Submitted:
07 June 2024
Posted:
11 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Molecular mechanisms underlying SARS-CoV-2 induced autoimmunity
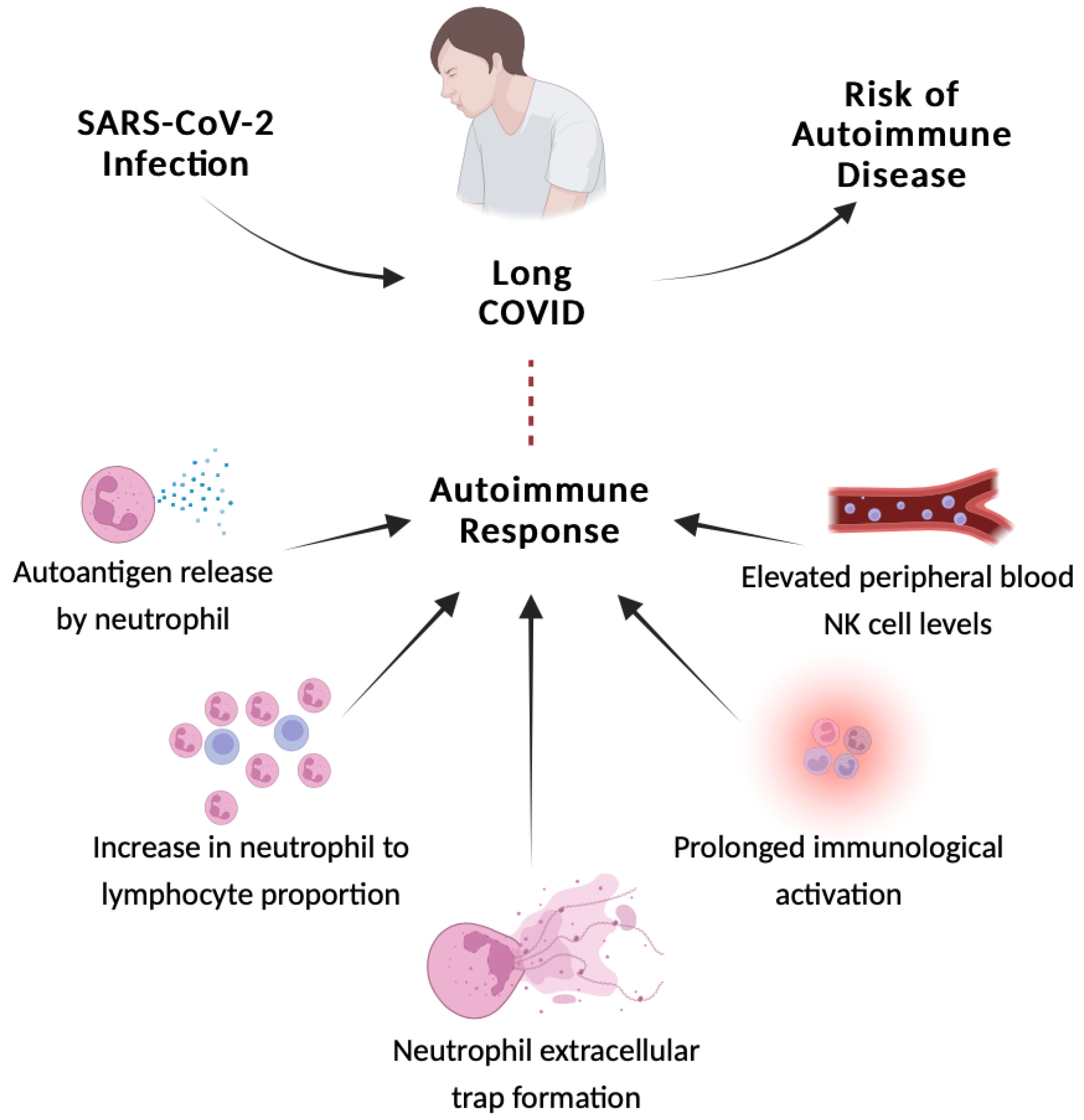
3. Autoimmune response
3.1 Adaptive immune cells
3.2 Innate immune cells
4. Risk of autoimmune diseases
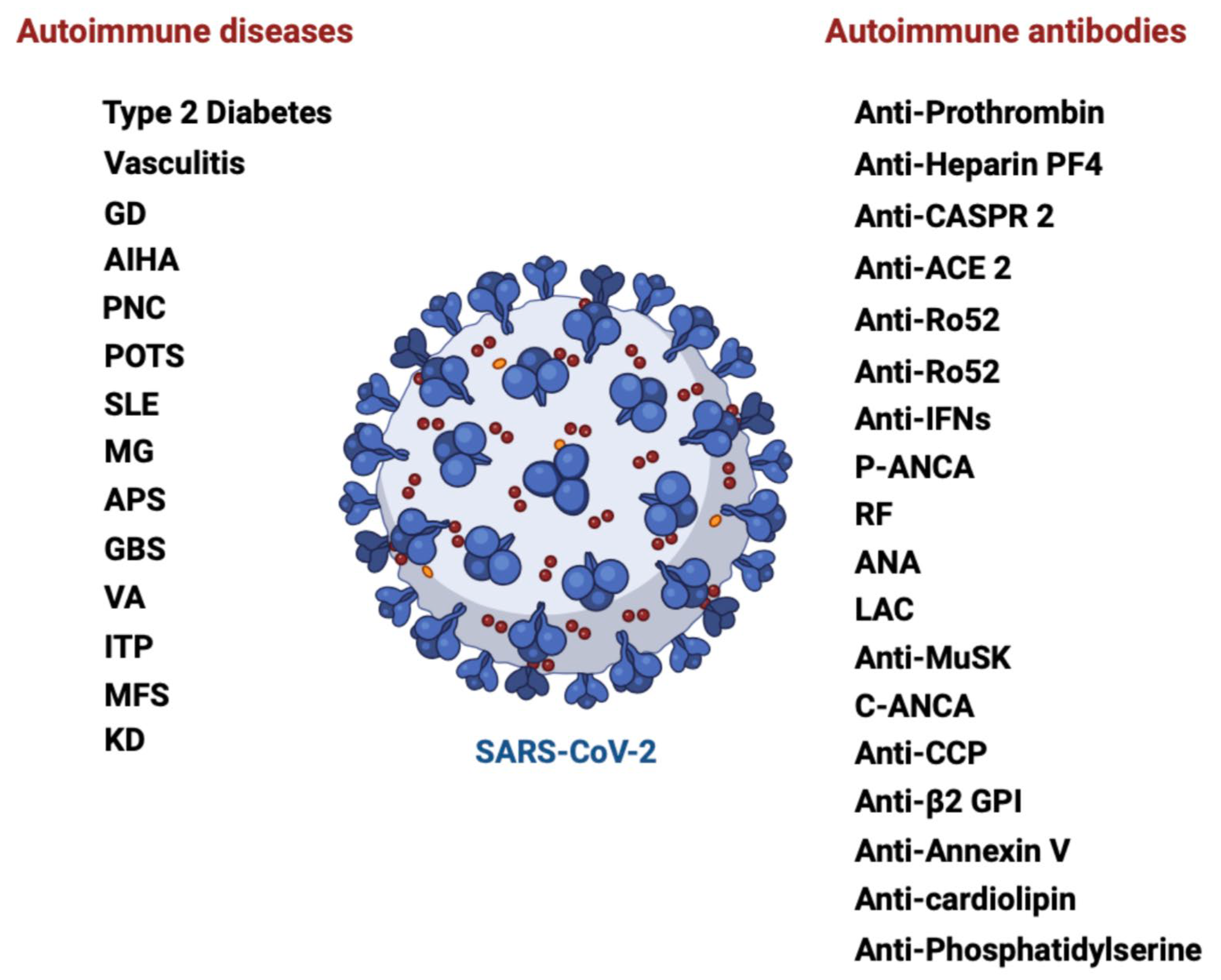
5. Elevated levels of autoantibodies and long COVID-19
6. The roles of the microbiota in the immune system and COVID-19 severity
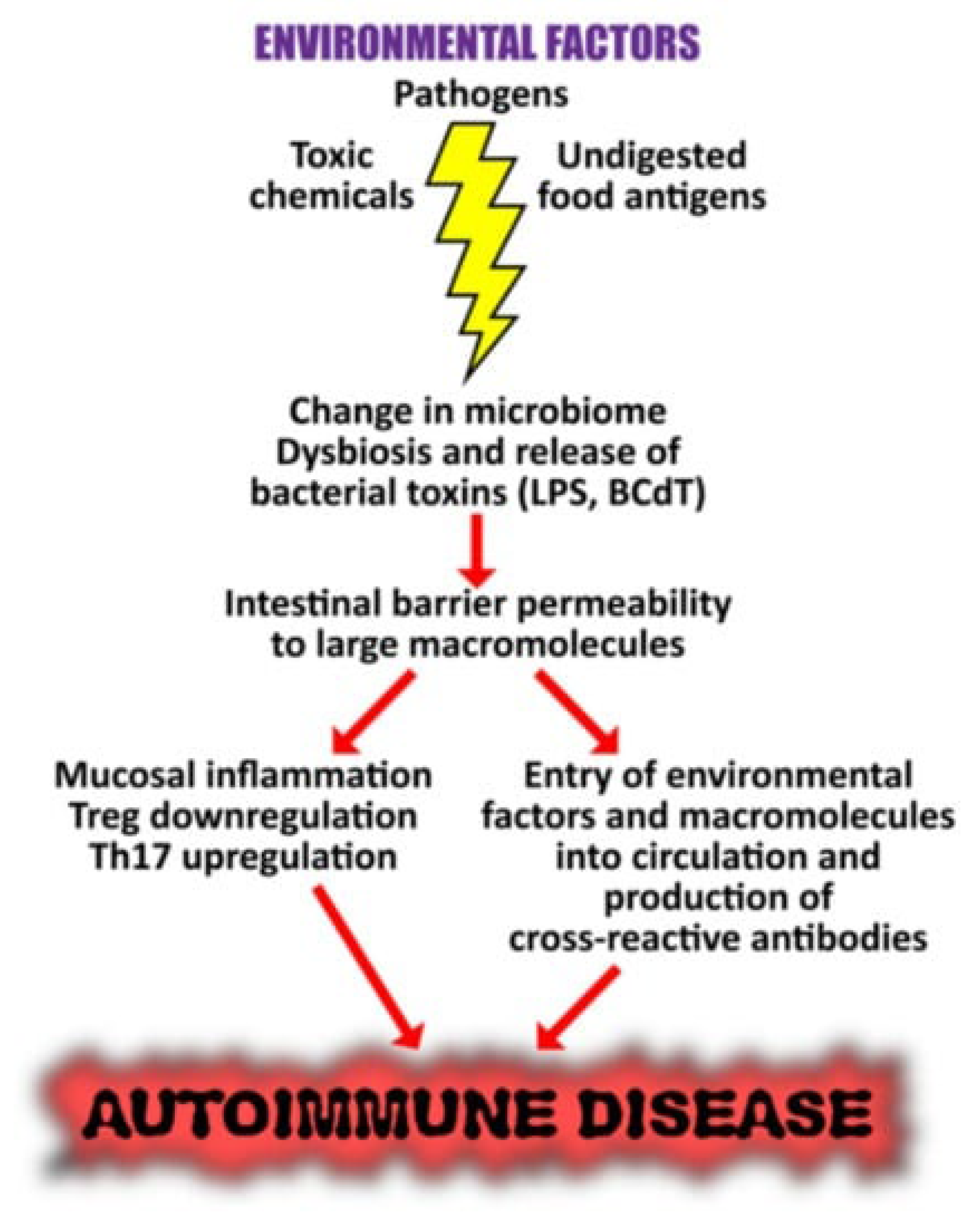
7. Altered microbiota in long COVID and its connection with autoimmunity
7.1. Connection between altered microbiome and the onset of autoimmunity
8. Reinstatement of microbiota as a potential treatment for long COVID-19
9. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hu, B.; Guo, H.; Zhou, P.; Shi, Z.L. Characteristics of SARS-CoV-2 and COVID-19. Nat Rev Microbiol 2021, 19, 141–154. [Google Scholar] [CrossRef]
- Lundstrom, K.; Hromić-Jahjefendić, A.; Bilajac, E.; Aljabali, A.A.A.; Baralić, K.; Sabri, N.A.; Shehata, E.M.; Raslan, M.; Ferreira, A.; Orlandi, L.; et al. COVID-19 signalome: Pathways for SARS-CoV-2 infection and impact on COVID-19 associated comorbidity. Cell Signal 2023, 101, 110495. [Google Scholar] [CrossRef] [PubMed]
- Hromić-Jahjefendić, A.; Barh, D.; Uversky, V.; Aljabali, A.A.; Tambuwala, M.M.; Alzahrani, K.J.; Alzahrani, F.M.; Alshammeri, S.; Lundstrom, K. Can COVID-19 Vaccines Induce Premature Non-Communicable Diseases: Where Are We Heading to? Vaccines (Basel) 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Hromić-Jahjefendić, A.; Barh, D.; Ramalho Pinto, C.H.; Gabriel Rodrigues Gomes, L.; Picanço Machado, J.L.; Afolabi, O.O.; Tiwari, S.; Aljabali, A.A.A.; Tambuwala, M.M.; Serrano-Aroca, Á.; et al. Associations and Disease-Disease Interactions of COVID-19 with Congenital and Genetic Disorders: A Comprehensive Review. Viruses 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- El-Baky, N.A.; Amara, A.A.; Uversky, V.N.; Redwan, E.M. Intrinsic factors behind long COVID: III. Persistence of SARS-CoV-2 and its components. J Cell Biochem 2024, 125, 22–44. [Google Scholar] [CrossRef]
- El-Maradny, Y.A.; Rubio-Casillas, A.; Mohamed, K.I.; Uversky, V.N.; Redwan, E.M. Intrinsic factors behind long-COVID: II. SARS-CoV-2, extracellular vesicles, and neurological disorders. J Cell Biochem 2023, 124, 1466–1485. [Google Scholar] [CrossRef] [PubMed]
- El-Maradny, Y.A.; Rubio-Casillas, A.; Uversky, V.N.; Redwan, E.M. Intrinsic factors behind long-COVID: I. Prevalence of the extracellular vesicles. J Cell Biochem 2023, 124, 656–673. [Google Scholar] [CrossRef]
- Saad, M.H.; Badierah, R.; Redwan, E.M.; El-Fakharany, E.M. A Comprehensive Insight into the Role of Exosomes in Viral Infection: Dual Faces Bearing Different Functions. Pharmaceutics 2021, 13. [Google Scholar] [CrossRef]
- Elrashdy, F.; Aljaddawi, A.A.; Redwan, E.M.; Uversky, V.N. On the potential role of exosomes in the COVID-19 reinfection/reactivation opportunity. J Biomol Struct Dyn 2021, 39, 5831–5842. [Google Scholar] [CrossRef]
- Ballering, A.V.; van Zon, S.K.R.; Olde Hartman, T.C.; Rosmalen, J.G.M. Persistence of somatic symptoms after COVID-19 in the Netherlands: an observational cohort study. Lancet 2022, 400, 452–461. [Google Scholar] [CrossRef]
- Ceban, F.; Ling, S.; Lui, L.M.W.; Lee, Y.; Gill, H.; Teopiz, K.M.; Rodrigues, N.B.; Subramaniapillai, M.; Di Vincenzo, J.D.; Cao, B.; et al. Fatigue and cognitive impairment in Post-COVID-19 Syndrome: A systematic review and meta-analysis. Brain Behav Immun 2022, 101, 93–135. [Google Scholar] [CrossRef]
- Al-Aly, Z.; Bowe, B.; Xie, Y. Long COVID after breakthrough SARS-CoV-2 infection. Nat Med 2022, 28, 1461–1467. [Google Scholar] [CrossRef]
- Davis, H.E.; McCorkell, L.; Vogel, J.M.; Topol, E.J. Long COVID: major findings, mechanisms and recommendations. Nat Rev Microbiol 2023, 21, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Greenhalgh, T.; Knight, M.; A'Court, C.; Buxton, M.; Husain, L. Management of post-acute covid-19 in primary care. Bmj 2020, 370, m3026. [Google Scholar] [CrossRef] [PubMed]
- Garrigues, E.; Janvier, P.; Kherabi, Y.; Le Bot, A.; Hamon, A.; Gouze, H.; Doucet, L.; Berkani, S.; Oliosi, E.; Mallart, E.; et al. Post-discharge persistent symptoms and health-related quality of life after hospitalization for COVID-19. J Infect 2020, 81, e4–e6. [Google Scholar] [CrossRef] [PubMed]
