Submitted:
20 May 2024
Posted:
21 May 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Incretin Hormones
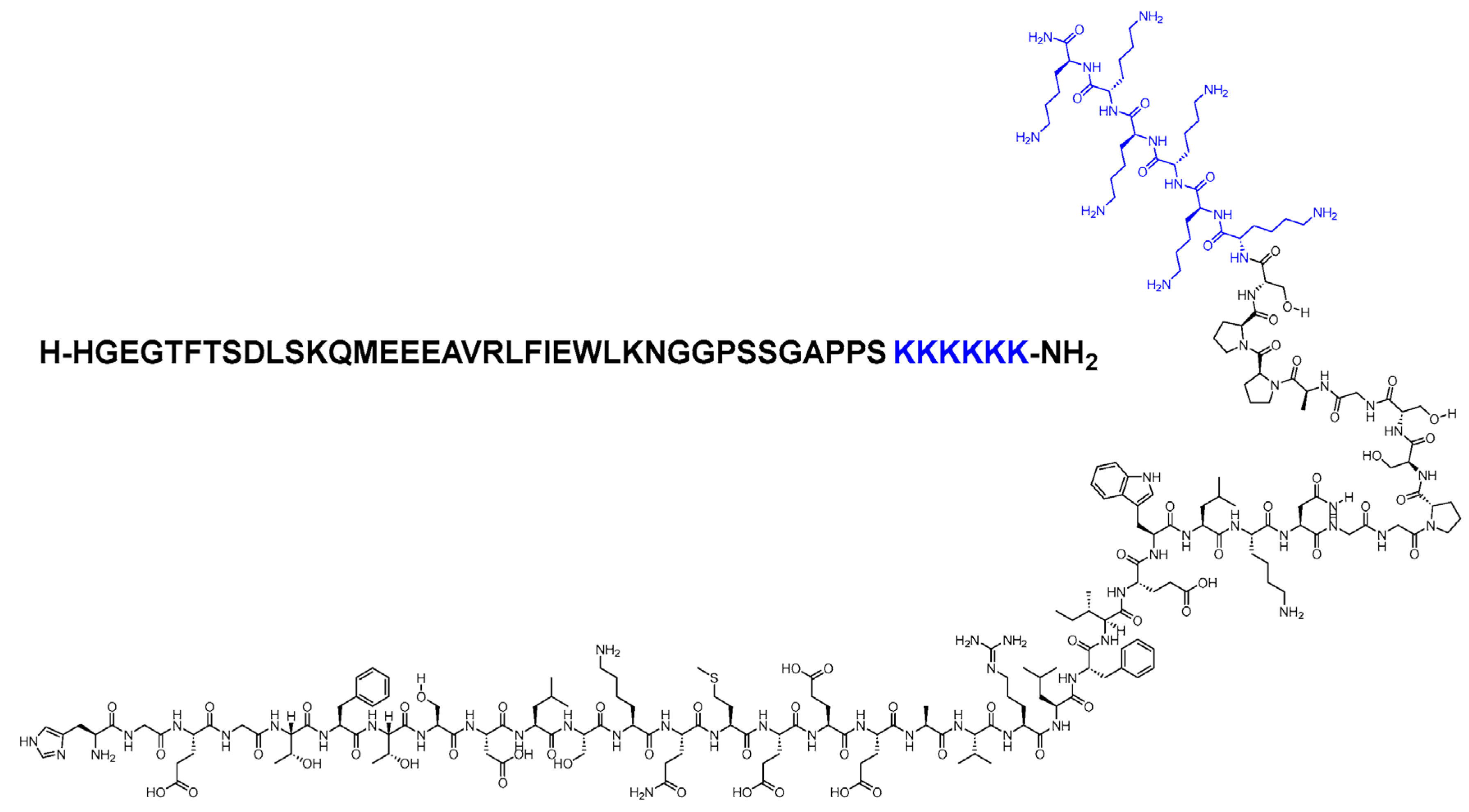
Glucagon-like Peptide-1 Receptor Agonists (GLP-1RAs)
Incretin Mimetics Classes
Exenatide
Liraglutide
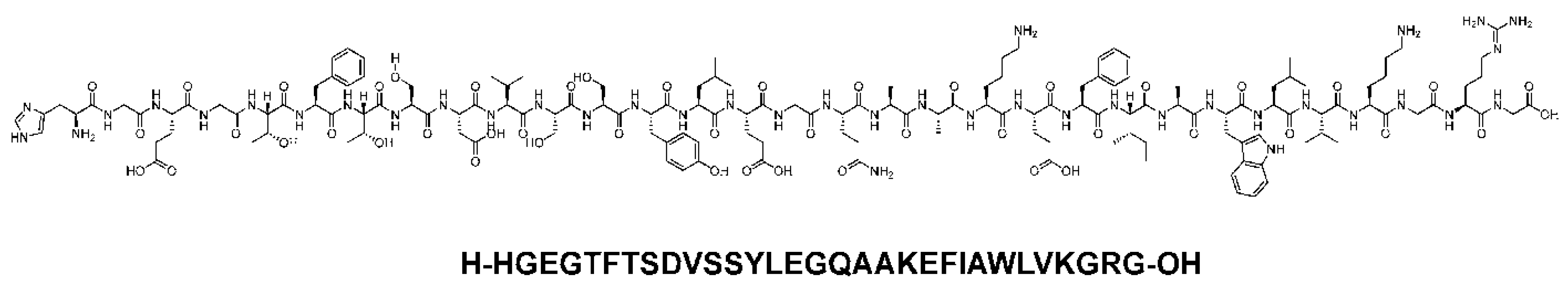
