digenio, V.J. Alexander, A. Hsieh, I. Gouni-Berthold, E. Bruckert,E. Stroes, R.S. Gear y, S.G. Hughes, D. Gaudet. TREATMENT WITH VOLANESORSEN (VLN) REDUCED TRIGLYCERIDES AND PANCREATITIS IN PATIENTS WITH FAMILIAL CHYLOMICRONEMIA SYNDROME (FCS) AND SEVERE HYPERTRIGLYCERIDEMIA (SHTG) VS PLACEBO: RESULTS OF THE APPROACH AND COMPASS STUDIES. Giornale Italiano dell’arteriosclerosi 2019, 4, 100. [Google Scholar]
- Macchi, C.; Sirtori, C.R.; Corsini, A.; Santos, R.D.; Watts, G.F.; Ruscica, M. A new dawn for managing dyslipidemias: The era of rna-based therapies. Pharmacological research 2019, 150, 104413. [Google Scholar] [CrossRef]
- Paik, J.; Duggan, S. Volanesorsen: First Global Approval. Drugs 2019, 79, 1349–1354. [Google Scholar] [CrossRef]
- Witztum, J.L.; Gaudet, D.; Freedman, S.D.; Alexander, V.J.; Digenio, A.; Williams, K.R.; Yang, Q.; Hughes, S.G.; Geary, R.S.; Arca, M. , et al. Volanesorsen and Triglyceride Levels in Familial Chylomicronemia Syndrome. The New England journal of medicine 2019, 381, 531–542. [Google Scholar] [CrossRef]
- Gaudet, D.B. , Alexis & Tremblay, Karine & Brisson, Diane & Laflamme, Nathalie & Paquette, Martine & Dufour, Robert & Bergeron, Jean. Natural History (up to 15 years) of Platelet Count in 84 Patients with Familial Hyperchylomicronemia Due to Lipoprotein Lipase Deficiency. Journal of clinical lipidology 2017, 11, 797–798. [Google Scholar] [CrossRef]
- Tripodi, A. Thrombin Generation Assay and Its Application in the Clinical Laboratory. Clin Chem 2016, 62, 699–707. [Google Scholar] [CrossRef]
- Born, G.V. Aggregation of blood platelets by adenosine diphosphate and its reversal. Nature 1962, 194, 927–929. [Google Scholar] [CrossRef]
- Bennett, C.F. Therapeutic Antisense Oligonucleotides Are Coming of Age. Annu Rev Med 2019, 70, 307–321. [Google Scholar] [CrossRef]
