Submitted:
22 May 2024
Posted:
23 May 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Material and Methods
2.1. Chemistry
2.2. Biology
2.2.1. Experimental Animals
2.2.2. Acute Toxicty in Mice
2.2.3. Sub-Acute Toxicity in Mice
2.2.4. Experimental Design
2.2.5. Histological Evaluation of Tissue Specimens
2.3. Assessment of the Oxidative Stress Biomarkers
2.4. InhA Inhibition Assay
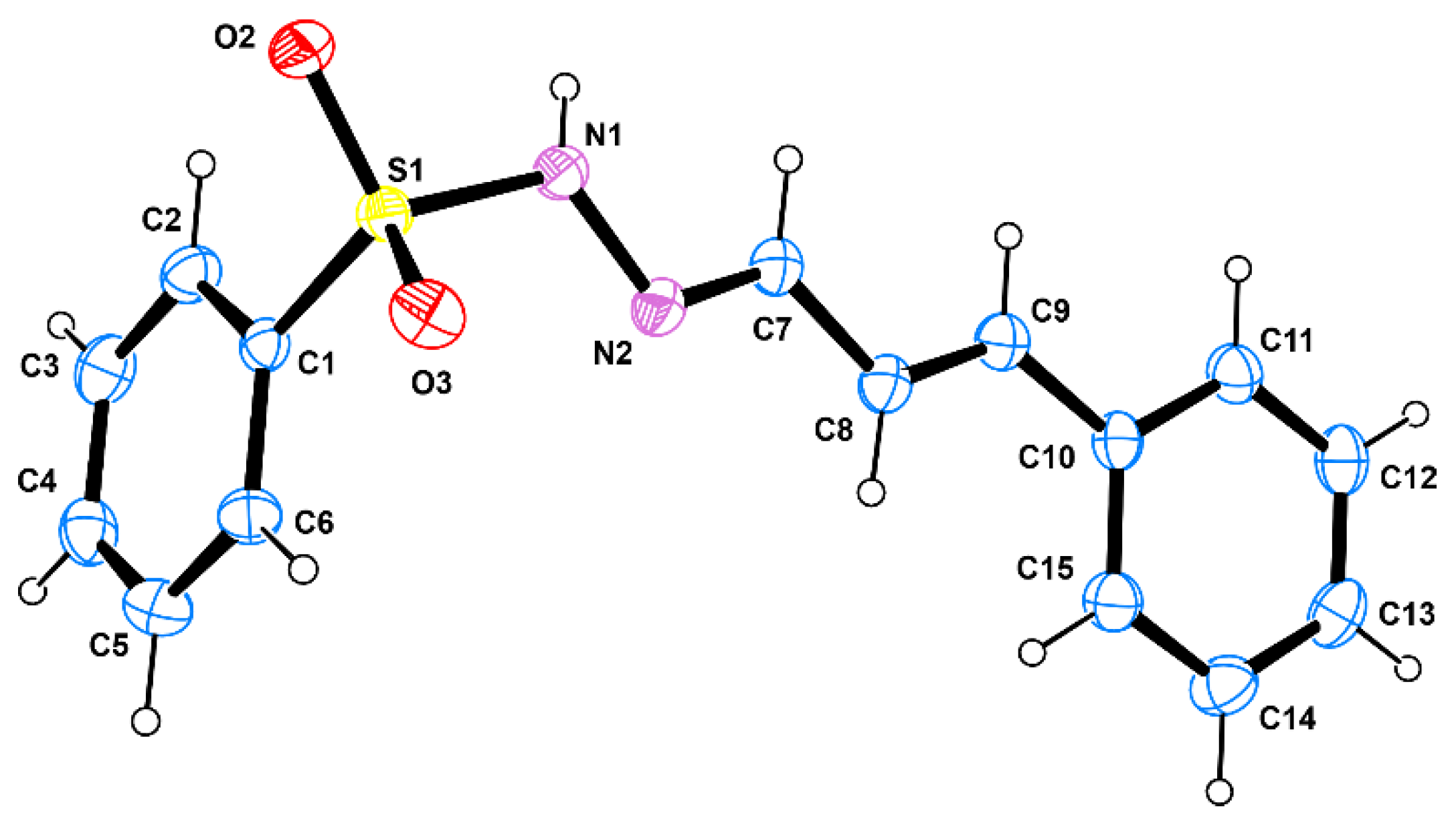
2.5. Single-Crystal X-ray Analysis
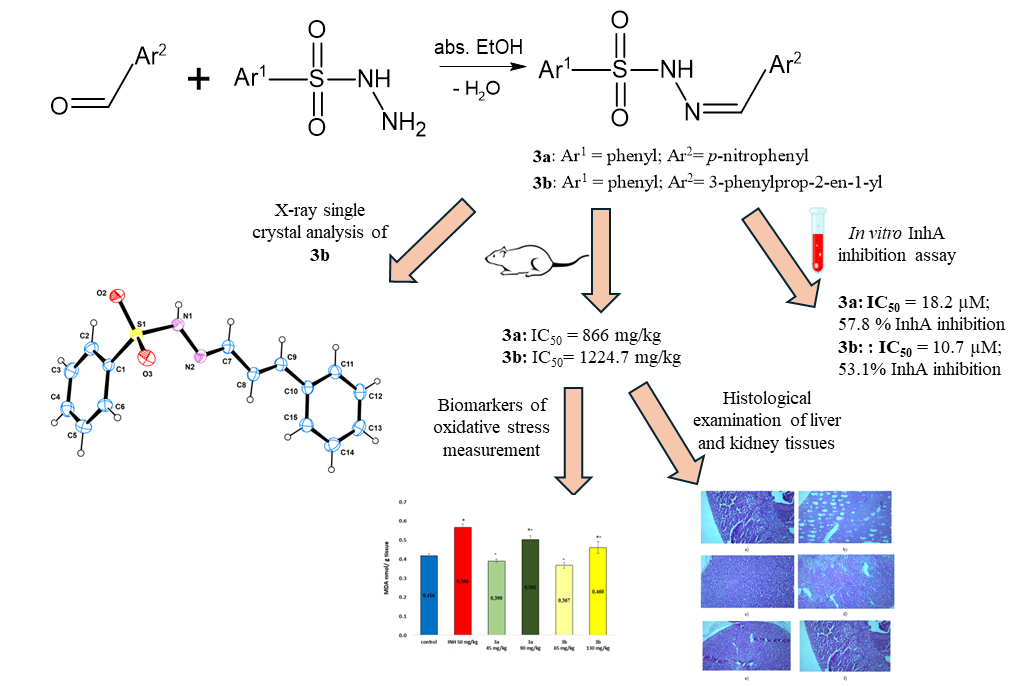
3. Results and Discussion