Dulaglutide
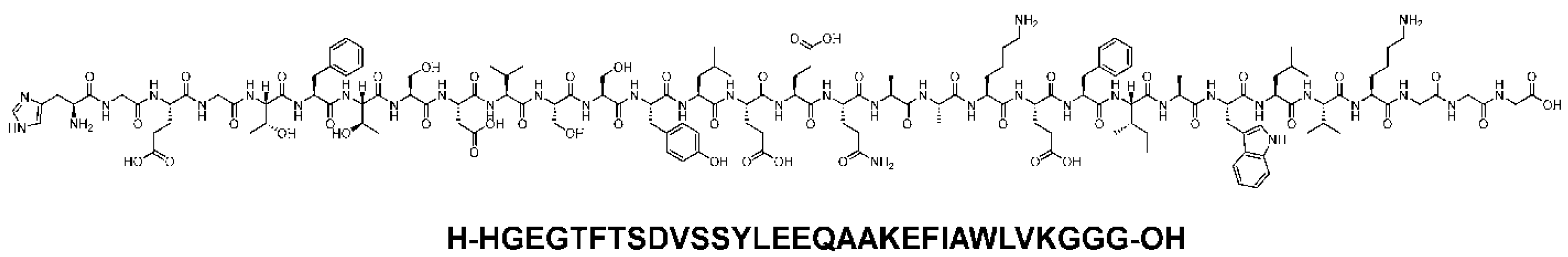
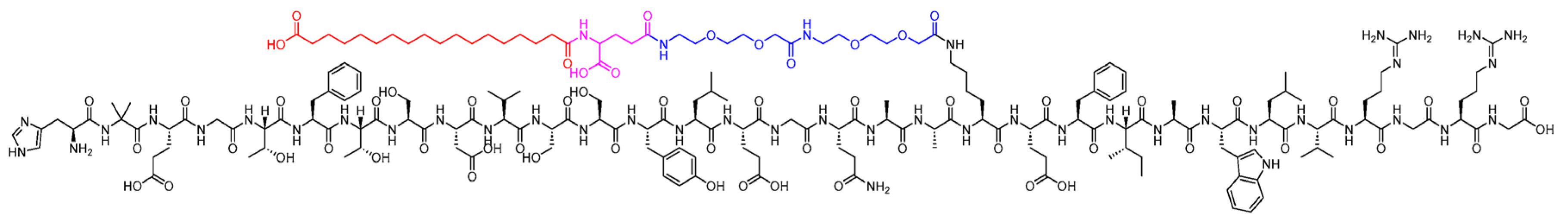
Semaglutide
Albiglutide
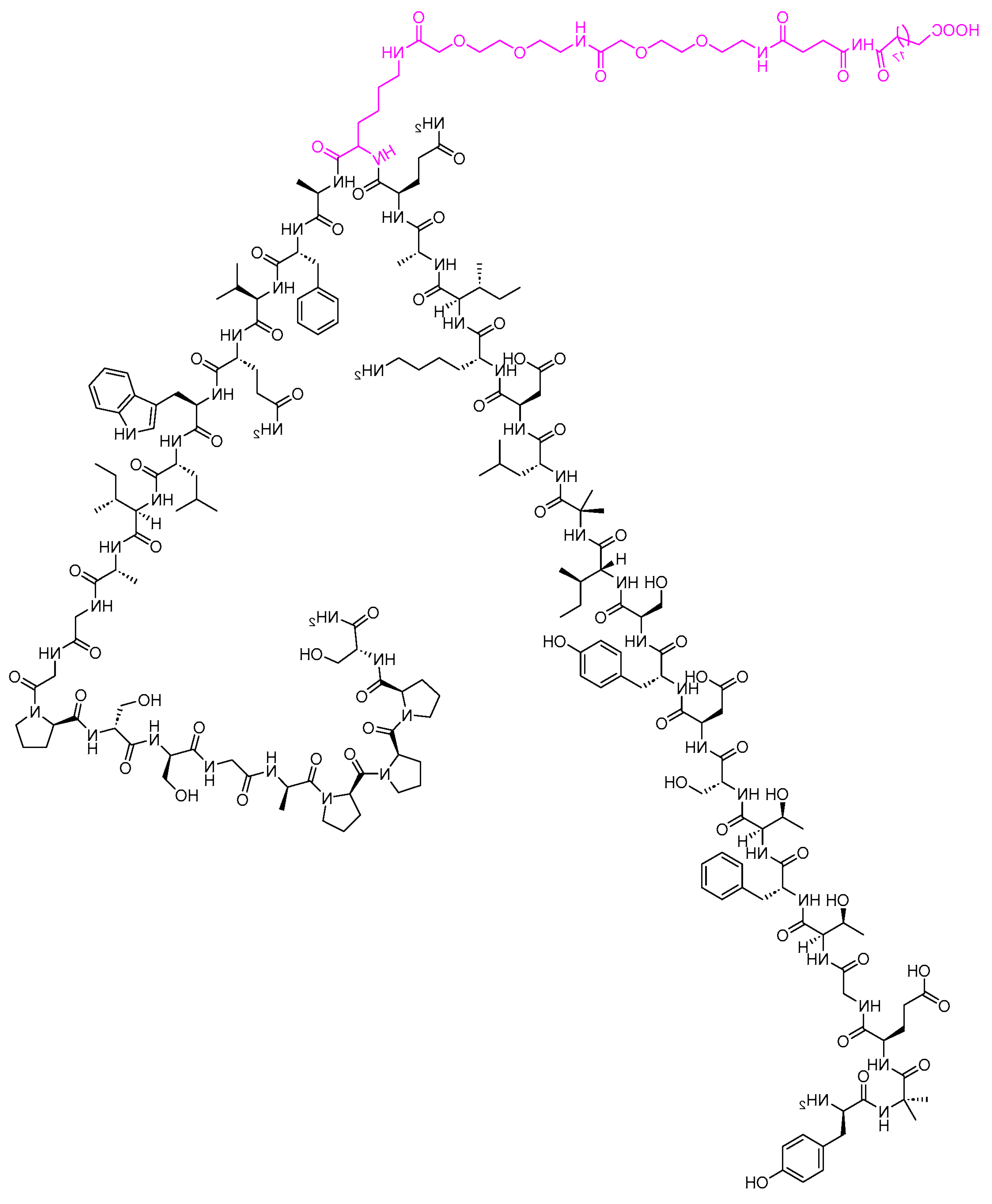
Tirzepatide
Lixisenatide
Conclusions
References
- Tan Q, Akindehin SE, Orsso CE, Waldner RC, DiMarchi RD, Müller TD and Haqq AM (2022) Recent Advances in Incretin-Based Pharmacotherapies for the Treatment of Obesity and Diabetes. Front. Endocrinol. 13:838410. [CrossRef]
- Ahmad, E., Lim, S., Lamptey, R., Webb, D. R., & Davies, M. J. (2022). Type 2 diabetes. The Lancet, 400(10365), 1803-1820.s.
