Submitted:
03 July 2024
Posted:
04 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Motor Function
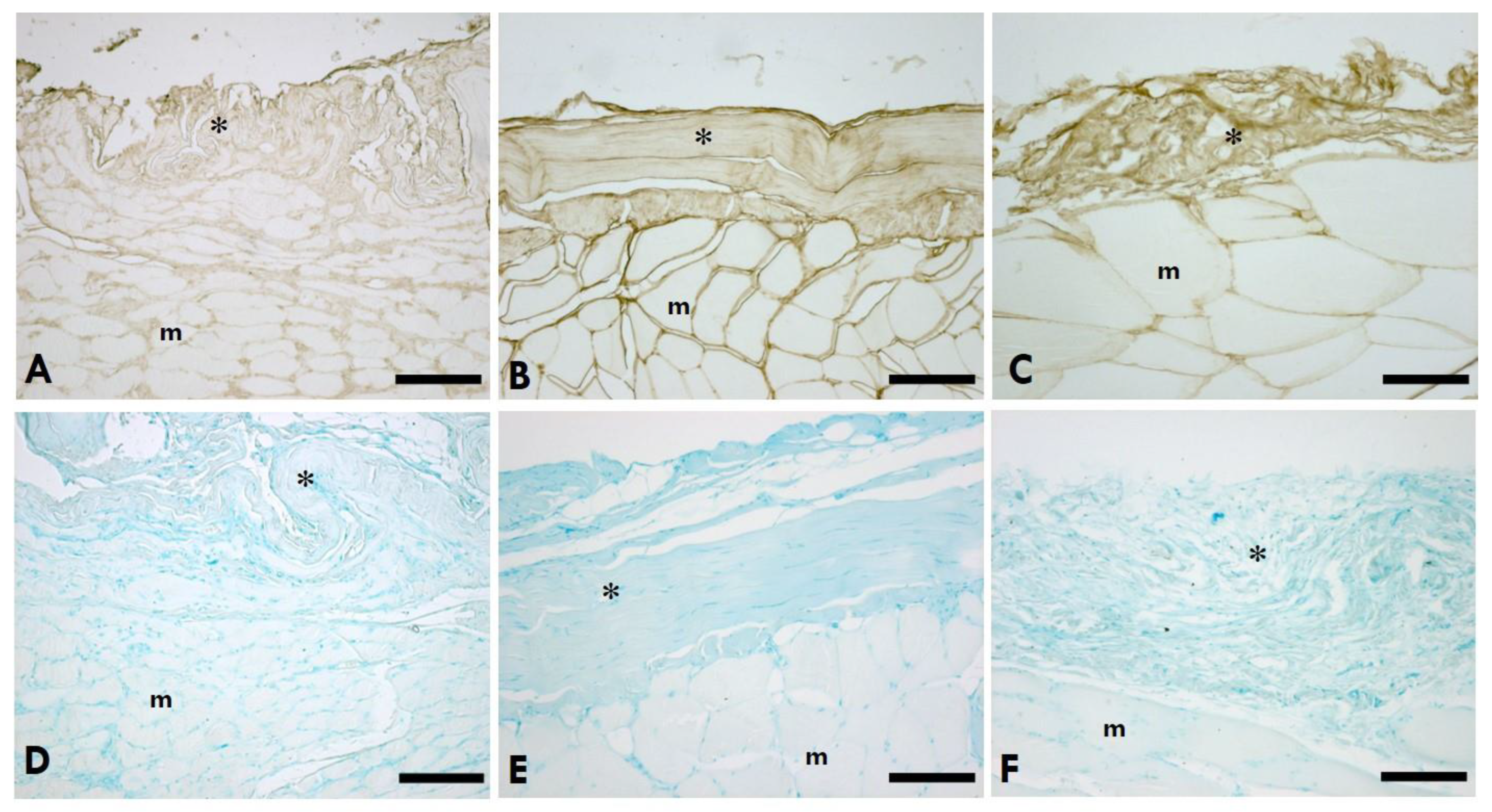
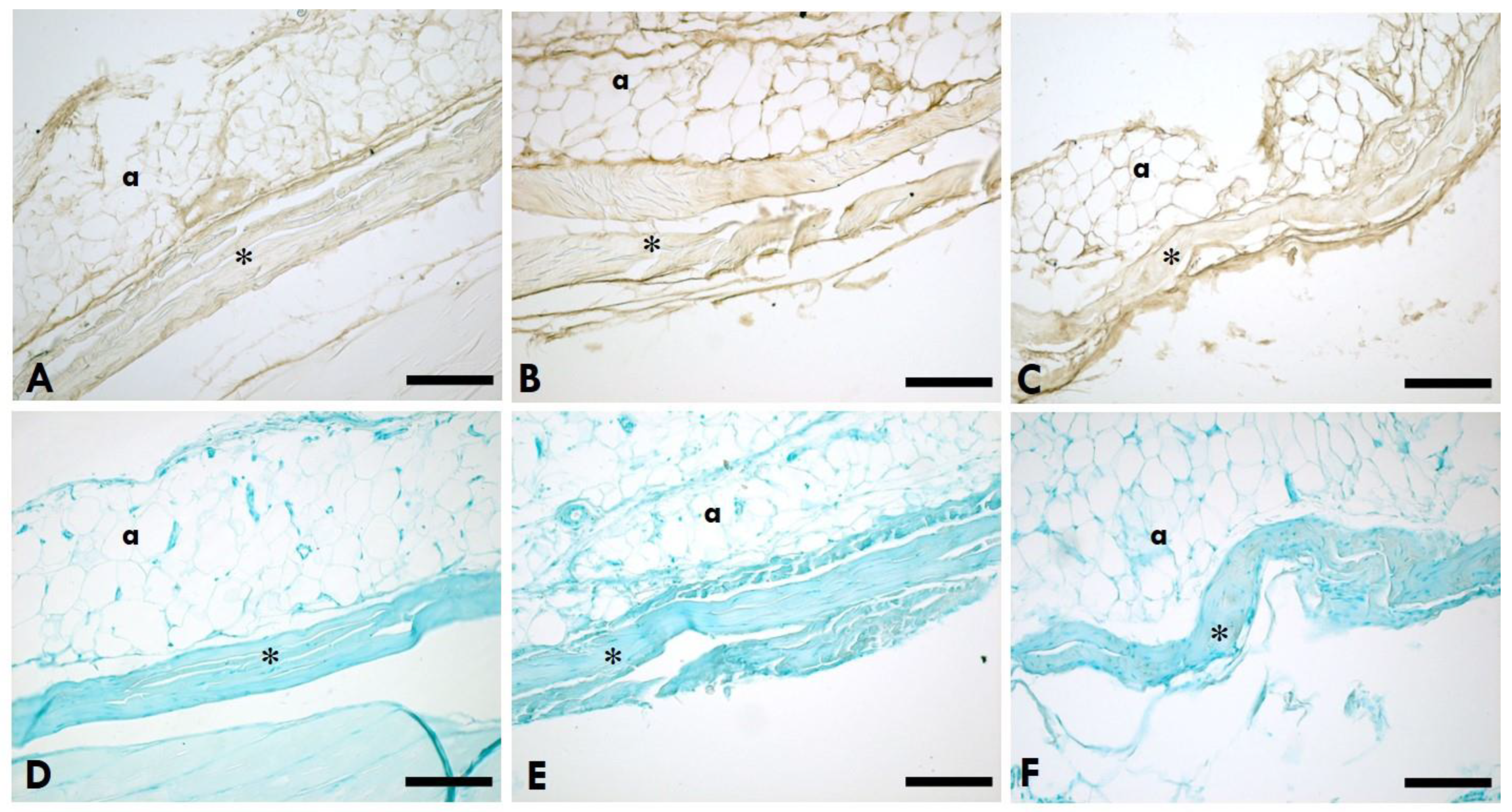
2.2. Distribution of HA
2.3. Distribution of Collagen
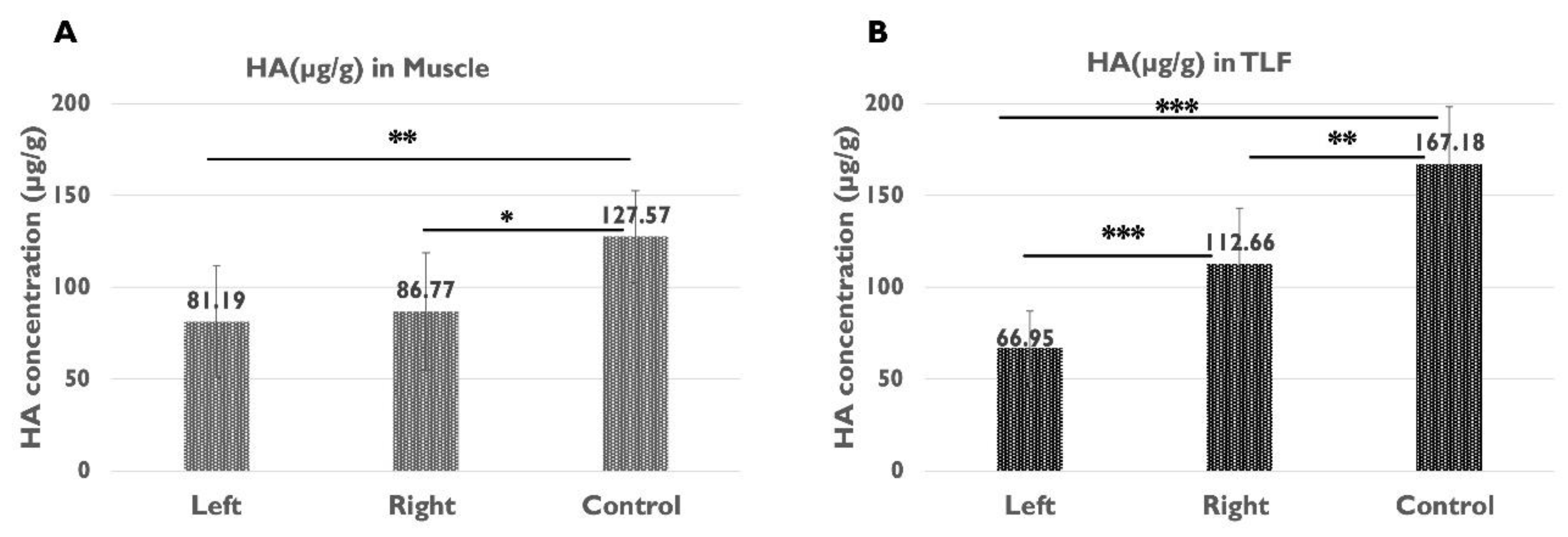
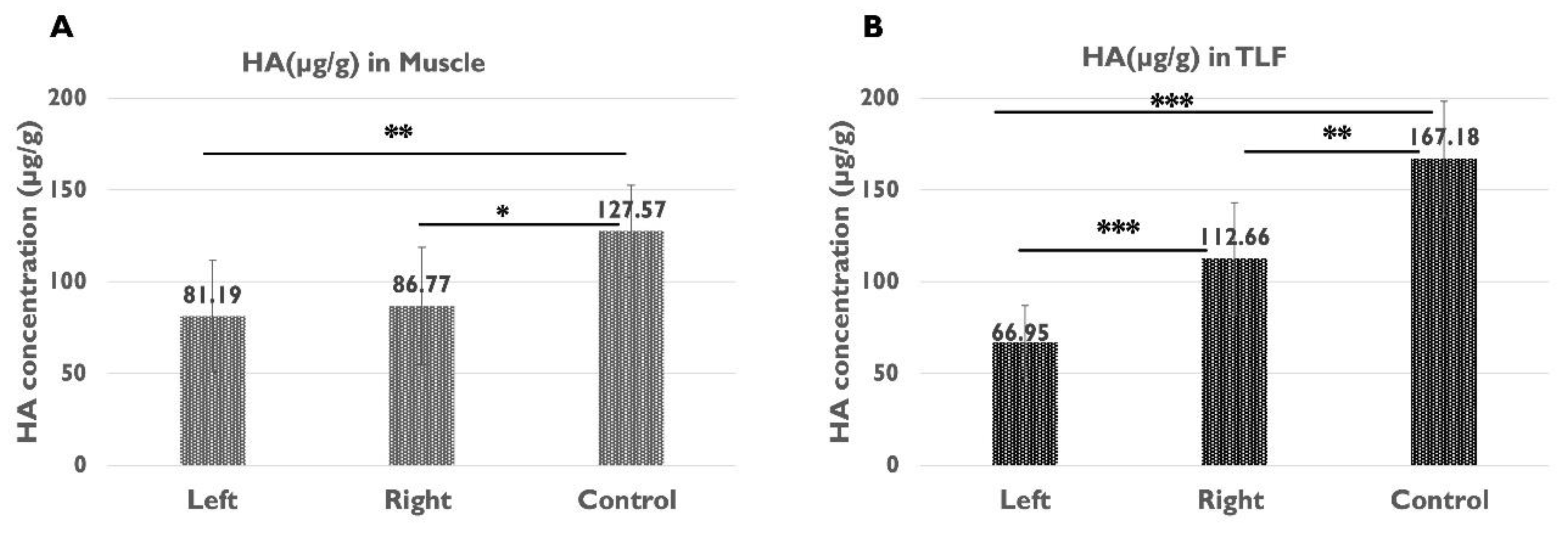
2.4. Quantification of HA in the Muscle and TLF
