Submitted:
28 May 2024
Posted:
28 May 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cell Line, Reagents, and Chemicals
2.2. Cell Culture and Drug Treatment
2.3. Cell Viability Assay
2.4. Transwell Migration and Transwell Invasion Assay
2.5. Isobologram Analysis
2.6. Gap closure Assay
2.7. Western Blot Analysis
2.8. Antibodies
2.9. Gelatin Zymography Analysis
2.10. Effect of p-Akt Overexpression on the Parecoxib/5-FU Combination
2.11. Intracellular ROS Analysis
2.12. Statistical Analysis
3. Results
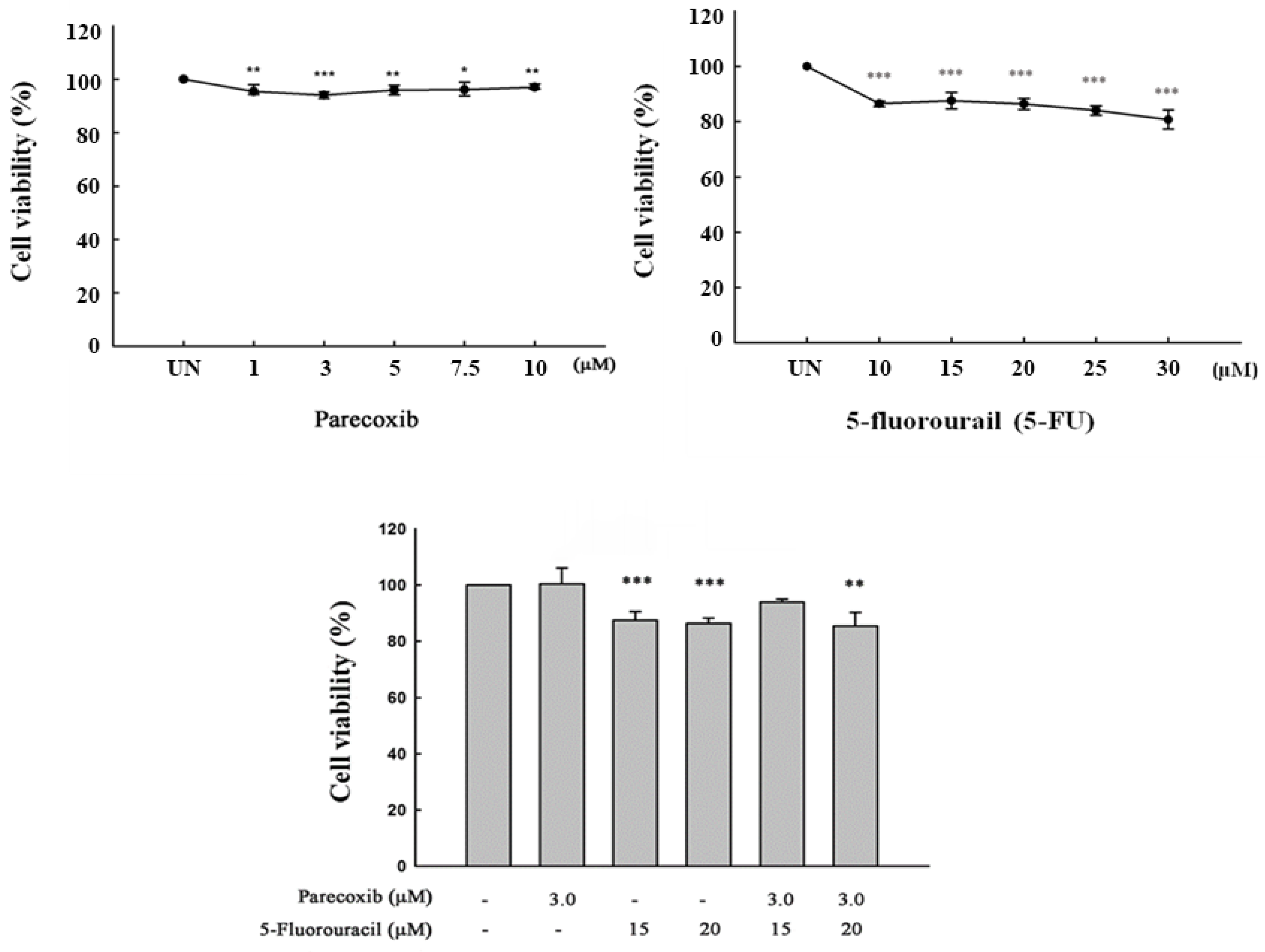
3.1. Cell Viability Assay of Parecoxib and 5-FU Treatment in DLD-1 Cells
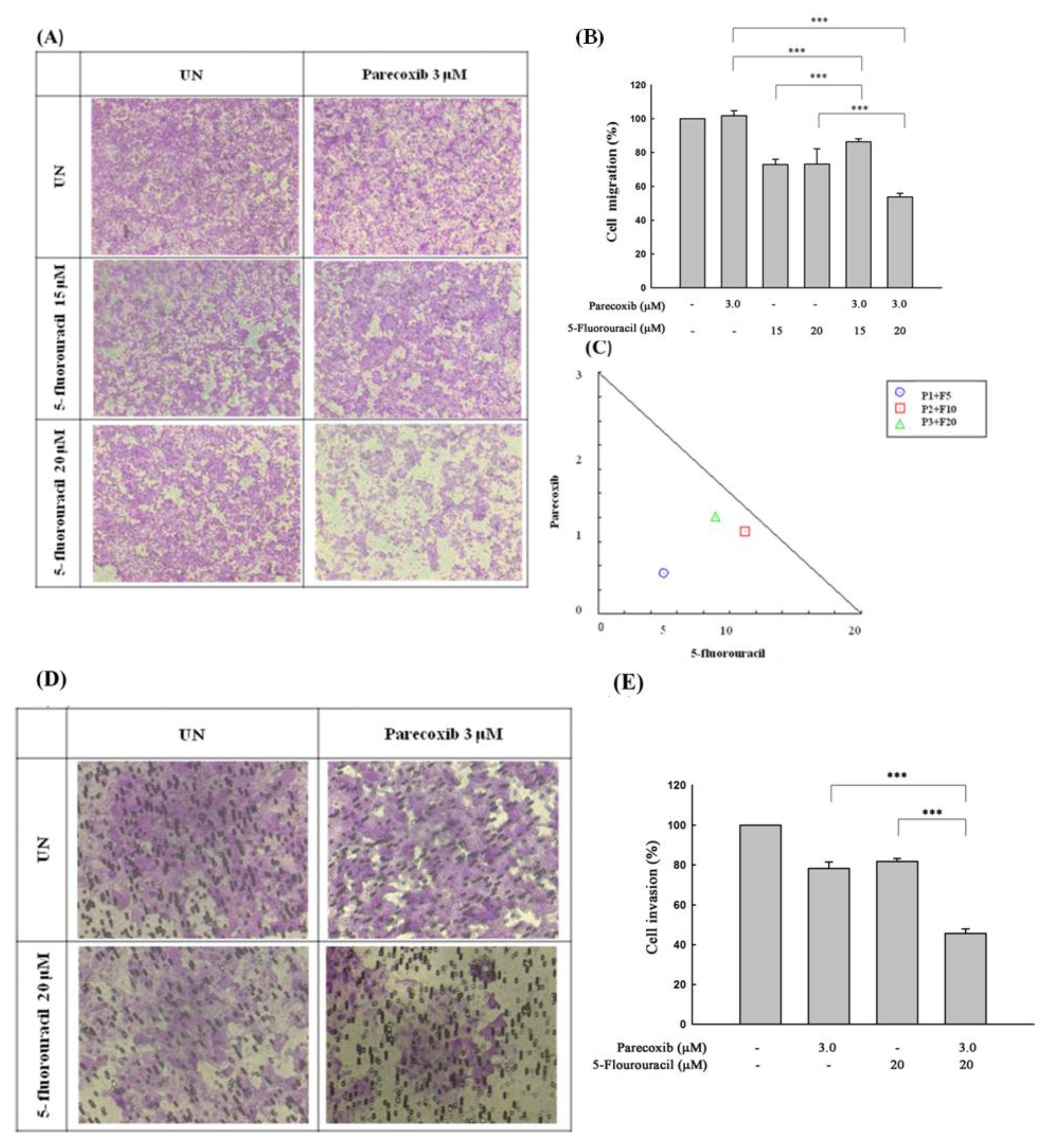
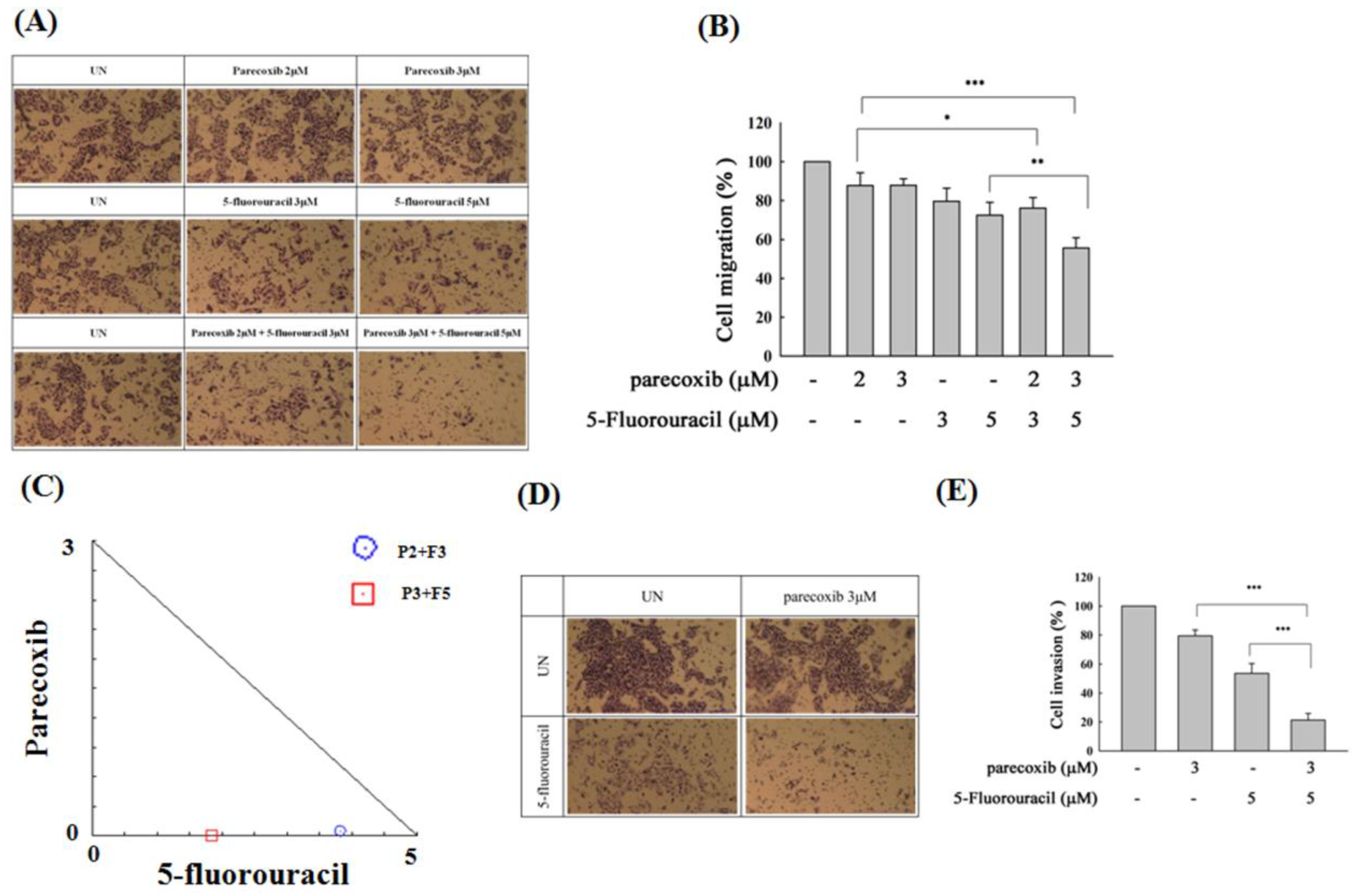
3.2. Synergism of Parecoxib and 5-FU Combination in Anti-Metastasis in DLD-1 Cells
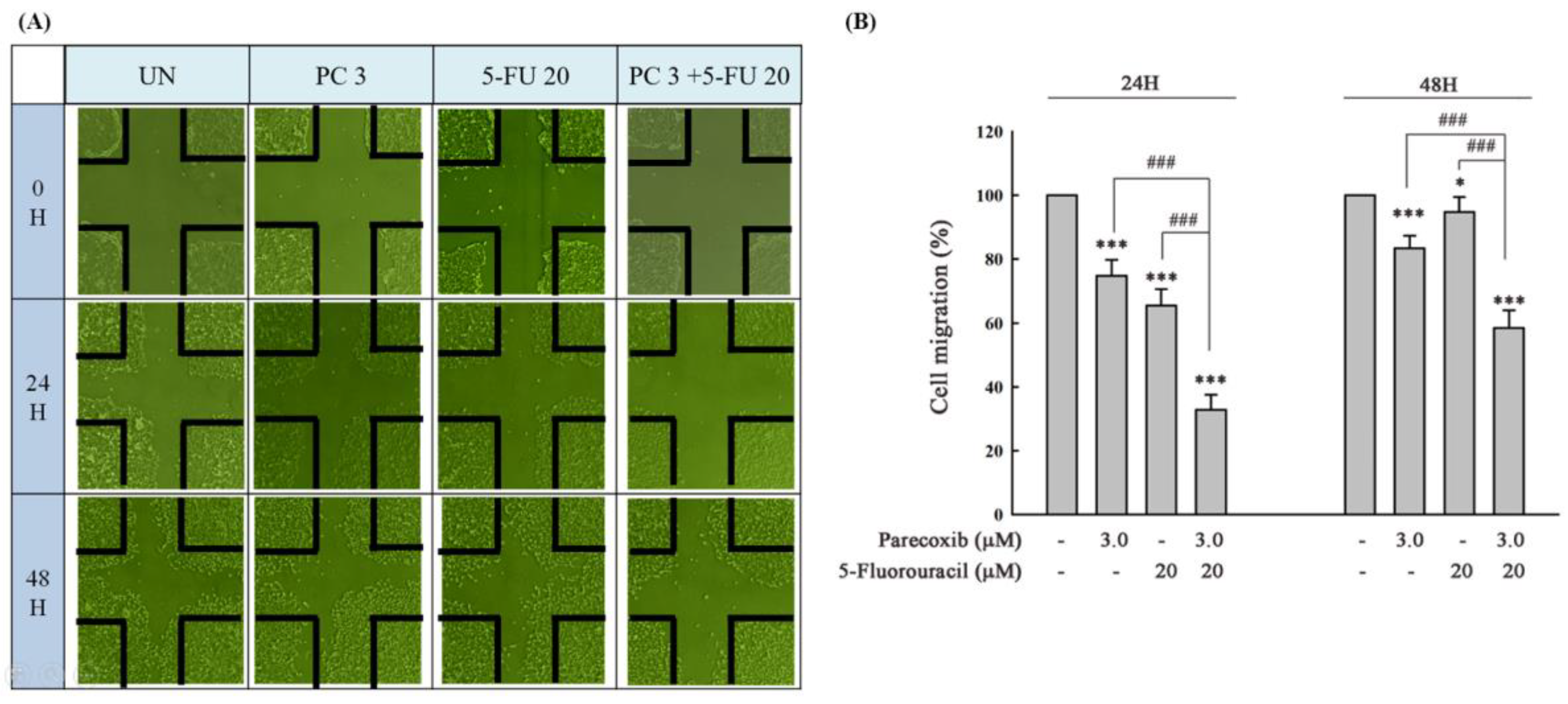
3.3. Parecoxib Enhances Gap Closure Inhibition of 5-FU Treatment in DLD-1 Cells
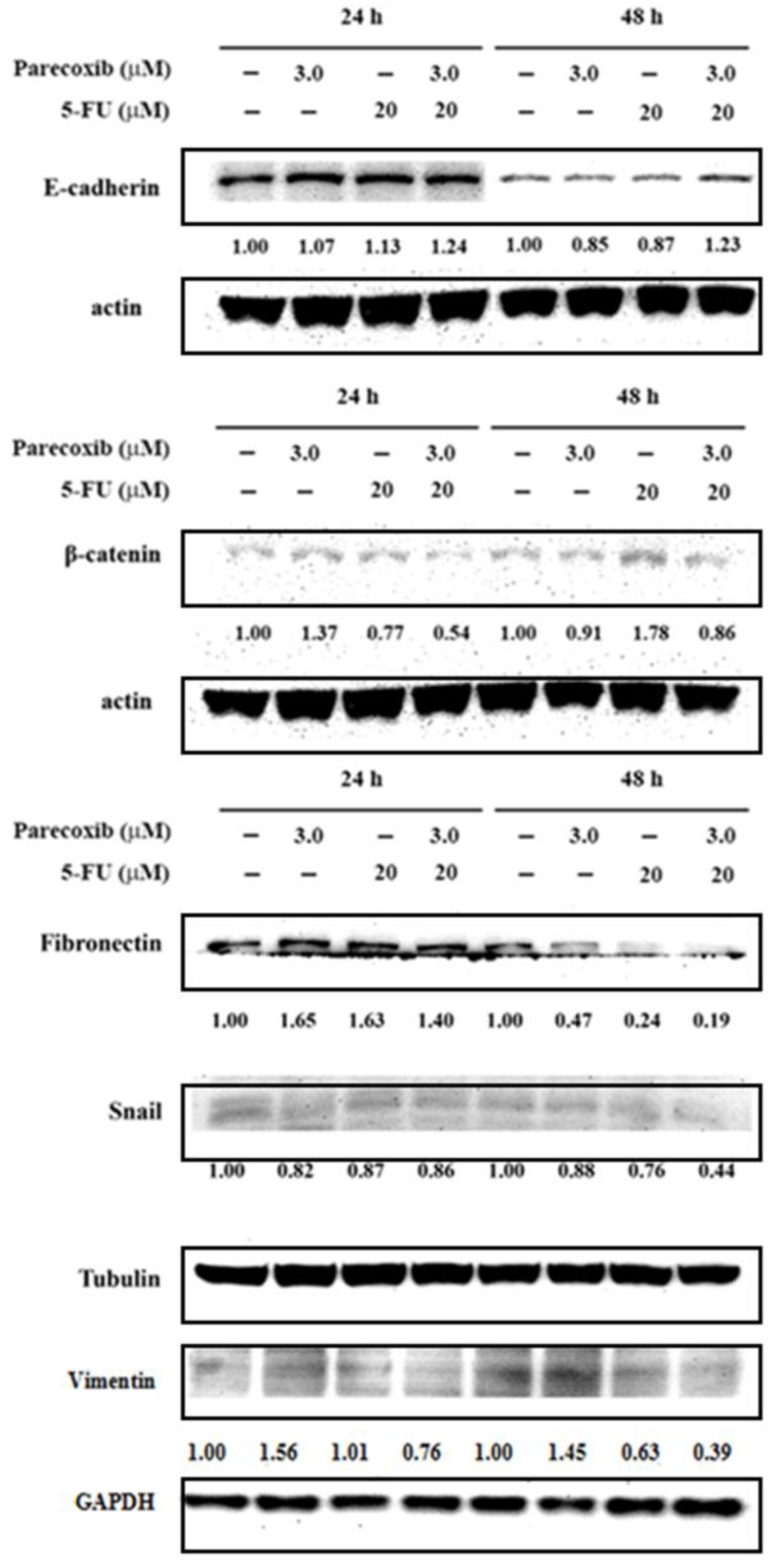
3.4. EMT on Parecoxib/5-FU Combination
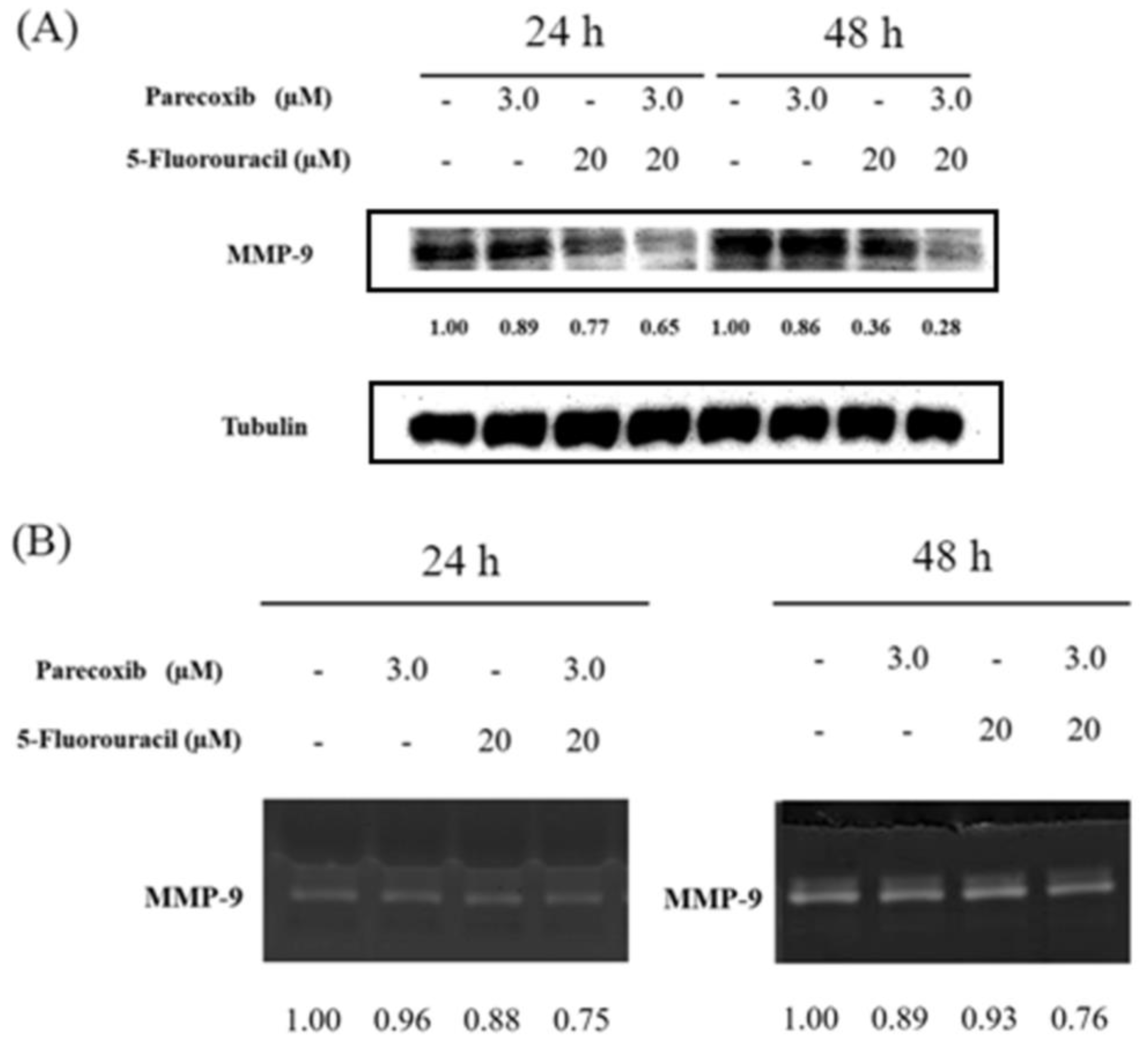
3.5. Parecoxib Enhances MMP-9 Inhibition of 5-FU Treatment
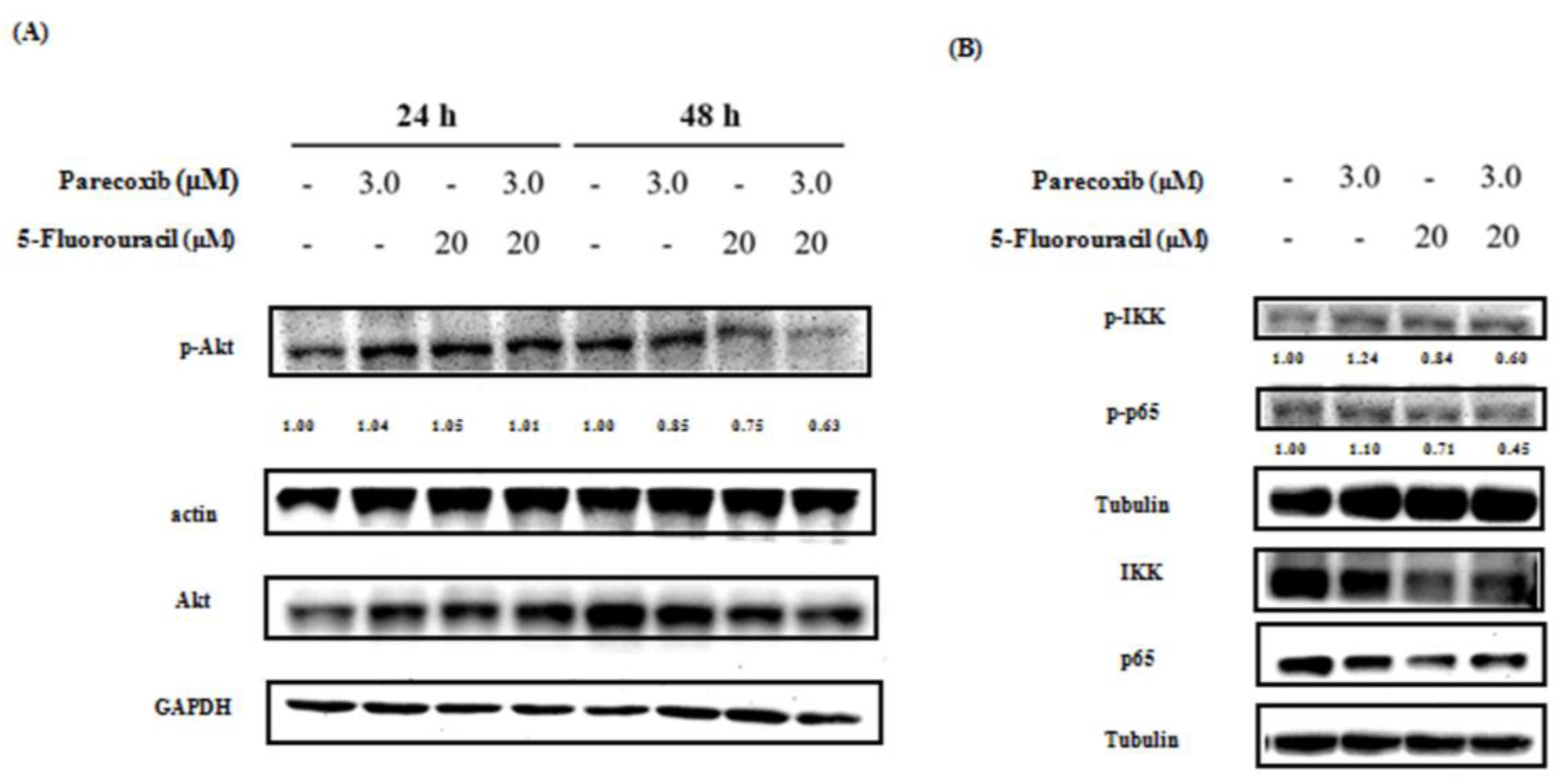
3.6. Parecoxib Enhances the PI3K/Akt/NF-κB Pathway in 5-FU Treatment
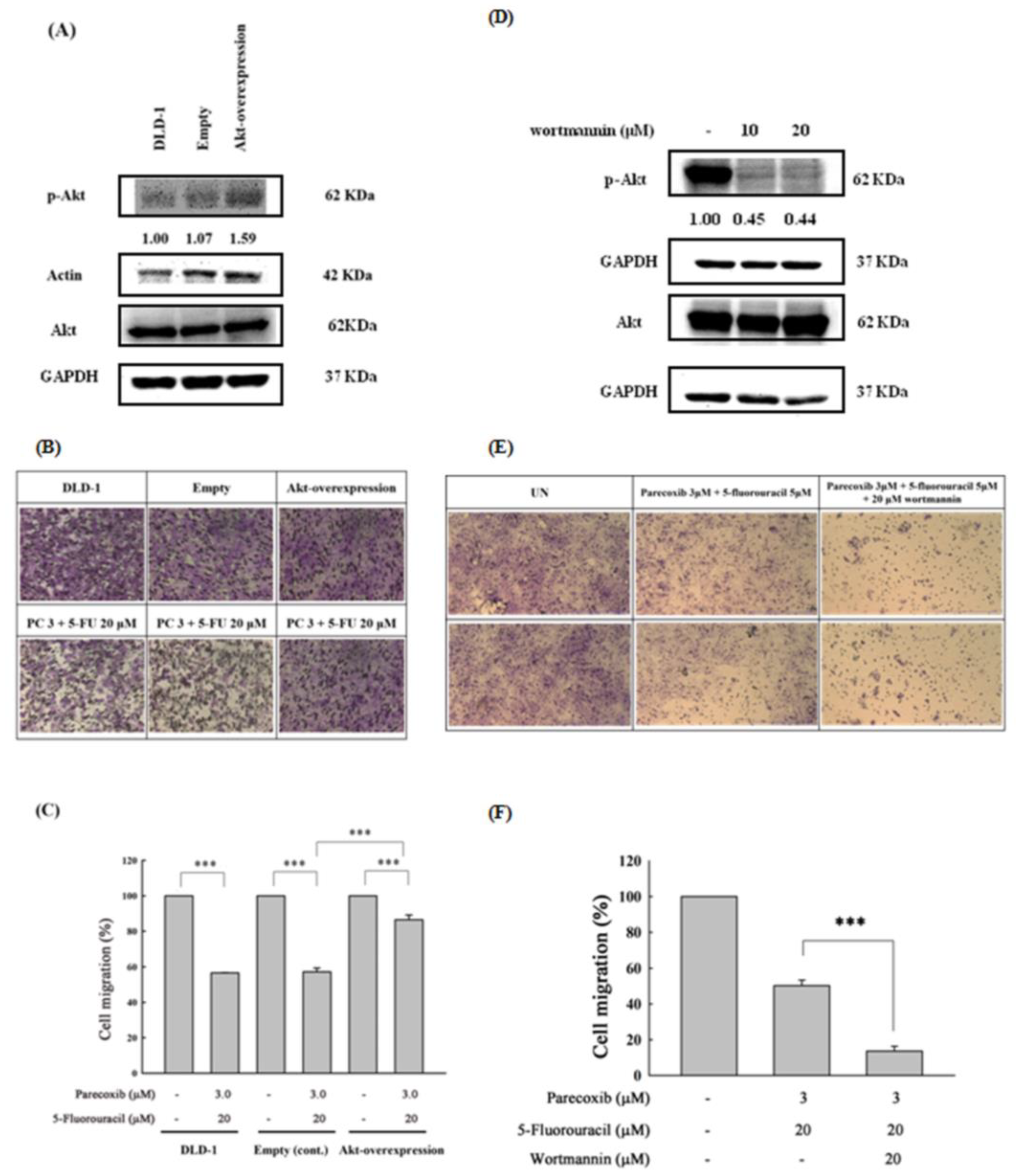
3.7. Akt Phosphorylation as the Critical Mediator by Which Parecoxib Can Potentiate the Anti-Migration Ability of 5-FU
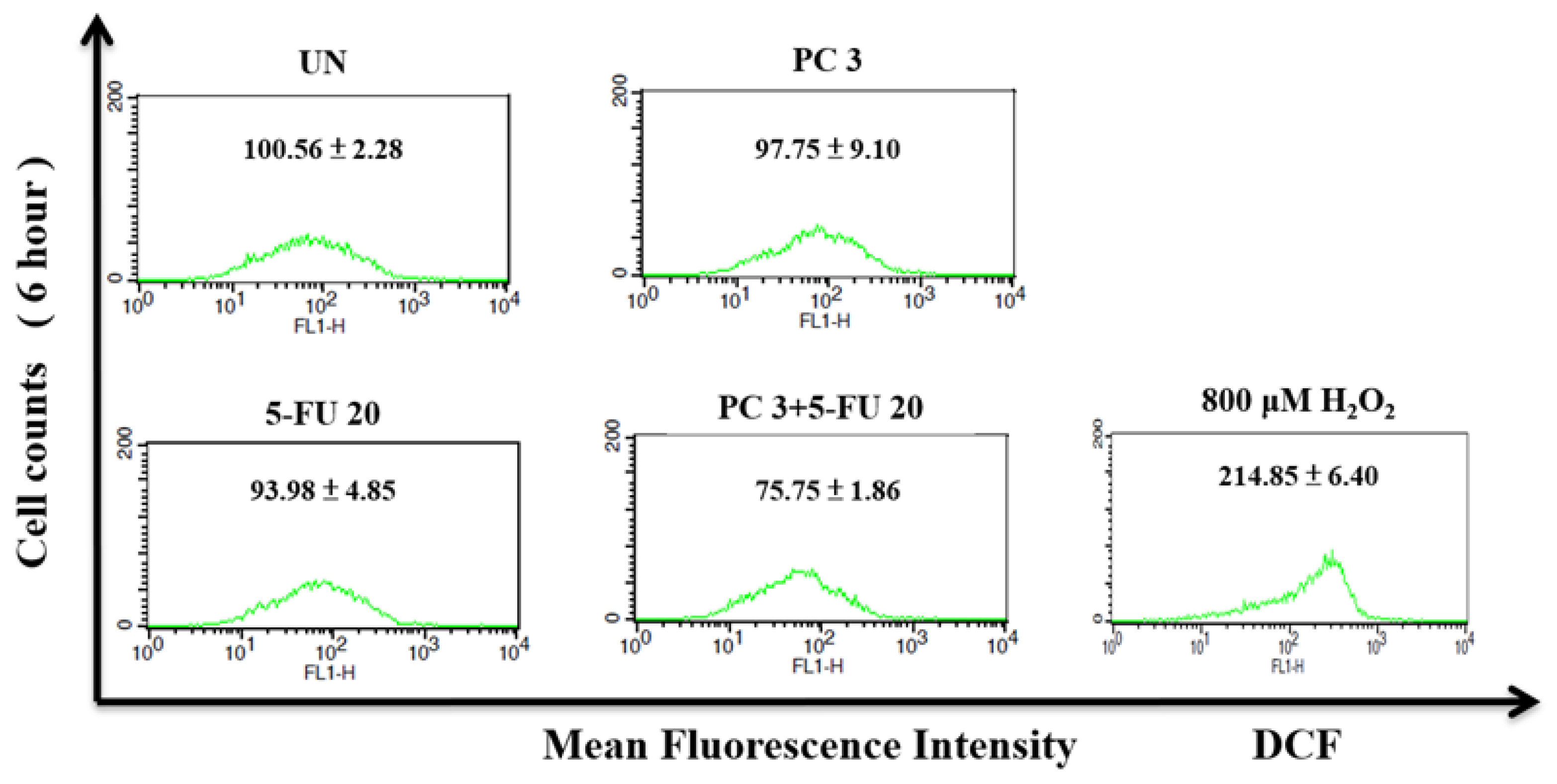
3.8. Parecoxib/5-FU Combination Inhibits Intracellular ROS
3.9. Synergism of Parecoxib and 5-FU Combination in Antimetastatic Effect in SW480 Cells