- Collins, L., & Costello, R. A. (2023). Glucagon-Like Peptide-1 Receptor Agonists. In StatPearls. StatPearls Publishing.
- Teague, M., Martinez, A., Walker, E., El-Rifai, M., & Carris, N. W. (2023). Use and Interchange of Incretin Mimetics in the Treatment of Metabolic Diseases: A Narrative Review. Clinical Therapeutics, 45(3), 248-261. [CrossRef]
- Hussain, U., Das, A. K., Ghosh, S., and Sil, P. C. An overview on the role of bioactive α-glucosidase inhibitors in ameliorating diabetic complications. Food and Chemical Toxicology, Volume 145, November 2020, 111738.
- Ruze, R., Liu, T., Zou, X., Song, J., Chen, Y., Xu, R., ... & Xu, Q. (2023). Obesity and type 2 diabetes mellitus: Connections in epidemiology, pathogenesis, and treatments. Frontiers in endocrinology, 14, 1161521.
- Usman, M. S., Davies, M., Hall, M. E., Verma, S., Anker, S. D., Rosenstock, J., & Butler, J. (2023). The cardiovascular effects of novel weight loss therapies. European heart journal, 44(48), 5036–5048. [CrossRef]
- Yashi, K., & Daley, S. F. (2023). Obesity and Type 2 Diabetes. In StatPearls. StatPearls Publishing.
- Collins, L., & Costello, R. A. (2023). Glucagon-Like Peptide-1 Receptor Agonists. In StatPearls. StatPearls Publishing.
- Tulp OL. Effect of delayed luminal carbodhydrate uptake via α-glucosidase inhibition on plasma glycemic parameters and lipid profiles in adult wistar fatty rats. Gastroenterol Hepatol Open Access. 2022;13(5):168‒173. [CrossRef]
- Hossain U, Das AK, Ghosh S, Sil PC. An overview on the role of bioactive α-glucosidase inhibitors in ameliorating diabetic complications. Food Chem Toxicol. 2020;145:111738. [CrossRef]
- Al Musaimi, O (2024). Exploring FDA-Approved Frontiers: Insights into Natural and Engineered Peptide Analogues in the GLP-1, GIP, GHRH, CCK, ACTH, and α-MSH Realms. Biomolecules, 14, 264. [CrossRef]
- Nauck, M. A., Mirna, A. E. A., & Quast, D. R. (2023). Meta-analysis of head-to-head clinical trials comparing incretin-based glucose-lowering medications and basal insulin: An update including recently developed glucagon-like peptide-1 (GLP-1) receptor agonists and the glucose-dependent insulinotropic polypeptide/GLP-1 receptor co-agonist tirzepatide. Diabetes, Obesity and Metabolism, 25(5), 1361-1371.
- Jinnouchi H, Sugiyama S, Yoshida A; et al. Liraglutide, a glucagon-like peptide-1 analog, increased insulin sensitivity assessed by hyperinsulinemic-euglycemic clamp examination in patients with uncontrolled type 2 diabetes mellitus. J Diabetes Res. 2015;2015:706416. [CrossRef]
- Boer, G. A., & Holst, J. J. (2020). Incretin Hormones and Type 2 Diabetes—Mechanistic Insights and Therapeutic Approaches. Biology, 9(12). [CrossRef]
- Jia, S., Wang, Z., Han, R., Zhang, Z., Li, Y., Qin, X., … & Yang, J. (2021). Incretin mimetics and sodium-glucose co-transporter 2 inhibitors as monotherapy or add-on to metformin for treatment of type 2 diabetes: A systematic review and network meta-analysis. Acta Diabetologica, 58, 5-18.
- Sposito, A. C., Berwanger, O., de Carvalho, L. S. F., & Saraiva, J. F. K. (2018). GLP-1RAs in type 2 diabetes: Mechanisms that underlie cardiovascular effects and overview of cardiovascular outcome data. Cardiovascular diabetology, 17(1), 157. [CrossRef]
- Nauck, M. A., Quast, D. R., Wefers, J., & Meier, J. J. (2021). GLP-1 receptor agonists in the treatment of type 2 diabetes–state-of-the-art. Molecular metabolism, 46, 101102.