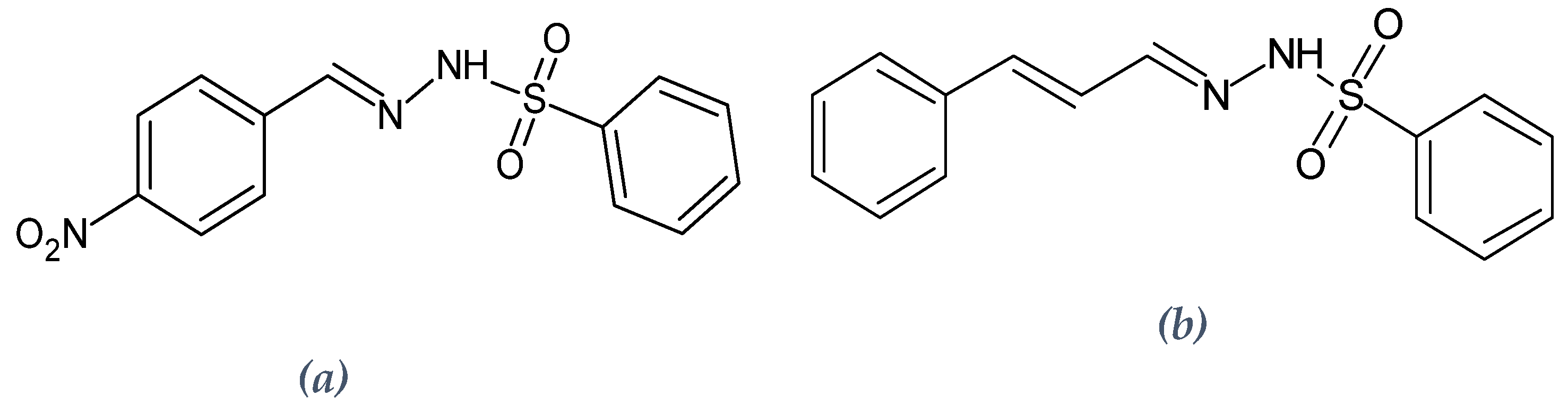
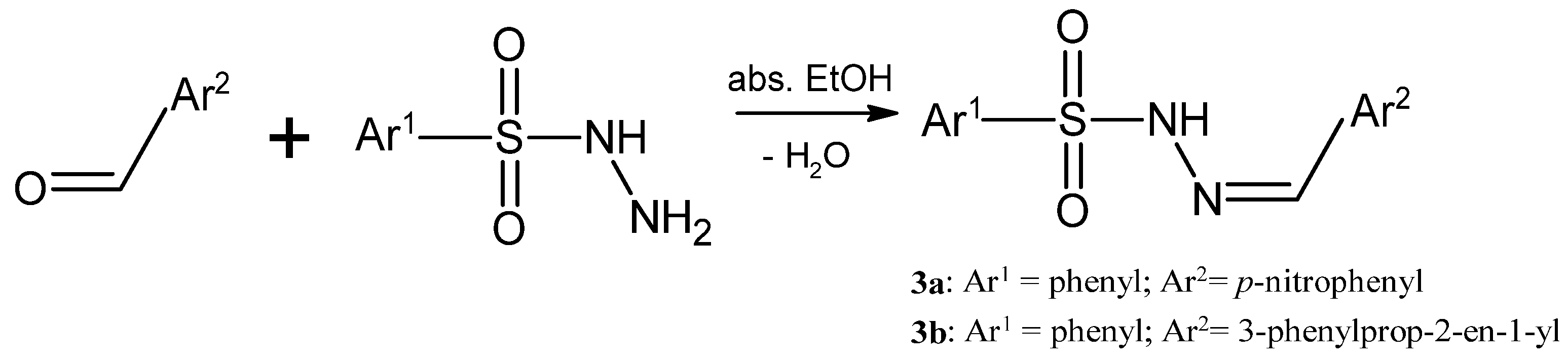
3.1. Synthesis of the Sulfonyl Hydrazone Derivatives
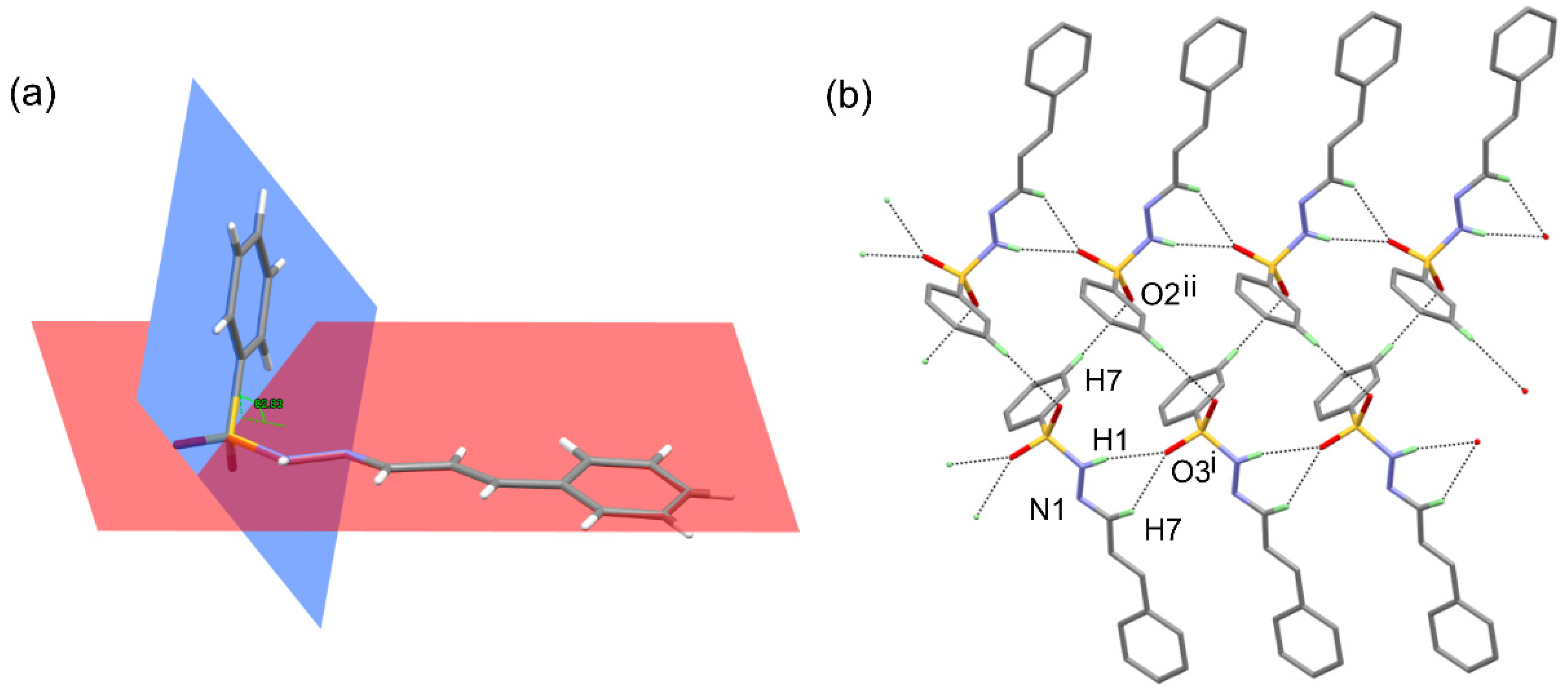
3.2. X-ray Crystallography
3.3. Acute Toxicity in Mice
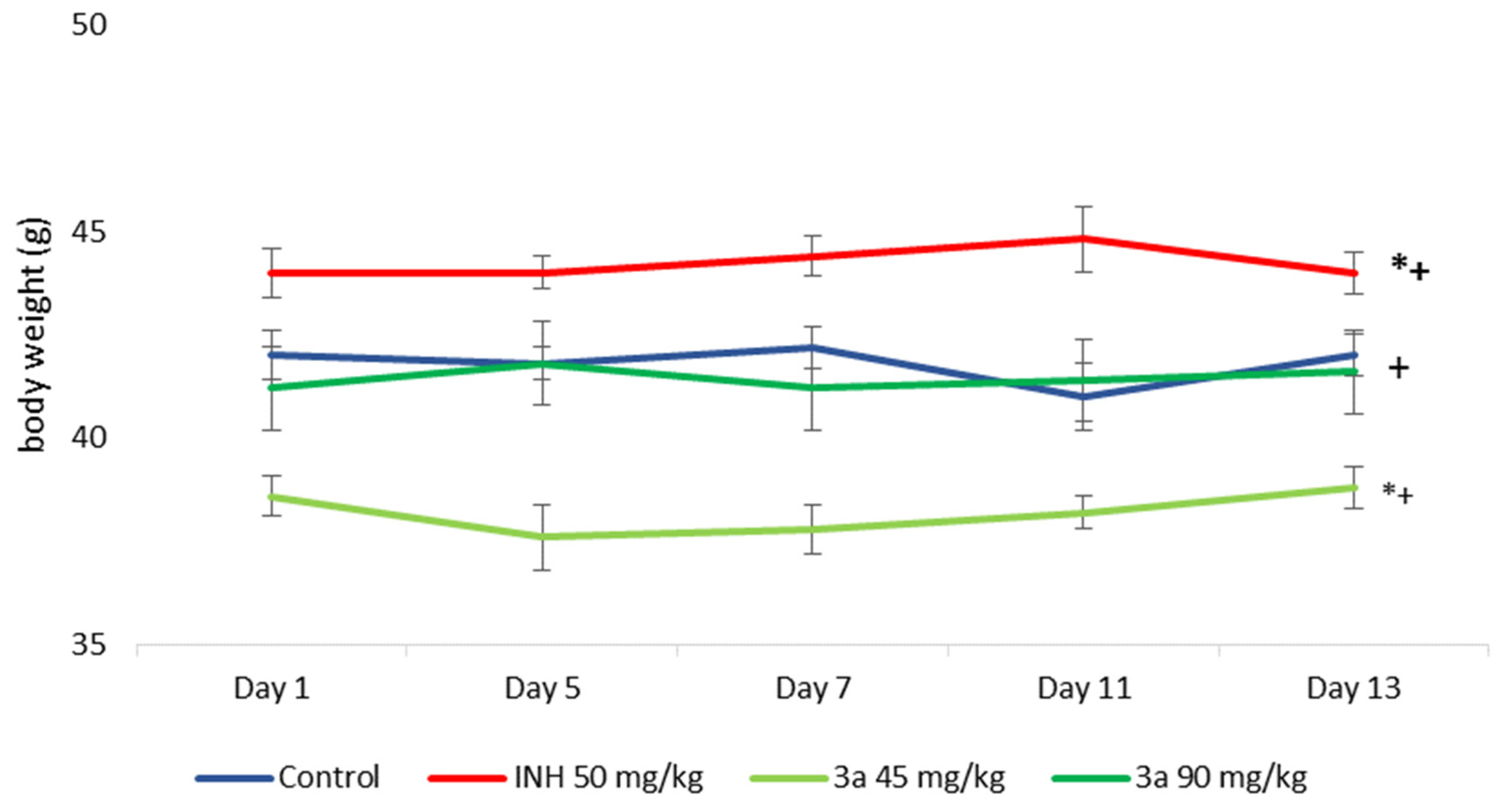
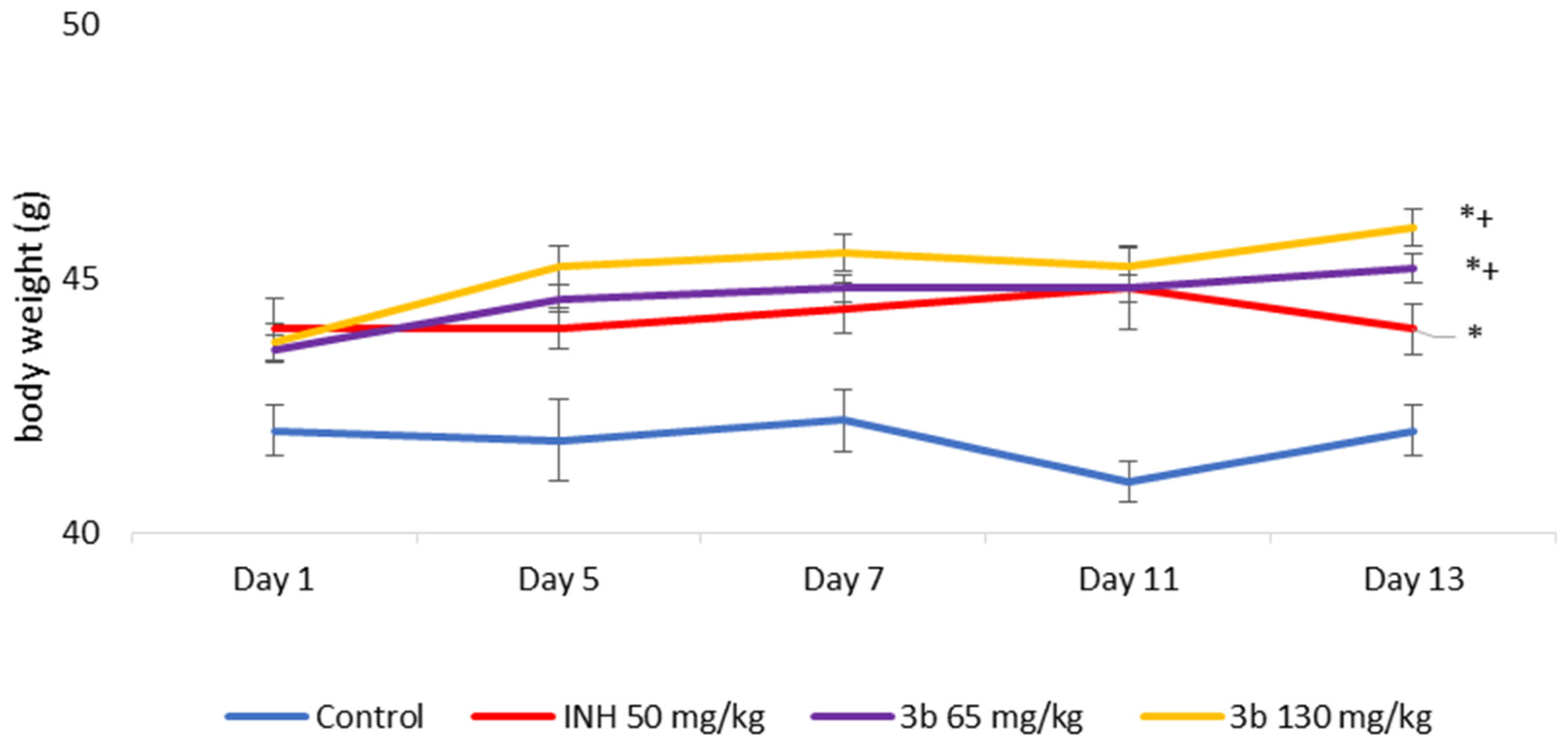
3.4. Sub-Acute Toxicity of Mice, Hematological, Biochemical Parameters amd Markes of Oxidative Stress after Sub-Acute Toxicity Study
3.5. Complete Blood Count (CBC) and Biochemistry in the Blood of Mice
