Submitted:
03 July 2024
Posted:
04 July 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Post-COVID Syndrome (PCS) and Symptom Prevalence
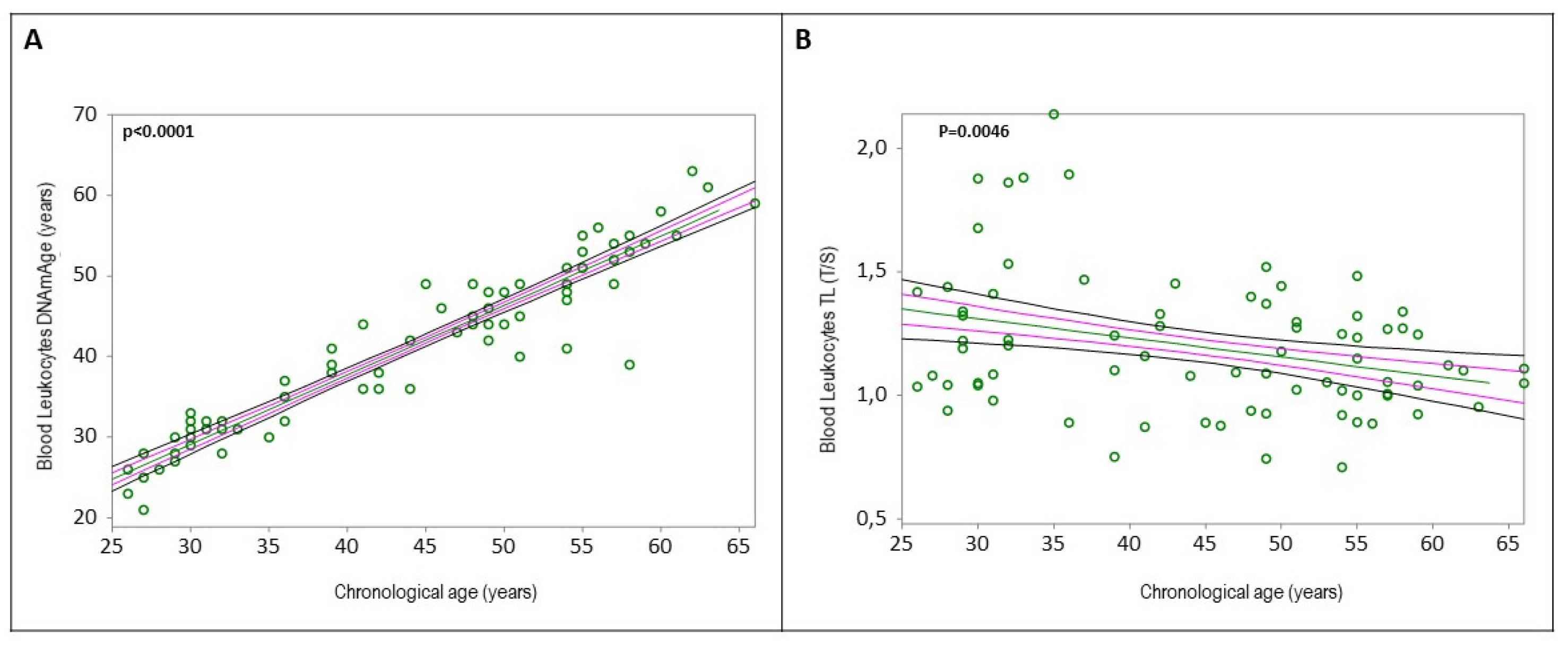
2.2. Blood Leukocytes Biological Age
2.3. Determinants of Blood Leukocytes DNAmAge and TL
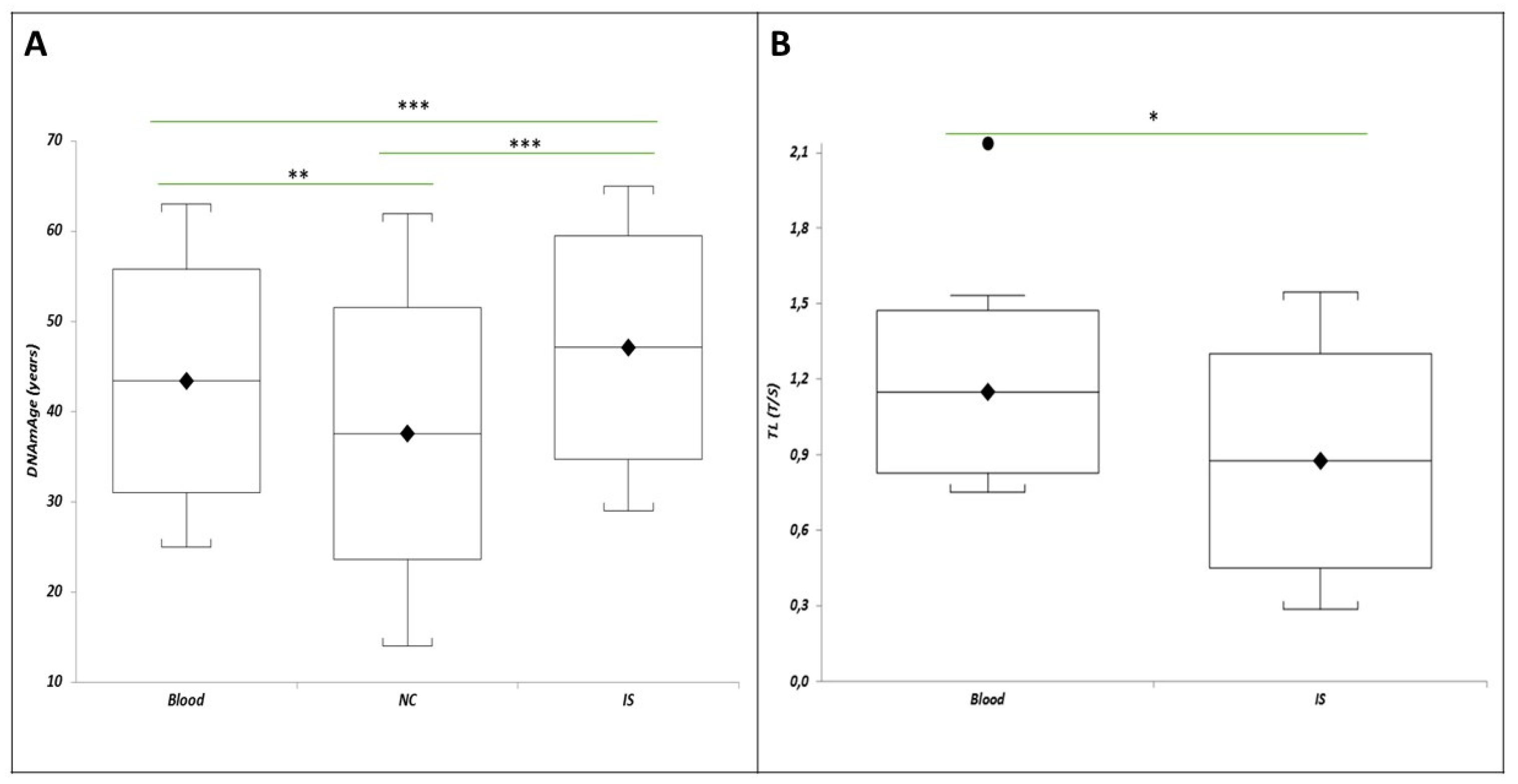
2.4. Biological Age of Blood Leukocytes, IS Cells, and NC
2.5. Correlations between Biological Aging Indicators
2.6. Comparison of Biological Aging in HCWs and COPD Patients
3. Discussion
3.1. PCS and Symptom Prevalence
3.2. Determinants of Increased Blood Leukocyte Dnamage
3.2.1. Sex-Related DNAmAge Differences
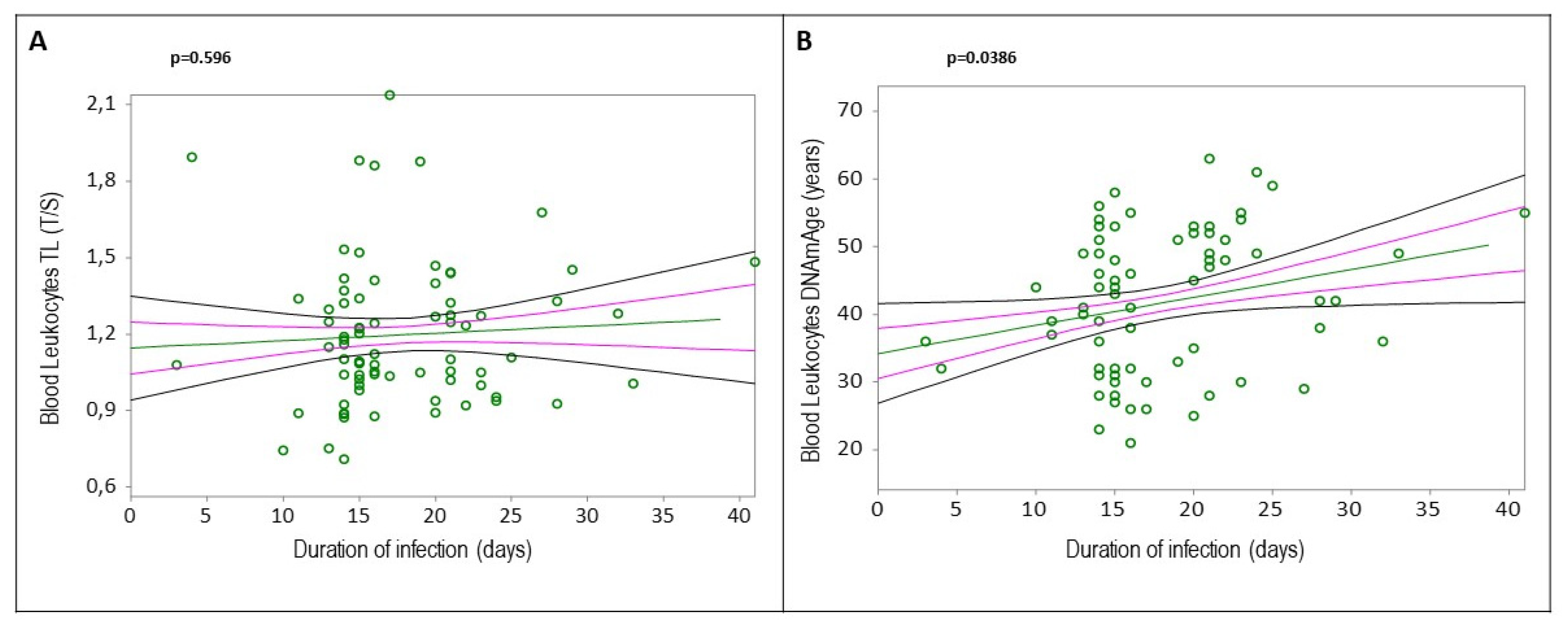
3.2.2. Impact of SARS-CoV-2 Infection
3.2.4. Chronic Diseases and DNAmAge
3.2.5. Lung Function and DNAmAge
3.2.6. Lipid Levels and DNAmAge
3.2.7. Blood Glucose and DNAmAge
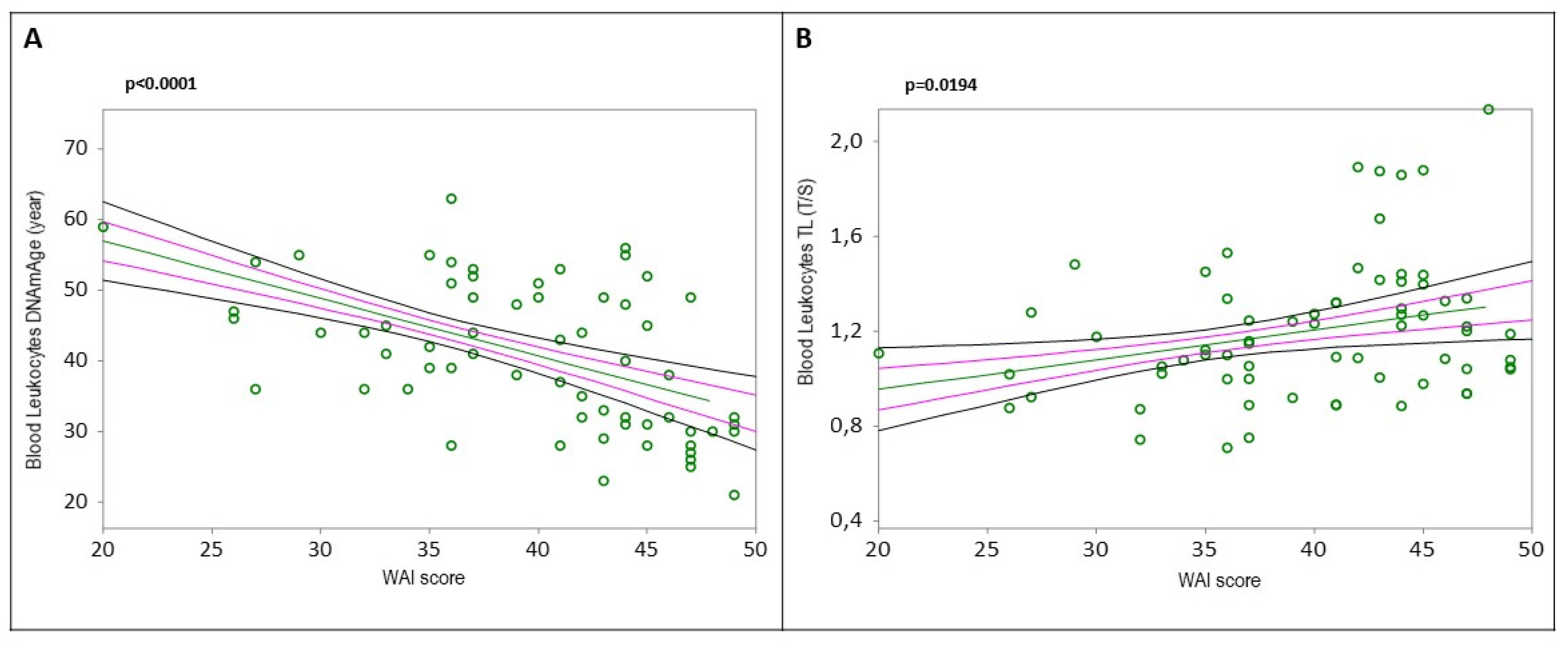
3.2.8. Work Capacity and DNAmAge
3.2.9. Lymphocyte Counts and DNAmAge
3.2.10. Haemoglobin Levels and DNAmAge
3.2.11. HR, HRV and DNAmAge
3.3. Determinants of Shorter Blood Leukocytes TL
3.3.1. WAI
3.3.2. LDL Levels and Cardiovascular Disease
3.3.3. Blood Leukocyte TL and Job Position
3.3.4. Lymphocyte Numbers
3.4. Biological Age of the Blood Leukocytes, IS Cells, and NC Determined by DNAmAge AND TL
3.4.1. Tissue-Specific Aging Rates
3.4.2. COVID-19 Impact on DNAmAge and TL
3.4.3. Biological Implications of Telomere Shortening in IS
3.4.4. Epigenetic Aging in IS cells, NC and Implications for Surrogate Tissue Use
3.5. Comparison of Biological Aging (AgeAcc and TL) in HCWs and COPD Patients
3.6. Limitations and Strengths
4. Materials and Methods
4.1. Study Design
4.2. Information Acquired through Questionnaires
4.3. Work Ability Assessment
4.4. Respiratory FUNCTION TESTS
4.5. Assessment of Autonomic Cardiac Balance and HRV Parameters
4.6. Samples Collection and IS Procedure
4.7. Basic Biochemistry Analyses
4.8. DNA Extraction (from Biological Samples)
4.9. DNAmAge Analysis and AgeAcc Estimation
4.10. TL Analysis
4.11. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- COVID-19 Cases | WHO COVID-19 Dashboard. Available online: https://data.who.int/dashboards/covid19/cases (accessed on 17 June 2024).
- Gorna, R.; MacDermott, N.; Rayner, C.; O’Hara, M.; Evans, S.; Agyen, L.; Nutland, W.; Rogers, N.; Hastie, C. Long COVID Guidelines Need to Reflect Lived Experience. The Lancet 2021, 397, 455–457. [Google Scholar] [CrossRef] [PubMed]
- Fernández-de-las-Peñas, C.; Palacios-Ceña, D.; Gómez-Mayordomo, V.; Cuadrado, M.L.; Florencio, L.L. Defining Post-COVID Symptoms (Post-Acute COVID, Long COVID, Persistent Post-COVID): An Integrative Classification. Int J Environ Res Public Health 2021, 18, 2621. [Google Scholar] [CrossRef] [PubMed]
- Parotto, M.; Gyöngyösi, M.; Howe, K.; Myatra, S.N.; Ranzani, O.; Shankar-Hari, M.; Herridge, M.S. Post-Acute Sequelae of COVID-19: Understanding and Addressing the Burden of Multisystem Manifestations. The Lancet Respiratory Medicine 2023, 11, 739–754. [Google Scholar] [CrossRef] [PubMed]
- Alkodaymi, M.S.; Omrani, O.A.; Fawzy, N.A.; Shaar, B.A.; Almamlouk, R.; Riaz, M.; Obeidat, M.; Obeidat, Y.; Gerberi, D.; Taha, R.M.; et al. Prevalence of Post-Acute COVID-19 Syndrome Symptoms at Different Follow-up Periods: A Systematic Review and Meta-Analysis. Clinical Microbiology and Infection 2022, 28, 657–666. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.; Zheng, B.; Daines, L.; Sheikh, A. Long-Term Sequelae of COVID-19: A Systematic Review and Meta-Analysis of One-Year Follow-Up Studies on Post-COVID Symptoms. Pathogens 2022, 11, 269. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Deng, J.; Liu, Q.; Du, M.; Liu, M.; Liu, J. Long-Term Consequences of Asymptomatic SARS-CoV-2 Infection: A Systematic Review and Meta-Analysis. International Journal of Environmental Research and Public Health 2023, 20, 1613. [Google Scholar] [CrossRef] [PubMed]
- Boscolo-Rizzo, P.; Hummel, T.; Hopkins, C.; Dibattista, M.; Menini, A.; Spinato, G.; Fabbris, C.; Emanuelli, E.; D’Alessandro, A.; Marzolino, R.; et al. High Prevalence of Long-Term Olfactory, Gustatory, and Chemesthesis Dysfunction in Post-COVID-19 Patients: A Matched Case-Control Study with One-Year Follow-up Using a Comprehensive Psychophysical Evaluation. Rhinology 2021, 59, 517–527. [Google Scholar] [CrossRef] [PubMed]
- Boscolo-Rizzo, P.; Guida, F.; Polesel, J.; Marcuzzo, A.V.; Capriotti, V.; D’Alessandro, A.; Zanelli, E.; Marzolino, R.; Lazzarin, C.; Antonucci, P.; et al. Sequelae in Adults at 12 Months after Mild-to-moderate Coronavirus Disease 2019 (COVID-19). Int Forum Allergy Rhinol 2021, 11, 1685–1688. [Google Scholar] [CrossRef]
