Submitted:
24 June 2024
Posted:
26 June 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Functional Organization of Proximal Straight Tubules
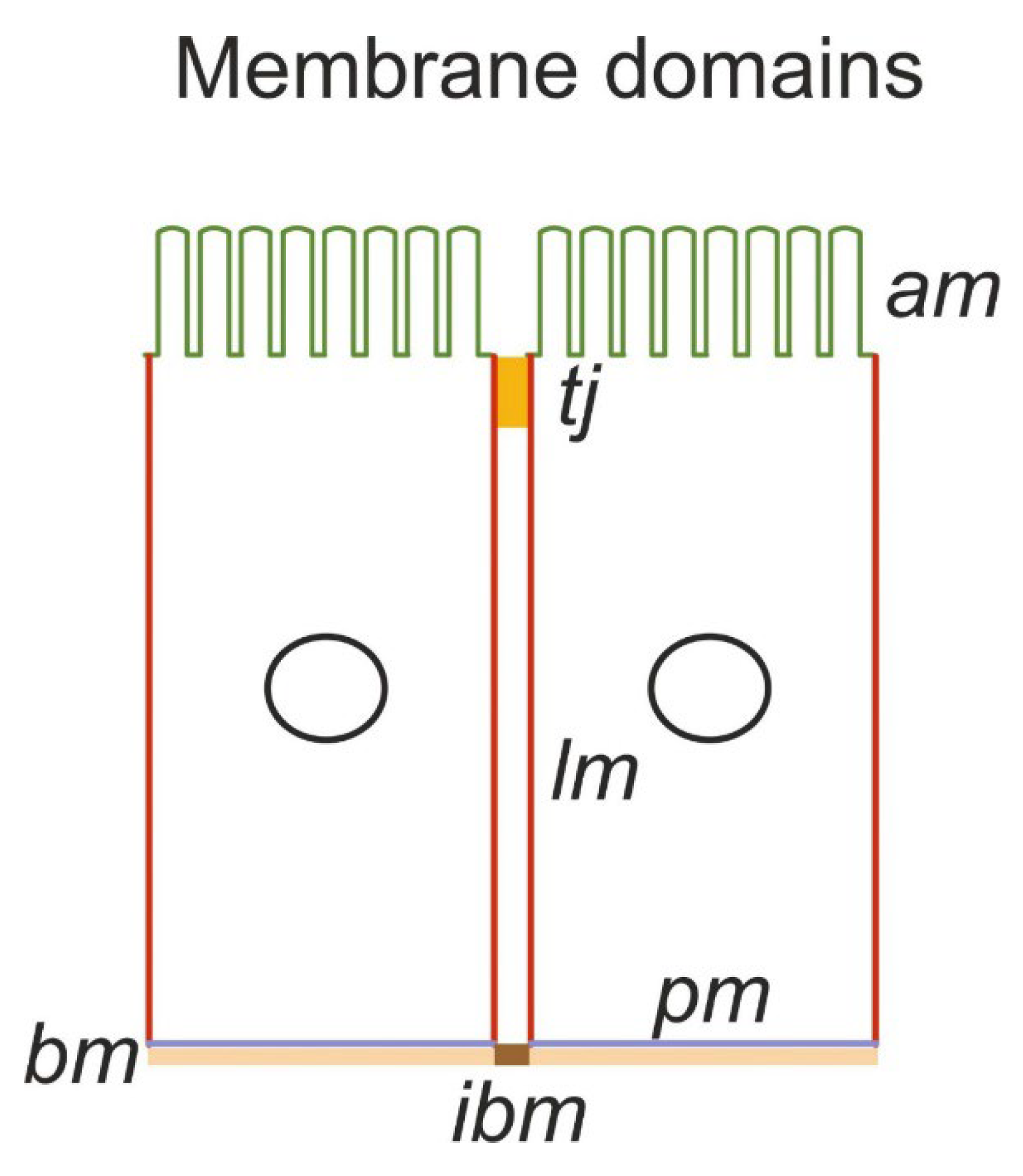
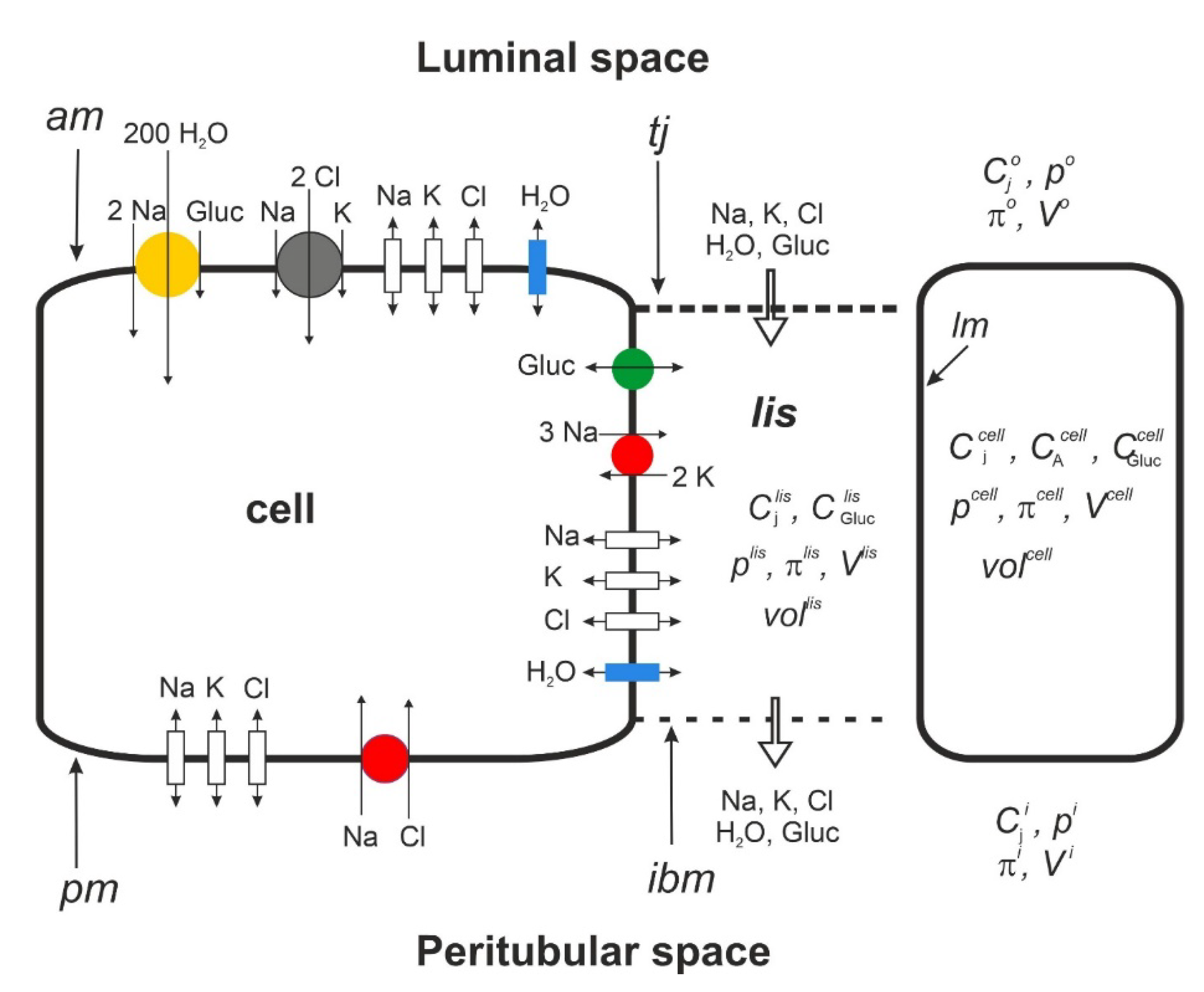
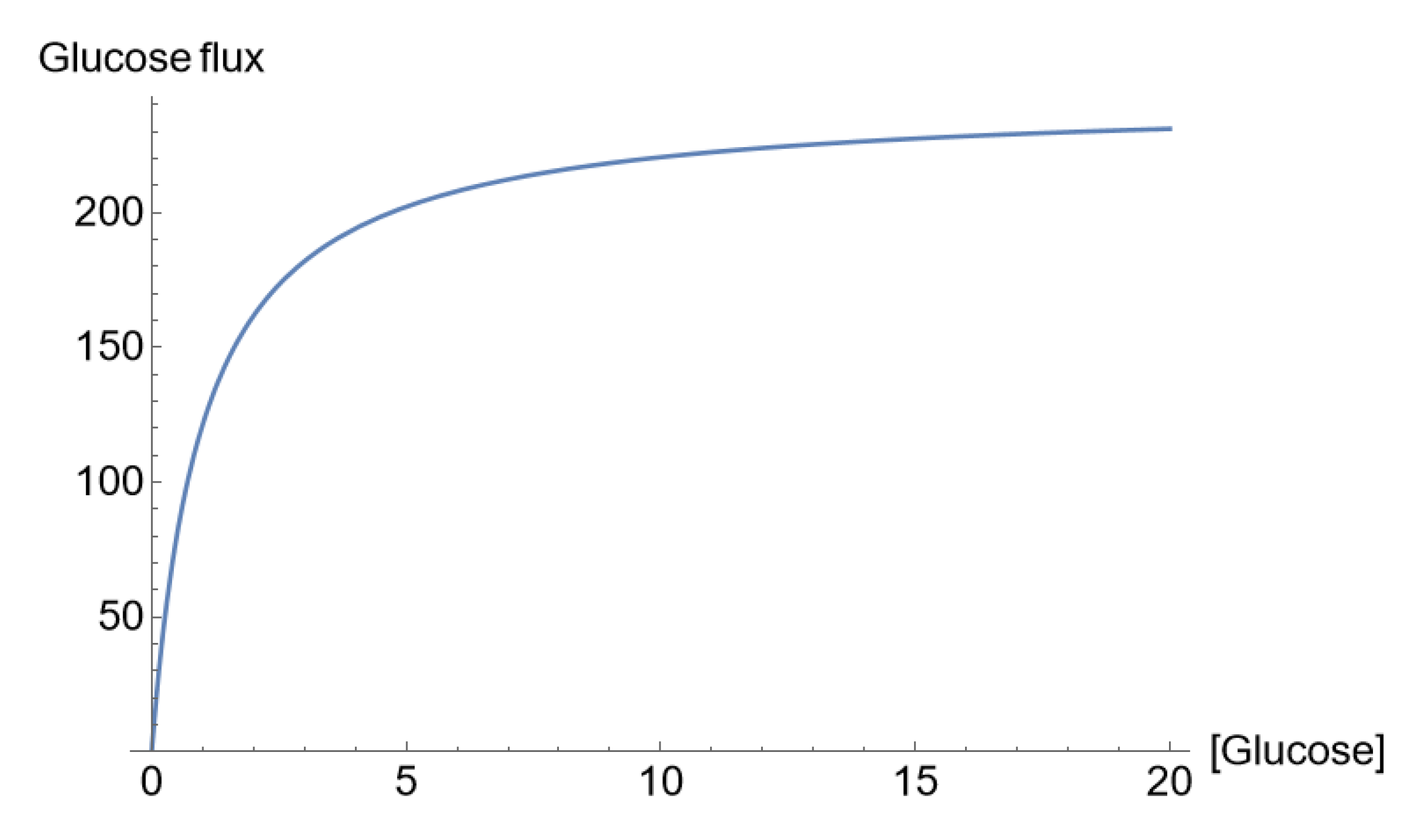
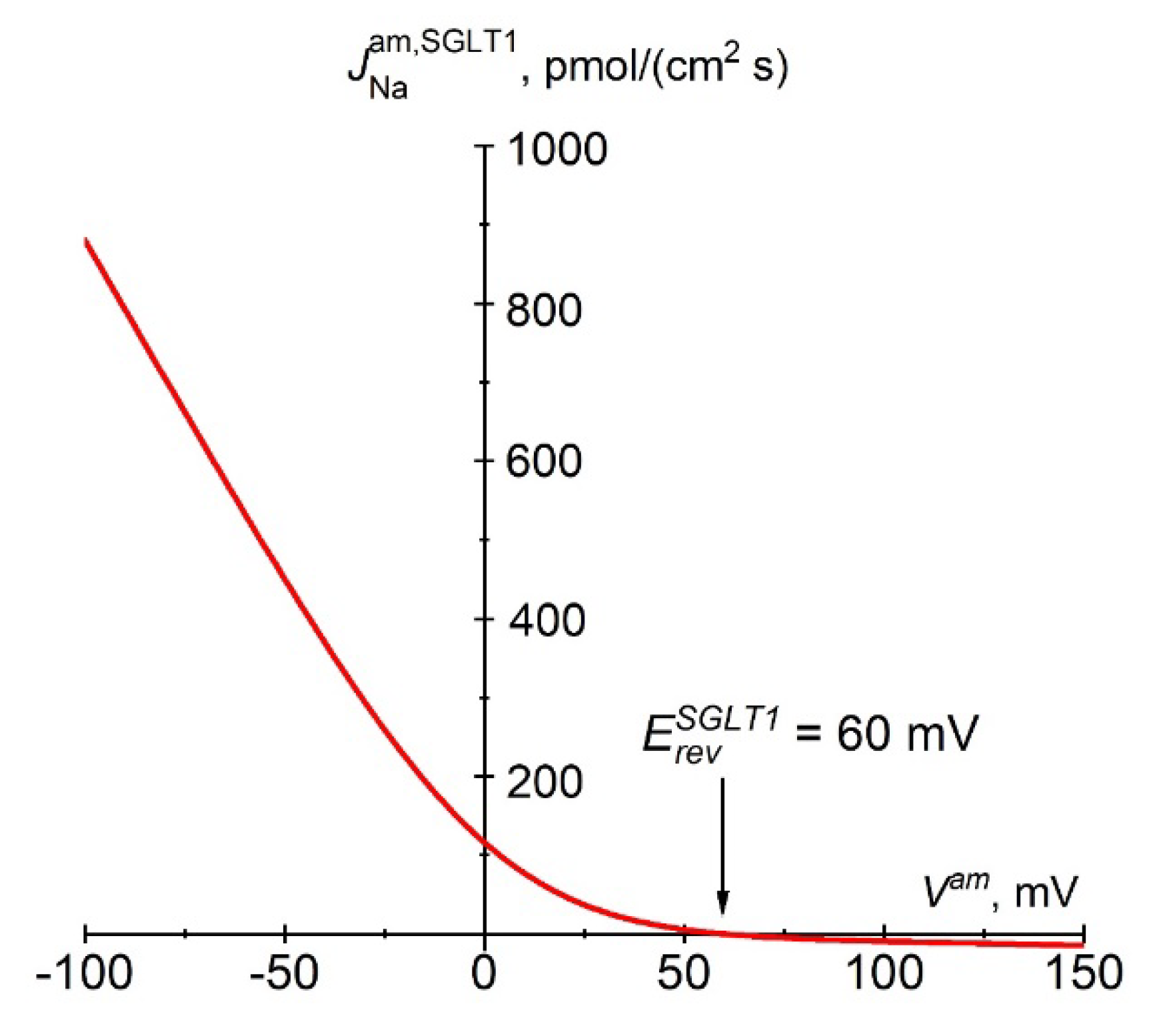
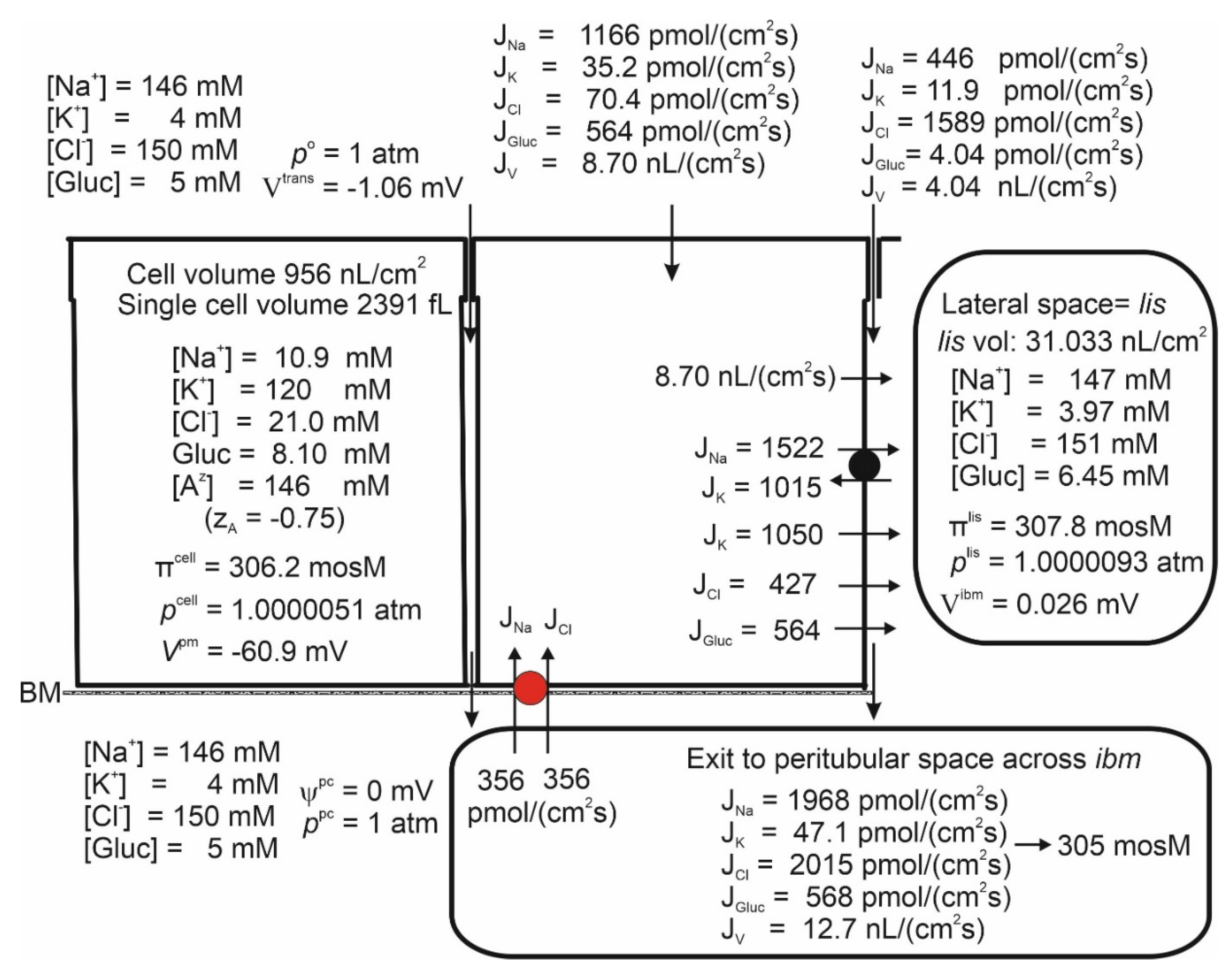
Mathematical Description
Prelude
Results
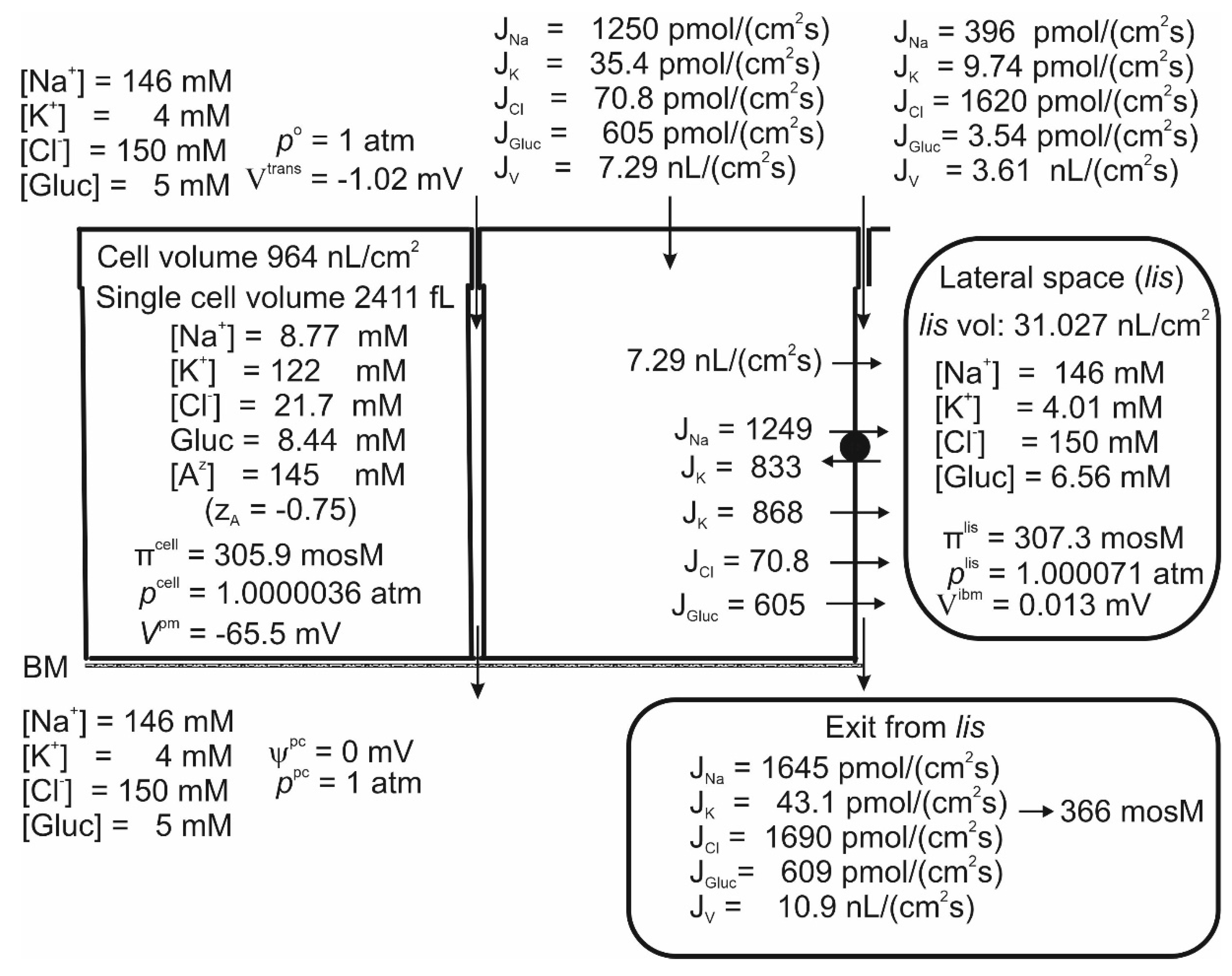
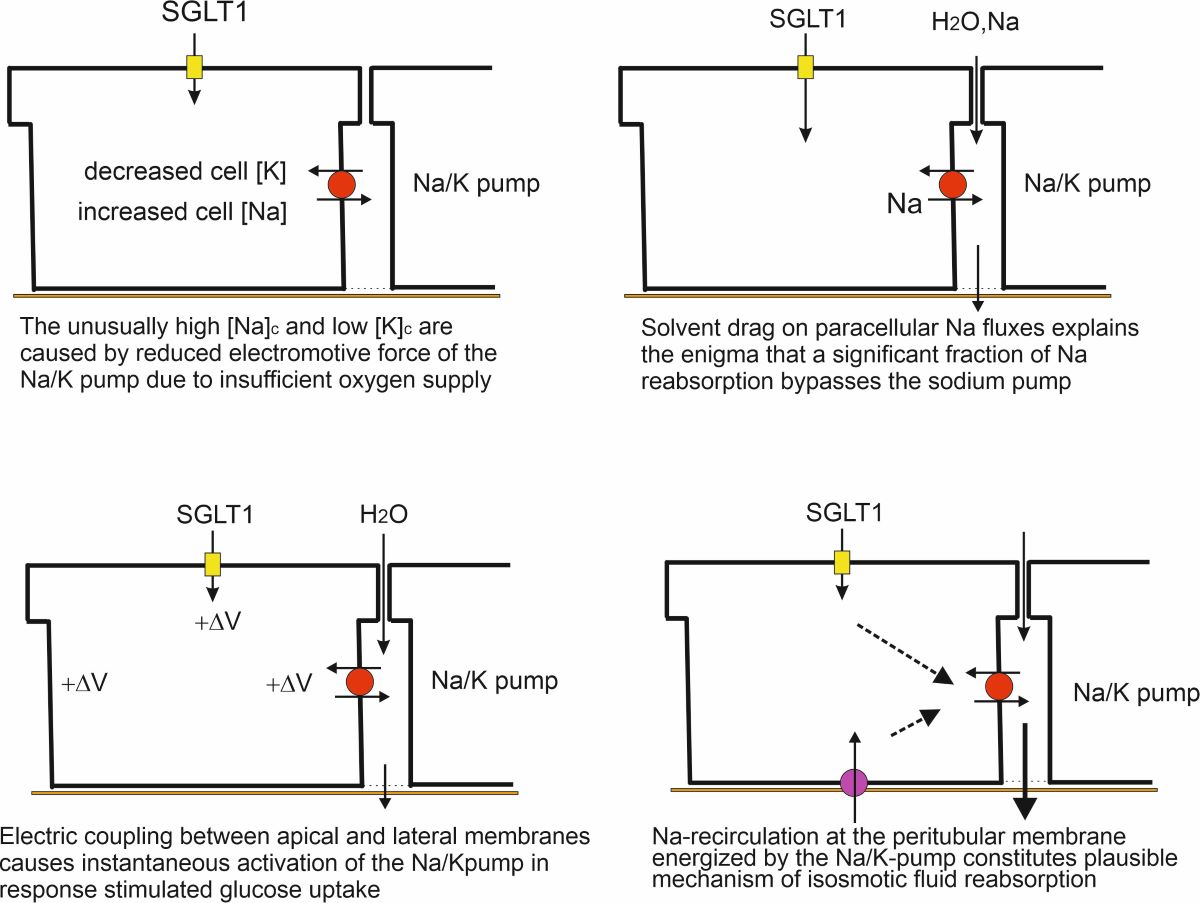
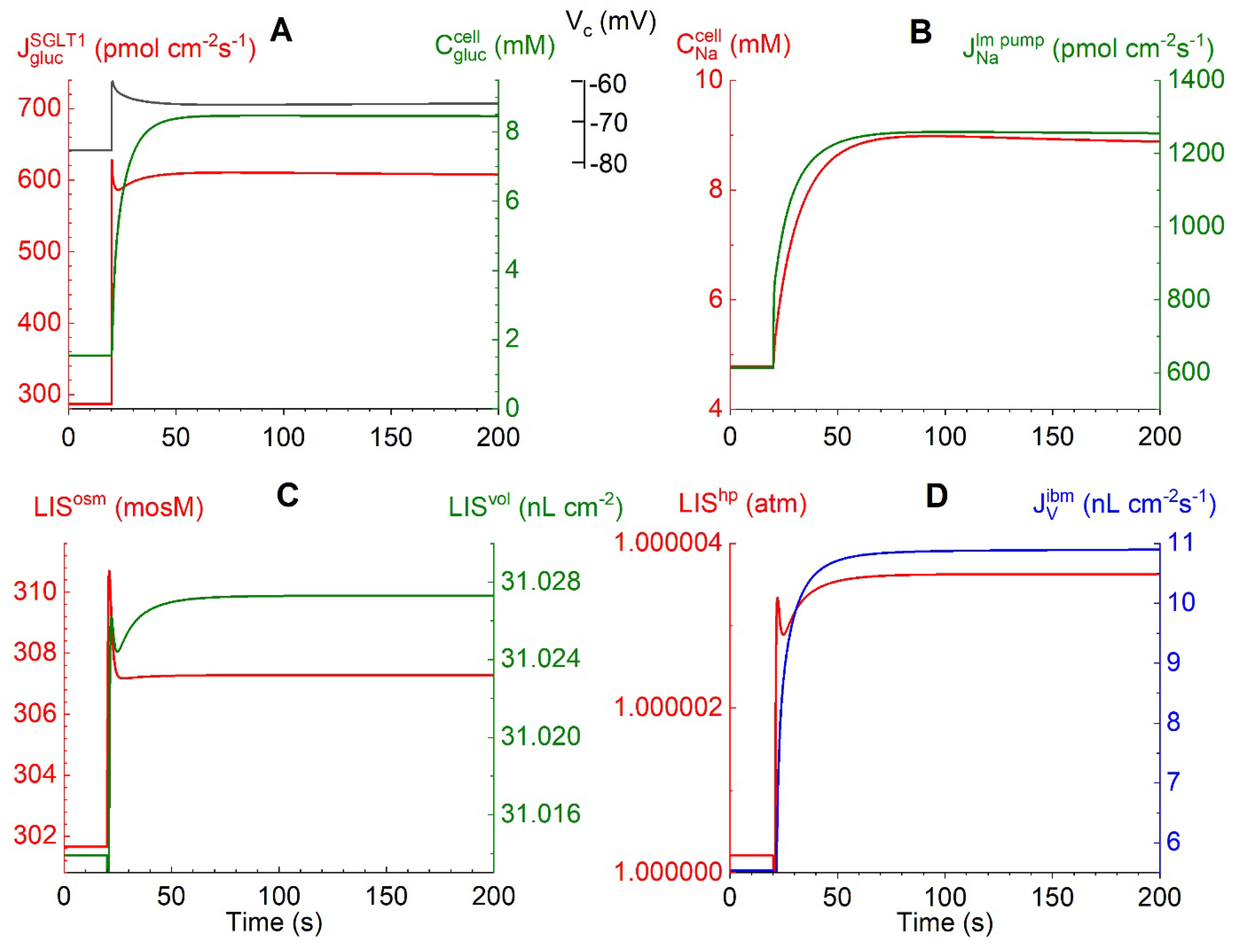
Crosstalk between Electrogenic Apical SGLT1 and Electrogenic Lateral Na/K Pumps
Contributions of Epithelial Water Pathways to Transepithelial Water Absorption
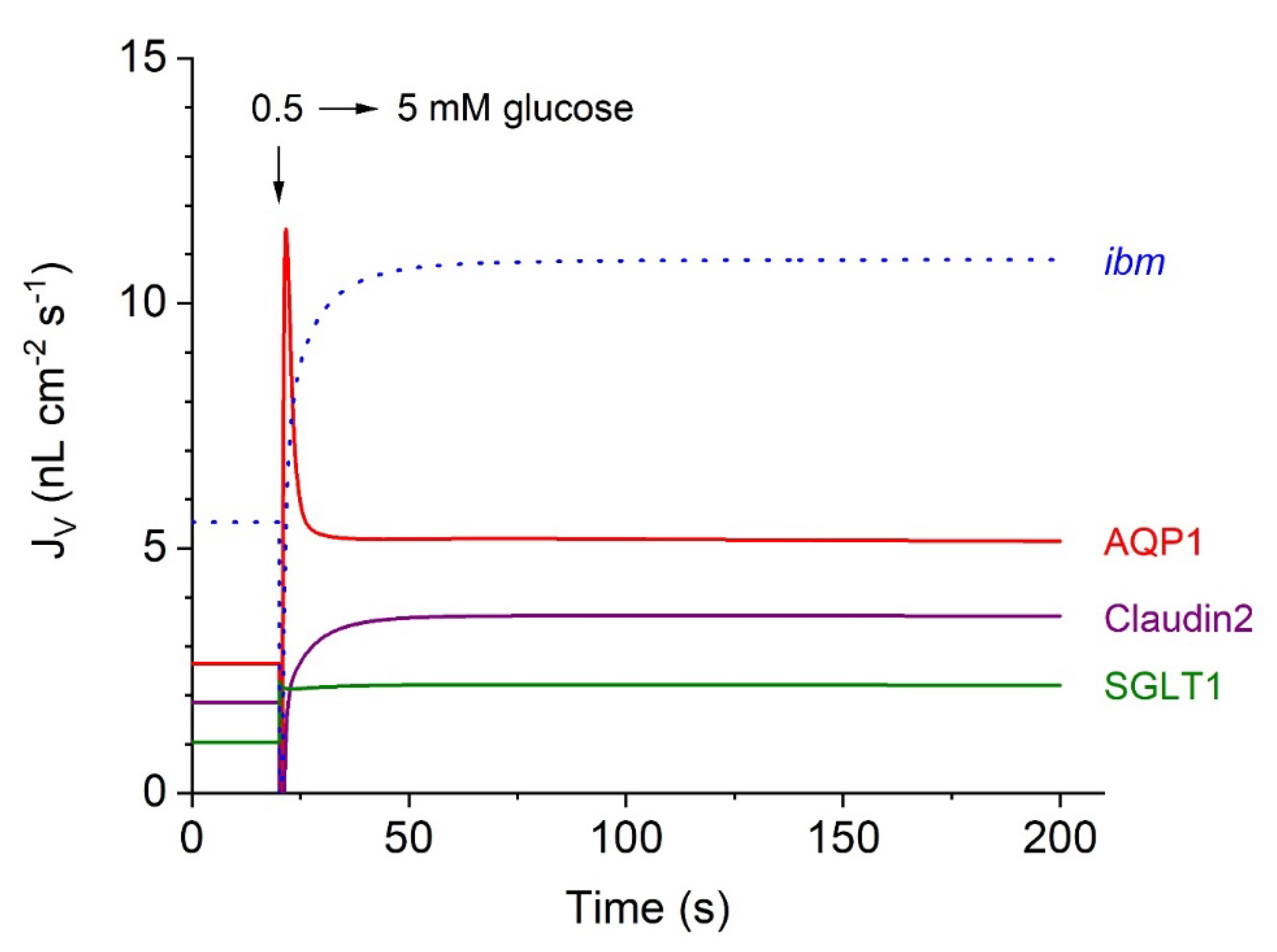
Isosmotic Transport
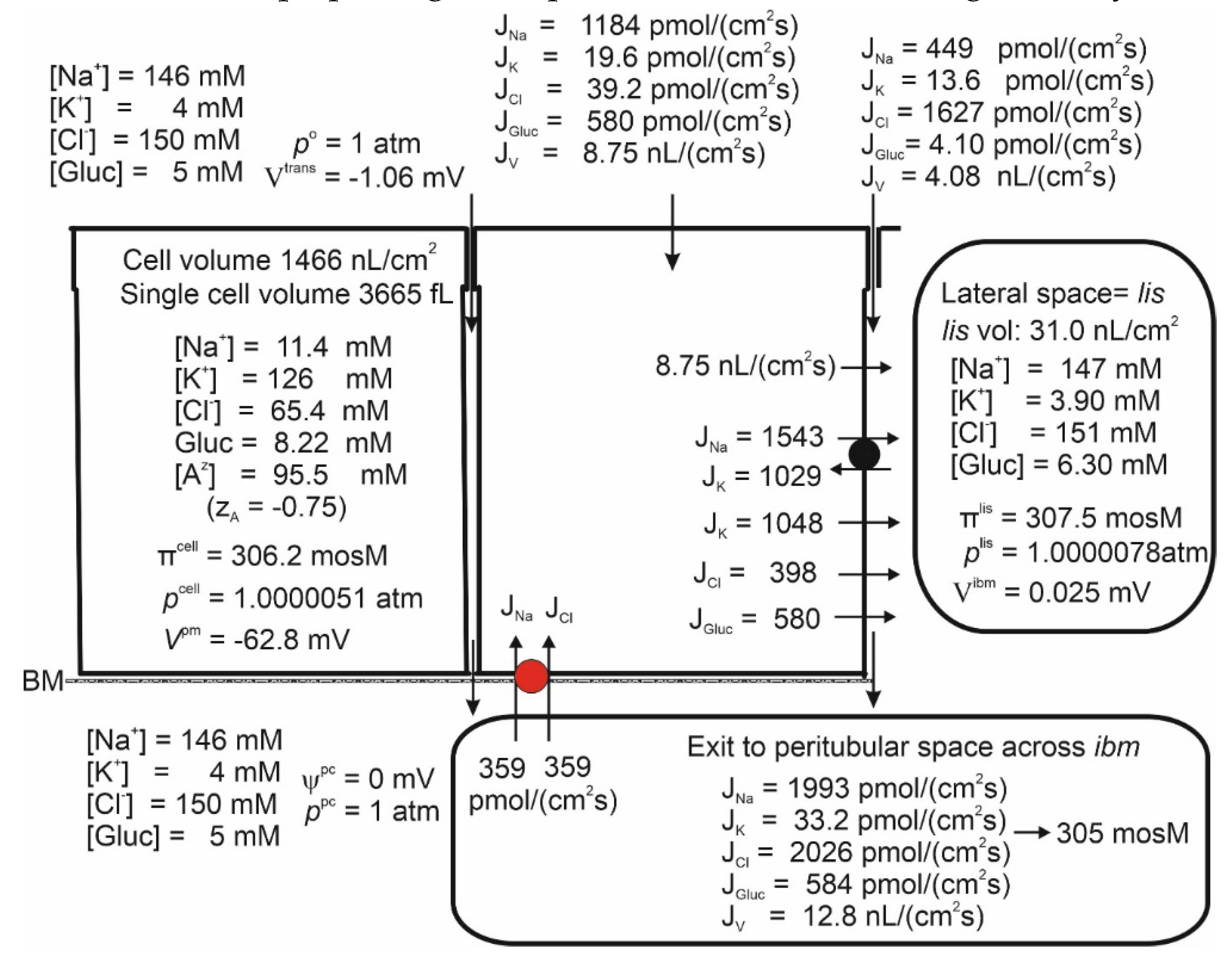
Isosmotic Transport Requires Cell Volume Regulation to Maintain Cellular Homeostasis
Discussion
Appendix 1: Definitions and Symbols of Variables and Constants
| Concentration of j (Na+, K+, Cl−, A-, or glucose) in compartment comp (o, cell, lis or ps) | |
| Osmolarity of compartment indicated by superscript (o, cell, lis or ps) | |
| Hydrostatic pressure of compartment indicated (o, cell, lis or ps) | pcomp |
| Electrical potential of compartment indicated (o, cell, lis or ps) with | |
| Transepithelial potential difference, | Vtrans |
| Electrical potential difference between lumen and cell, | Vam |
| Electrical potential difference between cell and lis, | Vlm |
