Submitted:
22 May 2024
Posted:
24 May 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
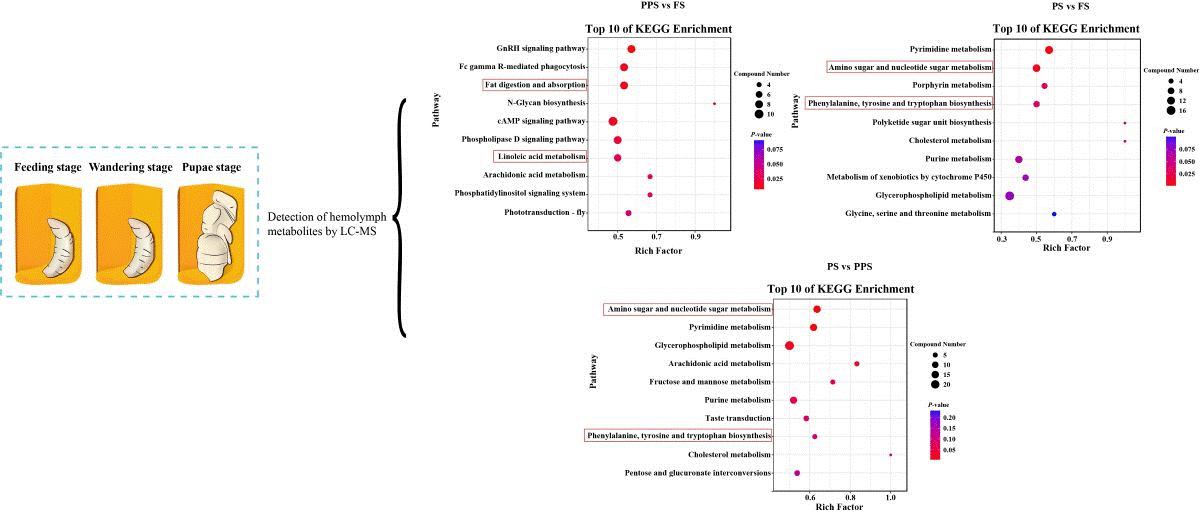
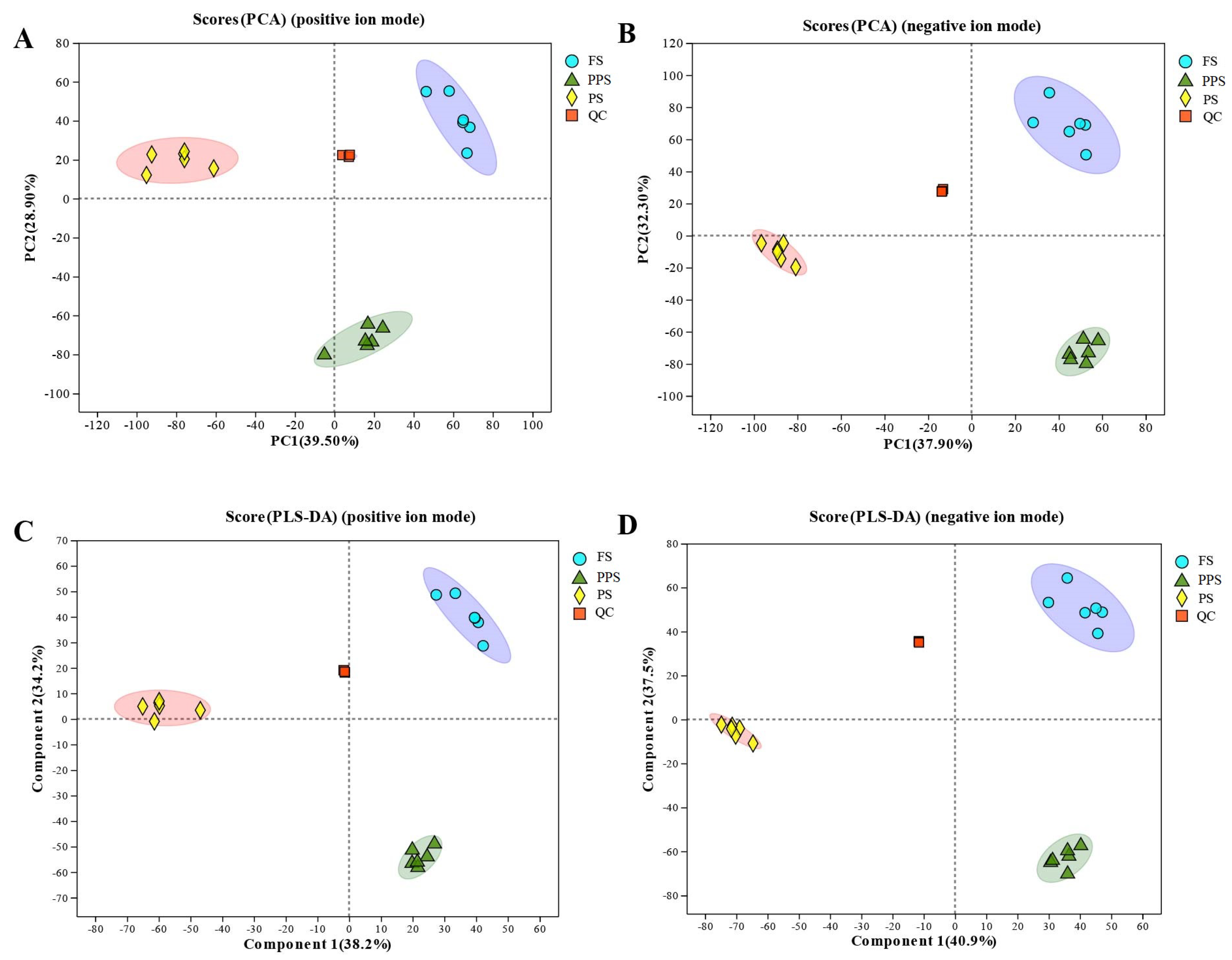
2.1. PCA, PLS-DA, and Ingredient of Metabolomics in Hemolymph of Apis mellifera Larvae
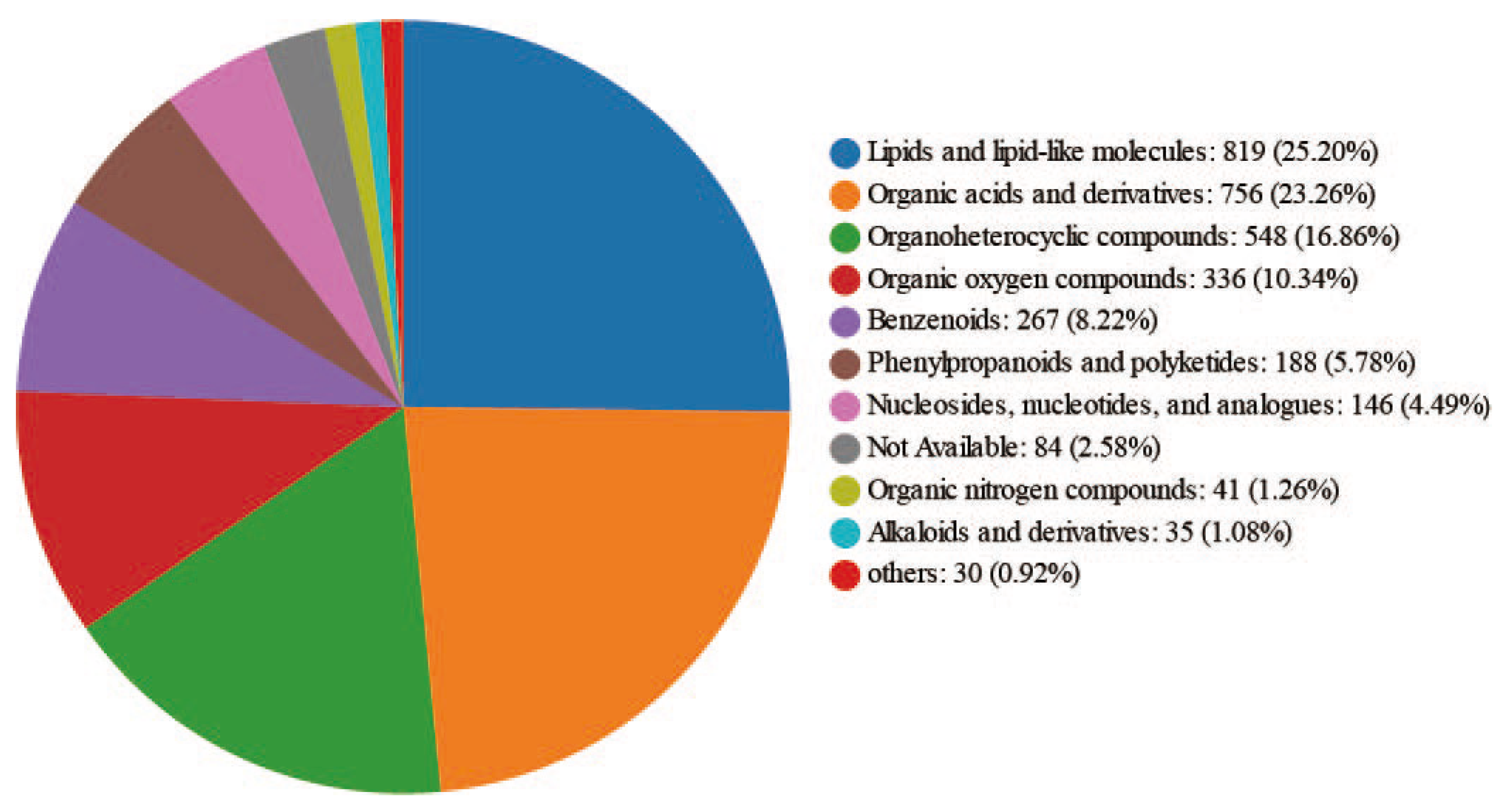
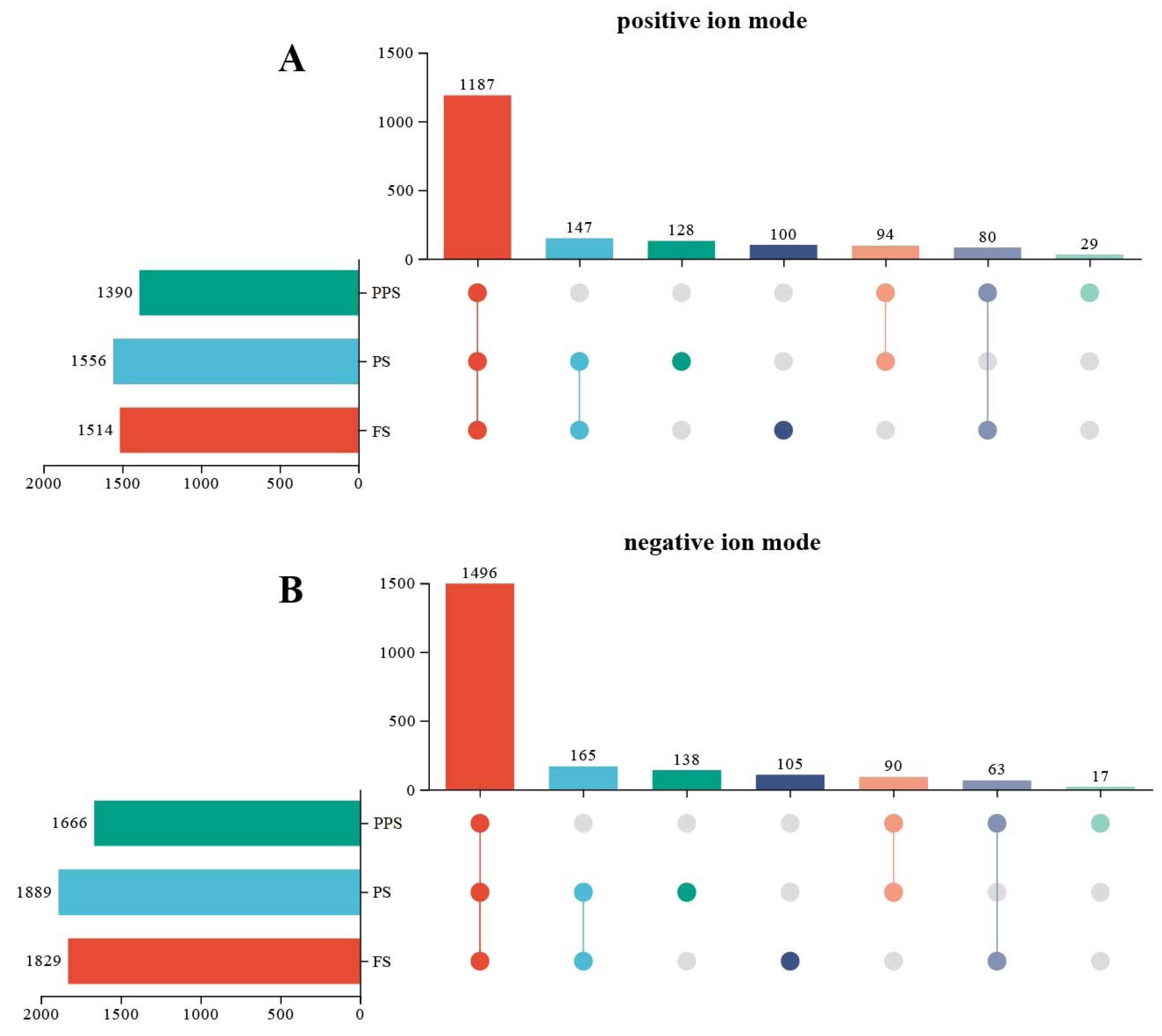
2.2. Metabolite Identification of the Hemolymph at Different Physiological Stages of Apis mellifera
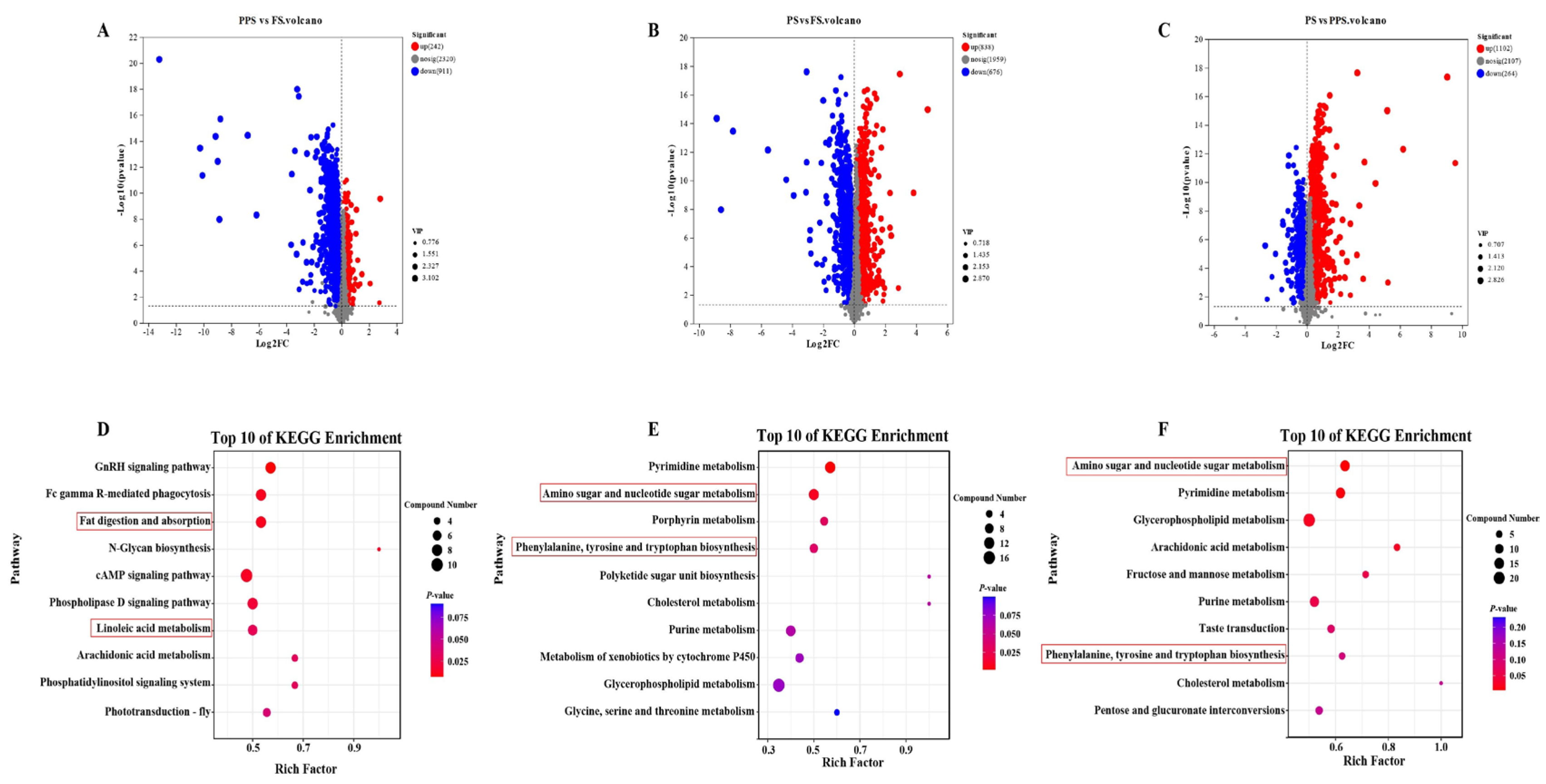
2.3. Differential Metabolite Analysis of Hemolymph at Different Physiological Stages of Apis mellifera
3. Discussion
4. Materials and Methods
4.1. Insect Rearing
4.2. Chemicals and Reagents
