Submitted:
06 June 2024
Posted:
11 June 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Strains, plasmids, and constructs
Growth, solubilisation and activation of Cry toxins
Cell culture
Fluorescence microscopy using GFP-labelled Cry41Aa
Cellular competition assays
Cellular binding assays
Western blotting
Aedes aegypti bioassays
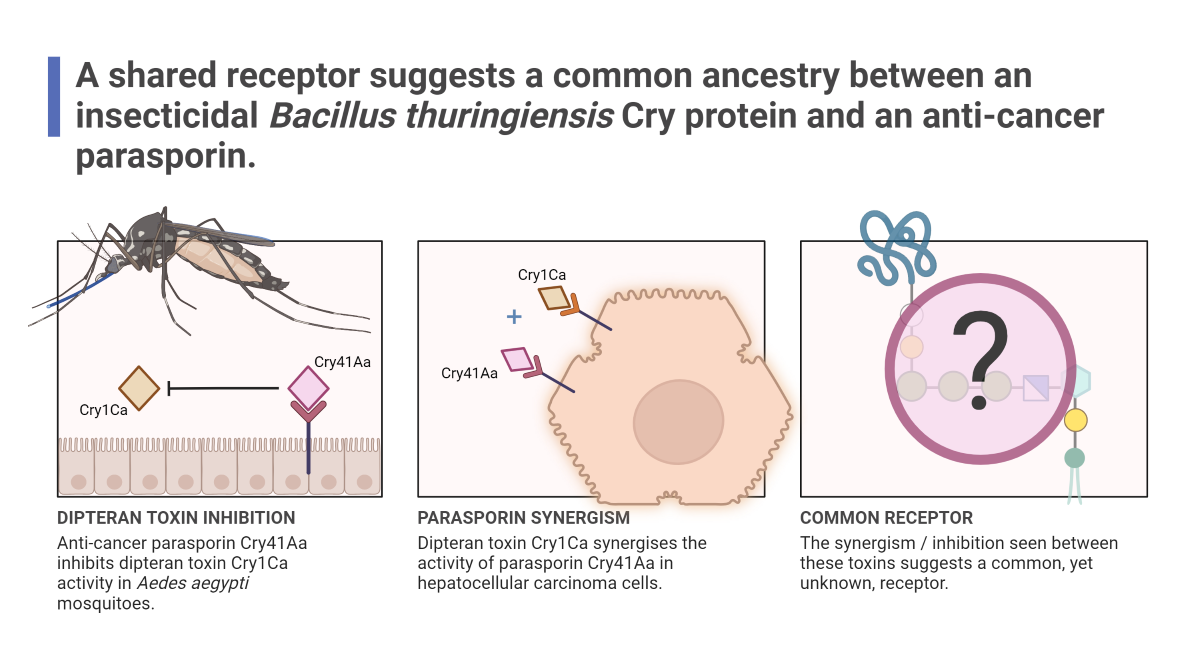
3. Results
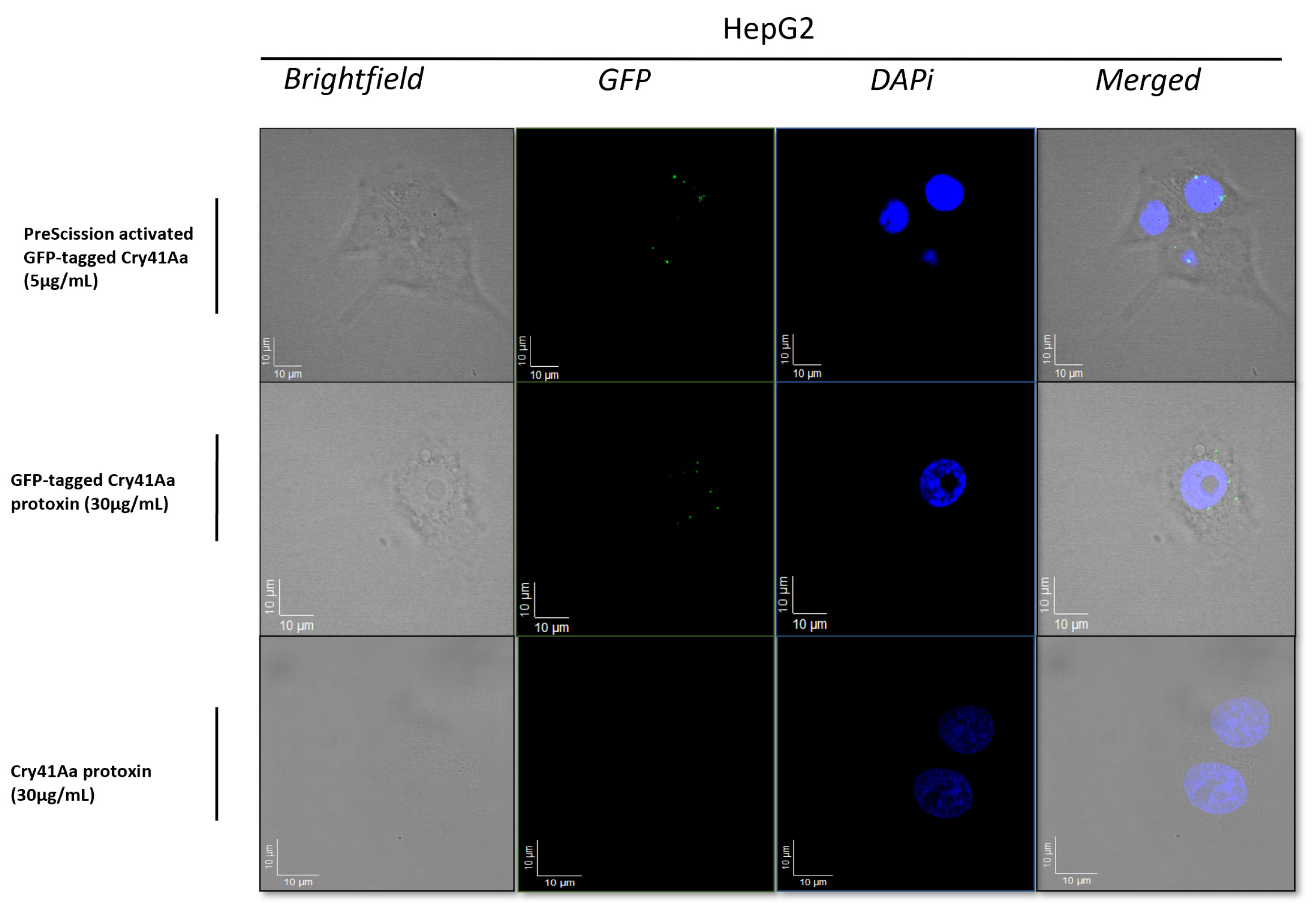
3.1. Fluorescence Microscopy Analysis of Binding of the GFP-Tagged Cry41Aa to the Surface of HepG2 Cells
3.2. Immunoblotting Confirms Cry41Aa’s Binding Capability
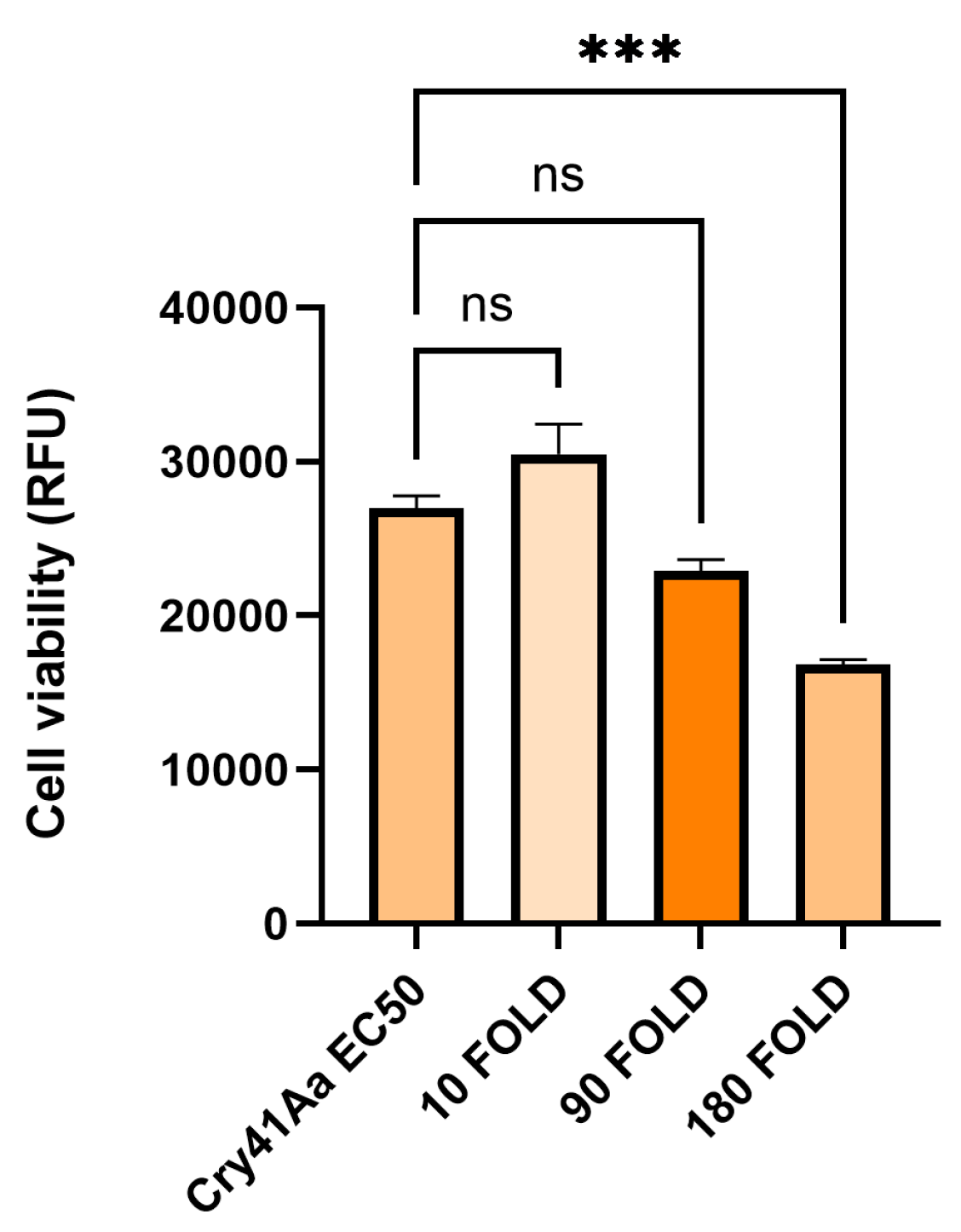
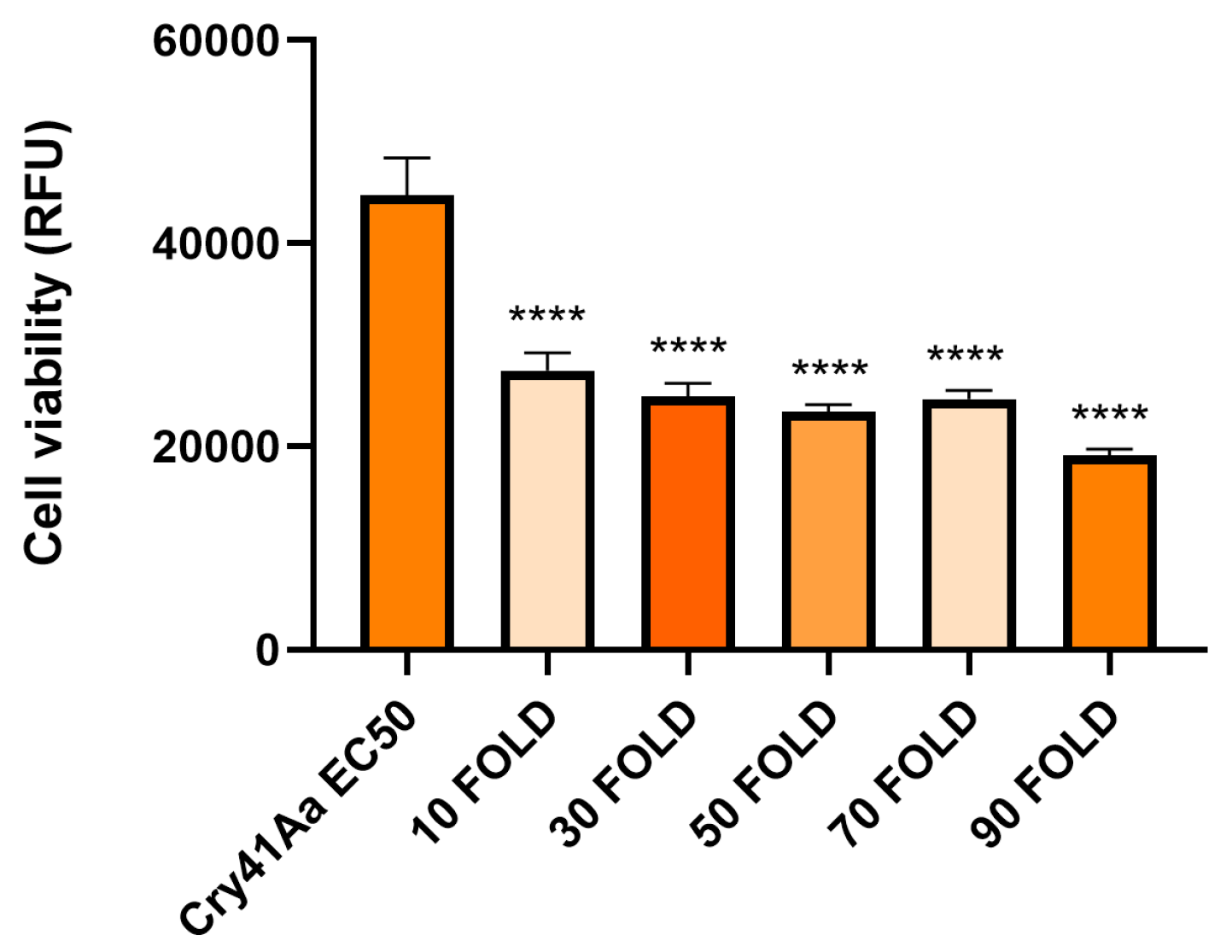
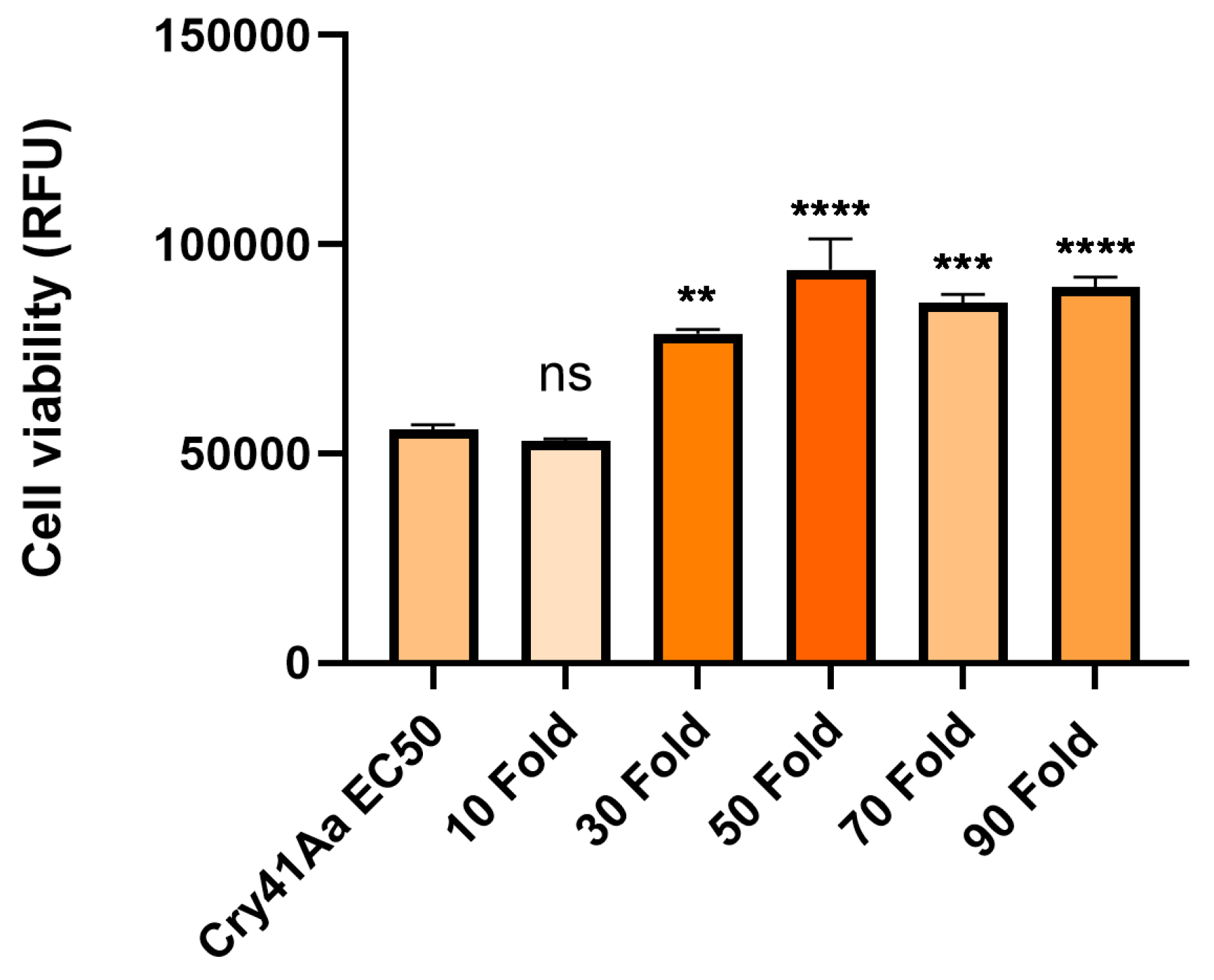
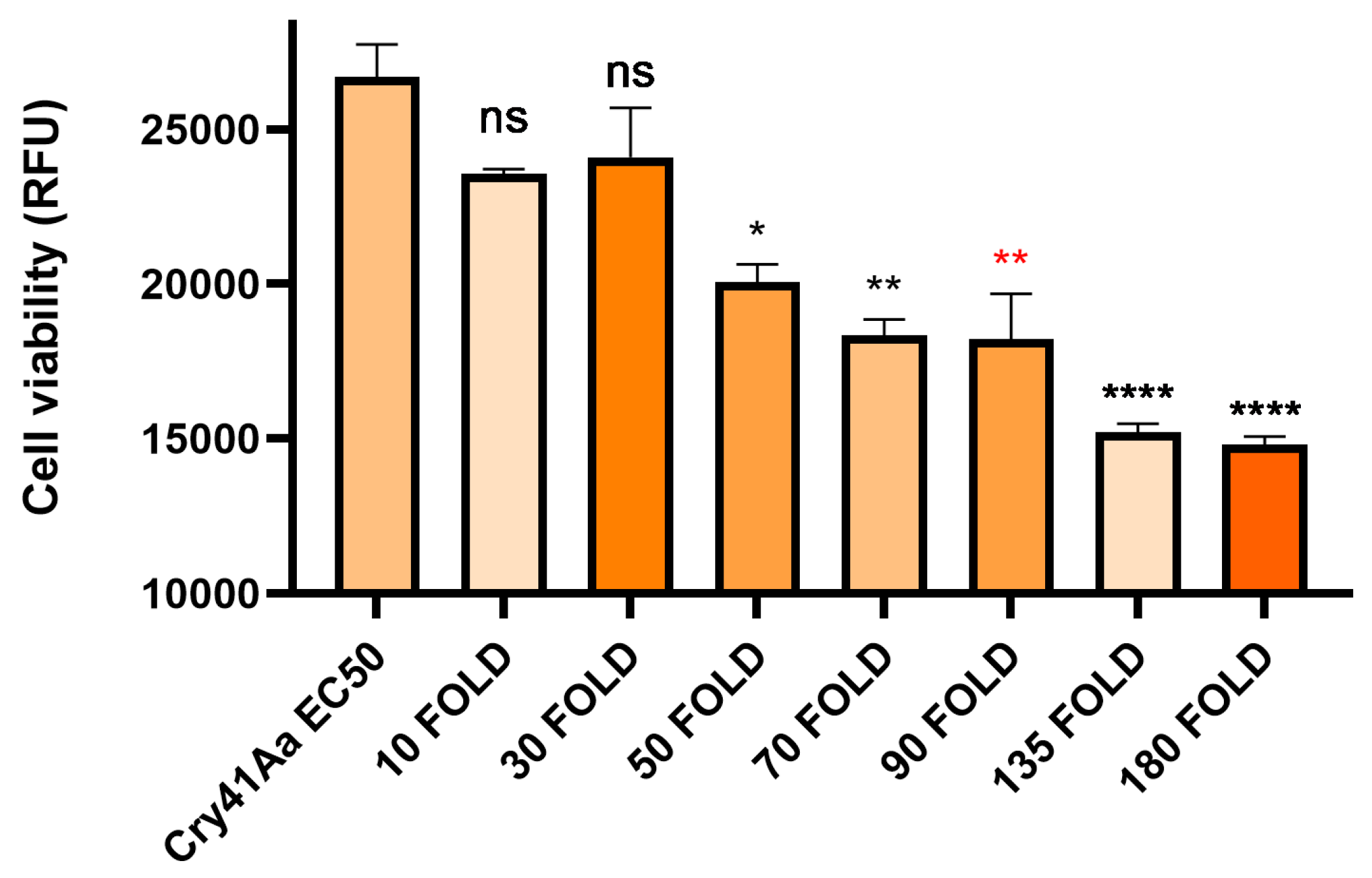
3.3. Cry41Aa and Insecticidal Cry Toxin Synergism
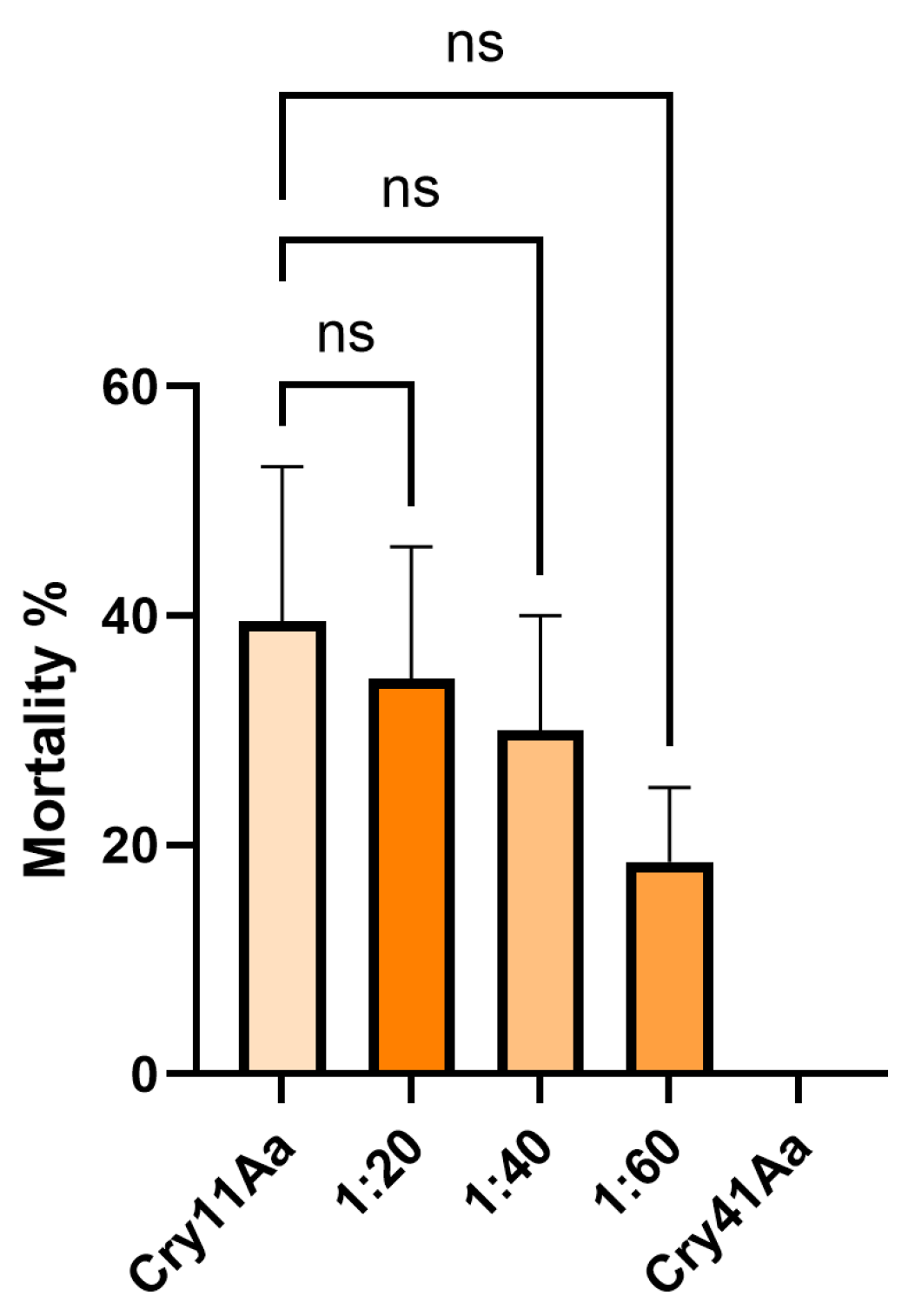