- Wright, S. C., Motso, A., Koutsilieri, S., Beusch, C. M., Sabatier, P., Berghella, A., ... & Lauschke, V. M. (2023). GLP-1R signaling neighborhoods associate with the susceptibility to adverse drug reactions of incretin mimetics. Nature communications, 14(1), 6243.
- Hansen, K. B., Vilsbøll, T., & Knop, F. K. (2010). Incretin mimetics: A novel therapeutic option for patients with type 2 diabetes – a review. Diabetes, Metabolic Syndrome and Obesity, 3, 155–163. [CrossRef]
- Ruan, Z., Yang, L., Shi, H., Yue, X., Wang, Y., Liang, M., & Hu, H. (2021). The cost-effectiveness of once-weekly semaglutide compared with other GLP-1 receptor agonists in type 2 Diabetes: A systematic literature review. Expert review of pharmacoeconomics & outcomes research, 21(2), 221-233.
- Frandsen, C. S., Dejgaard, T. F., Andersen, H. U., Holst, J. J., Hartmann, B., Thorsteinsson, B., & Madsbad, S. (2017). Liraglutide as adjunct to insulin treatment in type 1 diabetes does not interfere with glycaemic recovery or gastric emptying rate during hypoglycaemia: A randomized, placebo-controlled, double-blind, parallel-group study. Diabetes, Obesity and Metabolism, 19(6), 773-782not interfere with glycaemic recovery or gastric emptying rate during hypoglycaemia: A randomized, placebo-controlled, double-blind, parallel-group study.
- Hvarchanova, N. (2024, January). Incretin-mimetic drugs—Nature, benefits, and risks. In Varna Medical Forum (Vol. 13, No. 1).
- Zobel, E. H., Ripa, R. S., Von Scholten, B. J., Rotbain Curovic, V., Kjaer, A., Hansen, T. W., Rossing, P., & Størling, J. (2021). Effect of Liraglutide on expression of inflammatory genes in type 2 diabetes. Scientific Reports, 11(1), 1-8. [CrossRef]
- Wronka M, Krzemińska J, Młynarska E, Rysz J, Franczyk B. New Insights into the Use of Liraglutide-Impact on Cardiovascular Risk and Microvascular Outcomes. Biomedicines. 2023;11(4):1159. [CrossRef]
- Scott LJ. Dulaglutide: A Review in Type 2 Diabetes. Drugs. 2020;80(2):197-208. [CrossRef]
- Gao X, Di Y, Lv Y; et al. A pharmacokinetic study comparing the biosimilar HEC14028 and Dulaglutide (Trulicity®) in healthy Chinese subjects. Clin Transl Sci. 2024;17(4):e13775. [CrossRef]
- Arslanian SA, Hannon T, Zeitler P; et al. Once-Weekly Dulaglutide for the Treatment of Youths with Type 2 Diabetes. N Engl J Med. 2022;387(5):433-443. [CrossRef]
- Wang Y, Deng F, Zhong X; et al. Dulaglutide provides protection against sepsis-induced lung injury in mice by inhibiting inflammation and apoptosis. Eur J Pharmacol. 2023;949:175730. [CrossRef]
- Wang, R., Wang, N., Han, Y., Xu, J., & Xu, Z. (2021). Dulaglutide alleviates LPS-induced injury in cardiomyocytes. ACS omega, 6(12), 8271-8278.
- Xie D, Li Y, Xu M, Zhao X, Chen M. Effects of dulaglutide on endothelial progenitor cells and arterial elasticity in patients with type 2 diabetes mellitus. Cardiovasc Diabetol. 2022;21(1):200. [CrossRef]
- Kosiborod, M. N., Petrie, M. C., Borlaug, B. A., Butler, J., Davies, M. J., Hovingh, G. K., ... & Shah, S. J. (2024). Semaglutide in patients with obesity-related heart failure and type 2 diabetes. New England Journal of Medicine.
- Okamoto, A., Yokokawa, H., Nagamine, T., Fukuda, H., Hisaoka, T., & Naito, T. (2021). Efficacy and safety of semaglutide in glycemic control, body weight management, lipid profiles and other biomarkers among obese type 2 diabetes patients initiated or switched to semaglutide from other GLP-1 receptor agonists. Journal of Diabetes & Metabolic Disorders, 20, 2121-2128.
