Submitted:
01 July 2024
Posted:
01 July 2024
You are already at the latest version
Abstract
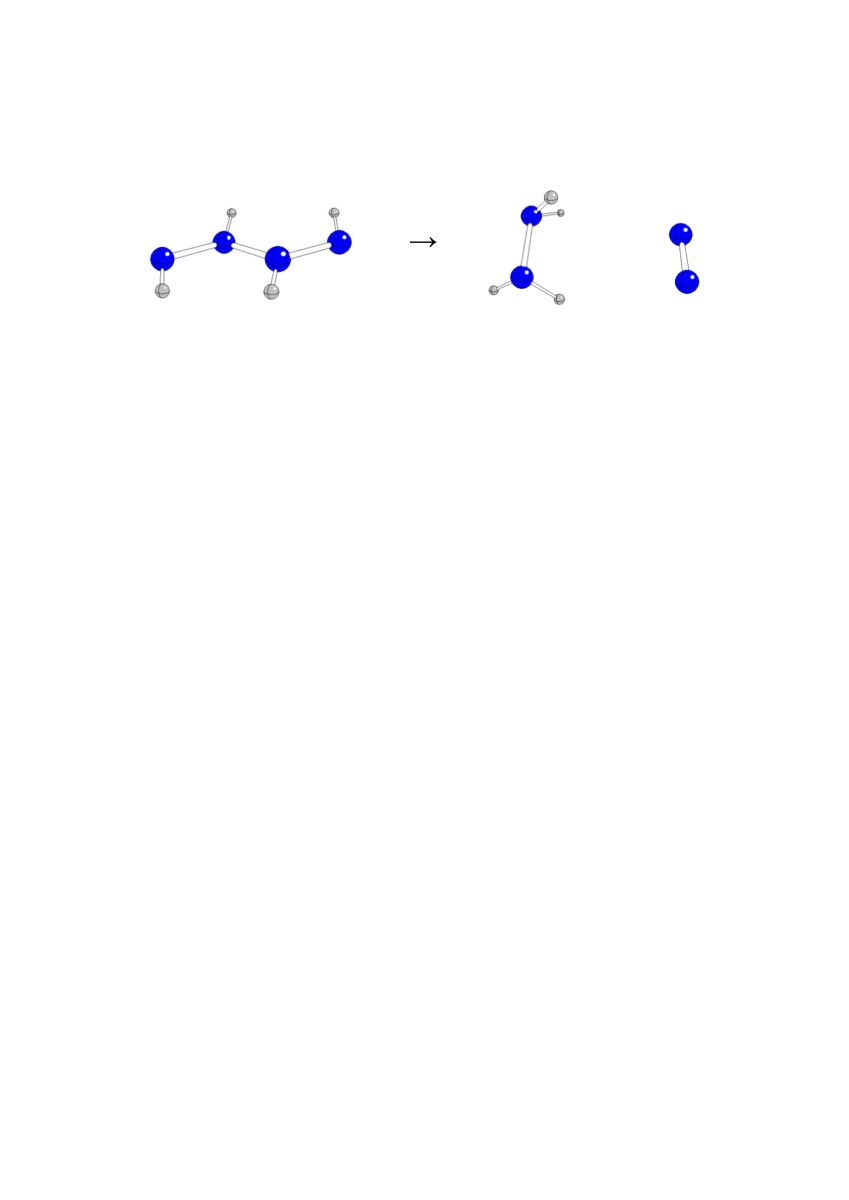
Keywords:
1. Introduction
2. Results
3. Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Davis, S. M.; Yilmaz, N. Advances in Hypergolic Propellants: Ignition, Hydrazine, and Hydrogen Peroxide Research. Adv. Aerospace Eng. 2014, 729313. [CrossRef]
- Lauko, L.; Hudec, R.; Lenghartova, K.; Manova, A.; Cacho, F.; Beinrohr, E. Simple Electrochemical Determination of Hydrazine in Water. Pol. J. Environ. Stud. 2015, 24(4), 1659-1666. [CrossRef] [PubMed]
- Nguyen, H. V. N.; Chenoweth, J. A.; Bebarta, V. S.; Albertson, T. E.; Nowadly, C. D. The Toxicity, Pathophysiology, and Treatment of Acute Hydrazine Propellant Exposure: A Systematic Review. Military Med. 2021, 186, e319–e326. [CrossRef] [PubMed]
- Gutbier, A.; Neudlinger, K. Katalyse des Hydrazins durch Platinmohr. Z. Phys. Chem. 1913, 84A, 203-249. [CrossRef]
- Heitbaum, J.; Vielstich, W. Untersuchungen zur Ammoniakbildung bei der Hydrazinzersetzung, Electrochim. Acta 1972, 17, 1529-1542.
- Petek, M.; Bruckenstein, S. An Isotopic Labeling Investigation of the Mechanism of the Electrooxidation of Hydrazine at Platinum. An Electrochemical Mass Spectrometric Study. Electroanal. Chem. Interrac. Electrochem. 1973, 47, 329-333. [CrossRef]
- Higginson, W. C. E.; Sutton, D. The Oxidation of Hydrazine in Aqueous Solution. Part II. The Use of 15N as a Tracer in the Oxidation of Hydrazine. J. Chem. Soc. 1953, 1402 -1406.