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Vogel, J.D.; Felder, S.I.; Bhama, A.R.; Hawkins, A.T.; Langenfeld, S.J.; Shaffer, V.O.; Thorsen, A.J.; Weiser, M.R.; Chang, G.J.; Lightner, A.L. The American Society of Colon and Rectal Surgeons clinical practice guidelines for the management of colon cancer. Diseases of the Colon & Rectum 2022, 65, 148–177. [Google Scholar] [CrossRef] [PubMed]
- Shinji, S.; Yamada, T.; Matsuda, A.; Sonoda, H.; Ohta, R.; Iwai, T.; Takeda, K.; Yonaga, K.; Masuda, Y.; Yoshida, H. Recent advances in the treatment of colorectal cancer: a review. Journal of Nippon Medical School 2022, 89, 246–254. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Guillén, L.; Arroyo, A. Immunonutrition in patients with colon cancer. 2020, 12, 5–8. [Google Scholar] [CrossRef] [PubMed]
- Ge, H.; Yao, Q. Traditional Chinese medicines as effective reversals of epithelial-mesenchymal transition induced-metastasis of colorectal cancer: Molecular targets and mechanisms. Frontiers in pharmacology 2022, 13, 842295. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, N.; Guo, H.; Yin, X.; Zheng, Y. Correlation Analysis of Gene and Radiomic Features in Colorectal Cancer Liver Metastases. Computational and Mathematical Methods in Medicine 2022, 2022. [Google Scholar] [CrossRef]
- Engstrand, J.; Nilsson, H.; Strömberg, C.; Jonas, E.; Freedman, J. Colorectal cancer liver metastases–a population-based study on incidence, management and survival. BMC cancer 2018, 18, 1–11. [Google Scholar] [CrossRef]