- Tulp OL, Einstein GP. Review: Obesity and its associated inflammatory cytokines pose significant risk factors for COVID-19 outcomes. Advances in Obesity, Weight Management and Control. 2022;12(1):14‒20. [CrossRef]
- Ruan Z, Yang L, Shi H; et al. The cost-effectiveness of once-weekly semaglutide compared with other GLP-1 receptor agonists in type 2 Diabetes: A systematic literature review. Expert Rev Pharmacoecon Outcomes Res. 2021;21(2):221-233. [CrossRef]
- Rakhat Y, Wang L, Han W; et al. Oral Semaglutide under Human Protocols and Doses Regulates Food Intake, Body Weight, and Glycemia in Diet-Induced Obese Mice. Nutrients. 2023;15(17):3765. Published 2023 Aug 28. [CrossRef]
- Bucheit, J. D., Pamulapati, L. G., Carter, N., Malloy, K., Dixon, D. L., & Sisson, E. M. (2020). Oral semaglutide: A review of the first oral glucagon-like peptide 1 receptor agonist. Diabetes technology & therapeutics, 22(1), 10-18.
- Sodhi, M., Rezaeianzadeh, R., Kezouh, A., & Etminan, M. (2023). Risk of Gastrointestinal Adverse Events Associated With Glucagon-Like Peptide-1 Receptor Agonists for Weight Loss. JAMA, 330(18), 1795–1797. [CrossRef]
- Okeke, I. G., Camarda, A. R., Okeke, R., & Chaughtai, S. (2024). Semaglutide-induced Hyperemesis Gravidarum. JCEM case reports, 2(2), luad167. [CrossRef]
- Tobaiqy, M., & Elkout, H. (2024). Psychiatric adverse events associated with semaglutide, liraglutide and tirzepatide: A pharmacovigilance analysis of individual case safety reports submitted to the EudraVigilance database. International journal of clinical pharmacy, 46(2), 488–495. [CrossRef]
- Rubino, D. M., Greenway, F. L., Khalid, U., O'Neil, P. M., Rosenstock, J., Sørrig, R., Wadden, T. A., Wizert, A., Garvey, W. T., & STEP 8 Investigators (2022). Effect of Weekly Subcutaneous Semaglutide vs Daily Liraglutide on Body Weight in Adults With Overweight or Obesity Without Diabetes: The STEP 8 Randomized Clinical Trial. JAMA, 327(2), 138–150. [CrossRef]
- Singh, S., Chang, H. Y., Richards, T. M., Weiner, J. P., Clark, J. M., & Segal, J. B. (2013). Glucagonlike peptide 1-based therapies and risk of hospitalization for acute pancreatitis in type 2 diabetes mellitus: A population-based matched case-control study. JAMA internal medicine, 173(7), 534–539.
- Casella, S., & Galli, K. (2024). Appendicitis: A Hidden Danger of GLP-1 Receptor Agonists?. The Journal of pharmacy technology : jPT : Official publication of the Association of Pharmacy Technicians, 40(2), 108–111. [CrossRef]
- Gudin, B., Ladhari, C., Robin, P., Laroche, M. L., Babai, S., Hillaire-Buys, D., & Faillie, J. L. (2020). Incretin-based drugs and intestinal obstruction: A pharmacovigilance study. Therapie, 75(6), 641–647. [CrossRef]
- Kalas, M. A., Galura, G. M., & McCallum, R. W. (2021). Medication-Induced Gastroparesis: A Case Report. Journal of investigative medicine high impact case reports, 9, 23247096211051919. [CrossRef]
- Newsome, P., Francque, S., Harrison, S., Ratziu, V., Gaal, L. V., Calanna, S., Hansen, M., Linder, M., & Sanyal, A. (2019). Effect of semaglutide on liver enzymes and markers of inflammation in subjects with type 2 diabetes and/or obesity. Alimentary Pharmacology & Therapeutics, 50(2), 193-203. [CrossRef]
- Nowell, J., Blunt, E., & Edison, P. (2023). Incretin and insulin signaling as novel therapeutic targets for Alzheimer’s and Parkinson’s disease. Molecular Psychiatry, 28(1), 217-229.