- Peluso, M.J.; Deeks, S.G. Early clues regarding the pathogenesis of long-COVID. Trends Immunol 2022, 43, 268–270. [Google Scholar] [CrossRef] [PubMed]
- Al-Hadrawi, D.S.; Al-Rubaye, H.T.; Almulla, A.F.; Al-Hakeim, H.K.; Maes, M. Lowered oxygen saturation and increased body temperature in acute COVID-19 largely predict chronic fatigue syndrome and affective symptoms due to Long COVID: A precision nomothetic approach. Acta Neuropsychiatr 2023, 35, 76–87. [Google Scholar] [CrossRef] [PubMed]
- Wigerblad, G.; Kaplan, M.J. NETs spread ever wider in rheumatic diseases. Nat Rev Rheumatol 2020, 16, 73–74. [Google Scholar] [CrossRef] [PubMed]
- Muller, S.; Radic, M. Oxidation and mitochondrial origin of NET DNA in the pathogenesis of lupus. Nat Med 2016, 22, 126–127. [Google Scholar] [CrossRef]
- Thierry, A.R.; Roch, B. Neutrophil Extracellular Traps and By-Products Play a Key Role in COVID-19: Pathogenesis, Risk Factors, and Therapy. J Clin Med 2020, 9. [Google Scholar] [CrossRef]
- Zuo, Y.; Yalavarthi, S.; Shi, H.; Gockman, K.; Zuo, M.; Madison, J.A.; Blair, C.; Weber, A.; Barnes, B.J.; Egeblad, M.; et al. Neutrophil extracellular traps in COVID-19. JCI Insight 2020, 5. [Google Scholar] [CrossRef] [PubMed]
- Dotan, A.; Muller, S.; Kanduc, D.; David, P.; Halpert, G.; Shoenfeld, Y. The SARS-CoV-2 as an instrumental trigger of autoimmunity. Autoimmun Rev 2021, 20, 102792. [Google Scholar] [CrossRef] [PubMed]
- Ortona, E.; Malorni, W. Long COVID: to investigate immunological mechanisms and sex/gender related aspects as fundamental steps for tailored therapy. Eur Respir J 2022, 59. [Google Scholar] [CrossRef] [PubMed]
- Lenti, M.V.; Rossi, C.M.; Melazzini, F.; Gastaldi, M.; Bugatti, S.; Rotondi, M.; Bianchi, P.I.; Gentile, A.; Chiovato, L.; Montecucco, C.; et al. Seronegative autoimmune diseases: A challenging diagnosis. Autoimmun Rev 2022, 21, 103143. [Google Scholar] [CrossRef] [PubMed]
- Halpert, G.; Shoenfeld, Y. SARS-CoV-2, the autoimmune virus. Autoimmun Rev 2020, 19, 102695. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Han, T.; Chen, J.; Hou, C.; Hua, L.; He, S.; Guo, Y.; Zhang, S.; Wang, Y.; Yuan, J.; et al. Clinical and Autoimmune Characteristics of Severe and Critical Cases of COVID-19. Clin Transl Sci 2020, 13, 1077–1086. [Google Scholar] [CrossRef] [PubMed]
- Bastard, P.; Rosen, L.B.; Zhang, Q.; Michailidis, E.; Hoffmann, H.H.; Zhang, Y.; Dorgham, K.; Philippot, Q.; Rosain, J.; Béziat, V.; et al. Autoantibodies against type I IFNs in patients with life-threatening COVID-19. Science 2020, 370. [Google Scholar] [CrossRef] [PubMed]
- Acosta-Ampudia, Y.; Monsalve, D.M.; Rojas, M.; Rodríguez, Y.; Zapata, E.; Ramírez-Santana, C.; Anaya, J.M. Persistent Autoimmune Activation and Proinflammatory State in Post-Coronavirus Disease 2019 Syndrome. J Infect Dis 2022, 225, 2155–2162. [Google Scholar] [CrossRef]
- Rojas, M.; Rodríguez, Y.; Acosta-Ampudia, Y.; Monsalve, D.M.; Zhu, C.; Li, Q.Z.; Ramírez-Santana, C.; Anaya, J.M. Autoimmunity is a hallmark of post-COVID syndrome. J Transl Med 2022, 20, 129. [Google Scholar] [CrossRef]
- Peluso, M.J.; Mitchell, A.; Wang, C.Y.; Takahashi, S.; Hoh, R.; Tai, V.; Durstenfeld, M.S.; Hsue, P.Y.; Kelly, J.D.; Martin, J.N.; et al. Low Prevalence of Interferon α Autoantibodies in People Experiencing Symptoms of Post-Coronavirus Disease 2019 (COVID-19) Conditions, or Long COVID. J Infect Dis 2023, 227, 246–250. [Google Scholar] [CrossRef]
- Son, K.; Jamil, R.; Chowdhury, A.; Mukherjee, M.; Venegas, C.; Miyasaki, K.; Zhang, K.; Patel, Z.; Salter, B.; Yuen, A.C.Y.; et al. Circulating anti-nuclear autoantibodies in COVID-19 survivors predict long COVID symptoms. Eur Respir J 2023, 61. [Google Scholar] [CrossRef] [PubMed]
- Wallukat, G.; Hohberger, B.; Wenzel, K.; Fürst, J.; Schulze-Rothe, S.; Wallukat, A.; Hönicke, A.S.; Müller, J. Functional autoantibodies against G-protein coupled receptors in patients with persistent Long-COVID-19 symptoms. J Transl Autoimmun 2021, 4, 100100. [Google Scholar] [CrossRef]
- Vergelli, M.; Hemmer, B.; Muraro, P.A.; Tranquill, L.; Biddison, W.E.; Sarin, A.; McFarland, H.F.; Martin, R. Human autoreactive CD4+ T cell clones use perforin- or Fas/Fas ligand-mediated pathways for target cell lysis. J Immunol 1997, 158, 2756–2761. [Google Scholar] [CrossRef] [PubMed]