| Electrical potential difference between cell and peritubular space, | Vps |
| Passive permeability of j in membrane m (am, lm, pm, tj, ibm) | |
| Reflection coefficient of j (Na+, K+, Cl−, glucose) in m (tj, ibm) | |
| Flux of j (= Na+, K+, Cl−, glucose) across m (am, lm, pm, tj, ibm) | |
| Electrical current carried by j (Na+, K+, Cl−) across m (am, lm, pm, tj, ibm) | |
| Integral ion (j) conductance of membrane m (am, lm, pm, tj, ibm) | |
| Water volume flux across m (am, lm, pm, tj, ibm) | |
| Hydraulic conductance of membrane m (am, lm, pm, tj, ibm) | |
| Osmotic permeability of m (am, lm, pm, tj, ibm) | |
| Relative compliance constant of membrane m (am, lm, pm) | |
| Absolute compliance constant of lm | |
| Empirical constant of 1Na:1K:2Cl cotransporter of membrane m | |
| Apparent dissociation constants of K+ binding of Na/K pump at lm | |
| Empirical apparent dissociation constant of Na+:glucose transporter at am | |
| Empirical apparent dissociation constant for glucose of Na+:glucose transporter at am | |
| Maximum turnover of glucose exchanger at lm | |
| App dissociation const. of symmetrical carrier at lm | |
| Turnover constant of 2 Na+:1 glucose transporter at am | |
| Temperature in K | T |
| Faraday | F |
| Universal gas constant | R |
Appendix 2: Independent Variables
| Name | Symbol | Value | MKSA-unit |
| Hydraulic conductance of am | Lam | 0.2125d-10 | m3∙s-1∙N-1 |
| Hydraulic conductance of pm | Lpm | 0.350d-15 | m3∙s-1∙N-1 |