- Cahn, J. W.; Powell, R. E. Oxidation of Hydrazine in Solution. J. Am. Chem. Soc, 1954, 76, 2568-2572. [CrossRef]
- Karp, S.; Meites, L. The Voltammetric Characteristics and Mechanism of Electrooxidation of Hydrazine, J. Am. Chem. Soc. 1962, 84, 906 – 912. [CrossRef]
- Wiberg, N.; Bachhuber, H.; Fischer, G. Isolation of Pure Diimine. Angew. Chem. Int. Ed. 1972, 11, 829-830. [CrossRef]
- Wiberg, N.; Bayer, H.; Bachhuber, H. Isolation of Tetrazene, N4H4. Angew. Chem. Int. Ed. 1975, 14, 177 – 178. [CrossRef]
- Veith, M.; Schlemmer, G. Die Kristall- und Molekulstruktur von trans-Tetrazen-2 (N4H4) bei -90°C. Z. anorg. allg. Chem. 1982, 494, 7-19. [CrossRef]
- Zhao, M.; Gimarc, B. M. Strain Energies of (NH), Rings, n = 3-8. J. Phys. Chem. 1994, 98, 7497-7503. [CrossRef]
- Ball, D. W. High-level ab initio calculations on hydrogen– nitrogen compounds. Thermochemistry of tetrazetidine, N4H4. J. Mol. Struct. (Theochem) 2002, 619, 37–43. [CrossRef]
- Feng, Y.; Zhu, H.; Zhang, Q.; Zhao, Q.; Zhao, H.; Suo, B.; Zhai, G.; Zou, W.; Han, H.; Song, Q.; Li, J.; Li, Y. Theoretical study on the two novel planar-type all-nitrogen N44- anions: Structures, stability, reaction rate and their stable mechanisms via protonation. Chem. Phys. Let. 2021, 771, 138519. [CrossRef]
- Hu, A.; Zhang, F. A hydronitrogen solid: high pressure ab initio evolutionary structure searches. J. Phys. Cond. Mat. 2011, 23, 022203. [CrossRef] [PubMed]
- Breza, M.; Manova, A. Hydrazine Oxidation in Aqueous Solutions I: N4H6 Decomposition. Inorganics 2023, 11, 413. [CrossRef]
- Bader, R.F.W. Atoms in Molecules: A Quantum Theory; Clarendon Press: Oxford, UK, 1990; ISBN 9780198558651.
- Scuseria, G.E.; Janssen, C.L.; Schaefer, H.F., III. An efficient reformulation of the closed-shell coupled cluster single and double excitation (CCSD) equations. J. Chem. Phys. 1988, 89, 7382–7387. [CrossRef]
- Dunning, T.H., Jr. Gaussian basis sets for use in correlated molecular calculations. I. The atoms boron through neon and hydrogen. J. Chem. Phys. 1989, 90, 1007–1023.
- Marenich, A.V.; Cramer, C.J.; Truhlar, D.G. Universal solvation model based on solute electron density and a continuum model of the solvent defined by the bulk dielectric constant and atomic surface tensions. J. Phys. Chem. B 2009, 113, 6378–6396. [CrossRef] [PubMed]