- Andrei Appelt, P.; Taciana Sisconetto, A.; Baldo Sucupira, K.S.M.; Neto, E. de M.; Chagas, T. de J.; Bazan, R.; Moura Cabral, A.; Andrade, A. de O.; de Souza, L.A.P.S.; José Luvizutto, G. Changes in Electrical Brain Activity and Cognitive Functions Following Mild to Moderate COVID-19: A One-Year Prospective Study After Acute Infection. Clin EEG Neurosci 2022, 53, 543–557. [Google Scholar] [CrossRef]
- Rank, A.; Tzortzini, A.; Kling, E.; Schmid, C.; Claus, R.; Löll, E.; Burger, R.; Römmele, C.; Dhillon, C.; Müller, K.; et al. One Year after Mild COVID-19: The Majority of Patients Maintain Specific Immunity, But One in Four Still Suffer from Long-Term Symptoms. J Clin Med 2021, 10, 3305. [Google Scholar] [CrossRef]
- Maestre-Muñiz, M.M.; Arias, Á.; Mata-Vázquez, E.; Martín-Toledano, M.; López-Larramona, G.; Ruiz-Chicote, A.M.; Nieto-Sandoval, B.; Lucendo, A.J. Long-Term Outcomes of Patients with Coronavirus Disease 2019 at One Year after Hospital Discharge. J Clin Med 2021, 10, 2945. [Google Scholar] [CrossRef]
- Lombardo, M.D.M.; Foppiani, A.; Peretti, G.M.; Mangiavini, L.; Battezzati, A.; Bertoli, S.; Martinelli Boneschi, F.; Zuccotti, G.V. Long-Term Coronavirus Disease 2019 Complications in Inpatients and Outpatients: A One-Year Follow-up Cohort Study. Open Forum Infectious Diseases 2021, 8, ofab384. [Google Scholar] [CrossRef]
- Seeßle, J.; Waterboer, T.; Hippchen, T.; Simon, J.; Kirchner, M.; Lim, A.; Müller, B.; Merle, U. Persistent Symptoms in Adult Patients 1 Year After Coronavirus Disease 2019 (COVID-19): A Prospective Cohort Study. Clin Infect Dis 2022, 74, 1191–1198. [Google Scholar] [CrossRef] [PubMed]
- Fischer, A.; Zhang, L.; Elbéji, A.; Wilmes, P.; Oustric, P.; Staub, T.; Nazarov, P.V.; Ollert, M.; Fagherazzi, G. Long COVID Symptomatology After 12 Months and Its Impact on Quality of Life According to Initial Coronavirus Disease 2019 Disease Severity. Open Forum Infect Dis 2022, 9, ofac397. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Li, W.; Wang, T.; Ran, D.; Davalos, V.; Planas-Serra, L.; Pujol, A.; Esteller, M.; Wang, X.; Yu, H. Accelerated Biological Aging in COVID-19 Patients. Nat Commun 2022, 13, 2135. [Google Scholar] [CrossRef] [PubMed]
- Jylhävä, J.; Pedersen, N.L.; Hägg, S. Biological Age Predictors. EBioMedicine 2017, 21, 29–36. [Google Scholar] [CrossRef]
- Pavanello, S.; Campisi, M.; Tona, F.; Lin, C.D.; Iliceto, S. Exploring Epigenetic Age in Response to Intensive Relaxing Training: A Pilot Study to Slow Down Biological Age. Int J Environ Res Public Health 2019, 16, 3074. [Google Scholar] [CrossRef] [PubMed]
- Mongelli, A.; Barbi, V.; Gottardi Zamperla, M.; Atlante, S.; Forleo, L.; Nesta, M.; Massetti, M.; Pontecorvi, A.; Nanni, S.; Farsetti, A.; et al. Evidence for Biological Age Acceleration and Telomere Shortening in COVID-19 Survivors. International Journal of Molecular Sciences 2021, 22, 6151. [Google Scholar] [CrossRef] [PubMed]
- Franzen, J.; Nüchtern, S.; Tharmapalan, V.; Vieri, M.; Nikolić, M.; Han, Y.; Balfanz, P.; Marx, N.; Dreher, M.; Brümmendorf, T.H.; et al. Epigenetic Clocks Are Not Accelerated in COVID-19 Patients. International Journal of Molecular Sciences 2021, 22, 9306. [Google Scholar] [CrossRef]
- Campisi, M.; Liviero, F.; Maestrelli, P.; Guarnieri, G.; Pavanello, S. DNA Methylation-Based Age Prediction and Telomere Length Reveal an Accelerated Aging in Induced Sputum Cells Compared to Blood Leukocytes: A Pilot Study in COPD Patients. Front. Med. 2021, 8. [Google Scholar] [CrossRef]
- COVID-19 Rapid Guideline: Managing the Long-Term Effects of COVID-19; National Institute for Health and Care Excellence: Clinical Guidelines; National Institute for Health and Care Excellence (NICE): London, 2020; ISBN 978-1-4731-3943-5.
- Tudorache, E.; Fildan, A.P.; Frandes, M.; Dantes, E.; Tofolean, D.E. Aging and Extrapulmonary Effects of Chronic Obstructive Pulmonary Disease. CIA 2017, 12, 1281–1287. [Google Scholar] [CrossRef] [PubMed]
- Ortona, E.; Buonsenso, D.; Carfi, A.; Malorni, W.; Group, T.L.C.K. study Long COVID: An Estrogen-Associated Autoimmune Disease? Cell Death Discovery 2021, 7. [Google Scholar] [CrossRef]
- Value Gender and Equity in the Global Health Workforce. Available online: https://www.who.int/activities/value-gender-and-equity-in-the-global-health-workforce (accessed on 19 June 2024).
- Khamsi, R. Rogue Antibodies Could Be Driving Severe COVID-19. Nature 2021, 590, 29–31. [Google Scholar] [CrossRef]