- Tschöp M, Nogueiras R, Ahrén B. Gut hormone-based pharmacology: Novel formulations and future possibilities for metabolic disease therapy. Diabetologia. 2023;66(10):1796-1808. [CrossRef]
- Pandey S, Mangmool S, Parichatikanond W. Multifaceted Roles of GLP-1 and Its Analogs: A Review on Molecular Mechanisms with a Cardiotherapeutic Perspective. Pharmaceuticals (Basel). 2023;16(6):836. [CrossRef]
- Willard, F. S., Douros, J. D., Gabe, B. N., Showalter, A. D., Wainscott, D. B., Suter, T. M., Capozzi, M. E., Stutsman, C., Cardona, G. R., Urva, S., Emmerson, P. J., Holst, J. J., Coghlan, M. P., Rosenkilde, M. M., Campbell, J. E., & Sloop, K. W. (2020). Tirzepatide is an imbalanced and biased dual GIP and GLP-1 receptor agonist. JCI Insight, 5(17). [CrossRef]
- Aronne, L. J., Sattar, N., Horn, D. B., Bays, H. E., Wharton, S., Lin, W. Y., Ahmad, N. N., Zhang, S., Liao, R., Bunck, M. C., Jouravskaya, I., Murphy, M. A., & SURMOUNT-4 Investigators (2024). Continued Treatment With Tirzepatide for Maintenance of Weight Reduction in Adults With Obesity: The SURMOUNT-4 Randomized Clinical Trial. JAMA, 331(1), 38–48. [CrossRef]
- Tobaiqy, M., & Elkout, H. (2024). Psychiatric adverse events associated with semaglutide, liraglutide and tirzepatide: A pharmacovigilance analysis of individual case safety reports submitted to the EudraVigilance database. International journal of clinical pharmacy, 46(2), 488–495. [CrossRef]
- Leon, N., LaCoursiere, R., Yarosh, D., & Patel, R. S. (2017). Lixisenatide (Adlyxin): A Once-Daily Incretin Mimetic Injection for Type-2 Diabetes. Pharmacy and Therapeutics, 42(11), 676. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5642155/.
- Lee J, Kim R, Kim MH; et al. Weight loss and side-effects of liraglutide and lixisenatide in obesity and type 2 diabetes mellitus. Prim Care Diabetes. 2023;17(5):460-465. [CrossRef]
- Niedermier V, Ayers G, Springer S. Lixisenatide (Adlyxin) for Type 2 Diabetes Mellitus. Am Fam Physician. 2017;96(4):257-258.
- Eleftheriadou A, Riley D, Zhao SS; et al. Risk of diabetic retinopathy and diabetic macular oedema with sodium-glucose cotransporter 2 inhibitors and glucagon-like peptide 1 receptor agonists in type 2 diabetes: A real-world data study from a global federated database. Diabetologia. [CrossRef]
- Wai KM, Mishra K, Koo E; et al. Impact of GLP-1 Agonists and SGLT-2 Inhibitors on Diabetic Retinopathy Progression: An Aggregated Electronic Health Record Data Study. Am J Ophthalmol. [CrossRef]
- Liu, C., Zou, Y., & Qian, H. (2020). GLP-1R agonists for treating obesity: A patent review (2015-present). Expert Opinion on Therapeutic Patents, 30(10), 781–794. [CrossRef]
- Gong, B., Yao, Z., Zhou, C., Wang, W., Sun, L., & Han, J. (2024). Glucagon-like peptide-1 analogs: Miracle drugs are blooming? European Journal of Medicinal Chemistry, 269, 116342. [CrossRef]
- Cowart, K. (2019). Oral Semaglutide: First-in-Class Oral GLP-1 Receptor Agonist for the Treatment of Type 2 Diabetes Mellitus. Annals of Pharmacotherapy. [CrossRef]
- Rizvi SAA, Einstein GP, Tulp OL, Sainvil F, Branly R. Introduction to Traditional Medicine and Their Role in Prevention and Treatment of Emerging and Re-Emerging Diseases. Biomolecules. 2022;12(10):1442. Published 2022 Oct 9. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