- Frisch, G.W.; Trucks, M.J.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 16, Revision B.01; Gaussian, Inc.: Wallingford, CT, USA, 2016.
- Biegler-König, F.; Schönbohm, J.; Bayles, D. AIM2000—A Program to Analyze and Visualize Atoms in Molecules. J. Comput. Chem. 2001, 22, 545–559.
- Ugliengo, P. MOLDRAW: A Program to Display and Manipulate Molecular and Crystal Structures, University Torino, Torino. 2012. Available online: https://www.moldraw.software.informer.com (accessed on 9 September 2019).
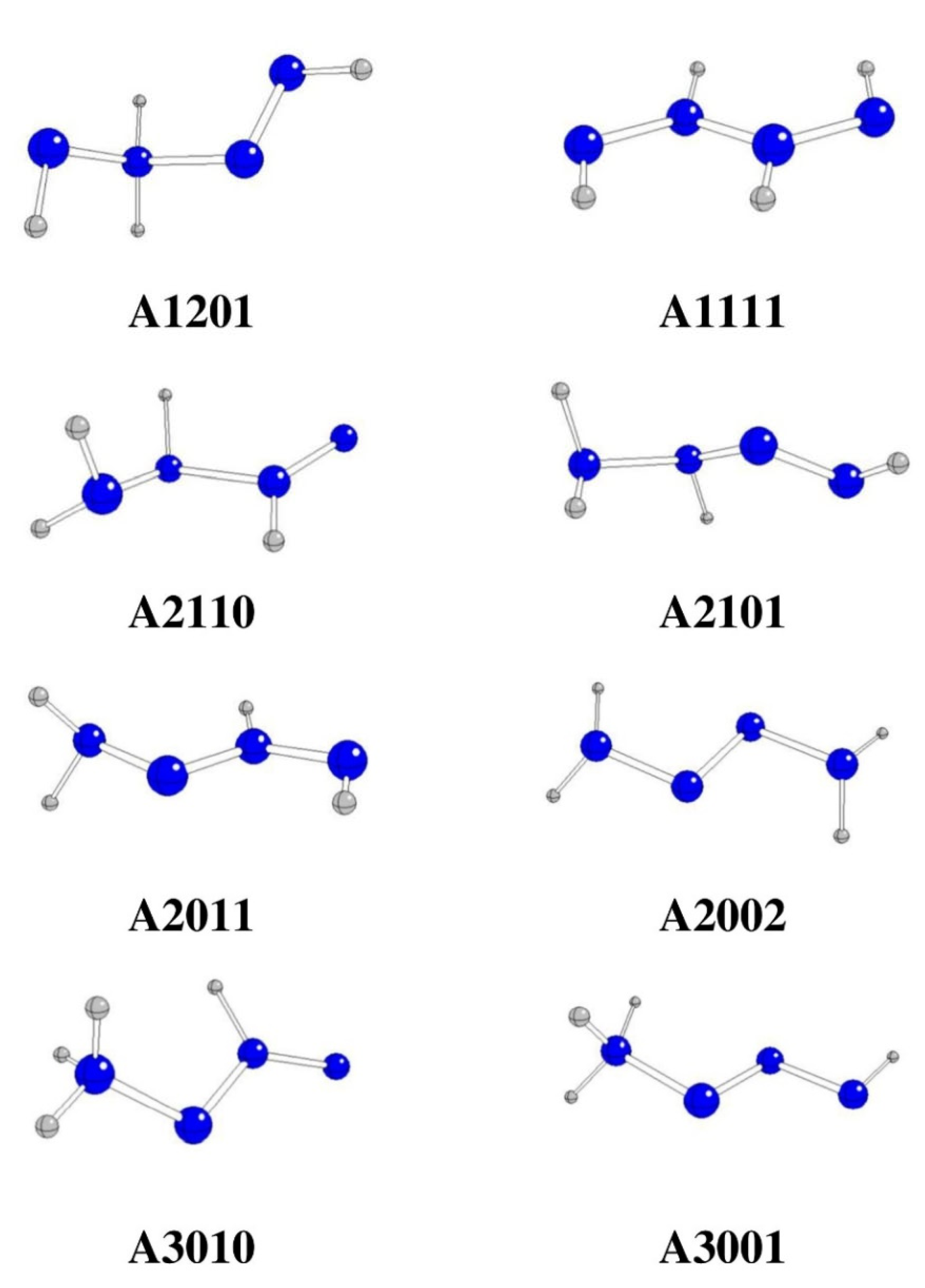
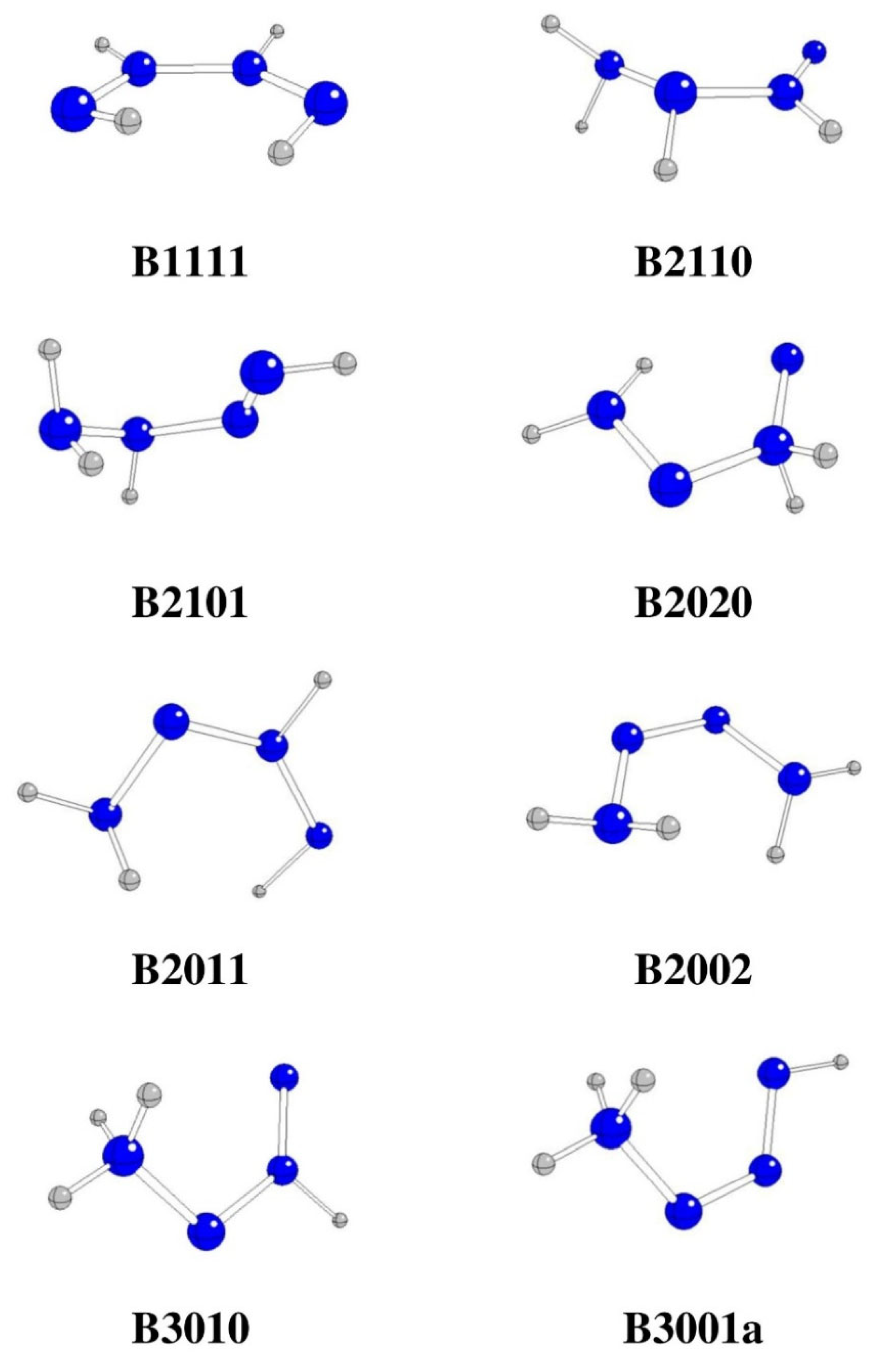
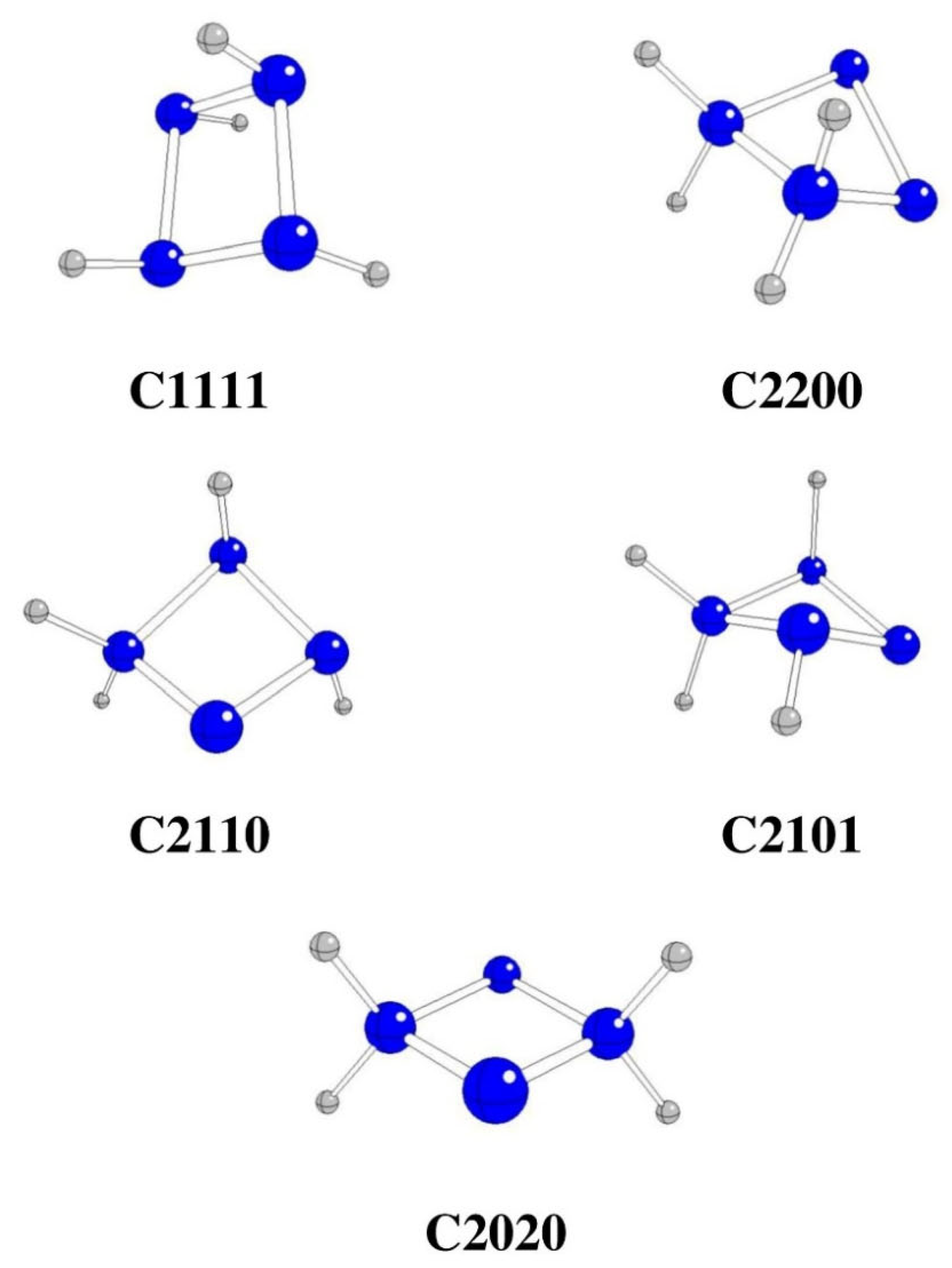
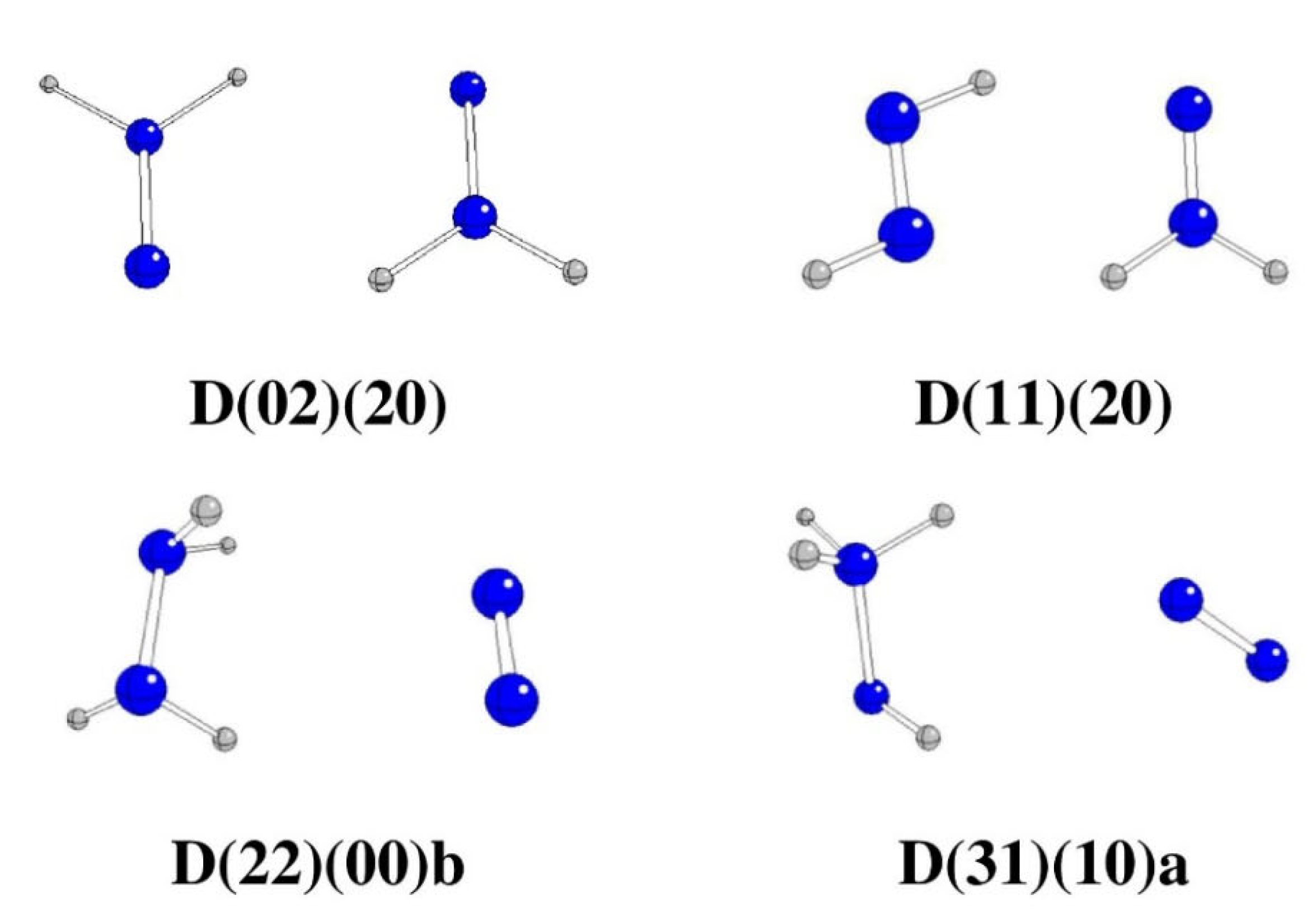
| Starting | Optimized | Θ1234 [o] | G298 [Hartree] | ΔG298 [kJ/mol] | Remarks |
|---|---|---|---|---|---|
| A0220 | D(02)(20) | -180.0 | -220.84657 | -251.26 | N=NH2 + H2N=N |
| A1210 | D(22)(00)a | 31.8 | -221.02068 | -4822.61 | H2N-NH2 + N2 |
| A1201 | A1201 | -106.4 | -220.84934 | -323.99 | |
| A1120 | D(11)(20)a | -179.1 | -220.87553 | -1011.62 | HN=NH + H2N=N |
| A1111 | A1111 | 180.0 | -220.83700 | 0.00 | |
| A2200 | D(22)(00)b | -176.2 | -221.02115 | -4834.95 | H2N-NH2 + N2 |
| A2110 | A2110 | -152.4 | -220.87955 | -1117.17 | |
| A2101 | A2101 | 153.9 | -220.91446 | -2033.75 | |
| A2020 | A2011 | 180.0 | -220.89237 | -1453.77 | 3→4 H rearrangement |
| A2011 | A2011 | -180.0 | -220.89237 | -1453.77 | |
| A2002 | A2002 | 173.0 | -220.93100 | -2468.02 | |
| A3100 | D(31)(00)a | 176.3 | -220.96823 | -3445.51 | H3N-NH + N2 |
| A3010 | A3010 | -180.0 | -220.84234 | -140.20 | |
| A3001 | A3001 | 180.0 | -220.89414 | -1500.24 | |
| A4000 | A3010 | 180.0 | -220.84232 | -139.87 | 1→3 H rearrangement |
| B0220 | D(02)(20) | 0.0 | -220.84657 | -251.26 | N=NH2 +H2N=N |
| B1210 | E12)(10 | -9.9 | -220.91726 | -2107.27 | N2-N3 fission, N1-N4 bonding, → B2101 |
| B1201 | D(22)(00)c | -2.3 | -221.02062 | -4821.03 | H2N-NH2 + N2 |
| B1120 | D(11)(20)b | -180.0 | -220.87565 | -1014.78 | HN=NH + H2N=N |
| B1111 | B1111 | -2.7 | -220.84994 | -339.75 | |
| B2200 | D(22)(00)d | 17.8 | -221.02057 | -4819.72 | H2N-NH2 + N2 |