3.4. Cry41Aa Inhibits Cry1Ca Activity in Aedes aegypti Larvae
4. Discussion
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- R. de Maagd, A. Bravo, and N. Crickmore, “How Bacillus thuringiensis has evolved specific toxins to colonize the insect world,” Trends in Genetics, vol. 17, no. 4. pp. 193–199, Apr. 2001. [CrossRef]
- J. L. Jurat-Fuentes and N. Crickmore, “Specificity determinants for Cry insecticidal proteins: Insights from their mode of action,” Journal of Invertebrate Pathology, vol. 142, pp. 5–10, Jan. 2017. [CrossRef]
- V. Krishnan, B. Domanska, A. Elhigazi, F. Afolabi, M. J. West, and N. Crickmore, “The human cancer cell active toxin Cry41Aa from Bacillus thuringiensis acts like its insecticidal counterparts,” Biochemical Journal, vol. 474, no. 10, pp. 1591–1602, Apr. 2017. [CrossRef]
- W. Souissi, A. Kaloki, S. Etherington, B. Domanska, M. J. West, and N. Crickmore, “Differential proteolytic activation of the Bacillus thuringiensis Cry41Aa parasporin modulates its anticancer effect,” Biochemical Journal, vol. 476, no. 24, pp. 3805–3816, Dec. 2019. [CrossRef]
- R. Sato, “Utilization of Diverse Molecules as Receptors by Cry Toxin and the Promiscuous Nature of Receptor-Binding Sites Which Accounts for the Diversity,” Biomolecules, vol. 14, no. 4, pp. 425-425, Apr. 2024. [CrossRef]
- T. Akiba and S. Okumura, “Parasporins 1 and 2: Their structure and activity,” Journal of Invertebrate Pathology, vol. 142, pp. 44–49, Jan. 2017. [CrossRef]
- H. Endo, “Molecular and Kinetic Models for Pore Formation of Bacillus thuringiensis Cry Toxin,” Toxins, vol. 14, no. 7, p. 433, Jun. 2022. [CrossRef]
- O. Arantes and D. Lereclus, “Construction of cloning vectors for Bacillus thuringiensis,” Gene, vol. 108, no. 1, pp. 115–119, Dec. 1991. [CrossRef]