- Ando, K.; Hiroishi, K.; Kaneko, T.; Moriyama, T.; Muto, Y.; Kayagaki, N.; Yagita, H.; Okumura, K.; Imawari, M. Perforin, Fas/Fas ligand, and TNF-alpha pathways as specific and bystander killing mechanisms of hepatitis C virus-specific human CTL. J Immunol 1997, 158, 5283–5291. [Google Scholar] [CrossRef] [PubMed]
- Praper, T.; Sonnen, A.; Viero, G.; Kladnik, A.; Froelich, C.J.; Anderluh, G.; Dalla Serra, M.; Gilbert, R.J. Human perforin employs different avenues to damage membranes. J Biol Chem 2011, 286, 2946–2955. [Google Scholar] [CrossRef] [PubMed]
- Stewart, S.E.; Kondos, S.C.; Matthews, A.Y.; D'Angelo, M.E.; Dunstone, M.A.; Whisstock, J.C.; Trapani, J.A.; Bird, P.I. The perforin pore facilitates the delivery of cationic cargos. J Biol Chem 2014, 289, 9172–9181. [Google Scholar] [CrossRef] [PubMed]
- Law, R.H.; Lukoyanova, N.; Voskoboinik, I.; Caradoc-Davies, T.T.; Baran, K.; Dunstone, M.A.; D'Angelo, M.E.; Orlova, E.V.; Coulibaly, F.; Verschoor, S.; et al. The structural basis for membrane binding and pore formation by lymphocyte perforin. Nature 2010, 468, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Ruenjaiman, V.; Sodsai, P.; Kueanjinda, P.; Bunrasmee, W.; Klinchanhom, S.; Reantragoon, R.; Tunvirachaisakul, C.; Manothummetha, K.; Mejun, N.; Liengswangwong, K.; et al. Impact of SARS-CoV-2 infection on the profiles and responses of innate immune cells after recovery. J Microbiol Immunol Infect 2022, 55, 993–1004. [Google Scholar] [CrossRef] [PubMed]
- Queiroz, M.A.F.; Neves, P.; Lima, S.S.; Lopes, J.D.C.; Torres, M.; Vallinoto, I.; Bichara, C.D.A.; Dos Santos, E.F.; de Brito, M.; da Silva, A.L.S.; et al. Cytokine Profiles Associated With Acute COVID-19 and Long COVID-19 Syndrome. Front Cell Infect Microbiol 2022, 12, 922422. [Google Scholar] [CrossRef]
- Arthur, J.M.; Forrest, J.C.; Boehme, K.W.; Kennedy, J.L.; Owens, S.; Herzog, C.; Liu, J.; Harville, T.O. Development of ACE2 autoantibodies after SARS-CoV-2 infection. PLoS One 2021, 16, e0257016. [Google Scholar] [CrossRef]
- Wang, E.Y.; Mao, T.; Klein, J.; Dai, Y.; Huck, J.D.; Jaycox, J.R.; Liu, F.; Zhou, T.; Israelow, B.; Wong, P.; et al. Diverse functional autoantibodies in patients with COVID-19. Nature 2021, 595, 283–288. [Google Scholar] [CrossRef] [PubMed]
- Gusev, E.; Sarapultsev, A.; Solomatina, L.; Chereshnev, V. SARS-CoV-2-Specific Immune Response and the Pathogenesis of COVID-19. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef] [PubMed]
- Hammoudeh, S.M.; Hammoudeh, A.M.; Bhamidimarri, P.M.; Al Safar, H.; Mahboub, B.; Künstner, A.; Busch, H.; Halwani, R.; Hamid, Q.; Rahmani, M.; et al. Systems Immunology Analysis Reveals the Contribution of Pulmonary and Extrapulmonary Tissues to the Immunopathogenesis of Severe COVID-19 Patients. Front Immunol 2021, 12, 595150. [Google Scholar] [CrossRef] [PubMed]
- Chang, R.; Yen-Ting Chen, T.; Wang, S.I.; Hung, Y.M.; Chen, H.Y.; Wei, C.J. Risk of autoimmune diseases in patients with COVID-19: A retrospective cohort study. EClinicalMedicine 2023, 56, 101783. [Google Scholar] [CrossRef] [PubMed]
- Visvabharathy, L.; Zhu, C.; Orban, Z.S.; Yarnoff, K.; Palacio, N.; Jimenez, M.; Lim, P.H.; Penaloza-MacMaster, P.; Koralnik, I.J. Autoantibody production is enhanced after mild SARS-CoV-2 infection despite vaccination in individuals with and without long COVID. medRxiv 2023. [Google Scholar] [CrossRef] [PubMed]
- Peluso, M.J.; Deveau, T.M.; Munter, S.E.; Ryder, D.; Buck, A.; Beck-Engeser, G.; Chan, F.; Lu, S.; Goldberg, S.A.; Hoh, R.; et al. Chronic viral coinfections differentially affect the likelihood of developing long COVID. J Clin Invest 2023, 133. [Google Scholar] [CrossRef]
- Merad, M.; Blish, C.A.; Sallusto, F.; Iwasaki, A. The immunology and immunopathology of COVID-19. Science 2022, 375, 1122–1127. [Google Scholar] [CrossRef]
- Stegbauer, J.; Lee, D.H.; Seubert, S.; Ellrichmann, G.; Manzel, A.; Kvakan, H.; Muller, D.N.; Gaupp, S.; Rump, L.C.; Gold, R.; et al. Role of the renin-angiotensin system in autoimmune inflammation of the central nervous system. Proc Natl Acad Sci U S A 2009, 106, 14942–14947. [Google Scholar] [CrossRef]