4.3. Collection of Hemolymph from Larvae and Pupae
4.4. Pre-Treatment of Hemolymph Samples
4.5. LC-MS Method
4.6. Quality Control
4.7. Identification of Metabolites in Hemolymph
4.8. Data Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang, Y.; Ma, L.-T.; Xu, B.-H. , Diversity in life history of queen and worker honey bees, Apis mellifera L. J. Asia Pac. Entomol 2015, 18, 145–149. [Google Scholar] [CrossRef]
- Vázquez, D. E.; Farina, W. M. , Locomotion and searching behaviour in the honey bee larva depend on nursing interaction. Apidologie 2021, 52, 1368–1386. [Google Scholar] [CrossRef]
- Barie, K.; Amsalem, E. , The influence of the social environment on larval development and resulting caste in Bombus impatiens. Anim Behav. 2023, 200, 49–57. [Google Scholar] [CrossRef]
- da Cruz-Landim, C.; Patrício, K.; Antonialli Jr, W. F. , Cell death and ovarian development in highly eusocial bees (Hymenoptera, Apidae): Caste differentiation and worker egg laying. J Morphol Sci. 2017, 23, 27–42. [Google Scholar]
- Noël, A.; Dumas, C.; Rottier, E.; Beslay, D.; Costagliola, G.; Ginies, C.; Nicolè, F.; Rau, A.; Le Conte, Y.; Mondet, F. , Detailed chemical analysis of honey bee (Apis mellifera) worker brood volatile profile from egg to emergence. Plos one 2023, 18, e0282120. [Google Scholar] [CrossRef]