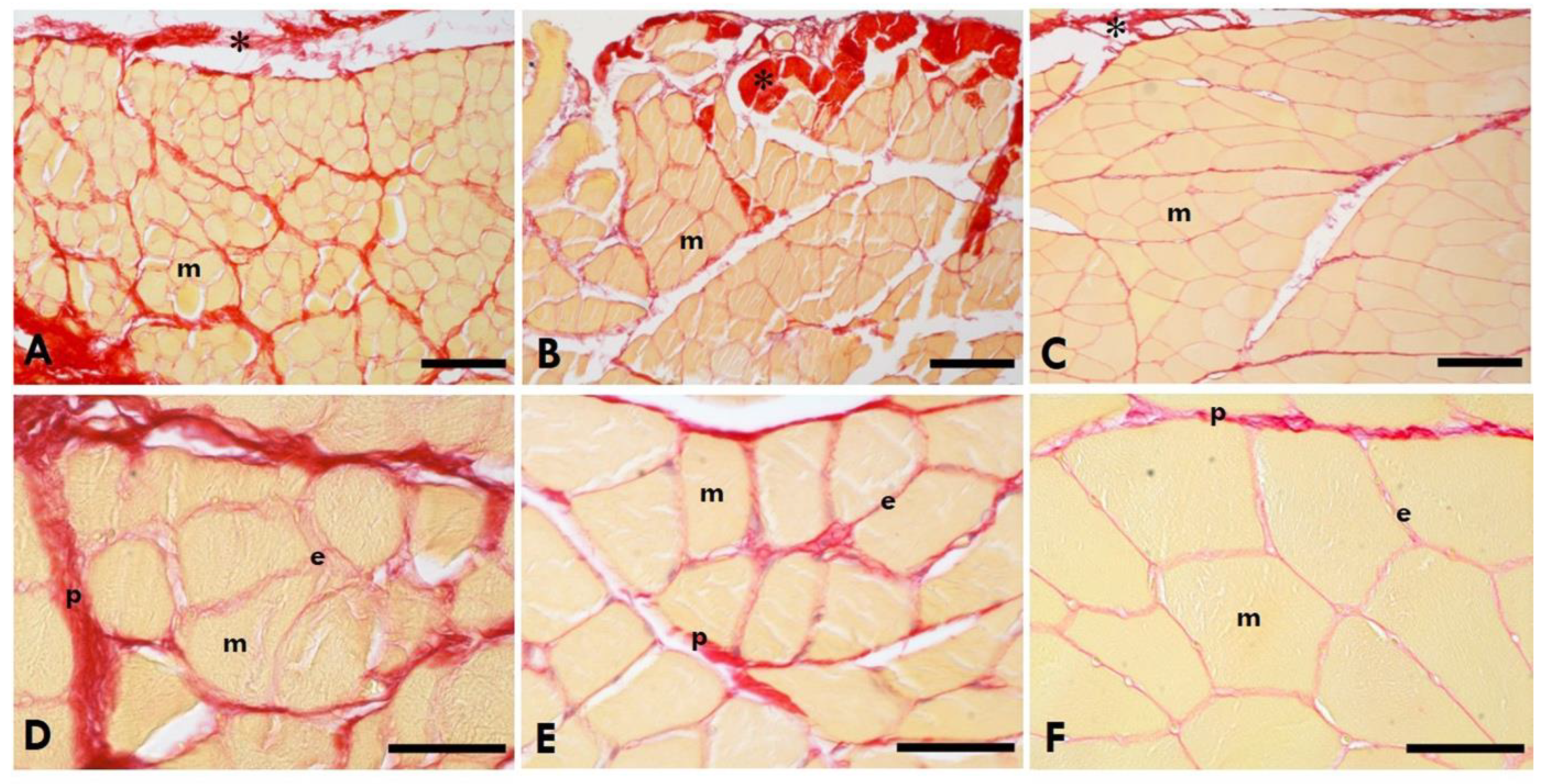
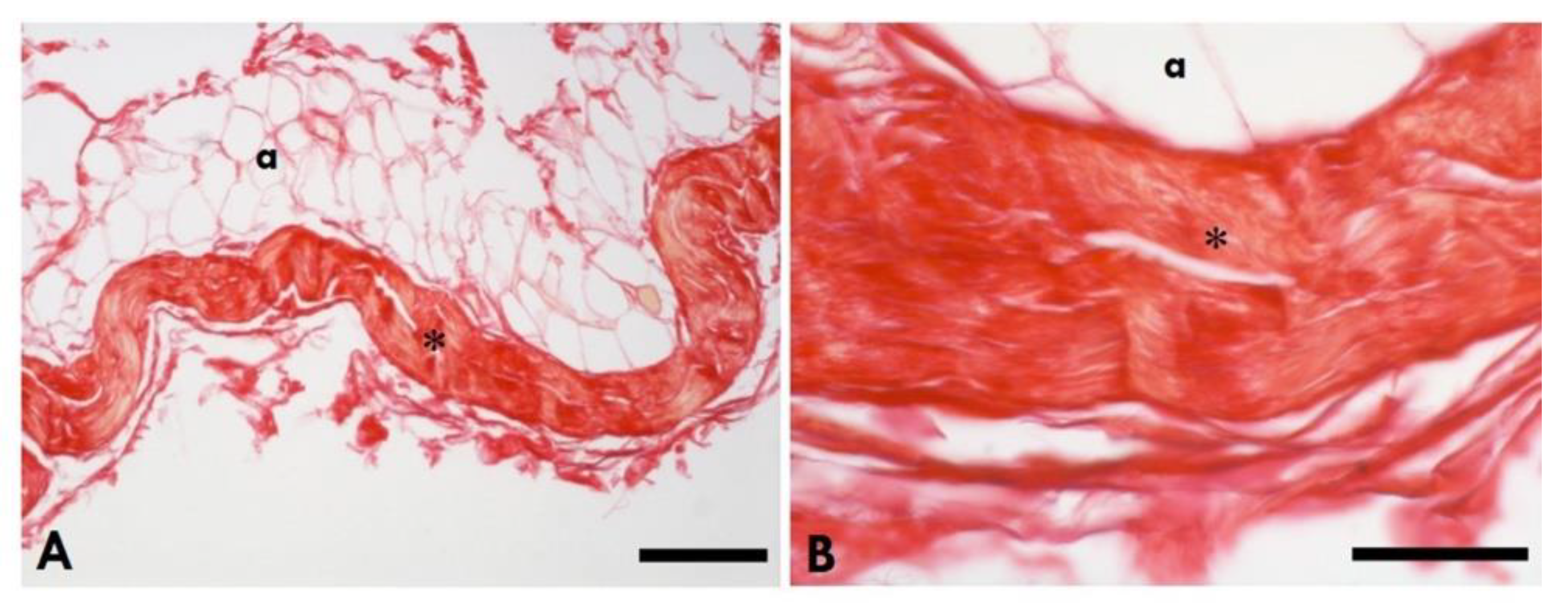
2.5. Quantification of Collagen in the Muscle and TLF
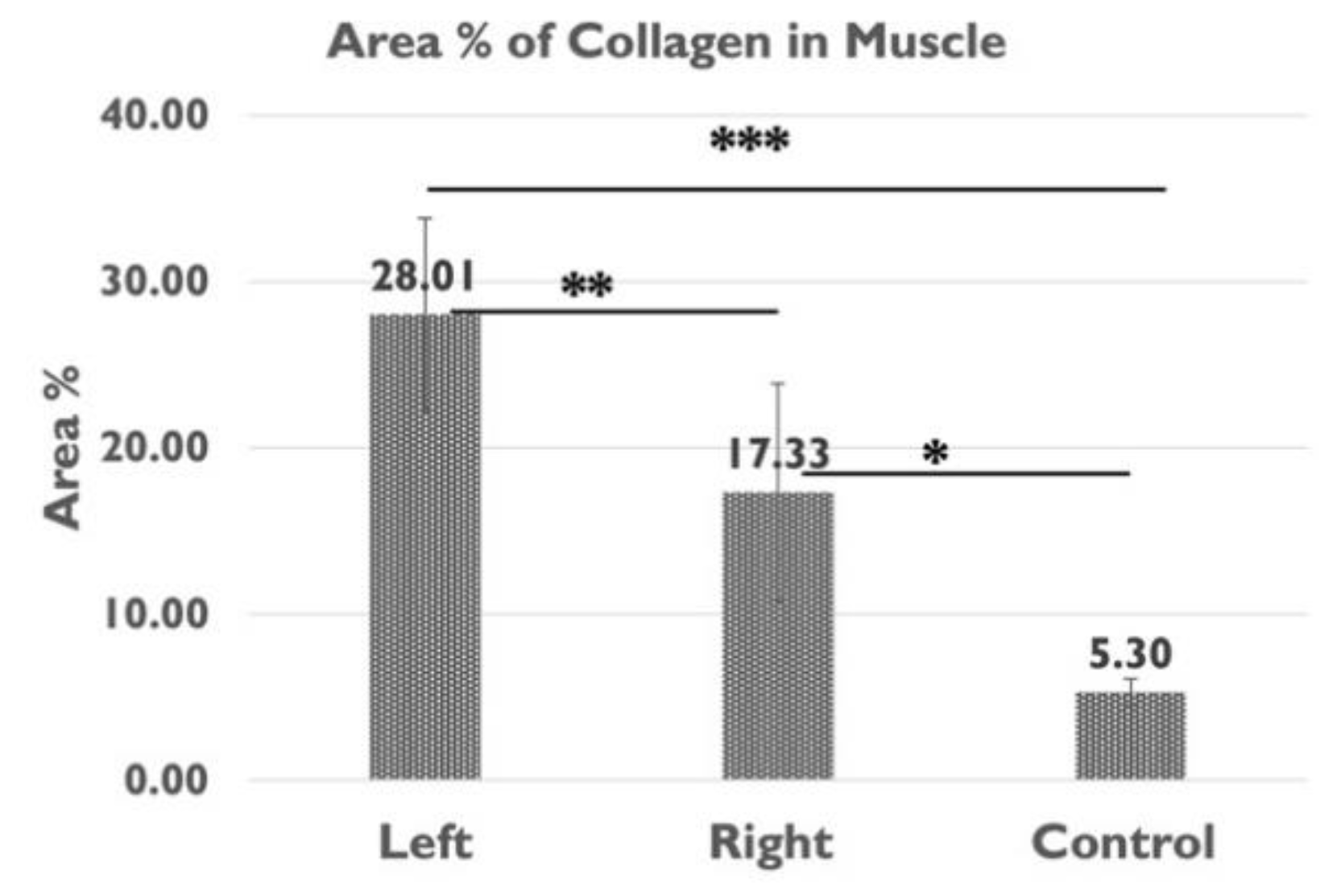
2.6. Area Percentage of Collagen Content in IMCT in the Gastrocnemius Muscle
3. Discussion
Limitations and Further Research
4. Materials and Methods
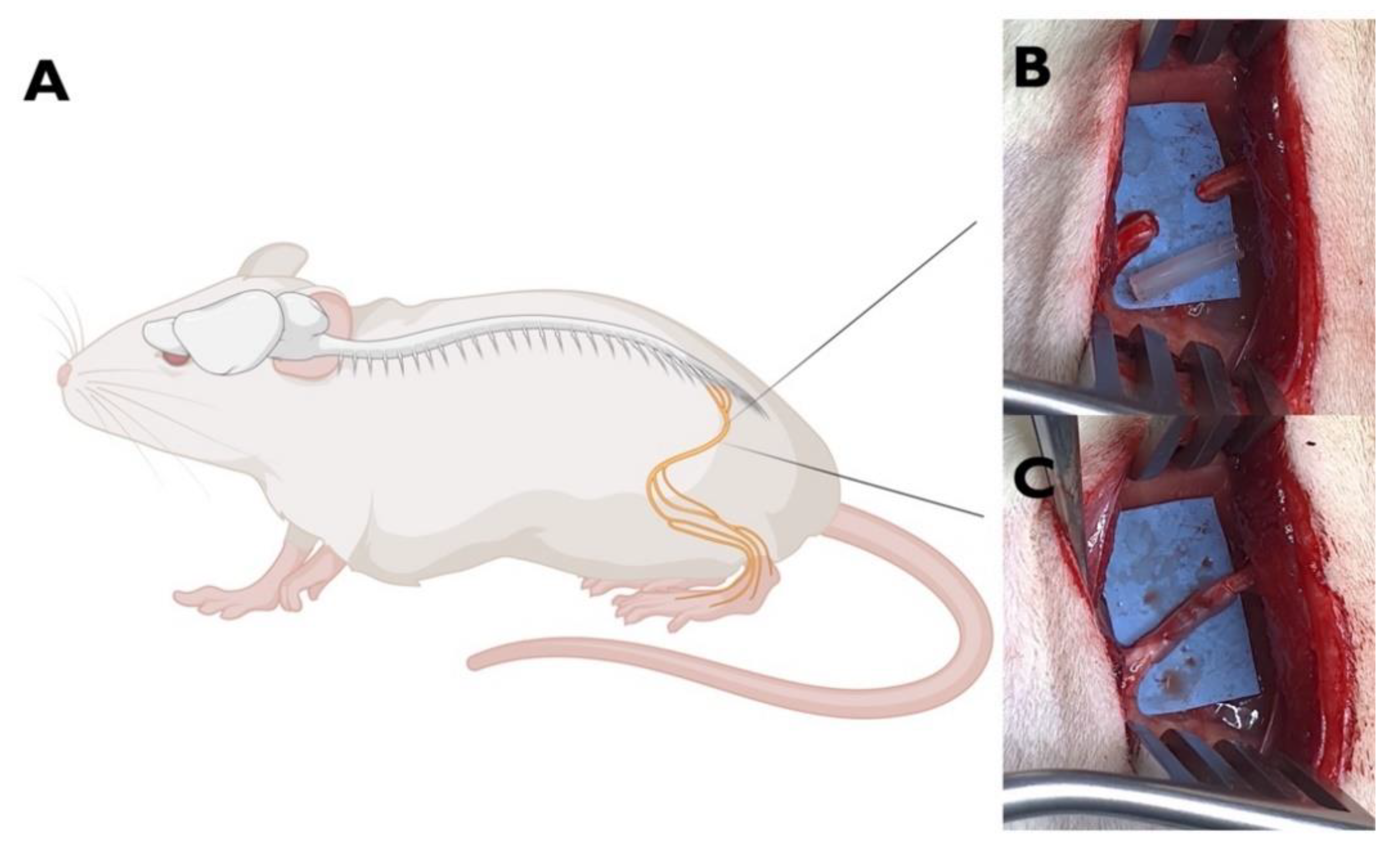
4.1. Animal Model of PNI