- F. A. Alzahrani and N. Crickmore, “N-terminal proteolysis determines the differential activity of Bacillus thuringiensis Cry2A toxins towards Aedes aegypti,” Journal of Invertebrate Pathology, vol. 204, pp. 108100–108100, Mar. 2024. [CrossRef]
- N. Crickmore and D. J. Ellar, “Involvement of a possible chaperonin in the efficient expression of a cloned CryIIA delta-endotoxin gene in Bacillus thuringiensis,” Molecular Microbiology, vol. 6, no. 11, pp. 1533–1537, Jun. 1992. [CrossRef]
- A. Bravo, J. Sánchez, T. Kouskoura, and N. Crickmore, “N-terminal Activation Is an Essential Early Step in the Mechanism of Action of the Bacillus thuringiensis Cry1Ac Insecticidal Toxin,” Journal of Biological Chemistry, vol. 277, no. 27, pp. 23985–23987, Jul. 2002 . [CrossRef]
- S. E. González-Villarreal, M. García-Montelongo, and J. E. Ibarra, “Insecticidal Activity of a Cry1Ca toxin of Bacillus thuringiensis Berliner (Firmicutes: Bacillaceae) and Its Synergism with the Cyt1Aa Toxin Against Aedes aegypti (Diptera: Culicidae),” Journal of Medical Entomology, vol. 57, no. 6, pp. 1852–1856, Jun. 2020. [CrossRef]
- J.-L. Schwartz et al., “Restriction of intramolecular movements within the CrylAa toxin molecule of Bacillus thuringiensis through disulfide bond engineering,” FEBS Letters, vol. 410, no. 2–3, pp. 397–402, Jun. 1997. [CrossRef]
- S. Pacheco, J. P. J. Quiliche, I. Gómez, J. Sánchez, M. Soberón, and A. Bravo, “Rearrangement of N-Terminal α-Helices of Bacillus thuringiensis Cry1Ab Toxin Essential for Oligomer Assembly and Toxicity,” Toxins, vol. 12, no. 10, p. 647, Oct. 2020. [CrossRef]