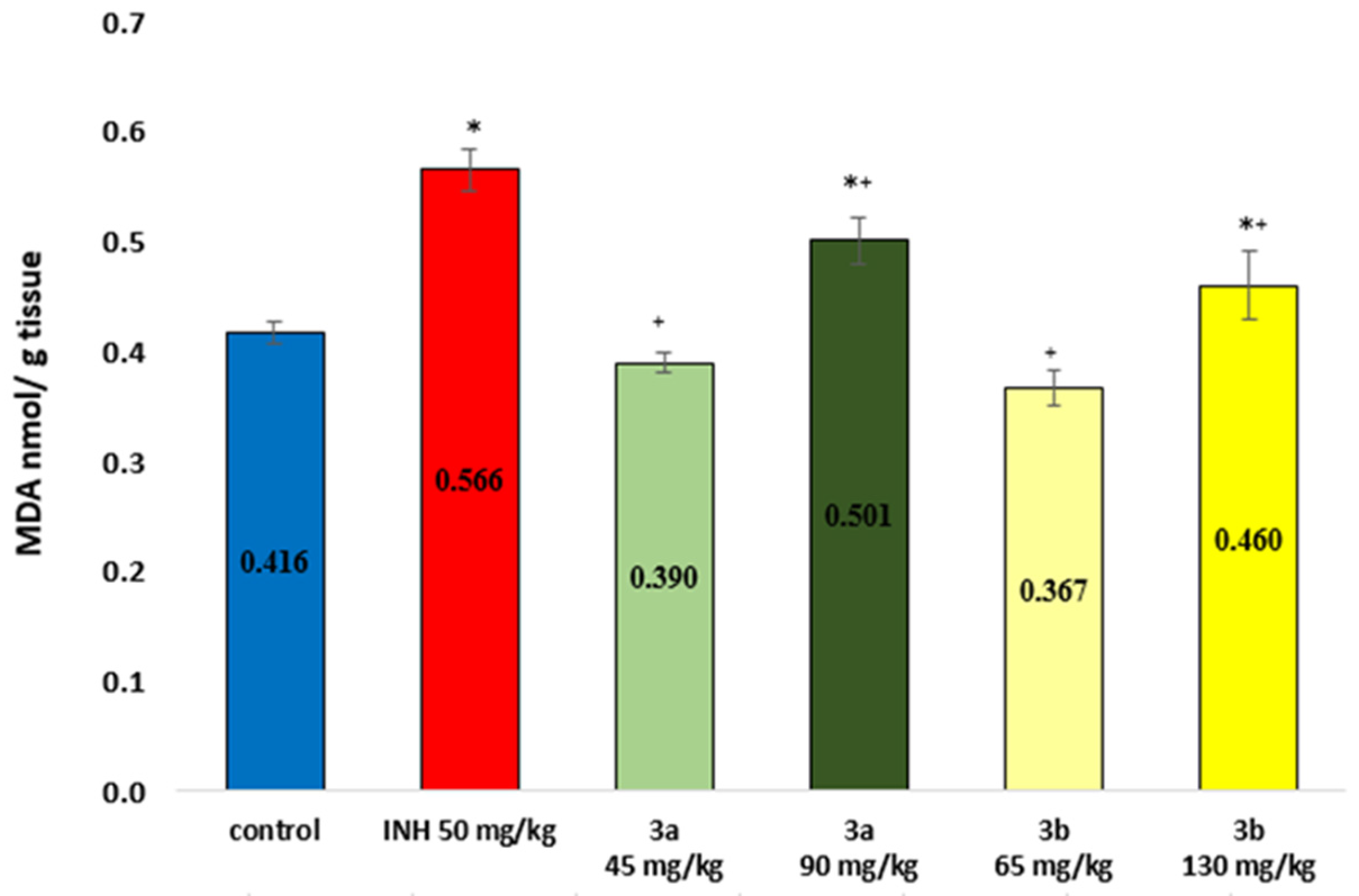
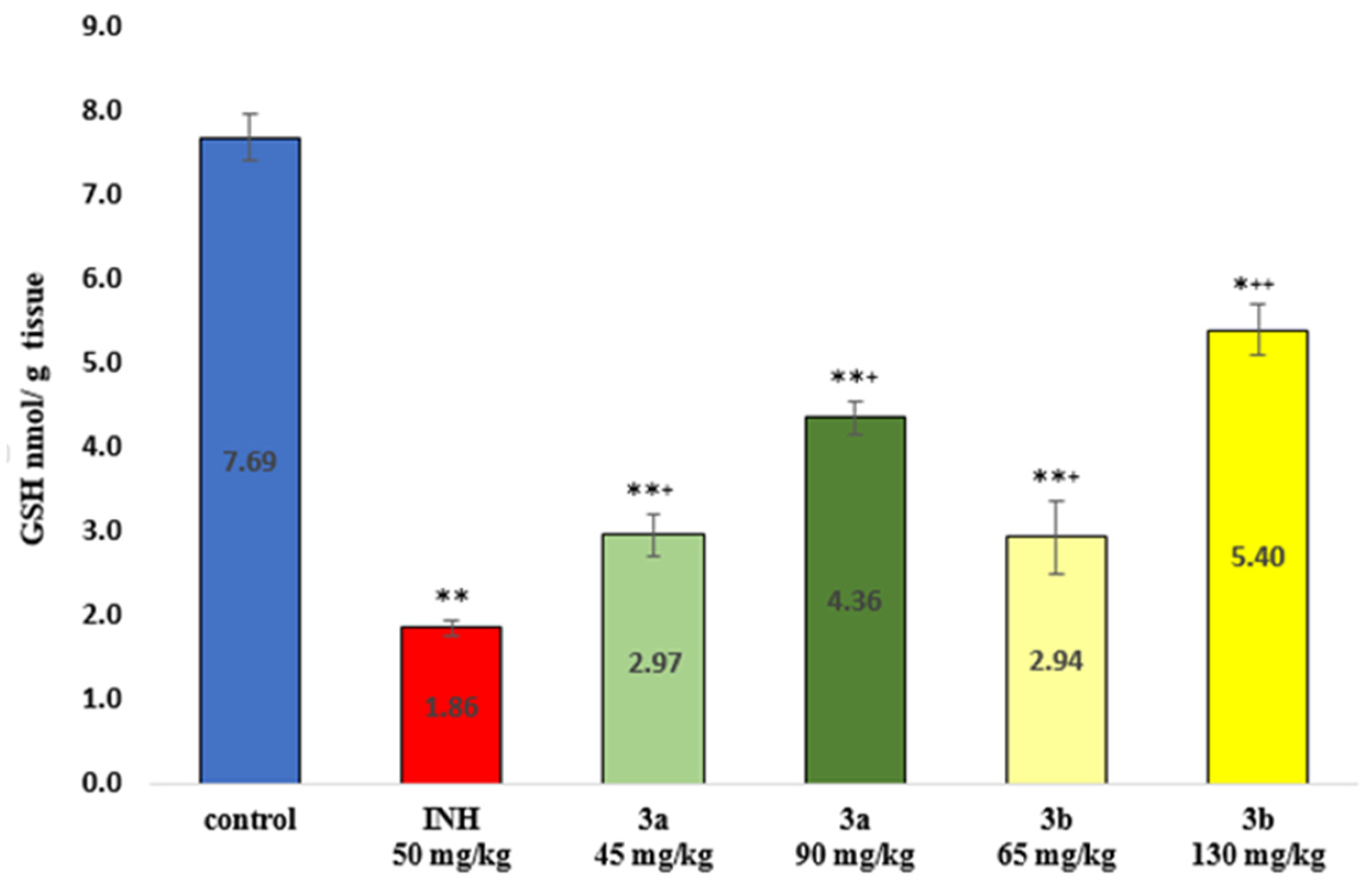
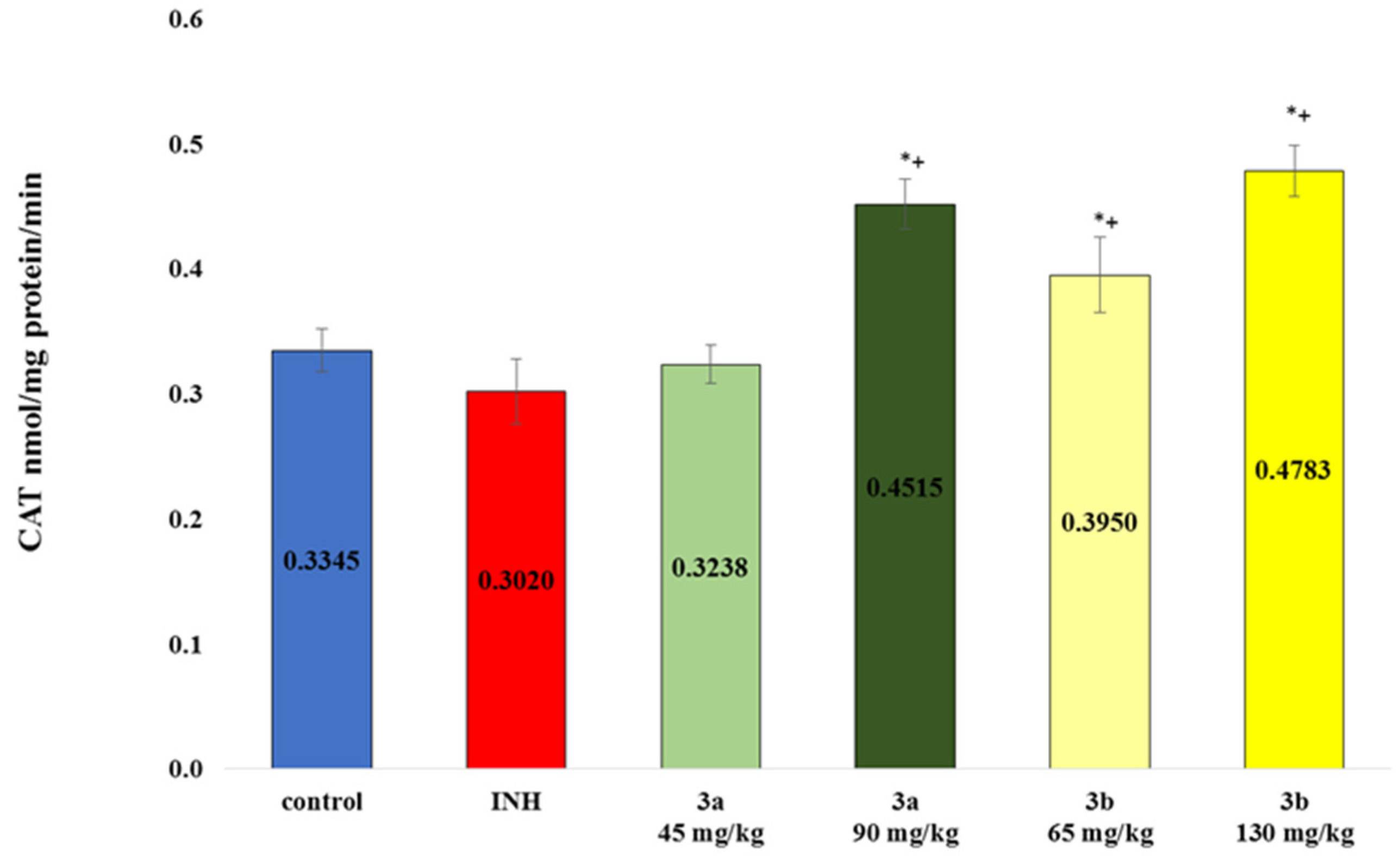
3.6. Markers of Oxidative Stress
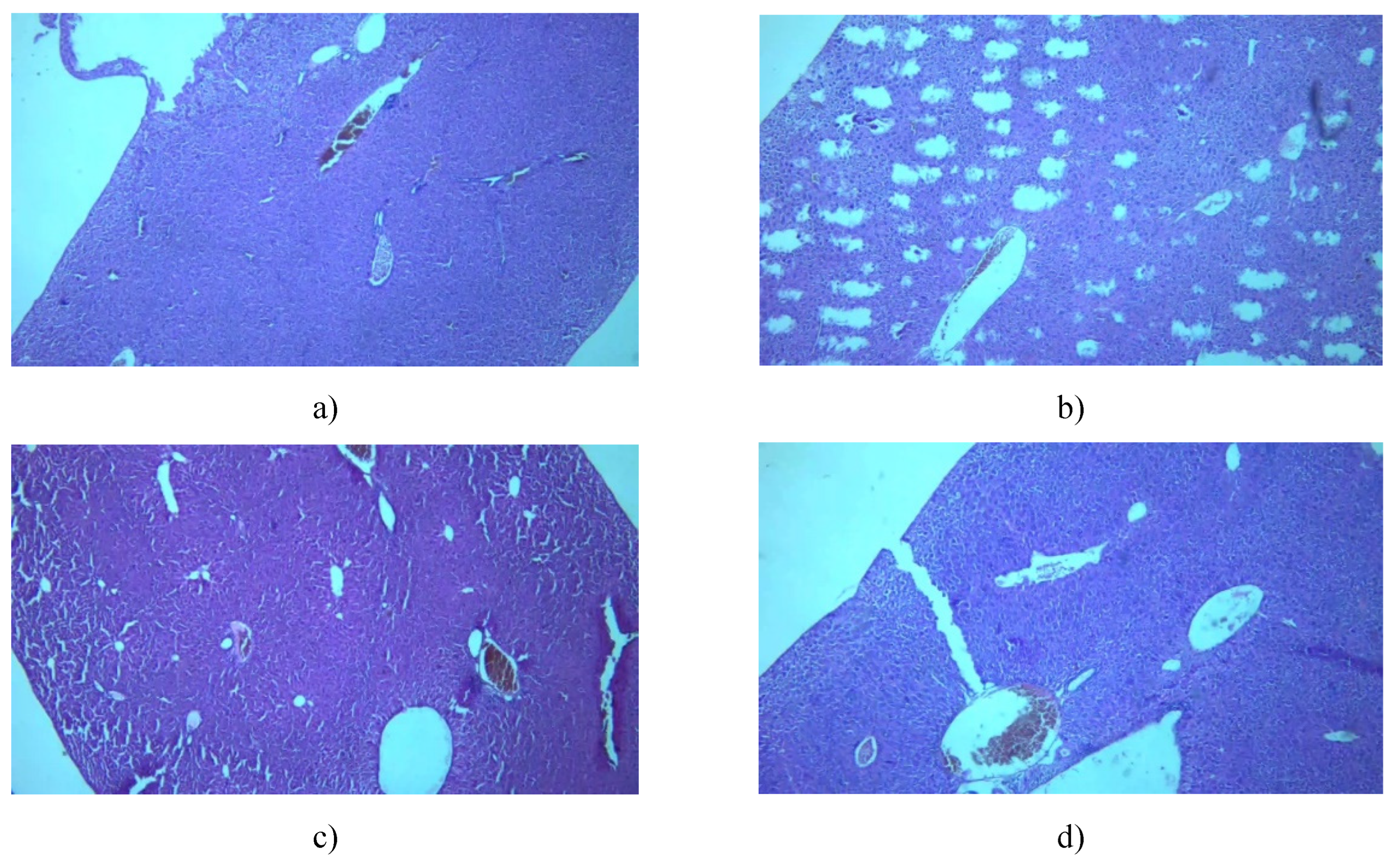
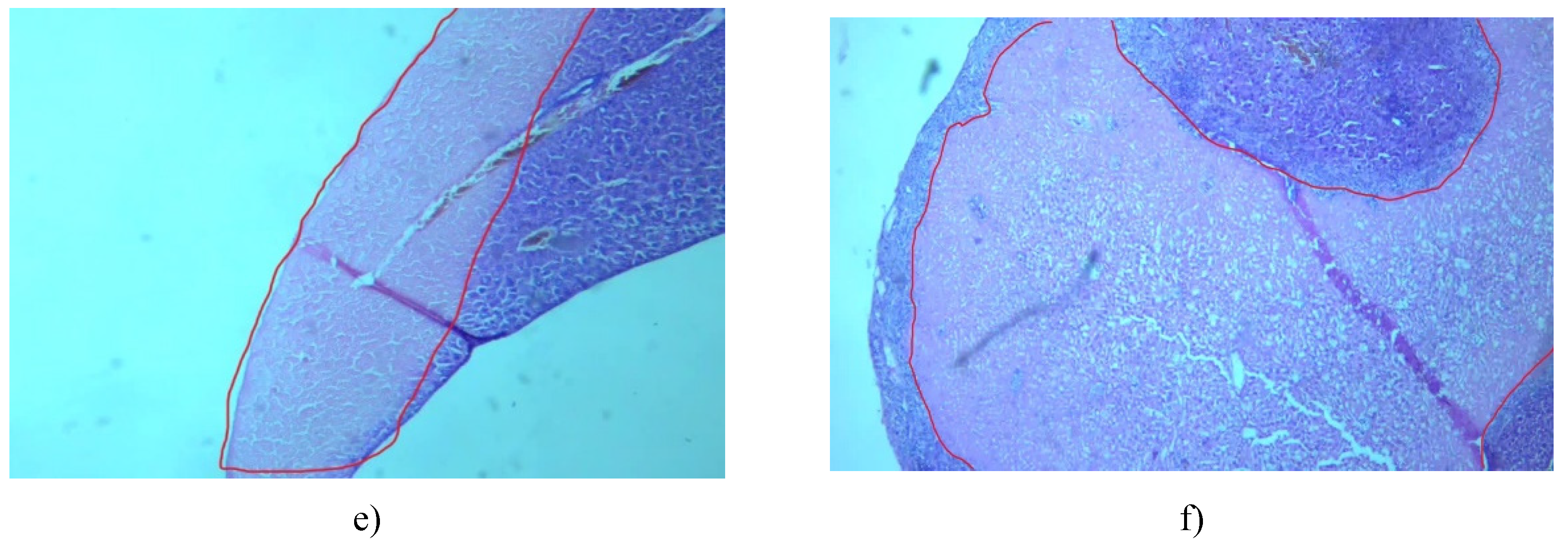
3.7. Histological Examinationof Tissue Specimens Post-Mortem
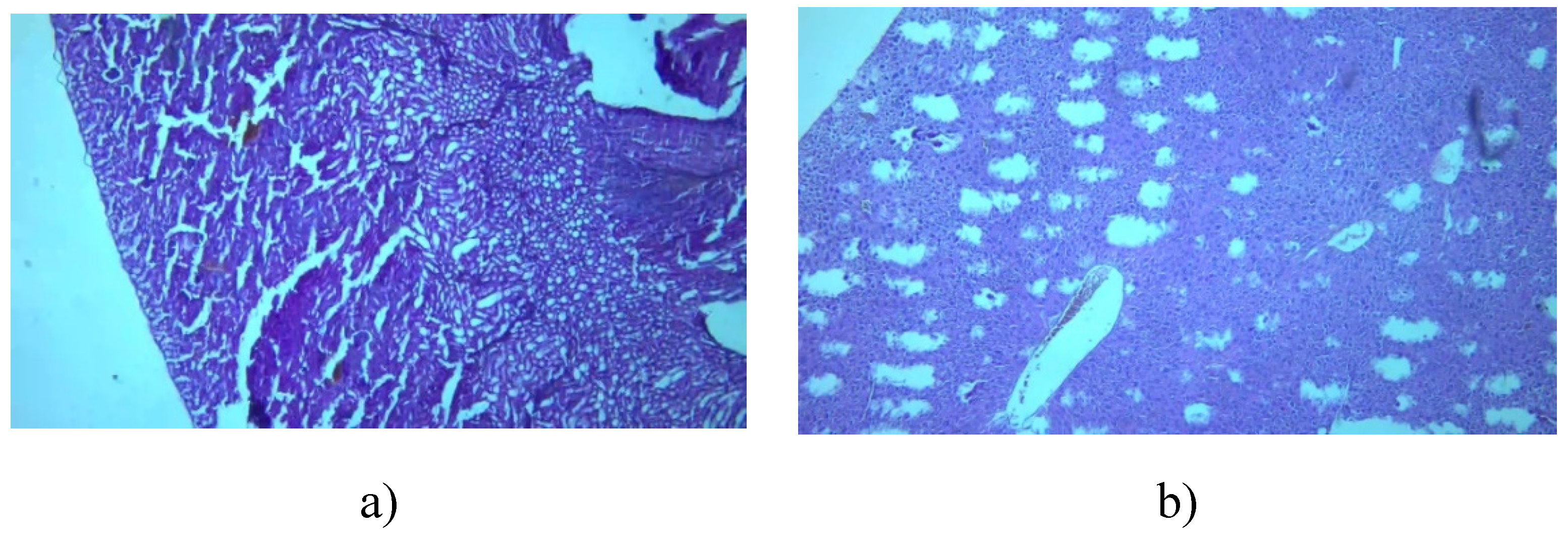
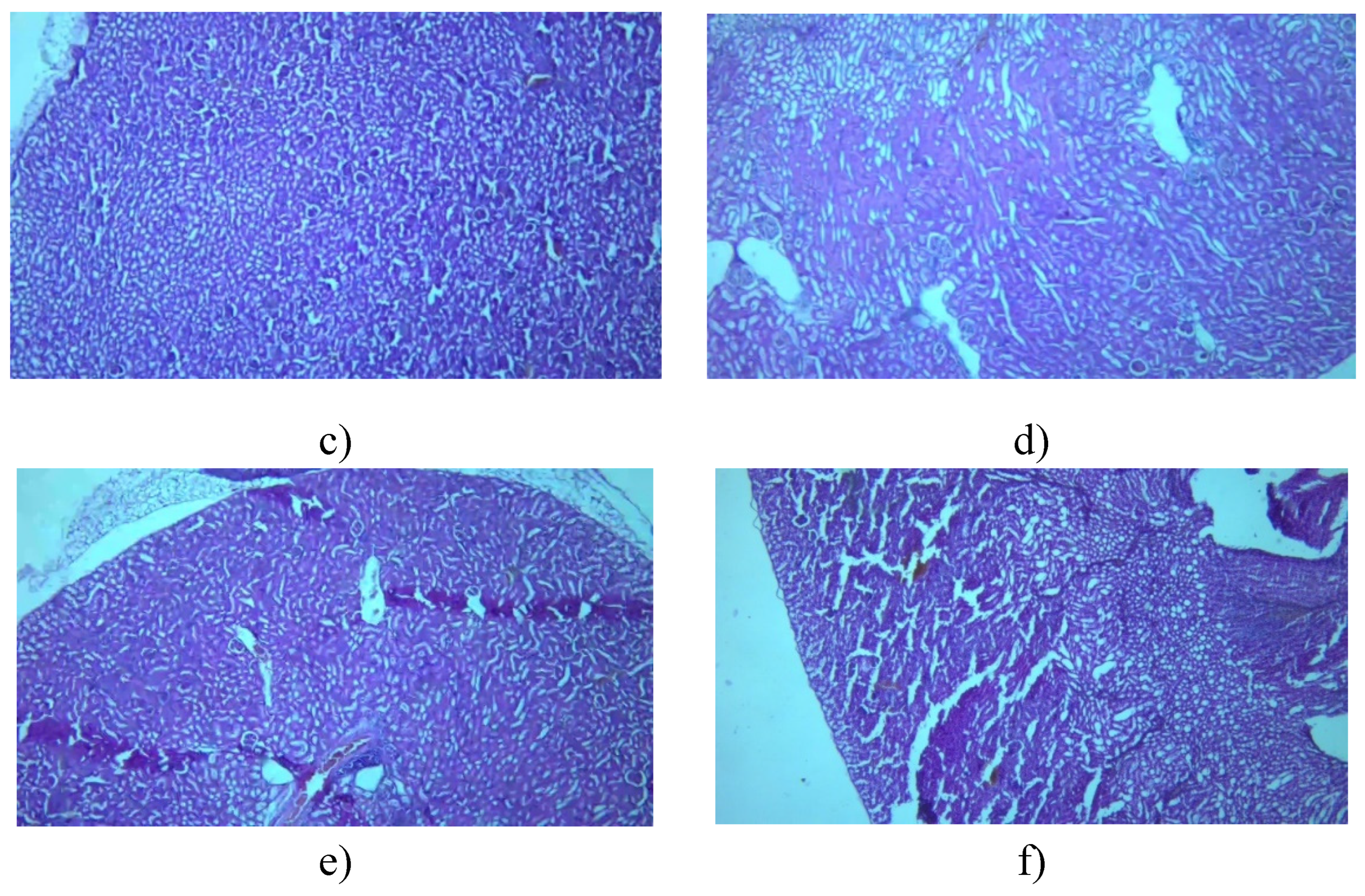
3.7.1. Kidney
3.7.2. Liver
3.8. In Vitro InhA Inhibition Assay
4. Conclusions
Supplementary Materials
Author Contributions