- Ballouz, T.; Menges, D.; Anagnostopoulos, A.; Domenghino, A.; Aschmann, H.E.; Frei, A.; Fehr, J.S.; Puhan, M.A. Recovery and Symptom Trajectories up to Two Years after SARS-CoV-2 Infection: Population Based, Longitudinal Cohort Study. BMJ 2023, 381, e074425. [Google Scholar] [CrossRef] [PubMed]
- Fumagalli, C.; Zocchi, C.; Tassetti, L.; Silverii, M.V.; Amato, C.; Livi, L.; Giovannoni, L.; Verrillo, F.; Bartoloni, A.; Marcucci, R.; et al. Factors Associated with Persistence of Symptoms 1 Year after COVID-19: A Longitudinal, Prospective Phone-Based Interview Follow-up Cohort Study. European Journal of Internal Medicine 2022, 97, 36–41. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-de-las-Peñas, C.; Notarte, K.I.; Macasaet, R.; Velasco, J.V.; Catahay, J.A.; Ver, A.T.; Chung, W.; Valera-Calero, J.A.; Navarro-Santana, M. Persistence of Post-COVID Symptoms in the General Population Two Years after SARS-CoV-2 Infection: A Systematic Review and Meta-Analysis. Journal of Infection 2024, 88, 77–88. [Google Scholar] [CrossRef]
- Oblak, L.; van der Zaag, J.; Higgins-Chen, A.T.; Levine, M.E.; Boks, M.P. A Systematic Review of Biological, Social and Environmental Factors Associated with Epigenetic Clock Acceleration. Ageing Research Reviews 2021, 69, 101348. [Google Scholar] [CrossRef]
- Alberts, S.C.; Archie, E.A.; Gesquiere, L.R.; Altmann, J.; Vaupel, J.W.; Christensen, K. The Male-Female Health-Survival Paradox: A Comparative Perspective on Sex Differences in Aging and Mortality. In Sociality, Hierarchy, Health: Comparative Biodemography: A Collection of Papers; National Academies Press (US), 2014. [Google Scholar]
- Corley, M.J.; Pang, A.P.S.; Dody, K.; Mudd, P.A.; Patterson, B.K.; Seethamraju, H.; Bram, Y.; Peluso, M.J.; Torres, L.; Iyer, N.S.; et al. Genome-Wide DNA Methylation Profiling of Peripheral Blood Reveals an Epigenetic Signature Associated with Severe COVID-19. Journal of Leukocyte Biology 2021, 110, 21–26. [Google Scholar] [CrossRef]
- Schäfer, A.; Baric, R.S. Epigenetic Landscape during Coronavirus Infection. Pathogens 2017, 6, 8. [Google Scholar] [CrossRef]
- Castro de Moura, M.; Davalos, V.; Planas-Serra, L.; Alvarez-Errico, D.; Arribas, C.; Ruiz, M.; Aguilera-Albesa, S.; Troya, J.; Valencia-Ramos, J.; Vélez-Santamaria, V.; et al. Epigenome-Wide Association Study of COVID-19 Severity with Respiratory Failure. EBioMedicine 2021, 66, 103339. [Google Scholar] [CrossRef]
- Menachery, V.D.; Schäfer, A.; Burnum-Johnson, K.E.; Mitchell, H.D.; Eisfeld, A.J.; Walters, K.B.; Nicora, C.D.; Purvine, S.O.; Casey, C.P.; Monroe, M.E.; et al. MERS-CoV and H5N1 Influenza Virus Antagonize Antigen Presentation by Altering the Epigenetic Landscape. Proc Natl Acad Sci U S A 2018, 115, E1012–E1021. [Google Scholar] [CrossRef] [PubMed]
- Esteban-Cantos, A.; Rodríguez-Centeno, J.; Barruz, P.; Alejos, B.; Saiz-Medrano, G.; Nevado, J.; Martin, A.; Gayá, F.; De Miguel, R.; Bernardino, J.I.; et al. Epigenetic Age Acceleration Changes 2 Years after Antiretroviral Therapy Initiation in Adults with HIV: A Substudy of the NEAT001/ANRS143 Randomised Trial. The Lancet HIV 2021, 8, e197–e205. [Google Scholar] [CrossRef] [PubMed]
- Horvath, S.; Levine, A.J. HIV-1 Infection Accelerates Age According to the Epigenetic Clock. The Journal of Infectious Diseases 2015, 212, 1563–1573. [Google Scholar] [CrossRef] [PubMed]
- Horvath, S.; Stein, D.J.; Phillips, N.; Heany, S.J.; Kobor, M.S.; Lin, D.T.S.; Myer, L.; Zar, H.J.; Levine, A.J.; Hoare, J. Perinatally Acquired HIV Infection Accelerates Epigenetic Aging in South African Adolescents. AIDS 2018, 32, 1465. [Google Scholar] [CrossRef] [PubMed]
- Gale, C.R.; Marioni, R.E.; Harris, S.E.; Starr, J.M.; Deary, I.J. DNA Methylation and the Epigenetic Clock in Relation to Physical Frailty in Older People: The Lothian Birth Cohort 1936. Clin Epigenet 2018, 10, 101. [Google Scholar] [CrossRef] [PubMed]
- Dugué, P.-A.; Bassett, J.K.; Joo, J.E.; Jung, C.-H.; Ming Wong, E.; Moreno-Betancur, M.; Schmidt, D.; Makalic, E.; Li, S.; Severi, G.; et al. DNA Methylation-Based Biological Aging and Cancer Risk and Survival: Pooled Analysis of Seven Prospective Studies. International Journal of Cancer 2018, 142, 1611–1619. [Google Scholar] [CrossRef] [PubMed]
- Grant, C.D.; Jafari, N.; Hou, L.; Li, Y.; Stewart, J.D.; Zhang, G.; Lamichhane, A.; Manson, J.E.; Baccarelli, A.A.; Whitsel, E.A.; et al. A Longitudinal Study of DNA Methylation as a Potential Mediator of Age-Related Diabetes Risk. GeroScience 2017, 39, 475–489. [Google Scholar] [CrossRef]
- Roetker, N.S.; Pankow, J.S.; Bressler, J.; Morrison, A.C.; Boerwinkle, E. Prospective Study of Epigenetic Age Acceleration and Incidence of Cardiovascular Disease Outcomes in the ARIC Study (Atherosclerosis Risk in Communities). Circulation: Genomic and Precision Medicine 2018, 11, e001937. [Google Scholar] [CrossRef] [PubMed]
