Submitted:
27 June 2024
Posted:
05 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Design and Sample Selection
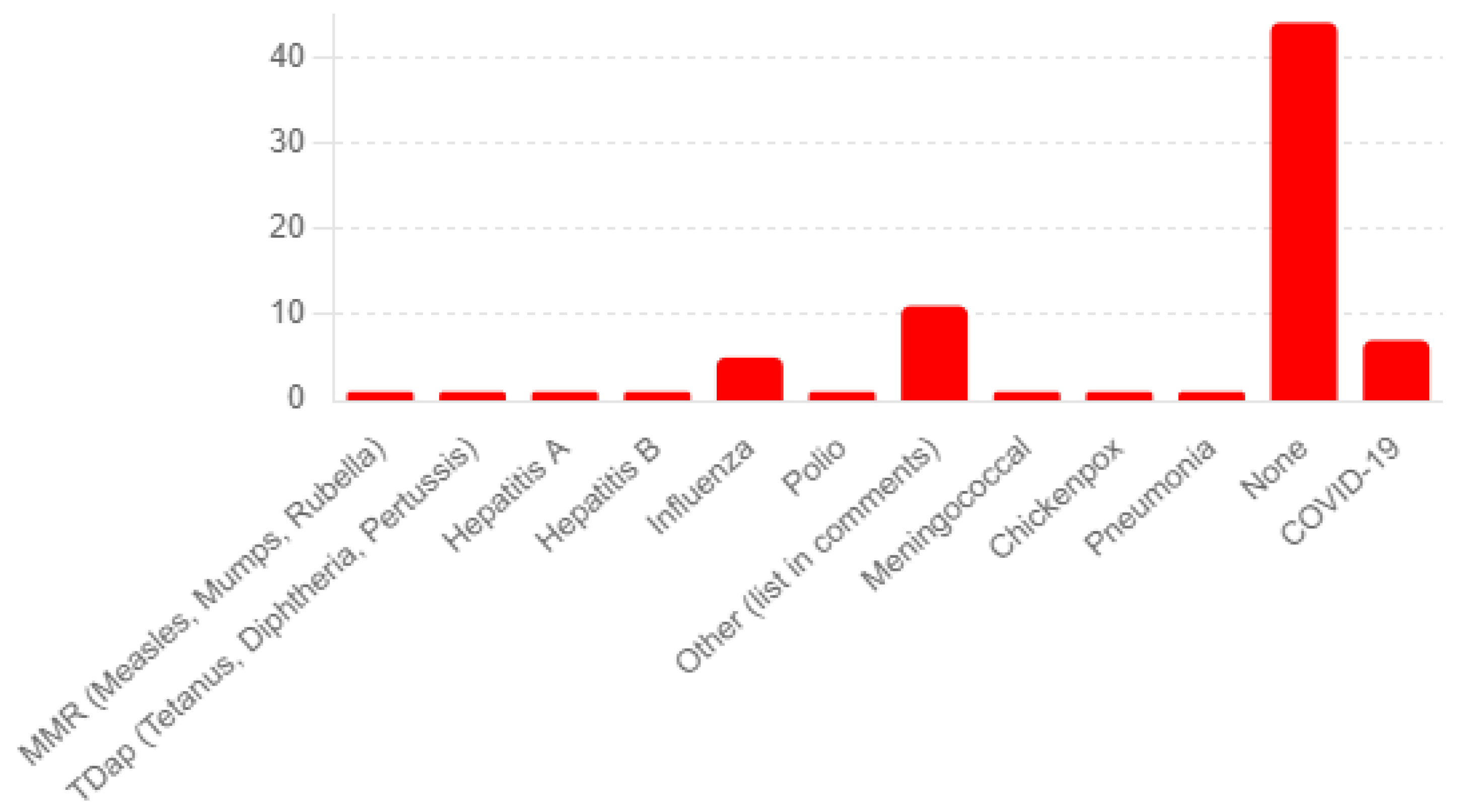
2.2. Questionnaire on Adverse Events following Shingrix Immunization
2.3. Study Period
2.4. Statistical Analysis
3. Results
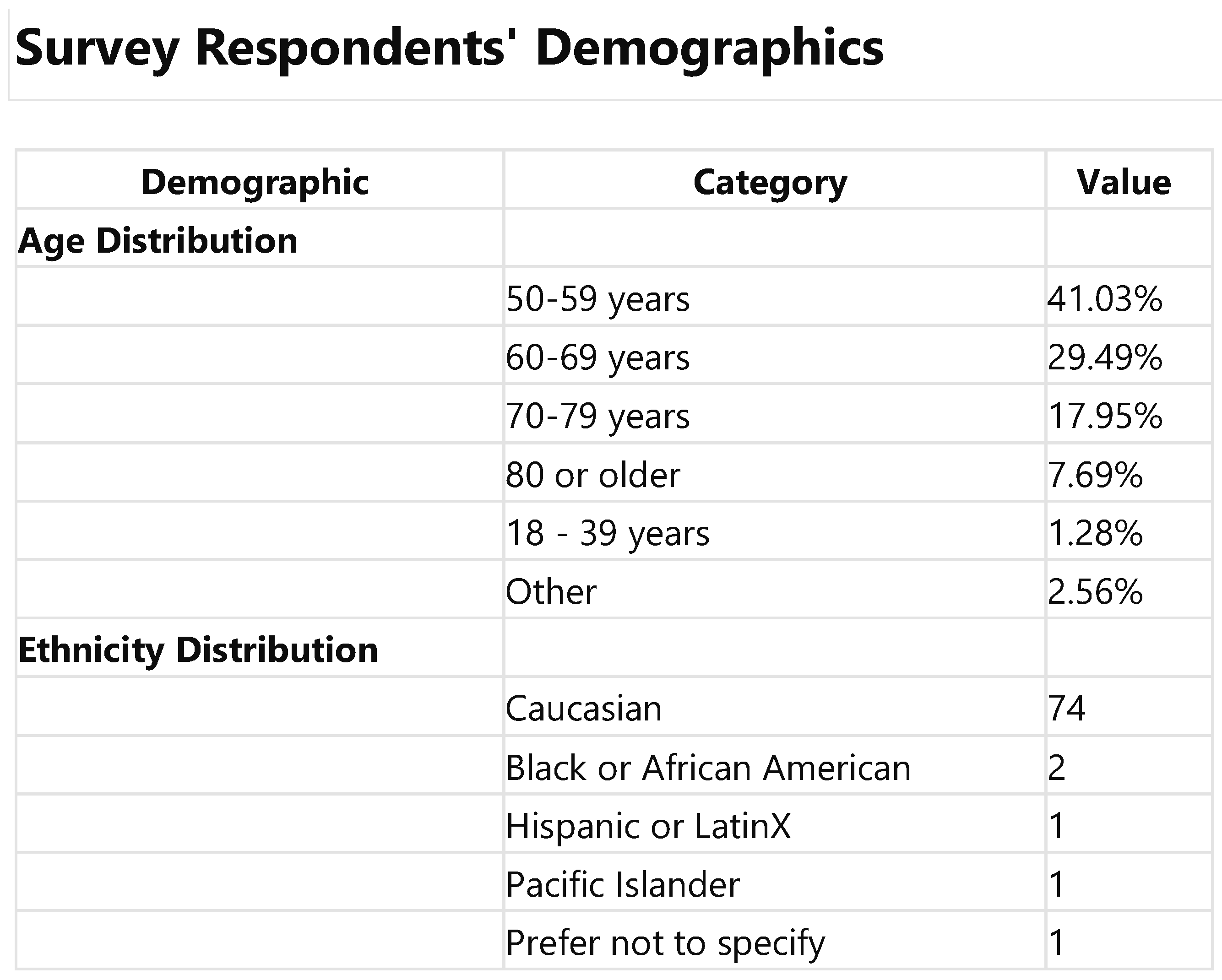
3.1. Demographic Analysis
3.2. Health History Prior to the Shingles Vaccination
3.3. Shingrix Vaccine Administration
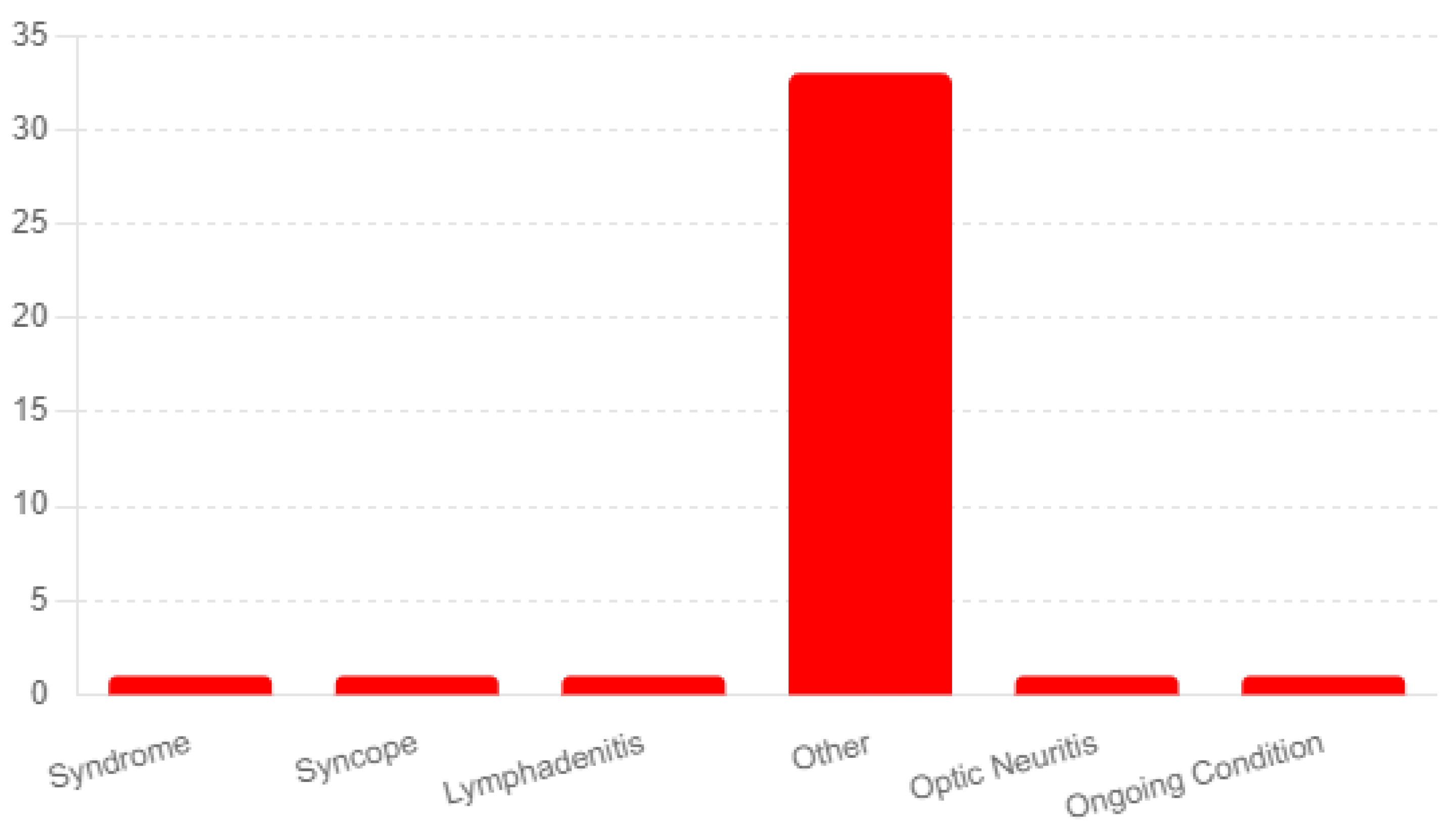
3.4. Shingrix Vaccination Side-Effects and Their Severity
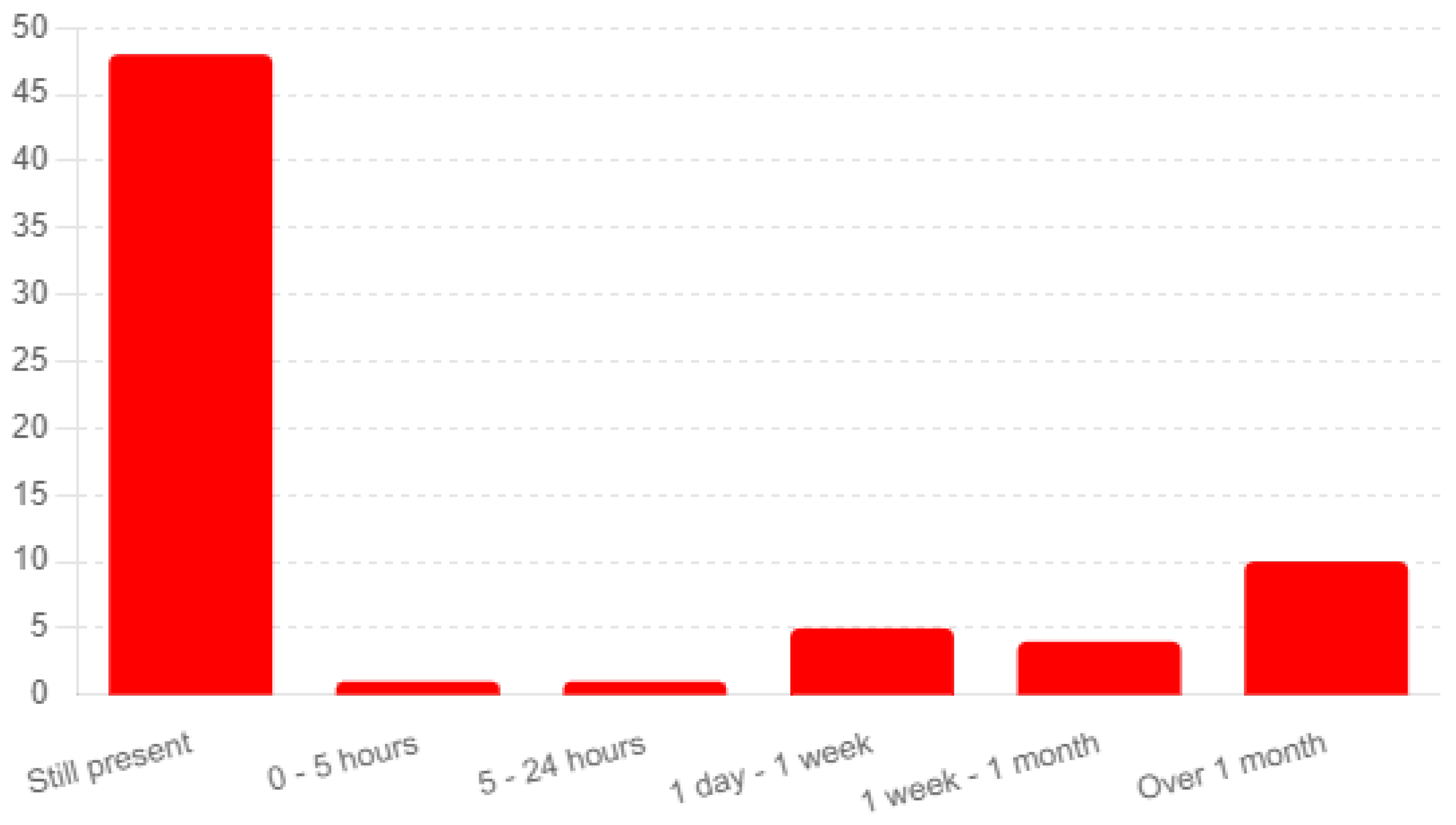
3.5. Long-term Shingrix Vaccination Side-Effects and Their Severity