- Vásquez-Ibarra, L.; Iriarte, A.; Rebolledo-Leiva, R.; González-Araya, M. C.; Angulo-Meza, L. , Determining the key factors that contribute to the eco-efficiency level of honey production using life cycle approaches. INT J Life Cycle Ass. 2023, 1–11. [Google Scholar] [CrossRef]
- Zariman, N. A.; Omar, N. A.; Huda, A. N. , Plant Attractants and Rewards for Pollinators: Their Significant to Successful Crop Pollination. I J Life Sciand Biotech. 2022, 5, 270–293. [Google Scholar] [CrossRef]
- VanEngelsdorp, D.; Evans, J. D.; Saegerman, C.; Mullin, C.; Haubruge, E.; Nguyen, B. K.; Frazier, M.; Frazier, J.; Cox-Foster, D.; Chen, Y. , Colony collapse disorder: a descriptive study. PloS one 2009, 4, e6481. [Google Scholar] [CrossRef]
- Malita, A.; Rewitz, K. , Interorgan communication in the control of metamorphosis. Curr Opin Insect Sci. 2021, 43, 54–62. [Google Scholar] [CrossRef]
- Schooley, D. A.; Horodyski, F. M.; Coast, G. M. , Hormones controlling homeostasis in insects. In Insect endocrinology, Elsevier: 2012; pp 366-429.
- Gäde, G. , Regulation of intermediary metabolism and water balance of insects by neuropeptides. Annu Rev Entomol. 2004, 49, 93–113. [Google Scholar] [CrossRef]
- Wang, Y.; Li-Byarlay, H. , Physiological and molecular mechanisms of nutrition in honey bees. In Advances in Insect Physiology, Elsevier: 2015; Vol. 49, pp 25-58.