| Hydraulic conductance of lm | Llm | 0.2125d-10 | m3∙s-1∙N-1 |
| Hydraulic conductance of tj | Ltj | 0.900d-11 | m3∙s-1∙N-1 |
| Hydraulic conductance of ibm | Libm | 0.231d-06 | m3∙s-1∙N-1 |
| Na+ concentration of outside (luminal) compartment | 146 | mol∙m-3 | |
| K+ concentration of outside (luminal) compartment | 4 | mol∙m-3 | |
| Cl− concentration of outside (luminal) compartment | 150 | mol∙m-3 | |
| Glucose concentration of luminal compartment Vary in Table |
5 | mol∙m-3 | |
| Na+ concentration of peritubular (inside) compartment | 146 | mol∙m-3 | |
| K+ concentration of peritubular (inside) compartment | 4 | mol∙m-3 | |
| Cl− concentration of peritubular (inside) compartment | 150 | mol∙m-3 | |
| Na+ GHK permeability of am | 0.100d-9 | m∙s-1 | |
| K+ GHK permeability of am | 0.100d-13 | m∙s-1 | |
| Cl− GHK permeability of am | 0.100d-13 | m∙s-1 | |
| Na+ GHK permeability of pm | 0.100d-12 | m∙s-1 | |
| K+ GHK permeability of pm | 0.100d-12 | m∙s-1 | |
| Cl− GHK permeability of pm | 0.100d-12 | m∙s-1 | |
| Na+ GHK permeability of lm | 0.300d-14 | m∙s-1 | |
| K+ GHK permeability of lm | 0.500d-6 | m∙s-1 | |
| Cl− GHK permeability of lm |
Figure 6 |
0.300d-7 0.600d-6 |
m∙s-1 m∙s-1 |
| Na+ GHK permeability of tj (Eqn. 6) | 0.150d-6 | m∙s-1 | |
| K+ GHK permeability of tj (Eqn. 6) | 0.2205d-6 | m∙s-1 | |
| Cl− GHK permeability of tj (Eqn. 1) | 0.270d-5 | m∙s-1 | |
| Glucose permeability of tj (Eqn. 7a) | 0.500d-9 | m∙s-1 | |
| Na+ GHK permeability of ibm (Eqn. 6) | 0.120d-5 | m∙s-1 | |