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. WHO consolidated guidelines on tuberculosis Module 4: Treatment Drug-susceptible tuberculosis treatment. World Health Organization 2022; 2022.
- Ghiano DG, Recio-Balsells A, Bortolotti A, Defelipe LA, Turjanski A, Morbidoni HR, et al. New one-pot synthesis of anti-tuberculosis compounds inspired on isoniazid. European journal of medicinal chemistry. 2020;208:112699.
- Ghiya S, Joshi YC. Synthesis and antimicrobial evaluation of hydrazones derived from 4-methylbenzenesulfonohydrazide in aqueous medium. Medicinal Chemistry Research. 2016;25(5):970-6.
- Mascarello A, Mori M, Chiaradia-Delatorre LD, Menegatti ACO, Monache FD, Ferrari F, et al. Discovery of Mycobacterium tuberculosis Protein Tyrosine Phosphatase B (PtpB) Inhibitors from Natural Products. PLOS ONE. 2013;8(10):e77081.
- Navakoski de Oliveira K, Chiaradia LD, Alves Martins PG, Mascarello A, Sechini Cordeiro MN, Carvalho Guido RV, et al. Sulfonyl-hydrazones of cyclic imides derivatives as potent inhibitors of the Mycobacterium tuberculosisprotein tyrosine phosphatase B (PtpB). MedChemComm. 2011;2(6):500-4.