- Larouche, M.; Brisson, D.; Morissette, M.C.; Gaudet, D. Post-prandial analysis of fluctuations in the platelet count and platelet function in patients with the familial chylomicronemia syndrome. Orphanet J Rare Dis 2023, 18, 167. [Google Scholar] [CrossRef]
- Mead, J.R.; Irvine, S.A.; Ramji, D.P. Lipoprotein lipase: structure, function, regulation, and role in disease. J Mol Med (Berl) 2002, 80, 753–769. [Google Scholar] [CrossRef]
- Ofosu, F.A.; Modi, G.J.; Smith, L.M.; Cerskus, A.L.; Hirsh, J.; Blajchman, M.A. Heparan sulfate and dermatan sulfate inhibit the generation of thrombin activity in plasma by complementary pathways. Blood 1984, 64, 742–747. [Google Scholar] [CrossRef]
- Vogtle, T.; Sharma, S.; Mori, J.; Nagy, Z.; Semeniak, D.; Scandola, C.; Geer, M.J.; Smith, C.W.; Lane, J.; Pollack, S. , et al. Heparan sulfates are critical regulators of the inhibitory megakaryocyte-platelet receptor G6b-B. Elife 2019, 8. [Google Scholar] [CrossRef]
- Lutz, E.P.; Merkel, M.; Kako, Y.; Melford, K.; Radner, H.; Breslow, J.L.; Bensadoun, A.; Goldberg, I.J. Heparin-binding defective lipoprotein lipase is unstable and causes abnormalities in lipid delivery to tissues. The Journal of clinical investigation 2001, 107, 1183–1192. [Google Scholar] [CrossRef]
- Haubenwallner, S.; Horl, G.; Shachter, N.S.; Presta, E.; Fried, S.K.; Hofler, G.; Kostner, G.M.; Breslow, J.L.; Zechner, R. A novel missense mutation in the gene for lipoprotein lipase resulting in a highly conservative amino acid substitution (Asp180-->Glu) causes familial chylomicronemia (type I hyperlipoproteinemia). Genomics 1993, 18, 392–396. [Google Scholar] [CrossRef]