| K+ GHK permeability of ibm (Eqn. 6) | 0.176d-5 | m∙s-1 | |
| Cl− GHK permeability of ibm (Eqn. 6) | 0.350d-4 | m∙s-1 | |
| Glucose permeability if ibm (Eqn. 7b) | 0.350d-5 | m∙s-1 | |
| 1Na:1K:2Cl cotransporter constant of am | 0.28d-13 | m10∙s-1∙mol-3 | |
| 1Na:1Cl cotransporter constant of pm | ~0, dependent | m10∙s-1∙mol-3 | |
| Turnover constant of lateral 3Na+/2K+ pump | 0.350d-3 | mol∙m-2∙s-1 | |
| Reversal potential of lateral 3Na+/2K+ pump | Epump | -0.200 | volt |
| Apparent dissociation constants of Na+ binding | 3.40d0 | mol∙m-3 | |
| Apparent dissociation constants of K+ binding | 0.75d0 | mol∙m-3 | |
| Turnover constant of SGLT1 at am | 0.70d-12 | mol∙m-2∙s-1 | |
| Apparent dissociation constant for glucose at SGLT1 | 1.0 | mol∙m-3 | |
| Maximum turnover of glucose exchanger at lm | 0.30d-9 | mol∙m-2∙s-1 | |
| App dissociation const. of symmetry carrier at lm | 5.0d1 | mol∙m-3 | |
| Hydrostatic pressure of outer (luminal) compartment | po | 1.01325e5 | Pa |
| Hydrostatic pressure of peritubular compartment | pps | 1.01325e5 | Pa |
| Mean valence of nondiffusible intracellular anions | zA | -0.75 | |
| Na+ reflection coefficient of tj | 0.70 | ||
| K+ reflection coefficient of tj | 0.70 | ||
| Cl− reflection coefficient of tj | 0.45 | ||
| Glucose reflection coefficient of tj | 0.80 | ||
| Na+ reflection coefficient of ibm | 0.0001 | ||
| K+ reflection coefficient of ibm | 0.0001 | ||