- Horvath, S.; Ritz, B.R. Increased Epigenetic Age and Granulocyte Counts in the Blood of Parkinson’s Disease Patients. Aging 2015, 7, 1130–1142. [Google Scholar] [CrossRef]
- Lowery, E.M.; Brubaker, A.L.; Kuhlmann, E.; Kovacs, E.J. The Aging Lung. CIA 2013, 8, 1489–1496. [Google Scholar] [CrossRef]
- Sharma, G.; Goodwin, J. Effect of Aging on Respiratory System Physiology and Immunology. Clinical Interventions in Aging 2006, 1, 253. [Google Scholar] [CrossRef] [PubMed]
- Thomas, E.T.; Guppy, M.; Straus, S.E.; Bell, K.J.L.; Glasziou, P. Rate of Normal Lung Function Decline in Ageing Adults: A Systematic Review of Prospective Cohort Studies. BMJ Open 2019, 9, e028150. [Google Scholar] [CrossRef] [PubMed]
- Ammous, F.; Zhao, W.; Ratliff, S.M.; Mosley, T.H.; Bielak, L.F.; Zhou, X.; Peyser, P.A.; Kardia, S.L.R.; Smith, J.A. Epigenetic Age Acceleration Is Associated with Cardiometabolic Risk Factors and Clinical Cardiovascular Disease Risk Scores in African Americans. Clinical Epigenetics 2021, 13, 55. [Google Scholar] [CrossRef] [PubMed]
- Peters, S.A.E.; Singhateh, Y.; Mackay, D.; Huxley, R.R.; Woodward, M. Total Cholesterol as a Risk Factor for Coronary Heart Disease and Stroke in Women Compared with Men: A Systematic Review and Meta-Analysis. Atherosclerosis 2016, 248, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Nagasawa, S.; Okamura, T.; Iso, H.; Tamakoshi, A.; Yamada, M.; Watanabe, M.; Murakami, Y.; Miura, K.; Ueshima, H.; et al. Relation Between Serum Total Cholesterol Level and Cardiovascular Disease Stratified by Sex and Age Group: A Pooled Analysis of 65 594 Individuals From 10 Cohort Studies in Japan. Journal of the American Heart Association 2012, 1, e001974. [Google Scholar] [CrossRef] [PubMed]
- The Lipid Research Clinics Coronary Primary Prevention Trial Results: I. Reduction in Incidence of Coronary Heart Disease. JAMA 1984, 251, 351–364. [CrossRef]
- Castelli, W.P.; Garrison, R.J.; Wilson, P.W.F.; Abbott, R.D.; Kalousdian, S.; Kannel, W.B. Incidence of Coronary Heart Disease and Lipoprotein Cholesterol Levels: The Framingham Study. JAMA 1986, 256, 2835–2838. [Google Scholar] [CrossRef] [PubMed]
- Castelli, W.P.; Anderson, K.; Wilson, P.W.F.; Levy, D. Lipids and Risk of Coronary Heart Disease The Framingham Study. Annals of Epidemiology 1992, 2, 23–28. [Google Scholar] [CrossRef] [PubMed]
- Doucet, J.; Gourdy, P.; Meyer, L.; Benabdelmoumene, N.; Bourdel-Marchasson, I. Management of Glucose-Lowering Therapy in Older Adults with Type 2 Diabetes: Challenges and Opportunities. Clin Interv Aging 2023, 18, 1687–1703. [Google Scholar] [CrossRef]
- Alavinia, S.M.; van Duivenbooden, C.; Burdorf, A. Influence of Work-Related Factors and Individual Characteristics on Work Ability among Dutch Construction Workers. Scand J Work Environ Health 2007, 33, 351–357. [Google Scholar] [CrossRef]
- Ilmarinen, J.E. Aging Workers. Occup Environ Med 2001, 58, 546–552. [Google Scholar] [CrossRef] [PubMed]
- McGonagle, A.K.; Fisher, G.G.; Barnes-Farrell, J.L.; Grosch, J.W. Individual and Work Factors Related to Perceived Work Ability and Labor Force Outcomes. J Appl Psychol 2015, 100, 376–398. [Google Scholar] [CrossRef] [PubMed]
- Alcover, C.-M.; Topa, G. Work Characteristics, Motivational Orientations, Psychological Work Ability and Job Mobility Intentions of Older Workers. PLoS One 2018, 13, e0195973. [Google Scholar] [CrossRef] [PubMed]
- Guidetti, G.; Viotti, S.; Bruno, A.; Converso, D. Teachers’ Work Ability: A Study of Relationships between Collective Efficacy and Self-Efficacy Beliefs. Psychol Res Behav Manag 2018, 11, 197–206. [Google Scholar] [CrossRef] [PubMed]
- McElhaney, J.E.; Effros, R.B. Immunosenescence: What Does It Mean to Health Outcomes in Older Adults? Curr Opin Immunol 2009, 21, 418–424. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Reynolds, S.R.; Stolrow, H.G.; Chen, J.-Q.; Christensen, B.C.; Salas, L.A. Deciphering the Role of Immune Cell Composition in Epigenetic Age Acceleration: Insights from Cell-Type Deconvolution Applied to Human Blood Epigenetic Clocks. Aging Cell 2024, 23, e14071. [Google Scholar] [CrossRef] [PubMed]
- Wacka, E.; Nicikowski, J.; Jarmuzek, P.; Zembron-Lacny, A. Anemia and Its Connections to Inflammation in Older Adults: A Review. J Clin Med 2024, 13, 2049. [Google Scholar] [CrossRef]
- Bradycardia: Slow Heart Rate Available online:. Available online: https://www.heart.org/en/health-topics/arrhythmia/about-arrhythmia/bradycardia--slow-heart-rate (accessed on 2 July 2024).