- Lookene, A.; Beckstead, J.A.; Nilsson, S.; Olivecrona, G.; Ryan, R.O. Apolipoprotein A-V-heparin interactions: implications for plasma lipoprotein metabolism. J Biol Chem 2005, 280, 25383–25387. [Google Scholar] [CrossRef] [PubMed]
- Aoki, I.; Aoki, N.; Kawano, K.; Shimoyama, K.; Maki, A.; Homori, M.; Yanagisawa, A.; Yamamoto, M.; Kawai, Y.; Ishikawa, K. Platelet-dependent thrombin generation in patients with hyperlipidemia. J Am Coll Cardiol 1997, 30, 91–96. [Google Scholar] [CrossRef] [PubMed]
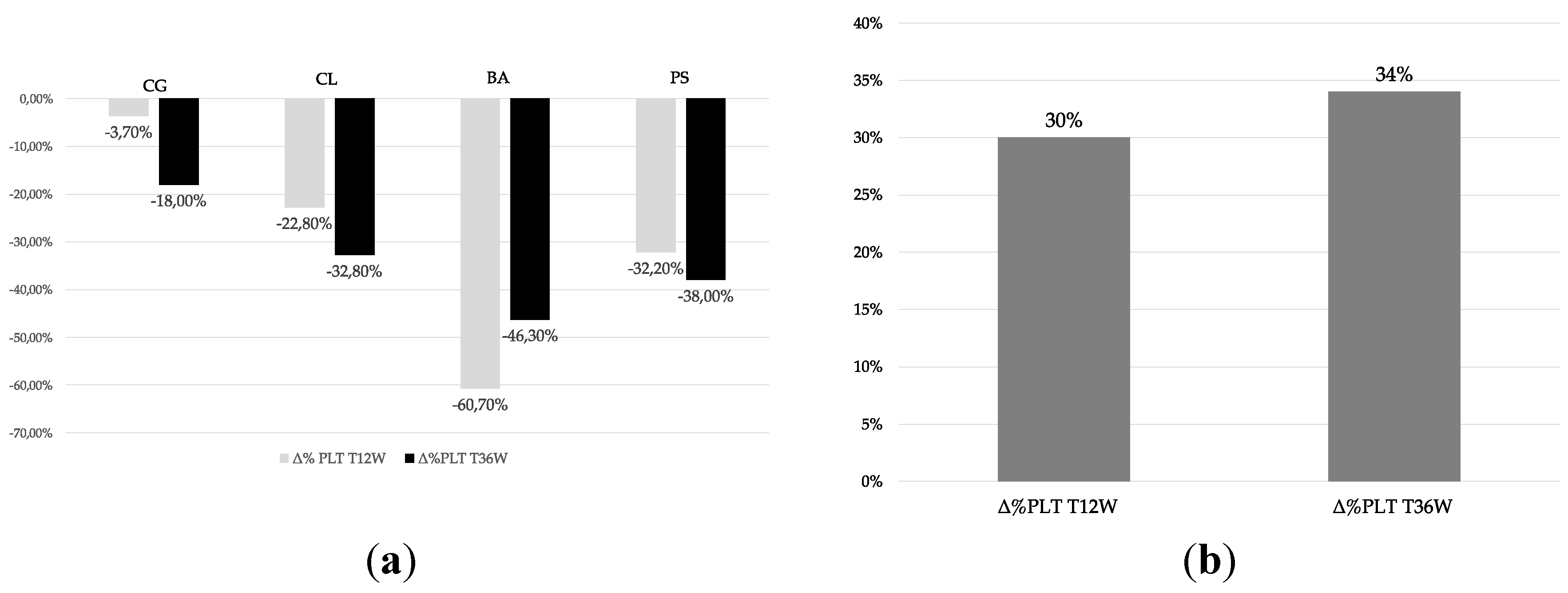
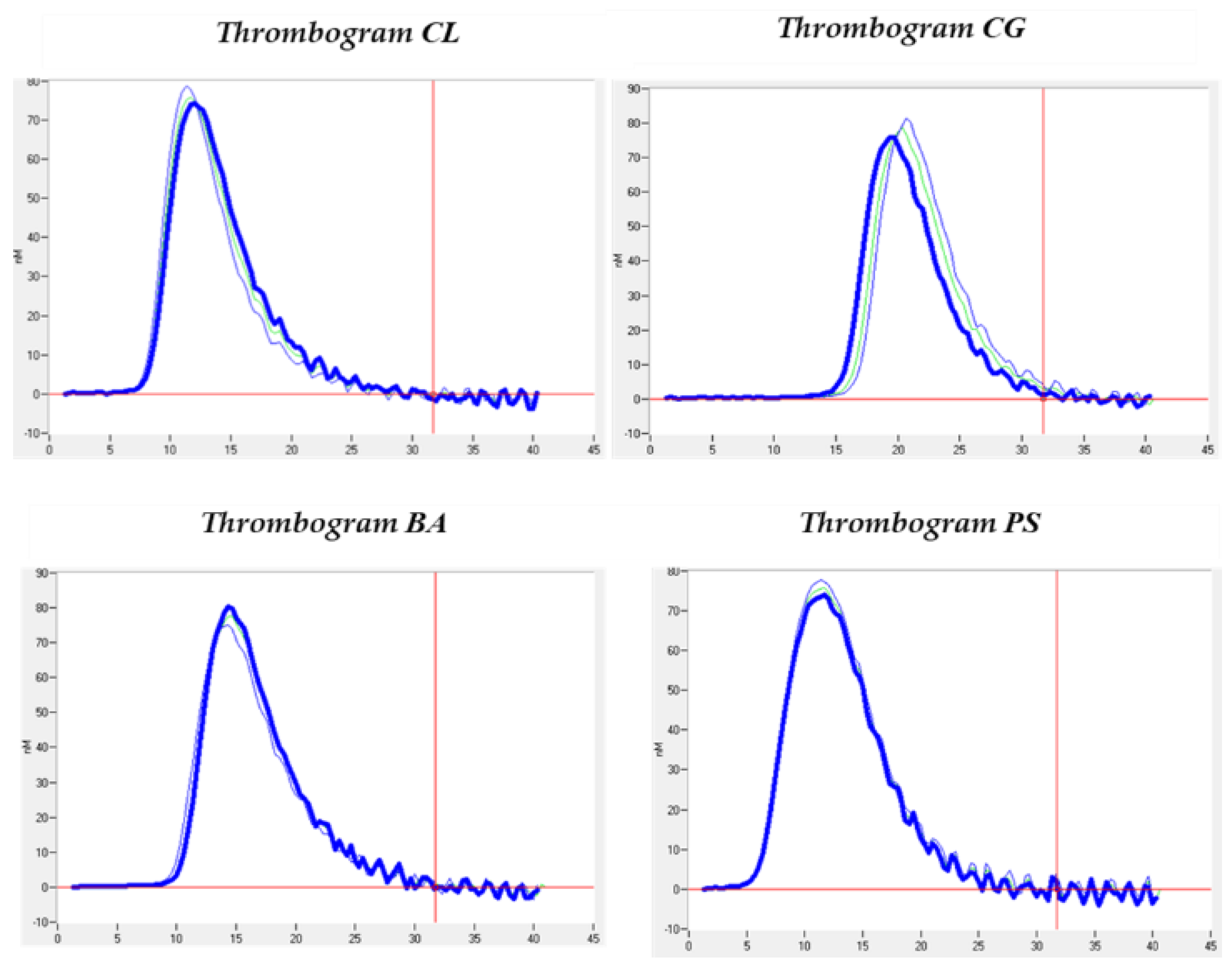
| Patients | CG | CL | BA | PS | Mean ± SD |
|---|---|---|---|---|---|
| Age | 62 | 71 | 53 | 40 | 56.5 ± 13.2 |
| Sex (M=1) | 0 | 1 | 1 | 0 | N.A. |
| BMI (kg/m2) | 20.8 | 22 | 24.8 | 20.8 | 22.1 ± 1.9 |
| Age at diagnosis | 20 | 22 | 6 | 4 | 13 ± 9.3 |
| Pathogenic variants | LPL c.844G>T (p.Glu282*) - stop gained |
LPL c.844G>T (p.Glu282*) - stop gained |
LPL c.621C>G (p.Asp207Glu) - missense |
APOA5 c.427delC p.(Arg143Alafs*57) c.49+5G>A |
|
| Previous pancreatitis | 4 | 10 | 20 | 3 | 9.25 |
| Pancreatitis in last 5 years | 1 | 2 | 10 | 2 | 3.75 |
| Chronic pancreatitis (Yes=1) | 0 | 1 | 1 | 0 | 50% |
| Diabetes | 1 | 1 | 0 | 1 | 75% |
| Treatment (fibrates, PUFA and MCT oil) | Gemfibrozil 600 mg bid, PUFA (6 gr), MCT oil | Fenofibrate 145 mg/daly, PUFA (6 gr), MCT oil | Fenofibrate 145 mg/daly, PUFA (6 gr) MCT oil | Fenofibrate 145 mg/daly, PUFA (6 gr) MCT oil |
-- |
| Parameter | FCS Patients | Controls | p-value |
| LagTime | 10.42 ± 4.40 | 9.25 ± 0.99 | 0.685 |
| ETP | 599.80 ± 73.47 | 900.2 ± 210.99 | 0.102 |
| Peak | 76.84 ± 1.07 | 123.30 ± 39.45 | 0.104 |
| ttPeak | 14.33 ± 4.01 | 13.10 ± 0.67 | 0.620 |
| StartTail | 32.13 ± 3.54 | 29.46 ± 1.69 | 0.143 |
| Velocity Index | 20.21 ± 3.63 | 33.05 ± 13.21 | 0.149 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