| Cl− reflection coefficient of ibm | 0.0001 | ||
| Glucose reflection coefficient of ibm | 0.0001 | ||
| Temperature | T | 310 | K |
| Faraday | F | 96485 | C∙mol-1 |
| Absolute compliance constant of lm | 0.250d-2 | Pa-1 | |
| Reference volume of lis | Vollis,ref | 3.10d-7 | m3∙m-2 |
| Cell density | Dc | 4.00d+9 | # cells∙m-2 |
| Nondiffusible anions in cell | MA | 3.50d-13 | mol∙cell-1 |
References
- Hummel, C. S. et al. Glucose transport by human renal Na+/D-glucose cotransporters SGLT1 and SGLT2. Am J Physiol Cell Physiol 300, C14-21. (2011). [CrossRef]
- Loo, D. D., Zeuthen, T., Chandy, G. & Wright, E. M. Cotransport of water by the Na+/glucose cotransporter. Proc Natl Acad Sci U S A 93, 13367-13370. (1996). [CrossRef]
- Zeuthen, T. General models for water transport across leaky epithelia. Int Rev Cytol 215, 285-317. (2002). [CrossRef]
- Han, L. et al. Structure and mechanism of the SGLT family of glucose transporters. Nature 601, 274-279. (2022). [CrossRef]
- Larsen, E. H. & Sørensen, J. N. Stationary and nonstationary ion and water flux interactions in kidney proximal tubule: Mathematical analysis of isosmotic transport by a minimalistic model. Rev Physiol Biochem Pharmacol 177, 101-147, (2020). [CrossRef]
- Larsen, E. H. & Sørensen, J. N. Ion and water absorption by the kidney proximal tubule: Computational analysis of isosmotic transport. Function (Oxf) 1, zqaa014. (2020). [CrossRef]