| B2110 | B2110 | 17.9 | -220.87548 | -1010.31 | |
| B2101 | B2101 | -18.7 | -220.91727 | -2107.53 | |
| B2020 | B2020 | -0.2 | -220.74152 | 2506.88 | |
| B2011 | B2011 | -2.4 | -220.89749 | -1588.20 | |
| B2002 | B2002 | -4.2 | -220.93084 | -2463.82 | |
| B3100 | D(31)(00)b | 29.0 | -220.96692 | -3411.11 | H3N=NH + N2 |
| B3010 | B3010 | 0.0 | -220.85489 | -469.71 | |
| B3001 | B3001a | 0.0 | -220.90266 | -1723.94 | |
| B4000 | B3001b | 0.0 | -220.89253 | -1457.97 | 1→4 H rearrangement |
| C1111 | C1111 | 11.8 | -220.84534 | -218.97 | |
| C2200 | C2200 | -31.2 | -220.71366 | 3238.35 | |
| C2110 | C2110 | -18.7 | -220.79133 | 1199.09 | |
| C2101 | C2101 | 27.8 | -220.77308 | 1678.25 | |
| C2020 | C2020 | 0.0 | -220.77314 | 1676.68 |
| Structure | N1-N2 | N2-N3 | N3-N4 | N1-H | N2-H | N3-H | N4-H |
|---|---|---|---|---|---|---|---|
| A1201 | 1.409 | 1.501 | 1.225 | 1.017 | 1.021(2×) | - | 1.025 |
| A1111 | 1.324 | 1.288 | 1.324 | 1.019 | 1.015 | 1.019 | 1.015 |
| A2110 | 1.414 | 1.463 | 1.190 | 1.009 1.012 |
1.016 | 1.035 | - |
| A2101 | 1.410 | 1.370 | 1.243 | 1.011 1.014 |
1.013 | - | 1.020 |
| A2011 | 1.437 | 1.280 | 1.276 | 1.015(2×) | - | 1.023 | 1.022 |
| A2002 | 1.389 | 1.236 | 1.389 | 1.010 1.015 |
- | - | 1.010 1.015 |
| A3010 | 1.459 | 1.396 | 1.210 | 1.016 1.022(2×) |
- | 1.040 | - |
| A3001 | 1.458 | 1.306 | 1.278 | 1.018(2×) 1.022 |
- | - | 1.016 |
| B1111 | 1.311 | 1.294 | 1.311 | 1.018 | 1.012 | 1.012 | 1.018 |
| B2110 | 1.405 | 1.485 | 1.187 | 1.009 1.012 |
1.017 | 1.031 | - |
| B2101 | 1.408 | 1.359 | 1.246 | 1.013 1.018 |
1.007 | - | 1.019 |
| B2020 | 1.352 | 1.433 | 1.342 | 1.004 1.025 |
- | 1.026 1.038 |
- |
| B2011 | 1.400 | 1.276 | 1.294 | 1.010 1.021 |
- | 1.014 | 1.017 |
| B2002 | 1.388 | 1.243 | 1.388 | 1.009 1.016 |
- | - | 1.009 1.016 |
| B3010 | 1.437 | 1.382 | 1.218 | 1.015 1.023(2×) |
- | 1.030 | - |
| B3001a | 1.485 | 1.288 | 1.286 | 1.014 1.021(2×) |
- | - | 1.015 |
| B3001b | 1.507 | 1.289 | 1.277 | 1.015 1.021(2×) |
- | - | 1.025 |
| Structure | N1-N2 | N2-N3 | N3-N4 | N1-N4 | N1-H | N2-H | N3-H | N4-H |
|---|---|---|---|---|---|---|---|---|
| C1111 | 1.469 | 1.469 | 1.469 | 1.469 | 1.018 | 1.018 | 1.018 | 1.018 |