- Rong, D.; Sun, G.; Zheng, Z.; Liu, L.; Chen, X.; Wu, F.; Gu, Y.; Dai, Y.; Zhong, W.; Hao, X. MGP promotes CD8+ T cell exhaustion by activating the NF-κB pathway leading to liver metastasis of colorectal cancer. International Journal of Biological Sciences 2022, 18, 2345. [Google Scholar] [CrossRef] [PubMed]
- Nagashima, F.; Hamaguchi, T.; Furuse, J. Activity of the JCOG geriatric study committee and chemotherapy of colorectal cancer in older patients. Gan to Kagaku ryoho. Cancer & Chemotherapy 2015, 42, 16–20. [Google Scholar] [PubMed]
- Vodenkova, S.; Buchler, T.; Cervena, K.; Veskrnova, V.; Vodicka, P.; Vymetalkova, V. 5-fluorouracil and other fluoropyrimidines in colorectal cancer: Past, present and future. Pharmacology & therapeutics 2020, 206, 107447. [Google Scholar] [CrossRef]
- Cho, Y.-H.; Ro, E.J.; Yoon, J.-S.; Mizutani, T.; Kang, D.-W.; Park, J.-C.; Il Kim, T.; Clevers, H.; Choi, K.-Y. 5-FU promotes stemness of colorectal cancer via p53-mediated WNT/β-catenin pathway activation. Nature communications 2020, 11, 5321. [Google Scholar] [CrossRef]
- Glynne-Jones, R.; Wyrwicz, L.; Tiret, E.; Brown, G.; Rödel, C.d.; Cervantes, A.; Arnold, D. Rectal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of Oncology 2017, 28, iv22–iv40. [Google Scholar] [CrossRef] [PubMed]
- Rejhová, A.; Opattová, A.; Čumová, A.; Slíva, D.; Vodička, P. Natural compounds and combination therapy in colorectal cancer treatment. European journal of medicinal chemistry 2018, 144, 582–594. [Google Scholar] [CrossRef] [PubMed]