- Burg, M., Patlak, C., Green, N. & Villey, D. Organic solutes in fluid absorption by renal proximal convoluted tubules. Am J Physiol 231, 627-637. (1976). [CrossRef]
- Windhager, E. E., Whittembury, G., Oken, D. E., Schatzmann, H. J. & Solomon, A. K. Single proximal tubules of the Necturus kidney. III. Dependence of H2O movement on NaCl concentration. Am J Physiol 197, 313-318. (1959). [CrossRef]
- Welling, L. W. & Welling, D. J. Relationship between structure and function in renal proximal tubule. J Electron Microsc Tech 9, 171-185. (1988). [CrossRef]
- Maunsbach, A. B. Observations on the segmentation of the proximal tubule in the rat kidney. Comparison of results from phase contrast, fluorescence and electron microscopy. J Ultrastruct Res 16, 239-258. (1966). [CrossRef]
- Kashgarian, M., Biemesderfer, D., Caplan, M. & Forbush, B., 3rd. Monoclonal antibody to Na,K-ATPase: immunocytochemical localization along nephron segments. Kidney Int 28, 899-913. (1985). [CrossRef]
- Borgnia, M., Nielsen, S., Engel, A. & Agre, P. Cellular and molecular biology of the aquaporin water channels. Annu Rev Biochem 68, 425-458. (1999). [CrossRef]
- Nielsen, S., Smith, B. L., Christensen, E. I., Knepper, M. A. & Agre, P. CHIP28 water channels are localized in constitutively water-permeable segments of the nephron. J Cell Biol 120, 371-383. (1993). [CrossRef]
- Murer, H., Hopfer, U. & Kinne, R. Sodium/proton antiport in brush-border-membrane vesicles isolated from rat small intestine and kidney. Biochem J 154, 597-604. (1976).