| C2200 | 1.434 | 1.491 | 1.511 | 1.495 | 1.032 1.023 |
1.032 1.023 |
- | - |
| C2110 | 1.459 | 1.467 | 1.488 | 1.484 | 1.019 1.024 |
1.020 | 1.023 | - |
| C2101 | 1.457 | 1.479 | 1.530 | 1.456 | 1.022 1.023 |
1.023 | - | 1.020 |
| C2020 | 1.468 | 1.468 | 1.468 | 1.468 | 1.019(2×) | - | 1.019(2×) | - |
| D(02)(20) | 1.226 | 3.133 | 1.226 | 3.375 | - | 1.028 1.032 |
1.028 1.032 |
- |
| D(11)(20)a | 1.245 | 3.386 | 1.224 | 3.314 | 1.027 | 1.029 | 1.028(2×) | - |
| D(11)(20)b | 1.245 | 3.064 | 1.223 | 3.168 | 1.029 | 1.027 | 1.028(2×) | - |
| D(22)(00)a | 1.445 | 3.709 | 1.096 | 3.492 | 1.011 1.014 |
1.011 1.014 |
- | - |
| D(22)(00)b | 1.445 | 3.756 | 1.096 | 3.893 | 1.011 1.014 |
1.011 1.014 |
- | - |
| D(22)(00)c | 1.445 | 3.758 | 1.096 | 3.461 | 1.011 1.014 |
1.011 1.014 |
- | - |
| D(22)(00)d | 1.445 | 3.705 | 1.096 | 3.706 | 1.011 1.014 |
1.011 1.014 |
- | - |
| D(31)(00)a | 1.466 | 3.418 | 1.096 | 4.353 | 1.014 1.020(2×) |
1.018 | - | - |
| D(31)(00)b | 1.466 | 3.545 | 1.096 | 3.742 | 1.014 1.020(2×) |
1.018 | - | - |
| Structure | N1-N2 | N2-N3 | N3-N4 | N1-H | N2-H | N3-H | N4-H |
|---|---|---|---|---|---|---|---|
| A1201 | 0.3088 | 0.5150 | 0.3426 | 0.3426 | 0.3505(2×) | - | 0.3441 |
| A1111 | 0.3805 | 0.4345 | 0.3805 | 0.3415 | 0.3491 | 0.3491 | 0.3415 |
| A2110 | 0.3203 | 0.2944 | 0.5366 | 0.3535 0.3503 |
0.3504 | 0.3350 | - |
| A2101 | 0.3233 | 0.3662 | 0.4891 | 0.3525 0.3495 |
0.3506 | - | 0.3494 |
| A2011 | 0.3054 | 0.4439 | 0.4393 | 0.3489(2×) | - | 0.3461 | 0.3428 |
| A2002 | 0.3442 | 0.5015 | 0.3442 | 0.3514 0.3462 |
- | - | 0.3514 0.3462 |
| A3010 | 0.2815 | 0.3389 | 0.5083 | 0.3478 0.3442(2×) |
- | 0.3325 | - |
| A3001 | 0.2866 | 0.4237 | 0.4459 | 0.3447(2×) 0.3421 |
- | - | 0.3504 |
| B1111 | 0.3959 | 0.4279 | 0.3959 | 0.3429 | 0.3506 | 0.3506 | 0.3429 |
| B2110 | 0.3267 | 0.2784 | 0.5386 | 0.3530 0.3505 |
0.3497 | 0.3388 | - |
| B2101 | 0.3236 | 0.3726 | 0.4848 | 0.3511 0.3456 |
0.3564 | - | 0.3498 |
| B2020 | 0.3717 | 0.3104 | 0.3743 | 0.3536 0.3307 |
- | 0.3359 0.3168 |
(0.0466) |
| B2011 | 0.3338 | 0.4460 | 0.4172 | 0.3524 0.3401 |
- | 0.3548 | 0.3469 |
| B2002 | 0.3435 | 0.4883 | 0.3435 | 0.3531 0.3435 |
- | - | 0.3531 0.3435 |