- Rasini, E.; Cosentino, M.; Marino, F.; Legnaro, M.; Ferrari, M.; Guasti, L.; Venco, A.; Lecchini, S. Angiotensin II type 1 receptor expression on human leukocyte subsets: a flow cytometric and RT-PCR study. Regul Pept 2006, 134, 69–74. [Google Scholar] [CrossRef]
- Castleman, M.J.; Stumpf, M.M.; Therrien, N.R.; Smith, M.J.; Lesteberg, K.E.; Palmer, B.E.; Maloney, J.P.; Janssen, W.J.; Mould, K.J.; Beckham, J.D.; et al. SARS-CoV-2 infection relaxes peripheral B cell tolerance. J Exp Med 2022, 219. [Google Scholar] [CrossRef]
- Lee, A.R.; Woo, J.S.; Lee, S.Y.; Lee, Y.S.; Jung, J.; Lee, C.R.; Park, S.H.; Cho, M.L. SARS-CoV-2 spike protein promotes inflammatory cytokine activation and aggravates rheumatoid arthritis. Cell Commun Signal 2023, 21, 44. [Google Scholar] [CrossRef] [PubMed]
- Patel, V.B.; Mori, J.; McLean, B.A.; Basu, R.; Das, S.K.; Ramprasath, T.; Parajuli, N.; Penninger, J.M.; Grant, M.B.; Lopaschuk, G.D.; et al. ACE2 Deficiency Worsens Epicardial Adipose Tissue Inflammation and Cardiac Dysfunction in Response to Diet-Induced Obesity. Diabetes 2016, 65, 85–95. [Google Scholar] [CrossRef]
- Shah, W.; Hillman, T.; Playford, E.D.; Hishmeh, L. Managing the long term effects of covid-19: summary of NICE, SIGN, and RCGP rapid guideline. Bmj 2021, 372, n136. [Google Scholar] [CrossRef] [PubMed]
- Gyöngyösi, M.; Alcaide, P.; Asselbergs, F.W.; Brundel, B.; Camici, G.G.; Martins, P.D.C.; Ferdinandy, P.; Fontana, M.; Girao, H.; Gnecchi, M.; et al. Long COVID and the cardiovascular system-elucidating causes and cellular mechanisms in order to develop targeted diagnostic and therapeutic strategies: a joint Scientific Statement of the ESC Working Groups on Cellular Biology of the Heart and Myocardial and Pericardial Diseases. Cardiovasc Res 2023, 119, 336–356. [Google Scholar] [CrossRef] [PubMed]
- Pisareva, E.; Badiou, S.; Mihalovičová, L.; Mirandola, A.; Pastor, B.; Kudriavtsev, A.; Berger, M.; Roubille, C.; Fesler, P.; Klouche, K.; et al. Persistence of neutrophil extracellular traps and anticardiolipin auto-antibodies in post-acute phase COVID-19 patients. J Med Virol 2023, 95, e28209. [Google Scholar] [CrossRef] [PubMed]
- Di Iorio, M.; Cook, C.E.; Vanni, K.M.M.; Patel, N.J.; D'Silva, K.M.; Fu, X.; Wang, J.; Prisco, L.C.; Kowalski, E.; Zaccardelli, A.; et al. DMARD disruption, rheumatic disease flare, and prolonged COVID-19 symptom duration after acute COVID-19 among patients with rheumatic disease: A prospective study. Semin Arthritis Rheum 2022, 55, 152025. [Google Scholar] [CrossRef]
- Seeßle, J.; Waterboer, T.; Hippchen, T.; Simon, J.; Kirchner, M.; Lim, A.; Müller, B.; Merle, U. Persistent Symptoms in Adult Patients 1 Year After Coronavirus Disease 2019 (COVID-19): A Prospective Cohort Study. Clin Infect Dis 2022, 74, 1191–1198. [Google Scholar] [CrossRef]
- Zhang, Q.; Bastard, P.; Cobat, A.; Casanova, J.L. Human genetic and immunological determinants of critical COVID-19 pneumonia. Nature 2022, 603, 587–598. [Google Scholar] [CrossRef]
- van der Wijst, M.G.P.; Vazquez, S.E.; Hartoularos, G.C.; Bastard, P.; Grant, T.; Bueno, R.; Lee, D.S.; Greenland, J.R.; Sun, Y.; Perez, R.; et al. Type I interferon autoantibodies are associated with systemic immune alterations in patients with COVID-19. Sci Transl Med 2021, 13, eabh2624. [Google Scholar] [CrossRef]
- Guo, L.; Wang, G.; Wang, Y.; Zhang, Q.; Ren, L.; Gu, X.; Huang, T.; Zhong, J.; Wang, Y.; Wang, X.; et al. SARS-CoV-2-specific antibody and T-cell responses 1 year after infection in people recovered from COVID-19: a longitudinal cohort study. Lancet Microbe 2022, 3, e348–e356. [Google Scholar] [CrossRef]
- Kundura, L.; Cezar, R.; André, S.; Campos-Mora, M.; Lozano, C.; Vincent, T.; Muller, L.; Lefrant, J.Y.; Roger, C.; Claret, P.G.; et al. Low perforin expression in CD8+ T lymphocytes during the acute phase of severe SARS-CoV-2 infection predicts long COVID. Front Immunol 2022, 13, 1029006. [Google Scholar] [CrossRef] [PubMed]
- Galán, M.; Vigón, L.; Fuertes, D.; Murciano-Antón, M.A.; Casado-Fernández, G.; Domínguez-Mateos, S.; Mateos, E.; Ramos-Martín, F.; Planelles, V.; Torres, M.; et al. Persistent Overactive Cytotoxic Immune Response in a Spanish Cohort of Individuals With Long-COVID: Identification of Diagnostic Biomarkers. Front Immunol 2022, 13, 848886. [Google Scholar] [CrossRef]