- Hammond, W.A.; Swaika, A.; Mody, K. Pharmacologic resistance in colorectal cancer: a review. Therapeutic advances in medical oncology 2016, 8, 57–84. [Google Scholar] [CrossRef] [PubMed]
- Dong, S.; Liang, S.; Cheng, Z.; Zhang, X.; Luo, L.; Li, L.; Zhang, W.; Li, S.; Xu, Q.; Zhong, M. ROS/PI3K/Akt and Wnt/β-catenin signalings activate HIF-1α-induced metabolic reprogramming to impart 5-fluorouracil resistance in colorectal cancer. Journal of Experimental & Clinical Cancer Research 2022, 41, 15. [Google Scholar] [CrossRef]
- Wang, W.; Wang, Q.; Wang, L.; Li, X.; Liu, D. Enhanced antitumor effect via combination of triptolide with 5-fluorouracil in pancreatic cancer. Translational Cancer Research 2018, 7. [Google Scholar] [CrossRef]
- Hao, Y.; Yin, G.; Wang, X.; Jiang, Z.; Zhang, G.; Feng, Z.; Sun, Q. Application of ICH Guidelines for the Assessment and Control of Elemental Impurities in Parecoxib Sodium by Graphite-Digestion and ICP-MS. International Journal of Analytical Chemistry 2022, 2022. [Google Scholar] [CrossRef] [PubMed]
- Xiong, W.; Li, W.-H.; Jiang, Y.-X.; Liu, S.; Ai, Y.-Q.; Liu, R.; Chang, L.; Zhang, M.; Wang, X.-L.; Bai, H. Parecoxib: an enhancer of radiation therapy for colorectal cancer. Asian Pacific Journal of Cancer Prevention 2015, 16, 627–633. [Google Scholar] [CrossRef]