3.6. Treatment Effectiveness for Shingrix Vaccination Side-Effects
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- CDC. Shingles Symptoms and Complications. Shingles (Herpes Zoster). Published May 14, 2024. Accessed June 18, 2024. https://www.cdc.gov/shingles/signs-symptoms/index.html.
- Viruses | Free Full-Text | Herpes zoster: A Review of Clinical Manifestations and Management. Accessed June 18, 2024. https://www.mdpi.com/1999-4915/14/2/192.
- Johnson RW, Alvarez-Pasquin MJ, Bijl M, et al. Herpes zoster epidemiology, management, and disease and economic burden in Europe: a multidisciplinary perspective. Ther Adv Vaccines. 2015;3(4):109-120. [CrossRef]
- Gonzalez Chiappe S, Sarazin M, Turbelin C, et al. Herpes zoster: Burden of disease in France. Vaccine. 2010;28(50):7933-7938. [CrossRef]
- Yawn BP, Itzler RF, Wollan PC, Pellissier JM, Sy LS, Saddier P. Health Care Utilization and Cost Burden of Herpes Zoster in a Community Population. Mayo Clin Proc. 2009;84(9):787-794.
- Anderson, TC. Use of Recombinant Zoster Vaccine in Immunocompromised Adults Aged ⪰19 Years: Recommendations of the Advisory Committee on Immunization Practices — United States, 2022. MMWR Morb Mortal Wkly Rep. 2022;71. [CrossRef]
- Xia Y, Zhang X, Zhang L, Fu C. Efficacy, effectiveness, and safety of herpes zoster vaccine in the immunocompetent and im-munocompromised subjects: A systematic review and network meta-analysis. Front Immunol. 2022;13:978203. [CrossRef]
- Maltz F, Fidler B. Shingrix: A New Herpes Zoster Vaccine. P T. 2019;44(7):406-433.
- Safety Information for Shingles (Herpes Zoster) Vaccines | Vaccine Safety | CDC. Published December 22, 2022. Accessed June 18, 2024. https://www.cdc.gov/vaccinesafety/vaccines/shingles-herpes-vaccine.html.
- Heydari-Kamjani M, Vante I, Uppal P, Demory Beckler M, Kesselman MM. Uveitis Sarcoidosis Presumably Initiated After Administration of Shingrix Vaccine. Cureus. 11(6):e4920. [CrossRef]
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