- Fjelltveit, E.B.; Blomberg, B.; Kuwelker, K.; Zhou, F.; Onyango, T.B.; Brokstad, K.A.; Elyanow, R.; Kaplan, I.M.; Tøndel, C.; Mohn, K.G.I.; et al. Symptom Burden and Immune Dynamics 6 to 18 Months Following Mild Severe Acute Respiratory Syndrome Coronavirus 2 Infection (SARS-CoV-2): A Case-control Study. Clin Infect Dis 2023, 76, e60–e70. [Google Scholar] [CrossRef]
- Wiech, M.; Chroscicki, P.; Swatler, J.; Stepnik, D.; De Biasi, S.; Hampel, M.; Brewinska-Olchowik, M.; Maliszewska, A.; Sklinda, K.; Durlik, M.; et al. Remodeling of T Cell Dynamics During Long COVID Is Dependent on Severity of SARS-CoV-2 Infection. Front Immunol 2022, 13, 886431. [Google Scholar] [CrossRef] [PubMed]
- Hornig, M.; Montoya, J.G.; Klimas, N.G.; Levine, S.; Felsenstein, D.; Bateman, L.; Peterson, D.L.; Gottschalk, C.G.; Schultz, A.F.; Che, X.; et al. Distinct plasma immune signatures in ME/CFS are present early in the course of illness. Sci Adv 2015, 1. [Google Scholar] [CrossRef] [PubMed]
- Tesch, F.; Ehm, F.; Vivirito, A.; Wende, D.; Batram, M.; Loser, F.; Menzer, S.; Jacob, J.; Roessler, M.; Seifert, M.; et al. Incident autoimmune diseases in association with SARS-CoV-2 infection: a matched cohort study. Clin Rheumatol 2023, 42, 2905–2914. [Google Scholar] [CrossRef] [PubMed]
- Sharma, C.; Ganigara, M.; Galeotti, C.; Burns, J.; Berganza, F.M.; Hayes, D.A.; Singh-Grewal, D.; Bharath, S.; Sajjan, S.; Bayry, J. Multisystem inflammatory syndrome in children and Kawasaki disease: a critical comparison. Nat Rev Rheumatol 2021, 17, 731–748. [Google Scholar] [CrossRef] [PubMed]
- Altmann, D.M.; Reynolds, C.J.; Joy, G.; Otter, A.D.; Gibbons, J.M.; Pade, C.; Swadling, L.; Maini, M.K.; Brooks, T.; Semper, A.; et al. Persistent symptoms after COVID-19 are not associated with differential SARS-CoV-2 antibody or T cell immunity. Nat Commun 2023, 14, 5139. [Google Scholar] [CrossRef]
- Peng, K.; Li, X.; Yang, D.; Chan, S.C.W.; Zhou, J.; Wan, E.Y.F.; Chui, C.S.L.; Lai, F.T.T.; Wong, C.K.H.; Chan, E.W.Y.; et al. Risk of autoimmune diseases following COVID-19 and the potential protective effect from vaccination: a population-based cohort study. EClinicalMedicine 2023, 63, 102154. [Google Scholar] [CrossRef]
- Cavalli, E.; Bramanti, A.; Ciurleo, R.; Tchorbanov, A.I.; Giordano, A.; Fagone, P.; Belizna, C.; Bramanti, P.; Shoenfeld, Y.; Nicoletti, F. Entangling COVID-19 associated thrombosis into a secondary antiphospholipid antibody syndrome: Diagnostic and therapeutic perspectives (Review). Int J Mol Med 2020, 46, 903–912. [Google Scholar] [CrossRef]
- Trahtemberg, U.; Rottapel, R.; Dos Santos, C.C.; Slutsky, A.S.; Baker, A.; Fritzler, M.J. Anticardiolipin and other antiphospholipid antibodies in critically ill COVID-19 positive and negative patients. Ann Rheum Dis 2021, 80, 1236–1240. [Google Scholar] [CrossRef]
- Juanes-Velasco, P.; Landeira-Viñuela, A.; García-Vaquero, M.L.; Lecrevisse, Q.; Herrero, R.; Ferruelo, A.; Góngora, R.; Corrales, F.; Rivas, J.L.; Lorente, J.A.; et al. SARS-CoV-2 Infection Triggers Auto-Immune Response in ARDS. Front Immunol 2022, 13, 732197. [Google Scholar] [CrossRef]
- Taeschler, P.; Cervia, C.; Zurbuchen, Y.; Hasler, S.; Pou, C.; Tan, Z.; Adamo, S.; Raeber, M.E.; Bächli, E.; Rudiger, A.; et al. Autoantibodies in COVID-19 correlate with antiviral humoral responses and distinct immune signatures. Allergy 2022, 77, 2415–2430. [Google Scholar] [CrossRef]
- Baiocchi, G.C.; Vojdani, A.; Rosenberg, A.Z.; Vojdani, E.; Halpert, G.; Ostrinski, Y.; Zyskind, I.; Filgueiras, I.S.; Schimke, L.F.; Marques, A.H.C.; et al. Cross-sectional analysis reveals autoantibody signatures associated with COVID-19 severity. J Med Virol 2023, 95, e28538. [Google Scholar] [CrossRef] [PubMed]
- Shome, M.; Chung, Y.; Chavan, R.; Park, J.G.; Qiu, J.; LaBaer, J. Serum autoantibodyome reveals that healthy individuals share common autoantibodies. Cell Rep 2022, 39, 110873. [Google Scholar] [CrossRef] [PubMed]
- Silverman, G.J.; Grönwall, C.; Vas, J.; Chen, Y. Natural autoantibodies to apoptotic cell membranes regulate fundamental innate immune functions and suppress inflammation. Discov Med 2009, 8, 151–156. [Google Scholar]
- Lynch, S.V.; Pedersen, O. The human intestinal microbiome in health and disease. New England journal of medicine 2016, 375, 2369–2379. [Google Scholar] [CrossRef] [PubMed]
- Baquero, F.; Nombela, C. The microbiome as a human organ. 2012, <italic>18</italic>, 2-4.