- Huang, H.-M.; Huang, X.-Y.; Wu, S.-P.; Chen, C.-K.; He, X.-H.; Zhang, Y.-F. RETRACTED ARTICLE: Parecoxib inhibits esophageal squamous cell carcinoma progression via the PDK1–AKT pathway. Cellular & molecular biology letters 2022, 27, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Wong, C.H.; Chang, W.L.; Lu, F.J.; Liu, Y.W.; Peng, J.Y.; Chen, C.H. Parecoxib expresses anti-metastasis effect through inhibition of epithelial-mesenchymal transition and the Wnt/β-catenin signaling pathway in human colon cancer DLD-1 cell line. Environmental Toxicology 2022, 37, 2718–2727. [Google Scholar] [CrossRef]
- Aggarwal, V.; Tuli, H.S.; Varol, A.; Thakral, F.; Yerer, M.B.; Sak, K.; Varol, M.; Jain, A.; Khan, M.A.; Sethi, G. Role of reactive oxygen species in cancer progression: molecular mechanisms and recent advancements. Biomolecules 2019, 9, 735. [Google Scholar] [CrossRef]
- Pan, C.; Zhang, Y.; Meng, Q.; Dai, G.; Jiang, Z.; Bao, H. Down regulation of the expression of ELMO3 by COX2 inhibitor suppresses tumor growth and metastasis in non-small-cell lung cancer. Frontiers in Oncology 2019, 9, 363. [Google Scholar] [CrossRef] [PubMed]
- Güllü, N.; Smith, J.; Herrmann, P.; Stein, U. MACC1-dependent antitumor effect of curcumin in colorectal cancer. Nutrients 2022, 14, 4792. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Wang, W.; Cai, H.; Du, B.; Zhang, L.; Ma, W.; Hu, Y.; Feng, S.; Miao, G. MACC1 facilitates chemoresistance and cancer stem cell-like properties of colon cancer cells through the PI3K/AKT signaling pathway. Molecular Medicine Reports 2017, 16, 8747–8754. [Google Scholar] [CrossRef] [PubMed]