- A. Bravo, S. S. Gill, and M. Soberón, “Mode of action of Bacillus thuringiensis Cry and Cyt toxins and their potential for insect control,” Toxicon, vol. 49, no. 4, pp. 423–435, Mar. 2007. [CrossRef]
- B. E. Tabashnik et al., “Dual mode of action of Bt proteins: protoxin efficacy against resistant insects,” Scientific Reports, vol. 5, no. 1, Oct. 2015. [CrossRef]
- D. Wu and F. N. Chang, “Synergism in mosquitocidal activity of 26 and 65 kDa proteins from Bacillus thuringiensis subsp. israelensis crystal,” FEBS Letters, vol. 190, no. 2, pp. 232–236, Oct. 1985. [CrossRef]
- R. Monnerat et al., “Synergistic activity of Bacillus thuringiensis toxins against Simulium spp. larvae,” Journal of Invertebrate Pathology, vol. 121, pp. 70–73, Sep. 2014. [CrossRef]
- N. Crickmore, E. J. Bone, J. A. Williams, and D. J. Ellar, “Contribution of the individual components of the δ-endotoxin crystal to the mosquitocidal activity of Bacillus thuringiensis subsp. israelensis,” FEMS Microbiology Letters, vol. 131, no. 3, pp. 249–254, Sep. 1995. [CrossRef]
- D. Pinos, N. Joya, S. Herrero, J. Ferré, and P. Hernández-Martínez, “Hetero-oligomerization of Bacillus thuringiensis Cry1A proteins enhance binding to the ABCC2 transporter of Spodoptera exigua,” Biochemical Journal, vol. 478, no. 13, pp. 2589–2600, Jul. 2021. [CrossRef]
- C. Perez et al., “Bacillus thuringiensis subsp. israelensis Cyt1Aa synergizes Cry11Aa toxin by functioning as a membrane-bound receptor,” Proceedings of the National Academy of Sciences, vol. 102, no. 51, pp. 18303–18308, Dec. 2005. [CrossRef]
- P. Hernández-Martínez et al., “Comparison of in vitro and in vivo binding site competition of Bacillus thuringiensis Cry1 proteins in two important maize pests,” Pest Management Science, vol. 78, no. 4, pp. 1457–1466, Jan. 2022. [CrossRef]
- Luisa E. Fernandez, Karlygash G. Aimanova, Sarjeet S. Gill, A. Bravo, and M. Soberón, “A GPI-anchored alkaline phosphatase is a functional midgut receptor of Cry11Aa toxin in Aedes aegypti larvae,” Biochemical Journal, vol. 394, no. 1, pp. 77–84, Jan. 2006. [CrossRef]


| Insecticidal Cry Toxin | Insecticidal Cry toxin (excess by weight) | Cry41Aa concentration | Effect when combined with Cry41Aa |
| Cry1Ca | 10-180 fold | 2 µg/ml | Synergism |
| Cry11Aa | 10-90 fold | 2 µg/ml | Synergism |
| Cry4Aa | 10-180 fold | 2 µg/ml | Synergism |
| Cry1Ac | 10-180 fold | 2 µg/ml | No effect |
| Cry1Da | 10-180 fold | 2 µg/ml | No effect |
| Cry2Aa | 10-90 fold | 2 µg/ml | No effect |
| Cry2Ab | 10-90 fold | 2 µg/ml | No effect |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