- Lee, S. K. et al. Distinguishing among HCO3- , CO32- , and H+ as substrates of proteins that appear to be "bicarbonate" transporters. J Am Soc Nephrol 34, 40-54. (2023). [CrossRef]
- Gögelein, H. & Greger, R. Na+ selective channels in the apical membrane of rabbit late proximal tubules (pars recta). Pflugers Arch 406, 198-203. (1986). [CrossRef]
- Willmann, J. K. et al. Amiloride-inhibitable Na+ conductance in rat proximal tubule. Pflugers Arch 434, 173-178. (1997). [CrossRef]
- Fromter, E. Electrophysiological analysis of rat renal sugar and amino acid transport. I. Basic phenomena. Pflugers Arch 393, 179-189. (1982). [CrossRef]
- Sten-Knudsen, O. Biological Membranes. Theory of transport, potentials and electric impulses. pp 1-671 (Cambridge University Press, 2002).
- Hodgkin, A. L. & Katz, B. The effect of sodium ions on the electrical activity of giant axon of the squid. J Physiol 108, 37-77. (4). [CrossRef]
- Wilson, R. W., Wareing, M. & Green, R. The role of active transport in potassium reabsorption in the proximal convoluted tubule of the anaesthetized rat. J Physiol 500 ( Pt 1), 155-164. (1997). [CrossRef]
- Spring, K. R. & Kimura, G. Chloride reabsorption by renal proximal tubules of Necturus. J Membr Biol 38, 233-254, doi:10.1007/BF01871924 (1978). (1978). [CrossRef]
- Spring, K. R. & Kimura, G. Intracellular ion activities in Necturus proximal tubule. Fed Proc 38, 2729-2732 (1979).
- Kimura, G. & Spring, K. R. Luminal Na+ entry into Necturus proximal tubule cells. Am J Physiol 236, F295-301. (1979). [CrossRef]
- Lucci, M. S. & Warnock, D. G. Effects of anion-transport inhibitors on NaCl reabsorption in the rat superficial proximal convoluted tubule. J Clin Invest 64, 570-579. (1979). [CrossRef]
- Tune, B. M. & Burg, M. B. Glucose transport by proximal renal tubules. Am J Physiol 221, 580-585. (1971). [CrossRef]
- Loo, D. D., Wright, E. M. & Zeuthen, T. Water pumps. J Physiol 542, 53-60. (2002). [CrossRef]
- Zeuthen, T., Gorraitz, E., Her, K., Wright, E. M. & Loo, D. D. Structural and functional significance of water permeation through cotransporters. Proc Natl Acad Sci U S A 113, E6887-E6894. (2016). [CrossRef]
- Goldman, D. E. Potential, Impedance, and Rectification in Membranes. J Gen Physiol 27, 37-60. (1943). [CrossRef]
- Bazzone, A., Zerlotti, R., Barthmes, M. & Fertig, N. Functional characterization of SGLT1 using SSM-based electrophysiology: Kinetics of sugar binding and translocation. Front Physiol 14, 1058583. (2023). [CrossRef]
- Fuchs, W., Larsen, E. H. & Lindemann, B. Current-voltage curve of sodium channels and concentration dependence of sodium permeability in frog skin. Journal of Physiology 267, 137-166. (1977). [CrossRef]
- Birnir, B., Loo, D. D. & Wright, E. M. Voltage-clamp studies of the Na+/glucose cotransporter cloned from rabbit small intestine. Pflugers Arch 418, 79-85. (1991). [CrossRef]
- Dominguez, J. H., Camp, K., Maianu, L. & Garvey, W. T. Glucose transporters of rat proximal tubule: differential expression and subcellular distribution. Am J Physiol 262, F807-812. (1992). [CrossRef]
- Stein, W. D. The movement of molecules acoss cell membranes. (Academic Press, 1967).
- Skou, J. C. Enzymatic Basis for Active Transport of Na+ and K+ across Cell Membrane. Physiol Rev 45, 596-617. (1965). [CrossRef]
- Thomas, R. C. Electrogenic sodium pump in nerve and muscle cells. Physiol Rev 52, 563-594. (1972). [CrossRef]
- Gadsby, D. C. & Nakao, M. Steady-state current-voltage relationship of the Na/K pump in guinea pig ventricular myocytes. J Gen Physiol 94, 511-537, (1989). [CrossRef]
- Jørgensen, P. L. Sodium and potassium ion pump in kidney tubules. Physiol Rev 60, 864-917. (1980). [CrossRef]
- Goldin, S. M. Active transport of sodium and potassium ions by the sodium and potassium ion-activated adenosine triphosphatase from renal medulla. Reconstitution of the purified enzyme into a well defined in vitro transport system. J Biol Chem 252, 5630-5642. (1977).
- Eskesen, K. & Ussing, H. H. Determination of the electromotive force of active sodium transport in frog skin epithelium (Rana temporaria) from presteady-state flux ratio experiments. J Membr Biol 86, 105-111. (1985). [CrossRef]
- Freeman, D., Bartlett, S., Radda, G. & Ross, B. Energetics of sodium transport in the kidney. Saturation transfer 31P-NMR. Biochim Biophys Acta 762, 325-336, (1983). [CrossRef]
- Slater, E. C., Rosing, J. & Mol, A. The phosphorylation potential generated by respiring mitochondria. Biochim Biophys Acta 292, 534-553. (1973). [CrossRef]
- Wackerhage, H.; et al. Recovery of free ADP, Pi, and free energy of ATP hydrolysis in human skeletal muscle. J Appl Physiol ( 1985) 85, 2140–2145. [CrossRef]
- Veech, R. L., Lawson, J. W., Cornell, N. W. & Krebs, H. A. Cytosolic phosphorylation potential. J Biol Chem 254, 6538-6547 (1979).
- Dørup, J. & Maunsbach, A. B. Three-dimensional organization and segmental ultrastructure of rat proximal tubules. Exp Nephrol 5, 305-317 (1997).
- Hertz, G. Ein neues verfahren zur trennung von gasgemischen durch diffusion. Physikalische Zeitschrift 23, 433-434 (1922).
- Larsen, E. H., Sorensen, J. B. & Sorensen, J. N. A mathematical model of solute coupled water transport in toad intestine incorporating recirculation of the actively transported solute. Journal of General Physiology 116, 101-124. (2000). [CrossRef]
- Larsen, E. H., Sørensen, J. B. & Sørensen, J. N. Analysis of the sodium recirculation theory of solute-coupled water transport in small intestine. J Physiol 542, 33-50. (2002). [CrossRef]
- Finkelstein, A. Water movement through lipid bilayers, pores, and plasma membranes. Theory and reality., pp. 1-228 (Wiley-Interscience, 1987).
- Fromm, M., Piontek, J., Rosenthal, R., Gunzel, D. & Krug, S. M. Tight junctions of the proximal tubule and their channel proteins. Pflugers Arch 469, 877-887. (2017). [CrossRef]
- Rosenthal, R. et al. Claudin-2-mediated cation and water transport share a common pore. Acta Physiol (Oxf) 219, 521-536, (2017). [CrossRef]
- Krug, S. M. et al. Claudin-17 forms tight junction channels with distinct anion selectivity. Cell Mol Life Sci 69, 2765-2778, (2012). [CrossRef]
- Whittembury, G., de Martinez, C. V., Linares, H. & Paz-Aliaga, A. Solvent drag of large solutes indicates paracellular water flow in leaky epithelia. Proc R Soc Lond B Biol Sci 211, 63-81. (1980). [CrossRef]
- Whittembury, G., Malnic, G., Mello-Aires, M. & Amorena, C. Solvent drag of sucrose during absorption indicates paracellular water flow in the rat kidney proximal tubule. Pflugers Arch 412, 541-547. (1988). [CrossRef]
- Barfuss, D. W. & Schafer, J. A. Differences in active and passive glucose transport along the proximal nephron. Am J Physiol 241, F322-332, (1981). [CrossRef]
- Ullrich, K. J. E. Permebility characteristics of the mammalian nephron American Physiological Society, Bethesda (1973).