- Treese, C.; Werchan, J.; von Winterfeld, M.; Berg, E.; Hummel, M.; Timm, L.; Rau, B.; Daberkow, O.; Walther, W.; Daum, S. Inhibition of MACC1-induced metastasis in esophageal and gastric adenocarcinomas. Cancers 2022, 14, 1773. [Google Scholar] [CrossRef] [PubMed]
- Kortüm, B.; Radhakrishnan, H.; Zincke, F.; Sachse, C.; Burock, S.; Keilholz, U.; Dahlmann, M.; Walther, W.; Dittmar, G.; Kobelt, D. Combinatorial treatment with statins and niclosamide prevents CRC dissemination by unhinging the MACC1-β-catenin-S100A4 axis of metastasis. Oncogene 2022, 41, 4446–4458. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.; Chen, H.; Liang, J.; Li, Y.; Yang, J.; Luo, C.; Tang, Y.; Ding, Y.; Liu, X.; Yuan, Q. Dual role of reactive oxygen species and their application in cancer therapy. Journal of Cancer 2021, 12, 5543. [Google Scholar] [CrossRef]
- Nakamura, H.; Takada, K. Reactive oxygen species in cancer: Current findings and future directions. Cancer science 2021, 112, 3945–3952. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, H.; Takada, K. Reactive oxygen species in cancer: Current findings and future directions. Cancer science 2021, 112, 3945–3952. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, R.; Mukherjee, S.; Biswas, J.; Roy, M. Phenethyl isothiocyanate, by virtue of its antioxidant activity, inhibits invasiveness and metastatic potential of breast cancer cells: HIF-1α as a putative target. Free Radical Research 2016, 50, 84–100. [Google Scholar] [CrossRef]