- Bhat M, Poojary B, Kumar SM, Hussain MM, Pai N, Revanasiddappa BC, et al. Structural, crystallographic, Hirshfeld surface, thermal and antimicrobial evaluation of new sulfonyl hydrazones. Journal of Molecular Structure. 2018;1159:55-66.
- Ozmen UO, Olgun G. Synthesis, characterization and antibacterial activity of new sulfonyl hydrazone derivatives and their nickel(II) complexes. Spectrochim Acta A Mol Biomol Spectrosc. 2008;70(3):641-5.
- Popiołek, Ł. The bioactivity of benzenesulfonyl hydrazones: A short review. Biomedicine & Pharmacotherapy. 2021;141:111851.
- Siemann S, Evanoff DP, Marrone L, Clarke AJ, Viswanatha T, Dmitrienko GI. N-Arylsulfonyl Hydrazones as Inhibitors of IMP-1 Metallo-β-Lactamase. Antimicrobial Agents and Chemotherapy. 2002;46(8):2450-7.
- Angelova VT, Pencheva T, Vassilev N, K-Yovkova E, Mihaylova R, Petrov B, et al. Development of New Antimycobacterial Sulfonyl Hydrazones and 4-Methyl-1,2,3-thiadiazole-Based Hydrazone Derivatives. Antibiotics (Basel, Switzerland). 2022;11(5):562.