| B3010 | 0.2990 | 0.3503 | 0.4972 | 0.3496 0.3430(2×) |
- | 0.3424 | - |
| B3001a | 0.2670 | 0.4397 | 0.4356 | 0.3504 0.3432(2×) |
- | - | 0.3506 |
| B3001b | 0.2517 | 0.4336 | 0.4395 | 0.3484 0.3422(2×) |
- | - | 0.3395 |
| Structure | N1-N2 | N2-N3 | N3-N4 | N1-N4 | N1-H | N2-H | N3-H | N4-H |
|---|---|---|---|---|---|---|---|---|
| C1111 | 0.2916 | 0.2916 | 0.2916 | 0.2916 | 1.018 | 1.018 | 1.018 | 1.018 |
| C2200 | 0.3175 | 0.2566 | 0.2620 | 0.2542 | 0.3384 0.3450 |
0.3383 0.3451 |
- | - |
| C2110 | 0.2964 | 0.2917 | 0.2759 | 0.2665 | 0.3498 0.3467 |
0.3522 | 0.3496 | - |
| C2101 | 0.2940 | 0.2946 | 0.2470 | 0.2785 | 0.3465 0.3464 |
0.3513 | - | 0.3522 |
| C2020 | 0.2815 | 0.2808 | 0.2819 | 0.2817 | 0.3530 0.3532 |
- | 0.3536(2×) | - |
| D(02)(20)a | 0.4964 | - | 04964 | - | (0.0233) | 0.3422 0.3383 |
0.3422 0.3383 |
(0.0233) |
| D(11)(20)a | 0.4863 | - | 0.4986 | - | 0.3484 (0.0158) |
0.3471 | 0.3426(2×) | (0.0194) |
| D(11)(20)b | 0.4864 | - | 0.4986 | - | 0.3471 | 0.3484 (0.0157) |
0.3426(2×) | (0.0193) |
| D(22)(00)a | 0.2955 | - | 0.7139 | 0.0036 | 0.3530 0.3500 |
0.3530 0.3503 |
(0.0027) | - |
| D(22)(00)b | 0.2957 | -(a) | 0.7138 | - | 0.3530 0.3502 |
0.3530 0.3500 |
(0.0038) | (0.0027) |
| D(22)(00)c | 0.2957 | - | 0.7139 | 0.0039 | 0.3530 0.3503 |
0.3530 0.3500 |
(0.0026) | - |
| D(22)(00)d | 0.2958 | 0.0023 | 0.7140 | - | 0.3530 0.3503 |
0.3531 0.3503 |
- | (0.0027) |
| D(31)(00)a | 0.2685 | 0.0048 | 0.7139 | - | 0.3525 0.3491 0.3486 |
0.3408 | (0.0057) | - |
| D(31)(00)b | 0.2681 | - | 0.7139 | - | 0.3525 0.3492 0.3483 |
0.3412 | (0.0043) | (0.0024) |
| Structure | N1-N2 | N2-N3 | N3-N4 | N1-H | N2-H | N3-H | N4-H |
|---|---|---|---|---|---|---|---|
| A1201 | 0.184 | 0.023 | 0.176 | 0.063 | 0.006(2×) | - | 0.009 |
| A1111 | 0.216 | 0.354 | 0.216 | 0.030 | 0.032 | 0.032 | 0.030 |
| A2101 | 0.011 | 0.134 | 0.187 | 0.045 0.049 |
0.052 | - | 0.002 |
| A2110 | 0.034 | 0.172 | 0.067 | 0.046 0.050 |
0.041 | 0.032 | - |
| A2011 | 0.022 | 0.300 | 0.223 | 0.041(2×) | - | 0.026 | 0.013 |
| A2002 | 0.101 | 0.238 | 0.105 | 0.041 0.045 |
- | - | 0.041 0.045 |
| A3010 | 0.146 | 0.277 | 0.036 | 0.007 0.012(2×) |
- | 0.030 | - |
| A3001 | 0.094 | 0.214 | 0.189 | 0.006 0.010(2×) |