- Guarner, F.; Malagelada, J.-R. Gut flora in health and disease. The lancet 2003, 361, 512–519. [Google Scholar] [CrossRef]
- Gensollen, T.; Iyer, S.S.; Kasper, D.L.; Blumberg, R.S. How colonization by microbiota in early life shapes the immune system. Science 2016, 352, 539–544. [Google Scholar] [CrossRef]
- Schirmer, M.; Smeekens, S.P.; Vlamakis, H.; Jaeger, M.; Oosting, M.; Franzosa, E.A.; Ter Horst, R.; Jansen, T.; Jacobs, L.; Bonder, M.J. Linking the human gut microbiome to inflammatory cytokine production capacity. Cell 2016, 167, 1125–1136. e1128. [Google Scholar] [CrossRef]
- Arpaia, N.; Campbell, C.; Fan, X.; Dikiy, S.; Van Der Veeken, J.; Deroos, P.; Liu, H.; Cross, J.R.; Pfeffer, K.; Coffer, P.J. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature 2013, 504, 451–455. [Google Scholar] [CrossRef] [PubMed]
- Weaver, L.K.; Minichino, D.; Biswas, C.; Chu, N.; Lee, J.-J.; Bittinger, K.; Albeituni, S.; Nichols, K.E.; Behrens, E.M. Microbiota-dependent signals are required to sustain TLR-mediated immune responses. JCI insight 2019, 4. [Google Scholar] [CrossRef]
- Zuo, T.; Zhang, F.; Lui, G.C.; Yeoh, Y.K.; Li, A.Y.; Zhan, H.; Wan, Y.; Chung, A.C.; Cheung, C.P.; Chen, N. Alterations in gut microbiota of patients with COVID-19 during time of hospitalization. Gastroenterology 2020, 159, 944–955. e948. [Google Scholar] [CrossRef]
- Yeoh, Y.K.; Zuo, T.; Lui, G.C.-Y.; Zhang, F.; Liu, Q.; Li, A.Y.; Chung, A.C.; Cheung, C.P.; Tso, E.Y.; Fung, K.S. Gut microbiota composition reflects disease severity and dysfunctional immune responses in patients with COVID-19. Gut 2021, 70, 698–706. [Google Scholar] [CrossRef] [PubMed]
- Mostafa, H.H.; Fissel, J.A.; Fanelli, B.; Bergman, Y.; Gniazdowski, V.; Dadlani, M.; Carroll, K.C.; Colwell, R.R.; Simner, P.J. Metagenomic next-generation sequencing of nasopharyngeal specimens collected from confirmed and suspect COVID-19 patients. MBio 2020, 11, 10–1128. [Google Scholar] [CrossRef] [PubMed]
- Maes, M.; Higginson, E.; Pereira-Dias, J.; Curran, M.D.; Parmar, S.; Khokhar, F.; Cuchet-Lourenço, D.; Lux, J.; Sharma-Hajela, S.; Ravenhill, B. Correction to: Ventilator-associated pneumonia in critically ill patients with COVID-19. Critical Care 2021, 25. [Google Scholar] [CrossRef]
- Tay, M.Z.; Poh, C.M.; Rénia, L.; MacAry, P.A.; Ng, L.F. The trinity of COVID-19: immunity, inflammation and intervention. Nature Reviews Immunology 2020, 20, 363–374. [Google Scholar] [CrossRef]
- Liu, Y.; Chan, M.T.; Chan, F.K.; Wu, W.K.; Ng, S.C.; Zhang, L. Lower gut abundance of Eubacterium rectale is linked to COVID-19 mortality. Frontiers in Cellular and Infection Microbiology 2023, 13. [Google Scholar] [CrossRef]
- Rueca, M.; Fontana, A.; Bartolini, B.; Piselli, P.; Mazzarelli, A.; Copetti, M.; Binda, E.; Perri, F.; Gruber, C.E.M.; Nicastri, E. Investigation of nasal/oropharyngeal microbial community of COVID-19 patients by 16S rDNA sequencing. International journal of environmental research and public health 2021, 18, 2174. [Google Scholar] [CrossRef]
- Trøseid, M.; Holter, J.C.; Holm, K.; Vestad, B.; Sazonova, T.; Granerud, B.K.; Dyrhol-Riise, A.M.; Holten, A.R.; Tonby, K.; Kildal, A.B. Gut microbiota composition during hospitalization is associated with 60-day mortality after severe COVID-19. Critical Care 2023, 27, 69. [Google Scholar] [CrossRef]
- Camargo, S.M.; Singer, D.; Makrides, V.; Huggel, K.; Pos, K.M.; Wagner, C.A.; Kuba, K.; Danilczyk, U.; Skovby, F.; Kleta, R. Tissue-specific amino acid transporter partners ACE2 and collectrin differentially interact with hartnup mutations. Gastroenterology 2009, 136, 872–882882. e873. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, T.; Perlot, T.; Rehman, A.; Trichereau, J.; Ishiguro, H.; Paolino, M.; Sigl, V.; Hanada, T.; Hanada, R.; Lipinski, S. ACE2 links amino acid malnutrition to microbial ecology and intestinal inflammation. Nature 2012, 487, 477–481. [Google Scholar] [CrossRef]
- Perlot, T.; Penninger, J.M. ACE2–From the renin–angiotensin system to gut microbiota and malnutrition. Microbes and infection 2013, 15, 866–873. [Google Scholar] [CrossRef]
- Viana, S.D.; Nunes, S.; Reis, F. ACE2 imbalance as a key player for the poor outcomes in COVID-19 patients with age-related comorbidities–role of gut microbiota dysbiosis. Ageing research reviews 2020, 62, 101123. [Google Scholar] [CrossRef]
- Liu, Q.; Mak, J.W.Y.; Su, Q.; Yeoh, Y.K.; Lui, G.C.-Y.; Ng, S.S.S.; Zhang, F.; Li, A.Y.; Lu, W.; Hui, D.S.-C. Gut microbiota dynamics in a prospective cohort of patients with post-acute COVID-19 syndrome. Gut 2022, 71, 544–552. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Zhou, Y.; Ma, Y.; Chen, P.; Tang, J.; Yang, B.; Li, H.; Liang, M.; Xue, Y.; Liu, Y. Gut microbiota dysbiosis correlates with long COVID-19 at one-year after discharge. Journal of Korean Medical Science 2023, 38. [Google Scholar] [CrossRef]
- Vojdani, A.; Vojdani, E.; Rosenberg, A.Z.; Shoenfeld, Y. The role of exposomes in the pathophysiology of autoimmune diseases II: Pathogens. Pathophysiology 2022, 29, 243–280. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.F.; Wang, G. Environmental agents, oxidative stress and autoimmunity. Current opinion in toxicology 2018, 7, 22–27. [Google Scholar] [CrossRef]
- Dehner, C.; Fine, R.; Kriegel, M.A. The microbiome in systemic autoimmune disease: mechanistic insights from recent studies. Current opinion in rheumatology 2019, 31, 201–207. [Google Scholar] [CrossRef]
- De Luca, F.; Shoenfeld, Y. The microbiome in autoimmune diseases. Clinical & Experimental Immunology 2019, 195, 74–85. [Google Scholar]
- Li, B.; Selmi, C.; Tang, R.; Gershwin, M.; Ma, X. The microbiome and autoimmunity: a paradigm from the gut–liver axis. Cellular & molecular immunology 2018, 15, 595–609. [Google Scholar]