4.2. Behavioural Tests of Motor Function
4.2.1. Sciatic Functional Index
4.2.2. Gait Analysis
4.3. Sample Collection
4.4. Quantification of Hyaluronan
4.5. Quantification of Collagen
4.6. Histological Analysis
4.7. Image Analysis
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Aberg, M.; et al. Clinical evaluation of a resorbable wrap-around implant as an alternative to nerve repair: A prospective, assessor-blinded, randomised clinical study of sensory, motor and functional recovery after peripheral nerve repair. J Plast Reconstr Aesthet Surg. 2009, 62, 1503–1509. [Google Scholar] [CrossRef] [PubMed]
- Menorca, R.M.; Fussell, T.S.; Elfar, J.C. Nerve physiology: Mechanisms of injury and recovery. Hand Clin. 2013, 29, 317–330. [Google Scholar] [CrossRef] [PubMed]
- Lopes, B.; et al. Peripheral Nerve Injury Treatments and Advances: One Health Perspective. Int J Mol Sci. 2022, 23. [Google Scholar]
- Carlson, B.M. The Biology of Long-Term Denervated Skeletal Muscle. Eur J Transl Myol. 2014, 24, 3293. [Google Scholar] [CrossRef] [PubMed]
- Purslow, P.P. The Structure and Role of Intramuscular Connective Tissue in Muscle Function. Front Physiol. 2020, 11, 495. [Google Scholar] [CrossRef] [PubMed]
- Gillies, A.R.; Lieber, R.L. Structure and function of the skeletal muscle extracellular matrix. Muscle Nerve. 2011, 44, 318–331. [Google Scholar] [CrossRef] [PubMed]
- Rebolledo, D.L.; et al. Denervation-induced skeletal muscle fibrosis is mediated by CTGF/CCN2 independently of TGF-β.