- O’Brien, I.A.; O’Hare, P.; Corrall, R.J. Heart Rate Variability in Healthy Subjects: Effect of Age and the Derivation of Normal Ranges for Tests of Autonomic Function. Br Heart J 1986, 55, 348–354. [Google Scholar] [CrossRef] [PubMed]
- Fransquet, P.D.; Wrigglesworth, J.; Woods, R.L.; Ernst, M.E.; Ryan, J. The Epigenetic Clock as a Predictor of Disease and Mortality Risk: A Systematic Review and Meta-Analysis. Clinical Epigenetics 2019, 11, 62. [Google Scholar] [CrossRef]
- Liviero, F.; Scapellato, M.L.; Volpin, A.; Battistella, M.; Fabris, L.; Brischigliaro, L.; Folino, F.; Moretto, A.; Mason, P.; Pavanello, S. Long Term Follow-up of Heart Rate Variability in Healthcare Workers with Mild COVID-19. Front Neurol 2024, 15, 1403551. [Google Scholar] [CrossRef]
- Tiwari, R.; Kumar, R.; Malik, S.; Raj, T.; Kumar, P. Analysis of Heart Rate Variability and Implication of Different Factors on Heart Rate Variability. Curr Cardiol Rev 2021, 17, e160721189770. [Google Scholar] [CrossRef]
- Tang, B.; Li, X.; Wang, Y.; Sjölander, A.; Johnell, K.; Thambisetty, M.; Ferrucci, L.; Reynolds, C.A.; Finkel, D.; Jylhävä, J.; et al. Longitudinal Associations between Use of Antihypertensive, Antidiabetic, and Lipid-Lowering Medications and Biological Aging. Geroscience 2023, 45, 2065–2078. [Google Scholar] [CrossRef]
- Horvath, S. DNA Methylation Age of Human Tissues and Cell Types. Genome Biol 2013, 14, 3156. [Google Scholar] [CrossRef] [PubMed]
- Hannum, G.; Guinney, J.; Zhao, L.; Zhang, L.; Hughes, G.; Sadda, S.; Klotzle, B.; Bibikova, M.; Fan, J.-B.; Gao, Y.; et al. Genome-Wide Methylation Profiles Reveal Quantitative Views of Human Aging Rates. Molecular Cell 2013, 49, 359–367. [Google Scholar] [CrossRef]
- Müezzinler, A.; Zaineddin, A.K.; Brenner, H. A Systematic Review of Leukocyte Telomere Length and Age in Adults. Ageing Res Rev 2013, 12, 509–519. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Epel, E. Stress and Telomere Shortening: Insights from Cellular Mechanisms. Ageing Res Rev 2022, 73, 101507. [Google Scholar] [CrossRef]
- Ference, B.A.; Ginsberg, H.N.; Graham, I.; Ray, K.K.; Packard, C.J.; Bruckert, E.; Hegele, R.A.; Krauss, R.M.; Raal, F.J.; Schunkert, H.; et al. Low-Density Lipoproteins Cause Atherosclerotic Cardiovascular Disease. 1. Evidence from Genetic, Epidemiologic, and Clinical Studies. A Consensus Statement from the European Atherosclerosis Society Consensus Panel. Eur Heart J 2017, 38, 2459–2472. [Google Scholar] [CrossRef] [PubMed]
- Ornish, D.; Lin, J.; Daubenmier, J.; Weidner, G.; Epel, E.; Kemp, C.; Magbanua, M.J.M.; Marlin, R.; Yglecias, L.; Carroll, P.R.; et al. Increased Telomerase Activity and Comprehensive Lifestyle Changes: A Pilot Study. Lancet Oncol 2008, 9, 1048–1057. [Google Scholar] [CrossRef]
- Rehkopf, D.H.; Needham, B.L.; Lin, J.; Blackburn, E.H.; Zota, A.R.; Wojcicki, J.M.; Epel, E.S. Leukocyte Telomere Length in Relation to 17 Biomarkers of Cardiovascular Disease Risk: A Cross-Sectional Study of US Adults. PLoS Med 2016, 13, e1002188. [Google Scholar] [CrossRef]
- Mazidi, M.; Kengne, A.P.; Sahebkar, A.; Banach, M. Telomere Length Is Associated With Cardiometabolic Factors in US Adults. Angiology 2018, 69, 164–169. [Google Scholar] [CrossRef]
- Kim, D.; Li, A.A.; Ahmed, A. Leucocyte Telomere Shortening Is Associated with Nonalcoholic Fatty Liver Disease-Related Advanced Fibrosis. Liver Int 2018, 38, 1839–1848. [Google Scholar] [CrossRef] [PubMed]
- Révész, D.; Verhoeven, J.E.; Picard, M.; Lin, J.; Sidney, S.; Epel, E.S.; Penninx, B.W.J.H.; Puterman, E. Associations Between Cellular Aging Markers and Metabolic Syndrome: Findings From the CARDIA Study. J Clin Endocrinol Metab 2018, 103, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Baragetti, A.; Palmen, J.; Garlaschelli, K.; Grigore, L.; Pellegatta, F.; Tragni, E.; Catapano, A.L.; Humphries, S.E.; Norata, G.D.; Talmud, P.J. Telomere Shortening over 6 Years Is Associated with Increased Subclinical Carotid Vascular Damage and Worse Cardiovascular Prognosis in the General Population. J Intern Med 2015, 277, 478–487. [Google Scholar] [CrossRef] [PubMed]
- Zhan, Y.; Karlsson, I.K.; Karlsson, R.; Tillander, A.; Reynolds, C.A.; Pedersen, N.L.; Hägg, S. Exploring the Causal Pathway From Telomere Length to Coronary Heart Disease: A Network Mendelian Randomization Study. Circ Res 2017, 121, 214–219. [Google Scholar] [CrossRef]
- Rode, L.; Nordestgaard, B.G.; Bojesen, S.E. Peripheral Blood Leukocyte Telomere Length and Mortality among 64,637 Individuals from the General Population. J Natl Cancer Inst 2015, 107, djv074. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.; Janicki-Deverts, D.; Miller, G.E. Psychological Stress and Disease. JAMA 2007, 298, 1685–1687. [Google Scholar] [CrossRef]
- Bellon, M.; Nicot, C. Telomere Dynamics in Immune Senescence and Exhaustion Triggered by Chronic Viral Infection. Viruses 2017, 9, 289. [Google Scholar] [CrossRef]
- Chou, J.P.; Effros, R.B. T Cell Replicative Senescence in Human Aging. Curr Pharm Des 2013, 19, 1680–1698. [Google Scholar] [CrossRef] [PubMed]
- Dowd, J.B.; Bosch, J.A.; Steptoe, A.; Jayabalasingham, B.; Lin, J.; Yolken, R.; Aiello, A.E. Persistent Herpesvirus Infections and Telomere Attrition Over 3 Years in the Whitehall II Cohort. J Infect Dis 2017, 216, 565–572. [Google Scholar] [CrossRef]
- Aubert, G.; Baerlocher, G.M.; Vulto, I.; Poon, S.S.; Lansdorp, P.M. Collapse of Telomere Homeostasis in Hematopoietic Cells Caused by Heterozygous Mutations in Telomerase Genes. PLoS Genet 2012, 8, e1002696. [Google Scholar] [CrossRef]
- Al-Attiyah, R.; Safar, H.A.; Botras, L.; Botras, M.; Al-Kandari, F.; Chehadeh, W.; Mustafa, A.S. Immune Cells Profiles In The Peripheral Blood Of Patients With Moderate To Severe COVID-19 And Healthy Subjects With and Without Vaccination With The Pfizer-BioNTech mRNA Vaccine. Front Immunol 2022, 13, 851765. [Google Scholar] [CrossRef]
- Berentschot, J.C.; Drexhage, H.A.; Aynekulu Mersha, D.G.; Wijkhuijs, A.J.M.; GeurtsvanKessel, C.H.; Koopmans, M.P.G.; Voermans, J.J.C.; Hendriks, R.W.; Nagtzaam, N.M.A.; de Bie, M.; et al. Immunological Profiling in Long COVID: Overall Low Grade Inflammation and T-Lymphocyte Senescence and Increased Monocyte Activation Correlating with Increasing Fatigue Severity. Front. Immunol. 2023, 14. [Google Scholar] [CrossRef]
- Fasching, C.L. Telomere Length Measurement as a Clinical Biomarker of Aging and Disease. Crit Rev Clin Lab Sci 2018, 55, 443–465. [Google Scholar] [CrossRef]
- Pavanello, S.; Campisi, M.; Fabozzo, A.; Cibin, G.; Tarzia, V.; Toscano, G.; Gerosa, G. The Biological Age of the Heart Is Consistently Younger than Chronological Age. Sci Rep 2020, 10, 10752. [Google Scholar] [CrossRef] [PubMed]
- Pavanello, S.; Campisi, M.; Rigotti, P.; Bello, M.D.; Nuzzolese, E.; Neri, F.; Furian, L. DNA Methylation - and Telomere - Based Biological Age Estimation as Markers of Biological Aging in Donors Kidneys. Front Med (Lausanne) 2022, 9, 832411. [Google Scholar] [CrossRef]