- | - | 0.021 |
| B1111 | 0.200 | 0.343 | 0.200 | 0.028 | 0.036 | 0.036 | 0.028 |
| B2110 | 0.038 | 0.177 | 0.079 | 0.047 0.051 |
0.041 | 0.031 | - |
| B2101 | 0.016 | 0.149 | 0.182 | 0.039 0.043 |
0.057 | - | 0.003 |
| B2020 | 0.173 | 0.129 | 0.181 | 0.041(2×) | - | 0.007 0.022 |
(0.209) |
| B2011 | 0.094 | 0.292 | 0.211 | 0.039(2×) | - | 0.031 | 0.019 |
| B2002 | 0.095 | 0.241 | 0.102 | 0.040(2×) | - | - | 0.040(2×) |
| B3010 | 0.130 | 0.272 | 0.028 | 0.005(3×) | - | 0.033 | - |
| B3001a | 0.073 | 0.211 | 0.174 | 0.002(2×) 0.004 |
- | - | 0.021 |
| B3001b | 0.069 | 0.205 | 0.185 | 0.005(3×) | - | - | 0.012 |
| Structure | N1-N2 | N2-N3 | N3-N4 | N1-N4 | N1-H | N2-H | N3-H | N4-H |
|---|---|---|---|---|---|---|---|---|
| C1111 | 0.059 | 0.059 | 0.059 | 0.059 | 0.039 | 0.039 | 0.039 | 0.039 |
| C2200 | 0.022 | 0.209 | 0.270 | 0.209 | 0.005 0.004 |
0.005 0.004 |
- | - |
| C2110 | 0.052 | 0.052 | 0.163 | 0.232 | 0.002 0.005 |
0.029 | 0.038 | - |
| C2101 | 0.072 | 0.181 | 0.186 | 0.067 | 0.001 0.005 |
0.031 | - | 0.026 |
| C2020 | 0.222 | 0.222 | 0.222 | 0.222 | 0.003(2×) | - | 0.003(2×) | - |
| D(02)(20)a | 0.008 | - | 0.008 | - | (0.006) | 0.030(2×) | 0.030(2×) | (0.006) |
| D(11)(20)a | 0.161 | - | 0.018 | - | 0.049 (0.022) |
0.005 | 0.032(2×) | (0.005) |
| D(11)(20)b | 0.161 | - | 0.018 | - | 0.005 | 0.004 (0.020) |
0.032(2×) | (0.005) |
| D(22)(00)a | - | 0.005 | 0.832 | - | 0.049 0.045 |
0.049 0.045 |
(0.676) | - |
| D(22)(00)b | 0.011 | -(a) | 0.003 | - | 0.045 0.049 |
0.044 0.049 |
(0.637) | (0.621) |
| D(22)(00)c | 0.010 | - | 0.003 | 0.653 | 0.049 0.045 |
0.049 0.045 |
(0.629) | - |
| D(22)(00)d | 0.010 | 0.330 | 0.005 | - | 0.049 0.045 |
0.049 0.045 |
- | (0.354) |
| D(31)(00)a | 0.151 | 0.316 | 0.003 | - | 0.011(2×) 0.009 |
0.076 | (0.220) | - |
| D(31)(00)b | 0.145 | - | 0.004 | - | 0.009 0.011(2×) |
0.078 | (0.266) | (1.271) |
| Structure | N1 | N2 | N3 | N4 | H(N1) | H(N2) | H(N3) | H(N4) |
|---|---|---|---|---|---|---|---|---|
| A1201 | -0.77 | -0.42 | -0.03 | -0.35 | 0.32 | 0.47(2×) | - | 0.44 |
| A1111 | -0.55 | -0.21 | -0.21 | -0.55 | 0.37 | 0.51 | 0.51 | 0.37 |
| A2110 | -0.72 | -0.37 | -0.21 | -0.19 | 0.42(2×) | 0.42 | 0.44 | - |