- Ozawa, J.; et al. Regulation of connective tissue remodeling in the early phase of denervation in a rat skeletal muscle. Biomed Res. 2013, 34, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Savolainen, J.; et al. Effects of denervation and immobilization on collagen synthesis in rat skeletal muscle and tendon. Am J Physiol. 1988; 254, Pt 2, R897–R902. [Google Scholar]
- Arruda, E.M.; et al. Denervation does not change the ratio of collagen I and collagen III mRNA in the extracellular matrix of muscle. Am J Physiol Regul Integr Comp Physiol. 2007, 292, R983–R987. [Google Scholar] [CrossRef] [PubMed]
- Salonen, V.; et al. Changes in intramuscular collagen and fibronectin in denervation atrophy. Muscle Nerve. 1985, 8, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Coburn, J.; et al. Biomimetics of the Extracellular Matrix: An Integrated Three-Dimensional Fiber-Hydrogel Composite for Cartilage Tissue Engineering. Smart Struct Syst. 2011, 7, 213–222. [Google Scholar] [CrossRef]
- Fede, C.; et al. A Closer Look at the Cellular and Molecular Components of the Deep/Muscular Fasciae. Int J Mol Sci. 2021, 22. [Google Scholar]
- Singla, S.K. Nomenclature of hyaluronic acid. Biochem J. 1987, 242, 623. [Google Scholar] [CrossRef]
- Stecco, C.; et al. Hyaluronan within fascia in the etiology of myofascial pain. Surg Radiol Anat. 2011, 33, 891–896. [Google Scholar] [CrossRef] [PubMed]
- McCombe, D.; et al. The histochemical structure of the deep fascia and its structural response to surgery. J Hand Surg Br. 2001, 26, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Pratt, R.L. Hyaluronan andthe Fascial Frontier. Int J Mol Sci. 2021, 22. [Google Scholar]
- Cowman, M.K.; et al. Viscoelastic Properties of Hyaluronan in Physiological Conditions. F1000Res 2015. 4, 622. [CrossRef]
- Amir, A.; et al. Hyaluronan homeostasis and its role in pain and muscle stiffness. Pm r. 2022, 14, 1490–1496. [Google Scholar] [CrossRef] [PubMed]
- Okita, M.; et al. Effects of reduced joint mobility on sarcomere length, collagen fibril arrangement in the endomysium, and hyaluronan in rat soleus muscle. J Muscle Res Cell Motil. 2004, 25, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Menon, R.G.; Raghavan, P.; Regatte, R.R. Quantifying muscle glycosaminoglycan levels in patients with post-stroke muscle stiffness using T(1ρ) MRI. Sci. Rep. 2019, 9, 14513. [Google Scholar] [CrossRef] [PubMed]
- Fede, C.; et al. The Effects of Aging on the Intramuscular Connective Tissue. Int J Mol Sci. 2022, 23. [Google Scholar] [CrossRef] [PubMed]
- Fantoni, I.; et al. Fascia Lata Alterations in Hip Osteoarthritis: An Observational Cross-Sectional Study. Life 2021, 11. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.I.; Choe, M.A. Effects of unilateral sciatic nerve injury on unaffected hindlimb muscles of rats. J Korean Acad Nurs. 2009, 39, 393–400. [Google Scholar] [CrossRef]
- Choe, M.A.; et al. Hindlimb muscle atrophy occurs from peripheral nerve damage in a rat neuropathic pain model. Biol Res Nurs. 2011, 13, 44–54. [Google Scholar] [CrossRef]
- Stecco, C. Functional Atlas of the Human Fascial System; Elsevier Health Sciences: Edinburgh, 2015. [Google Scholar]
- Bain, J.R.; Mackinnon, S.E.; Hunter, D.A. Functional evaluation of complete sciatic, peroneal, and posterior tibial nerve lesions in the rat. Plast Reconstr Surg. 1989, 83, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Stocco, E.; et al. Development and preclinical evaluation of bioactive nerve conduits for peripheral nerve regeneration: A comparative study. Mater Today Bio. 2023, 22, 100761. [Google Scholar] [CrossRef] [PubMed]
- Litwiniuk, M.; et al. Hyaluronic Acid in Inflammation and Tissue Regeneration. Wounds 2016, 28, 78–88. [Google Scholar] [PubMed]
- Jiang, D.; Liang, J.; Noble, P.W. Hyaluronan in tissue injury and repair. Annu Rev Cell Dev Biol. 2007, 23, 435–461. [Google Scholar] [CrossRef] [PubMed]
- Leng, Y.; et al. Hyaluronic acid, CD44 and RHAMM regulate myoblast behavior during embryogenesis. Matrix Biol. 2019, 78-79, 236–254. [Google Scholar] [CrossRef] [PubMed]