- Binnie, A.; Walsh, C.J.; Hu, P.; Dwivedi, D.J.; Fox-Robichaud, A.; Liaw, P.C.; Tsang, J.L.Y.; Batt, J.; Carrasqueiro, G.; Gupta, S.; et al. Epigenetic Profiling in Severe Sepsis: A Pilot Study of DNA Methylation Profiles in Critical Illness. Crit Care Med 2020, 48, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Güiza, F.; Vanhorebeek, I.; Verstraete, S.; Verlinden, I.; Derese, I.; Ingels, C.; Dulfer, K.; Verbruggen, S.C.; Garcia Guerra, G.; Joosten, K.F.; et al. Effect of Early Parenteral Nutrition during Paediatric Critical Illness on DNA Methylation as a Potential Mediator of Impaired Neurocognitive Development: A Pre-Planned Secondary Analysis of the PEPaNIC International Randomised Controlled Trial. Lancet Respir Med 2020, 8, 288–303. [Google Scholar] [CrossRef]
- Balnis, J.; Madrid, A.; Hogan, K.J.; Drake, L.A.; Chieng, H.C.; Tiwari, A.; Vincent, C.E.; Chopra, A.; Vincent, P.A.; Robek, M.D.; et al. Blood DNA Methylation and COVID-19 Outcomes. Clin Epigenetics 2021, 13, 118. [Google Scholar] [CrossRef] [PubMed]
- Morselli, M.; Farrell, C.; Montoya, D.; Gören, T.; Sabırlı, R.; Türkçüer, İ.; Kurt, Ö.; Köseler, A.; Pellegrini, M. DNA Methylation Profiles in Pneumonia Patients Reflect Changes in Cell Types and Pneumonia Severity. Epigenetics 2022, 17, 1646–1660. [Google Scholar] [CrossRef]
- Saiz, M.L.; DeDiego, M.L.; López-García, D.; Corte-Iglesias, V.; Baragaño Raneros, A.; Astola, I.; Asensi, V.; López-Larrea, C.; Suarez-Alvarez, B. Epigenetic Targeting of the ACE2 and NRP1 Viral Receptors Limits SARS-CoV-2 Infectivity. Clin Epigenetics 2021, 13, 187. [Google Scholar] [CrossRef]
- Wang, Q.; Codd, V.; Raisi-Estabragh, Z.; Musicha, C.; Bountziouka, V.; Kaptoge, S.; Allara, E.; Angelantonio, E.D.; Butterworth, A.S.; Wood, A.M.; et al. Shorter Leukocyte Telomere Length Is Associated with Adverse COVID-19 Outcomes: A Cohort Study in UK Biobank. EBioMedicine 2021, 70, 103485. [Google Scholar] [CrossRef]
- Mahmoodpoor, A.; Sanaie, S.; Roudbari, F.; Sabzevari, T.; Sohrabifar, N.; Kazeminasab, S. Understanding the Role of Telomere Attrition and Epigenetic Signatures in COVID-19 Severity. Gene 2022, 811, 146069. [Google Scholar] [CrossRef] [PubMed]
- Aviv, A. Telomeres and COVID-19. FASEB J 2020, 34, 7247–7252. [Google Scholar] [CrossRef]
- Sanchez-Vazquez, R.; Guío-Carrión, A.; Zapatero-Gaviria, A.; Martínez, P.; Blasco, M.A. Shorter Telomere Lengths in Patients with Severe COVID-19 Disease. Aging (Albany NY) 2021, 13, 1–15. [Google Scholar] [CrossRef]
- Froidure, A.; Mahieu, M.; Hoton, D.; Laterre, P.-F.; Yombi, J.C.; Koenig, S.; Ghaye, B.; Defour, J.-P.; Decottignies, A. Short Telomeres Increase the Risk of Severe COVID-19. Aging (Albany NY) 2020, 12, 19911–19922. [Google Scholar] [CrossRef]
- Martínez, P.; Sánchez-Vazquez, R.; Saha, A.; Rodriguez-Duque, M.S.; Naranjo-Gonzalo, S.; Osorio-Chavez, J.S.; Villar-Ramos, A.V.; Blasco, M.A. Short Telomeres in Alveolar Type II Cells Associate with Lung Fibrosis in Post COVID-19 Patients with Cancer. Aging 2023, 15, 4625–4641. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, A.; Flores-Gonzalez, J.; Buendia-Roldan, I.; Chavez-Galan, L. Telomere Shortening and Its Association with Cell Dysfunction in Lung Diseases. International Journal of Molecular Sciences 2022, 23, 425. [Google Scholar] [CrossRef]
- Bejaoui, Y.; Humaira Amanullah, F.; Saad, M.; Taleb, S.; Bradic, M.; Megarbane, A.; Ait Hssain, A.; Abi Khalil, C.; El Hajj, N. Epigenetic Age Acceleration in Surviving versus Deceased COVID-19 Patients with Acute Respiratory Distress Syndrome Following Hospitalization. Clin Epigenetics 2023, 15, 186. [Google Scholar] [CrossRef] [PubMed]
- Raghuraman, S.; Donkin, I.; Versteyhe, S.; Barrès, R.; Simar, D. The Emerging Role of Epigenetics in Inflammation and Immunometabolism. Trends Endocrinol Metab 2016, 27, 782–795. [Google Scholar] [CrossRef]
- Bayarsaihan, D. Epigenetic Mechanisms in Inflammation. J Dent Res 2011, 90, 9–17. [Google Scholar] [CrossRef]
- Ahmad, S.; Manzoor, S.; Siddiqui, S.; Mariappan, N.; Zafar, I.; Ahmad, A.; Ahmad, A. Epigenetic Underpinnings of Inflammation: Connecting the Dots between Pulmonary Diseases, Lung Cancer and COVID-19. Seminars in Cancer Biology 2022, 83, 384–398. [Google Scholar] [CrossRef]
- Stukas, S.; Hoiland, R.L.; Cooper, J.; Thiara, S.; Griesdale, D.E.; Thomas, A.D.; Orde, M.M.; English, J.C.; Chen, L.Y.C.; Foster, D.; et al. The Association of Inflammatory Cytokines in the Pulmonary Pathophysiology of Respiratory Failure in Critically Ill Patients With Coronavirus Disease 2019. Crit Care Explor 2020, 2, e0203. [Google Scholar] [CrossRef] [PubMed]
- Sungnak, W.; Huang, N.; Bécavin, C.; Berg, M.; Queen, R.; Litvinukova, M.; Talavera-López, C.; Maatz, H.; Reichart, D.; Sampaziotis, F.; et al. SARS-CoV-2 Entry Factors Are Highly Expressed in Nasal Epithelial Cells Together with Innate Immune Genes. Nat Med 2020, 26, 681–687. [Google Scholar] [CrossRef] [PubMed]
- Wellford, S.A.; Moseman, E.A. Olfactory Immune Response to SARS-CoV-2. Cell Mol Immunol 2024, 21, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Meinhardt, J.; Radke, J.; Dittmayer, C.; Franz, J.; Thomas, C.; Mothes, R.; Laue, M.; Schneider, J.; Brünink, S.; Greuel, S.; et al. Olfactory Transmucosal SARS-CoV-2 Invasion as a Port of Central Nervous System Entry in Individuals with COVID-19. Nat Neurosci 2021, 24, 168–175. [Google Scholar] [CrossRef] [PubMed]
- Butowt, R.; von Bartheld, C.S. Anosmia in COVID-19: Underlying Mechanisms and Assessment of an Olfactory Route to Brain Infection. Neuroscientist 2021, 27, 582–603. [Google Scholar] [CrossRef] [PubMed]
- Barnes, P.J. Inflammatory Mechanisms in Patients with Chronic Obstructive Pulmonary Disease. Journal of Allergy and Clinical Immunology 2016, 138, 16–27. [Google Scholar] [CrossRef]
- Barnes, P.J. Cellular and Molecular Mechanisms of Asthma and COPD. Clinical Science 2017, 131, 1541–1558. [Google Scholar] [CrossRef]
- da Silva, C.O.; de Souza Nogueira, J.; do Nascimento, A.P.; Victoni, T.; Bártholo, T.P.; da Costa, C.H.; Costa, A.M.A.; Valença, S. dos S.; Schmidt, M.; Porto, L.C. COPD Patients Exhibit Distinct Gene Expression, Accelerated Cellular Aging, and Bias to M2 Macrophages. Int J Mol Sci 2023, 24, 9913. [Google Scholar] [CrossRef]
- Kukrety, S.P.; Parekh, J.D.; Bailey, K.L. Chronic Obstructive Pulmonary Disease and the Hallmarks of Aging. Lung India 2018, 35, 321–327. [Google Scholar] [CrossRef]
- Shah, W.; Hillman, T.; Playford, E.D.; Hishmeh, L. Managing the Long Term Effects of Covid-19: Summary of NICE, SIGN, and RCGP Rapid Guideline. BMJ 2021, 372, n136. [Google Scholar] [CrossRef] [PubMed]
- Pavanello, S.; Stendardo, M.; Mastrangelo, G.; Bonci, M.; Bottazzi, B.; Campisi, M.; Nardini, M.; Leone, R.; Mantovani, A.; Boschetto, P. Inflammatory Long Pentraxin 3 Is Associated with Leukocyte Telomere Length in Night-Shift Workers. Front Immunol 2017, 8, 516. [Google Scholar] [CrossRef] [PubMed]
- Pavanello, S.; Stendardo, M.; Mastrangelo, G.; Casillo, V.; Nardini, M.; Mutti, A.; Campisi, M.; Andreoli, R.; Boschetto, P. Higher Number of Night Shifts Associates with Good Perception of Work Capacity and Optimal Lung Function but Correlates with Increased Oxidative Damage and Telomere Attrition. Biomed Res Int 2019, 2019, 8327629. [Google Scholar] [CrossRef]
- Ilmarinen, J. The Work Ability Index (WAI). Occupational Medicine 2007, 57. [Google Scholar] [CrossRef]
- Miller, M.R.; Hankinson, J.; Brusasco, V.; Burgos, F.; Casaburi, R.; Coates, A.; Crapo, R.; Enright, P.; van der Grinten, C.P.M.; Gustafsson, P.; et al. Standardisation of Spirometry. Eur Respir J 2005, 26, 319–338. [Google Scholar] [CrossRef] [PubMed]