- Abe, K.; Takahashi, A.; Fujita, M.; Imaizumi, H.; Hayashi, M.; Okai, K.; Ohira, H. Dysbiosis of oral microbiota and its association with salivary immunological biomarkers in autoimmune liver disease. PloS one 2018, 13, e0198757. [Google Scholar] [CrossRef]
- Choi, S.-C.; Brown, J.; Gong, M.; Ge, Y.; Zadeh, M.; Li, W.; Croker, B.P.; Michailidis, G.; Garrett, T.J.; Mohamadzadeh, M. Gut microbiota dysbiosis and altered tryptophan catabolism contribute to autoimmunity in lupus-susceptible mice. Science translational medicine 2020, 12, eaax2220. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, G.L.V.; Leite, A.Z.; Higuchi, B.S.; Gonzaga, M.I.; Mariano, V.S. Intestinal dysbiosis and probiotic applications in autoimmune diseases. Immunology 2017, 152, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Harbison, J.E.; Roth-Schulze, A.J.; Giles, L.C.; Tran, C.D.; Ngui, K.M.; Penno, M.A.; Thomson, R.L.; Wentworth, J.M.; Colman, P.G.; Craig, M.E. Gut microbiome dysbiosis and increased intestinal permeability in children with islet autoimmunity and type 1 diabetes: A prospective cohort study. Pediatric diabetes 2019, 20, 574–583. [Google Scholar] [CrossRef] [PubMed]
- Horta-Baas, G.; Romero-Figueroa, M.d.S.; Montiel-Jarquín, A.J.; Pizano-Zárate, M.L.; García-Mena, J.; Ramírez-Durán, N. Intestinal dysbiosis and rheumatoid arthritis: a link between gut microbiota and the pathogenesis of rheumatoid arthritis. Journal of immunology research 2017, 2017. [Google Scholar] [CrossRef] [PubMed]
- Mousa, W.K.; Chehadeh, F.; Husband, S. Microbial dysbiosis in the gut drives systemic autoimmune diseases. Frontiers in Immunology 2022, 13, 906258. [Google Scholar] [CrossRef]
- Maeda, Y.; Kurakawa, T.; Umemoto, E.; Motooka, D.; Ito, Y.; Gotoh, K.; Hirota, K.; Matsushita, M.; Furuta, Y.; Narazaki, M. Dysbiosis contributes to arthritis development via activation of autoreactive T cells in the intestine. Arthritis & rheumatology 2016, 68, 2646–2661. [Google Scholar]
- Alghamdi, M.A.; Redwan, E.M. Interplay of microbiota and citrullination in the immunopathogenesis of rheumatoid arthritis. Probiotics and antimicrobial proteins 2022, 14, 99–113. [Google Scholar] [CrossRef] [PubMed]
- Moulton, V.R.; Suarez-Fueyo, A.; Meidan, E.; Li, H.; Mizui, M.; Tsokos, G.C. Pathogenesis of human systemic lupus erythematosus: a cellular perspective. Trends in molecular medicine 2017, 23, 615–635. [Google Scholar] [CrossRef]
- Mu, Q.; Zhang, H.; Liao, X.; Lin, K.; Liu, H.; Edwards, M.R.; Ahmed, S.A.; Yuan, R.; Li, L.; Cecere, T.E. Control of lupus nephritis by changes of gut microbiota. Microbiome 2017, 5, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hevia, A.; Arboleya, S.; Gueimonde Fernández, M.; Margolles Barros, A. Intestinal dysbiosis associated with Systemic Lupus Erythematosus highlights microbial targets for intervention strategies. 2014.
- Azzouz, D.; Omarbekova, A.; Heguy, A.; Schwudke, D.; Gisch, N.; Rovin, B.H.; Caricchio, R.; Buyon, J.P.; Alekseyenko, A.V.; Silverman, G.J. Lupus nephritis is linked to disease-activity associated expansions and immunity to a gut commensal. Annals of the rheumatic diseases 2019, 78, 947–956. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Zhang, Z.; Yu, A.M.; Wang, W.; Wei, Z.; Akhter, E.; Maurer, K.; Reis, P.C.; Song, L.; Petri, M. The SLE transcriptome exhibits evidence of chronic endotoxin exposure and has widespread dysregulation of non-coding and coding RNAs. PloS one 2014, 9, e93846. [Google Scholar] [CrossRef] [PubMed]
- Ogunrinde, E.; Zhou, Z.; Luo, Z.; Alekseyenko, A.; Li, Q.Z.; Macedo, D.; Kamen, D.L.; Oates, J.C.; Gilkeson, G.S.; Jiang, W. A link between plasma microbial translocation, microbiome, and autoantibody development in first-degree relatives of systemic lupus erythematosus patients. Arthritis & rheumatology 2019, 71, 1858–1868. [Google Scholar]
- Tursi, S.A.; Lee, E.Y.; Medeiros, N.J.; Lee, M.H.; Nicastro, L.K.; Buttaro, B.; Gallucci, S.; Wilson, R.P.; Wong, G.C.; Tükel, Ç. Bacterial amyloid curli acts as a carrier for DNA to elicit an autoimmune response via TLR2 and TLR9. PLoS pathogens 2017, 13, e1006315. [Google Scholar] [CrossRef] [PubMed]
- Gallo, P.M.; Rapsinski, G.J.; Wilson, R.P.; Oppong, G.O.; Sriram, U.; Goulian, M.; Buttaro, B.; Caricchio, R.; Gallucci, S.; Tükel, Ç. Amyloid-DNA composites of bacterial biofilms stimulate autoimmunity. Immunity 2015, 42, 1171–1184. [Google Scholar] [CrossRef] [PubMed]
- Glassner, K.L.; Abraham, B.P.; Quigley, E.M. The microbiome and inflammatory bowel disease. Journal of Allergy and Clinical Immunology 2020, 145, 16–27. [Google Scholar] [CrossRef]
- Boziki, M.K.; Kesidou, E.; Theotokis, P.; Mentis, A.-F.A.; Karafoulidou, E.; Melnikov, M.; Sviridova, A.; Rogovski, V.; Boyko, A.; Grigoriadis, N. Microbiome in multiple sclerosis: where are we, what we know and do not know. Brain sciences 2020, 10, 234. [Google Scholar] [CrossRef]
- Zhang, H.; Liao, X.; Sparks, J.B.; Luo, X.M. Dynamics of gut microbiota in autoimmune lupus. Applied and environmental microbiology 2014, 80, 7551–7560. [Google Scholar] [CrossRef]
- Abdelhamid, L.; Luo, X.M. Retinoic acid, leaky gut, and autoimmune diseases. Nutrients 2018, 10, 1016. [Google Scholar] [CrossRef]
- Rathi, A.; Jadhav, S.B.; Shah, N. A randomized controlled trial of the efficacy of systemic enzymes and probiotics in the resolution of post-COVID fatigue. Medicines 2021, 8, 47. [Google Scholar] [CrossRef] [PubMed]
- Kedor, C.; Freitag, H.; Meyer-Arndt, L.; Wittke, K.; Zoller, T.; Steinbeis, F.; Haffke, M.; Rudolf, G.; Heidecker, B.; Volk, H. Chronic COVID-19 Syndrome and Chronic Fatigue Syndrome (ME/CFS) following the first pandemic wave in Germany–a first analysis of a prospective observational study. <italic>MedRxiv </italic>2021, 2021.2002. 2006.21249256.