| A2101 | -0.68 | -0.35 | -0.02 | -0.39 | 0.41(2×) | 0.44 | - | 0.41 |
| A2011 | -0.71 | -0.14 | -0.19 | -0.45 | 0.41(2×) | - | 0.50 | 0.39 |
| A2002 | -0.68 | -0.07 | -0.07 | -0.68 | 0.42(2×) | - | - | 0.42(2×) |
| A3010 | -0.74 | -0.33 | -0.21 | -0.35 | 0.48(2×) 0.50 |
- | 0.38 | - |
| A3001 | -0.73 | -0.24 | -0.11 | -0.54 | 0.49(3×) | - | - | 0.45 |
| B1111 | -0.59 | -0.28 | -0.28 | -0.59 | 0.36 | 0.53 | 0.53 | 0.36 |
| B2110 | -0.70 | -0.37 | -0.23 | -0.19 | 0.41(2×) | 0.42 | 0.44 | - |
| B2101 | -0.68 | -0.34 | -0.04 | -0.42 | 0.40(2×) | 0.46 | - | 0.40 |
| B2020 | -0.74 | -0.19 | -0.48 | -0.39 | 0.46(2×) | - | 0.53(2×) | - |
| B2011 | -0.69 | -0.09 | -0.21 | -0.53 | 0.42(2×) | - | 0.51 | 0.38 |
| B2002 | -0.71 | -0.05 | -0.05 | -0.071 | 0.43(2×) | - | - | 0.43(2×) |
| B3010 | -0.72 | -0.31 | -0.22 | -0.41 | 0.47(2×) 0.500 |
- | 0.41 | - |
| B3001a | -0.76 | -0.20 | -0.09 | -0.59 | 0.49(3×) | - | - | 0.36 |
| B3001b | -0.79 | -0.19 | -0.08 | -0.53 | 0.50(3×) | - | - | 0.31 |
| Structure | N1 | N2 | N3 | N4 | H(N1) | H(N2) | H(N3) | H(N4) |
|---|---|---|---|---|---|---|---|---|
| C1111 | -0.36 | -0.36 | -0.36 | -0.36 | 0.40 | 0.40 | 0.40 | 0.40 |
| C2200 | -0.43 | -0.43 | -0.44 | -0.44 | 0.47 0.49 |
0.47 0.49 |
- | - |
| C2110 | -0.45 | -0.33 | -0.38 | -0.39 | 0.46 0.49 |
0.42 | 0.37 | - |
| C2101 | -0.44 | -0.33 | -0.44 | -0.37 | 0.50(2×) | 0.37 | - | 0.40 |
| C2020 | -0.43 | -0.35 | -0.43 | -0.35 | 0.46 0.49 |
- | 0.46 0.49 |
- |
| D(02)(20) | -0.28 | -0.52 | -0.52 | -0.27 | - | 0.46 0.42 |
0.46 0.42 |
- |
| D(11)(20)a | -0.36 | -0.36 | -0.51 | -0.26 | 0.48 | 0.40 | 0.43(2×) | - |
| D(11)(20)b | -0.36 | -0.36 | -0.51 | -0.26 | 0.40 | 0.38 | 0.43(2×) | - |
| D(22)(00)a | -0.70 | -0.69 | 0.01 | -0.02 | 0.38(2×) | 0.38(2×) | - | - |
| D(22)(00)b | -0.71 | -0.70 | 0.06 | 0.05 | 0.38(2×) | 0.37 0.39 |
- | - |
| D(22)(00)c | -0.71 | -0.70 | 0.06 | 0.07 | 0.38(2×) | 0.38(2×) | - | - |
| D(22)(00)d | -0.70 | -0.71 | 0.01 | 0.03 | 0.38(2×) | 0.38(2×) | - | - |
| D(31)(00)a | -0.71 | -0.84 | 0.05 | 0.10 | 0.44(2×) 0.47 |
0.289 | - | - |
| D(31)(00)b | -0.72 | -0.84 | -0.01 | 0.02 | 0.45(2×) 0.47 |
0.28 | - | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