- des Communautes Europeennes, C. Tables de Reference Pour Les Examens Spirometriques; Office Des Publications Officielle Des Communautes Europeennes: Luxembourg, 1971. [Google Scholar]
- Mancia, G.; Kreutz, R.; Brunström, M.; Burnier, M.; Grassi, G.; Januszewicz, A.; Muiesan, M.L.; Tsioufis, K.; Agabiti-Rosei, E.; Algharably, E.A.E.; et al. 2023 ESH Guidelines for the Management of Arterial Hypertension The Task Force for the Management of Arterial Hypertension of the European Society of Hypertension: Endorsed by the International Society of Hypertension (ISH) and the European Renal Association (ERA). Journal of Hypertension 2023, 41, 1874. [Google Scholar] [CrossRef] [PubMed]
- Tarvainen, M.P.; Niskanen, J.-P.; Lipponen, J.A.; Ranta-aho, P.O.; Karjalainen, P.A. Kubios HRV – Heart Rate Variability Analysis Software. Computer Methods and Programs in Biomedicine 2014, 113, 210–220. [Google Scholar] [CrossRef]
- Koizumi, K.; Terui, N.; Kollai, M. Effect of Cardiac Vagal and Sympathetic Nerve Activity on Heart Rate in Rhythmic Fluctuations. J Auton Nerv Syst 1985, 12, 251–259. [Google Scholar] [CrossRef]
- Moak, J.P.; Goldstein, D.S.; Eldadah, B.A.; Saleem, A.; Holmes, C.; Pechnik, S.; Sharabi, Y. Supine Low-Frequency Power of Heart Rate Variability Reflects Baroreflex Function, Not Cardiac Sympathetic Innervation. Heart Rhythm 2007, 4, 1523–1529. [Google Scholar] [CrossRef]
- Rahman, F.; Pechnik, S.; Gross, D.; Sewell, L.; Goldstein, D.S. Low Frequency Power of Heart Rate Variability Reflects Baroreflex Function, Not Cardiac Sympathetic Innervation. Clin Auton Res 2011, 21, 133–141. [Google Scholar] [CrossRef]
- Campisi, M.; Mastrangelo, G.; Mielżyńska-Švach, D.; Hoxha, M.; Bollati, V.; Baccarelli, A.A.; Carta, A.; Porru, S.; Pavanello, S. The Effect of High Polycyclic Aromatic Hydrocarbon Exposure on Biological Aging Indicators. Environmental Health 2023, 22, 27. [Google Scholar] [CrossRef] [PubMed]
- Pavanello, S.; Campisi, M.; Grassi, A.; Mastrangelo, G.; Durante, E.; Veronesi, A.; Gallucci, M. Longer Leukocytes Telomere Length Predicts a Significant Survival Advantage in the Elderly TRELONG Cohort, with Short Physical Performance Battery Score and Years of Education as Main Determinants for Telomere Elongation. J Clin Med 2021, 10, 3700. [Google Scholar] [CrossRef] [PubMed]
| PCS>12 weeks | PCS~1 year | P_Trend | |
|---|---|---|---|
| HCWs | 0.4605 | 0.3026 | 0.0663 |
| Women | 0.5741 | 0.3333 | 0.0204 |
| Men | 0.1818 | 0.2273 | 0.9999 |
| P_Trend | 0.0043 | 0.5238 |
| Symptoms | Up to 4 weeks after diagnosis n, (%) |
From 4 to 12 weeks after diagnosis n , (%) |
Over 12 weeks after diagnosis n, (%) |
P Chi2 | 1-year follow-up | P Chi2 | |
|---|---|---|---|---|---|---|---|
| General symptoms | |||||||
| Asthenia | 46 (60.53) | 34 (44.74) | 19 (25.00) | <0.0001 | 12 (15.79) | <0.0001 | |
| Fever | 46 (60.53) | 1 (1.32) | 0 (0.00) | <0.0001 | 0 (0.00) | Na | |
| Pain | 20 (26.32) | 8 (10.53) | 6 (7.89) | 0.0026 | 6 (7.89) | 0.0013 | |
| Respiratory symptoms | |||||||
| Dyspnoea | 11 (14.47) | 7 (9.21) | 7 (9.21) | 0.4873 | 5 (6.58) | 0.4217 | |
| Cough | 36 (47.37) | 7 (9.21) | 4 (5.26) | <0.0001 | 2 (2.63) | <0.0001 | |
| Rhinitis | 2 (2.63) | 1 (1.32) | 0 (0.00) | Na | 0 (0.00) | Na | |
| Cardiovascular symptoms | |||||||
| Chest pain | 8 (10.53) | 1 (1.32) | 1 (1.32) | 0.0059 | 0 (0.00) | Na | |
| Thoracic oppression | 14 (18.12) | 5 (6.58) | 2 (2.63) | 0.0022 | 0 (0.00) | Na | |
| Palpitations | 11 (14.47) | 7 (9.21) | 6 (7.89) | 0.3761 | 3 (3.95) | 0.1495 | |
| Neurological symptoms | |||||||
| Brain fog | 10 (13.16) | 7 (9.21) | 3 (3.95) | 0.1316 | 2 (2.63) | 0.0453 | |
| Headache | 37 (48.78) | 10 (13.16) | 5 (6.58) | <0.0001 | 3 (3.95) | <0.0001 | |
| Delirium | 2 (2.63) | 0 (0.00) | 0 (0.00) | Na | 0 (0.00) | Na | |
| Sleep disorders | 20 (26.32) | 11 (14.47) | 10 (13.16) | 0.0668 | 8 (10.53) | 0.0412 | |
| Peripheral neuropathies | 4 (5.26) | 4 (5.26) | 3 (3.95) | 0.9089 | 3 (3.95) | 0.9601 | |
| Loss of concentration | 14 (18.42) | 13 (17.11) | 11 (14.47) | 0.8017 | 9 (11.84) | 0.6858 | |
| Memory problems | 11 (14.47) | 14 (18.42) | 13 (17.11) | 0.8017 | 11 (14.47) | 0.8833 | |
| Dizziness | 5 (6.58) | 0 (0.00) | 0 (0.00) | 0.0056 | 0 (0.00) | Na | |
| Hypersomnia | 2 (2.63) | 0 (0.00) | 0 (0.00) | Na | 0 (0.00) | Na | |
| Gastrointestinal symptoms | |||||||
| Diarrhoea | 18 (23.68) | 1 (1.32) | 1 (1.32) | 0.0059 | 1 (1.32) | <0.0001 | |
| Abdominal pain | 5 (6.58) | 0 (0.00) | 0 (0.00) | 0.006 | 0 (0.00) | Na | |
| Nausea | 9 (11.84) | 0 (0.00) | 0 (0.00) | <0.0001 | 0 (0.00) | Na | |
| Anorexia | 16 (21.05) | 0 (0.00) | 0 (0.00) | <0.0001 | 0 (0.00) | Na | |
| Vomiting | 2 (2.63) | 0 (0.00) | 0 (0.00) | Na | 0 (0.00) | Na | |
| Other | 2 (2.63) | 0 (0.00) | 0 (0.00) | Na | 0 (0.00) | Na | |
| Musculoskeletal symptoms | |||||||
| Joint pain | 39 (51.32) | 15 (19.74) | 13 (17.11) | <0.0001 | 8 (10.53) | <0.0001 | |
| Muscle pain | 37 (48.68) | 17 (22.37) | 8 (10.53) | <0.0001 | 6 (7.89) | <0.0001 | |
| Psychological or psychiatric symptoms | |||||||
| Anxiety | 11 (14.47) | 9 (11.84) | 7 (9.21) | 0.6040 | 5 (6.58) | 0.4245 | |
| Depression | 7 (9.21) | 5 (6.58) | 4 (5.26) | 0.6247 | 0 (0.00) | Na | |
| Other | 1 (1.32) | 0 (0.00) | 0 (0.00) | Na | 0 (0.00) | Na | |
| Otorhinolaryngological symptoms | |||||||
| Ageusia | 40 (52.63) | 10 (13.16) | 3 (3.95) | <0.0001 | 2 (2.63) | <0.0001 | |
| Anosmia | 41 (53.95) | 12 (15.79) | 4 (5.26) | <0.0001 | 2 (2.63) | <0.0001 | |
| Sore throat | 8 (10.53) | 1 (1.32) | 1 (1.32) | 0.0059 | 0 (0.00) | Na | |
| Otalgia | 1 (1.32) | 0 (0.00) | 0 (0.00) | 0.3663 | 0 (0.00) | Na | |
| Dermatological signs | 3 (3.95) | 2 (2.63) | 3 (3.95) | 0.8785 | 2 (2.63) | 0.9374 | |
| Ocular symptoms | 11 (14.47) | 5 (6.58) | 4 (5.26) | 0.0947 | 2 (2.63) | 0.0318 | |
| Other | 10 (13.16) | 5 (6.58) | 4 (5.26) | 0.1687 | 3 (3.95) | 0.1280 | |
| Age | Blood leukocytes DNAmAge (years) | Blood leukocytes AgeAcc (years) | Blood leukocytes TL (T/S) | |
| Mean± SD | 46.00±12.88 | -2.59±3.47 | 1.12±4.37 | 1.20±0.06 |
| Variable | Mean±SD | N subjects | % |
|---|---|---|---|
| Age [years] | 44.64±11.75 | ||
| Gender [n (%)] | |||
| M | 22 | 28.95 | |
| F | 54 | 71.05 | |
| Marital status [n (%)] | |||
| Not married | 31 | 40.79 | |
| Married | 40 | 52.63 | |
| Divorced | 5 | 6.58 | |
| Widower | 0 | 0.00 | |
| Years of education [years] | 16.67±5.57 | ||
| BMI [Kg/m2] | 24.48±4.01 | ||
| Systolic blood pressure [mmHg] | 123.55±13.74 | ||
| Diastolic blood pressure [mmHg] | 77.43±10.55 | ||
| EMPLOYMENT ANAMNESIS | |||
| Professional position: [n (%)] | |||
| Healthcare assistant | 19 | 25.00 | |
| Nurse | 30 | 39.47 | |
| Doctor | 19 | 25.00 | |
| Resident | 0 | 0.00 | |
| Other | 8 | 10.53 | |
| Total years of work [years] | 19.74±12.65 | ||
| Years of work in the current job [years] | 9.78±9.23 | ||
| Performance of night shifts [n (%)] | 43 | 56.58 | |
| Frequency of night shifts/month | |||
| 0 | 33 | 43.42 | |
| From 1 to 4 | 13 | 17.11 | |
| >5 | 30 | 39.47 | |
| Work ability - WAI (n=68) [n (%)] | |||
| Poor (7-27) | 5 | 7.35 | |
| Moderate (28-36) | 15 | 22.06 | |
| Good (37-43) | 5 | 33.82 | |