- Nakka, K.; et al. JMJD3 activated hyaluronan synthesis drives muscle regeneration in an inflammatory environment. Science 2022, 377, 666–669. [Google Scholar] [CrossRef]
- Ricard-Blum, S. The collagen family. Cold Spring Harb Perspect Biol. 2011, 3, a004978. [Google Scholar] [CrossRef] [PubMed]
- Tang, V.W. Collagen, stiffness, and adhesion: The evolutionary basis of vertebrate mechanobiology. Mol Biol Cell 2020, 31, 1823–1834. [Google Scholar] [CrossRef]
- Zhang, W.; Liu, Y.; Zhang, H. Extracellular matrix: An important regulator of cell functions and skeletal muscle development. Cell Biosci. 2021, 11, 65. [Google Scholar] [CrossRef]
- Liu, J.; et al. Research on skeletal muscle impact injury using a new rat model from a bioimpact machine. Front Bioeng Biotechnol. 2022, 10, 1055668. [Google Scholar] [CrossRef]
- Francis, C.A.; et al. The modulation of forward propulsion, vertical support, and center of pressure by the plantarflexors during human walking. Gait Posture 2013, 38, 993–997. [Google Scholar] [CrossRef] [PubMed]
- Willard, F.H.; et al. The thoracolumbar fascia: Anatomy, function and clinical considerations. J Anat. 2012, 221, 507–536. [Google Scholar] [CrossRef] [PubMed]
- Benetazzo, L.; et al. 3D reconstruction of the crural and thoracolumbar fasciae. Surg Radiol Anat. 2011, 33, 855–862. [Google Scholar] [CrossRef] [PubMed]
- Tesarz, J.; et al. Sensory innervation of the thoracolumbar fascia in rats and humans. Neuroscience 2011, 194, 302–308. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; et al. Effects of Isometric Plantar-Flexion on the Lower Limb Muscle and Lumbar Tissue Stiffness. Front Bioeng Biotechnol. 2021, 9, 810250. [Google Scholar] [CrossRef] [PubMed]
- Langevin, H.M.; et al. Reduced thoracolumbar fascia shear strain in human chronic low back pain. BMC Musculoskelet Disord. 2011, 12, 203. [Google Scholar] [CrossRef] [PubMed]
- Langevin, H.M. Fascia Mobility, Proprioception, and Myofascial Pain. Life 2021, 11. [Google Scholar] [CrossRef] [PubMed]
- Kodama, Y.; et al. Response to Mechanical Properties and Physiological Challenges of Fascia: Diagnosis and Rehabilitative Therapeutic Intervention for Myofascial System Disorders. Bioengineering 2023, 10. [Google Scholar] [CrossRef]
- Stocco, E.; et al. New bioresorbable wraps based on oxidized polyvinyl alcohol and leukocyte-fibrin-platelet membrane to support peripheral nerve neurorrhaphy: Preclinical comparison versus NeuraWrap. Sci Rep. 2019, 9, 17193. [Google Scholar] [CrossRef]
- Stocco, E.; et al. Partially oxidized polyvinyl alcohol conduitfor peripheral nerve regeneration. Sci Rep. 2018, 8, 604. [Google Scholar] [CrossRef]
- J, I.J.-P.; et al. Transection of peripheral nerves, bridging strategies and effect evaluation. Biomaterials 2004, 25, 1583–1592. [Google Scholar]
- Fede, C.; et al. Quantification of hyaluronan in human fasciae: Variations with function and anatomical site. J Anat. 2018, 233, 552–556. [Google Scholar] [CrossRef] [PubMed]
- Stecco, C.; et al. The fasciacytes: A new cell devoted to fascial gliding regulation. Clin Anat. 2018, 31, 667–676. [Google Scholar] [CrossRef] [PubMed]
- Scott, J.E.; Dorling, J. Differential staining of acid glycosaminoglycans (mucopolysaccharides) by alcian blue in salt solutions. Histochemie 1965, 5, 221–233. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat Methods. 2012, 9, 671–675. [Google Scholar] [CrossRef]
| Variable | L (n=12) | R (n=12) | Ctrl (n=6) | p-value L vs. R |
p-value L vs. Ctrl |
p-value R vs. Ctrl |
|---|---|---|---|---|---|---|
| µg HA/g muscle | 81.19 ± 30.33 | 86.77 ± 31.96 | 127.57 ± 24.90 | 0.473 | 0.005** | 0.015* |
| µg HA/g TLF | 66.95 ± 20.08 | 112.66 ± 30.53 | 167.18 ± 31.13 | 0.000*** | 0.000*** | 0.003** |
| µg Col/g muscle | 32.92±11.34 | 20.54 ± 7.03 | 12.74 ± 4.83 | 0.001** | 0.001** | 0.035* |
| µg Col/g TLF | 115.89 ± 28.18 | 90.43 ± 20.83 | 47.51 ± 7.82 | 0.016* | 0.000*** | 0.000*** |
| Col area% muscle | 28.01 ± 5.78 | 17.33 ± 6.52% | 5.30 ± 0.82 % | 0.004** | 0.000*** | 0.011* |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