- Liberti, J.; Kay, T.; Quinn, A.; Kesner, L.; Frank, E. T.; Cabirol, A.; Richardson, T. O.; Engel, P.; Keller, L. , The gut microbiota affects the social network of honeybees. NAT Ecol Evol. 2022, 6, 1471–1479. [Google Scholar] [CrossRef] [PubMed]
- Pasquier, C.; Robichon, A. , Temporal and sequential order of nonoverlapping gene networks unraveled in mated female Drosophila. Life Sci Alliance. 2022, 5. [Google Scholar] [CrossRef] [PubMed]
- Gillette, C. M.; Tennessen, J. M.; Reis, T. , Balancing energy expenditure and storage with growth and biosynthesis during Drosophila development. Dev Biol. 2021, 475, 234–244. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A. C.; Rebelo, A. R.; Homem, C. C. , Integrating animal development: how hormones and metabolism regulate developmental transitions and brain formation. Dev Biol. 2021, 475, 256–264. [Google Scholar] [CrossRef] [PubMed]
- Tellis, M. B.; Kotkar, H. M.; Joshi, R. S. , Regulation of trehalose metabolism in insects: from genes to the metabolite window. Glycobiology 2023, 33, 262–273. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Gu, F.; Liu, Z.; Li, Z.; Wu, F. a.; Sheng, S. , The key role of fatty acid synthase in lipid metabolism and metamorphic development in a destructive insect pest, Spodoptera litura (Lepidoptera: Noctuidae). INT J Mol Sci 2022, 23, 9064. [Google Scholar] [CrossRef] [PubMed]
- Kaczmarek, A.; Boguś, M. , The metabolism and role of free fatty acids in key physiological processes in insects of medical, veterinary and forensic importance. PeerJ 2021, 9, e12563. [Google Scholar] [CrossRef] [PubMed]
- Pei, D. , How do biomolecules cross the cell membrane? Acc. Chem. Res. 2022, 55, 309–318. [Google Scholar] [CrossRef]
- Yan, B.; Fung, K.; Ye, S.; Lai, P.-M.; Wei, Y. X.; Sze, K.-H.; Yang, D.; Gao, P.; Kao, R. Y.-T. , Linoleic acid metabolism activation in macrophages promotes the clearing of intracellular Staphylococcus aureus. Chem Sci. 2022, 13, 12445–12460. [Google Scholar] [CrossRef]
- Unuofin, J. O.; Lebelo, S. L. , Antioxidant effects and mechanisms of medicinal plants and their bioactive compounds for the prevention and treatment of type 2 diabetes: an updated review. OXID Med Cell Longev. 2020, 2020. [Google Scholar] [CrossRef]
- Standifer, L. , Honey bee nutrition supplemental feeding. Beekeeping in the United States Agriculture Handbook 2003, 335, 39–45. [Google Scholar]
- Wang, X.; Qi, Y.; Zheng, H. , Dietary polyphenol, gut microbiota, and health benefits. Antioxidants 2022, 11, 1212. [Google Scholar] [CrossRef] [PubMed]
- Boucias, D. G.; Pendland, J. C. , Principles of insect pathology. Springer Science & Business Media: 2012.
- Yu, L.; Zhao, J.; Wang, W.; Zong, L.; Ge, S.; Yan, S. , Structural stabilization of honeybee wings based on heterogeneous stiffness. Soft Matter 2023, 19, 841–850. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Wang, H.; Chen, W.; Song, H.; Wang, Y.; Liu, Z.; Xu, B. , 20-Hydroxyecdysone and receptor interplay in the regulation of hemolymph glucose level in honeybee (Apis mellifera) larvae. Metabolites 2023, 13, 80. [Google Scholar] [CrossRef] [PubMed]
- Shi, J. F.; Fu, J.; Mu, L. L.; Guo, W. C.; Li, G. Q. , Two Leptinotarsa uridine diphosphate N-acetylglucosamine pyrophosphorylases are specialized for chitin synthesis in larval epidermal cuticle and midgut peritrophic matrix. Insect Biochem Molec 2016, 68, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Whitten, M. M.; Coates, C. J. , Re-evaluation of insect melanogenesis research: Views from the dark side. Pigm Cell Melanoma R. 2017, 30, 386–401. [Google Scholar] [CrossRef] [PubMed]
- Figon, F.; Casas, J. , Ommochromes in invertebrates: biochemistry and cell biology. Biological Reviews 2019, 94, 156–183. [Google Scholar] [CrossRef]
- Shamim, G.; RANjAN, S. K.; PANdEy, D. M.; RAmANI, R. , Biochemistry and biosynthesis of insect pigments. EJE 2014, 111, 149–164. [Google Scholar] [CrossRef]
| Ion mode | All peaks | Identified metabolites | Metabolites in library |
|---|---|---|---|
| pos | 11 896 | 1 765 | 1 620 |
| neg | 11 951 | 2 074 | 2 041 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