- Poenaru, S.; Abdallah, S.J.; Corrales-Medina, V.; Cowan, J. COVID-19 and post-infectious myalgic encephalomyelitis/chronic fatigue syndrome: a narrative review. Therapeutic advances in infectious disease 2021, 8, 20499361211009385. [Google Scholar] [CrossRef] [PubMed]
- Rudroff, T.; Fietsam, A.C.; Deters, J.R.; Bryant, A.D.; Kamholz, J. Post-COVID-19 fatigue: potential contributing factors. Brain sciences 2020, 10, 1012. [Google Scholar] [CrossRef] [PubMed]
- Perrin, R.; Riste, L.; Hann, M.; Walther, A.; Mukherjee, A.; Heald, A. Into the looking glass: Post-viral syndrome post COVID-19. Medical hypotheses 2020, 144, 110055. [Google Scholar] [CrossRef] [PubMed]
- Toogood, P.L.; Clauw, D.J.; Phadke, S.; Hoffman, D. Myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS): Where will the drugs come from? Pharmacological Research 2021, 165, 105465. [Google Scholar] [CrossRef] [PubMed]
- Azad, M.A.K.; Sarker, M.; Wan, D. Immunomodulatory effects of probiotics on cytokine profiles. BioMed research international 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Morshedi, M.; Hashemi, R.; Moazzen, S.; Sahebkar, A.; Hosseinifard, E.-S. Immunomodulatory and anti-inflammatory effects of probiotics in multiple sclerosis: a systematic review. Journal of neuroinflammation 2019, 16, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wu, Y.; Wang, Y.; Xu, H.; Mei, X.; Yu, D.; Wang, Y.; Li, W. Antioxidant properties of probiotic bacteria. Nutrients 2017, 9, 521. [Google Scholar] [CrossRef]
- Eguchi, K.; Fujitani, N.; Nakagawa, H.; Miyazaki, T. Prevention of respiratory syncytial virus infection with probiotic lactic acid bacterium Lactobacillus gasseri SBT2055. Scientific reports 2019, 9, 4812. [Google Scholar] [CrossRef]
- Venturini, L.; Bacchi, S.; Capelli, E.; Lorusso, L.; Ricevuti, G.; Cusa, C. Modification of immunological parameters, oxidative stress markers, mood symptoms, and well-being status in CFS patients after probiotic intake: observations from a pilot study. Oxidative medicine and cellular longevity 2019, 2019. [Google Scholar] [CrossRef]
- Kim, N.; Yun, M.; Oh, Y.J.; Choi, H.-J. Mind-altering with the gut: Modulation of the gut-brain axis with probiotics. Journal of microbiology 2018, 56, 172–182. [Google Scholar] [CrossRef] [PubMed]
- Marotta, A.; Sarno, E.; Del Casale, A.; Pane, M.; Mogna, L.; Amoruso, A.; Felis, G.E.; Fiorio, M. Effects of probiotics on cognitive reactivity, mood, and sleep quality. Frontiers in psychiatry 2019, 10, 427235. [Google Scholar] [CrossRef] [PubMed]
- Ng, Q.X.; Peters, C.; Ho, C.Y.X.; Lim, D.Y.; Yeo, W.-S. A meta-analysis of the use of probiotics to alleviate depressive symptoms. Journal of affective disorders 2018, 228, 13–19. [Google Scholar] [CrossRef] [PubMed]
| Relationship between autoimmunity and long-term COVID-19 manifestations | Reference | |
| Autoimmune response | Adaptive immunity: Low perforin expression in CD8+T cells More antiviral cytotoxicity in CD8+T; Higher levels of the PD-1 marker |
[33,34,35,36] [37,38,39] |
| Innate immunity: Higher levels of monocytes (1-3 months after recovery) Lower HLA class II marker HLA-DR in COVID-19 patients Higher number of NK cells | ||
| Cytokine profile: High serum levels of IL-6, IL-17 and IL-2 (one month after infection) Low serum levels of IL-4 and IL-10 (one month after infection) | ||
| Elevated autoantibodies | to inflammatory cytokines (such as IgG to IL-2, D8B, thyroglobulin, and IFN) | [29,30] |
| to antinuclear and extractable nuclear antigens | [31] | |
| against GPCRs (including 1- and 2-adrenoceptors, angiotensin receptor, nociception-like opioid receptor, and muscarinic M2-receptor) | [32] | |
| to ACE2 (the receptor for SARS-CoV-2 entrance), 2-adrenoceptor, muscarinic M2 receptor, angiotensin II AT1 receptor, and the angiotensin 1-7 MAS receptor | [32,40] | |
| targeting tissue (connective tissue, extracellular matrix elements, vascular endothelium, coagulation factors, and platelets) | [41] | |
| targeting systemic organs (including the lung, central nervous system, skin, and gastrointestinal tract) | ||
| targeting immune-modulating proteins (cytokines, chemokines, complement components, and cell-surface proteins) | ||
| Immune dysregulation | Viral protein mimicry | [42,43] |
| Systemic display and multiorgan commitment of COVID-19 due to prevalent expression of the SARS-CoV-2 receptor ACE2 | ||
| Bystander triggering of immune cells | ||
| Release of autoantigens from virus-damaged tissue | ||
| Superantigen-mediated activation of lymphocytes | ||
| Epitope spreading | ||
| Risk of autoimmune diseases | Rheumatoid arthritis (aHR 2.98; 95% CI 2.78-3.20) | [44,45] |
| Systemic lupus erythematosus (aHR 2.99; 95% CI 2.68-3.34) | ||
| Vasculitis (aHR 1.96; 95% CI 1.74-2.20) | ||
| Inflammatory bowel disease (aHR 1.78; 95% CI 1.72-1.84) | ||
| Type 1 diabetes mellitus (aHR 2.68; 95% CI 2.51–2.85) | ||
| Systemic sclerosis (aHR:2.58, 95% CI:2.02-3.28) | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