- Zeng, J.; Xu, H.; Fan, P.z.; Xie, J.; He, J.; Yu, J.; Gu, X.; Zhang, C.j. Kaempferol blocks neutrophil extracellular traps formation and reduces tumour metastasis by inhibiting ROS-PAD4 pathway. Journal of Cellular and Molecular Medicine 2020, 24, 7590–7599. [Google Scholar] [CrossRef]
- Yang, J.-T.; Lee, I.; Lu, F.-J.; Chung, C.-Y.; Lee, M.-H.; Cheng, Y.-C.; Chen, K.-T.; Chen, C.-H. Propyl gallate exerts an antimigration effect on temozolomide-treated malignant glioma cells through inhibition of ROS and the NF-κB pathway. Journal of immunology research 2017, 2017. [Google Scholar] [CrossRef] [PubMed]
- Capeloa, T.; Van de Velde, J.A.; d’Hose, D.; Lipari, S.G.; Derouane, F.; Hamelin, L.; Bedin, M.; Vazeille, T.; Duhoux, F.P.; Murphy, M.P. Inhibition of Mitochondrial Redox Signaling with MitoQ Prevents Metastasis of Human Pancreatic Cancer in Mice. Cancers 2022, 14, 4918. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.-H.; Choi, S.-Y.; Choi, H.-J.; Ryu, H.-M.; Kim, Y.-J.; Jung, H.-Y.; Cho, J.-H.; Kim, C.-D.; Park, S.-H.; Kwon, T.-H. The emerging role of xanthine oxidase inhibition for suppression of breast cancer cell migration and metastasis associated with hypercholesterolemia. The FASEB Journal 2019, 33, 7301–7314. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Zhang, M.; Xu, L.; Zhang, X.; Zhao, Y. Gypensapogenin H suppresses tumor growth and cell migration in triple-negative breast cancer by regulating PI3K/AKT/NF-κB/MMP-9 signaling pathway. Bioorganic Chemistry 2022, 126, 105913. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Cheng, T.; Zhao, Y.; Qu, Y. HMGA1 exacerbates tumor progression by activating miR-222 through PI3K/Akt/MMP-9 signaling pathway in uveal melanoma. Cellular signalling 2019, 63, 109386. [Google Scholar] [CrossRef] [PubMed]