| Excellent (44-49) | 25 | 36.76 | |
| PHYSIOLOGICAL ANAMNESIS AND LIFESTYLE | |||
| Chronic diseases [n (%)] | |||
| 0 | 28 | 36.84 | |
| 1 | 16 | 21.05 | |
| ≥2 | 32 | 42.11 | |
| Tobacco habit [n (%)] | |||
| Smoker | 10 | 13.16 | |
| Ex-smoker | 13 | 17.11 | |
| Non-smoker | 53 | 69.74 | |
| Pack/years [(cigarettes/20) per years of smoking] | 1.63±4.26 | ||
| Alcohol consumption [n (%)] | 56 | 73.68 | |
| Alcohol consumption [u.a./die] | 106.89±155.76 | ||
| Binge drinking habit [n (%)] | 1 | 1.32 | |
| Meals with grilled meat or pizza cooked in a wood-fired oven/year [n of meals/year] | 80.63±59.72 | ||
| Frequency of fruit meals/day [n (%)] | |||
| <2 | 39 | 51.32 | |
| >2 | 37 | 48.68 | |
| Frequency of vegetable meals/day [n (%)] | |||
| <2 | 32 | 42.11 | |
| >2 | 44 | 57.89 | |
| IPAQ score (n=69) [n (%)] | |||
| <700 | 11 | 15.94 | |
| ≥700; ≤ 2519 | 20 | 28.99 | |
| >2520 | 38 | 55.07 | |
| Indoor pollution* | |||
| 0 | 52 | 68.42 | |
| 1 | 19 | 25.00 | |
| 2 | 5 | 6.58 | |
| 3 | 0 | 0.00 | |
| Living area [n (%)] | |||
| Urban/ peripheral area | 50 | 65.79 | |
| Rural area | 26 | 34.21 | |
| Traffic in the living area [n (%)] | |||
| Continuous intense for a good part of the day | 23 | 30.26 | |
| Intermittent intense | 30 | 39.47 | |
| Scarce or absent | 23 | 30.26 | |
| BASIC BIOCHEMISTRY PARAMETERS | |||
| Leukocytes (103/ml) | 6.19±1.59 | ||
| Blood red cells (103/ml) | 4.70±0.43 | ||
| Platelet count (103/ml) | 267.08±57.13 | ||
| Neutrophils (103/ml) | 3.41±1.18 | ||
| Lymphocytes (103/ml) | 2.17±0.58 | ||
| Monocytes (103/ml) | 0.51±0.13 | ||
| Eosinophils (103/ml) | 0.27±0.97 | ||
| Basophils (103/ml) | 0.03±0.03 | ||
| Hemoglobin (g/dl) | 138.18±13.79 | ||
| Glycemia (mg/dl) | 92.12±14.31 | ||
| Cholesterol (mg/dl) | 194.00±14.27 | ||
| Triglycerides (mg/dl) | 93.11±42.18 | ||
| HDL (mg/dl) | 59.85±15.58 | ||
| LDL (mg/dl) | 123.04±30.74 | ||
| Creatinine (mg/dl) | 1.49±6.26 | ||
| Bilirubin (umol/L) | 9.60±10.58 | ||
| LIVER FUNCTION | |||
| AST/GOT (U/L) | 23.23±6.85 | ||
| ALT/GPT (U/L) | 21.8±12.73 | ||
| GGT (U/L) | 9.60±10.58 | ||
| INFLAMMATION | |||
| PCR (mg/L) | 5.19±3.23 | ||
| LUNG FUNCTION | |||
| FEV1 (L) | 3.30±0.88 | ||
| FEV1 (%) | 101.76±13.54 | ||
| FVC (L) | 4.10±0.92 | ||
| FVC (%) | 95.56±12.68 | ||
| FEV1/VC (%) | 0.82±0.06 | ||
| TLC (L) | 5.55±1.26 | ||
| TLC (%) | 96.47±15.70 | ||
| RV (L) | 1.59±0.40 | ||
| RV (%) | 107.36±23.43 | ||
| HEART RATE | |||
| Mean HR | 68.05±9.71 | ||
| HEART RATE VARIABILITY | |||
| nLF: 0.04 - 0.15 Hz | 52.38±18.20 | ||
| nHF: 0.15 - 0.40 Hz | 47.31±18.41 | ||
| LF/HF ratio | 1.54±1.30 | ||
| SDNN | 35.16±25.45 | ||
| RMSSD | 36.89±34.19 |
| Blood Leukocytes DNAmAge (years) | b | r | t value | P | |
| Age | b1 = 0.74399 | r = 0.911647 | t = 17.884209 | <0.0001 | |
| Gender (male) | b2 = 2.703646 | r = 0.298937 | t = 2.525593 | 0.014 | |
| Chronic diseases (0=no; 1=yes) | b3 = 1.860584 | r = 0.266824 | t = 2.23213 | 0.0291 | |
| FEV1 (L) | b4 = -2.219589 | r = -0.382602 | t = -3.338666 | 0.0014 | |
| Lymphocytes (109/L) | b5 = -2.017434 | r = -0.36957 | t = -3.206587 | 0.0021 | |
| Blood Leukocytes TL (T/S) | Age | b1 = -0.011446 | r = -0.363521 | t = -3.146031 | 0.0025 |
| Gender (male) | b2 = 0.004352 | r = 0.005765 | t = 0.046482 | 0.9631 | |
| Chronic diseases (0=no; 1=yes) | b3 = 0.131573 | r = 0.218459 | t = 1.804868 | 0.0757 | |
| FEV1 (L) | b4 = -0.023505 | r = -0.05008 | t = -0.404266 | 0.6873 | |
| Lymphocytes (109/L) | b5 = 0.120086 | r = 0.261295 | t = 2.182445 | 0.0327 |
| Blood Leukocytes DNAmAge (years) | b | r | t value | P | |
| Occupation (0=HA; 1=N; 2=D; 3=R; 4=T and A) | b1 = -0.727891 | r = -0.097697 | t = -0.785337 | 0.4352 | |
| Night shift work (0=no; 1=yes) | b2 = -5.367146 | r = -0.263551 | t = -2.185686 | 0.0325 | |
| WAI | b3 = -0.617807 | r = -0.382432 | t = -3.311157 | 0.0015 | |
| Blood Leukocytes TL (T/S) | Occupation (0=HA; 1=N; 2=D; 3=R; 4=T and A) | b1 = 0.064564 | r = 0.268124 | t = 2.226516 | 0.0295 |
| Night shift work (0=no; 1=yes) | b2 = 0.102616 | r = 0.164683 | t = 1.3357 | 0.1864 | |
| WAI | b3 = 0.007125 | r = 0.150818 | t = 1.220504 | 0.2268 |
| Blood Leukocytes DNAmAge (years) | b | r | t value | P | |
| haemoglobin (g/dL) | b1 = -0.205578 | r = -0.300885 | t = -2.484288 | 0.0157 | |
| glycaemia (mg/dL) | b2 = 0.2006 | r = 0.329513 | t = 2.748063 | 0.0078 | |
| cholesterol (mg/dL) | b3 = 0.006919 | r = 0.022835 | t = 0.179853 | 0.8579 | |
| triglycerides (mg/dL) | b4 = 0.003211 | r = 0.01292 | t = 0.101744 | 0.9193 | |
| HDL (mg/dL) | b5 = 0.063761 | r = 0.098495 | t = 0.779342 | 0.4387 | |
| LDL (mg/dL) | b6 = 0.172631 | r = 0.388744 | t = 3.322289 | 0.0015 | |
| creatinine (mg/dL) | b7 = 0.267526 | r = 0.205048 | t = 1.649598 | 0.1041 | |
| bilirubin (mg/dL) | b8 = 0.022068 | r = 0.028541 | t = 0.224826 | 0.8229 | |
| Blood Leukocytes TL (T/S) | haemoglobin (g/dL) | b1 = 0.003138 | r = 0.141452 | t = 1.125107 | 0.2649 |
| glycaemia (mg/dL) | b2 = -0.00175 | r = -0.089968 | t = -0.711294 | 0.4796 | |
| cholesterol (mg/dL) | b3 = -0.000016 | r = -0.001611 | t = -0.012687 | 0.9899 | |
| triglycerides (mg/dL) | b4 = 0.001514 | r = 0.177891 | t = 1.42342 | 0.1596 | |
| HDL (mg/dL) | b5 = -0.000176 | r = -0.008083 | t = -0.06365 | 0.9495 | |
| LDL (mg/dL) | b6 = -0.003492 | r = -0.245463 | t = -1.993777 | 0.0506 | |
| creatinine (mg/dL) | b7 = -0.0017 | r = -0.039478 | t = -0.311094 | 0.7568 | |
| bilirubin (mg/dL) | b8 = -0.000485 | r = -0.018608 | t = -0.146549 | 0.884 |
| Blood Leukocytes DNAmAge (years) | b | r | t value | P | |
| SDNN | b1 = -0.311272 | r = -0.186422 | t = -1.564708 | 0.1223 | |
| RMSSD | b2 = 0.14605 | r = 0.114644 | t = 0.95165 | 0.3446 | |
| Mean HR | b3 = -0.403812 | r = -0.356467 | t = -3.14618 | 0.0025 | |
| Drugs affecting HRV (0=no; 1=yes) | b4 = 8.905208 | r = 0.297306 | t = 2.567761 | 0.0124 | |
| Blood Leukocytes TL (T/S) | SDNN | b1 = 0.008358 | r = 0.166005 | t = 1.388176 | 0.1696 |
| MSSD | b2 = -0.006522 | r = -0.167858 | t = -1.404117 | 0.1648 | |
| Mean HR | b3 = 0.005025 | r = 0.154982 | t = 1.293644 | 0.2002 | |
| Drugs affecting HRV (0=no; 1=yes) | b4 = 0.154968 | r = 0.176234 | t = 1.476368 | 0.1445 |
| Post COVID-19 subjects | |||||
| N=17 | Age | Blood AgeAcc (years) | IS AgeAcc (years) | Predicted Blood TL (T/S) | Predicted IS TL (T/S) |
| Mean±SD | 46.00±12.88 | -2.59±3.47*§ | 1.12±4.37§ | 1.20±0.06*§ | 0.93±0.02§ |
| COPD patients | |||||
| N=7 | Age | Blood AgeAcc (years) | IS AgeAcc (years) | Predicted Blood TL (T/S) | Predicted IS TL (T/S) |
| Mean±SD | 72.43±6.00 | -10.29±3.50* | -4.29±5.15 | 1.31±0.03* | 0.97±0.01 |
| Variable | Mean±SD | N subjects | % |
| Duration of SARS-CoV-2 infection [days] | 17.81±6.03 | ||
| Diagnosis of SARS-CoV-2 pneumonia | |||
| Yes | 4 | 5.26 | |
| No | 72 | 94.74 | |
| Hospitalisation for COVID-19 [n (%)] | 5 | 6.58 | |
| Drug therapy during acute infection [n (%)] | |||
| Antibiotics | 14 | 18.42 | |
| Inhaled Corticosteroids | 2 | 2.63 | |
| Systemic Corticosteroids | 4 | 5.26 | |
| NSAID or paracetamol | 49 | 64.47 | |
| Hydroxychloroquine | 8 | 10.53 | |
| Tocilizumab | 1 | 1.32 | |
| Antiviral | 2 | 2.63 | |
| Anticoagulant | 3 | 3.95 | |
| Other | 7 | 9.21 | |
| None | 22 | 28.95 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