- Knepper, M. & Burg, M. Organization of nephron function. Am J Physiol 244, F579-589. (1983). [CrossRef]
- Schafer, J. A., Patlak, C. S. & Andreoli, T. E. Fluid absorption and active and passive ion flows in the rabbit superficial pars recta. Am J Physiol 233, F154-167. (1977). [CrossRef]
- Welling, L. W., Welling, D. J. & Ochs, T. J. Video measurement of basolateral membrane hydraulic conductivity in the proximal tubule. Am J Physiol 245, F123-129. (1983). [CrossRef]
- Carpi-Medina, P., Lindemann, B., Gonzalez, E. & Whittembury, G. The continuous measurement of tubular volume changes in response to step changes in contraluminal osmolality. Pflugers Arch 400, 343-348. (1984). [CrossRef]
- Carpi-Medina, P. & Whittembury, G. Comparison of transcellular and transepithelial water osmotic permeabilities (Pos) in the isolated proximal straight tubule (PST) of the rabbit kidney. Pflugers Arch 412, 66-74. (1988). [CrossRef]
- Kubota, T., Biagi, B. A. & Giebisch, G. Intracellular potassium activity measurements in single proximal tubules of Necturus kidney. J Membr Biol 73, 51-60. (1983). [CrossRef]
- Biagi, B., Sohtell, M. & Giebisch, G. Intracellular potassium activity in the rabbit proximal straight tubule. Am J Physiol 241, F677-686. (1981). [CrossRef]
- Edelman, A., Curci, S., Samarzija, I. & Fromter, E. Determination of intracellular K+ activity in rat kidney proximal tubular cells. Pflugers Arch 378, 37-45. (1978). [CrossRef]
- Gullans, S. R., Avison, M. J., Ogino, T., Giebisch, G. & Shulman, R. G. NMR measurements of intracellular sodium in the rabbit proximal tubule. Am J Physiol 249, F160-168. (1985). [CrossRef]
- Yoshitomi, K. & Fromter, E. How big is the electrochemical potential difference of Na+ across rat renal proximal tubular cell membranes in vivo? Pflugers Arch 405 Suppl 1, S121-126. (1985). [CrossRef]
- Wang, Z. et al. Specific metabolic rates of major organs and tissues across adulthood: evaluation by mechanistic model of resting energy expenditure. Am J Clin Nutr 92, 1369-1377. (2010). [CrossRef]
- Singh, H. Mitochondrial ion channels in cardiac function. Am J Physiol Cell Physiol 321, C812-C825. (2021). [CrossRef]
- Nelson, D. L. & Cox, M. Lehninger Principles of Biochemistry. 8th edn, (Macmillan Higher Education).
- Boron, W. F. & Boulpaep, E. L. Medical Physiology. (Elsevier, 2017).
- Schafer, J. A., Troutman, S. L., Watkins, M. L. & Andreoli, T. E. Volume absorption in the pars recta. I. "Simple" active Na+ transport. Am J Physiol 234, F332-339. (1978). [CrossRef]
- Andreoli, T. E., Schafer, J. A., Troutman, S. L. & Watkins, M. L. Solvent drag component of Cl- flux in superficial proximal straight tubules: evidence for a paracellular component of isotonic fluid absorption. Am J Physiol 237, F455-462. (1979). [CrossRef]
- Thaysen, J. H., Lassen, N. A. & Munck, O. Sodium transport and oxygen consumption in the mammalian kidney. Nature 190, 919-921. (1961). [CrossRef]
- Lassen, N. A., Munck, O. & Thaysen, J. H. Oxygen consumption and sodium reabsorption in the kidney. Acta Physiol Scand 51, 371-384. (1961). [CrossRef]
- Hall, J. E. Guyton and Hall Textbook of Medical Physiology. 13th edn, (Elsevier, 2016).
- Bennett, C. M., Clapp, J. R. & Berliner, R. W. Micropuncture study of the proximal and distal tubule in the dog. Am J Physiol 213, 1254-1262. (1967). [CrossRef]
- Kokko, J. P., Burg, M. B. & Orloff, J. Characteristics of NaCl and water transport in the renal proximal tubule. J Clin Invest 50, 69-76, (1971). [CrossRef]
- Schafer, J. A., Troutman, S. L. & Andreoli, T. E. Volume reabsorption, transepithelial potential differences, and ionic permeability properties in mammalian superficial proximal straight tubules. J Gen Physiol 64, 582-607. (1974). [CrossRef]
- Boron, W. F. & Boulpaep, E. L. Medical Physiology, Chapter 36 Giebisch, G., Windhager, E. E,, and Aronson P. S. Transport of Urea Glucose, Phophate, Calcium, Magnesium, and Organis Solutes. Third edn, (Elsevier, 2017).
- Diamond, J. M. & Bossert, W. H. Standing-gradient osmotic flow. A mechanism for coupling of water and solute transport in epithelia. Journal of General Physiology 50, 2061-2083. (1967). [CrossRef]
- Nedergaard, S., Larsen, E. H. & Ussing, H. H. Sodium recirculation and isotonic transport in toad small intestine. J Membr Biol 168, 241-251. (1999). [CrossRef]
- Sten-Knudsen, O. & Ussing, H. H. The flux ratio equation under nonstationary conditions. J Membr Biol 63, 233-242, doi:10.1007/BF01870984 (1981). (1981). [CrossRef]
- Ussing, H. H., Lind, F. & Larsen, E. H. Ion secretion and isotonic transport in frog skin glands. J Membr Biol 152, 101-110, (1996). [CrossRef]
- Sørensen, J. B., Nielsen, M. S., Nielsen, R. & Larsen, E. H. Luminal ion channels involved in isotonic secretion by Na+-recirculation in exocrine gland-acini. Biol. Skr. Dan. Vid. Selsk. 49, 179-191. (1998).
- Parsons, D. S. & Wingate, D. L. The effect of osmotic gradients on fluid transfer across rat intestine in vitro. Biochim Biophys Acta 46, 170-183. (1961). [CrossRef]
- Kirk, K. L., Schafer, J. A. & DiBona, D. R. Cell volume regulation in rabbit proximal straight tubule perfused in vitro. Am J Physiol 252, F922-932. (1987). [CrossRef]
- Larsen, E. H. & Hoffmann, E. K. in Ion channels and transporters of epithelia in health and disease (eds K. L. Hamilton & Devor, D. C.) Ch. 4, (American Physiological Society, 2016).
- Kirk, K. L., DiBona, D. R. & Schafer, J. A. Regulatory volume decrease in perfused proximal nephron: evidence for a dumping of cell K+. Am J Physiol 252, F933-942. (1987). [CrossRef]
- Morel, F. & Murayama, Y. Simultaneous measurement of undirectional and net sodium fluxes in microperfused rat proximal tubules. Pflugers Arch 320, 1-23. (1970). [CrossRef]
| Vc | Vtrans | ||||||||
| mV | kJ/mol | mM | mM | mV | mV | mV | mV | pmol∙cm-2∙s-1 | nL∙cm-2∙s-1 |
| -200 | -58.0 | 8.77 | 122 | -65.5 | 141 | -25.7 | -1.02 | 1249 | 10.9 |
| -180 | -52.2 | 10.7 | 120 | -65.1 | 135 | -25.7 | -1.02 | 1243 | 10.9 |
| -160 | -46.4 | 13.9 | 117 | -64.4 | 127 | -25.7 | -1.01 | 1230 | 10.7 |
| -140 -120 -100 |
-40.6 -35.2 -29.0 |
20.4 35.2 43.9 |
110 95.9 71.6 |
-63.0 -59.5 -52.3 |
116 97.5 75.9 |
-25.6 25.3 -24.7 |
-0.99 -0.93 -0.82 |
1204 1138 1004 |
10.5 9.94 8.77 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).