- Bolignano, D.; Donato, V.; Lacquaniti, A.; Fazio, M.R.; Bono, C.; Coppolino, G.; Buemi, M. Neutrophil gelatinase-associated lipocalin (NGAL) in human neoplasias: a new protein enters the scene. Cancer letters 2010, 288, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Jacob, A.; Jing, J.; Lee, J.; Schedin, P.; Gilbert, S.M.; Peden, A.A.; Junutula, J.R.; Prekeris, R. Rab40b regulates trafficking of MMP2 and MMP9 during invadopodia formation and invasion of breast cancer cells. Journal of cell science 2013, 126, 4647–4658. [Google Scholar] [CrossRef] [PubMed]
- Hung, C.-Y.; Lee, C.-H.; Chiou, H.-L.; Lin, C.-L.; Chen, P.-N.; Lin, M.-T.; Hsieh, Y.-H.; Chou, M.-C. Praeruptorin-B inhibits 12-O-tetradecanoylphorbol-13-acetate-induced cell invasion by targeting AKT/NF-κB via matrix metalloproteinase-2/-9 expression in human cervical cancer cells. Cell Physiol Biochem 2019, 52, 1255–1266. [Google Scholar] [CrossRef]
- Huang, Y.; Hong, W.; Wei, X. The molecular mechanisms and therapeutic strategies of EMT in tumor progression and metastasis. Journal of hematology & oncology 2022, 15, 129. [Google Scholar] [CrossRef]
- Fan, X.; Tong, Y.; Chen, Y.; Chen, Y. Sunitinib Reduced the Migration of Ectopic Endometrial Cells via p-VEGFR-PI3K-AKT-YBX1-Snail Signaling Pathway. Analytical Cellular Pathology 2022, 2022. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