- Valcheva V, Simeonova R, Mileva M, Philipov S, Petrova R, Dimitrov S, et al. In Vivo Toxicity, Redox-Modulating Capacity and Intestinal Permeability of Novel Aroylhydrazone Derivatives as Anti-Tuberculosis Agents. Pharmaceutics. 2022;15(1):79.
- Teneva Y, Simeonova R, Valcheva V, Angelova VT. Recent Advances in Anti-Tuberculosis Drug Discovery Based on Hydrazide–Hydrazone and Thiadiazole Derivatives Targeting InhA. Pharmaceuticals. 2023;16(4):484.
- Europe Co. European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes (ETS 123). In: Europe Co, editor. Strasbourg, France1991.
- Lorke, D. A new approach to practical acute toxicity testing. Arch Toxicol. 1983;54(4):275-87.
- Chinedu E, Arome D, Ameh FS. A new method for determining acute toxicity in animal models. Toxicol Int. 2013;20(3):224-6.
- Chen C, Wicha SG, de Knegt GJ, Ortega F, Alameda L, Sousa V, et al. Assessing Pharmacodynamic Interactions in Mice Using the Multistate Tuberculosis Pharmacometric and General Pharmacodynamic Interaction Models. CPT Pharmacometrics Syst Pharmacol. 2017;6(11):787-97.
- Lillie, R.D. STUDIES ON HISTOCHEMICAL ACYLATION PROCEDURES. I. PHENOLS. J Histochem Cytochem. 1964;12:522-9.
- Polizio AH, Peña C. Effects of angiotensin II type 1 receptor blockade on the oxidative stress in spontaneously hypertensive rat tissues. Regulatory peptides. 2005;128(1):1-5.
- Bump EA, Taylor YC, Brown JM. Role of glutathione in the hypoxic cell cytotoxicity of misonidazole. Cancer Research. 1983;43(3):997-1002.
- Aebi, H. [13] Catalase in vitro. Methods in enzymology. 105: Elsevier; 1984. p. 121-6.
- Chetty S, Armstrong T, Sharma Kharkwal S, Drewe WC, De Matteis CI, Evangelopoulos D, et al. New InhA Inhibitors Based on Expanded Triclosan and Di-Triclosan Analogues to Develop a New Treatment for Tuberculosis. Pharmaceuticals. 2021;14(4):361.
- Doğan H, Doğan Ş D, Gündüz MG, Krishna VS, Lherbet C, Sriram D, et al. Discovery of hydrazone containing thiadiazoles as Mycobacterium tuberculosis growth and enoyl acyl carrier protein reductase (InhA) inhibitors. European Journal of Medicinal Chemistry. 2020;188:112035.
- Doğan ŞD, Gündüz MG, Doğan H, Krishna VS, Lherbet C, Sriram D. Design and synthesis of thiourea-based derivatives as Mycobacterium tuberculosis growth and enoyl acyl carrier protein reductase (InhA) inhibitors. European journal of medicinal chemistry. 2020;199:112402.
- Bruker, A. APEX 2. Bruker Advanced X-ray Solutions. Madison, Wisconsin, USA; 2004.
- Bruker, A. Saint and SADABS. Bruker AXS Inc, Madison, Wisconsin, USA. 2009.
- Sheldrick, G.M. SHELXT–Integrated space-group and crystal-structure determination. Acta Crystallographica Section A: Foundations and Advances. 2015;71(1):3-8.
- Cunha MR, Tavares MT, Carvalho CF, Silva NA, Souza AD, Pereira GJ, et al. Environmentally safe condition for the synthesis of aryl and alkyl sulfonyl hydrazones via one-pot reaction. ACS Sustainable Chemistry & Engineering. 2016;4(4):1899-905.
- Ozochukwu IS, Okpareke OC, Izuogu DC, Ibezim A, Ujam OT, Asegbeloyin JN. N’-(Pyridin-3-ylmethylene) benzenesulfonohydrazide: Crystal structure, DFT, Hirshfeld surface and in silico anticancer studies. European Journal of Chemistry. 2021;12(3):256-64.
- Rahman MM, Hussain MM, Arshad MN, Awual MR, Asiri AM. Arsenic sensor development based on modification with (E)-N′-(2-nitrobenzylidine)-benzenesulfonohydrazide: a real sample analysis. New Journal of Chemistry. 2019;43(23):9066-75.
- Blatova OA, Asiri AM, Al-Amshany ZM, Arshad MN, Blatov VA. Molecular packings and specific-bonding patterns in sulfonamides. New Journal of Chemistry. 2014;38(9):4099-106.
- Hodge HC, Sterner JH. Tabulation of Toxicity Classes. American Industrial Hygiene Association Quarterly. 1949;10(4):93-6.
- Ogawa K, Ito M. Appetite-enhancing Effects of trans-Cinnamaldehyde, Benzylacetone and 1-Phenyl-2-butanone by Inhalation. Planta Med. 2016;82(1-2):84-8.
- Niedernhofer LJ, Daniels JS, Rouzer CA, Greene RE, Marnett LJ. Malondialdehyde, a Product of Lipid Peroxidation, Is Mutagenic in Human Cells *. Journal of Biological Chemistry. 2003;278(33):31426-33.
- Bains VK, Bains R. The antioxidant master glutathione and periodontal health. Dent Res J (Isfahan). 2015;12(5):389-405.
- Chen Y, Dong H, Thompson DC, Shertzer HG, Nebert DW, Vasiliou V. Glutathione defense mechanism in liver injury: Insights from animal models. Food and Chemical Toxicology. 2013;60:38-44.
- Nair A, Sharma P, Tiwary MK. Glutathione deficiency in COVID19 illness-does supplementation help? Saudi Journal of Anaesthesia. 2021;15(4):458-60.
- Nandi A, Yan LJ, Jana CK, Das N. Role of Catalase in Oxidative Stress- and Age-Associated Degenerative Diseases. Oxid Med Cell Longev. 2019;2019:9613090.

| Dose mg/kg b.w. | Effect/Lethality | Time of occurrence | Symptoms |
|---|---|---|---|
| 1500 | 2/3 (66%) | After 24h | Delayed reflexes, somnolence, lethal outcome |
| 1000 | 1/3 (33%) | After 7 days | Impaired coordination, rapid breathing, lethal outcome |
| 750 | 0/3 | - | - |
| 500 | 0/3 | - | - |
| 250 | 0/3 | - | - |
| Dose mg/kg b.w. | Effect/Lethality | Time of occurrence | Symptoms |
|---|---|---|---|
| 1500 | 3/3 (100%) | After 24h | Respiratory failure with long pauses, ataxia, piloerection, seizures, lethal outcome. |
| 1000 | 0/3 | - | - |
| 750 | 0/3 | - | - |
| 500 | 0/3 | - | - |
| 250 | 0/3 | - | - |
| Compound | Concentration (µM) | % inhibition of InhA |
|---|---|---|
| 3a | 1 | 35.2 |
| 10 | 54.1 | |
| 25 | 52.9 | |
| 50 | 64.7 | |
| 100 | 82.3 | |
| 3b | 1 | 29.3 |
| 10 | 54.1 | |
| 25 | 58.8 | |
| 50 | 64.7 | |
| 100 | 58.8 | |
| Triclosan | 100 | 82.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




